Manuscript accepted on :06-04-2026
Published online on: 13-05-2026
Plagiarism Check: Yes
Reviewed by: Dr. Raju Thenge
Second Review by: Dr. Gowri Burle
Final Approval by: Dr. Patorn Piromchai
Mohammed Hasan Mukhtar 1 , Wesam Ahmed Nasif 1,3
, Wesam Ahmed Nasif 1,3 , Abdullatif Taha Babakr1
, Abdullatif Taha Babakr1 , Sameer Hasan Fatani1
, Sameer Hasan Fatani1 , Mohammad Althubiti1
, Mohammad Althubiti1 , Soud Abdulraof Khogeer1
, Soud Abdulraof Khogeer1 and Mohamed Mahmoud Nour Eldein1,2*
and Mohamed Mahmoud Nour Eldein1,2*
1Department of Biochemistry, Faculty of Medicine, Umm Al-Qura University, Makkah, K.S.A
2Faculty of Medicine, Ain-Shams University, Egypt
3Molecular Biology Department, Genetic Engineering and Biotechnology Research Institute, Sadat City University, Sadat City, Egypt.
Corresponding Author E-mail:mmsayed@uqu.edu.sa
Abstract
The search for reliable biomarkers to predict the severity and outcomes of COVID-19 remains critical. Aim: to evaluate the diagnostic and prognostic value of LDH in patients with COVID-19. A retrospective cohort study was conducted on 2184 confirmed COVID-19 patients from hospitals in Makkah, Saudi Arabia. Demographic, clinical, and laboratory data, including LDH levels, were collected. Statistical analyses were performed using SPSS. The overall recovery rate was 96.4%, with a 3.6% mortality rate. Mean LDH levels were significantly higher in severe cases (399.8 ± 80.60 U/L) compared to non-severe cases (272.9 ± 72.4 U/L) and in deceased patients (488 ± 77 U/L) compared to survivors (250 ± 45 U/L) (p < 0.001). LDH levels were also significantly elevated in COVID-19 patients versus controls. ROC curve analysis demonstrated that LDH could discriminate between COVID-19 patients. Furthermore, LDH showed significant positive correlations with CRP, D-dimer, and ferritin. So Elevated LDH is strongly associated with COVID-19 severity and mortality.
Keywords
COVID-19; Disease Severity; Lactate Dehydrogenase (LDH); Prognostic Biomarker; Risk Stratification
| Copy the following to cite this article: Mukhtar M. H, Nasif W. A, Babakr A. T, Fatani S. H, Althubiti M, Khogeer S. A. Eldein M. M. N. Longitudinal Trends in Lactate Dehydrogenase Levels and COVID-19 Outcomes. Biomed Pharmacol J 2026;19(2). |
| Copy the following to cite this URL: Mukhtar M. H, Nasif W. A, Babakr A. T, Fatani S. H, Althubiti M, Khogeer S. A. Eldein M. M. N. Longitudinal Trends in Lactate Dehydrogenase Levels and COVID-19 Outcomes. Biomed Pharmacol J 2026;19(2). Available from: https://bit.ly/4u7CPFM |
Introduction
Severe Acute Respiratory Syndrome (SARS) SARS-CoV-2 is the causative agent of coronavirus disease 2019 (COVID-19).According to a WHO epidemiological report, the COVID-19 pandemic expanded quickly, claimed many lives, and affected numerous nations.1A new coronavirus known as SARS coronavirus (SARS-CoV) evolved from SARS coronaviruses found in bats (SARSr-CoVs).2The SARS pandemic began when the merged virus infected civets and then humans.3,4Bats likely introduced the Middle East respiratory syndrome coronavirus (MERS-CoV) to dromedary camels thirty years ago.5
Despite widespread awareness, the exact manifestation of severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2)-caused coronavirus illness 2019 (COVID-19) cannot be predicted for any one person. The symptoms of symptomatic COVID-19 can range greatly, from a cough and mild temperature (>37.5°C) to acute respiratory distress syndrome (ARDS) and even death. The disease also has an erratic course. This unpredictability has prompted a desperate hunt for disease severity biomarkers to treat patients effectively and avoid life-threatening consequences. Cytokine storm, or enormous inflammatory activation in response to infection, is a common inflammatory pathophysiology shared by severe COVID-19 and other important disorders. Furthermore, reports of COVID-19 individuals experiencing multi-organ failure (MOF) and organ damage as a result of vasculitis are not uncommon.6 As a result, the majority of biomarkers examined in COVID-19 patients, including interleukin (IL)-6, D-dimer, lymphocyte count (LC), neutrophil count (NC), neutrophil\lymphocyte ratio (NLR), activated partial thromboplastin time (aPTT), and prothrombin time (PT) are associated with coagulation and the immune-inflammatory pathways. Transaminases and Lactate dehydrogenase (LDH) are further supposed non-specific biomarkers of cellular inflammation and damage.7,8
Enzyme LDH converts pyruvate to lactate as glucose is broken down. Secretion of lactate dehydrogenase (LDH) is triggered by cell membrane necrosis, which may indicate viral infection or lung injury, such as pneumonia caused by SARS-CoV-2.9 LDH levels and the onset of COVID-19 illness are strongly correlated.10 According to a study, ICU patients had considerably higher levels of LDH than non-ICU patients. LDH may be a predictive biomarker of severe disease since high levels persisted in ICU patients for days after admission. The one-center study, however, might be subject to selection bias, which might compromise its validity.11 The extent of tissue injury and inflammation was found to be correlated with rising levels of LDH in a multi-center study with 1099 patients . Additionally, considerably greater LDH levels reflected the severity of pneumonia when LDH levels and CT scans were connected [104]. LDH is becoming a more reliable biomarker for assessing the severity of COVID-19 infection. Another investigation discovered that refractory COVID-19 patients had significantly higher LDH levels.12
Elevated serum LDH levels reflect widespread cellular injury and hypoxia, both hallmarks of severe SARS-CoV-2 infection.13,14 In COVID-19, LDH elevation is driven by multiple pathophysiological mechanisms. First, the direct viral cytotoxicity, where SARS-CoV-2 infection induces pyroptosis and necrosis in pulmonary, cardiac, and renal tissues, releasing LDH into circulation.15Additionally, it may be elevated due to the mechanism that involves hypoxia-induced anaerobic metabolism, as severe pneumonia and acute respiratory distress syndrome (ARDS) reduce oxygen delivery, shifting cells to glycolysis and increasing LDH production. Furthermore, it may be elevated due to hyperinflammatory responses, where cytokine storms (e.g., IL-6, TNF-α) exacerbate tissue damage, further elevating LDH. The present study was conducted to evaluate the diagnostic and prognostic value of LDH in Covid-19 patients.
Materials and Methods
Study Design
This study utilized a retrospective cohort design to investigate the demographic, clinical, and laboratory biomarkers associated with COVID-19 severity among patients in various hospitals in Makkah, Saudi Arabia. It differentiated between severe and non-severe cases, as well as recovery versus mortality outcomes, aiming to uncover the complex relationships between specific biomarkers and patient prognoses. Through the comprehensive integration and analysis of electronic health records, our approach facilitated a deep exploration of the multifactorial influences on disease severity and patient survival. By focusing on correlations among diverse markers, ranging from inflammatory responses to organ function indicators, this study aims to enhance the accuracy of predicting COVID-19 outcomes. Ultimately, it seeks to contribute to more targeted and effective patient management strategies, spanning from mild to critical cases, with the overarching goal of improving recovery rates and reducing fatalities. The study was approved by Umm al-Qura University’s Faculty of Medicine Institutional Review Board (IRB number: HAPO-02-K012-2022-09-1183) on 23 September 2022.
Participant Selection
The study included 2184 confirmed COVID-19 patients. Inclusion criteria were aged less than 19 years and above, with a confirmed diagnosis of SARS-CoV-2 infection, selected based on positive RT-PCR tests. Patients were excluded if they had incomplete medical records. This selection strategy ensured a diverse participant pool, reflecting a wide range of demographic backgrounds, comorbidities, and disease severities.
Data Collection
Data was thoroughly collected from patients’ electronic health records from different hospitals in Makkah-Mukarromah, KSA, including demographic information, medical history, clinical symptoms, and laboratory results. The laboratory results, including inflammatory biomarkers, coagulation factors, liver function, kidney function, cardiac function, lipid profile, blood glucose level, and electrolyte levels, were analyzed to determine their association with disease severity and predict prognosis and mortality.
Statistical Analysis
Statistical analyses were conducted with IBM SPSS Statistics. Categorical factors were examined utilizing the chi-square test to evaluate connections between demographic features and COVID-19 results. Continuous variables were assessed using one-way analysis of variance (ANOVA) to compare clinical and laboratory data among patient groups.
Before analysis, the data distribution was evaluated for normality utilizing suitable techniques (e.g., Shapiro–Wilk test). ANOVA was utilized for variables that satisfied normality assumptions, followed by post-hoc multiple comparison tests (e.g., Tukey’s test) to discern intergroup differences.
To address potential confounding variables, multivariate analysis was performed utilizing regression models, controlling for essential characteristics such as age and comorbidities. A p-value of less than 0.05 was deemed statistically significant.
This thorough analytical method allowed the discovery of independent predictive biomarkers, encompassing signs of inflammation and organ function, linked to illness severity and clinical outcomes. These findings offer significant insights into disease development and facilitate risk assessment and clinical decision-making for patients with COVID-19.
Ethical Considerations
The study was conducted with approval from the Umm Al-Qura University Ethics Committee. Given the retrospective design, all data were stored securely, accessible only to the research team. Ethical protocol underscored the importance of balancing scientific inquiry with respect for patient rights and privacy.
Results
Demographics and clinical characteristics
In this study, we collected data from a total of 2184 individuals who were hospitalized due to SARS-CoV-2 infection, which was confirmed using RT-PCR. Among the cohort of COVID-19 patients, 2106 individuals (96.4%) exhibited signs of clinical recovery, while 78 individuals (3.6%) deceased during their hospitalization. The COVID-19 pandemic has highlighted the complex relationship between demographics and clinical outcomes on a global scale. Age, gender, nationality, and blood group are vital determinants that affect the varied clinical expressions of SARS-CoV-2 infection. Moreover, an in-depth analysis of additional biomarkers, including inflammatory biomarkers, coagulation factors, CBC profile, liver function, renal function, cardiac function, lipid profile, glucose blood level, electrolyte and mineral levels, has revealed associations with both severity and mortality in COVID-19 cases.
Demographics of the Patient Population
From the total studied population, the gender distribution was significantly different, with 1482 (67.9%) being male and 702 (32.1%) being female (p<0.001), as shown in Table 1. Furthermore, the analysis revealed a gender disparity within both the survived and deceased groups. Males constituted a higher proportion of those who survived (68%) compared to females (32%), whereas a higher proportion of deceased individuals were male (62.8%) compared to females (37.2%). However, despite these trends, statistical tests indicated that gender did not significantly influence survival or mortality rates, with p-values exceeding 0.2 for both males and females, as illustrated in Table 1.
Table 1: Demographics of COVID-19 survived and deceased cases among gender and age.
| Demographics | Total (2184) | Survived (2106) | Deceased (78) | |||
| No. | % | No. | % | No. | % | |
| Gender | ||||||
| Male | 1482 | 67.9 | 1433 | 68 | 49 | 62.8 |
| Female | 702 | 32.1 | 673 | 32 | 29 | 37.2 |
| *P-value | <0.001 | 0.2 | ||||
| Age | ||||||
| ≤ 19 | 25.0 | 1.14 | 24 | 1.14 | 1.0 | 1.30 |
| 20 – 45 | 963 | 44.1 | 944 | 44.8 | 19 | 24.4 |
| 46 – 60 | 789 | 36.1 | 761 | 36.1 | 28 | 35.9 |
| > 60 | 570 | 26.1 | 534 | 25.4 | 36 | 46.2 |
| *P-value | <0.0001 | <0.001 | ||||
* Significance P-value: (P≤0.05).
The demographic breakdown further detailed the age categories of the individuals. Those aged 19 years old or below constituted 1.14% of the total cases, with a slight difference between the survived rate (1.14%) and deceased (1.30%) cases. The 20- to 45-year age group represented 44.1% of the total cases, with a slightly higher recovery rate (44.8%) compared to the deceased in this bracket (24.4%). Individuals between 46 and 60 years made up 36.1% of the cases, with a consistent survival rate of 36.1% and a closely matching mortality rate of 35.9%. The demographic of individuals aged 60 and above accounted for 26.1% of the overall population. Among them, (25.4%) exhibited a recovery rate, while (46.2%) died due to the disease. This high mortality rate.
Among the older age group, suggests a notable correlation between age and the severity of COVID-19 outcomes in older adults, as seen in Table 1.
The results of the current study demonstrated that while most individuals recovered across all age groups, a significant disparity in outcomes is only evident in those aged 19 years old and below. For patients aged 20 years and above, the differences in the percentage of recovered versus deceased cases were not statistically significant. This suggests that younger individuals with COVID-19 have a significantly higher probability of recovery compared to older individuals. However, the outcomes for older age groups do not significantly differ from one another based on age alone, as shown in Table 1.
Comorbidity Illnesses
Individuals with comorbidities, such as hypertension, diabetes, Asthma, CKD, CVD, and lung diseases, are at increased risk of severe COVID-19 and death. This is because co-morbid illnesses can weaken the immune system and make it more difficult for the body to fight off the virus. Additionally, some co-morbid illnesses can damage the lungs and other organs, which can make them more vulnerable to COVID-19 infection. Table 2 indicates that the mortality rate for COVID-19 infection is markedly elevated in patients with hypertension (p<0.0001), diabetes (p<0.001), and asthma (p<0.0001), in comparison to those who recovered. No significant difference in death rate was noted between persons with CKD, CVD, or lung illnesses and those who recovered (P > 0.05).
Table 2: COVID-19 survived and deceased group, including Comorbid Illnesses.
| Co-morbidity Illnesses | Total (2184) | Survived (2106) | Deceased (78) | *P-value | |||
| No. | % | No. | % | No. | % | ||
| Hypertension | 387 | 17.7 | 355 | 16.9 | 32 | 41.0 | 0.0001 |
| Diabetes | 467 | 21.4 | 434 | 20.6 | 33 | 42.3 | 0.001 |
| Asthma | 33 | 1.51 | 27.0 | 1.28 | 6.0 | 85.7 | 0.0001 |
| CKD | 144 | 6.59 | 142 | 6.74 | 11 | 14.1 | 0.182 |
| CVD | 119 | 5.45 | 110 | 5.22 | 9.0 | 11.5 | 0.217 |
| Lung diseases | 36 | 1.65 | 31.0 | 1.47 | 5.0 | 6.41 | 0.240 |
*Significance P-value: (P≤0.05).
CKD: Chronic Kidney Disease, CVD: Cardiovascular Disease.
Muscle, liver, and heart are among the many tissues that contain the enzyme lactate dehydrogenase (LDH). Its release into the bloodstream may indicate tissue damage or inflammation. Individuals with severe COVID-19 had significantly higher mean LDH levels (399.8 ± 80.60 U/L) than those with non-severe cases (272.9 ± 72.4 U/L; p < 0.001), suggesting greater tissue damage or inflammation associated with disease severity. This association extended to mortality, with deceased individuals exhibiting significantly elevated LDH levels (488 ± 77 U/L) compared to recovered individuals (250 ± 45 U/L), p < 0.001, as shown in fig.1.
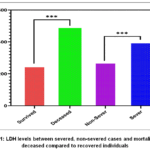 |
Figure 1: LDH levels between severed, non-severed cases and mortality with deceased compared to recovered individuals |
COVID-19 patients presented with significantly higher mean LDH levels (328.1 ±137.60 U/L) compared to negative cases (248.9 ± 81.4 U/L), p < 0.001 fig.2.
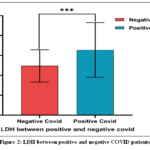 |
Figure 2: LDH between positive and negative COVID patients |
COVID-19 patients who deceased presented with significantly higher mean LDH levels (416.8 ±279.1 U/L) compared to improved patients (324.9 ± 129.1 U/L), p < 0.001 within COVID-19 positive groups, Fig. 3.
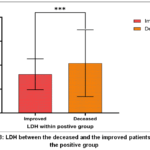 |
Figure 3: LDH between the deceased and the improved patients within the positive group. |
Patients infected with COVID-19 had considerably higher blood levels of LDH-5 as compared to Creatine Kinase (CK), with a mean value of 328.1 U/L and 278.6 U/L (P < 0.001).
Association between LDH-1 and LDH-2 isoenzymes with heart enzymes among COVID-19 Infection.
LDH-1 (4H) and LDH-2 (3H1M) are the primary isoenzymes found in the heart muscle. Elevated levels of LDH-1 and LDH-2 in the blood can be indicative of heart damage. The serum levels of LDH-1 and LDH-2 were significantly and substantially increased in patients with COVID-19 compared with those of Creatine Kinase (CK) and creatine kinase MB (CK-MB), with mean values of 328.1 U/L, 278.6 U/L, and 28.3 U/L, respectively (P < 0.001).
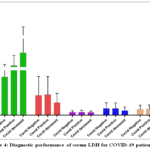 |
Figure 4: Diagnostic performance of serum LDH for COVID-19 patients. |
Correlation of LDH with CRP, D-Dimer, and Ferritin in COVID-19 patients, the LDH isoenzymes studied showed a significant positive correlation with CRP (r = 0.130; P < .001), fig.5a, ferritin (r = 0.152; P < .001) fig.5b, and D-Dimer (r = 0.016; P < .001) fig.5c.
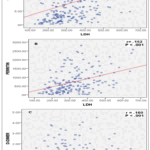 |
Figure 5: Correlation of LDH with CRP, D-Dimer, and Ferritin in COVID-19 patients. |
To determine if serum LDH levels could distinguish between COVID-19-positive and -negative samples, the ROC curve was analyzed. You can see the AUC in Figure 6. A statistically significant AUC of 0.765 and a P-value of less than 0.001 were shown by the data analysis of the ROC curve. The sensitivity was found to be 86.6% and the specificity to be 49.4% when a cutoff value of LDH equal to 241 U/mL was used.
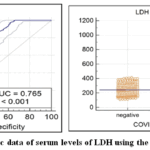 |
Figure 6: Diagnostic data of serum levels of LDH using the ROC curve. |
| Variable | AUC | SE | 95% CI | Sensitivity | Specificity | Cutoff value |
| LDH | 0.765 | 0.0189 | 0.731 to 0.797 | 86.6 | 49.4 | 241 |
Discussion
This study provides a comprehensive analysis of the multifaceted impact of COVID-19, examining demographic factors, comorbidities, laboratory markers, and other factors related to disease outcomes. A detailed examination of hospitalized COVID-19 patients reveals critical insights into how demographic variables, such as age and gender, and laboratory parameters influence disease prognosis and severity. Key findings highlight the high recovery rate among patients, gender, and age-related disparities in outcomes. Additionally, the study delves into the prognostic value of laboratory markers, with a focus on Lactate dehydrogenase as a diagnostic and prognostic biomarker in COVID-19 patients. Lactate dehydrogenase (LDH), a cytoplasmic enzyme critical for anaerobic glycolysis, has emerged as a robust biomarker for predicting disease severity and mortality in COVID-19 patients.
In the present study, a significant increase in LDH levels in cases of COVID-19 compared to normal controls was observed, as shown in Figure 2. Moreover, LDH presented a stronger biomarker for both COVID-19 severity and mortality.IndividualswithsevereCOVID-19 showed significantlyelevated LDH levelscompared to non-severe cases, with deceased patients exhibitingnotablyhigherLDHlevelscomparedtothosewhorecovered, as shown in Figure 1. This pattern underlines LDH’s potential as a predictive marker for both the severity of infection and mortality risk among COVID-19 patients.Our findings align with existing literature, where elevated CK and LDH levels have been notedfortheirassociationswithCOVID-19outcomes.Forinstance,researchbyJohnson,Harris.16 on CK levels among different COVID-19 severity groups support our observations regarding the enzyme’s elevated levels in severe cases. In parallel, the work by Grivas, Khaki.17 on LDH as an indicator of tissue damage and its prognostic significance in COVID-19 further validates our conclusions regarding LDH’s association with both disease severity and mortality.
Compared to other studied biomarkers such as CK, CK-MB, AST, and ALT, the levels of LDH showed marked elevation in deceased cases of COVID-19 patients, as shown in Figure 3. This may point to the association of this biomarker with severity and mortality scenarios of the disease. The enzyme’s broad tissue distribution (lungs, heart, liver, kidneys) also makes it a nonspecific but sensitive indicator of multiorgan involvement, common in fatal COVID-19 cases. The present study showed that there is a significant correlation between LDH levels and CRP, a nonspecific indicator of inflammation, D-dimer, and ferritin, which was suggested as a strong prognostic marker for COVID-19 in previous studies.18, as shown in Figure 5. Clinically, LDH levels correlate strongly with disease progression, with studies showing that patients with LDH levels >350–400 U/L have higher risks of ICU admission, mechanical ventilation, and mortality.19,20 Meta-analyses reveal that elevated LDH is one of the most consistent laboratory predictors of poor outcomes, often outperforming other biomarkers like CRP or D-dimer in multivariate models. For instance, Szabolcs Kiss and colleagues in a systematic review and meta-analysis found that LDH >500 U/L was associated with a 6-fold increase in mortality risk. 21
The data strongly highlights Lactate Dehydrogenase (LDH) as the most significantly altered biomarker in the context of severe and fatal COVID-19. This finding aligns with established clinical observations that elevated LDH, an indicator of tissue damage and cell death, is a robust predictor of disease severity and mortality in COVID-19 patients. In contrast, the other enzymes show less dramatic or more complex patterns. The dead group’s Creatine Kinase (CK) levels are shockingly lower than those of the positive and negative groups. This finding defies logic and might be due to a small sample size effect or a particular stage of the disease. Alanine Aminotransferase (ALT) and Aspartate Aminotransferase (AST) are prevalent indicators of hepatic damage, and remain relatively low across all cohorts, with only minor elevations in the COVID-positive and deceased groups. This suggests that while some degree of liver involvement may occur, it is not the primary or most pronounced systemic damage reflected by these specific biomarkers in this dataset. The overall pattern underscores the utility of LDH as a critical prognostic tool, differentiating between mild/moderate infection and the severe, life-threatening systemic pathology characteristic of fatal COVID-19 cases.
While LDH lacks disease specificity, its dynamic rise during hospitalization can signal worsening clinical status, guiding interventions such as oxygen therapy or immunomodulators. Future research should explore LDH isoenzymes (e.g., LDH-3 for lung injury) to enhance prognostic precision. In summary, LDH’s role as a biomarker underscores its utility in risk stratification, reflecting the interplay of cellular hypoxia, inflammation, and organ damage in COVID-19 pathogenesis.
Limitations
Notwithstanding the significant findings, this study had numerous critical limitations. The retrospective methodology naturally includes possible selection and information biases, thereby compromising the accuracy and completeness of the obtained data and constraining causal inference. The lack of multivariate analysis is a notable methodological restriction, since it hinders the identification of independent predictors and the control of concurrent effects from many variables. Therefore, the documented correlations must be regarded with prudence.
Insufficient adjustment for potential confounding variables, notably age and underlying comorbidities, may have affected the reported correlations between biomarkers and clinical outcomes. The investigation was ultimately performed at a single center and within a defined demographic, potentially introducing population-specific bias and constraining the external validity and generalizability of the results to other contexts or groups.
Conclusion
This study confirms that elevated lactate dehydrogenase (LDH) is a significant biomarker for predicting COVID-19 severity and mortality. Patients with severe disease and fatal outcomes exhibited markedly higher LDH levels, which also correlated strongly with inflammatory markers like CRP and ferritin. The enzyme’s rise reflects widespread cellular injury and multi-organ involvement characteristic of advanced COVID-19. Therefore, serial monitoring of LDH is clinically valuable for risk stratification, early identification of high-risk patients, and guiding timely medical interventions to improve outcomes and reduce mortality.
Acknowledgement
The authors extend their appreciation to Al Noor Hospital, Makkah, Saudi Arabia, for permission to collect the data
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This study complied with the ethical standards specified in the 1975 Declaration of Helsinki, and clearance was obtained from the medical ethics committee of the College of Medicine at Umm Al-Qura University in Makkah, Kingdom of Saudi Arabia (Approval Number: HAPO-02-K012-2022-09-1183).
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Author contributions
- Wesam Ahmed Nasif: design of the study, clinical selection, diagnosis, and classification of the cases, interpreted biochemical laboratory investigations, preparing tables and figures, editing, styling, writing, and revising of the main manuscript text.
- Mohammed Hasan Mukhtar: design of the study, clinical selection, diagnosis and classification of the cases, revising of the main manuscript text revising of the main manuscript text.
- Mohamed Mahmoud Nour Eldein: design of the study, statistical analysis, interpretation of biochemical results, preparing tables and figures, writing, and revising of the main manuscript text.
- Sameer Hasan Fatani: design of the study, styling, diagnosis and classification of the cases, and revising of the main manuscript text.
- Abdullatif Taha Babakr: Conceptualization, Investigation, Data curation, Writing—original draft, Writing—review & editing.
- Mohammad Althubiti: Conceptualization, Investigation, Data curation, Writing—original draft, Writing—review & editing.
- Soud Abdulroof Khogeer: Data collection,Data processing, and preparing tables and figures
References
- WHO, World Health Organization coronavirus disease (COVID-19) dashboard. World Health Organization, 2020.
- Alagaili, Abdulaziz N et al. “Middle East respiratory syndrome coronavirus infection in dromedary camels in Saudi Arabia.” mBio.Feb. 2014;(5)2:e01002-14 .
CrossRef - Song, Huai-Dong et al. “Cross-host evolution of severe acute respiratory syndrome coronavirus in palm civet and human.” Proceedings of the National Academy of Sciences of the United States of America.2005;(102)7: 2430-5.
- Chinese SARS Molecular Epidemiology Consortium. “Molecular evolution of the SARS coronavirus during the course of the SARS epidemic in China.” Science (New York, N.Y.). 2004;(303)5664:1666-9.
CrossRef
- Müller, Marcel A et al. “MERS coronavirus neutralizing antibodies in camels, Eastern Africa, 1983-1997.” Emerging infectious diseases. 2014;(20)12:2093-5.
CrossRef
- Bohn, M.K., et al., Molecular, serological, and biochemical diagnosis and monitoring of COVID-19: IFCC taskforce evaluation of the latest evidence. Clinical Chemistry and Laboratory Medicine (CCLM). 2020; 58(7): 1037-1052.
CrossRef
- Lin, L., et al., Hypothesis for potential pathogenesis of SARS-CoV-2 infection–a review of immune changes in patients with viral pneumonia. Emerging microbes & infections.2020;9(1):727-732.
CrossRef
- Tang, N., et al., Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. Journal of thrombosis and haemostasis. 2020;18(4): 844-847.
CrossRef
- Ferrari, D., et al., Routine blood tests as a potential diagnostic tool for COVID-19. Clinical chemistry and laboratory medicine (CCLM).2020; 58(7): 1095-1099.
CrossRef
- Luo, W., et al., Clinical findings of 35 cases with novel coronavirus pneumonia outside of Wuhan. 2020.
CrossRef - Guan, W.J., et al., Clinical Characteristics of Coronavirus Disease 2019 in China. N Engl J Med. 2020; 382(18):1708-1720.
CrossRef
- Xiong, Y., et al., Clinical and high-resolution CT features of the COVID-19 infection: comparison of the initial and follow-up changes. Investigative radiology, 2020.
CrossRef - Wu J, Stefaniak J, Hafner C, et al. Intermittent Hypoxia Causes Inflammation and Injury to Human Adult Cardiac Myocytes. Anesth Analg. Feb 2016;122(2):373-80.
CrossRef
- Ochani R, Asad A, Yasmin F, et al. COVID-19 pandemic: from origins to outcomes. A comprehensive review of viral pathogenesis, clinical manifestations, diagnostic evaluation, and management. Infez Med. Mar 1 2021;29(1):20-36.
- Karki R, Sharma BR, Tuladhar S, et al. Synergism of TNF-α and IFN-γ Triggers Inflammatory Cell Death, Tissue Damage, and Mortality in SARS-CoV-2 Infection and Cytokine Shock Syndromes. Cell. Jan 7 2021;184(1):149-168.e17.
CrossRef
- Grivas, P., et al., Association of clinical factors and recent anticancer therapy with COVID-19 severity among patients with cancer: a report from the COVID-19 and Cancer Consortium. Annals of oncology. 2021;32(6): 787-800.
CrossRef
- Johnson, A.K., Ghazarian, Z., Cendrowski, K. D., & Persichino, J. G. , Pulmonary aspergillosis and mucormycosis in a patient with COVID-19. Medical mycology case reports. 2021;32: 64-67.
CrossRef
- Para O, Caruso L, Pestelli G, et al. Ferritin as prognostic marker in COVID-19: the FerVid study. Postgrad Med. Jan 2022;134(1):58-63.
CrossRef
- Battaglini D, Lopes-Pacheco M, Castro-Faria-Neto HC, Pelosi P, Rocco PRM. Laboratory Biomarkers for Diagnosis and Prognosis in COVID-19. Front Immunol. 2022;13:857573. doi:10.3389/fimmu.2022.857573
CrossRef - Pezoulas VC, Kourou KD, Mylona E, et al. ICU admission and mortality classifiers for COVID-19 patients based on subgroups of dynamically associated profiles across multiple timepoints. Comput Biol Med. Feb 2022;141:105176.
CrossRef
- Kiss S, Gede N, Hegyi P, et al. Early changes in laboratory parameters are predictors of mortality and ICU admission in patients with COVID-19: a systematic review and meta-analysis. Med Microbiol Immunol. Feb 2021;210(1):33-47.
CrossRef







