Manuscript accepted on :07-May-2025
Published online on: 24-07-2025
Plagiarism Check: Yes
Reviewed by: Dr. Mohamad R. Abdullah
Second Review by: Dr. Aparna Gupta
Final Approval by: Dr. Anton R Keslav
Utkarsh Upadhyay1 , Vivek Srivastava1*
, Vivek Srivastava1* , Neha Mathur1
, Neha Mathur1 and Somesh Thapliyal2
and Somesh Thapliyal2
1Amity Institute of Pharmacy, Amity University Lucknow Campus, Lucknow, India.
2Department of Pharmaceutical Sciences, Hemvati Nandan Bahuguna Garhwal University, Srinagar, Uttarakhand, India.
Corresponding Author E-mail: srivastavav696@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3246
Abstract
Breast carcinoma is the world's most common cancer and the top cause of death for women among all cancers. Cancer immunotherapy has made significant clinical progress, particularly in adoptive cell transfer therapy and immune checkpoint inhibition in the treatment of breast cancer. The treatment of breast cancer has evolved a lot because of several advancements in the understanding of the pathways as well as of molecular approach. Chimeric antigen receptor target (CART), Program lethal ligand 1 & 2(PD-1/PDL-2), and the cytotoxic T-lymphocyte–associated factor 4 (CTLA-4) checkpoint inhibitors are the prime targets with positive results for the treatment using immunotherapy. Only four drugs of this category have been approved by the FDA till now. Several vaccines are under clinical trials in mono as well as in combination therapy for the treatment of the same and their response data are summarized in this review .In this review clinical trial data on CART therapy, PD- 1/PDL-1, CTLA-4 checkpoint inhibitors, and their therapeutic approach have been explored and to provide all the treatment options available for the management of breast cancer in immunotherapy.
Keywords
Breast Cancer; CART-Therapy; CTLA-4; Immunotherapy; PD-1/PDL-1; Vaccines
Download this article as:| Copy the following to cite this article: Upadhyay U, Srivastava V, Mathur N, Thapliyal S. Recent Clinical Advancements in Immunotherapy for the Management of Breast Cancer. Biomed Pharmacol J 2025;18(3). |
| Copy the following to cite this URL: Upadhyay U, Srivastava V, Mathur N, Thapliyal S. Recent Clinical Advancements in Immunotherapy for the Management of Breast Cancer. Biomed Pharmacol J 2025;18(3). Available from: https://bit.ly/4h7zroV |
Introduction
Breast cancer (BC) is metastatic cancer if detected at the initial stage higher percentage of survival and an excellent prognosis can be achieved, as the disease progresses, it can spread to distant organs i.e., the brain, lungs, liver, and bones which makes it quite impossible to cure.1 There are 2.3 million women diagnosed, and 30 percent deaths with breast cancer take place globally as per the WHO report in 2020.2 About sixty percent of the world’s population is diagnosed and seventy percent of breast cancer deaths occur in America, Central America, Africa, and Asia.3 Indeed, the rates of BC mortality are higher in many developing countries as shown in Fig no. 01. In the last five years, BC has been diagnosed in 7.8 lacs women, making it the commonly diagnosed cancer in the world as of the end of 2020.4
Cells in our body derive cancerous cells, which gain the characteristics of malignancy through genetic alterations. These malignant cells frequently evade the immune system and grow into a significant clinical mass. HoweverTargeting regulated mortality-1 (PD-1) and programmed mortality the ligand-1 (PD-L1), along with cytotoxicity T-lymphocyte linked antigen-4 (CTLA-4), can restore pre-existing anti-cancer immunity and provide long-term clinical responses in various types of solid tumors that are resistant to standard therapies. 5,6 These immune checkpoint inhibitors show anti-cancer immunity’s existence in malignant tumors. According to evidence from the last ten years, relapse and metastases of breast cancer remain a significant concern in clinical practice, shown by advanced endocrine therapy and anti-human epidermal receptor-2 (HER-2) therapy.7,8 Therefore new therapeutic approaches are still needed for the treatment of the disease, and intensive interest has been shown in immunotherapy for the treatment of BC.9,10
Methods
The primary goal of this review is to compile recent advances in immunotherapy drugs used for the treatment of BC that are present in trials and only four are approved by FDA for clinical applications. We have done an immense literature survey from pubmed.gov to compile this review by using various keywords like “BC in immunotherapy,” “CART”, “PD-1/PDL-1”, and “CTLA-4 in BC.”
Drugs targeting the immune checkpoints CART, PD-1/PDL-1, and CTLA-4 are summarized in this review in a detailed manner as these targets have a potential benefit in cancer therapy.
Immune system and Breast cancer
Immune cells play an important role in both BC recognition and early eradication,11 as well as tumor progression. Immunoediting is characterized by evolving the interactions between mammary tumors and host immunity.12 It refers to the complex interaction between a tumor and the body’s immune defenses. Cancer cell interactions with the immune network progress through three stages: Eliminating, the point of equilibrium, and escapes.13 During the elimination phase, cancer is detected and removed by both the adaptive and innate immune systems. Rare cells of cancer that escape the elimination stage are predicted to move into an equilibrium phase, in which the immune system’s adaptive mechanism stops tumor development and keeps them dormant without eliminating the cancer. It develops the ability to avoid immune detection and destruction during the phases in a variety of ways.14
Hence, the evasion of tumors in the immune system is an indication of cancer. BC progresses through the accumulation of mutational changes, which occur mostly in the ductal epithelium. Multiple elements in the ductal microenvironment can influence the development and progression of these changes.14 Cells that play an important role in breast carcinogenesis, are immune cells, they begin with immunosurveillance in the tissue of the normal breast and continue through primary and metastatic BC.15 In the normal breast, the ductal cellular layer contains a significant immune cell population that includes B-cells and macrophages, NK Cells with CD4 and CD8+ T cells, and other immune cell subtypes.16 To provide innate and acquired immunity to the epithelial layer, as well as to protect against exogenous and endogenous agents, and for the elimination of transformed cells, cells of the immune system together. There are significant implications in the understanding of the cellular population in the normal tissues of the breast, for the prevention of BC, regulation of carcinogenesis of the breast, and improved risk assessment methods.17,18,19
Genes responsible for Breast cancer
Breast Cancer Genes 1&2 (BRCA1& BRSA2)- The BRCA1 & BRSA2 genes code for the production of a protein that acts as a tumor suppressor and both are located on chromosomes 17q21 and 13q12, and both genes code for tumor suppressor proteins. BRCA1 causes aberrant duplication of centrosomes, instability in genetic material, dysregulation in cycle checkpoint, and apoptosis. BRSA1 proteins help to repair damaged DNA.20,21,22 By aiding in DNA repair, the BRCA1 protein helps to maintain the equilibrium of a cell’s DNA. The BRCA2 protein controls recombinational healing in double-strand breaks in DNA through its interactions with RAD51 and DMC1.23
Ataxia-telangiectasia (ATM): Normally, the ATM gene helps repair damaged DNA, or it might cause the cell to die if the damage cannot be fixed. The serine-threonine kinase protein that is encoded by the ATM gene is essential for triggering barrier signaling in reaction to DNA damage24 (double-strand breaks) by proteins in activating checkpoint signaling such as BRCA, p53, and Chk2 in response to DNA damage.25 By inheriting 2 abnormal copies of this gene Ataxia-telangiectasia is caused and this gene in some families has been linked to the history of having an increased risk of BC.26
Partner And Localizer of BRSA2(PALB2)- A protein produced by the PALB2 gene functions as an essential partner with the protein produced by the BRCA2 gene and promotes BRCA1 and BRCA2 specific binding domain to the locations of DNA damage.27 Mutations in this gene have been related to an increased incidence of BC.28
Tumor protein53(TP53)- The TP53 gene aids in the arrest of cell growth in the presence of damaged DNAThe syndrome known as Li-Fraumeni is caused by hereditary abnormalities in this gene. This syndrome increases the risk of developing breast cancer.29 This variant is a rather rare cause of BC. Checkpoint kinase-2(CHEK2)- a gene that normally used in DNA repair is the CHEK2 gene. BC risk is raised by a CHEK2 mutation.30
Phosphatase and TENsin (PTEN)- The PTEN gene aids in the control of cell proliferation. The syndrome of Cowden is caused by genetic mutations of this gene This raises the chance for growths in the thyroid, uterus, ovaries, and digestive tract in addition to cancer and benign (non-cancerous) breast tumors.31
Cadherin1(CDH1)- By inheriting mutations in the gene diffused hereditary gastric carcinoma (a rare type of stomach cancer) is caused, and women having this kind of mutation are at higher risk of developing invasive lobular BC.32
Syndrome Serine/Threonine kinase 11(STK11)- Peutz-Jeghers can be caused by the deficiency of this gene and it gives rise to various cancers including BC.33
Immunotherapy, a treatment that harness the bodys immune system to fight cancer, shows promise in breast cancer, with some strategies targeting specific immune related genes and cells.
As shown in Fig no.01, there are several environmental factors that contribute to BC in addition to genes.
 |
Figure 1: Breast cancer is caused by an abnormal balance of estrogen and progesterone in the woman’s body and all the agents triggering these changes are said to be the risk factors responsible for the occurrence of Breast Cancer.23Click here to view Figure |
Immune-mediated pathogenesis
Breast cancer’s specific mechanism is unclear. However, Significant immune suppression has been detected by the cell’s inability to respond to proliferative agents like Con A and phytohemagglutinin, It has been linked to tumor incidence in the development and growth of carcinogen-induced BC. Thymic atrophy causes a decrease in the quantity of T-cells as well as a decrease in the production of the interleukin-2 two (IL-2) receptor.34 The immune system plays a dual function in tumor genesis and progression, capable of both preventing and promoting tumor growth. When cytokines such as interferons (IFN), tumor necrosis factors (TNF), or growth factors transforming are created during the early phases of cancer development, they have an anti-tumor effect in an inflammatory setting, but they actively encourage tumor growth and spread when they accumulate during prolonged inflammation (i.e., after the tumor has formed).[34]. Before surgery and adjuvant therapy, BC patients also had a general immune system malfunction favoring a Th2 response, as seen by a decreased percentage of CD4+ and CD8+ T cells generating type 1 antibodies (lL-2, IFN-, or TNF-) or type 2 lymphocytes (IL-4).35 cytokines in comparison to healthy controls. In the initial stages of tumorigenesis in BC, a key role is played by NK-impaired cells.36 These immune alterations directly influence treatment decisions by guiding the use of immunotherapies, such as immune checkpoint inhibitors or cytokine-based treatments, and help determine the timing and suitability of immunomodulatory strategies alongside surgery and chemotherapy. For instance, therapies aiming to restore Th1 responses or enhance NK cell activity are being considered to improve patient outcomes.37
Recent Clinical Advancements in Immunotherapy
Immune checkpoint inhibitors targeting Chimeric Antigen Receptor T-cells therapy (CART), and Program Death-1 (PD-1) induce long-lasting responses that can lead to improved survival in a variety of cancers.37 Although the FDA has approved several drugs for various cancers, only a few drugs, are approved for BC.
Chimeric Antigen Receptor T-cell therapy (CART)
CAR-T cell therapy is a type of adoptive immunotherapy that uses T cells to boost anti-tumor activity. These are synthetic compounds outside of the cell, inside the cell, across the membrane, and gap domains, all of the components that make up CARs. Most commonly, a single-chain polymorphic fragment of an antibody with a particular target antigen as its antigen is used to construct the extracellular domain.39 The intracellular region transmits the signals into the cell and as a result, there are now four generations of CARs that may be divided into groups based on how many costimulatory domains they include.40 The difficulty still lies in finding a tumor-associated antigen that permits at least a modest identification of the antigens on normal cells, even though a perfect CAR structure is still lacking.41 Patients with hematologic malignancies experience long-lasting and curative effects from CART-cell therapy, while solid tumors have had only limited success with this therapeutic approach.42
The First Generation
Only the intracellular portion of the CD3 domain is used by first-generation CAR-T cells to relay activation signals. Due to a lack of costimulatory signaling, which causes more rapid CAR-T cell death, they have shown limited benefit in clinical 43
The Second Generation
The second generation of CAR-T cell therapy included the addition of the costimulatory signal domain 4-1BB or CD28; the simultaneous activation of these two signals increased the effectiveness of CART cells in eliminating Multiple clinical trials prove that second-generation CART-cells do exhibit tumorigenic properties in B-cell acute lymphoblastic leukemia. 44,45
The Third generation
In the third generation of CAR-T cells, costimulatory signaling molecules like CD27, 4-1BB, CD28, OX40, and ICOS are used commonly, which gives them better antitumor activity than its earlier 29 The widest technology used for creating CAR structures is the third generation of CAR-T cells.
In vitro, this generation of Egf CAR-T cells efficiently and selectively suppressed the growth of TNBC cells; however, healthy breast epithelial cells and breast cancer cells only displayed modest cytotoxicity that were estrogen receptor-positive (ER+). With negligible off-tumor harm, this ability was also proven in vivo utilizing a transgenic mouse model. In vitro treatment with TNBC cells promoted the development and durability of naive-associated EGFR-expressing CAR-T cells. Furthermore, in TNBC cells, EGFR CAR-T cells enhanced the messenger pathway for the interferon, granzyme-perforin-PARP, and Fas-FADD-caspase.47
The Fourth generation CART-cell targets folate receptor
Due to the combined advantages of the three costimulatory domains, the 4th generation of FR-targeted CAR-T cells with the costimulatory domains CD27, CD-28, and 4-1BB showed superior therapeutic success in
In the study, fourth-generation FR-CAR T cells lysed 88.7 10.6% of target cells when mixed using the FR-expressing MDA-MB-231 BC cell type at a 20:1 effector to target ratio. It’s worth noting that the target cells with higher surface FR expression demonstrated greater FR-CAR T cell lethal lysis. FR-negative MCF10A healthy breast-like cell type at a comparable ratio (34.3 4.7%), where the CAR T cells’ specific apoptosis was not seen. When mixed with MDA-MD-231 spheroid, FR-CAR T cells demonstrated anticancer activity as evidenced by spheroid size reduction and breakage.47 A list of drugs under clinical trials is given in table no.01.
Table 1: Listed drugs are under clinical trials in phase-1/2 and phase-1 for the treatment of Breast cancer through CART-mediated cell therapy.110
| Antigen targeted | No of enrolled Candidates | The phase of the clinical trial | Id-of clinical trial | Type of BC |
| HER2/GD2/CD44v6 | 100 | Phase1/2 | NCT04430595 | BC |
| MUC1 | 20 | Phase1/2 | NCT04107142 | TNBC |
| CEA | 40 | Phase1/2 | NCT04348643 | BC |
| CD133 | 20 | Phase1/2 | NCT02541370 | BC |
| CD44v6 | 100 | Phase1/2 | NCT04427449 | CD44v6 +ve BC |
| MUC1 | 69 | Phase 1 | NCT04020575 | Metastatic BC |
| cMET | 06 | Phase 1 | NCT01837602 | TNBC, Metastatic BC |
| EpCAM | 30 | Phase 1 | NCT02915445 | Recurrent BC |
| MSLN | 186 | Phase 1 | NCT02792114 | BC |
| NKG2DL | 10 | Phase 1 | NCT04107142 | TNBC |
| HER-2 | 220 | Phase 1 | NCT04650451 | HER2 +ve BC |
| HER-2 | 45 | Phase 1 | NCT03740256 | BC |
| CEA | 75 | Phase 1 | NCT02349724 | BC |
| TnMUC1 | 112 | Phase 1 | NCT04025216 | TNBC |
| ROR1 | 60 | Phase 1 | NCT02706392 | BC Stage 4 |
| MSLN | 20 | Phase 1 | NCT02580747 | TNBC |
| C7R/GD2 | 94 | Phase 1 | NCT03635632 | BC |
Immune Checkpoint Inhibitors
Program Death -1 (PD-1) and Program Death Ligand-1(PDL-1) Pathway
The PDCD1 gene produces human PD-1 (CD279) and belongs to the superfamily of immunoglobulin genes. It has been shown that apoptotic stimuli can increase the expression of this Mucin-domain with-3, lymphocyte-activation gene-3, PD-1, as well as T-cell immunoglobin are T-cell intrinsic checkpoints that mediate T-cell signaling. This factor is found in two distinct cell lines (2B4.11 and LyD9t), and as it is involved in apoptosis, it is known as programmed cell death protein 1.49
Structure of PD-1 PD-1 PD-1 is a type I membrane polypeptide with a size of 50-55 kD. It additionally has a hydrophilic transmembrane domain a tail in the cytoplasm structure domain and a single extracellular IgV domain. The 20 amino acids that make up the IgV domain are not part of the plasma membrane, yet it shares 23% of its amino acid sequence with CTLA-4. 50 2-tyrosine motifs, an immunological receptors inhibitory tyrosine-based switching motif, and an immune cell tyrosine-based inhibitory motif can be found in the cytoplasm (ITSM). 51
This is followed by phosphorylation of the downstream proteins phospholipid inositol-3-kinase (PI3K) and spleen tyrosine kinase and hinders biological activities of T cells like cytokine secretion, lymphocyte proliferation and cytotoxicity of T lymphocytes.51 Through this interaction, tumor-specific T cells become exhausted and undergo death, allowing cancer cells to escape T cell-mediated immune surveillance.52 Checkpoint inhibitors, such as anti-PD-1 and anti-PD-L1 therapies, differ significantly from conventional treatments like chemotherapy, targeted therapy, and hormonal therapy in both mechanism and clinical impact. These immunotherapies work by reactivating exhausted T-cells through blocking inhibitory checkpoint signals in the tumor microenvironment. This restores the immune system’s ability to recognize and attack cancer cells. In contrast, chemotherapy acts non-specifically by killing all rapidly dividing cells, which often results in significant side effects such as immunosuppression, nausea, and hair loss.
On the other hand, targeted therapies are designed to inhibit specific molecular pathways or mutations (e.g., HER2 or PIK3CA in breast cancer), making them effective but only in patients with those particular biomarkers. Hormonal therapies are limited to hormone receptor-positive cancers and function by interfering with hormonal signaling, such as estrogen or progesterone, which drives tumor growth in certain breast cancer subtypes.
Checkpoint inhibitors offer the advantage of specificity to the immune environment, often providing a durable response due to the generation of memory T-cells. However, they may cause immune-related adverse events such as colitis, dermatitis, or endocrinopathies. While chemotherapy and hormonal therapy are widely used across many types of breast cancer, checkpoint inhibitors are particularly beneficial in triple-negative breast cancer (TNBC) patients who express PD-L1. These therapies are increasingly being combined with chemotherapy to enhance their effectiveness. Thus, treatment decisions must consider tumor subtype, biomarker expression, patient immune status, and potential toxicities. Checkpoint inhibitors offer a novel and powerful therapeutic avenue, especially when conventional treatments are less effective or not well-tolerated.
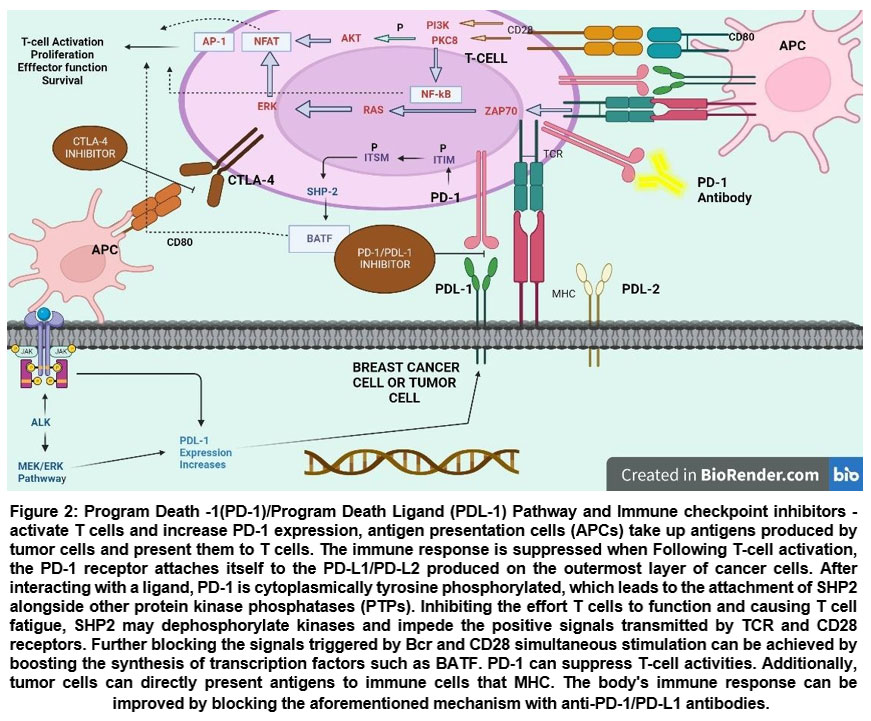 |
Figure 2: Program Death -1(PD-1)/Program Death Ligand (PDL-1) Pathway and Immune checkpoint inhibitors – activate T cells and increase PD-1 expression, antigen presentation cells (APCs) take up antigens produced by tumor cells and present them to T cells.Click here to view Figure |
Tumor microenvironment expressions
PD-1 is expressed by activating B cells, T cells, naturally occurring killer T cells, regulating T cells (Tregs), cells known as dendritic cells, and monocytes. PD-L1 is frequently elevated in cells from tumors, including haemangiomas and solid tumors. B cells, T cells, and DCs,
bone marrow-derived mast cells, macrophages, and some non-immune cells also express PD-L1. The PD-1/PD-L1 pathway (as shown in Fig no. 03) is essential for the development of tumor immunity, viral infection, transplantation immunology, and autoimmune diseases.53,54 However, tumors develop and occur, resulting in 1) blocking the activation of lymphocytes that invade tumors and causing their apoptosis, 2) blocking the CTL granule enzyme and generating perforin, and 3) inhibiting PD-1 and PD-L1 expression. 3) increasing the release of the immune inhibitory cytokine IL-10 while reducing the release of pro-inflammatory cytokines including IFN-, IL-2, and TNF-; 4) slowing the T cell cycle, This leads to the buildup of cells during the G0/G1 phase, and 5) promotes the creation of cancerous epithelial cells, tumor metastasis, and tumor infiltration.55,56
Signaling pathways that regulate PD-1/PDL-1 expressions
Mitogen-activated protein kinase (MAPK) pathway- This pathway is an essential regulator of cellular processes like survival of cells and proliferation. 57,58 Additionally, MAPK pathway activation encourages immune evasion in patients with TNBC, which results in resistance to chemotherapeutic drugs and a low survival rate.59,60 It has been shown that the MAPK pathway regulates PD-L1 expression in different types of cancer cells.61 In vivo and in vitro investigations on TNBC cells revealed that blocking this signaling pathway increased the production of whereas it was discovered that Blocking MAPK and PD-1/PD-L1 increased the effectiveness of immune system checkpoint inhibitors62 and in BC cells, PD-L1/PD-1 association promotes MAPK phosphorylation, activating MAPK pathways and raising the expression of multidrug resistance protein (MDR1)63
PI3K/PTEN/Akt/mTOR Pathway- The intracellular signaling system PI3K/AKT/mTOR controls the cell cycle and is associated with angiogenesis, apoptosis, cell metabolism, and proliferation.64,65 AKT is phosphorylated and activated by PI3K activation, which results in its localization in the blood plasma membrane. Receptor tyrosine kinases initiate the pathway of signaling by activating PI3K, which in turn causes AKT and mTOR complex 1 (mTORC1) to become phosphorylated.66 The most common genomic abnormalities Mutations in the PI3K/AKT/mTOR pathway, which include the gene called mutation in the gene, loss of functionality alterations, or epigenetic inhibition of phosphates, tensin homolog (PTEN), occur in several breast cancer subtypes.67,68
PDL-1 expression control
JAK/STAT pathway- In TNBC, the main transcription factors that have a significant impact on tumor cell survival, growth, intrusion, metastatic disease, and surveillance of immunity are phosphorylated. signal transducers and activators of translation 1/3 (pSTAT1/3) with JAK/STAT signaling pathway activation.69,70,71,72,73 It is noteworthy that STATs regulate the immune response through a variety of pathways, including modulation of PD-L1 expression when binding to the PD-L1 75 In addition, solo suppression of STAT1 or STAT3 leads to a partial reduction of PD-L1 expression, simultaneous suppression of these transcription variables causes a complete downregulation.76 Inhibiting JAK/STAT signaling may therefore show a promising therapeutic strategy for TNBC.77
Nuclear factor k-B (NF-kB) Signalling Pathway – The nuclear factor kappa-light chain-enhancer of active B (NF-kB) is a DNA-binding protein that consists of two main subunits: p50 and p65. NFB is used as a gene regulator which controls cell division, and Proteins of this pathway are overexpressed in cancer which results in weak coordination between cancer cells and organisms. NF-kB activation leads to DNA activation and a variety of gene expressions. According to reports, NF-kappa B is constitutively active in ER-negative BC cell lines, and as a result, it has recently emerged as a key target in the treatment of BC.
Hypoxia-Inducible Factor 1α (HIF-1α)- It is widely known that the hypoxic characteristic acts as an adaptation in the low oxygen environment in BC and other forms of In BC, hypoxia-induced activation of HIF-1α and HIF-2α78,79,80 results in poor prognosis and antioestrogen resistance.81 HIF-1 activates PD-L1 transcription by attaching to the hypoxic response components (HRE) promoter.82 Indeed, earlier research showed a concomitant upregulation of HIF-1, elevated PD-L1 expression, and decreased T-cell activity.83,84,85 The level of expression in the TNBC in vivo model acts as a biomarker for determining the degree of hypoxia.86 Blockade of HIF-1/PD-1/PD-L1 has been promoted as a possible therapeutic target to prevent the immunosuppressive activity of malignancies as a result of this discovery.87
Anti – Program Death 1 Pathway (PD-1) & Program Death Ligand-1 (PDL-1) antibodies
On activated T cells, The PD-1, which (a surface of the cell receptor that operates as a T cell barrier) is increased and binds to two known ligands, PD-L1 and PD-L2. (as shown in fig no 2). PD-1 signals inhibit T-cells throughout the “effector” phase of the immune system reaction by binding to PD-L1 on the outermost layer of tumor and immune cells. Humanized mAbs targeting PD-L1 (nivolumab as well as atezolizumab) and PD-1, which (pembrolizumab) are effective in treating metastatic-BC (pembrolizumab). To date, the side effects associated with these agents’ use in BC have been consistent with those expected for the other drugs of the same class.
Avelumab-Agents that target PD-1 or its ligand show antitumor activity in the treatment of BC by inhibiting PD-1 and PDL-1. 10mg/kg of the drug intravenous, when administered to 168 individuals for fourteen days, shows good tolerability. It shows an acceptable safety profile and modest clinical profile with tumor shrinkage in 9% of treated patients.88
Atezolizumab-In the multicentre study (NCT002620280) drug was tested on around 280 patients with TNBC and showed little or no effect when treated with Atezolizumab plus when not treated with the same in combination with nab- paclitaxel and carboplatin. pCR rate was 48.6 % without drugs and 44.4%with It also showed liver transaminase abnormality.89
Pembrolizumab-Addition of chemotherapy with Pembrolizumab in phase three trials shows that the survival of patients with TNBC is increased when compared to chemotherapy alone. 68.1% of those enrolled in the pembrolizumab-treatment control and in the placebo group suffered 66.9% of serious side effects of grade 3, 4, or 5 associated with the study schedule, with 0.4% of individuals for the pembrolizumab-treatment arm dying compared to none in the group receiving placebo.90
Durvalumab and Olaparib-Polymerase inhibitors(olaparib) when combined with immunotherapy have shown potential antitumor The effectiveness and safety were similar to those observed in the drug and durvalumab monotherapy trials. To ascertain therapeutic benefit predictors and whether adding durvalumab to olaparib monotherapy improves long-term clinical outcomes, more study in a randomized context is required. (32%) of volunteers reported worse adverse effects, with pancreatitis (two [6%]), anemia (four [12%]), and neutropenia (three [9%]) being the most prevalent. Four (12%) individuals had a total of six major adverse events, and three (9%) patients stopped treatment because of side effects. No one passed away as a result of the treatment.91
Nivolumab-Multicenter phase II study shows that there is a clinical benefit of this drug for patients, and can be used as a therapeutic option for Nivolumab’s clinical effectiveness appeared to be improved in tumors with higher levels of microsatellite instability, higher levels of tumor mutation, and higher levels of programmed death-ligand 1 expression. On the other hand, depending on the estimated tissue of origin, there were no apparent changes in efficacy amongst tumor categories. Adverse events noted are by nivolumab’s well-known safety profile. No deaths associated with treatment were noted.92
Cytotoxic T-lymphocyte-associated antigen 4 (CTLA-)4 Inhibitors
Just after T-cell activation, CTLA-4 is upregulated, binds to CD80/CD86, and negative feedback is provided to CD28 stimulation which limits T-cell.
Tremelimumab-Anti–CTLA-4 agent, which was studied in all cancer types. In 26 patients with HER-2/ER+ BC, tremelimumab was tested with exemestane. Dose-limiting toxicity occurred in five patients, Accompanied by diarrhea (four counts) and high blood transcription factors levels (one count). The best response for 42 percent of the patients was 12 weeks of stable illness, and the majority of patients exhibited a significant rise in the proportion of ICOS+/FoxP3+ CD4+ T cells.93
Ipilimumab- Ipilimumab is being researched both alone and in conjunction with PD-1/PD-L1 inhibition in a no. of different tumor types. It has been cleared by a single drug for both earlier and delay-stage carcinoma. 84
Combination Therapy
Immunotherapy paired with chemotherapy has shown promising results in the treatment of a variety of cancers and their subtypes. Clinical trials study shows that there are various combinations of targeted drugs, hormonal drugs, PDL-1 checkpoint inhibitors, CTLA-4 targets, and vaccines that show better result and fewer chances of re-occurrences.
Atezolimumab and Nab-Paclitaxel combination- In a randomized, placebo-controlled, double-blind, phase 3 experiment was undertaken in 246 educational institutions in 41 countries. who were aged 18 and above had a history of metastatic- BC,(gov, NCT02425891)and it is interpreted from the study that Atezolimumab plus nab-paclitaxel is a good therapeutic option for patients of TNBC but this combination has not indicated much difference in the rate of survival of patients. 85
Durvalumab and tremelimumab combination with an Olaparib- Phase-II study and registration number NCT04169841 were made to check the efficiency of the Olaparib drug (300mgBID), durvalumab (1500 mg Q4W), and tremelimumab drug (75 mg IV Q4W) in the treatment of tumors, for four months and this study has revealed that if immune system targeting therapy is combined with Olaparib it acts synergistically and can be a good option for the treatment of solid tumors and this study is continued to check the further benefits and stability of this 86
Mesothelin and Pembrolizumab- Phase-1 study conducted to know the efficacy of CAR-T cell therapy derived Mesothelin and PD-1 inhibitor Pembrolizumab combination for the treatment of tumors of BC. As per the data of this trial, this combination carries enough potential to treat solid tumors without showing severe adverse 89
Eribulin and Pembrolizumab- Eribulin(1.4mg) and pembrolizumab (200mg) combination have shown promising results in metastatic-TNBNC. Phase- 1b(n=7)/II(n=160) clinical trial conducted to “September 21, 2015, to July 31, 2019” in 167 patients’ it has shown that this combination is well tolerated and shows anticancer activity in a 1-3L setting in metastatic-TNBC and shows limited side effects like nausea, fatigue, alopecia, constipation, and peripheral sensory neuropathy.91
Selinexor and Carboplatin/Paclitaxel- Open-label and single-center phase 1b study (ClinicalTrial.gov identifier: NCT02419495) was conducted to assess the safety profile, dose-limiting toxicity, and the maximum amount of dose that can be administered to the patients with solid tumors. Selinexor (60mg orally twice every week) is well tolerated when combined with carboplatin/Paclitaxel (175mg/intra-venous every 3 weeks) and shows visible clinical activity against solid 90
Ipilimumab and Nivolumab- Clinical trial, ICON (CA209-9FN), Phase IIb study conducted to examine the tolerability and efficacy to the anti-PD-1 drug) (Nivolumab and (anti-CTLA-4 drug) Ipilimumab. This study was conducted on seventy-five subjects and if this trial will show well tolerability and positive clinical effects against cancer this study will be conducted further and can prove to be the best therapeutic option for the 80
Vaccines for Breast cancer
Clinical trials for different types of vaccines as mentioned in Table no 02, are getting evaluated for BC, but it hasn’t given significant benefits. Despite the unsatisfied data obtained at present, studies of vaccines used in combination with anti-HER2 monoclonal antibodies have shown positive outcomes.
Table 2: Types of vaccines which are under clinical trial in different phases for the treatment of as Breast cancer well as for the prevention of reoccurrences of Breast cancer.
| S.No | Type of Vaccine |
| 1 | Peptide-based vaccine93 |
| 2 | Whole tumor-cell vaccine94 |
| 3 | Gene-based vaccine95 |
| 4 | Dendritic-cell-based vaccine96 |
| 5 | Nanotechnology on BC vaccine97 |
| 6 | Carbohydrate-Antigen vaccine98 |
Peptide Vaccines
Breast cancer patients are being studied for the role of CD8+ T cell-eliciting vaccines.
Human Epidermal Receptor-2(HER2) Peptide Vaccine- Randomized, single-blinded, and multi-center phase II trial, of this vaccine, confirms the data about GP2 &AE37 (both are HER2-derived vaccines) GP2 induces CD8+ response and AE37 induces CD4+ response against the HER This study shows that GP2 and AE37 are safe and are associated with improved. If GP2 and AE37 vaccines are given with or differently for specific categories of BC patients based on their condition there are chances, that these vaccines can show positive results.92
FRα Peptide Vaccine- Phase-I clinical trial of this vaccine, conducted at Mayo Clinic in Rochester, and this vaccine was administered to 8 women suffering from BC along with cyclophosphamide. This vaccine targets CD4Tcells whose activity is regulated by HLA class II which is specifically expressed on blood-stem cells and this ensures the safety profile of the There are minimal untoward and no severe unanticipated toxicities. Anti-tumor immunity of the vaccine can’t be verified from the data, but it supports the continuation of the production of this vaccine to prevent re-occurrence or used in combination with immune checkpoint inhibitors.90
Glycopeptide Vaccine- Mag-Tn-3 vaccine induces responses against tumor Tn (tumor-with carbohydrate antigen) which is found on carcinoma This vaccine induces levels of Tn-specific antibodies in patients of BC. The vaccine immunized seven patients and results showed that patients.94
Folate binding protein (FBP) peptide Vaccines- FBP-derived vaccines E39 &E39՛ elicit strong immune results in both in-vitro and vivo and can overstimulate the immune E39 possesses more immunostimulatory properties than then the other. These vaccines are safe with a low toxicity profile that is less than 2.93
Dendritic-cell Vaccines
Antigen-presenting cells called dendritic cells participate in the activation of the acquired immune system, these cells recognize antigens on tumor cells and initiate phagocytosis. These cells can be activated in vitro which can be modified by using recombinant viral vectors.96
Dendritic cell vaccines are proven to be a game changer in anti-tumor immunity and play a huge role in immunotherapy. Results of phase-1 and phase-2 studies are satisfactory and it is safe, well tolerated, and has a practical approach for the treatment of tumors, however confirmation of the safety of this vaccine can be assessed after mass immunization in a bigger population,97however manufacturing dendritic cell vaccine is a time consuming and expensive process and faces technical challenges with limited efficacy.
Gene-based Vaccines
Recombinant Viral Vector
The customized vaccine Ankara (MVA), which contains the Twist gene and the TRIad of co-stimulant substances (B7-1, ICAM-1 and LFA-3; TRICOM), has been evaluated in BC patients and has been proven to produce CD8+ as well as CD4+ T cell responses against the Twist factor and ultimately treats BC by reducing the growth of the 107
Bacterial Plasmid Vector
Immunostimulatory molecules, such as toll-like receptors, are combined with DNA vaccines. DNA vaccines that target HER2 with the TLR9 agonist exhibited anti-cancer activity and dependent cytotoxicity in animals.108 To help Antigen-presenting cells to be able to recognize tumor antigens, another DNA vaccine has been designed by joining the cellular part of CTLA-4 to HER-2/Neu.
Conclusion
Breast cancer remains the most commonly diagnosed cancer worldwide, as per the 2020 WHO report, and conventional systemic therapies continue to offer limited long-term outcomes. Immunotherapy, currently the fifth most commonly used treatment modality in oncology, has demonstrated promising results in various tumor types. In breast cancer, particularly in metastatic triple-negative cases, PD-1/PD-L1 checkpoint inhibitors have shown encouraging clinical outcomes. Since 2014, the FDA has approved four immunotherapeutic agents in this category, with additional checkpoint inhibitors—such as CTLA-4 inhibitors—currently under clinical investigation.
Combination therapies involving immunotherapy alongside targeted or hormonal agents have yielded enhanced clinical benefits, showing synergy in treatment response. Furthermore, cancer vaccines are under active research and clinical trials, holding potential as palliative or even preventive options in breast cancer management.
However, despite these advancements, immunotherapy faces several limitations and challenges. These include variability in patient response, lack of predictive biomarkers, immune-related adverse events, and the high cost of treatment. In breast cancer, especially, immunotherapy is not yet universally effective across all subtypes. Tumor heterogeneity and an immunosuppressive tumor microenvironment may limit therapeutic success. Moreover, access to immunotherapy is often restricted by regulatory, logistical, and economic barriers. Continued research is essential to optimize patient selection, refine combination strategies, and overcome resistance mechanisms. With ongoing advancements and clinical trials, immunotherapy is expected to play an increasingly integral role in the future of breast cancer treatment.
Acknowledgement
Authors thanks to Amity Institute of Pharmacy, Amity University Lucknow, for all the support required for the compilation of this review paper.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Authors Contributions
- Utkarsh Upadhyay: Writing, reviewing and editing
- Vivek Srivastava: Conceptualizing, writing & review
- Neha Mathur: Writing, reviewing
- Somesh Thapliyal: Data collection, editing
References
- DeSantis CE, Fedewa SA, Goding Sauer A, Kramer JL, Smith RA, Jemal A. Breast cancer statistics: Convergence of incidence rates between Black and White women. CA Cancer J Clin. 2016;66(1):31-42.
CrossRef - Liao L. Inequality in breast cancer: global statistics from 2022 to 2050. The Breast. 2025; 79:103851.
CrossRef - Balenger A, Seth G, Bhattarai S, Collin LJ, McCullough L, Gogineni K, Subhedar P, Ellison C, Khan U, Swahn MH, Aneja R. Barriers to breast cancer screening in Atlanta, GA: results from the Pink Panel survey at faith-based institutions. Cancer Causes & Control. 2022;33(12):1465-1472.
CrossRef - Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394-424.
CrossRef - Jelinek T, Mihalyova J, Kascak M, Duras J, Hajek R. PD-1/PD-L1 inhibitors in hematological malignancies: update 2017. 2017;152(3):357-370.
CrossRef - Topalian SL, Hodi FS, Brahmer JR. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med. 2012;366(26):2443-2454.
CrossRef - Savas P, Salgado R, Denkert C, Sotiriou C, Darcy PK, Smyth MJ, Loi S. Clinical relevance of host immunity in breast cancer: from TILs to the clinic. Nat Rev Clin Oncol. 2016;13(4):228-241.
CrossRef - Criscitiello C, Esposito A, Trapani D, Curigliano G. Prognostic and predictive value of tumor infiltrating lymphocytes in early breast cancer. Cancer Treat Rev. 2016;50:205-207.
CrossRef - Emens LA. Breast cancer immunobiology driving immunotherapy: vaccines and immune checkpoint blockade. Expert Rev Anticancer Ther. 2012;12(12):1597-1611.
CrossRef - Dzinic SH, Bernardo MM, Oliveira DSM, Wahba M, Sakr W, Sheng S. Tumor suppressor maspin as a modulator of host immune response to cancer. Bosn J Basic Med Sci. 2015;15(4):1.
CrossRef - Smyth MJ, Dunn GP, Schreiber RD. Cancer immunosurveillance and immunoediting: the roles of immunity in suppressing tumor development and shaping tumor immunogenicity. Adv Immunol. 2006;90:1-50.
CrossRef - Mittal D, Gubin MM, Schreiber RD, Smyth MJ. New insights into cancer immunoediting and its three component phases—elimination, equilibrium, and escape. Curr Opin Immunol. 2014;27:16-25.
CrossRef - Schreiber RD, Old LJ, Smyth MJ. Cancer immunoediting: integrating immunity’s roles in cancer suppression and promotion. 2011;331(6024):1565-1570.
CrossRef - Zumwalde NA, Haag JD, Sharma D, Mirrielees JA, Wilke LG, Gould MN, Gumperz JE. Analysis of immune cells from human mammary ductal epithelial organoids reveals Vδ2+ T cells that efficiently target breast carcinoma cells in the presence of bisphosphonate. Cancer Prev Res. 2016;9(4):305-316.
CrossRef - Azizi E, Carr AJ, Plitas G, Cornish AE, Konopacki C, Prabhakaran S, Nainys J, Wu K, Kiseliovas V, Setty M, Choi K. Single-cell map of diverse immune phenotypes in the breast tumor microenvironment. 2018;174(5):1293-1308.
CrossRef - Hussein MR, Hassan HI. Analysis of the mononuclear inflammatory cell infiltrate in the normal breast, benign proliferative breast disease, in situ and infiltrating ductal breast carcinomas: preliminary observations. J Clin Pathol. 2006;59(9):972-977.
CrossRef - Dieci MV, Mathieu MC, Guarneri V, Conte P, Delaloge S, Andre F, Goubar A. Prognostic and predictive value of tumor-infiltrating lymphocytes in two phase III randomized adjuvant breast cancer trials. Ann Oncol. 2015;26(8):1698-1704.
CrossRef - Ibrahim EM, Al-Foheidi ME, Al-Mansour MM, Kazkaz GA. The prognostic value of tumor-infiltrating lymphocytes in triple-negative breast cancer: a meta-analysis. Breast Cancer Res Treat. 2014;148(3):467-476.
CrossRef - Dine J, Deng CX. Mouse models of BRCA1 and their application to breast cancer research. Cancer Metastasis Rev. 2013;32(1-2):25-37.
CrossRef - Tan-Wong SM, French JD, Proudfoot NJ, Brown MA. Dynamic interactions between the promoter and terminator regions of the mammalian BRCA1 gene. Proc Natl Acad Sci U S A. 2008;105(13):5160-5165.
CrossRef - Hegan DC, Lu Y, Stachelek GC, Crosby ME, Bindra RS, Glazer PM. Inhibition of poly (ADP-ribose) polymerase down-regulates BRCA1 and RAD51 in a pathway mediated by E2F4 and p130. Proc Natl Acad Sci U S A. 2010;107(5):2201-2206.
CrossRef - Sánchez H, Paul MW, Grosbart M, van Rossum-Fikkert SE, Lebbink JH, Kanaar R, Houtsmuller AB, Wyman C. Architectural plasticity of human BRCA2–RAD51 complexes in DNA break repair. Nucleic Acids Res. 2017;45(8):4507-4518.
CrossRef - Ahmed M, Rahman N. ATM, and breast cancer susceptibility. 2006; 25:5906-5911.
CrossRef - Goldgar DE, Healey S, Dowty JG. Rare variants in the ATM gene and risk of breast cancer. Breast Cancer Res. 2011;13(4).
CrossRef - Taylor AMR, Byrd PJ, McConville CM. Molecular pathology of ataxia telangiectasia. J Clin Pathol. 2005; 58:1009-1015.
CrossRef - Kwong A, Ho CY, Au CH, Tey SK, Ma ES. Germline RAD51C and RAD51D mutations in high-risk Chinese breast and/or ovarian cancer patients and families. J Pers Med. 2024;14(8):866.
CrossRefv - Xia B, Dorsman JC, Ameziane N. Fanconi anemia is associated with a defect in the BRCA2 partner PALB2. Nat Genet. 2007;39(2):159-161.
CrossRef - Liu C, Zheng Y, Lu Y. Application of next-generation sequencing in detection of breast cancer susceptibility genes BRCA1/2, TP53, and PTEN germline mutation. Chin J Lab Med. 2009;42(2):98-103.
- Kurian AW, Ward KC, Hamilton AS. Genetic testing and results in a population-based cohort of breast cancer patients and ovarian cancer patients. J Clin Oncol. 2019;37(15):1305-1315.
CrossRef - Gao X, Huang W, Zhang Y. PTENP1/miR-20a/PTEN axis contributes to breast cancer progression by regulating PTEN via PI3K/AKT pathway. J Exp Clin Cancer Res. 2019;38(1).
CrossRef - Celebiler Cavusoglu A, Kilic S, Kaya EO. Predicting invasive phenotype with CDH1, CDH13, CD44, and TIMP3 gene expression in primary breast cancer. Cancer Sci. 2009;100(12):2341-2345.
CrossRef - Chen J, Lindblom A. Germline mutation screening of the STK11/LKB1 gene in familial breast cancer with LOH on 19p. Clin Genet. 2000;57(5):394-397.
CrossRef - Gallo F, Morale MC, Sambataro D. The immune system response during development and progression of carcinogen-induced rat mammary tumors: Prevention of tumor growth and restoration of immune system responsiveness by thymopentin. Breast Cancer Res Treat. 1993;27(3):221-237.
CrossRef - Campbell MJ, Scott J, Maecker HT, Park JW, Esserman LJ. Immune dysfunction and micrometastases in women with breast cancer. Breast Cancer Res Treat. 2005;91(2):163-171.
CrossRef - Welte T, Kim IS, Tian L. Oncogenic mTOR signalling recruits myeloid-derived suppressor cells to promote tumour initiation. Nat Cell Biol. 2016;18(6):632-644.
CrossRef - Mutebi M, Edge J, Stedman M. Breast cancer treatment: A phased approach to implementation. 2020;126(S10):2365-2378.
CrossRef - Fridman W H, Zitvogel L, Sautès-Fridman C, Kroemer G. The immune contexture in cancer prognosis and treatment. Nature Reviews Clinical Oncology. 2017; 14(12): 717-734.
CrossRef - Gong J, Chehrazi-Raffle A, Reddi S, Salgia R. Development of PD-1 and PD-L1 inhibitors as a form of cancer immunotherapy: a comprehensive review of registration trials and future considerations. J Immunother Cancer. 2018; 6:1-8.
CrossRef - Rafiq S, Hackett CS, Brentjens RJ. Engineering strategies to overcome the current roadblocks in CAR T cell therapy. Nat Rev Clin Oncol. 2020;17(3):147-167.
CrossRef - Yang M, Tang X, Fang Y. Tandem CAR-T cells targeting CD70 and B7-H3 exhibit potent preclinical activity against multiple solid tumors. 2020;10(17):7622-7634.
CrossRef - Zah E, Lin MY, Anne SB, Jensen MC, Chen YY. T cells expressing CD19/CD20 bispecific chimeric antigen receptors prevent antigen escape by malignant B cells. Cancer Immunol Res. 2016;4(6):498-508.
CrossRef - Kagoya Y, Tanaka S, Guo T. A novel chimeric antigen receptor containing a JAK-STAT signaling domain mediates superior antitumor effects. Nat Med. 2018;24(3):352-359.
CrossRef - Till B, Jensen M, Wang J. Adoptive immunotherapy for indolent non-Hodgkin lymphoma and mantle cell lymphoma using genetically modified autologous CD20-specific T cells. 2008;112(6):2261.
CrossRef - Frey NV, Shaw PA, Hexner EO. Optimizing chimeric antigen receptor T-cell therapy for adults with acute lymphoblastic leukemia. J Clin Oncol. 2020;38(5):415-422.
CrossRef - Lee DW, Kochenderfer JN, Stetler-Stevenson M, et al. T cells expressing CD19 chimeric antigen receptors for acute lymphoblastic leukemia in children and young adults: a phase 1 dose-escalation trial. 2015;385(9967):517-528.
CrossRef - Xia L, Zhang D, Du R. EGFR-targeted CAR-T cells are potent and specific in suppressing triple-negative breast cancer both in vitro and in vivo. Clin Transl Immunol. 2020;9(5):e1135.
CrossRef - Luangwattananun P, Junking M, Chaicharoenaudomrung N. Fourth-generation chimeric antigen receptor T cells targeting folate receptor alpha antigen expressed on breast cancer cells for adoptive T cell therapy. Breast Cancer Res Treat. 2021;186(1):25-36.
CrossRef - Neel BG, Gu H, Pao L. The ‘Shp’ing news: SH2 domain-containing tyrosine phosphatases in cell signaling. Trends Biochem Sci. 2003;28(6):284-293.
CrossRef - Carreno BM, Collins M. The B7 family of ligands and its receptors: new pathways for costimulation and inhibition of immune responses. Annual review of immunology. 2002;20(1):29-53.
CrossRef - Li C, Ge B, Nicotra T. ADAP and SKAP55 deficiency suppresses PD-1 expression in CD8+ cytotoxic T lymphocytes for enhanced anti-tumor immunotherapy. EMBO Mol Med. 2015;7(6):754-769.
CrossRef - Sanmamed MF, Chen L. Inducible expression of B7-H1 (PD-L1) and its selective role in tumor site immune modulation. Cancer J. 2014;20(4):256-261.
CrossRef - Salmaninejad A, Valilou SF, Shabgah AG. PD-1, and cancer: Molecular mechanisms and polymorphisms. 2022;70(2):73-86.
CrossRef - Dai S, Jia R, Zhang X, Fang Q, Huang L. The PD-1/PD-Ls pathway and autoimmune diseases. Cell Immunol. 2014;290(1):72-79.
CrossRef - McNally B, Ye F, Willette M, Flaño E. Local blockade of epithelial PD-L1 in the airways enhances T cell function and viral clearance during influenza virus infection. J Virol. 2013;87(23):12916-12924.
CrossRef - Rollins MR, Gibbons Johnson RM. CD80 expressed by CD8+ T cells contributes to PD-L1-induced apoptosis of activated CD8+ T cells. J Immunol Res. 2017;2017(1):7659462.
CrossRef - Mansfield AS, Aubry MC, Moser JC. Temporal and spatial discordance of programmed cell death-ligand 1 expression and lymphocyte tumor infiltration between paired primary lesions and brain metastases in lung cancer. Ann Oncol. 2016;27(10):1953-8.
CrossRef - Kolch W. Coordinating ERK/MAPK signalling through scaffolds and inhibitors. Nat Rev Mol Cell Biol. 2005 Nov;6(11):827-37.
CrossRef - Herrero A, Pinto A, Colón-Bolea P, Casar B, Jones M, Agudo-Ibáñez L, Vidal R, Tenbaum SP, Nuciforo P, Valdizán EM, Horvath Z. Small molecule inhibition of ERK dimerization prevents tumorigenesis by RAS-ERK pathway oncogenes. Cancer Cell. 2015;28(2):170-182.
CrossRef - Marotta LL, Almendro V, Marusyk A, Shipitsin M, Schemme J, Walker SR, Bloushtain-Qimron N, Kim JJ, Choudhury SA, Maruyama R, Wu Z. The JAK2/STAT3 signaling pathway is required for growth of CD44+ CD24–stem cell–like breast cancer cells in human tumors. J Clin Invest. 2011;121(7):2723-2735.
CrossRef - Zerdes I, Wallerius M, Sifakis E, Wallmann T. STAT3 activity promotes programmed-death ligand 1 expression and suppresses immune responses in breast cancer. Cancers. 2020;12(5):1458.
CrossRef - C F-J. Transcription factor STAT3 as a prognostic marker and therapeutic target in cancer. J Clin Oncol. 2013;31(8):918–927.
CrossRef - Yu H, R J-N. The STATs of cancer—new molecular targets come of age. Nat Rev Cancer. 2004;4(3):97–105.
CrossRef - Schindler C, Levy DE. JAK-STAT signaling: From interferons to cytokines. J Biol Chem. 2007;282(28):20189–20193.
CrossRef - Koromilas AE, Sexl V. The tumor suppressor function of STAT1 in breast cancer. JAK-STAT. 2013;2(2):e23353.
CrossRef - Marzec M, Zhang Q, Goradia A, Raghunath PN, Liu X, Paessler M, Wang HY, Wysocka M, Cheng M, Ruggeri BA, Wasik MA. Oncogenic kinase NPM/ALK induces through STAT3 expression of immunosuppressive protein CD274 (PD-L1, B7-H1). Proc Natl Acad Sci U S A. 2008;105(52):20852-7.
CrossRef - Sasidharan Nair V, Toor SM, Ali BR, Elkord E. Dual inhibition of STAT1 and STAT3 activation downregulates expression of PD-L1 in human breast cancer cells. Expert Opin Ther Targets. 2014;22(6):547-57.
CrossRef - Jing N, Tweardy DJ. Targeting Stat3 in cancer therapy. Anti-cancer Drugs. 2005;16(6):601-7.
CrossRef - Jögi A, Ehinger A, Hartman L, Alkner S. Expression of HIF-1α is related to a poor prognosis and tamoxifen resistance in contralateral breast cancer. PLoS One. 2019;14(12): e0226150.
CrossRef - Ortmann B, Druker J, Rocha S. Cell cycle progression in response to oxygen levels. Cell Mol Life Sci. 2014;71(18):3569-3582.
CrossRef - G S. Defining the role of hypoxia-inducible factor 1 in cancer biology and therapeutics. Oncogene. 2010;29(10):1856-1863.
CrossRef - Bos R. Levels of hypoxia-inducible factor-1α independently predict prognosis in patients with lymph node-negative breast carcinoma. Cancer. 2003;97(6):1573-1581.
CrossRef - Shehade H, O.-E. G. Hypoxia in the intestine or solid tumors: A beneficial or deleterious alarm signal? Eur J Immunol. 2014;44(9):2550-2557.
CrossRef - Noman MZ, Chouaib S. Targeting hypoxia at the forefront of anticancer immune responses. Oncoimmunology. 2014;3(12): e954463.
CrossRef - Noman MZ, Desantis G, Janji B. PD-L1 is a novel direct target of HIF-1α, and its blockade under hypoxia enhanced MDSC-mediated T cell activation. J Exp Med. 2014;211(5):781-790.
CrossRef - Polizzi K, Reboldi JP-N. Integrating canonical and metabolic signaling programs in the regulation of T cell responses. Nat Rev Immunol. 2022;22(5):369-383.
- Brown J, W-N R. Exploiting tumor hypoxia in cancer treatment. Nat Rev Cancer. 2004;4(6):437-447.
CrossRef - Chatterjee A, Rodger EJ, Ahn A. Marked global DNA hypomethylation is associated with constitutive PD-L1 expression in melanoma. iScience. 2018; 4:312-325.
CrossRef - Dirix LY. Avelumab, an anti-PD-L1 antibody, in patients with locally advanced or metastatic breast cancer: A phase 1b JAVELIN Solid Tumor study. Breast Cancer Res Treat. 2018;167(3):671-686.
CrossRef - Gianni L. Pathologic complete response (pCR) to neoadjuvant treatment with or without atezolizumab in triple-negative, early high-risk, and locally advanced breast cancer: NeoTRIP Michelangelo randomized study. Ann Oncol. 2022;33(5):534-543.
CrossRef - Cortes J. Pembrolizumab plus chemotherapy in advanced triple-negative breast cancer. N Engl J Med. 2022;387(3):217-226.
CrossRef - Domchek SM. Olaparib and durvalumab in patients with germline BRCA-mutated metastatic breast cancer (MEDIOLA): An open-label, multicentre, phase 1/2, basket study. Lancet Oncol. 2020;21(9):1155-1164.
CrossRef - Tanizaki J. Open-label phase II study of the efficacy of nivolumab for cancer of unknown primary. Ann Oncol. 2022;33(2):216-226.
CrossRef - Vonderheide RH. Tremelimumab in combination with exemestane in patients with advanced breast cancer and treatment-associated modulation of inducible costimulator expression on patient T cells. Clin Cancer Res. 2010;16(13):3485-3494.
CrossRef - Adams S. A multicenter phase II trial of ipilimumab and nivolumab in unresectable or metastatic metaplastic breast cancer: Cohort 36 of dual anti-CTLA-4 and anti-PD-1 blockade in rare tumors (DART, SWOG S1609). Clin Cancer Res. 2022;28(2):271-278.
CrossRef - Schmid P. Atezolizumab plus nab-paclitaxel as first-line treatment for unresectable, locally advanced, or metastatic triple-negative breast cancer (IMpassion130): Updated efficacy results from a randomized, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2022;21(1):44-59.
CrossRef - Fumet JD, Limagne E, Thibaudin M. Precision medicine phase II study evaluating the efficacy of double immunotherapy by durvalumab and tremelimumab combined with olaparib in patients with solid cancers and carriers of homologous recombination repair genes mutation in response or stable after olaparib treatment. BMC Cancer. 2020;20(1):748.
CrossRef - Adusumilli PS, Zauderer MG, Rusch VW. A phase I trial of regional mesothelin-targeted CAR T-cell therapy in patients with malignant pleural disease, in combination with the anti–PD–1 agent pembrolizumab. Cancer Discov. 2021;11(11):2748–2763.
CrossRef - Tolaney SM, Barroso-Sousa R, Keating NL. Eribulin plus pembrolizumab in patients with metastatic triple-negative breast cancer (ENHANCE 1): A phase Ib/II study. Clin Cancer Res. 2021;27(11):3061–3068.
CrossRef - Thein KZ, Twardowski PW, Greco SJ. Selinexor in combination with carboplatin and paclitaxel in patients with advanced solid tumors: Results of a single-center, multi-arm phase Ib study. Invest New Drugs. 2022;40(2):290–299.
CrossRef - Kyte JA, Andresen NK, Russnes HG. ICON: A randomized phase IIb study evaluating immunogenic chemotherapy combined with ipilimumab and nivolumab in patients with metastatic hormone receptor-positive breast cancer. J Transl Med. 2020; 18:1–10.
CrossRef - Brown TA, Mittendorf EA, Perez SA. Prospective, randomized, single-blinded, multi-center phase II trial of two HER2 peptide vaccines, GP2 and AE37, in breast cancer patients to prevent recurrence. Breast Cancer Res Treat. 2020;181(2):391–401.
CrossRef - Kalli KR, Block MS, Kasi PM. The folate receptor alpha peptide vaccine generates immunity in breast and ovarian cancer patients. Clin Cancer Res. 2018;24(13):3014.
CrossRef - Rosenbaum P, Schick J, Wijnen J. The fully synthetic glycopeptide MAG-Tn3 therapeutic vaccine induces tumor-specific cytotoxic antibodies in breast cancer patients. Cancer Immunol Immunother. 2020;69(5):703-716.
CrossRef - Vreeland TJ, Clifton GT, Hale DF. Phase Ib trial of folate binding protein (FBP)-derived peptide vaccines, E39 and an attenuated version, E39′: An analysis of safety and immune response. Clin Immunol. 2018; 192:6.
CrossRef - Gelao L, Criscitiello C, Esposito A. Dendritic cell-based vaccines: Clinical applications in breast cancer. 2014; 6(3):349-360.
CrossRef - Zhang W, Qiao S, Liu Y. Phase I/II clinical trial of a Wilms’ tumor 1-targeted dendritic cell vaccination-based immunotherapy in patients with advanced cancer. Cancer Immunol Immunother. 2019;68(1):121-130.
CrossRef - Kwilas AR, Ardiani A, Dirmeier U, Wottawah C, Schlom J, Hodge JW. A poxviral-based cancer vaccine targeting the transcription factor twist inhibits primary tumor growth and metastases in a model of metastatic breast cancer and improves survival in a spontaneous prostate cancer model. 2015;6(29):28194.
CrossRef - Aurisicchio L, Peruzzi D, Conforti A. Treatment of mammary carcinomas in HER-2 transgenic mice through a combination of genetic vaccine and an agonist of Toll-like receptor 9. Clin Cancer Res. 2009;15(5):1575.
CrossRef
Abbreviations List
Breast Cancer-BC
World health Organization-WHO
Food and Drug administration-FDA
Breast Cancer gene 1-BRCA1
Breast Cancer gene 2- BRCA2
Ataxia-telangiectasia-ATM
Partner And Localizer of BRCA2-PALB2
Tumor Protein 53-TP53
Checkpoint Kinase-2-CHEK2
Phosphatase and TENsin homolog deleted on chromosome10-PTEN
Cadherin 1-CDH1
Serine/Threonine kinase 11-STK11
Interlukin2-IL-2
Transforming growth factor-TGF







