Manuscript accepted on :25-09-2025
Published online on: 29-09-2025
Plagiarism Check: Yes
Reviewed by: Dr. Praveen Kumar S E
Second Review by: Dr. Soujania Singh Gunasingh
Final Approval by: Dr. Anton R Kiselev
Chimedragchaa Chimedtseren1 , Dejidmaa Buyantogtokh1*
, Dejidmaa Buyantogtokh1* , Myadagbadam Urtnasan2
, Myadagbadam Urtnasan2 , Anu Altangerel1
, Anu Altangerel1 , Uuganbayar Baatartsogt1
, Uuganbayar Baatartsogt1 and Erdenechimeg Chuluunbaatar2
and Erdenechimeg Chuluunbaatar2
1Department of Pharmacology, Research center, Institute of Traditional Medicine and Technology, Ulaanbaatar, Mongolia
2Department of Chemistry and Technology, Research center, Institute of Traditional Medicine and Technology, Ulaanbaatar, Mongolia
Corresponding Author E-mail: b.dejidmaa@yahoo.com
DOI : https://dx.doi.org/10.13005/bpj/3240
Abstract
Traditional Mongolian medicine encompasses various practices for treating hemorrhoids. One of the pharmaceutical formulations is Zidraga-6, which is composed of six active remedies. Aim of the study: The study sought to explore the phytochemical composition of Zidraga-6 and to thoughtfully evaluate its potential effects in a hemorrhoid model using rats. Methods: Thin-layer chromatography (TLC) was employed to identify biologically active compounds, and their concentrations were quantified using UV spectrophotometric methods. The experiment was conducted using a methodology established with a croton oil mixture. In a designated group, a hemorrhoid model was successfully induced in healthy animals using croton oil. Following a 24-hour period after the induction of the model, the Zidraga-6 herbal formulation was administered once daily over the course of eight days. Results: Using thin-layer chromatography (TLC), we identified oleonalic acid, gallic acid, quercetin, capsaicin, and p-methoxycinnamate in the Zidraga-6 capsules. UV spectrophotometric analysis determined the content of bioactive compounds in Zidarga‑6 capsules to be 1.89 ± 0.19 % total triterpene saponins, 1.30 ± 0.12 % flavonoids, and 12.59 ± 0.68 % phenolic compounds. Using thin-layer chromatography (TLC), we identified oleonalic acid, gallic acid, quercetin, capsaicin, and p-methoxycinnamate in the Zidraga-6 capsules. UV spectrophotometric analysis determined the content of bioactive compounds in Zidarga‑6 capsules to be 1.89 ± 0.19 % total triterpene saponins, 1.30 ± 0.12 % flavonoids, and 12.59 ± 0.68 % phenolic compounds. The results of this study indicated that all doses of Zidraga-6 capsules effectively reduced the inflammatory cytokine IL-6 in the rat hemorrhoid model. Furthermore, the levels of the cytokine IL-1β were significantly lowered with the 130 mg/kg and 325 mg/kg doses. Histopathological examination of the recto anal tissue demonstrated that the structural changes, inflammation, and necrosis were significantly diminished with the 130 mg/kg and 325 mg/kg doses of Zidraga-6 capsules when compared to the untreated hemorrhoid model. Conclusions: Zidraga‑6 capsules contain biologically active constituents such as flavonoids, phenolic compounds, and triterpene saponin. Zidraga-6 has demonstrated efficacy in treating hemorrhoids by reducing pro-inflammatory cytokines and decreasing rectal damage.
Keywords
Croton oil; Hemorrhoid model; Inflammatory cytokine; Total flavonoid; Triterpene saponin
Download this article as:| Copy the following to cite this article: Chimedtseren C, Buyantogtokh D, Urtnasan M, Altangerel A, Baatartsogt U, Chuluunbaatar E. Evaluation of Anti-Hemorrhoidal Activity and Phytochemical Study of Zidarga-6 Capsule. Biomed Pharmacol J 2025;18(3). |
| Copy the following to cite this URL: Chimedtseren C, Buyantogtokh D, Urtnasan M, Altangerel A, Baatartsogt U, Chuluunbaatar E. Evaluation of Anti-Hemorrhoidal Activity and Phytochemical Study of Zidarga-6 Capsule. Biomed Pharmacol J 2025;18(3). Available from: https://bit.ly/4o0gNSc |
Introduction
Hemorrhoids are the most common disease in the world, and it is estimated that 55-60% of then all population will suffer from some symptom of hemorrhoids at some point in their lives. Several factors can contribute to the development of hemorrhoids, including a sedentary lifestyle, obesity, constipation, and pregnancy.1-3 These conditions can result in increased pressure in the pelvic and rectal veins, causing blood to leak from surrounding tissues due to the dilation and distortion of the blood vessel walls. Consequently, this may lead hematochezia. This disease occurs due to muscle contractions in the inner lining and migration of the vascular fossa of the anus. Symptoms are manifested by anal bleeding, palpable nodules, pain, itching, and pathological exude. In modern medicine, hemorrhoids treatment is based on lifestyle changes, local and topical medications, and surgery.4-5
Four Tantra of Medicine, which serves as the foundation of Mongolian traditional medicine, identifies several conditions that can lead to the development of hemorrhoids. These factors include injuries to the anal canal, difficulties with bowel and urinary movements, and prolonged periods of horseback riding. Each of these elements can significantly contribute to the formation of hemorrhoids in the rectum.6-7 Zidraga-6 is a commonly prescribed medication for this disease. Zidraga-6 is known for its yellowish color and bitter taste. It has been used in traditional medicine to treat superficial and internal hemorrhoids and those related to imbalances of wind, bile, and blood. This formulation is recognized for its effectiveness in alleviating various hemorrhoid conditions.7-9 The ‘Zidraga-6’ formulation, widely utilized in traditional medicine, was selected, technologically developed, and subsequently transformed into a capsule dosage form, named ‘Zidraga-6’. 6,8
 |
Figure 1: Herbal Ingredients of Zidraga-6 CapsuleClick here to view Figure |
The berries of Embelia ribes contain several chemical constituents, including embelin, volatile oil, fixed oil, resin, tannins, christembine, and phenolic acids such as gallic acid, caffeic acid, vanillic acid, chlorogenic acid, cinnamic acid, and o-coumaric acid.10 “The primary constituents of the raw material of Kaempferia galanga L are volatile compounds. The essential oil extracted from it contains p-methoxycinnamate, borneol, cineole, and δ-3-carene. A chemical analysis conducted by Archana P. and colleagues identified 38 different essential oil components, with p-methoxycinnamate being the most predominant.11
Cynanchum thesioides contains diverse phytochemical constituents, including triterpenoids, flavonols, steroids, phenolic acids, amyrin, and oleanolic acid.12-14 Extensive phytochemical investigations of Terminalia chebula have confirmed the presence of a diverse array of compounds, including flavonoids, gallic acid, ellagic acid, chebulic acid, tannins, and other bioactive constituents.15 “The fruits of Capsicum annuum are notably rich in bioactive compounds, including capsaicinoids, carotenoids, essential vitamins, minerals, and a variety of flavonoids such as quercetin, luteolin, kaempferol, apigenin, and myricetin, as well as more complex glycosylated forms like quercetin 3-O-α-L-rhamnopyranoside, luteolin 6-C-β-D-glucopyranoside-8-C-α-L-arabinopyranoside, and luteolin apiosyl-6-acetyl-glucoside.16-17
Atragene sibirica has been reported to contain a diverse array of bioactive constituents. The aerial parts of the plant are particularly rich in flavonoids (185 mg%), including kaempferol, quercetin, and isoquercitrin, as well as vitamin C (229 mg%). In addition, the plant contains several coumarins—such as esculetin, esculin, umbelliferone, and scopoletin—alongside dulcitol, four spirostanol glycosides, and a total alkaloid content of approximately 0.35%, comprising compounds such as saponins, delphinium alkaloids, and aconitine. Furthermore, cardiac glycosides are distributed throughout the plant, with a concentration of approximately 0.10%.18
The study aimed to investigate the phytochemical composition of Zidraga-6 and to evaluate its potential impact on the hemorrhoid model in rats.
Materials and Methods
Chemicals and reagents
The study utilized oleonalic acid, gallic acid, quercetin, capsaicin, and p‑methoxycinnamate as reference standards sourced from Macklin Biochemical Co., Ltd (Shanghai, China). The ELISA kits for IL-6 (interleukin-6), IL-1β (interleukin-1β) were purchased from MLBIO Biotechnology Co.Ltd (Shanghai, China) and used in this study.
Chemical analysis
Sample preparation
Preparation of the test extract: Add 1.0 g of Zidraga-6 sample to 10 ml of methanol, heat under reflux for 30 min, and filter. 8-10 drops of the filtrate were used for TLC.11 The system for performing TLC is shown in Table 1.
Table 1: The mobile phase for performing thin-layer chromatography
| № | Solvent system | Standard substance | Detection solution |
| 1. | Hexane-ethyl acetate (18:1) | P-Methoxycinnamate | Anisaldehyde-sulfuric acid solution |
| 2. | Toluone- ethyl acetate (7:3) | Oleanolic acid | Vanillin sulfuric acid solution |
| 3. | Hexane-ethyl acetate-formic acid (10:5:1) | Gallic acid | Ferric chloride solution |
| 4. | Toluene-chloroform-acetone (4:2:30) | Capsaicin | Iodine vapour |
| 5. | Hexane-ethyl acetate-acetic acid (12:6:1) | Quercetin | Aluminum chloride solution |
Sample preparation
Add 1.0 g of Zidraga-6 sample to 50 ml of 70% ethanol, heat under reflux for 60 minutes, and filter. The extract was diluted to 50 mL with 96% ethanol (solution A)
Amount of total triterpene saponins
0.4 ml of solution A was measured in a 10 ml tube, 0.4 ml of 5% solution of vanillin in acetic acid, and 2.4 ml of perchloric acid, and heated at 70°C for 15 min, and diluted to volume with ethyl acetate. The solution was measured at 550 nm. Triterpene saponins were expressed as equivalents [mg oleanolic acid/g extract].19
Amount of total phenolic
0.5 ml of solution A was measured in a 25 ml tube, 1 ml of Folin-Ciocalteu, 10 ml of distilled water, and 13.5 ml of 10.75% Na2CO3 solution were added. The solution was calculated at 760 nm. The polyphenolic was indicated as GA equivalents [mg GA/g extract].20
Amount of total flavonoids
1 g of the Zidraga-6 drug, which was extracted with 0.5% hydrochloric acid and 50 mL of 70% ethanol, and then ultrasonicated for 1 hour, and filtered. 4 mL test solution to a 25 mL volumetric flask, and added 2 mL of a 3% aluminium chloride solution and 19 mL of 70% ethanol. Allow the solution to react for 20 minutes before measuring the absorbance at 430 nm using a spectrophotometer. Use 70% ethanol as the reference solution. The total flavonoids were expressed as quercetin equivalents [mg quercetin/g extract].21,22
Ethics statement
This study utilised experimental animals, and the research was approved by the Medical Ethics Committee, as detailed in protocol No. 23/029 from the Ministry of Health of Mongolia. This study did not involve human participants, and therefore, informed consent was not required.
Pharmacological method
Two sets of experiments were undertaken to explore the potential anti-hemorrhoid effects. The first set focused on evaluating plasma inflammatory cytokine activity, while the second set aimed to assess the rectal index and the degree of inflammation through histopathological analysis. We divided the experimental .
In this study, male Wistar rats weighing between 200 and 250 grams were utilized for experimental purposes. To induce a model of hemorrhoid, a prepared mixture containing Croton oil was applied to the control group and experimental groups of animals. This mixture consisted of deionized water, pyridine, diethyl ether, and 6% Croton oil, combined in a ratio of 1:4:5:10. To ensure the integrity of the experiment, the rats were fasted the night before the induction of the hemorrhoid model. A sterile cotton swab, measuring 4 mm in diameter and soaked in 100 μl of the croton oil mixture, was inserted into the rectum of each rat to a depth of approximately 20 mm, where it was held for 10 seconds.23.24 Following this procedure, a linear oedema was observed to develop within 7 to 8 hours.
Twenty-four hours after the induction of the haemorrhoid model, all experimental groups of rats received their respective Zidraga-6 capsule (Zid) treatments at doses of 65, 130, and 325 mg/kg once daily for eight days. The group with hemorrhoids was administered only a saline solution.
On the 8th day, a procedure was conducted to collect 2-3 ml of blood through cardiac puncture. Subsequently, the levels of IL-1β and IL-6 cytokines in the blood samples were assessed using the ELISA method, providing valuable insights into the effects of the treatments.
In the second set of experiments, Zidraga-6 was administered orally once daily for a duration of eight days in three doses after the induction of the hemorrhoid model. On the eighth day, one hour post-treatment, the animals were euthanised under deep anaesthesia with isoflurane. Subsequently, the colon and rectum were carefully removed and weighed, with measurements taken at 20 mm. Additionally, the colon-rectum coefficient was assessed using an established formula.
Rectal coefficient =
Following determining the recto anal coefficient, the recto anal tissue was fixed in 10% formalin and subsequently analyzed through histopathological examination. This evaluation considered various factors, including inflammatory cell infiltration, necrosis, hemorrhage, and vasodilation.
Statistical analysis
The data are presented as mean ± standard deviation (SD). Statistical analyses were performed using SPSS version 22.0 (SPSS Inc., Chicago, IL, USA). The statistical significance of differences was evaluated using One-way ANOVA and the Kruskal-Wallis test, with a p-value of less than 0.05 indicating a statistically significant difference.
Results
Results of the detection of biologically active substances in Zidraga-6 capsule medicine.
The results of detecting biologically active substances such as p-methoxycinnamate, oleanolic acid, gallic acid, and capsaicin in Zidraga-6 capsule medicine by TLC are shown in Table 2.
Table 2: Identification of Bio-Active Compounds in The Zidraga-6 capsule
| № | Solvent system | Standard substance | Detection solution | Visible color | Rf value |
| 1. | Hexane-ethyl acetate (18:1) | P-Methoxycinnamate | Anisaldehyde-sulfuric acid solution | Purple | 0.26 |
| 2. | Toluone- ethyl acetate (7:3) | Oleanolic acid | Vanillin sulfuric acid solution | Pink | 0.82 |
| 3. | Hexane-ethyl acetate-formic acid (10:5:1) | Gallic acid | Ferric chloride solution | Dark blue | 0.24 |
| 4. | Toluene-chloroform-acetone (4:2:30) | Capsaicin | Iodine vapour | Yellow | 0.49 |
| 5. | Hexane-ethyl acetate-acetic acid (12:6:1) | Quercetin | Aluminum chloride solution | Yellow fluorescence | 0.31 |
The presence of p-methoxycinnamate, oleanolic acid, gallic acid, quercetin and capsaicin in the Zidraga-6 capsule was confirmed through Thin Layer Chromatography (TLC), with the following Rf values: 0.26 for p-methoxycinnamate, 0.82 for oleanolic acid, 0.24 for gallic acid, 0.31 for quercetin and 0.49 for capsaicin.
Polyphenolic compound, total triterpene saponin, and flavonoid contents
The results of the spectrophotometric determination of biologically active compounds in Zidraga-6 capsules are shown in Table 3.
Table 3: Total phenolics, flavonoids, and triterpene saponins in the Zidraga-6 capsule
| Biologically active compounds | Standard reagent | Standard curve equation | Content, % |
| Total flavonoids | Quercetin | y = 0. 9.6326x – 0.0075, R2=0.9976 | 10.55 ±0.16 |
| Total phenolic compounds | Gallic acid | y=0.083x +0.0013,R2=0.9957 | 12.59±0.68% |
| Total triterpene saponin | Oleanolic acid | y = 0.0416x + 0.0071, R2=0.9907 | 1.89 ± 0.16% |
The total triterpene saponin compound content in the Zidraga-6 capsule, measured from the regression equation of the calibration curve (y = 0.0463x + 0.049, R² = 0.9907), was 1.89 ± 0.16%. The content of flavonoids in the Zidraga-6 capsule formulation, in quercetin equivalent, measured using the reliable calibration curve’s regression equation (y = 9.6326x – 0.0075, r2 =0.9976), and is represented in quercetin equivalent was 1.3±0.12%. The total phenolic compound content in Zidraga-6 capsules was determined using the spectrophotometric method and expressed as gallic acid equivalents. The equation of the linear curve established with gallic acid was y=0.083x+0.0013, and the correlation coefficient R2=0.9957. The amount of polyphenolic compounds in the Zidraga-6 capsule preparation was 12.59±0.68%, as determined by spectrophotometry.
Effect of Zidraga-6 capsule on a Croton oil-induced hemorrhoid model
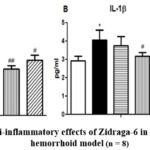 |
Figure 2: The anti-inflammatory effects of Zidraga-6 in a croton oil-induced hemorrhoid model (n = 8).Click here to view Figure |
All values are presented as means ± standard deviation (SD). The mean values across all groups were compared using one-way ANOVA, followed by post hoc tests. Statistical significance is indicated as follows: *p < 0.05 and **p < 0.01 compared to the normal control group; #p < 0.05 and ##p < 0.02 compared to the hemorrhoid model group. Zid- Zidraga-6 capsule.
Zidraga-6 appears to have a noteworthy effect on reducing IL-6 cytokines in the hemorrhoids model, with results indicating a statistically significant impact at doses ranging from 65 to 325 mg/kg. Moreover, Zidraga-6 appears to have a promising effect on reducing IL-1β cytokine levels in a hemorrhoid model, particularly at doses ranging from 130 to 325 mg/kg. This suggests a potential benefit in managing inflammation associated with this condition.
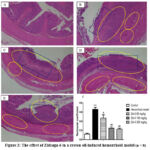 |
Figure 3: The effect of Zidraga-6 in a croton oil-induced hemorrhoid model (n = 8)Click here to view Figure |
Oedema and inflammatory cell infiltration (indicated by a circle) alongside necrosis (indicated by a square) in the layers of the recto anal. Stained with hematoxylin and eosin, magnification x40. A-control group, B-hemorrhoid model group, C-Zidraga-6 65 mg/kg, D-Zidraga-6 130 mg/kg, E-Zidraga-6 325 mg/kg, F-Rectoanal coefficient of experiment. Histopathological examination revealed that the epithelial cells of the rectal mucosa, the submucosal layer, and the outer muscularis mucosa in the control group of rats exhibited a standard structure. In the hemorrhoid model group, observations indicated that the epithelial layer of the rectal-anal mucosa experienced damage, accompanied by an inflammatory cell infiltrate at its base, primarily composed of neutrophils. Furthermore, the submucosal layer revealed a notable presence of inflammatory cell infiltration, along with oedema and clusters of inflammatory cells between the smooth muscle fibres of the muscularis mucosa. It was also noted that the blood vessels in both the mucosal and submucosal layers exhibited dilation and hyperemia, suggesting an active inflammatory response. In the Zidraga-6 65 mg/kg group, there were some observations of epithelial cell loss in specific areas of the mucosal layer, which coincided with the presence of inflammatory cell infiltration. Additionally, inflammatory cells were noted in the submucosal layer, and a small number were found infiltrating between the muscle fibres of the muscularis mucosa. In the Zidraga-6 130 mg/kg group, observations indicated that the epithelial cells in the rectoanal section experienced some damage, accompanied by an infiltration of inflammatory cells within the mucosa and submucosa. In the group of rats treated with Zidraga-6 at a dosage of 325 mg/kg, some changes were observed, including a reduction in epithelial cells and an increase in inflammatory cell infiltration in the rectum and anus region. Notably, a considerable presence of inflammatory cells was noted within the submucosal layer, while a smaller number of these cells were detected among the muscle fibres of the muscularis mucosa. The rectoanal coefficient was significantly higher in the haemorrhoid model group than in the control group. However, this index showed a decrease in the low, medium, and high-dose Zidraga-6 groups compared to the haemorrhoid model group.
Table 4: Findings of pathological changes in the experimental groups
| Groups | Inflammatory cell infiltration | Hyperemia | Edema | Necrosis | Degeneration | Vasodilation |
| Control | – | – | – | – | – | – |
| Hemorrhoid model | +++ | +++ | +++ | +++ | ++ | +++ |
| Zid 65 mg/kg | ++ | + | + | +++ | ++ | + |
| Zid 130 mg/kg | ++ | + | + | ++ | + | + |
| Zid 325 mg/kg | ++ | + | + | + | + | + |
– None, + Less, ++ Moderate, +++ Severe
The histopathological analysis results indicate that the pathological changes—such as inflammation, haemorrhage, oedema, and vasodilation were notably less pronounced in the groups treated with Zidarga at doses of 65 mg/kg, 130 mg/kg, and 325 mg/kg, compared to the haemorrhoid model group. These findings may suggest a potentially beneficial effect of the treatment.
Discussion
In this study, croton oil was utilised as a stimulant and an inflammatory agent to induce experimental hemorrhoids. Croton oil triggers inflammation by releasing a variety of factors, including inflammatory lipid metabolites like prostaglandins, leukotrienes, and lipoxins. It also generates kinins such as bradykinins, chemokines, nitric oxide, and cytokines, including IL-1β and IL-6.25.26
In traditional Mongolian medicine, various herbal tinctures, powders, and oils have long been utilized for the treatment of hemorrhoids. At present, we are engaged in a study to examine the phytochemical composition of the Zidraga-6 capsule and its potential therapeutic effects on this condition.7.9
Many symptoms of hemorrhoids are closely related to inflammation and inflammatory cytokines, including interleukin-6 (IL-6), interleukin-17 (IL-17), tumor necrosis factor-alpha (TNF-α), nitric oxide (NO), inducible nitric oxide synthase (iNOS), and matrix metalloproteinases (MMPs), are known to contribute significantly to the development of hemorrhoids. Understanding the role of these factors may be essential for advancing our approach to treatment and prevention. Inflammatory cells are also found in hemorrhoidal tissue. Thus, anti-inflammatory activity is a crucial criterion for evaluating the effectiveness of traditional formulations designed to alleviate hemorrhoid conditions.25.26
The acute toxicity of Zidarga-6 was evaluated in white mice via intraperitoneal injection, adhering to the methodology established by V.B. Prozorovsky in 1978. The lethal dose (LD50) was determined to be 3.25 grams per kilogram (g/kg), with a confidence interval ranging from 2.8 to 3.8 g/kg. Based on the classification criteria established by K.K. Sidorov, Zidarga-6 is categorized as non-toxic. Furthermore, the effective dose (ED) was identified as 65 mg/kg, in accordance with the method outlined by I.P. Zapadnyuk in 1983, with a defined range of 32.5 to 260 mg/kg. We investigated the anti-hemorrhoidal activity at doses of 130 mg/kg and 325 mg/kg based on the findings from acute toxicity and pharmacological screening studies.27-29
The results of this study indicated that all doses of Zidraga-6 capsules effectively reduced the inflammatory cytokine IL-6 in the rat hemorrhoid model. Furthermore, the levels of the cytokine IL-1β were significantly lowered with the 130 mg/kg and 325 mg/kg doses. Additionally, histopathological examination of the rectoanal tissue demonstrated that the structural changes, inflammation, and necrosis were significantly diminished with the 130 mg/kg and 325 mg/kg doses of Zidraga-6 capsules when compared to the untreated hemorrhoid model.
The histopathological analysis results indicate that the pathological changes—such as inflammation, haemorrhage, oedema, and vasodilation — were notably less pronounced in the groups treated with Zidarga at all doses (65 mg/kg, 130 mg/kg, and 325 mg/kg) compared to the hemorrhoid model group. These findings may suggest a potentially beneficial effect of the treatment.
The findings of the study suggest that treatment with Zidraga-6 may effectively reduce levels of IL-6 and IL-1β cytokines in rats with induced haemorrhoids. Additionally, this treatment appears to decrease the rectoanal coefficient and improve inflammatory conditions, as evidenced by reductions in tissue oedema, vasodilation, and haemorrhage.
According to the Chinese Pharmacopoeia, Kaempferia galanga is standardized based on the presence of p-methoxycinnamate, a major volatile compound.30 In the present study, p-methoxycinnamate was identified in Zidarga-6 capsules using thin-layer chromatography (TLC), with an Rf value of 0.26. Raw materials from Terminalia chebula15 and Embelia ribes10 are known to be rich in organic acids, particularly polyphenolic compounds such as gallic acid, caffeic acid, vanillic acid, and chebulic acid. Other herbal components of the formulation, including Capsicum annuum16-17 and Cynanchum thesioides13-14, are recognized for their high flavonoid content. As a result, this study quantified for the first time that Zidarga-6 capsules contain 12.59 ± 0.68% of total phenolic compounds and 1.30 ± 0.12% of total flavonoids, as determined by UV spectrophotometric analysis. The biologically active compounds in herbal medicines vary.35,36
The principal bioactive constituent of Capsicum annuum (red pepper) is capsaicin, which is also listed as a marker compound in the Chinese Pharmacopoeia.30-32 In this study, capsaicin was detected in Zidarga-6 capsules at an Rf value of 0.49, corresponding closely to its typical Rf value in the raw material (~0.59). 17
Furthermore, Atragene sibirica, another key component of the formulation, contains a significant amount of triterpene saponins, with oleanolic acid identified as a major active compound33-34. TLC analysis confirmed the presence of oleanolic acid in Zidarga-6 capsules, showing an Rf value of 0.82. In addition, UV spectrophotometric analysis validated that the total triterpene saponin content in the capsules was 1.89 ± 0.19%.
The effectiveness of Zidraga-6 in treating hemorrhoids may be linked to its content of biologically active substances, disease, reduction of blood pressure, and immunosuppression. The use of purified flavonoid fractions like diosmin and hesperidin is increasingly recognised as a non-surgical treatment option for alleviating the symptoms of hemorrhoids, including bleeding, swelling, and itching. 5,37.39
Specific triterpene saponins have demonstrated anti-inflammatory effects on the mucosa and are particularly effective against hemorrhoids, especially in treating rectal ulcers.40
A limitation of this study is that it does not incorporate qualitative studies on pro-inflammatory cytokine proteins, and it lacks a mechanism for elucidating these findings.
Conclusion
Zidraga‑6 capsules contain biologically active constituents such as flavonoids, phenolic compounds, and saponins, p-Methoxycinnamate, Oleanolic acid, Gallic acid, Quercetin and Capsaicin detected in Zidraga-6 capsule. Zidraga-6 has demonstrated a significant ability to attenuate inflammation-related cytokines, particularly interleukin-6 (IL-6) and interleukin-1 beta (IL-1β). In a controlled model of hemorrhoids, it has also been effective in mitigating edema, necrosis, and vasodilation.
Acknowledgment
We thank to regard special thanks to the Mongolian Foundation for Science and Technology. We sincerely appreciate the invaluable support provided by the research workers at the Institute of Traditional Medicine and Technology in the conduct of this study.
Funding source
This project was funded by the Mongolian Foundation for Science and Technology. The agreement number of the project was ShUTBIKhKhZG-2022/136.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Author Contributions
- Chimedragchaa Chimedtseren: Data collection, Analysis, Writing- Original Draft
- Dejidmaa Buyantogtokh: Data collection, Analysis, review and & Editing, and Project
- Myadagbadam Urtnasan: Data collection, Analysis, Review, & Editing
- Anu Altangerel: Data collection, Analysis.
- Uuganbayar Baatartsogt: Data collection, Analysis.
- Erdenechimeg Chuluunbaatar: Funding Acquisition, Resources, Supervision
References
- Brown SR. Haemorrhoids: an update on management. Therapeutic Advances in Chronic Disease. 2017;8(10):141-147.
CrossRef - Lohsiriwat V. Hemorrhoids: from basic pathophysiology to clinical management. World J Gastroenterol. 2012;18(17):2009-2017.
CrossRef - Mott T., Latimer K., Edwards C. Hemorrhoids: Diagnosis and Treatment Options. Am Fam Physician. 2018;97(3):172-179.
- Akinmoladun O., Oh W. Management of Hemorrhoids and Anal Fissures. Surg Clin North Am. 2024;104(3):473-490.
CrossRef - Wei X., Qin H., Wei T., et al. The Potential Mechanism of Kushen Decoction in Treating Haemorrhoids: An Integration of Network Pharmacology, Molecular Docking and Molecular Dynamics Simulation. IET Syst Biol. 2025;19(1).
CrossRef - Juru dosil. State Publishing House. 1991;35-36.
- Yuteg Youndongombo. The for medical tantra. State Publishing House. 1991;35-38.
- Chimedragchaa C, Dagvatseren B, Khishigjargal L. Reference book of control of traditional medicinal substances and drugs. Ulaanbaatar, Mongolia: Lsom Press. 2025;468-469.
- Baavgai C, Boldsaikhan B. Mongolian traditional medicine. State Publishing House. 1990;121-123.
- Rout S., Sahoo G., Padhy D., Mishra., et al. Embelia ribes Burm F.(Vai Vidanga)-An Overview. International Journal of Modern Agriculture. 2021;10(2),4588-4594.
- Wang SY., Cai L., Yang N.,et al. Chemical composition of the Kaempferia galanga L. essential oil and its in vitro and in vivo antioxidant activities. Frontiers in Nutrition. 2023;2(10):1080487.
CrossRef - Zhai X.J., Yang Z.R.; Zhang, F.L., et al. Study on the flowering biology and pollination adaptability of Cynanchum thesioides (Freyn) K. Schum. J. Chinese Hortic. 2017;1:2616.
CrossRef - Wang, D., Chen, G., Qiao, L., et al. The chemical constituents from the fruits of Cynanchum thesioides (Freyn) K. Schum. J. Med. Chem. 2007;17:101–103.
CrossRef - Lyu T., Kim W. Study on Bioactive Components of Aromatic Cynanchum thesioides (Freyn) K. Schum by Solvent Fractionation. 2024;13(22):3123.
CrossRef - Juang LJ., Sheu SJ., Lin TC. Determination of hydrolyzable tannins in the fruit of Terminalia chebula Retz. by high-performance liquid chromatography and capillary electrophoresis. J Sep Sci. 2004;27(9):718-724.
CrossRef - Antonio A.S., Wiedeman, L.S.M., Junior V.V. The genus Capsicum: a phytochemical review of bioactive secondary metabolites. RSC advances. 2018;8(45):25767-25784.
CrossRef - Nadi M.S., Fikri F., Purnama, M.T.E. Determination of capsaicin levels in Capsicum annuum Linn Ethanolic extract using thin layer chromatography analysis. 2020;70(14):1831-1842.
- Ligaa U., Davaasuren B., Ninjil N. The use of Mongolian medicinal plants in Western and Eastern medicine. JKC printing. 2006;91-92.
- Wagner H., Bladt S., Rickl V. Plant Drug Analysis: A Thin Layer Chromatography atlas. Springer Berlin Heidelberg; 1996.
CrossRef - Chimedtseren, C., Baatartsogt, U., Tsagaanbaatar, N., et al. Results of Chemical and Pharmacological Study of the Mongolian Traditional Prescription “Indra-4”. Journal of Complementary and Alternative Medical Research, 2024;25(12):165-178.
CrossRef - Singleton VL., Orthofer R., Lamuela-Raventós RM. Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Methods in enzymology. 1999;299:152-178.
CrossRef - Aryal S., Baniya M. K., Danekhu K., et al. Total phenolic content, flavonoid content and antioxidant potential of wild vegetables from Western Nepal. Plants, 2019;8(4): 96.
CrossRef - Azeemuddin M., Viswanatha GL., Rafiq M., et al. An improved experimental model of hemorrhoids in rats: evaluation of antihemorrhoidal activity of an herbal formulation. ISRN Pharmacol. 2014;2014(1):7.
CrossRef - Hu D., Zeng Q., Wang H.. Arginine Hydrochloride Reduce Rectal Mucosal Irritation of Sodium Aescinate: Molecular Docking, Physical Properties, Anti-Hemorrhoidal Activity, Safety and Topical Gel Formulations Investigation. Pharmaceutics. 2024;16(12):1498.
CrossRef - Liang Y., Ren T., Li R., et al. Natural Products with Potential Effects on Hemorrhoids: A Review. Molecules. 2024 ;29(11):2673.
CrossRef - Razdar S., Panahi Y., Mohammadi R.Evaluation of the efficacy and safety of an innovative flavonoid lotion in patients with haemorrhoid: a randomised clinical trial. BMJ Open Gastroenterol. 2023;10(1):1158.
CrossRef - Prozorovskiĭ V, B., and Prozorovskaia M, P. Table method of determining the ED50 (LD50) of substances with low biological activity. Farmakol Toksikol, 1978;43(6): 733–735.
- Sidorov K, K. Classification of toxicities of parenteral drugs. Toxilogy new industrial chemical properties, 197; (13): 47-51.
- Zapadnyuk I.P., Zapadnyuk V.I., Zakhariya E.A. Laboratory animals. Breeding. Maintenance, use in the experiment.. Kiev.1983;383-385.
- Pharmacopoeia of the people’s republic of China. Vol 1. People’s Medical Publishing House. 2015.
- Mandal SK., Rath SK., Logesh R. Capsicum annuum L. and its bioactive constituents: A critical review of a traditional culinary spice in terms of its modern pharmacological potentials with toxicological issues. Phytother Res. 2023;37(3):965-1002.
CrossRef - Nadi MS., Fikri F., Purnama MT. Determination of capsaicin levels in Capsicum annum Linn Ethanolic extract using thin layer chromatography analysis. Drugs. 2020;70(14):1831-42.
- Kuznetsova S. B. Biomorphology of Atragene sibirica L. shrubby liana. Contemporary Problems of Ecology. 2015;8(2):598 606.
CrossRef - Yudin S. I. Atragene sibirica L. – ornamental liana of Altai flora. Introduction of Plants.2006; 30: 46 48.
- Altangerel A., Chimedtseren C., Urtnasan M.. The Antihypertensive Effect of Marchin-13 Tang on L-NAME-induced Hypertension in Rats. Biomed Pharmacol J. 2024;17(2).1203-1212.
CrossRef - Myadagbadam U., Purevsuren S. Standardization study of khurtsiin deed-6 traditional medicine. Pharmacognosy Journal. 2022; 14(3):610-621.
CrossRef - Vidyarthi S., Samant S.S., Sharma P. Traditional and indigenous uses of medicinal plants by local residents in Himachal Pradesh, North Western Himalaya, India. J. Biodivers. Sci., Ecosyst. Serv. Manage. 2013;9(3):185-200.
CrossRef - Ullah A., Munir S., Badshah SL. Important Flavonoids and Their Role as a Therapeutic Agent. Molecules. 2020;25(22):5243.
CrossRef - Kusumo MHB., Prayitno A., Soetrisno, et al. Synergistic therapeutic approach for hemorrhoids: integrating mesenchymal stem cells with diosmin-hesperidin to target tissue edema and inflammation. Arch Med Sci. 2024;20(5):1556-1566.
CrossRef - Baky MH, Elsaid MB, Farag MA. Phytochemical and biological diversity of triterpenoid saponins from family Sapotaceae: A comprehensive review. 2022;202:113345.
CrossRef







