Manuscript accepted on :15-01-2026
Published online on: 17-04-2026
Plagiarism Check: Yes
Reviewed by: Dr. Tetty A
Second Review by: Dr. Ana Golez
Final Approval by: Dr. Mariia Shanaida
Nitya Kala1, 2* , Ravi Saranyan1
, Ravi Saranyan1 , Dorairaj Jayachandran1
, Dorairaj Jayachandran1 , Mythili Gnanamangai3
, Mythili Gnanamangai3 and Harikrishnan Prasad1,4
and Harikrishnan Prasad1,4
1Department of Periodontology, Vinayaka Mission’s Sankarachariyar Dental College, Vinayaka Mission’s Research Foundation (Deemed to be University), Salem, Tamil Nadu, India.
2Department of Periodontics, KSR Institute of Dental Science and Research, The Tamil Nadu Dr. M.G.R. Medical University, Thokkavadi, Tiruchengode, Tamil Nadu, India.
3Department of Biotechnology, K.S.Rangasamy College of Technology, Anna University, Tiruchengode, Tamil Nadu, India.
4Department of Oral and Maxillofacial Pathology and Oral Microbiology, KSR Institute of Dental Science and Research, The Tamil Nadu Dr. M.G.R. Medical University, Tiruchengode, Tamil Nadu, India.
Corresponding Author E-mail: nityakala@ksridsr.edu.in
Abstract
Porphyromonas gingivalis is an anaerobic Gram-negative coccus known to play a significant role in periodontal disease. Although tetracycline is a potent antibiotic with excellent antibacterial activity against this organism, it should be used judiciously owing to its side effects and the possibility of antibiotic resistance. In the search for alternate treatment strategies against P.gingivalis, herbal products show sufficient promise. Therefore, we performed a scoping review to understand our current knowledge on the efficacy of herbal extracts against P.gingivalis. Using specific keywords, databases like PubMed, Medline, Lilacs, Web of science, and Scopus were searched for in vitro studies reporting the efficacy of various herbal products against Porphyromonas gingivalis. After screening and elimination of irrelevant and duplicate articles, 94 articles were included in this review. Data was extracted from these articles, and the findings are summarised. Numerous herbal extracts have been evaluated for their efficacy against P.gingivalis. Extracts from coffee beans, Moringa oleifera leaves and Allium sativum pods were effective at low concentrations. Artemisia herba-alba showed a maximum zone of inhibition against the organism. Our literature search and review suggest that a wide range of herbal extracts have been evaluated for their effectiveness against P.gingivalis. However, a majority of such extracts have never been evaluated in more than one study, and some do not have proper positive and negative controls, prompting us to view their results with extreme caution. Further research in the form of animal studies and clinical trials is essential to identify and develop a potent alternative to tetracycline for use in periodontal disease management.
Keywords
Antibacterial activity; Herbal extract; Inhibition zone diameter; Minimum inhibitory concentration; Porphyromonas gingivalis
| Copy the following to cite this article: Kala N, Saranyan R, Jayachandran J, Gnanamangai M, Prasad H. Plant-Based Antibacterial Strategies Against Porphyromonas gingivalis: A Systematic Review. Biomed Pharmacol J 2026;19(2). |
| Copy the following to cite this URL: Kala N, Saranyan R, Jayachandran J, Gnanamangai M, Prasad H. Plant-Based Antibacterial Strategies Against Porphyromonas gingivalis: A Systematic Review. Biomed Pharmacol J 2026;19(2). Available from: https://bit.ly/4enl7Jq |
Introduction
Porphyromonas gingivalis is an anaerobic Gram-negative coccus which plays a key role in the initiation and progression of periodontal disease. This microbe invades epithelial tissue and modifies the host defence by chemokine paralysis (inhibiting secretion of IL-8). It also possesses a battery of enzymes like collagenase and gingipains that adds to its virulence.1 This cannot be eliminated from the periodontal pocket by mechanical debridement alone (scaling and root planing) as they can invade the periodontal tissue. Use of antimicrobials has become an important part of anti-infective therapy in the management of periodontal disease. The antibiotic of choice used here is tetracycline which has good antibacterial efficacy and anti-collagenase activity. Side effects of the drug (like nausea, headache, stomach upset) and emergence of antimicrobial resistant microbe are vital factors to be considered in their usage.2
In recent years herbal alternatives are being researched for management of diseases. These products have bioactive compounds that could be used in anti-infective therapy against P.gingivalis.3 In this review we have analysed the existing data on the antibacterial efficacy of herbal extracts against P.gingivalis to find a promising alternative to antibiotics in the management of periodontal disease.
Materials and Methods
This systematic review was prospectively registered with the Open Science Framework (OSF.IO/H8K43). A comprehensive literature search was conducted across PubMed, MEDLINE, Web of Science, and Scopus databases using the keywords “Plant extract”, “Herbal extract”, AND “Porphyromonas gingivalis“. Articles published up to and including 30th June 2024 were considered for inclusion.
Inclusion criteria
In vitro studies specifically evaluating crude plant extracts against Porphyromonas gingivalis
Studies reporting quantitative antimicrobial endpoints: minimum inhibitory concentration (MIC) and/or inhibition zone diameter (IZD) using agar diffusion methods
Primary research articles published in peer-reviewed journals
Exclusion criteria
Studies evaluating commercially available herbal products or formulations
Research on combined extracts from multiple plant species
Investigations of isolated chemical compounds or purified phytochemicals from plant extracts
Clinical studies, animal studies, or reviews/meta-analyses
Studies lacking specific quantitative MIC or IZD data against P. gingivalis
From 823 identified records, articles were independently screened by two reviewers (Authors 1 and 5) following PRISMA-SCR guidelines. After title/abstract screening, full-text assessment, and resolution of discrepancies by consensus discussion, 88 studies met all inclusion criteria and were included in the qualitative synthesis (selection flowchart: Fig-1).
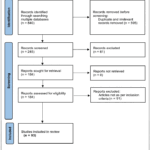 |
Figure 1: PRISMA-SCR Flowchart |
Results
The summary of the data extracted is tabulated (Table 1). The research for antibacterial activity of plant extract against Porphyromonas gingivalis started by Clark et al. in 1993 using Acacia senegal (arabica gum).4 Studies have been conducted on plants native to Brazil, China, India, Sudan and United States of America. There has been a lot of research on this domain but there are very few repetitions. It can be observed that five studies were conducted on Punica granatum L. (Pomegranate) and four studies on Pistacia lentiscus (fruit or mastic gum) and Azadirachta indica (Neem). Minimum inhibitory concentration data reveal that green coffee bean extract has the least value of 0.2 µgm/mL by Bharath N et al.,38 followed by leaves of Moringa oleifera (Drumstick tree) with 0.8µgm/mL by Ida MJ et al.,84 and pods of Allium sativum with 1.2 µgm/mL by Alirezaei S et al.55 The results of agar disc diffusion method showed that Artemisia herba-alba (white wormwood) has the maximum inhibition diameter of 49 mm by Arbia L et al.4
Table 1: Quantitative synthesis of published studies evaluating plant-based antimicrobials against oral pathogens (n = 115)
| Domain | Parameter | Value |
| Study characteristics | Total studies 4-96 | 115 |
| Publication period | 1993–2024 | |
| Studies per decade | 1990s-3; 2000s-9; 2010s-49; 2020 to 2024-54 | |
| Outcome measures | Studies reporting MIC/MBC | 96 (83.5%) |
| Studies reporting IZD | 52 (45.2%) | |
| Studies reporting both | 33 (28.7%) | |
| Studies reporting neither | 6 (5.2%) | |
| Plant material | Most studied plant part | Leaves (57; 49.6%) |
| Second most studied part | Fruits (21; 18.3%) | |
| Bark / stem bark | 14 (12.2%) | |
| Seeds / galls / gum / resin | 13 (11.3%) | |
| Rhizome / tuber | 7 (6.1%) | |
| Part not specified | 18 (15.7%) | |
| Extraction method | Ethanol-based extracts | 46 (40.0%) |
| Aqueous / water-based | 24 (20.9%) | |
| Methanol | 18 (15.7%) | |
| Dichloromethane | 9 (7.8%) | |
| Acetone | 8 (7.0%) | |
| Non-polar solvents (hexane, chloroform) | 7 (6.1%) | |
| Solvent not specified | 14 (12.2%) | |
| Antimicrobial potency | MIC range reported | 0.13 – 55,000 µg/mL |
| Studies reporting MIC ≤100 µg/mL | 38 (33.0%) | |
| Studies reporting MIC >1,000 µg/mL | 29 (25.2%) | |
| Comparators (IZD studies) | Any positive control used | 46 / 52 (88.5%) |
| Chlorhexidine (0.12–0.2%) | 27 (51.9%) | |
| Antibiotic controls (any) | 19 (36.5%) | |
| No control stated | 6 (11.5%) | |
| Reporting quality indicators | Unit-standardized MIC values | 72 (62.6%) |
| Multiple plants per study | 29 (25.2%) | |
| Multiple solvents per study | 21 (18.3%) |
Discussion
Porphyromonas gingivalis, the primary focus of this review, belongs to Socransky’s “red complex”—a highly pathogenic complex comprising P. gingivalis, Tannerella forsythia, and Treponema denticola, that strongly correlates with deep periodontal pockets (>6 mm), attachment loss, and bleeding on probing.97 These species form synergistic biofilms characterized by proteolytic activity, nutrient sharing, and immune evasion, driving dysbiosis from health-associated communities toward tissue-destructive inflammation. Recent metagenomic analyses confirm red-complex enrichment in severe periodontitis, alongside orange-complex species (Campylobacter rectus, Fusobacterium nucleatum) and emerging pathogens like Parvimonas micra and Filifactor alocis.
Sonets et al. applied whole-genome shotgun metagenomics to mild vs. severe cases, revealing that red-complex dominance, coupled with specific metabolic pathways (like amino acid degradation) and network interactions, predicts disease progression.98 Non-microbial factors, including cardiovascular comorbidities and recent antibiotic exposure, further amplified severity signatures, underscoring periodontitis as a polymicrobial, multifactorial disorder rather than a monopathogen infection.
P. gingivalis acts as a keystone pathogen, using gingipains, fimbriae, and outer membrane vesicles to orchestrate community virulence and host immune dysregulation, despite low abundance.97 Plant extracts targeting P. gingivalis may thus indirectly modulate broader dysbiotic consortia, aligning with Ecological plaque hypotheses.
The investigation of plant extracts for antibacterial activity against Porphyromonas gingivalis began in the early 1990s. For more than a decade, the number of publications in this field remained relatively limited. However, a marked increase in research output has been observed after 2010. This surge in interest may be attributed to growing concerns regarding the adverse effects associated with conventional antimicrobials and the escalating problem of antibiotic resistance in periodontal pathogens. A detailed year‑wise distribution of published studies is provided in Table 2.
Table 2: Year-wise distribution of the published studies
| Year | Number of studies |
| 1997 to 2000 | 4 4-7 |
| 2001 to 2005 | 3 8-10 |
| 2006 to 2010 | 4 11-14 |
| 2011 to 2015 | 24 15- 38 |
| 2016 to 2020 | 29 39-67 |
| 2021 to 2025 | 29 68- 96 |
The studies included in this review investigated a wide variety of plant extracts, with only a limited number of repetitions across different herbs. Interestingly, in cases where the same plant was evaluated by multiple authors, the results often showed notable variability in both minimum inhibitory concentration (MIC) values and inhibition zone diameters. These discrepancies may be explained by differences in the plant part utilized for extraction, the choice of solvent, or variations in extraction methodology.
For example, Carrol et al. in 2020 reported that Pistacia lentiscus demonstrated an MIC of 8 µg/mL when an ethanolic extract of the fruit was tested, compared with 32 µg/mL when the woody portion of the plant was used and 64 µg/mL when water served as the extraction solvent.61 Similarly, Gawade et al. in 2025 observed pronounced variability in Sphaeranthus indicus, where the MIC for leaf extract was 12.5 µg/mL, whereas the flower extract yielded an MIC as high as 100 µg/mL.96
These variations highlight the importance of standardizing extraction protocols in phytochemical and antimicrobial research. Since MIC is defined as the lowest concentration of an antimicrobial agent that completely inhibits the visible growth of a microorganism after incubation, even modest methodological differences can significantly influence reported outcomes.
Furthermore, such inconsistencies emphasize the critical need for quality control and reproducibility standards in the development of herbal-based therapeutic agents. Establishing uniform guidelines for plant part selection, extraction solvent, and preparation methods would not only enhance comparability across studies but also strengthen the translational potential of promising plant extracts for clinical applications. A summary of the most frequently investigated plant extracts, along with their MIC values and inhibition diameters, is presented in Table 3.
Table 3: Commonly researched plants
| Plant studied | Author | MIC (in µgm/ml) | IZD (in mm) |
| Punica granatum L. (Pomegranate) | Rosas-Pinon et al. 21 | 125 | — |
| Veloso et al. 64 | 170 | 3.0 ± 0.0 | |
| Abullais et al. 69 | 5210 | 14 ± 0.45 | |
| Ghafar and Dahham, 78 | — | 41 | |
| Madhloom AF, 77 | 6.25 mg/ml | 14.02 | |
| Pistacia lentiscus (Mastic gum/ fruit) | Sterer N 11 | — | 10.5-13.7 |
| Karygianni et al. 29 | 20 | — | |
| Koychev et al. 47 | — | 25 | |
| Carrol et al. 61 | 64(Woody part) | — | |
| Carrol et al. 61 | 8 (Fruit) | — | |
| Azadirachta indica (Neem) | Heyman et al. 44 | 10 μM | — |
| Carrol DH et al. 61 | 64 | — | |
| Saquib et al.69 | 640 | 13 ± 0.36 | |
| Müller-Heupt LK et al.72 | 256 (Acetone) | — | |
| Müller-Heupt LK et al. 72 | 1024 (70% Ethanol) | — |
The reported MIC values of commonly used antimicrobials against Porphyromonas gingivalis are 0.4 µg/mL for chlorhexidine,43 0.016 µg/mL for tetracycline,10 and 0.313 µg/mL for doxycycline.30 Analysis of the extracted data (Table 4) demonstrates that among the plant-derived preparations, Moringa oleifera leaves exhibited the lowest MIC at 0.8 µg/mL.84 This value is of particular relevance as it is comparable to the inhibitory range of some standard antimicrobial agents currently used in clinical practice. Importantly, these findings suggest that selected plant extracts, particularly Moringa oleifera, may serve as promising adjuncts or potential alternatives to conventional antimicrobials in the management of periodontal infections, especially in the context of increasing antibiotic resistance. A consolidated list of the plant extracts demonstrating the lowest MIC values against P. gingivalis is presented in Table 4.
Table 4: Plant extracts with the least minimum inhibitory concentration (MIC)
| Plant | Part | MIC (µgm/mL) | Author |
| Coffea canephora | Bean | 0.2 | Bharath et al. 38 |
| Moringa oleifera | Leaf | 0.8 | Ida MJ et al. 84 |
| Allium sativum | Pods | 1.21±0.37 | Alirezaei et al. 55 |
| Rheum palmatum | Root | 4 | Müller-Heupt et al. 72 |
| Lycium barbarum | Fruit | 6.25 | Sanghavi AD et al. 71 |
| Copaifera reticulata | Oleoresin | 6.25 | Bardaji DK et al. 42 |
µgm/mL- microgram per millilitre
The inhibition zone diameter (IZD) is defined as the maximum linear distance across the clear zone of microbial growth inhibition surrounding a test compound in the agar diffusion assay. In several studies, plant extracts demonstrated IZDs that were comparable to, and in some cases exceeded, those of standard antimicrobial agents. For example, extracts have produced inhibition zones within the range of 32.25-40 mm, which is similar to the activity observed with doxycycline (32.25-40 mm),39 while others achieved IZDs between 16-33.5 mm,29 comparable to 0.2% chlorhexidine.
These results highlight the potential of certain plant extracts to provide inhibitory effects against P. gingivalis comparable to conventional antimicrobials. Nonetheless, the variability in outcomes across studies emphasizes the importance of methodological standardization to validate these findings. The plant extracts demonstrating the highest inhibition zone diameters are summarized in Table 5
Table 5: Plant extracts with the highest inhibition zone diameter in agar diffusion test.
| Plant | Inhibition diameter (mm) | Author |
| Artemisia herba-alba | 49.00 ± 0.82 | Arbia et al. 45 |
| Opuntia ficus-indica | 48.83 ± 1.03 | Arbia et al. 45 |
| Camellia sinensis | 42.00 ± 0.41 | Arbia et al. 45 |
| Ocimum sanctum | 33.79 ± 1.82 | Jayanti et al. 48 |
| Camellia sinensis | 26.65±1.56 | Araghizadeh et al. 23 |
Conceptualizing periodontitis as a “continuum”, with antecedents (genetic/systemic risks), triggers (dysbiosis), mediators (inflammation/oxidative stress), and outcomes, demands multimodal therapies beyond mechanical debridement alone. Benahmed et al. advocated integrating biofilm control with host modulation, locally delivered antimicrobials, probiotics/prebiotics for microbiome reshaping, and nanoparticle drug delivery to target matrix metalloproteinases and cytokines.99
Emerging antimicrobials exemplify this shift. Tolentino et al. tested cannabidiol (CBD) against a 33-species subgingival biofilm model, finding 500–1000 µg/mL CBD significantly reduced total biomass and red-complex proportions (P. gingivalis, T. forsythia). This was comparable to 0.12% chlorhexidine, while relatively enriching green-complex health-associated species. CBD’s efficacy was greatest when applied pre-biofilm formation (post-debridement mimicry), suggesting ecological rebalancing over broad suppression, though clinical trials remain needed.100
Recent work in this platform has found that Camellia sinensis polyphenols inhibited gingipain activity, a key virulence factor of P. gingivalis.101 The modulation of inflammatory cytokines and reduced ROS production by Moringa oleifera extracts was also observed. These factors strengthen the mechanistic insights and the pharmacological rationale for incorporating these botanicals as adjuncts in periodontal therapy.102,103
Emerging molecular studies have also shed light on the mechanisms of action underlying plant-derived antibacterial activity. Wu et al. investigated dihydrochalcone flavonoids such as phloretin and phlorizin, showing these compounds cause morphological disruption, increase membrane permeability, and induce apoptosis in P. gingivalis. 104 Transcriptomic profiling revealed interference with DNA function and oxidative stress pathways, supporting phytochemicals as multi-target agents capable of disrupting bacterial survival and resistance mechanisms.
These frameworks contextualize the plant extracts reviewed here as adjuncts within continuum-based care. Extracts matching chlorhexidine/doxycycline MICs (e.g., Moringa oleifera 0.8 µg/mL, Coffea canephora 0.2 µg/mL) or the ones with anti-gingipain activity could integrate with strategies like antimicrobial photodynamic therapy, host modulation (e.g., via polyphenols inhibiting gingipains/NF-κB), or microbiome-targeted probiotics. Future studies should evaluate phytochemicals against multi-species biofilms, clinical isolates across red/orange complexes, and systemic inflammatory markers to validate their role in precision periodontal management.
In terms of clinical relevance, mouthwash formulations containing Punica granatum L. have undergone randomized clinical trials, confirming comparable or superior efficacy to chlorhexidine in reducing gingival inflammation and plaque indices.101 Despite modest in vitro activity against P. gingivalis measured by some studies, the clinical benefits likely stem from synergistic anti-inflammatory and antimicrobial effects of multiple phytochemicals such as punicalagin and quercetin, which inhibit key virulence enzymes of P. gingivalis.
Limitations of Existing Studies
Although numerous studies have investigated the antibacterial activity of plant extracts against Porphyromonas gingivalis, several limitations were evident. First, many studies did not incorporate appropriate positive controls (such as chlorhexidine, doxycycline, or tetracycline). The absence of controls undermines the reliability, comparability, and overall significance of the reported findings. Second, the majority of the research was conducted using laboratory stock strains, particularly ATCC 33277, with relatively few studies evaluating other strains such as W381, W50, or W83. Only a limited number utilized clinical isolates, which are more representative of pathogens encountered in patients.
Notably, differences in strain type influenced reported antimicrobial activity. For example, Mendes et al. in 2020 found that fractions of Salvia officinalis demonstrated an MIC of 50 µg/mL against ATCC 33277, but a higher MIC of 100 µg/mL against clinical isolates.62 Similarly, Allium sativum aqueous extract showed remarkable variation, with an MIC of 1.2 µg/mL against ATCC 33277,55 compared with 4.4 mg/mL against the W50 strain.93 Nakao et al. in 2024 reported comparable discrepancies for Camellia sinensis extracts, with MIC values of 250 µg/mL for ATCC 33277 and W50, but a significantly lower 125 µg/mL for the W83 strain.92 These findings highlight that MIC values are highly strain-dependent, and reliance solely on stock cultures may not accurately reflect clinical efficacy.
Therefore, to improve translational relevance, future research should prioritize standardized methodologies, inclusion of appropriate positive controls, and testing across multiple strains, particularly clinical isolates. Such measures will enhance the reproducibility and clinical applicability of plant-derived antibacterial agents in periodontal therapy.
Future research should focus on standardizing extraction methodologies, incorporating appropriate positive controls, and testing across multiple P. gingivalis strains, particularly clinical isolates. In addition, mechanistic studies, toxicity assessments, and clinical trials are essential to validate efficacy and ensure the translational potential of plant-derived extracts as safe and sustainable adjuncts in periodontal therapy.
Conclusion
The potential of plant-derived compounds as alternatives to conventional therapies has garnered significant attention within the medical field. To effectively transition these botanical extracts into clinical practice, comprehensive in vitro characterization and subsequent human trials are imperative. Antibacterial properties are particularly crucial for addressing periodontal diseases, such as those caused by P. gingivalis. This review underscores the diverse array of plant species investigated globally for their antimicrobial efficacy against this pathogen. While certain plant extracts have demonstrated comparable antibacterial activity to existing antimicrobial agents, further research is warranted to isolate the specific phytochemicals responsible for these effects and to explore their potential for drug development.
Acknowledgement
The author would like to thank Vinayaka Mission’s Research Foundation (Deemed to be University) for permitting this Ph.D. research.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not applicable
Author contributions
- Nitya Kala: Conceptualization, Methodology, Data Collection, Analysis, Writing – Original Draft
- Ravi Saranyan: Conceptualization, Methodology, Supervision.
- Dorairaj Jayachandran: Visualization, Methodology, Supervision.
- Mythili Gnanamangai : Writing – Review & Editing, Supervision.
- Harikrishnan Prasad: Conceptualization, Methodology, Analysis, Writing – Review & Editing
References
- How KY, Song KP, Chan KG. Porphyromonas gingivalis: An overview of periodontopathic pathogen below the gum line. Front Microbiol. 2016;7:53. doi: 10.3389/fmicb.2016.00053.
CrossRef - Shutter MC, Akhondi H. Tetracycline. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Updated 2023 Jun 5. Available from: https://www.ncbi.nlm.nih.gov/books/NBK549905/
- Vaou N, Stavropoulou E, Voidarou C, Tsigalou C, Bezirtzoglou E. Towards advances in medicinal plant antimicrobial activity: a review study on challenges and future perspectives. Microorganisms. 2021;9(10):2041.
CrossRef - Clark DT, Gazi MI, Cox SW, Eley BM, Tinsley GF. The effects of Acacia arabica gum on the in vitro growth and protease activities of periodontopathic bacteria. J Clin Periodontol. 1993 Apr;20(4):238-43. doi: 10.1111/j.1600-051x.1993.tb00351.x.
CrossRef - Sote EO, Wilson M. In-vitro antibacterial effects of extracts of Nigerian tooth-cleaning sticks on periodontopathic bacteria. Afr Dent J. 1995;9:15-9.
- Kim JH. Anti-bacterial action of onion (Allium cepa L.) extracts against oral pathogenic bacteria. J Nihon Univ Sch Dent. 1997 Sep;39(3):136-41. doi: 10.2334/josnusd1959.39.136.
CrossRef - Hu JP, Takahashi N, Yamada T. Coptidis rhizoma inhibits growth and proteases of oral bacteria. Oral Dis. 2006; 297-302. Doi: 10.1111/j.1601-0825.2000.tb00142.x
CrossRef - Bakri IM, Douglas CW. Inhibitory effect of garlic extract on oral bacteria. Arch Oral Biol. 2005 Jul;50(7):645-51. doi: 10.1016/j.archoralbio.2004.12.002. Epub 2005 Feb 5.
CrossRef - Ndukwe KC, Okeke IN, Lamikanra A, Adesina SK, Aboderin O. Antibacterial activity of aqueous extracts of selected chewing sticks. J Contemp Dent Pract. 2005 Aug 15;6(3):86-94.
CrossRef - Al-hebshi N, Al-haroni M, Skaug N. In vitro antimicrobial and resistance-modifying activities of aqueous crude khat extracts against oral microorganisms. Arch Oral Biol. 2006 Mar;51(3):183-8. doi: 10.1016/j.archoralbio.2005.08.001. Epub 2005 Oct 24.
CrossRef - Sterer N. Antimicrobial effect of mastic gum methanolic extract against Porphyromonas gingivalis. J Med Food. 2006 Summer;9(2):290-2. doi: 10.1089/jmf.2006.9.290.
CrossRef - Rohrer U, Kunz EM, Lenkeit K, Schaffner W, Meyer J. Antimicrobial activity of Mahonia aquifolium and two of its alkaloids against oral bacteria. Schweiz Monatsschr Zahnmed. 2007;117(11):1126-31.
- Alviano WS, Alviano DS, Diniz CG et al. In vitro antioxidant potential of medicinal plant extracts and their activities against oral bacteria based on Brazilian folk medicine. Arch Oral Biol. 2008 Jun;53(6):545-52. doi: 10.1016/j.archoralbio.2007.12.001.
CrossRef - Wong RW, Hägg U, Samaranayake L, Yuen MK, Seneviratne CJ, Kao R. Antimicrobial activity of Chinese medicine herbs against common bacteria in oral biofilm. A pilot study. Int J Oral Maxillofac Surg. 2010 Jun;39(6):599-605. doi: 10.1016/j.ijom.2010.02.024.
CrossRef - Seneviratne CJ, Wong RW, Hägg U et al. Prunus mume extract exhibits antimicrobial activity against pathogenic oral bacteria. Int J Paediatr Dent. 2011 Jul;21(4):299-305. doi: 10.1111/j.1365-263X.2011.01123.x.
CrossRef - Basri DF, Tan LS, Shafiei Z, Zin NM. In Vitro Antibacterial Activity of Galls of Quercus infectoria Olivier against Oral Pathogens. Evid Based Complement Alternat Med. 2012;2012:632796. doi: 10.1155/2012/632796.
CrossRef - Kuvatanasuchati J, Laphookhieo S, Rodanant P. Antimicrobial activity against periodontopathic bacteria and cytotoxic study of Cratoxylum formosum and Clausena lansium. Journal of Medicinal Plants Research. 2011;5(25)5988:92. Doi: 10.5897/JMPR11.328
- Zhao Y, Li P, Ge L, et al. Effect of electrolyzed reduced water on chiral theanine and polyphenols in tea. Food Chem. 2012;134, 1761-6. 10.1016/j.foodchem.2012.03.075.
CrossRef - Shafiei Z, Shuhairi NN, Shah MF, Yap N, Sibungkil CA, Latip J. Antibacterial Activity of Myristica fragrans against Oral Pathogens. Evid Based Complement Alternat Med. 2012;2012:825362. doi: 10.1155/2012/825362.
CrossRef - Choi JS, Ha YM, Joo CU, Cho KK, Kim SJ, Choi IS. Inhibition of oral pathogens and collagenase activity by seaweed extracts. J Environ Biol. 2012 Jan;33(1):115-21.
- Rosas-Piñón Y, Mejía A, Díaz-Ruiz G, Aguilar MI, Sánchez-Nieto S, Rivero-Cruz JF. Ethnobotanical survey and antibacterial activity of plants used in the Altiplane region of Mexico for the treatment of oral cavity infections. J Ethnopharmacol. 2012 Jun 14;141(3):860-5. doi: 10.1016/j.jep.2012.03.020.
CrossRef - Patel M, Naidoo R, Owotade FJ. Inhibitory Effect of Dodonaea viscosa var. angustifolia on the Virulence Properties of the Oral Pathogens Streptococcus mutans and Porphyromonas gingivalis. Evid Based Complement Alternat Med. 2013;2013:624089. doi: 10.1155/2013/624089.
CrossRef - Araghizadeh A, Kohanteb J, Fani MM. Inhibitory activity of green tea (Camellia sinensis) extract on some clinically isolated cariogenic and periodontopathic bacteria. Med Princ Pract. 2013;22(4):368-72. doi: 10.1159/000348299.
CrossRef - Kim Y, Kim JH, Jin H, Lee SY. Antimicrobial Activity of Ethanol Extracts of Xanthium strumarium L. against Oral Microorganisms. J Pure Appl Microbio. 2014;8:95-101.
- Teanpaisan R, Teanpaisan R, Senapong S, Puripattanavong J. In vitro Antimicrobial and Antibiofilm Activity of Artocarpus Lakoocha (Moraceae) Extract against Some Oral Pathogens. Tropical Journal of Pharmaceutical Research. 2014;13(7):1149-55. DOI:10.4314/tjpr.v13i7.20
CrossRef - Mehta VV, Rajesh G, Rao A, Shenoy R, Mithunpai BH. Antimicrobial Efficacy of Punica granatum mesocarp, Nelumbo nucifera Leaf, Psidium guajava Leaf and Coffea Canephora Extract on Common Oral Pathogens: An In-vitro Study. J Clin Diagn Res. 2014;8:65-8. Doi: 10.7860/JCDR/2014/9122.4629
CrossRef - Patra JK, Kim ES, Oh K, Kim HJ, Kim Y, Baek KH. Antibacterial effect of crude extract and metabolites of Phytolacca americana on pathogens responsible for periodontal inflammatory diseases and dental caries. BMC Complement Altern Med. 2014;14:343. Doi: 10.1186/1472-6882-14-343
CrossRef - Bersan SM, Galvão LC, Goes VF et al. Action of essential oils from Brazilian native and exotic medicinal species on oral biofilms. BMC Complement Altern Med. 2014 Nov 18;14:451. doi: 10.1186/1472-6882-14-451.
CrossRef - Karygianni L, Cecere M, Skaltsounis AL et al. High-level antimicrobial efficacy of representative Mediterranean natural plant extracts against oral microorganisms. Biomed Res Int. 2014;2014:839019. doi: 10.1155/2014/839019.
CrossRef - Ocheng F, Bwanga F, Joloba M, Borg-Karlson AK, Gustafsson A, Obua C. Antibacterial activities of extracts from Ugandan medicinal plants used for oral care. J Ethnopharmacol. 2014;155, 852:5. 10.1016/j.jep.2014.06.027
CrossRef - Oh JH, Jeong YJ, Koo HJ, et al. Antimicrobial activities against periodontopathic bacteria of Pittosporum tobira and its active compound. Molecules. 2014;19:3607-16. Doi: 10.3390/molecules19033607
CrossRef - Kapadia SP, Pudakalkatti PS, Shivanaikar S. Detection of antimicrobial activity of banana peel (Musa paradisiaca L.) on Porphyromonas gingivalis and Aggregatibacter actinomycetemcomitans: An in vitro study. Contemp Clin Dent. 2015;6:496-9. Doi: 10.4103/0976-237X.169864
CrossRef - Gunjal S, Ankola AV, Bhat K. In vitro antibacterial activity of ethanolic extract of Morus alba leaf against periodontal pathogens. Indian J Dent Res. 2015 Sep-Oct;26(5):533-6. doi: 10.4103/0970-9290.172082.
CrossRef - Rodriguez-Garcia A, Peixoto IT, Verde-Star MJ, Torre-Zavala DS, Aviles-Arnaut H, Ruiz AL. In Vitro Antimicrobial and Antiproliferative Activity of Amphipterygium adstringens. Evid Based Complement Alternat Med. 2015;175497. doi: 10.1155/2015/175497.
CrossRef - Bedran LTB, Morin MP, Spolidorio PD, Grenier D. Black Tea Extract and Its Theaflavin Derivatives Inhibit the Growth of Periodontopathogens and Modulate Interleukin-8 and β-Defensin Secretion in Oral Epithelial Cells. PLoS One. 2015 Nov 18;10(11):e0143158. doi: 10.1371/journal.pone.0143158.
CrossRef - Hussain KA, Tarakji B, Kandy BP, et al. Antimicrobial effects of citrus sinensis peel extracts against periodontopathic bacteria: an in vitro study. Rocz Panstw Zakl Hig. 2015;66(2): 173-8.
- Patra JK, Kim ES, Oh K et al. Bactericidal effect of extracts and metabolites of Robinia pseudoacacia L. on Streptococcus mutans and Porphyromonas gingivalis causing dental plaque and periodontal inflammatory diseases. Molecules. 2015 Apr 8;20(4):6128-39. doi: 10.3390/molecules20046128.
CrossRef - Bharath N, Sowmya NK, Mehta DS. Determination of antibacterial activity of green coffee bean extract on periodontogenic bacteria like Porphyromonas gingivalis, Prevotella intermedia, Fusobacterium nucleatum and Aggregatibacter actinomycetemcomitans: An in vitro study. Contemp Clin Dent. 2015 Apr-Jun;6(2):166-9. doi: 10.4103/0976-237X.156036.
CrossRef - Mallikarjun S, Rao A, Rajesh G, Shenoy R, Pai M. Antimicrobial efficacy of Tulsi leaf (Ocimum sanctum) extract on periodontal pathogens: An in vitro study. J Indian Soc Periodontol. 2016;20(2):145-50. Doi: 10.4103/0972-124X.175177
CrossRef - Leandro LF, Moraes TDS, de Oliveira PF et al. Assessment of the antibacterial, cytotoxic and mutagenic potential of the phenolic-rich hydroalcoholic extract from Copaifera trapezifolia Hayne leaves. J Med Microbiol. 2016 Sep;65(9):937-950. doi: 10.1099/jmm.0.000316.
CrossRef - Sekita Y, Murakami K, Yumoto H et al. Anti-bacterial and anti-inflammatory effects of ethanol extract from Houttuynia cordata poultice. Biosci Biotechnol Biochem. 2016 Jun;80(6):1205-13. doi: 10.1080/09168451.2016.1151339.
CrossRef - Bardají DK, da Silva JJ, Bianchi TC et al. Copaifera reticulata oleoresin: Chemical characterization and antibacterial properties against oral pathogens. Anaerobe. 2016 Aug;40:18-27. doi: 10.1016/j.anaerobe.2016.04.017. E
CrossRef - Mohieldin EAM, Muddathir AM, Mitsunaga T. Inhibitory activities of selected Sudanese medicinal plants on Porphyromonas gingivalis and matrix metalloproteinase-9 and isolation of bioactive compounds from Combretum hartmannianum (Schweinf) bark. BMC Complement Altern Med. 2017 Apr 20;17(1):224. doi: 10.1186/s12906-017-1735-y.
CrossRef - Heyman L, Houri-Haddad Y, Heyman SN, Ginsburg I, Gleitman Y, Feuerstein O. Combined antioxidant effects of Neem extract, bacteria, red blood cells and Lysozyme: possible relation to periodontal disease. BMC Complement Altern Med. 2017 Aug 10;17(1):399. doi: 10.1186/s12906-017-1900-3.
CrossRef - Arbia L, Chikhi-Chorfi N, Betatache I et al. Antimicrobial activity of aqueous extracts from four plants on bacterial isolates from periodontitis patients. Environ Sci Pollut Res Int. 2017 May;24(15):13394-13404. doi: 10.1007/s11356-017-8942-4.
CrossRef - Moreti DLC, Leandro LF, da Silva Moraes T et al. Mikania glomerata Sprengel extract and its major compound ent-kaurenoic acid display activity against bacteria present in endodontic infections. Anaerobe. 2017 Oct;47:201-208. doi: 10.1016/j.anaerobe.2017.06.008.
CrossRef - Koychev S, Dommisch H, Chen H, Pischon N. Antimicrobial Effects of Mastic Extract Against Oral and Periodontal Pathogens. J Periodontol. 2017 May;88(5):511-517. doi: 10.1902/jop.2017.150691.
CrossRef - Jayanti I, Jalaluddin M, Avijeeta A, Ramanna PK, Rai PM, Nair RA. In vitro Antimicrobial Activity of Ocimum sanctum (Tulsi) Extract on actinomycetemcomitans and Porphyromonas gingivalis. J Contemp Dent Pract. 2018 Apr 1;19(4):415-419.
CrossRef - Reddy PRT, Vandana KV, Prakash S. Antibacterial and anti-inflammatory properties of Plantago ovata Forssk. leaves and seeds against periodontal pathogens: An in vitro study. Ayu. 2018 Oct-Dec;39(4):226-229. doi: 10.4103/ayu.AYU_176_16.
CrossRef - Kohli D, Hugar SM, Bhat KG, Shah PP, Mundada MV, Badakar CM. Comparative evaluation of the antimicrobial susceptibility and cytotoxicity of husk extract of Cocos nucifera and chlorhexidine as irrigating solutions against Enterococcus Faecalis, Prevotella Intermedia and Porphyromonas Gingivalis – An in-vitro study. J Indian Soc Pedod Prev Dent. 2018 Apr-Jun;36(2):142-150. doi: 10.4103/JISPPD.JISPPD_1176_17.
CrossRef - Shetty YS, Shankarapillai R, Vivekanandan G et al. Evaluation of the Efficacy of Guava Extract as an Antimicrobial Agent on Periodontal Pathogens. J Contemp Dent Pract. 2018 Jun 1;19(6):690-697.
CrossRef - Hickl J, Argyropoulou A, Sakavitsi ME, et al. Mediterranean herb extracts inhibit microbial growth of representative oral microorganisms and biofilm formation of Streptococcus mutans. PLoS ONE. 2018;13, 207574:10.
CrossRef - Kumar V, Ankola AV, Sankeshwari R, Jalihal S, Deepak V, Jois HS. Assessment of the antimicrobial efficacy of hydroalcoholic fruit extract of cranberry against Socransky complexes and predominant cariogenic, mycotic and endodontic climax communities of the oral cavity: An extensive in-vitro study. J Oral Maxillofac Pathol. 2019 Sep-Dec;23(3):407-411. doi: 10.4103/jomfp.JOMFP_3_18.
CrossRef - Azeez SH, Gaphor SM. Evaluation of Antibacterial Effect against Porphyromonas gingivalis and Biocompatibility of Essential Oil Extracted from the Gum of Pistacia atlantica Kurdica. Biomed Res Int. 2019 May 29;2019:9195361. doi: 10.1155/2019/9195361.
CrossRef - Alirezaei S, Godarzi H, Ghadim MN, Maheri A. Antimicrobial Activity of Aqueous Garlic Extract (Allium sativum) Against Porphyromonas gingivalis: An In-Vitro Study. J Res Dent Maxillofac Sci. 2019;4(4):17-22.
CrossRef - Bankur PK, Mathew M, Almalki SA, Jalaluddin M, Jayanti I, Durgaraju M. An In Vitro Evaluation of Antibacterial Efficacy of Various Concentration of Eucalyptus globulus Leaf Extract on Periodontal Pathogens. J Contemp Dent Pract. 2019 Sep 1;20(9):1041-1044.
CrossRef - Hanafiah RA, Kamaruddin KAC. Saikin NAA et al. Antibacterial Properties of Clinacanthus nutans Extracts Against Porphyromonas gingivalis and Aggregatibacter actinomycetemcomitans: An In-Vitro Study. J Int Dent Med Res. 2019;12, 401:4.
- Nagarajappa R, Bhanushali NV, Ramesh G, Bhanushali PV, Aapaliya P, Pujara P: Antimicrobial Activity of Moringa Oleifera Extracts against Common Periodontal Pathogens: Potential Application in the Prevention and Treatment of Oral Diseases. Indian Journal of Public Health Research & Development. 2019;10, 243-10.
CrossRef - Shekar BRC, Nagarajappa R, Jain R, Suma S, Mruthunjaya K, Thakur R. Evaluating the Antimicrobial Efficacy of an Innovative, Novel Herbal Formulation on Dental Caries and Plaque Microorganisms – A Clinical Research. Biomed Pharmacol J. 2019; 12(4).
CrossRef - Muddathir AM, Mohieldin EAM, Mitsunaga T. In vitro activities of Acacia nilotica (L.) Delile bark fractions against Oral Bacteria, Glucosyltransferase and as antioxidant. BMC Complement Med Ther. 2020; 20:360. Doi: 10.1186/s12906-020-03147-4
CrossRef - Carrol DH, Chassagne F, Dettweiler M, Quave CL. Antibacterial activity of plant species used for oral health against Porphyromonas gingivalis. PLoS One. 2020 Oct 8;15(10):e0239316. doi: 10.1371/journal.pone.0239316.
CrossRef - Mendes FSF, Garcia LM, Moraes TDS, Casemiro LA, Alcântara CB, Ambrósio SR, Veneziani RCS, Miranda MLD, Martins CHG. Antibacterial activity of salvia officinalis L. against periodontopathogens: An in vitro study. Anaerobe. 2020 Jun;63:102194. doi: 10.1016/j.anaerobe.2020.102194.
CrossRef - Satoh Y, Ishihara K. Investigation of the antimicrobial activity of Bilberry (Vaccinium myrtillus L.) extract against periodontopathic bacteria. J Oral Biosci. 2020;62, 169-174. 10.1016/j.job.2020.01.009
CrossRef - Veloso DJ, Abrão F, Martins CHG et al. Potential antibacterial and anti-halitosis activity of medicinal plants against oral bacteria. Arch Oral Biol. 2020 Feb;110:104585. doi: 10.1016/j.archoralbio.2019.104585.
CrossRef - Souissi M, Azelmat J, Chaieb K, Grenier D. Antibacterial and anti-inflammatory activities of cardamom (Elettaria cardamomum) extracts: Potential therapeutic benefits for periodontal infections. Anaerobe. 2020 Feb;61:102089. doi: 10.1016/j.anaerobe.2019.102089.
CrossRef - Kranz S, Guellmar A, Olschowsky P et al. Antimicrobial Effect of Natural Berry Juices on Common Oral Pathogenic Bacteria. Antibiotics (Basel). 2020 Aug 24;9(9):533. doi: 10.3390/antibiotics9090533.
CrossRef - Omwenga EO, Goycoolea FM, Hensel A, Shitandi A. Antimicrobial, cytotoxicity and preliminary phytochemical determination of commonly used medicinal plants to treat oral cavity, urinary tract and gut infections by inhabitants of Borabu sub-county, Nyamira County, Kenya. Malaysian Journal of Microbiology. 2020;16(4):312-322.
CrossRef - Moon SH, Shin SJ, Tae HJ, Oh SH, Bae JM. Effects of Colocasia orum var. Esculenta Extract In Vitro and In Vivo against Periodontal Disease. Medicina (Kaunas). 2021 Oct 2;57(10): 1054. doi: 10.3390/medicina57101054.
CrossRef - Saquib AS, AlQahtani AN, Ahmad I et al. Synergistic antibacterial activity of herbal extracts with antibiotics on bacteria responsible for periodontitis. J Infect Dev Ctries. 2021; Nov 30;15(11):1685-1693. doi: 10.3855/jidc.14904.
CrossRef - Alzandi AA, Taher EA, Azizi M, Al-Sagheer NA, Al-Khulaidi AW, Naguib DM. Antibacterial Activity of Some Medicinal Plants in Al Baha Region, Saudi Arabia, Against Carcinogenic Bacteria Related to Gastrointestinal Cancers. J Gastrointest Cancer. 2023; Mar;54(1):51-55. doi: 10.1007/s12029-021-00793-w.
CrossRef - Sanghavi AD, Chopra A, Shah A, Lobo R, Shenoy PA. Antimicrobial, anti-adhesion, anti-biofilm properties of goji berry (Lycium barbarum) against periodontal bacteria: potential benefits for periodontal diseases. J Complement Integr Med. 2022 Nov 21;20(1):129-136. doi: 10.1515/jcim-2022-0214.
CrossRef - Müller-Heupt LK, Vierengel N, Groß J, Opatz T, Deschner J, von Loewenich FD. Antimicrobial Activity of Eucalyptus globulus, Azadirachta indica, Glycyrrhiza glabra, Rheum palmatum Extracts and Rhein against Porphyromonas gingivalis. Antibiotics (Basel). 2022 Jan 31;11(2):186. doi: 10.3390/antibiotics11020186.
CrossRef - Yoshino N, Ikeda T, Nakao R. Dual Inhibitory Activity of Petroselinic Acid Enriched in Fennel Against Porphyromonas gingivalis. Front Microbiol. 2022 May 19;13:816047. doi: 10.3389/fmicb.2022.816047.
CrossRef - Neto NFO, Bonvicini JFS, deSouza GL et al. Antibacterial activity of Brazilian red propolis and in vitro evaluation of free radical production. Arch Oral Biol. 2022 Nov;143:105520. doi: 10.1016/j.archoralbio.2022.105520.
CrossRef - Kaur J, Sanghavi AD, Chopra A, Lobo R, Saha S. Antimicrobial and cytotoxicity properties of Plumeria alba flower extract against oral and periodontal pathogens: A comparative in vitro study. J Indian Soc Periodontol. 2022 Jul-Aug;26(4):334-341. doi: 10.4103/jisp.jisp_329_21.
CrossRef - Assis DMA, Ramos DPL, Hasna AA et al. Antimicrobial and Antibiofilm Effect of Brazilian Green Propolis Aqueous Extract against Dental Anaerobic Bacteria. Molecules. 2022 Nov 22;27(23):8128. doi: 10.3390/molecules27238128.
CrossRef - Madhloom FA, Al-Taweel BHF, Sha AM, Abdulbaqi RH. Antimicrobial Effect of Moringa Oleifera L. and Red Pomegranate against Clinically Isolated Porphyromonas gingivalis: in vitro Study. Arch Razi Inst. 2022 Aug 31;77(4):1405-1419. doi: 10.22092/ARI.2022.357513.2051.
- Ghafar AMT, Dahham SN. Determination of the inhibitory activity of some plant extracts against the obligate anaerobic bacteria Porphyromonas gingivalis that causes dental caries. Bionatura. 2023;4:1. Doi: 10.21931/RB/CSS/2023.08.04.83
CrossRef - Nayak A, Sowmya BR, Gandla H, Kottrashetti V, Ingalagi P, Srinivas VSC. Determination and comparison of antimicrobial activity of aqueous and ethanolic extracts of Amorphophallus paeoniifolius on periodontal pathogens: An in vitro study. J Indian Soc Periodontol. 2023 Jan-Feb;27(1):40-44. doi: 10.4103/jisp.jisp_182_22.
CrossRef - Rahayu YC, Setiawatie EM, Rahayu RP, et al. Effects of Cocoa Pod Husk Extract (Theobroma Cacao L.) on Alveolar Bone in Experimental Periodontitis Rats. Trends Sci Internet. 2023;20(6):6535.
CrossRef - Ibrahim SM, Al-Mizraqchi AS, Haider J. Metronidazole Potentiation by Panax Ginseng and Symphytum officinale. A New Strategy for P. gingivalis Infection Control. Antibiotics (Basel. 2023;12:1288 doi:10.3390/antibiotics12081288
CrossRef - Güler Ş, Torul D, Kurt-Bayrakdar S, Tayyarcan EK, Çamsarı Ç, Boyacı İH. Evaluation of antibacterial efficacy of Lawsonia inermis Linn (henna) on periodontal pathogens using agar well diffusion and broth microdilution methods: An in-vitro study. Biomedicine (Taipei). 2023 Sep 1;13(3):25-30. doi: 10.37796/2211-8039.1411.
CrossRef - de Melo RN, de Goes VFF, Canelli AP et al. Antibacterial activity of barbatimão (Stryphnodendron adstringens) against periodontopathogens and cytotoxic effects on fibroblasts. Lett Appl Microbiol. 2023 Jun 1;76(6):ovad054. doi: 10.1093/lambio/ovad054. PMID: 37291703.
CrossRef - Ida MJ, Shetty B, Khan SF, Yadalam U, Nambiar M. Development and in vitro characterization of a mucoadhesive gel with Moringa oleifera extract for periodontal drug delivery. J Indian Soc Periodontol. 2023 Mar-Apr;27(2):146-153. doi: 10.4103/jisp.jisp_710_21.
CrossRef - Peeran SW, Murugan M, Fageeh H et al. In-vitro Evaluation of Wild Bitter Melon (Momordica charantia) Extracts against Periodontopathic Bacteria (Prevotella intermedia and Porphyromonas gingivalis) using Antibacterial, Anti-Inflammatory and Molecular Docking Analysis. J Pharm Bioallied Sci. 2024 Apr;16(Suppl 2):S1554-S1564. doi: 10.4103/jpbs.jpbs_512_23.
CrossRef - Shivananda S, Doddawad VG, Bhuyan L, Shetty A, Pushpa VH. Assessment of the Antibacterial Activity of Spilanthes acmella Against Bacteria Associated with Dental Caries and Periodontal Disease: An In-vitro Microbiological Study. J Pure Appl Microbiol. 2024;18(1):476-82. Doi:10.22207/JPAM.18.1.31
CrossRef - Shirai T, Satoh Y, Ishihara K. Antibacterial activity of mulberry extracts and purified fractions against oral pathogenic bacteria. J Oral Biosci. 2024; 66:439-46. Doi: 10.1016/j.job.2023.12.009
CrossRef - Antoniadou M, Rozos G, Vaiou N et al. The In Vitro Assessment of Antibacterial and Antioxidant Efficacy in Rosa damascena and Hypericum perforatum Extracts against Pathogenic Strains in the Interplay of Dental Caries, Oral Health, and Food Microbiota. Microorganisms. 2023 Dec 28;12(1):60. doi: 10.3390/microorganisms12010060.
CrossRef - Hindi NKK, Hadi BH, Al-Shalah LAM, Abbas AS. In vitro study of the antibacterial effects of the Cydonia oblonga extract. Rev. Clin. Pharmacol. Pharmacokinet. Int. 2024; 38(Supp2):117-119.
CrossRef - Astuti LA, Fikriah I, Yani S. The effect of Sarang Semut (Myrmecodia pendens and Myrmecodia tuberosa jack) as Antibacterial for Periodontal Pocket Therapy. J Int Dent Med Res. 2023;16(4):1436-42.
- Lu C, Qing L, Yina L. Phyllanthus emblica fruit extract alleviates halitosis and reduces the inflammatory response to oral bacteria. J Appl Oral Sci. 2024 Jun 21;32:e20240047. doi: 10.1590/1678-7757-2024-0047.
CrossRef - Nakao R, Takatsuka A, Mandokoro K et al. Multimodal inhibitory effect of matcha on Porphyromonas gingivalis. Microbiol Spectr. 2024 Jul 2;12(7):e0342623. doi: 10.1128/spectrum.03426-23.
CrossRef - Bakri HH, Rahman SN, Bakri ZS, Munadziroh E, Harun WHA. Antimicrobial activity of Ruta angustifolia L. Pers against periodontal pathogen: Porphyromonas gingivalis. PeerJ. 2024 Dec 18;12:e18751. doi: 10.7717/peerj.18751.
CrossRef - Nair AR, Rajula BP, Geddam SSS. Antibacterial Activity of Cissus quadrangularis (Veldt Grape) Against Porphyromonas gingivalis, a Keystone Pathogen in Periodontal Disease: An In Vitro Study. Cureus. 2024 Aug 7;16(8):e66377. doi: 10.7759/cureus.66377.
CrossRef - Sebe M, Senoura S, Miura K, et al. Antibacterial Activity of Banglene Extracted from Indonesian Ginger “Bangle” Against Porphyromonas gingivalis. Int J Mol Sci. 2025; 26(5):1787. Doi:10.3390/ijms26051787
CrossRef - Gawade MR, Agrawal AA, Sreeram M, Mohan M. Antimicrobial efficacy of extracts obtained from aerial part, leaves, and flowers of Sphaeranthus indicus on different periodontal pathogens: An in vitro study. J Indian Soc Periodontol. 2024 Sep-Oct;28(5):544-550. doi: 10.4103/jisp.jisp_397_23.
CrossRef - Mohanty R, Asopa SJ, Joseph MD, et al. Red complex: Polymicrobial conglomerate in oral flora: A review. J Family Med Prim Care. 2019 Nov 15;8(11):3480-3486. doi: 10.4103/jfmpc.jfmpc_759_19.
CrossRef - Sonets IV, Galeeva IS, Krivonos DV, et al. In-Depth Multi-Approach Analysis of WGS Metagenomics Data Reveals Signatures Potentially Explaining Features in Periodontitis Stage Severity. Dent J (Basel). 2025;13(12):590. doi: 10.3390/dj13120590.
CrossRef - Benahmed AG, Tippairote T, Gasmi A, et al. Periodontitis Continuum: Antecedents, Triggers, Mediators, and Treatment Strategies. Curr Med Chem. 2024;31(41):6775-6800.
CrossRef - Tolentino PHMP, Dinelli RG, Garzón HS, et al. Evaluation of the antimicrobial effect of cannabidiol (CBD) in a multispecies subgingival biofilm model. J Oral Microbiol. 2025;18(1):2603706
CrossRef - Wu D, Li X, Zhao G, Hao L, Liu X. Discovery of Gingipains and Porphyromonas gingivalis Inhibitors from Food-Derived Natural Products: A Narrative Review. Foods. 2025 Aug 19;14(16):2869. doi: 10.3390/foods14162869.
CrossRef - da Silva Parente TSJ, Sarandy MM, de Araújo ERD, Gonçalves RV, Zucolotto SM. Effect of Moringa oleifera on inflammatory diseases: an umbrella review of 26 systematic reviews. Front Pharmacol. 2025 May 19;16:1572337. doi: 10.3389/fphar.2025.1572337.
CrossRef - Pareek A, Pant M, Gupta MM, Kashania P, Ratan Y, Jain V, Pareek A, Chuturgoon AA. Moringa oleifera: An Updated Comprehensive Review of Its Pharmacological Activities, Ethnomedicinal, Phytopharmaceutical Formulation, Clinical, Phytochemical, and Toxicological Aspects. Int J Mol Sci. 2023 Jan 20;24(3):2098. doi: 10.3390/ijms24032098.
CrossRef - Wu D, Hao L, Liu X, Li X, Zhao G. Comparative transcriptomics reveals the mechanism of antibacterial activity of fruit-derived dihydrochalcone flavonoids against Porphyromonas gingivalis. Food Funct. 2024 Sep 30;15(19):9734-9749. doi: 10.1039/d4fo02854f.
CrossRef - Lima Bezerra JJ, Domingos da Silva JM. Dental applications of Punica granatum L. in the treatment of gingivitis: A review of ethnomedicinal uses, randomized clinical trials, and antibacterial potential against Porphyromonas gingivalis. J Ethnopharmacol. 2025 Jan 10;336:118701. doi: 10.1016/j.jep.2024.11870
CrossRef







