Manuscript accepted on :08-01-2026
Published online on: 27-01-2026
Plagiarism Check: Yes
Reviewed by: Dr. Karthikeyan
Second Review by: Dr. Karuna Priyachitra
Final Approval by: Dr. Patorn Piromchai
Zafar Isomiddinovich Sanoev1,4 , Sokhib Zamon ogli Rashidov1,4
, Sokhib Zamon ogli Rashidov1,4 , Tolmas Tolibovich Khamroev1,4*
, Tolmas Tolibovich Khamroev1,4* , Ibrokhimjon Tuychievich Abdinazarov1,5
, Ibrokhimjon Tuychievich Abdinazarov1,5 , Sukhrob Davlatyor ogli Rakhimboev2
, Sukhrob Davlatyor ogli Rakhimboev2 , Dilnoza Safaralievna Ismailova3
, Dilnoza Safaralievna Ismailova3 , Burkhon Zhuraevich Elmuradov3
, Burkhon Zhuraevich Elmuradov3 and Madina Aladdin kizi Shakhmurova4
and Madina Aladdin kizi Shakhmurova4
1Department of Pharmacology and Toxicology of the Institute of Chemistry of Plant Substances named after Academician S.Y. Yunusov of the Academy of Sciences of the Republic of Uzbekistan, Uzbekistan.
2Department of Biomedical Sciences, Faculty of Medicine of Kimyo international university in Tashkent, Uzbekistan.
3Department of Organic Synthesis of the Institute of Chemistry of Plant Substances named after Academician S.Y. Yunusov of the Academy of Sciences of the Republic of Uzbekistan, Uzbekistan.
4Department of Pharmacology of Tashkent State Medical University. Uzbekistan.
5Profi University, Bektemir dist, Tashkent, Uzbekistan
Corresponding Author; E-mail: tolmastht@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3381
Abstract
Many psychiatrists have consistently pointed out the link between stressors and the emergence of depressed moods. Both acute traumatic experiences and long-term psychogenic stress can contribute to the development of depressive disorders in people. The aim of this research is to develop a novel antidepressant medication based on compound-1, 5-(2-aminophenyl)-1,3,4-oxadiazole-2-(3H)-thione. The reference medication fluoxetine and 5-(2-aminophenyl)-1,3,4-oksadiazole-2(3H)-thione (compound 1) were the subjects of the investigation. Their effects were evaluated using models such as the FST and TST, which measure behavioral despair and immobility in response to stress. Other factors that were assessed included acute toxicity, locomotor activity, research activities, anti-anxiety activity, and the spectrum of antidepressant action. In all studies, chemical 1 was administered orally at dosages of 0.1, 0.5, 1.0, 5.0, 10.0, 30.0, and 60.0 mg/kg. Experiments conducted on models of stress-induced depression revealed that compound-1 has a strong antidepressant effect when given once to mice (male and female). Under compound-1's influence, the animals' total immobilization time decreased and the latency period before the onset of immobility increased statistically substantially in the "non-escapable forced Porsolt swimming" and "hanging mice by the tail" tests. Under the effect of the test drug, the number of jumps made by the animals and the total amount of time spent actively swimming increased statistically considerably in the "non-escapable forced swimming, but Porsolt" test. The uniqueness and benefits of compound-1 as a possible antidepressant medication have therefore been demonstrated by neuro- and psychopharmacological research comparing it to the well-known antidepressant fluoxetine.
Keywords
Depression models; Depressive states; Forced swim test; Fluoxetine; Immobilization; Tail suspension test
Download this article as:| Copy the following to cite this article: Sanoev Z. I, Rashidov S. Z. O, Khamroev T. T, Abdinazarov I. T, Rakhimboev S. D. O, Ismailova D. S, Elmuradov B. Z, Shakhmurova M. A. K. Synthesis and Antidepressant Activity of 5-(2-Aminophenyl)-1,3,4-Oxadiazole-2-(3H)-Thione. Biomed Pharmacol J 2026;19(1). |
| Copy the following to cite this URL: Sanoev Z. I, Rashidov S. Z. O, Khamroev T. T, Abdinazarov I. T, Rakhimboev S. D. O, Ismailova D. S, Elmuradov B. Z, Shakhmurova M. A. K. Synthesis and Antidepressant Activity of 5-(2-Aminophenyl)-1,3,4-Oxadiazole-2-(3H)-Thione. Biomed Pharmacol J 2026;19(1). Available from: https://bit.ly/45u1L0a |
Introduction
The connection between stress factors and the development of depressive states has been repeatedly noted by a number of psychiatrists. Depressive disorders in humans develop both as a result of an acute traumatic situation and under the influence of prolonged psychogenic stress.1 Depression is classified as an affective (emotional) syndrome in which a psychopathological condition manifests itself in the form of a persistent decrease in mood. In depression, the emotional, intellectual, volitional and physical spheres are involved in the pathological process. A reduction in the production and metabolism of monoamines in the central nervous system, including dopamine, serotonin, and norepinephrine, is linked to the mechanisms of depression development. Currently, special importance is attached to the serotonergic theory of depression. At the same time, it is believed that the main cause of the development of a depressive state is a deficiency of serotonin in the synaptic space. Depressive-like conditions that develop in animals with chronic repetition of traumatic effects can be considered as analogies of psychogenic (reactive) depressions in humans. Depressive disorders are among the most common mental disorders. According to various authors, the incidence of depressive syndrome among the population ranges from 6 to 20.6%. Depression is a chronic recurrent disease that worsens their professional and social functioning, as well as their quality of life. In 15% of cases, patients suffering from depression commit suicide attempts, which is about 66% of all suicide attempts.2 Despite the fact that a number of antidepressant drugs have been developed to date, the problem of pharmacotherapy of depression is far from being solved. It is known that the administration of most antidepressants during course therapy (6-8 weeks) in adequate therapeutic doses leads to a therapeutic effect in only 70% of patients.3 At the same time, the lack of clinical effect in the remaining patients is associated with significant impairments in cognitive and socially oriented activities. In addition, the antidepressants used have a number of disadvantages: a long latent period of action, an insufficient range of therapeutic effects, a high probability of side effects, and the risk of toxic reactions in case of overdose.4 All this determines the need for further search for effective antidepressant drugs. However, in order to study new antidepressants, it is necessary to recreate adequate and reproducible models of depressive states in animals.5 Antidepressants come in dozens of names worldwide, each of which belongs to a different structural class and has a unique mode of action. The patent6 describes compounds, heterobicyclo-substituted-[1,2,4] triazolo[1,5c] quinazoline 5-amine, having the properties of A2A antagonists acting on a disorder of the central nervous system, which is a motor disorder associated with Parkinson’s disease, or the treatment of this disorder using dopaminergic therapy. Recently, a combination is proposed that includes mirtazapine or its pharmacologically acceptable salt and one or more selective serotonin reuptake inhibitors7 or their pharmacologically acceptable salts, which can be used to treat various types of depression and persistent depression, such as endogenous depression, major depression, melancholia, etc. The most popular antidepressant, fluoxetine, was selected as the prototype and comparison medication.8 One of these drugs is fluoxetine (prozac, framex). It is a derivative of phenoxypropylamine based on its chemical structure. Like tricyclic antidepressants, it has a strong antidepressant effect. The effect takes one to four weeks to manifest. In contrast to tricyclic antidepressants, it has virtually no sedative effect, typically exhibits some psychostimulating impact, has no or very little M-holinoblocking effect, and has no effect on adrenoreceptors. Hemodynamics is stable when using fluoxetine. Reduced appetite, nausea, anxiety, headaches, sleeplessness, and skin rashes are among the side effects. It may result in weight reduction and has an anorexic impact. When used with indiscriminate monoamine oxidase (MAO) inhibitors, fluoxetine may cause the “serotonin syndrome,” which is linked to the build-up of high serotonin levels. This can show up as potentially fatal circulatory collapse, muscle rigidity, and heat. Having said that, fluoxetine and non-selective MAO inhibitors should be taken at least two weeks apart. Unlike non-selective MAO inhibitors, fluoxetine has not been shown to interact with dietary constituents. Fluoxetine has been selected as a near homologue since it is frequently used in medical practice to treat depressive conditions. Among the five-membered heterocyclic compounds, 1,3,4-oxadiazole derivatives are very promising structural fragments in the development of effective drugs.9,10,11,12,13 1,3,4-oxadiazole derivatives have a wide range of biological effects: analgesic, antifungal, antibacterial, anticonvulsant, anti-inflammatory, antiviral, antitumor, hypotensive, etc. As a result, it is fascinating to synthesize new heterocyclic compounds that contain pieces of this cycle and investigate their biological characteristics. In this regard, the objective of this study is to study the antidepressant activity based on 5-(2-aminophenyl)-1,3,4-oxadiazole-2-(3H)-thione (compound-1) of a new compound.
Materials and Methods
Mature male mice weighing 18–24 g were used in the studies, which were conducted during the day in natural light. Ten animals were utilized for each test’s preparations. Each lab animal was kept in a standard vivarium with unlimited access to water and a complete laboratory meal for the duration of the experiments. The studies were conducted in accordance with the international guidelines set forth in the “Guide for the Care and Use of Laboratory Animals” (National Research Council, 2011). The Republic of Uzbekistan’s Ethic Committee approved the experiment before to its start (protocol § 1/1-1628, 14/02/2023).
Compound 1 (5-(2-aminophenyl)-1,3,4-oksadiazole-2(3H)-thione) and the reference medication fluoxetine were the subjects of the investigation. The forced swim test (FST) and tail suspension test (TST), which measure behavioral despair and immobility in response to stress, were among the models used to assess acute toxicity, locomotor activity, research activities, anti-anxiety activity,17 and the spectrum of antidepressant action. Compound 1 was administered orally at doses of 0.1, 0.5, 1.0, 5.0, 10.0, 30.0, and 60.0 mg/kg in each experiment.
Statistical analysis. The standard software program BIOSTAT 2009 was used to perform a one-way analysis of variance on the obtained data, and the paired Student’s test was used to perform variation statistics and determine the significance of the indicators (Mean±Std error). When the 95% p-value was less than 0.05, differences between the groups being compared were considered significant.
Results
Chemical characterization of 5-(2-aminophenyl)-1,3,4-oksadiazole-2(3H)-thione (compound 1)
In order to obtain 5-(2-aminophenyl)-1,3,4-oxadiazole-2-(3H)-thion (2a), we cyclized 2-aminohydrazide (1) and the potassium salt of hexylxanthogenate by boiling equimolar amounts of reagents in an aqueous alcohol solution for 10 hours:
As a result, 5-(2-aminophenyl)-1,3,4-oxadiazole-2-(3H)-thion (2a) was obtained in almost quantitative (98%) yield in the form of a white powder (Figure 1).
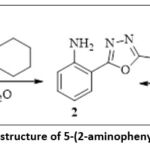 |
Figure 1: Formation and chemical structure of 5-(2-aminophenyl)-1,3,4-oxadiazole-2-(3H)-thione. |
Synthesis of 5-(2-aminophenyl)-1,3,4-oxadiazole-2-(3H)-thione (2a): a mixture of 31 g (0.2 mol) of 2-aminobenzohydrazide (1) and 45.36 g (0.21 mol) of potassium salt of hexylxanthogenate in 160 ml of a mixture of solvents – ethanol+water (3:1) boil for 10 hours. The reaction proceeds with the release of hydrogen sulfide (H2S). After the release of hydrogen sulfide was stopped, the alcohol was distilled under vacuum, and the remaining reaction mixture was diluted with twice the volume of water. Next, the resulting mixture is mixed by adding diluted HCl drop by drop to a pH of 2-3, resulting in a white precipitate that is filtered, rinsed several times with water and then dried at room temperature. As a result, 39 g (98%) of 5-(2-aminophenyl)-1,3,4-oxadiazole-2-(3H)-thion (2a) was obtained, with a volume of 199-2000S, Rf 0.4 (chloroform: ethanol – 10:1). IR spectrum (v, cm–1): 3482 (NH2), 3154 (NH), 1624 (N-N), 1224 (C=S). 1H Nuclear Magnetic Resonance (NMR) spectrum (CDCl3, δ (ppm), J (Hz)): 6.65 (1H, ddd, J=8.0, 7.2, 1.0, H-5′), 6.76 (1H, ddd, J=8.3, 1.0, H-3′ ), 7.19 (1H, ddd, J=8.0, 7.2, 1.5, H-4′), 7.63 (1H, ddd, J=8.0, 1.5, H-6′ ).
Detection of four (4 ArH) single-proton signals in the 1H NMR spectrum in the form of doublet doublets and doublet doublet doublets, as well as the absence in the IR spectrum of the appearance of an absorption band of one exocyclic amino (NH2) and endocyclic imino (NH) groups at 3482 cm-1 and 3154 cm-1, respectively, as well as thiocarbonyl. The groups (C=S) at 1224 cm-1 unambiguously confirm the proposed structure in Figure 1.
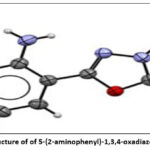 |
Figure 2: X-ray structure of of 5-(2-aminophenyl)-1,3,4-oxadiazole-2-(3H)-thione |
Pharmacological studies of 5-(2-aminophenyl)-1,3,4-oxadiazole-2-(3H)-thione
General effect and toxicity of compound-1 on white mice when administered orally.
When examining the general effects and toxicity of compound-1, it was found that mice’s behavior showed some activation of motor activity at oral doses of 1.0 to 10.0 mg/kg. Between dosages of 50 and 800 mg/kg, there were no discernible stable behavioral alterations. At doses between 1000 and 2000 mg/kg, mice displayed a general depression; nevertheless, if the mice were disturbed, they behaved appropriately. The mice exhibited more severe depression and a slow response to pain and sound irritation at dosages greater than 3000 mg/kg. Mice started to die at dosages higher than 3000 mg/kg, and the oral LD50 of compound-1 was 4558 mg/kg. In the context of respiratory depression, mice died 30 to 240 minutes after injection.
The effect of compound-1 on the motor activity (MA) of white mice with a single injection.
The suggested approach was used to conduct experiments on white mice, and the number of sex line crossovers by the mice in a minute was used to quantify motor activity, or MA.19 The doses used were from 0.1; 0.5; 1.0; 5.0; 10.0; 30.0 and 60.0 mg/kg orally. MA in all groups of mice on the background of compound-1 administration, it was significantly higher than in the group of control mice (see Table 1). Based on the experimental results, it can be concluded that compound-1 had an activating effect on MA, and a dose of 0.1 and 0.5 mg/kg enhanced MA to the greatest extent in mice. and the dose of 10 was slightly inferior in terms of increased activity to the first dose (Table 1).
Table 1: The effect of compound-1 on the motor activity of white mice with a single injection.
| Groups | Doses in mg/kg | Initial | 60 min. | 120 min. | 180 min. | 240 min. |
| Control group | 18,4±4,7 | 15,2±2,9 | 14,4±3,7 | 9.6±2,1 | 7,2±1,7 | |
| Compound-1 | 0.1 | 17,2±3,4 | 25,3±4,2* | 23,7±5,2* | 21,8±7,4* | 19,8±4,9* |
| 0.5 | 16,3±3,8 | 24,8±5,3* | 24,6±7,2* | 22.7±6,7* | 21,8±6,4* | |
| 1.0 | 15,7±6,2 | 23,8±6,8* | 21,7±5,9* | 19.8±6,4* | 17,8±5,3* | |
| 5.0 | 16,8±5,8 | 24,2±5,9* | 24,8±5,2* | 21.4±4,9* | 19,6±7,8* | |
| 10 | 16,3±3,8 | 23,6±3,6* | 21,2±3,9* | 19,7±2,4* | 13,4±2,3* | |
| 30 | 14,7±2,6 | 17,4±2,8* | 23,1±3,8* | 22,8±3,7* | 19,7±3,1* | |
| 60 | 19,6±3,1 | 22,6±4,3* | 21,9±3,4* | 17,6±3,2* | 15,4±2,9* |
Note: * – reliability compared to the data of the control group-P<0.05
The effect of compound 1 on the locomotor action of phenamine (amphetamine).15
As is well known, central alpha-adrenergic receptor activation is the primary cause of phenamine’s psychopharmacological effects. Thus, compound-1’s impact on phenamine-induced locomotor activity was investigated.
Compound-1 was given orally in the aforementioned dosages in mouse trials, and after an hour, phenamine 7 mg/kg was injected subcutaneously. Increased motor activity was seen in both control and experimental mice 1, 2, and 3 hours after phenamine was administered. Against the background of compound-1, there was a definite tendency to increased motor activity in all experiments when compared to the control group. In comparison to the control group, mice’s motor activity increased by 1.5–2 times two and three hours after phenamine was administered. In all studies, the dose of 30 mg was more active than 10 and 60 mg/kg. Based on the results obtained, it can be concluded that compound-1 significantly potentiates the locomotor action of phenamine (Figure 3).
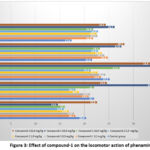 |
Figure 3: Effect of compound-1 on the locomotor action of phenamine. |
The effect of compound-1 on haloperidol catalepsy.16
As is known, D-receptors are involved in the manifestation of the antidepressant effect of drugs. It was decided to study the effect of compound-1 on haloperidol catalepsy, which is caused by the blockade of CNS D-receptors. In the experiments on mice, compound-1 was administered in the studied doses 1 hour before the administration of haloperidol 0.5 mg/kg subcutaneously. The study showed that haloperidol itself caused catalepsy lasting more than 120 seconds for 5 hours, while against the background of compound-1, the duration of catalepsy was less pronounced by 50-60%. It can be concluded that compound-1 in doses of 1 and 10 mg/kg partially blocks the action of haloperidol. It was also noted that doses significantly varying in magnitude have an antagonistic effect similar in severity to that of haloperidol. The results of this experiment are shown in Figure 4.
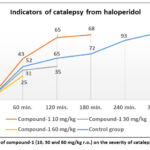 |
Figure 4: The effect of compound-1 (10, 30 and 60 mg/kg r.o.) on the severity of catalepsy from haloperidol. |
The effect of compound-1 on MA and research activities in the Hall “open field” test.17
Compound-1 MA was assessed with a single injection by counting the number of square line intersections, and research activity was measured by counting the number of looks into the mink’s lumen. Compound-1 enhanced its research activity from 1.5 to 2 times at all doses, with 1.0, 5.0, and 10 mg/kg being the most active. As can be seen, the MA indicators roughly matched the number of investigated minks (see Table 2).
Table 2: The effect of compound-1 on the motor and research activity of white mice in the “open field” test.
| Groups | Number of line intersections | Number of mink surveys |
| Control group (dis.water) | 14,3±3,2 | 13,8±2,9 |
| 0.1 mg/kg r.o. | 17,8±3,8* | 19,8±3,4* |
| 0.5 mg/kg r.o. | 20,8±4,2* | 22,8±4,2* |
| 1.0 mg/kg r.o. | 24,8±5,3* | 25,3±7,5* |
| 5.0 mg/kg r.o. | 28,9±9,2* | 27,8±6,8* |
| 10.0 mg/kg r.o. | 27,6 ±3,8* | 25,4±4,1* |
| 30.0 mg/kg r.o. | 24,8±2,9* | 23,4±3,1* |
| 60.0 mg/kg r.o. | 17,2±2,4* | 15,4±1,9* |
Note: * – reliability compared to the data of the control group-P<0.05
The effect of compound-1 on anxiety according to the Kilfoil method.18
In these experiments, when mice are placed in a 4-chamber maze with 2 dark and 2 light chambers, mice prefer to stay in dark chambers and, to a lesser extent, in light chambers with 2-5 camera shifts.19 With increased anxiety caused by anxiogens, in our case corazole 25 mg/kg subcutaneously, mice prefer dark cells even more and move less frequently from cell to cell. Against the background of substances with anxiolytic properties, mice prefer light chambers and change chambers more often. The duration of the experience is 1 min. Based on the ratio of time spent in light and dark cells, the “K Index” = Tlight/Tdark is calculated. (The quotient of dividing the time the mouse spent in the light compartment (Tlight) by the time it spent in the dark compartment (Tdark). As a rule, in control experiments (against the background of anxiety), the K index was usually less than 1.0 (0.2 – 0.7), and in a state of relaxation, i.e. in the absence of anxiety, it was more than 1, and more often 2-3. In the table 3 presents the data obtained, which indicate that all doses of compound-1 increase the “anti-anxiety index” K from 4 to 6 times compared with the control group.
Table 3: The effect of compound-1 on the feeling of anxiety with a single injection
| Groups | Time spent in light compartmentsin seconds | Time spent in dark compartmentsin seconds | Number of transitions from camera to cameraper second | The ratio of time spent in a light and dark cell |
| Control group(corazole 25 mg/kg per/cut) | 18,6±3,1 | 41,4±4,1 | 8,0±1,2 | 0,45±0,08 |
| 0.1 | 38,4±5,2* | 21,6±2,3* | 11,4±1,4* | 1,78±0,12* |
| 0.5 | 39,8±4,8* | 20,2±2,8* | 12,8±1,8* | 1,97±0,18* |
| 1.0 | 42,2±5,6* | 17,8±2,4* | 13,9±1,6* | 2,37±0,21* |
| 5.0 | 43,8±7,2* | 16,2±2,8* | 14,5±1,2* | 2,70±0,24* |
| 10.0 | 41,2±6,7* | 18,8±3,4* | 13,0±2,8* | 2,19±0,51* |
| 30.0 | 39,6±6,4* | 20,4±3,8* | 11,2±2,1* | 1,94±0,21* |
| 60.0 | 43.0±7,5* | 17,0±2,3* | 18,1±2,3* | 2,53±0,72* |
Note: * – reliability compared to the data of the control group-P<0.05
The effect of compound-1 on the resorptive effect of arecoline.
When studying the effect of psychotropic substances on neuroreceptors, experiments with the M-cholinostimulator arecoline are mandatory. For example, the instructions for use of fluoxetine do not have a direct effect on M-cholinergic receptors. The effect of compound-1 on M-cholinergic receptors has not been studied. This fragment of the work is devoted to a comparative study of the studied substances on the central tremor effect of arecoline.
The experiments were conducted on white male mice. Arecoline was administered at a dose of 10 mg/kg subcutaneously causing tremor (central effect) and salivation (peripheral effect). Attention was paid to the duration of the tremor. Against the background of compound 1, the duration of tremor was from 7.5 to 9.4 minutes, respectively, in the control groups 12.5 minutes. Salivation in the control group was 16.5 minutes. Against the background of compound 1, the duration of salivation was from 9 to 9.7 minutes, respectively. According to the data, compound-1 in all doses counteracted the function of arecoline, i.e. had an M-cholinolytic effect.
Study of antidepressant effect
Two traditional models of stress-induced depression—”The test of hanging mice by the tail”20 and “The test of non-escapable forced swimming along the Porsolt”—were used to assess the impact of the drug on the intensity of depressed behavior in animals.20The “Hanging mice by the tail” test (Table 4)
A 60 x 20 x 20 cm chamber with two sections was used for the experiment. The investigated compound-1 was administered into the animals 45 minutes before to the experiment (the time of peak pharmacological activity). A Band-Aid was used to suspend the mouse by its tail at a distance of 1.5 cm from its tip. Ten centimeters separated the animal’s nose from the ground. Two animals were concurrently recorded for the entire length of immobilization (motionless hovering) for six minutes. A clinical indication of depression is immobilization, which is a manifestation of behavioral despair. When animals in the experimental groups were immobilized for a shorter period of time than those in the control group, it was thought that the drug had antidepressant qualities.
Table 4: The effect of compound-1 on the “Hanging mice by the tail” test with a single injection.
| Groups | Doses in mg/kg | Mobility phase in sec. | Phase of immobilization in sec. |
| Control group | – | 135,8±15,36 | 224,2±15,36 |
| Compound-1 | 0,1 | 246,6±18,72*,a | 113,4±18,72*,a |
| 0,5 | 288,2±15,12*,a | 71,8±15,12*,a | |
| 1,0 | 299,6±7,68*,a | 60,4±7,68*,a | |
| 5,0 | 288,4±6,24*,a | 71,6±6,24*,a | |
| 10,0 | 266,1±12.15*,a | 93,9±12,1*,a | |
| 30,0 | 259,8±13,18*,a | 100,2±14,3*,a | |
| 60,0 | 231,3±14,12*,a | 128,7±22,4*,a | |
| Fluoxetine | 10,0 | 177,9±9,78* | 182,1±14,8* |
| 20,0 | 185,7±8.98* | 174,3±13,8* | |
| 30,0 | 216.9±10.15* | 143,1±9,8* | |
| Amitriptyline | 0,1 | 204,5±8.78* | 115,5±16,7* |
| 1,0 | 227,4±12,24* | 132,6±18,6* | |
| 10,0 | 249.9±13,12* | 110,1±12,8* |
Note: * – reliability compared to the data of the control group-P<0.05
a reliability compared to the data to the fluoxetine group (P>0.05)
The “Forced non-runaway swimming” test (Table 5)
The experiment was carried out in a glass pool that measured 22 cm in diameter and 50 cm in height. It was filled with water that was between 21 and 24 °C so that the animal’s rear legs could not touch the bottom. The technique was utilized to investigate compound-1’s antidepressant effects. ([Porsolt R. D.Bertin A. Yalfre M. Arch. Int. Pharmacodyn. Ther. 1977, v. 229, pp. 327-333] as modified by [Schmidt J.Biomed. Biochim. Acta. 1985, v. 44, p. 755-761] according to which the main indicator of the state of “behavioral despair” in mice is the time of immobilization (refusal to search for an exit from a vessel filled with water). The reduction in the duration of immobilization was regarded as a manifestation of the antidepressant activity of the substance. Compound-1 significantly reduced the duration of animal immobilization, which can be considered an increase in the activity of mice in finding a way out of an aversive situation. The dose-dependent nature of the antidepressant effect of compound-1 should be noted. When comparing the antidepressant activity of compound-1, the antidepressant fluoxetine, which is currently widely used in clinical practice, was chosen as the comparison drug. According to the “behavioral despair” test, which is most often used to assess antidepressant activity, fluoxetine has a pronounced antidepressant effect at doses of 10, 20 and 30 mg / kg, which is 1/24.8, 1/12.4 and 1/8.2 of LD50. Compound-1 exhibits antidepressant activity 3-4 times higher than that of fluoxetine, which is only 1/456-1/76 of LD50, i.e. at doses 18.4 times less toxic than that of the reference drug.
Table 5: Antidepressant activity of compound-1 in comparison with fluoxetine and amitriptyline in the forced swimming test
| Groups | Doses in mg/kg | Mobility phase in sec. | Phase of immobilization in sec. |
| Control group | – | 128,2±15,12 | 231,8±15,12 |
| Compound-1 | 0.1 | 306,4±9,12 | 51,6±11,52*,a |
| 0.5 | 332,0±6,0 | 28,0±7,2*,a | |
| 1.0 | 313,6±8,64 | 46,4±7,2*,a | |
| 5.0 | 331,2±6,96 | 28,8±6,96*,a | |
| 10,0 | 261,6±8,72 | 98,4±7,1*,a | |
| 30,0 | 244,2±7,94 | 115,8±18,3*,a | |
| 60,0 | 221,3±12,18 | 138,7±17,8*,a | |
| Fluoxetine | 10,0 | 191,3±11,71 | 168,7±21,8* |
| 20,0 | 165,2±9,82 | 194,8±23,4* | |
| 30,0 | 176,9±10,31 | 183,1±19,2* | |
| Amitriptyline | 0,1 | 194,5±9,82 | 165,5±16,2* |
| 1,0 | 167,4±8,94 | 192,6±23,6* | |
| 10,0 | 161,9±9,21 | 198,1±17,8* |
Note: * – reliability compared to the data of the control group-P<0.05
a reliability compared to the data to the fluoxetine group (P>0.05).
Discussion
Antidepressants are approved by the FDA to treat a variety of medical conditions, even though they may be the preferred medication for depression. Antidepressants, for instance, are used to treat panic disorder, generalized anxiety disorder (GAD), obsessive-compulsive disorder, social anxiety disorder, and post-traumatic stress disorder (PTSD). Additionally, antidepressants have off-label, non-FDA-approved uses. As it relates to the crucial information required by members of an interprofessional team overseeing the care of patients receiving antidepressant medications for conditions that respond to this medication class, this activity examines the indications, contraindications, action, adverse events, and other important aspects of antidepressant therapy in the clinical setting.23,24,25
Experiments on stress-induced depression models revealed that compound-1 has a strong antidepressant effect when given once to mice (male and female). In the “non-escapable forced Porsolt swimming” and “hanging mice by the tail” tests, compound-1 caused a statistically significant increase in the latency interval prior to the onset of immobility and a decrease in the animals’ overall immobilization time (Tables 5 and 6). In the “non-escapable forced swimming, but Porsolt” test, the test material considerably enhanced the animals’ overall amount of active swimming time as well as the number of jumps they made.. The antidepressant properties of compound-1 exceed the similar effect of fluoxetine. Thus, neuro- and psychopharmacological studies of compound-1 in comparison with the well-known antidepressant fluoxetine have revealed the originality and advantages of compound-1 as a potential antidepressant drug. Its originality lies in the fact that:
Firstly, the determination of the acute daily toxicity (LD50) of the studied substances in white mongrel mice, when administered orally, is 18.4 times less toxic than fluoxetine and exceeds its antidepressant activity by 3-7 times at doses of 10-30 mg /kg. Secondly, compound-1 in the doses used significantly potentiates, while fluoxetine does not have a direct effect on alpha-adrenergic receptors, the activating effect of phenamine on the motor activity of white mice. Therefore, the clinical use of compound-1 should be accompanied by less pronounced motor disorders known from fluoxetine.8 Thirdly, compound-1 potentiates the central cholinopositive properties of arecoline, while the reference drug fluoxetine is not known to have a direct effect on M-cholinergic receptors.8 This fact suggests an improvement in mental abilities when using compound-1.
These facts suggest that the more pronounced psychopharmacological properties of compound-1 in comparison with fluoxetine in clinical conditions will show advantages over fluoxetine. Moreover, the 3 differences listed above will suggest a special original property of compound-1, which in the long term is expressed in improving the cognitive functions of patients found in elderly patients whose treatment includes the use of M-cholinergic drugs – galantamine, arecoline and others.21, 22 The use of compound-1 should be accompanied to a lesser extent by marked or absent motor disorders that occur when taking antidepressants, including fluoxetine.
Conclusion
Thus, compound-1 was 3-7 times superior in antidepressant effect in experiments. The fundamental difference between compound-1 and fluoxetine lies in the M-choline and alpha-adrenopotentiating effects of the former, while fluoxetine does not have a direct effect on these receptors. Based on these data, it can be assumed that compound-1 will not disrupt the cognitive properties of the patient, and will also cause dyskinesia to a lesser extent or not at all, which can only be verified in clinical trials.
This, in turn, requires extensive research to find promising antidepressant drugs, in particular antipsychotics from among the derivatives of 5-(2-aminophenyl)-1,3,4-oxadiazole-2 -(3H) – Thione.
Acknowledgment
The authors’ acknowledgements go to the Institute of Chemistry of Plant Substances named after Academician S.Y. Yunusov of the Academy of Sciences of the Republic of Uzbekistan for supporting this work.
Funding source
The work financial supported by the Ministry of Higher Education, Science and Innovation of Republic of Uzbekistan (Grant № F-FA-2021-408 “Study of the laws of introduction of pharmacophore fragments into the molecule on the basis of modern cross-coupling and heterocyclization reactions” and Grant № ALM-2023031533-02 “Creation of anticonvulsant medicinal substance from a number of heterocyclic compounds synthesized on the basis of domestic raw materials”).
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
All research work in this article was carried out on the basis of the permission of the Ethics Committee under the Ministry of Health of the Republic of Uzbekistan No. 1/1-1628.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not applicable
Author Contributions
- Z.I.Sanoev: Conceptualization, Methodology, Writing – Original Draft. Pharmacological and toxicological experiments, statictical Analysis
- S.Z.Rashidov: Data Collection, Analysis, Writing – Review & Editing.
- T.Khamroev – Conceptualization, Methodology, Writing – Original Draft. Pharmacological and toxicological experiments, statictical Analysis
- I.T.Abdinazarov: Funding Acquisition, Resources, Supervision.
- S.D.Rakhimboev: Conceptualization, Methodology, Funding Acquisition.
- D.S.Ismailova: Conceptualization, Methodology, Funding Acquisition.
- B.Zh.Elmuradov: Data Collection, Analysis, Writing – Review & Editing.
- M.A.Shakhmurova: Data Collection, Analysis, Writing – Review & Editing.
References
- Garibova T.L., Kraineva V.A., Voronina T.A. Povedencheskie eksperimentalnыe modeli depressii. Farmakokinetika i farmAkodinamika. 2017, 3, 14-19.
- Parfenov VA. Primenenie antidepressantov v nevrologicheskoy praktike. Nevrologiya, neyropsixiatriya, psixosomatika. 2023;15(2):4–9.
CrossRef - Mudra Rakshasa-Loots A., Swiffen D., Steyn C., Marwick KFM., Smith DJ. Affective disorders and chronic inflammatory conditions: analysis of 1.5 million participants in Our Future Health. BMJ Ment Health. 2025; 10;28(1)1-7:e301706.
CrossRef - Nikitina A.V., Semina I.I., Nikitin D.O., Gavrilova E.L., Krutov I.A., Isaeva A.O. Experimental investigation of the behavioral effects of new phosphorylated thiosemicarbazides. Pharmacokinetics and Pharmacodynamics. 2023;(3):68-76.
CrossRef - Cuijpers P., Noma H., Karyotaki E., Vinkers C.H., Cipriani and Furukawa T.A. A network meta-analysis of the effects of psychotherapies, pharmacotherapies and their combination in the treatment of adult depression. World Psychiatry, 2020; 19: 92-107.
CrossRef - Miftakhova A.F., Nikitina I.L., Gabidullin R.A. Study of anti-depressive action mechanism of a new derivative of 1- (thietanyl-3) imidazoles in tests of neuropharmacological interaction. Bashkortostan Medical Journal. 2021;16(1):52-57.
- Hassanein E.H.M., Althagafy H.S., Baraka M.A. et al. Pharmacological update of mirtazapine: a narrative literature review. Naunyn-Schmiedeberg’s Arch Pharmacol 2024; 397, 2603–2619.
CrossRef - Xarkevich D.A. Farmakologiya. Uchebnik. Fluoksetin. 12-e izd. – Geotar-media. 2017. – 760 s.: il. ISBN 978-5-9704-3884-8.
- Yurttaş L.. Evren, A.E., Kubilay A., Aksoy M.O., Temel, H.E., Akalın Ç.G. Synthesis of Some New 1,3,4-Oxadiazole Derivatives and Evaluation of Their Anticancer Activity. American Chemical Society. 2023; 8(51); 49311-26.
CrossRef - Oliveira C.S., Lira B.F., Barbosa-Filho J.M., Lorenzo J.G.F., Athayde-Filho P.F. Synthetic approaches and pharmacological activity of 1,3,4-oxadiazoles: a review of the literature from 2000–2012 // Molecules. 2012. Vol. 17. № 9. P. 10192-10231.
CrossRef - Khamkar T., Kadam R., Mali S., Udugade B., Singh S. Recent Advances in Synthetic Approaches for 1,3,4-Oxadiazole Derivatives: A Comprehensive Review on Therapeutic Applications. Open Med Chem J, 2025; 19: e18741045372896.
CrossRef - Khalilullah H., Ahsan M.J., Hedaitullah Md., Khan S. 1,3,4-Oxadiazole: A Biologically Active Scaffold // Mini-Reviews in Medicinal Chemistry. -2012. 12. 789-801.
CrossRef - Sharma R., Kumar N., Yaday R. Chemistry and pharmacological importance of 1,3,4-oxadiazole derivatives // Research & Reviews: Journal of Chemistry -2015. 4.2.1-27.
- National Academies of Sciences, Engineering, and Medicine. 2011. Guide for the Care and Use of Laboratory Animals: Eighth Edition. Washington, DC: The National Academies Press. 2011; (8): 316.
- Rothman R., Baumann M., Dersch C. et al. Amphetamine-type central nervous system stimulants release norepinephrine more potently than they release dopamine and serotonin. // 2001;39(1):32-41.
CrossRef - Waku I., Reimer AE. and de Oliveira AR. Effects of Immediate Aversive Stimulation on Haloperidol-Induced Catalepsy in Rats. Behav. Neurosci. 2022;16:867180.
CrossRef - Hall C.S. Emotional behavior in the rat. III. The relationship between emotionality and ambulatory activity. Journal of Comparative Psychology, 1936; 22, 345-352.
CrossRef - Kilfoil T., Michel A Montgomery D., Whiting R. / Effect of anxyolyic and anxyogenic drugs on exploratory activity a simple model of anxiety in mice. // Psychopharmacology, 1989,28, (9), 901-905.
CrossRef - Sanoev Z. I., Ismailova D. S., Rakhimboev S. D., et al. Synthesis and Research Anticonvulsant Activity of Annulated Triazolo-Thiadiazine Derivative in Laboratory Animals. Biomed Pharmacol J 2023;16(4). 2457-2467.
CrossRef - Emily R.T., Rachel S., Julia B. et al. The need for guidance in antidepressant drug development: Revisiting the role of the forced swim test and tail suspension test. Regulatory Toxicology and Pharmacology 2024; 151. 105666-74.
CrossRef - Erkinjuntti T., Kurz A., Gauthier S. et al. Efficacy of galantamine in probable vascular dementia and Alzheimer’s disease combined with cerebrovascular disease: a randomised trial. 2002; 13;359:1283-90.
CrossRef - Taheri M., Afzali Mehr M. & Ghafouri H. The novel orthosteric agonist M1 muscarinic acetylcholine receptor reveals anti-Alzheimer’s disease activity. Sci Rep 2024; 14, 28824.
CrossRef - Gutiérrez-Rojas L, Porras-Segovia A, Dunne H, Andrade-González N, Cervilla JA. Prevalence and correlates of major depressive disorder: a systematic review. Braz J Psychiatry. 2020;42(6):657-672.
CrossRef - Cipriani A, Furukawa TA, Salanti G, et al. Comparative efficacy and acceptability of 12 new-generation antidepressants: a multiple-treatments meta-analysis. Lancet. 2009;373(9665):746-58.
CrossRef - Declercq PL, Eraldi JP, Beuzelin M, et al. Severe serotonin syndrome caused by an interaction between an antidepressant and a cough syrup. 2021;76(3):249-252.
CrossRef
Abbreviations list
FST – forced swim test
TST – tail suspension test
MAO – monoamine oxidase
MA – motor activity
Tlight – spent in the light compartment
Tdark – spent in the dark compartment
NMR – Nuclear Magnetic Resonance







