Manuscript accepted on :26-02-2026
Published online on: 27-04-2026
Plagiarism Check: Yes
Reviewed by: Dr. Kashif Zayan
Second Review by: Dr. Amit Panaskar
Final Approval by: Dr. Hanefi ÖZBEK
Yallamalli Indira Muzib* and Chella Shilpaja
and Chella Shilpaja
Department of Pharmaceutics, Institute of Pharmaceutical Technology, Sri Padmavati Mahila Visvavidyalayam (Women's University), Tirupati, Andhra Pradesh, India.
Corresponding Author E-mail:yindira1415@gmail.com
Abstract
Alzheimer’s disease (AD) is a chronic neurodegenerative condition characterized by progressive memory loss and cognitive dysfunction. Although donepezil (DPZ), an acetylcholinesterase inhibitor, is frequently prescribed to manage AD symptoms, its effectiveness is hindered by limited brain penetration due to the blood-brain barrier (BBB) and extensive first-pass metabolism. This research focused on formulating a nanoemulsion (DPZ-NE) for intranasal delivery to improve brain targeting and therapeutic outcomes. A pseudo-ternary phase diagram was developed to determine the optimal composition, using oleic acid as the oil phase, PEG 400 as a co-surfactant, and Tween 80 as a surfactant. The resulting nanoemulsion displayed an average droplet size of 24.9 nm, a polydispersity index (PDI) of 0.748 ± 0.04, and a zeta potential of -0.1 mV, indicating nanoscale size and colloidal stability. In vitro release studies showed a controlled release pattern of the drug over time. These findings suggest that intranasal administration of DPZ-NE could be a promising approach in Alzheimer's treatment, following Higuchi kinetics, suggesting a diffusion-controlled mechanism. In vivo neuroprotective activity was assessed using a scopolamine-induced dementia model in Wistar rats. Behavioral tests, including the Morris water maze and active avoidance response, demonstrated significant cognitive improvements in DPZ-NE-treated animals compared to the control. Biochemical assays showed reduced acetylcholinesterase activity and enhanced antioxidant enzyme levels, indicating neuroprotection. Histopathological analysis confirmed reduced amyloid plaque formation and neuronal degeneration. These findings suggest that intranasal DPZ-NE effectively enhances drug bioavailability, facilitates BBB penetration, making it a promising approach for Alzheimer’s treatment. Further studies are required to assess its clinical potential.
Keywords
: Alzheimer’s Disease; Blood-Brain Barrier; Donepezil; Intranasal Delivery; Neuroprotection; Nanoemulsion
| Copy the following to cite this article: Muzib Y. I, Shilpaja C. Development of a Brain-Targeted Donepezil Nanoemulsion via Intranasal Route with Pharmacodynamic Study for Alzheimer’s Management. Biomed Pharmacol J 2026;19(2). |
| Copy the following to cite this URL: Muzib Y. I, Shilpaja C. Development of a Brain-Targeted Donepezil Nanoemulsion via Intranasal Route with Pharmacodynamic Study for Alzheimer’s Management. Biomed Pharmacol J 2026;19(2). Available from: https://bit.ly/3P0eAtU |
Introduction
Alzheimer’s disease (AD) is a chronic neurodegenerative condition characterized by progressive memory loss and cognitive dysfunction.1It is associated with loss of synapsesand neurons,as well as the formation of abnormal protein aggregates.2 One of the most significant pathogenic feature of AD is cholinergic insufficiency,which is connected to the loss of cognition and memory.The production of free radicals is also thought to be an essential element in the pathogenesis of AD. The reversible cholinesterase inhibitors showed high selectivity for centrally active cholinesterase. Acetylcholinesterase inhibitors are available in the market to treat AD by various routes of administration.3Donepezil Hydrochloride, an inhibitor of acetylcholinesterase with a molecular weight of 379.492daltons, is primarily utilized for the treatment of Alzheimer’s disease. Donepezil has been authorized by the Food and Drug Administration (FDA) for managing dementia linked to mild, moderate, and severe stages of Alzheimer’s disease. Currently, no proof indicates that donepezil alters the progression of the disease.Commercially, oral tablets and transdermal patchesare available in doses of 5 and 10mg. Oral route of administration results in side effects includes anorexia,gastrointestinal hemorrhage,muscular convulsion,nausea and diarrhea, due to extensive first-pass metabolism. Also, BBB is another obstacle that causes the low quantity of drug available in the brain.Therefore, an alternative route of administration is necessary to reduce unwanted side effects.
The intranasal route is the most promising for the safe,non-invasive transport of medication to the brain. This route not only reduces the unwanted side effects, circumvents the BBB, but also directs the drug to the brain, and prevents the drug from reaching the GIT. Nasal mucosa is characterized by high vascularization, large absorptive surface area, and rapid onset of action. Delivery of the drug through nasal route will target either olfactory or trigeminal neuronal pathways, showing a significant link between the nasal mucosa and the brain.4 This route is also convenient for those who are unable to swallow the solid dosage forms. Intra-nasal route possesses some disadvantages like less invitro residence time,limited surface area,low epithelial permeability,enzymatic degradation,small volume of drug for administration and mucociliary clearance leads to removal of drug from the site.5 Therefore there is a need to develop the dosage form which overcome these disadvantages.
The use of nanocarriers as a promising tool to deliver the drugs through intra nasal route.
Various drug carriers like cubosomesRP Patil et al.,6nanoemulsionsKaur et al.,7 hydrogelsAI Harthi et al.,8 nanostructured lipid carriersTekade et al.,9 solid lipid nanocarriers (Yasir et al.,10nanosuspensions MDS et al.,11 liposomesRajput et al.,12 used to deliver the drug through intra nasal route.
Nanoemulsion is one of the most common dosage forms for delivering active substances to the target. Nanoemulsions are colloidal nanocarriers with a low droplet size is less than 250 nm and low viscosity.They are promising systems to deliver the drug from nose to brain due to their low droplet size,high permeability, solubilizing potential and their protective nature for lipophilic drugs.13 Therefore, the present research aims to formulate and evaluate donepezil-loaded nanoemulsion (DPZ-NE) intended for intranasal delivery. The nanoemulsion was optimized through pseudo-ternary phase diagram construction and subsequently assessed for its physicochemical attributes, drug release behaviour in vitro, and neuroprotective efficacy using a scopolamine-induced dementia model. The primary objective is to enhance the bioavailability and brain-targeting efficiency of DPZ, thereby improving its therapeutic effectiveness and offering a promising alternative strategy for the management of Alzheimer’s disease.
Materials and Methods
Materials
Cipla Pharmaceuticals Ltd., Ahmedabad, provided a free sample of donepezil hydrochloride.
Oleic acid was received from Gattefosse, France. PEG400 and PEG200 ethylene glycol were received from S.D Fine chemicals ltd. Tween 80, and Transistor-P received from Corel Pharma Chem, Ahmedabad. Other surfactants, co-surfactants, chemicals & reagents utilised were made of Analytical grade.
Methods
Determining the drug saturation mobility in oils, surfactants, and Co-surfactants:
Donepezil Hydrochloride solubility in castor oil, soya oil, oleic acid, etc., as well as surfactants and co-surfactants such as Tween-80, Tween-20, PEG-200, 400, and polypropylene glycol was assessed by introducing an excess quantity (100 mg) of the drug into 2 mL of the selected oil, co-surfactants, and surfactants each in separate 5 mL volume stoppered vials, and blending with a cyclo mixer for 15 minutes. These vials were maintained at 25°C ± 1.0°C in an isothermal (orbital) shaker with continuous shaking at 100 rpm for 24 hours, then equilibrated for 24 hours. The balanced samples were centrifuged for 15 minutes at 3000 rpm. The supernatant obtained was diluted with chloroform/methanol and measured spectrophotometrically for dissolved drug at 230 nm. The findings were shown as the average value (mg/mL) ± standard error of the mean.
Pseudo-ternary phase diagram construction
To identify the optimal region for nanoemulsion formation, pseudo-ternary phase diagrams were constructed using the aqueous phase titration method. Solubility studies involving oleic acid (as the oil phase), Tween 80 (surfactant), PEG 400 (co-surfactant), and distilled water (aqueous phase) facilitated the development of multiple phase diagrams.9,10Various volume ratios of surfactant to co-surfactant (Smix)—including 1:1, 1:2, 2:1, 3:1, 4:1, and 5:1—were prepared to generate different diagrams. For each system, mixtures of oil and Smix were combined in varying ratios from 1:9 to 9:1. Aqueous phase was gradually titrated into various mixtures of oil and Smix using a micropipette, with constant stirring, until a clear oil-in-water (O/W) nanoemulsion was achieved. The nanoemulsion region was identified and represented on a pseudo-ternary phase diagram, where each axis corresponds to one component: aqueous phase, oil, and Smix (a fixed ratio mixture of surfactant and co-surfactant). These diagrams were constructed and visualized using Fire software (version 3.60).14
Preparation of drug-loaded nanoemulsion
Donepezil solubility in surfactants, oils, and co-surfactants was assessed to formulate the nanoemulsion. Various mixtures were formulated by blending chosen oils, surfactants, and co-surfactants, and later vortexing them. The prepared combinations were kept overnight to ensure the stability concerning miscibility and clarity. Ultimately, the nanoemulsion was produced through a pre-homogenization process succeeded by ultrasonication. To prepare the final nanoemulsion, the pre-emulsion has undergone high shear homogenization with a Tissue Master 125 Homogenizer while in an ice bath, followed by high-energy ultrasonication using a Bench Top Ultrasonicator (Figure 1).14
Table 1: Composition of nanoemulsion with different ratios
| Formulation | Drug (mg) | Oil (ml) | Smix (surfactant and co-surfactant)ml | Water (ml) |
| F1 | 20 | 0.2 | 1:1 | 18 |
| F2 | 20 | 0.2 | 1:2 | 17 |
| F3 | 20 | 0.3 | 2:1 | 17 |
| F4 | 20 | 0.3 | 3:1 | 16 |
| F5 | 20 | 0.4 | 4:1 | 15 |
| F6 | 20 | 0.4 | 5:1 | 14 |
| F7 | 20 | 0.5 | 6:1 | 13 |
| F8 | 20 | 0.5l | 7:1 | 12 |
| F9 | 20 | 0.6 | 8:1 | 11 |
| F10 | 20 | 0.6 | 9:1 | 10 |
Evaluation Test
Measurement of Transmittance:
A UV/Vis spectrophotometer was employed to assess the transmittance percentage of the created NEs in triplicate and evaluate their clarity. Milli Q water served as a blank.15
pH determination of the prepared nanoemulsion:
This parameter was measured in triplicate with a calibrated pH meter.
Measurement of viscosity:
A Brookfield viscometer was used to measure the viscosity of the nanoemulsion. The measurement was done by the trial-and-error method using spindle no. 18 and a small sample adapter. The sample was filled in a sample adapter, and the rpm was optimised to obtain 80 to 100% torque. In this way, 50 rpm was considered optimum, and all measurements were done in triplicate. 16
Zeta potential
Zeta potential instrument (Horiba Scientific SZ-100) was used to measure the zeta potential. The electric field ranged from −120 to 120 V in the case of zeta potential. As a result, particles move at a zeta potential-dependent velocity.17
Particle size & Polydispersity index:
A Zetasizer (Horiba Scientific SZ-100) was employed to assess the size of the particles and PDI of the nanoemulsion, and this was further diluted using distilled water and filtered with a 0.45 μm membrane filter. Later, 1 mL of the NE sample was poured into transparent polystyrene cuvettes for measuring PDI and globule size. Dynamic light scattering is the principle employed in the Zetasizer.18
Scanning Electron Microscopy (SEM)
The surface characteristics of Donepezil hydrochloride, PEG 400, oleic acid, and Tween 80 were examined utilising SEM (Vega 3 Tescan). The specimens were analysed using electron microscopy, displayed in secondary electron mode.19,20
In-vitro release studies
The in vitro drug release profile was evaluated using a dialysis membrane method (molecular weight cut-off: 12,000–14,000 Da) in simulated cerebrospinal fluid (CSF) at pH 7.4 and 37 ± 2 °C. Prior to the release study, the dialysis membranes were pre-soaked in simulated CSF for 24 hours. Each formulation (2 mL, containing 0.474 mg of donepezil) was placed into individual dialysis bags, which were then submerged in 100 mL of simulated CSF containing 25% (w/v) methanol to maintain sink conditions. The system was stirred at 100 rpm throughout the experiment. At predetermined time intervals (0, 0.5, 1, 2, 3, 4, 8, 10, and 24 hours), 3 mL aliquots were withdrawn and immediately replaced with an equal volume of fresh medium to preserve sink conditions. Samples were appropriately diluted and analysed in triplicate using a UV-visible spectrophotometer at 230 nm.21,22
In vivo neuroprotective activity of Donepezil Nano emulsion (F1)
Animals employed
Healthy adult male Albino Wistar rats, weighing between 200 and 250 grams, were used for the study. The animals were housed in standard laboratory cages under controlled environmental conditions, adhering to proper hygienic practices. They were maintained on a standard pellet diet (Amrut Laboratory Animal Diet) with unrestricted access to clean drinking water. The IACE committee sanctioned the animal studies aimed at overseeing and regulating animal experiments (CPCSEA).
Experimental design
The subjected animals were divided into 4 groups (6 rats/group). Doses were administered orally for 14 consecutive days. Except for the control group, all animals received an intraperitoneal injection of scopolamine hydrobromide (5 mg/kg) one hour following the final dose of the test substances. Animals were grouped and treated as follows: Group I received normal saline; Group II received scopolamine (5 mg/kg); Groups III and IV were treated with Scopolamine + Donepezil Hydrochloride (2 mg/kg) and Scopolamine + Donepezil Nanoemulsion, respectively. 23
A total of 24 rats were involved, with six rats in each group:
Group I: Control (healthy rats with no scopolamine treatment)
Group II: Scopolamine-induced dementia group
Group III: Scopolamine + Donepezil-treated group (2 mg/kg/day, orally)
Group IV: Scopolamine + F1 Donepezil nanoemulsion group (intranasal Donepezil nanoemulsion at 1 mg/kg equivalent dose)
Evaluation parameters
At the end of the treatment period, rats were subjected to behavioural tests to assess their memory and learning using the Morris Water Maze and Elevated Plus Maze.24 At the conclusion of the experiment, the rats were sacrificed, and their brains were carefully extracted and split into two hemispheres.25 The right hemisphere was preserved in 10% formaldehyde for histopathological evaluation, while the left hemisphere was stored at –80 °C for biochemical analyses, including assessment of acetylcholinesterase (AChE) activity,26catalase levels,27malondialdehyde (MDA) content,28 and glutathione (GSH) levels.29
Statistical Research
Statistical analysis was performed using one-way ANOVA followed by Dunnett’s post hoc test, utilising GraphPad Prism software. Data are expressed as mean ± standard error of the mean (SEM), with a sample size of n = 6 per group. A p-value of ≤ 0.001 was considered statistically significant.
Results
Donepezil hydrochloride solubility was carried out on oils, surfactants & co-surfactants. Considering solubility, oleic acid was utilised as the oily phase, while PEG 400 served as the co-surfactant & Tween 80 as surfactant (Table 2).
Table 2: Evaluating the saturation solubility of a drug in oils, surfactants, and co-surfactants
| Oils | Solubility in mg/ml(n=3)
Mean±SD |
Surfactant | Solubility in mg/ml(n=3)
Mean±SD |
Co-surfactant | Solubility in mg/ml(n=3)
Mean±SD |
| Oleic Acid | 95 ± 2.64 | Tween 80 | 85 ± 2.0 | PEG200 | 70 ± 2.05 |
| Castor oil | 80 ± 1.01 | Tween 20 | 75 ± 2.0 | PEG400 | 80 ± 1.5 |
| Soybean oil | 45 ± 1.23 | Propylene glycol | 65 ± 1.5 |
Development of the Pseudo ternary phase diagram:
Pseudo-ternary phase diagrams were constructed for each Smix ratio using oleic acid as the oil phase, distilled water as the aqueous phase, Tween-80 as the surfactant, and PEG 400 as the co-surfactant. A total of six phase diagrams were generated with Smix ratios of 1:1, 2:1, 3:1, 4:1, and 5:1. The nanoemulsion (NE) regions were identified and highlighted within each diagram. Among the ratios tested, the Smix ratio of 1:2 demonstrated the largest nanoemulsion region, indicating superior emulsification capacity. The size and extent of the nanoemulsion region are largely influenced by the ability of the Smix to solubilise the oil phase. Moreover, minimising the system’s free energy is crucial for achieving stable formulations with optimal droplet size (Figure 1).
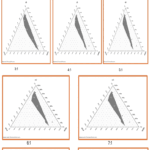 |
Figure 1: Pseudo-Ternary Phase Diagrams with Different ratios of Smix [Surfactant: Co-Surfactant] |
Prepared Nanoemulsion Characterization
Measurement of Transmittance
The percentage transmittance (%T) of the prepared nanoemulsion was determined using a UV-visible spectrophotometer at a wavelength of 270 nm, with distilled water used as the blank. The formulation exhibited a %T value of 98.21 ± 2.31%, indicating high clarity and transparency, characteristic of a well-formed nanoemulsion.
pH of the prepared Nano emulsion
pH of all prepared batches of Nano emulsion was found between 5.98 – 7.40.
Viscosity measurement
It was measured by a Brookfield viscometer under 80 to 100 % torque. The viscosity of all prepared batches was found in the range of 65.42 – 80.54 cp.
Zeta potential
A 1ml sample was placed in a folded disposable capillary cell. Zeta potential was measured using the instrument. The electric field ranges from -120 to 120 V. The optimised formulation had a zeta potential of -0.1mV (Figure 2).
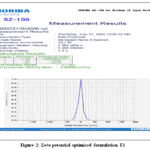 |
Figure 2: Zeta potential optimised formulation F1 |
Particle size and Poly dispersity index determination
A Zeta sizer was employed for measuring the globule size and PDI of nanoemulsion later diluting with the distilled water that was filtered through 0.45μm membrane. The optimised formulation had a mean 24.9 nm globule size & 0.748 PDI (Figure 3).
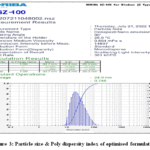 |
Figure 3: Particle size & Poly dispersity index of optimised formulation |
Scanning electron microscopy (SEM)
The surface properties of Donepezil hydrochloride, oleic acid, PEG 400, and Tween 80 were investigated using SEM (Vega 3 Tuscan). An acceleration potential of 10kV was applied by scanning the specimens using an electron beam and the images were captured in the secondary electron mode. Figure 4 illustrates the findings.
 |
Figure 4: Particle size determination of F1 |
In-Vitro release studies
The cumulative assessments for the release of Donepezil Hydrochloride from the nanoemulsion (NE) and the drug solution were conducted in a simulated phosphate-buffered saline (PBS) at a pH of 7.4. After 24 hours, the total amount of Donepezil Hydrochloride released from the NE was recorded at 88.90 ± 4.2, whereas the majority of the drug from the solution was released within only 4 hours. The graph illustrates that the NE exhibited an initial burst release, likely due to the presence of nanodroplets located near the NE’s surface, followed by a gradual release phase. The extended release period may result from the delayed release of the drug from the oily core at the oil-water interface, as the aqueous medium acts as a barrier to drug transport. the dialysis bag served as a physical barrier, permitting only the unbound drug to pass through its nanosized pores. The in vitro drug release data for the formulation were analysed using various release kinetic models, including zero-order, first-order, Higuchi, and Peppas models, to assess the release mechanism from the nanoemulsion. The results demonstrated that the drug release best fit the zero order and Higuchi model, as indicated by the highest correlation coefficient (R² = 0.961). This behavior suggests that the dialysis membrane acts as a controlling barrier, limiting diffusion within the reservoir system (Figure 5).
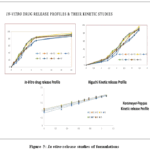 |
Figure 5: In vitro release studies of formulations |
Table 3 Kinetics release study:
| Zeroorder | Ist Order | Higuchi | Korsemeyer-Pepps | |
| R2 | R2 | R2 | R2 | n |
| 0.96 | 0.756 | 0.961 | 0.943 | 0.611 |
In vivo neuroprotective activity
Effect of F1 on Active avoidance response
Rats treated with scopolamine showed reduced performance in the retention trial compared to the acquisition trial, indicating impaired learning and memory relative to the control group. In contrast, rats receiving donepezil along with scopolamine exhibited a higher active avoidance response during the retention trial than during acquisition. Similarly, co-administration of the F1 formulation with scopolamine resulted in a significant improvement in retention performance compared to acquisition, suggesting enhanced learning and memory. The results are shown in Figure 6.
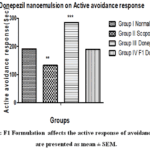 |
Figure 6: F1 Formulation affects the active response of avoidance. Values are presented as mean ± SEM. |
Animals in the scopolamine-treated group(GroupII) showed impaired performance in the active avoidance test. Treatment with Scopolamine + Donepezil(groupIII) and Scopolamine + F1 intranasal formulation(GroupIV) significantly improved active avoidance response, as evidenced by a significant reduction in avoidance latency (sec) compared to the scopolamine group (***p < 0.001).
F1 Formulation intranasal group effect on Escape latency time (ELT)
Engaging in the Morris water maze task over the course of seven days improves rats’ ability to find the concealed platform in the designated quadrant. Rats that received scopolamine treatment exhibited longer escape latencies (the duration required to locate a platform) relative to those given the vehicle–inoculated controls through the training days, thereby suggesting more deficits. Groups that were treated with saline quickly acquired the location of the platform. F1 Formulation treated groups Trough nasal route, like the groups treated with donepezil, had significantly lowered the Scopolamine effects on Escape latency (EL). Results were shown in Figures 7 & 8.
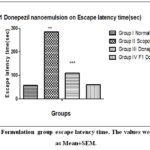 |
Figure 7: F1 Formulation group escape latency time. The values were expressed as Mean±SEM. |
Animals that were treated with scopolamine had a higher Escape latency time (**p<0.001) in comparison to the control group. In contrast, animals treated with scopolamine along with donepezil or the F1 intranasal formulation exhibited a significant reduction in escape latency time when compared to the scopolamine group (***p < 0.001).
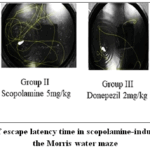 |
Figure 8: Recording of escape latency time in scopolamine-induced dementia by using the Morris water maze |
Effect of F1 Formulation intranasal group on acetylcholinesterase
Scopolamine-treated animals had higher levels of acetylcholinesterase (0.60±0.11) compared to the control group, while Scopolamine + donepezil-treated animals had lower levels of acetylcholinesterase levels (0.18±0.18) when compared to scopolamine-treated animals and animals treated with Scopolamine + F1 Formulation, the intranasal group had shown a significant decrease in Acetylcholinesterase levels (0.20±0.17) compared to scopolamine-treated animals. Results were shown in Figure 9.
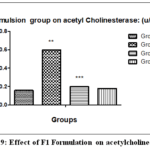 |
Figure 9: Effect of F1 Formulation on acetylcholinesterase |
Animals treated with scopolamine had higher levels of acetylcholinesterase (** p < 0.001) in comparison to the normal control. Scopolamine + Donepezil and the Scopolamine + F1 Formulation intranasal groups had significantly lower levels of acetylcholinesterase (*** p<0.001) when compared to the Scopolamine group.
F1 Formulation effect on catalase levels (µM/mg/min)
Animals treated with scopolamine had lower catalase levels (21.76±13.90) as compared to controls. Scopolamine + Donepezil-treated animals had higher catalase levels (44.85±8.65) compared to scopolamine-treated animals. Animals treated with Scopolamine + F1 Donepezil nanoemulsion intranasally had significantly higher catalase levels than scopolamine-treated animals. Based on these findings, F1 Donepezil nanoemulsion significantly increases catalase levels (42.55±4.42). Results were shown in Figure 10.
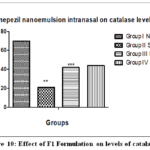 |
Figure 10: Effect of F1 Formulation on levels of catalase. |
The values were expressed in terms of mean ± SEM. Animals treated with scopolamine showed significantly reduced levels of catalase (** p< 0.001) in comparison to the normal control group. Scopolamine + Donepezil and Scopolamine + F1 Donepezil nanoemulsion intranasal treated animals possess significantly greater catalase values (***p<0.001) than the Scopolamine treated group.
F1 Formulation reduced Glutathione levels (μmol/g tissue) effect
Scopolamine-treated animals have lower Reduced Glutathione levels (35.36±1.21) compared to the control group. Scopolamine + Donepezil-treated animals have higher Reduced Glutathione levels (90.1±0.18) compared to scopolamine-treated animals. Scopolamine + F1 Donepezil nanoemulsion intranasal-treated animals have significantly higher reduced Glutathione levels than scopolamine-treated animals. Based on the results, F1 Donepezil nanoemulsion intranasal significantly increases the Reduced Glutathione levels (88.9±0.98). The results were shown in Figure 11.
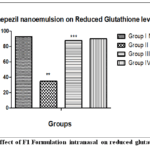 |
Figure 11: Effect of F1 Formulation intranasal on reduced glutathione levels. |
The values were represented as Mean ± SEM. Animal group treated with Scopolamine shows lowering of glutathione levels (**p<0.001) to that of normal control. Scopolamine + Donepezil & Scopolamine + F1 Formulation intranasal treated animals showed significantly higher reduced values of glutathione (***p <0.001) than to the group that was treated with Scopolamine.
Histopathological studies
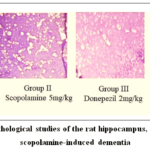 |
Figure 12: Histopathological studies of the rat hippocampus, part of the brain in scopolamine-induced dementia |
Normal control rat demonstrating histological hippocampal structure.
Rats treated with scopolamine exhibited notable congestion in the blood capillaries, which was accompanied by perivascular edema (Scars), along with the presence of edema and amyloid plaques in the hippocampus.
Dementia induced by scopolamine in rats undergoing donepezil treatment showed widespread gliosis and reduced nuclear size in hippocampal pyramidal cells.
In comparison to the control, rats treated with nanoemulsion exhibit reduced development of amyloid plaques in the hippocampal area of the brain.
Discussion
The development of a Donepezil nanoemulsion
(DPZ-NE) Intranasal delivery demonstrated significant potential for enhancing the drug’s therapeutic efficacy in Alzheimer’s disease management. The selection of oleic acid as the oily phase, PEG 400 as the co-surfactant, and Tween 80 as the surfactant was based on solubility studies, ensuring optimal formulation stability and drug dispersion. Pseudo-ternary phase diagrams indicated that the 1:2 Smix ratio provided the largest nanoemulsion region, suggesting an ideal composition for stable nanoemulsion formation. Characterisation studies confirmed that the optimised formulation exhibited desirable physicochemical properties, including a transparent appearance, Newtonian flow behaviour, and a nanosized globule diameter of approximately 24.9 nm.
In vitro release profile
The in vitro release profile of DPZ-NE adhered to Higuchi kinetics, suggesting a diffusion-driven release mechanism. In contrast to traditional Donepezil solutions, the nanoemulsion exhibited a more prolonged and controlled release, which could potentially improve therapeutic outcomes by sustaining drug concentrations in the brain. Additionally, the nanoemulsion’s enhanced permeation across the nasal mucosa was further validated by zeta potential analysis and its pseudoplastic characteristics, both of which promote improved mucosal adhesion and absorption.
In vivo studies
In vivo studies using scopolamine-induced dementia models provided strong evidence of DPZ-NE’s neuroprotective potential. Behavioral tests, including the Morris water maze and active avoidance response, showed that nanoemulsion-treated animals exhibited significantly improved cognitive function compared to those receiving oral Donepezil. Additionally, biochemical analysis revealed that DPZ-NE effectively reduced acetylcholinesterase activity while increasing antioxidant enzyme levels, such as catalase and glutathione. These findings suggest that the nanoemulsion not only enhances drug delivery to the brain but also plays a role in mitigating oxidative stress and neurodegeneration.
Histopathological analysis
Histopathological analysis of hippocampal tissues further confirmed the neuroprotective effects of DPZ-NE. The nanoemulsion-treated group exhibited reduced amyloid plaque deposition and neuronal damage compared to the scopolamine-induced dementia group, indicating a protective effect against Alzheimer’s pathology. These findings collectively highlight the potential of intranasal DPZ-NE as a promising strategy for improving Donepezil’s bioavailability and therapeutic impact in Alzheimer’s disease. Future studies should focus on clinical translation and long-term safety assessments to validate its effectiveness in human populations.
Conclusion
The study successfully developed a Donepezil-loaded nanoemulsion (DPZ-NE) aimed at improving brain-targeting and enhancing the therapeutic efficacy of Donepezil for Alzheimer’s disease management. The optimized nanoemulsion exhibited a stable and nanosized globule diameter (24.9 nm) and a zeta potential of -0.1 mV, indicative of its stability and favourable physicochemical properties. The in vitro drug release followed Higuchi’s diffusion-controlled model, providing sustained drug release compared to conventional formulations, which could enhance therapeutic outcomes by maintaining drug concentrations in the brain. In vivo neuroprotective studies using a scopolamine-induced dementia rat model demonstrated significant cognitive improvements in animals treated with the nanoemulsion compared to those receiving conventional Donepezil. The nanoemulsion treatment effectively reduced acetylcholinesterase levels, increased antioxidant enzyme levels (such as catalase and glutathione), and reduced oxidative stress, suggesting neuroprotective effects. Histopathological analysis confirmed the reduction of amyloid plaque deposition and neuronal damage, indicating the nanoemulsion’s potential for combating Alzheimer’s pathology.
This study underscores the potential of intranasal DPZ-NE as a promising strategy for enhancing Donepezil’s bioavailability and therapeutic efficacy in Alzheimer’s disease treatment. Future studies focusing on clinical translation will be essential to validate the effectiveness of this formulation in human populations.
Acknowledgement
All authors are thankful to the higher authorities of IPT, SPMVV, Tirupati, for providing facilities to conduct this research work.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
The study involves an experiment on animals; then the name of the authorizing body is CPCSEA, and the number is 1858/PO/Re/S/16/CPCSEA, Sandip Institute of Pharmaceutical Sciences.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
AuthorContributions
- Yallamalli Indira Muzib Conceived and designed the study, formal analysis, experimental design, and supervision.
- Chella Shilpaja performed the experiments, performed data analysis, contributed to data interpretation, and wrote the initial draft of the manuscript. Both authors critically revised the manuscript and approved the final version.
References
- Birks JS, Harvey RJ. Donepezil for dementia due to Alzheimer’s disease. Cochrane Database Syst Rev.2018;2018(6): CD001190. Doi: 10.1002/14651858.
CrossRef - Coman H, Nemeş B. New therapeutic targets in Alzheimer’s disease. International Journal of Gerontology. 2017;11(1):2-6https://doi.org/10.1016/j.ijge.2016.07.003
CrossRef - Chatzitaki AT, Jesus S, Karavasili C, Andreadis D, Fatouros DG, Borges O. Chitosan-coated PLGA nanoparticles for the nasal delivery of ropinirole hydrochloride: In vitro and ex vivo evaluation of efficacy and safety. International journal of pharmaceutics.2020; 589:119776.https://doi.org/10.1016/ j.ijpharm. 2020.119776
CrossRef - Crowe T.P., Greenlee M.H.W., Kanthasamy A.G., Hsu W.H. Mechanism of intranasal drug delivery directly to the brain. Life Sciences. 2018;195:44–52. doi:10.1016/j.lfs.2017.12.025.
CrossRef - Md S., Bhattmisra S.K., Zeeshan F., Shahzad N., Mujtaba M.A., Srikanth Meka V., Radhakrishnan A., Kesharwani P., Baboota S., Ali J. Nano-carrier enabled drug delivery systems for nose to brain targeting for the treatment of neurodegenerative disorders. Journal of Drug Delivery Science and Technology. 2018;43:295–310. doi:10.1016/j.jddst.2017.09.022.
CrossRef - Patil R.P., Pawara D.D., Gudewar C.S., Tekade A.R. Nanostructured cubosomes in an in situ nasal gel system: an alternative approach for the controlled delivery of donepezil HCl to brain. Journal of Liposome Research. 2019;29(3):264–273.
CrossRef - Kaur A., Nigam K., Bhatnagar I., Sukhpal H., Awasthy S., Shankar S., Tyagi A., Dang S. Treatment of Alzheimer’s disease using donepezil nanoemulsion: An intranasal approach. Drug Delivery and Translational Research. 2020;10(6):1862–1875. doi:10.1007/s13346-020-00754-z.
CrossRef - Al Harthi S., Alavi S.E., Radwan M.A., El Khatib M.M., AlSarra I.A. Nasal delivery of donepezil HCl-loaded hydrogels for the treatment of Alzheimer’s disease. Scientific Reports. 2019;9(1):9563.
CrossRef - Tekade A., Susar R., Kulkarni G., Surwade S., Gaikwad A. Nanostructured lipid carriers of donepezil hydrochloride for the treatment of Alzheimer’s disease. Current Alzheimer Research. 2024;21(10):710–722.
CrossRef - Yasir M., Sara U.V., Chauhan I., Gaur P.K., Singh A.P., Puri D., Ameeduzzafar. Solid lipid nanoparticles for nose to brain delivery of donepezil: Formulation, optimization by Box–Behnken design, in vitro and in vivo evaluation. Artificial Cells, Nanomedicine, and Biotechnology. 2018;46(8):1838–1851.
- Md S., Ali M., Ali R., Bhatnagar A., Baboota S., Ali J. Donepezil nanosuspension intended for nose to brain targeting: in vitro and in vivo safety evaluation. International Journal of Biological Macromolecules. 2014;67:418–425.
CrossRef - Rajput A., Butani S. Donepezil HCl liposomes: development, characterization, cytotoxicity, and pharmacokinetic study. AAPS PharmSciTech. 2022;23(2):74.
CrossRef - Wen M.M., El-Salamouni N.S., El-Refaie W.M., Hazzah H.A., Ali M.M., Tosi G., Farid R.M., Blanco-Prieto M.J., Billa N., Hanafy A.S. Nanotechnology-based drug delivery systems for Alzheimer’s disease management: Technical, industrial, and clinical challenges. Journal of Controlled Release. 2017;245:95–107. doi:10.1016/j.jconrel.2016.11.025.
CrossRef - Bonferoni M.C., Rossi S., Sandri G., . Intranasal delivery of drugs for CNS targeting: in vivo evidences and nanotechnological approaches for enhancement. Nanomaterials. 2021;11(3):698. doi:10.3390/nano11030698.
CrossRef - Alam T., Pandit J., Vohora D., Aqil M., Ali A., Sultana Y. Optimization of nanostructured lipid carriers of lamotrigine for brain delivery: in-vitro characterization and in vivo efficacy in epilepsy. Expert Opinion on Drug Delivery. 2015;12(2):181–194. doi:10.1517/17425247.2015.980831.
CrossRef - Lee D., Shen A.M., Shah M., Garbuzenko O.B., Minko T. In vivo evaluation of nose-to-brain delivery of liposomal donepezil, memantine, and BACE-1 siRNA for Alzheimer’s disease therapy. International Journal of Molecular Sciences. 2024;25(19):10357.
CrossRef - Kumar M., Misra A. Intranasal nanoemulsion-based brain targeting drug delivery system. International Journal of Pharmaceutics. 2008;358(1–2):285–291.
CrossRef - Shehata M.K., Ismail A.A., Kamel M.A. Combined donepezil with astaxanthin via nanostructured lipid carriers: effective delivery to the brain for Alzheimer’s disease in a rat model. International Journal of Nanomedicine. 2023;18:4193–4227. doi:10.2147/IJN.
CrossRef - Tekade A.R., Suryavanshi M.R., Shewale A.B., Patil V.S. Design and development of donepezil hydrochloride-loaded nanostructured lipid carriers for efficient management of Alzheimer’s disease. Drug Development and Industrial Pharmacy. 2023. doi:10.1080/03639045.2023.2262035.
CrossRef - Sokolova V., Mekky G., van der Meer S.B., et al. Transport of ultrasmall gold nanoparticles (2 nm) across the blood-brain barrier in a six-cell brain spheroid model. Scientific Reports. 2020;10(1):18033. doi:10.1038/s41598-020-75125-2.
CrossRef - Kamat P.K., Kalani A., Rai S., et al. Streptozotocin intracerebroventricular-induced neurotoxicity and brain insulin resistance: a therapeutic intervention for treatment of sporadic Alzheimer’s disease (AD)-like pathology. Molecular Neurobiology. 2016;53(7):4548–4562. doi:10.1007/s12035-015-9384-y.
CrossRef - Wang Z., Gonzalez K.M., Cordova L.E., Lu J. Nanotechnology-empowered therapeutics targeting neurodegenerative diseases. Wiley Interdisciplinary Reviews: Nanomedicine and Nanobiotechnology. 2023;15(5): e1907. doi:10.1002/wnan.1907.
CrossRef - Perna A.F., Ingrosso D., De Santo N.G. Homocysteine and oxidative stress. Amino Acids. 2003;25(3–4):409–417.
CrossRef - Morris R. Developments of a water-maze procedure for studying spatial learning in the rat. Journal of Neuroscience Methods. 1984;11(1):47–60. doi:10.1016/0165-0270(84)90007-4.
CrossRef - Paxinos G., Watson C. The Rat Brain in Stereotaxic Coordinates. 6th ed. Academic Press; 2007.
- Ellman G.L., Courtney K.D., Andres V. Jr., Featherstone R.M. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochemical Pharmacology. 1961;7(2):88–95. doi:10.1016/0006-2952(61)90145-9.
CrossRef - Aebi H. Catalase in vitro. Methods in Enzymology. 1984;105:121–126. doi:10.1016/S0076-6879(84)05016-3.
CrossRef - Ohkawa H., Ohishi N., Yagi K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Analytical Biochemistry. 1979;95(2):351–358. doi:10.1016/0003-2697(79)90738-3.
CrossRef - Ellman G.L. Tissue sulfhydryl groups. Archives of Biochemistry and Biophysics. 1959;82(1):70–77. doi:10.1016/0003-9861(59)90090-6.
CrossRef







