Manuscript accepted on :18-02-2026
Published online on: 18-03-2026
Plagiarism Check: Yes
Reviewed by: Dr. Inamullah Khan
Second Review by: Dr. Gowri Sankara Rao Burle
Final Approval by: Dr. Prabhishek Singh
Luh Putu Ratna Sundari1* , I Dewa Ayu Inten Dwi Primayanti1
, I Dewa Ayu Inten Dwi Primayanti1 , I Ketut Tunas2
, I Ketut Tunas2 and Made Alyashanti Radya Bulandari3
and Made Alyashanti Radya Bulandari3
1Department of Physiology, Faculty of Medicine, Udayana University, Bali, Indonesia.
2Department of Health Information Management, Faculty of Health Sciences, Bali International University, Bali, Indonesia.
3Medicine Study Program, Faculty of Medicine, Udayana University, Bali, Indonesia.
Corresponding Author E-mail: luhputu_ratnafk@unud.ac.id
DOI : https://dx.doi.org/10.13005/bpj/3365
Abstract
Vitamin D and insulin-like growth factor-1 (IGF-1) are important regulators of blood pressure, particularly in relation to aging. This study aims to examine whether reduced vitamin D and IGF-1 levels serve as risk factors for hypertension in older adults. Case–control study was used to investigate the differences between vitamin D and IGF-1 concentrations in plasma as risk factors for hypertension in the aging population. A total of 45 elderly people participated in the study and were divided into two groups: 23 hypertensive subjects as the case group and 22 normotensive subjects as the control. Both groups underwent measurements of vitamin D and IGF-1 levels. The association between vitamin D and IGF-1 levels and hypertension was evaluated using chi-square tests, followed by logistic regression analysis to determine the odds ratio. Statistical analysis revealed a significant association between low vitamin D levels and hypertension (p < 0.038), with an odds ratio (OR) of 4.000; 95% confident interval (CI) 1.155 – 13.855. For IGF-1 levels, the p-value was 0.016 (p<0.05), with an OR of 5.289; 95% CI 1.438 – 19.447. A significant association between vitamin D deficiency and IGF-1 was observed (p<0.05). In multivariate analysis adjusting for age, sex, and body mass index, low vitamin D (aOR=16.756; 95% CI: 2.05–136.93; p=0.009 and low IGF-1 (aOR=23.182; 95% CI 2.483–216.472; p=0.006) remained independent risk factors. In conclusion, the findings of this study indicate an association between reduced vitamin D and IGF-1 levels and an increased risk of hypertension in older adults.
Keywords
Case-control study; Hypertension; Insulin-like growth factor-1 (IGF-1); Older adults; Vitamin D
Download this article as:| Copy the following to cite this article: Sundari L. P. R, Primayanti I. D. A. I. D, Tunas I. K, Bulandari M. A. R. Low Serum Vitamin D And Insulin-Like Growth Factor-1 Level as Risk Factors for Hypertension Among Older Adults. Biomed Pharmacol J 2026;19(1). |
| Copy the following to cite this URL: Sundari L. P. R, Primayanti I. D. A. I. D, Tunas I. K, Bulandari M. A. R. Low Serum Vitamin D And Insulin-Like Growth Factor-1 Level as Risk Factors for Hypertension Among Older Adults. Biomed Pharmacol J 2026;19(1). Available from: https://bit.ly/47U3I71 |
Introduction
Hypertension is one of the major health problems among older adults and contributes significantly to morbidity and mortality associated with cardiovascular diseases. As age advances, the risk of developing hypertension increases due to progressive cardiovascular aging, stiffening of blood vessels, and altered hormonal balance.1
Vitamin D contributes not only to skeletal health but also to the maintenance of cardiovascular homeostasis. Inadequate vitamin D status has been associated with activation of the renin–angiotensin–aldosterone system (RAAS), increased insulin resistance, and impaired endothelial function, mechanisms that may promote elevations in blood pressure.2 In addition, insulin-like growth factor-1 (IGF-1), an anabolic hormone that naturally declines with age, plays a role in blood pressure regulation by influencing endothelial integrity, lipid metabolism, and insulin sensitivity.3 Evidence from multiple studies indicates that reduced IGF-1 levels are linked to a higher risk of hypertension and atherosclerotic disease among older adults.4
As people live longer, the proportion of older adults in the global population continues to rise. The aging process is characterized by a decline in endocrine and metabolic functions, which affects multiple aspects of health. Two biomarkers that consistently decrease with advancing age are 25-hydroxyvitamin D [25(OH)D] and insulin-like growth factor-1 (IGF-1).5 Vitamin D plays roles in calcium metabolism, bone health, muscle function, lipid metabolism, inflammation and immune regulation. 6 Meanwhile, IGF-1 is the main biological mediator of growth hormone (GH) activity, contributing to tissue growth, muscle anabolism, and cellular regeneration. Reduced physiological adaptability, functional impairment, and a higher risk of chronic diseases have all been linked to declines in these two biomarkers in older persons. 7,8 Aging is accompanied by reduced levels of vitamin D and insulin-like growth factor-1 (IGF-1). In older adults, insufficient vitamin D and reduced IGF-1 levels have been associated with musculoskeletal impairment, decreased muscle strength, and higher rates of morbidity.9 Preventive efforts, such as promoting physical activity, ensuring adequate nutrition, and correcting vitamin D deficiency are essential components of strategies to maintain quality of life in the older adult population.3
Two age-sensitive biomarkers, namely 25-hydroxyvitamin D and insulin-like growth factor-1, show a progressive decline in later life and may contribute to biological mechanisms underlying hypertension in older adults. This study will examine the level of vitamin D and IGF-1 in the blood of elderly subjects to evaluating the role of decreases in these two biomarkers in the development of hypertension among older adult populations. Experimental evidence indicates that vitamin D contributes to blood pressure regulation by inhibiting the renin–angiotensin–aldosterone system (RAAS) and preserving endothelial integrity. 10 In contrast, insulin-like growth factor-1 (IGF-1) supports vascular health by increasing nitric oxide bioavailability and enhancing endothelial function.11 Observational studies generally report associations between low vitamin D levels and elevated blood pressure; however, evidence from intervention trials on vitamin D supplementation for blood pressure management remains inconclusive.12 The relationship between IGF-1 levels and blood pressure also tends to be inverse higher IGF-1 levels are associated with lower blood pressure in several cohorts although non-linear patterns and population heterogeneity complicate causal interpretation.13 Current evidence supports the identification and correction of vitamin D deficiency in older adults at risk, as well as the strengthening of nutritional and exercise interventions to support IGF-1 status. However, evidence that these interventions directly reduce the incidence of hypertension in older adults remains inconclusive. 14
However, few studies have investigated the combined influence of low vitamin D and IGF-1 levels on hypertension in older adults, with data from Indonesia being especially limited. Several studies have examined the relationship between n vitamin D and hypertension in the elderly in Indonesia, but the results have been inconsistent. For example, a study by Hermawan and Andoko15 showed a significant association between vitamin D levels and hypertension, while a study by Ferawaty et al. 2024, showed an insignificant association, indicating that vitamin D supplementation had no association with hypertension in the elderly.16 To date, no studies have examined the relationship between vitamin D and IGF-1 levels and hypertension in the elderly in Indonesia. Therefore, this study is important to clarify this relationship so it may serve as a foundation for preventive measures and management strategies for hypertension based on hormonal and nutritional status.
Current studies suggest that vitamin D and IGF-1 may interact within shared biological pathways. Vitamin D may enhance IGF-1 receptor expression and modulate it’s signaling pathways, while IGF-1 appears to support the vasodilatory effects of vitamin D through increased nitric oxide (NO) bioavailability.17 The novelty of this study lies in examining whether the combined deficiency of these two factors may accelerate endothelial dysfunction and increase vascular resistance, positioning them as important biomarker candidates in the pathogenesis of hypertension among older adults. This research is conducted to substantiate the still-limited evidence on their direct causal relationship. If proven, these findings would place vitamin D and IGF-1 as biologically significant, interacting factors involved in regulating blood pressure through endothelial and RAAS-related mechanisms. Ultimately, a multidisciplinary approach incorporating targeted nutritional interventions, physical activity, and supplementation may serve as an effective measure for reducing the risk and improving the management of hypertension in older populations.
Method
This study employed a case–control design to examine differences in plasma vitamin D and IGF-1 levels as risk factors for hypertension among older adults. The sample consisted of older adults residing in Selat Village, Abiansemal District, Badung, Bali, Indonesia, who had signed informed consent forms. Eligible participants were enrolled consecutively based on predetermined inclusion and exclusion criteria for both study groups. The study received ethical approval from the Ethics Commission of the Faculty of Medicine at Udayana University, Denpasar-Bali, Indonesia No. 2808/UN.14.2.2.VII.14/LT/2025 with protocol number 2025.02.1.1250
Case and Control Criteria
Case group: Older adults residing in Abiansemal village who were diagnosed with hypertension.
Control group: Older adults residing in Abiansemal village who did not have hypertension.
Inclusion and Exclusion Criteria
Case inclusion: Older adults aged 60 years and above, classified as having hypertension based on the JNC VIII guidelines 18 (> 140/90) and willing to participate in the study
Control inclusion: Older adults aged 60 years and above, not diagnosed with hypertension (normotensive), and willing to participate in the study
Exclusion: Individuals with a history of severe chronic illnesses, kidney disorders, liver diseases, or cancer, individuals undergoing treatments that may influence vitamin D or IGF-1 levels (supplementation, hormonal therapy).
Criteria Level of Vitamin D and IGF-1
Level of Vitamin D: Low <30 ng/ml, Normal: 30-50 ng/ml, High>50ng/ml.19
Level of IGF-1: Low <80 ng/ml, Normal: >80 ng/ml.20
The sample size was calculated using the formula described by Kasiulevius. 21
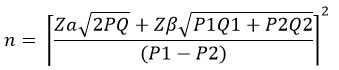
Materials and Methods
The measurement of vitamin D and IGF-1 concentration was performed using blood plasma samples analyzed with the ELISA method, employing the 25-Hydroxyvitamin D and IGF-1 assay kits from Diagnostic Biochem Canada Inc. Blood pressure was measured using an Omron digital sphygmomanometer. Venous blood (3 mL) was drawn from the cubital vein by qualified paramedical personnel using a sterile 3 mL spuit and a 21G Abbott catheter. Following collection, the samples were centrifuged for five to fifteen minutes at 3000 rpm to separate the serum, which was then analyzed for vitamin D and insulin-like growth factor-1 concentrations.
The collected data were expressed as quantitative values and examined through cross-tabulation and chi-square analysis to evaluate the relationships between vitamin D status, IGF-1 concentrations, and hypertension. A p-value of less than 0.05 was considered statistically significant, and odds ratios were reported with 95% confidence intervals.
Results
The subject characteristic is presented in Table 1.
Table 1: Participant Characteristics and Clinical Parameters
| Variable | Hypertension(n =23) | Non-Hypertension (n =22) | p-value |
| Age (yrs) + SD | 66.30 + 5,24 | 65.18 + 6.34 | 0.520 |
| BMI (kg/m2) mean + SD | 25.26 + 2.85 | 24.82 + 3.62 | 0.656 |
| Obesity (BMI>25kg/m2), n% | 13 (28.9%) | 10 (22.2%) | 0.458 |
| Systolic BP (mmHg), mean + SD | 158.13+13.51 | 120.5+11.37 | <0.001 * |
| Diastolic BP (mmHg), mean + SD | 88.6 + 10.53 | 76.4+ 8.01 | 0.007 * |
| 25(OH)D3 ng/ml, (mean + SD) | 28.2 | 34.6 | 0.052 |
| Vit D Insuf (<30 ng/ml), n% | 16 (35.6%) | 8 (17.8%) | 0.026 * |
| IGF-1 (ng/ml) (mean + SD) | 83.12 + 10.53 | 85.45+ 9.57 | 0.392 |
| Low IGF-1 (<80 ng/ml), n% | 14 (31.1%) | (11.1%) | 0.010 * |
*p significance (p<0.05)
This study involved 45 elderly participants (≥60 years) consisting of 23 subjects in the case group (hypertension) and 22 subjects in the control group (non-hypertension). Subjects were recruited through consecutive sampling at the elderly posyandu in Selat Village, Abiansemal IV Community Health Center. Both groups had a comparable number distribution to increase analytical power in calculating the odds ratio. The average age of all respondents was 66.26 years. The hypertension group had a slightly higher average age than the controls (66.30 ± 5.24 vs 65.18 ± 6.34 years), but the difference was not statistically significant, p = 0.520 (p>0.05). The average Body Mass Index (BMI) in the hypertensive group was higher than in the control group (25.26 ± 2.85 kg/m² vs. 24.83 ± 3.62 kg/m²; but not statistically significantly different, p=0.656), but the proportion of obesity was higher in the case group (28.9% vs. 22.2%).
The mean of blood pressure was clearly different, with values: 158.13/88.6 mmHg and 120.5/76.4 mmHg in the non-hypertensive group (p<0.05). Meanwhile, the mean of vitamin D and IGF-1 levels did not differ significantly between the two groups, with p-values of 0.052 and 0.392, respectively. However, the proportion of vitamin D insufficiency (<30 ng/ml) in the hypertensive group (35.6% and 17.8% in the non-hypertensive group) was significantly different (p<0.05). These findings indicate that vitamin D deficiency is more common in the case group than in the control group. The proportion of low IGF-1 (80 ng/ml) between the case and control groups, at 31.1% and 11.1%, was also significantly different. Descriptively, the hypertension group showed a higher proportion of obesity and significantly lower vitamin D and IGF-1 levels.
These preliminary findings suggest an association between low vitamin D and IGF-1 levels and hypertension in the elderly. However, further analysis using bivariate (chi-square) tests and logistic regression is needed to determine the magnitude of the risk (odds ratio) and to confirm whether these two biomarkers are independent risk factors after controlling for confounding variables.
To assess the occurrence of hypertension in individuals with low vitamin D levels, a chi-square analysis using a 2 × 2 contingency table was conducted. The findings are shown in Table 2 below.
Table 2: Vitamin D Deficiency and Hypertension Risk
| Variable | Group | OR | CI 95% | p | ||
| Case | Control | |||||
| Level of Vit D | Low | 16 | 8 | 4.000 | 1.155 – 13.855 | 0.038 |
| Normal | 7 | 14 | ||||
(p significance <0.05)
As presented in Table 2, a statistically significant association was observed between low vitamin D levels and hypertension among older adults. Participants with reduced vitamin D levels exhibited a four-fold increased risk of hypertension compared with the control group with normal or elevated vitamin D levels.
To examine the relationship between IGF-1 levels and hypertension, a chi-square analysis based on a 2 × 2 contingency table was performed, with the results summarized in Table 3.
Table 3: IGF-1 Deficiency and Hypertension Risk
| Variable | Group | OR | CI 95% | p | ||
| Case | Control | |||||
| Level of IGF-1 | Low | 14 | 5 | 5.289 | 1.438 – 19.447 | 0.016 |
| Normal | 9 | 17 | ||||
(p significance <0.05)
Table 3 demonstrates that low IGF-1 levels were also significantly associated with hypertension, with older adults showing reduced IGF-1 levels having a 5.289-fold higher risk of developing hypertension compared with controls with normal or higher IGF-1 levels.
Table 4: Association of Vitamin D and IGF-1 Level in Participant
| Variable | IGF-1 | OR | CI 95% | p | ||
| Low | Normal | |||||
| Level of Vit D | Low | 14 | 10 | 4.480 | 1.232 – 16.293 | 0.034 |
| Normal | 5 | 16 | ||||
(p significance <0.05)
In this study, association between vitamin D levels and IGF-1 levels was carried out using Chi Square analysis, obtaining a p value = 0.034 (p<0.05), which means there is a significant association between vitamin D and IGF-1 levels, where participants with low vitamin D levels have a four-fold risk of having low IGF-1 levels compared to participants with normal vitamin D levels (Table 4).
Table 5: Multivariate Logistic Regression Analysis for Hypertension in Elderly
| Variables | BCoefficient | Adjusted OR | 95% CI | p-value |
| Vit D insufficiency | 2.819 | 16.756 | 2.050 – 136.931 | 0.009 |
| Low IGF-1 | 3.143 | 23.182 | 2.483 – 216.472 | 0.006 |
| Age | -.067 | 0.936 | 0.806 – 1.086 | 0.381 |
| Sex | 1.919 | 6.817 | 1.046 – 44.423 | 0.045 |
| Body Mass Index(BMI) | 0.010 | 1.010 | 0.785 – 1.300 | 0.937 |
p- significance (p<0.05)
Multivariate logistic regression analysis demonstrated that vitamin D insufficiency remained independently associated with hypertension after adjustment for age, sex, and BMI (aOR=16.756; 95% CI 2.05–136.93; p=0.009). Similarly, low IGF-1 levels were independently associated with increased odds of hypertension (aOR=23.182; 95% CI 2.483–216.472; p=0.006). This analysis demonstrates that vitamin D insufficiency and low IGF-1 levels are independent risk factors, with their effects not entirely influenced by age and BMI. This supports the hypothesis that RAAS regulation disorders and endothelial dysfunction play a role in the pathogenesis of hypertension in the elderly.
Discussion
Vitamin D Deficiency and Hypertension Risk
Table 2 shows that low vitamin D levels are a risk factor for hypertension in the elderly, with an odds ratio (OR) of 4.000, p=0.038 (p<0.05) in this study. These results are supported by previous research showing that vitamin D levels are inversely related to blood pressure and the incidence of hypertension.22 Studies in both animals and humans have found that the development of hypertension in individuals with low vitamin D (1,25(OH)2D3) levels increases RAAS activity both systemically and in the kidneys. 23 A systematic review and meta-analysis concluded that serum vitamin D concentrations are inversely associated with the risk of hypertension and prehypertension in adults, both in cross-sectional and cohort studies.14 The association between vitamin D levels and blood pressure can be explained by several pathophysiological factors, as follows:
Dysregulation RAAS (Renin–Angiotensin–Aldosterone System).
Vitamin D has been proposed as a negative endocrine regulator of renin. Animal studies have shown that VDR deficiency increases renin and angiotensin II expression, contributing to increased blood pressure through RAAS activation, independent of normal calcium metabolism.24 Epidemiological studies also report a negative correlation between 25(OH)D levels and renin and blood pressure, supporting the association of vitamin D deficiency with a more active RAAS.25
Endothelial dysfunction and decreased NO (Nitric Oxide)
Vitamin D influences the regulation of endothelial nitric oxide synthase (eNOS), which is essential for the production of Nitric Oxide (NO), a key vasodilator mediator. In animal models without VDR, eNOS expression is reduced, leading to endothelial dysfunction and increased arterial stiffness. NO deficiency plays a role in increased peripheral vascular resistance and hypertension, which are more common in the elderly.26
Chronic inflammation and oxidative stress
Systemic inflammation levels increase in older adults (inflammaging), and vitamin D deficiency is associated with increased pro-inflammatory cytokines such as TNF-α and vascular oxidative stress, which impairs endothelial function and increases vasoconstriction. This contributes to vascular remodeling and increased blood pressure with age. 27
Several observational studies in adult and elderly populations have reported an inverse association between serum 25(OH)D levels and hypertension, with a higher risk of hypertension in individuals with low vitamin D levels. A large meta-analysis showed that higher vitamin D levels were associated with a reduced risk of hypertension. 28 However, it is important to note that clinical trials of vitamin D supplementation in older adults have not consistently shown reductions in blood pressure, indicating the complexity of the relationship between vitamin D status and blood pressure.
Vitamin D through its active form 1.25(OH)₂D and its receptor (VDR) suppresses renin expression in the kidneys, thereby downregulating RAAS activity. Vitamin D deficiency removes this inhibitory effect, potentially increasing vasoconstriction and sodium/volume retention.29 In addition, vitamin D influences endothelial function (via nitric oxide synthesis) and modulates chronic inflammation, both of which contribute to vascular dysfunction.30 Low levels of vitamin D are also associated with insulin resistance, where insulin resistance is involved in the pathogenesis of hypertension31, coupled with other evidence in a study that vitamin D supplementation can improve insulin sensitivity32, which will indirectly affect blood pressure.
IGF-1 Deficiency and Hypertension Risk
Table 3 shows low IGF-1 levels as a risk factor for hypertension in the elderly with an OR value of 5.289 with p = 0.016 (p <0.05) in this study. This is supported by several previous studies that found a relationship between low IGF-1 levels and the incidence of hypertension in a group of women with rheumatoid arthritis.33 The results of other studies also found that higher IGF-1 levels reduce the risk of hypertension in women without diabetes 34, and a systematic review and meta-analysis found that IGF-1 levels have a negative correlation with systolic and diastolic blood pressure.13 The mechanism explaining the relationship between IGF-1 levels and hypertension is as follows: there is strong evidence that IGF-1 is related to the regulation of endothelial function, where vascular endothelial and smooth muscle cells express IGF-1 receptors that play a role in angioprotective function.35 Local production of IGF-1 also supports the proliferation and migration of endothelial progenitor cells that are important for blood vessel repair and control of inflammation-induced oxidative stress.36 Insulin like Growth Factor-1 in circulation can induce vasodilation that contributes to the regulation of blood pressure and blood vessel tension. Low IGF-1 levels are associated with changes in blood vessels in aging and obesity, thus can increase cases of cardiovascular morbidity.37
The decline in insulin-like growth factor-1 (IGF-1) levels is a physiological phenomenon that occurs with aging, known as somatopause. With age, growth hormone (GH) secretion decreases, resulting in decreased IGF-1 production in both the liver and peripheral tissues. IGF-1 plays a crucial role in maintaining vascular function and cardiovascular homeostasis, so its decline in the elderly can contribute to an increased risk of hypertension.
Molecularly, IGF-1 exerts a vasoprotective effect through activation of IGF-1 receptors on endothelial cells and vascular smooth muscle cells. This activation stimulates the phosphatidylinositol-3-kinase (PI3K)/Akt pathway, which increases endothelial nitric oxide synthase (eNOS) activity and nitric oxide (NO) production. NO is a key mediator of vasodilation and maintains blood vessel elasticity. When IGF-1 levels are low, NO production decreases, resulting in endothelial dysfunction, increased peripheral vascular resistance, and ultimately increased blood pressure.4
In addition to causing endothelial dysfunction, IGF-1 deficiency also plays a role in increased arterial stiffness. IGF-1 has anti-apoptotic and anti-inflammatory effects on vascular tissue and plays a role in maintaining the integrity of vascular smooth muscle cells. Experimental studies have shown that low IGF-1 increases oxidative stress and vascular remodeling, leading to reduced arterial elasticity and increased systolic pressure—conditions often found in elderly hypertension. 38
IGF-1 also interacts with the renin–angiotensin–aldosterone system (RAAS). Physiologically, IGF-1 can reduce the vasoconstrictor response to angiotensin II. However, in conditions of IGF-1 deficiency, vascular sensitivity to angiotensin II increases, triggering vasoconstriction and sodium retention, leading to increased blood pressure.35 In the elderly, where RAAS regulation is already altered due to aging, low IGF-1 levels can exacerbate activation of this system.
Furthermore, low IGF-1 levels are associated with increased low-grade systemic inflammation and oxidative stress. Observational studies have shown an inverse relationship between IGF-1 levels and inflammatory markers such as C-reactive protein (CRP) and interleukin-6 (IL-6). This chronic inflammation contributes to endothelial damage and vascular dysfunction, increasing the risk of hypertension in the elderly population. 39
Epidemiologically, various cohort and population studies have shown that lower IGF-1 levels are significantly associated with increased systolic blood pressure and the prevalence of hypertension. Elderly people with IGF-1 levels in the lowest quartile were reported to have higher arterial stiffness and a greater risk of hypertension than groups with higher IGF-1 levels, even after adjusting for confounding factors such as age and body mass index.40 Thus, low IGF-1 levels can be understood as a relevant biological risk factor for hypertension in the elderly, particularly in the context of vascular aging.
Hypertension Risk in Older Adults Due to Declining Vitamin D and IGF-1 Concentrations
Hypertension is a prevalent condition among older adults and represents a major risk factor for cardiovascular disease. Aging influences blood pressure regulation through vascular changes (arterial stiffening), reduced renal function, and hormonal alterations. Two biological factors that decline with age, vitamin D (25[OH]D) and IGF-1 are hypothesized to contribute to the increased risk of hypertension through distinct yet interconnected pathways.2
This case-control study found that low vitamin D and IGF-1 levels were significantly associated with an increased risk of hypertension in the elderly. Subjects with 25(OH)D levels <30 ng/mL had an OR of 4.00 (95% CI: 1.155–13.855) higher risk of hypertension compared to those with adequate levels (Table 2). Meanwhile, IGF-1 levels <80 ng/mL were associated with an OR of 5.289 (95% CI: 1.438–19.447) higher risk of hypertension (Table 3). Analysis was also conducted to test the relationship between vitamin D and IGF-1 levels, in this study the results showed a significant relationship between vitamin D and IGF-1 levels, where participants with low Vit D levels had a 4-fold risk of having low IGF-1 levels compared to participants with normal levels (Table 4). Furthermore, multivariate logistic regression analysis obtained significant results for both variables (vitamin D and IGF-1) after adjustment for age, gender and BMI, both biomarkers remained independent predictors of hypertension (Table 5). This original research answers this hypothesis, where in this research the results obtained were low levels of vitamin D and IGF-1 as risk factors for hypertension in the elderly (p<0.05).
The age-related decline in 25(OH)D and IGF-1 has a biological basis that may elevate the risk of hypertension by promoting activation of the renin–angiotensin–aldosterone system (RAAS), impairing endothelial function, and reducing vascular anabolic capacity.36
Interestingly, the results of this study showed a significant association between vitamin D and IGF-1 levels, with participants with low vitamin D levels being at higher risk of having low IGF-1 levels. This suggests a possible synergistic effect. Biologically, both factors contribute to decreased NO bioavailability, increased RAAS activation, and increased chronic low-grade inflammation, which are key characteristics of hypertension in the elderly. Vitamin D is also known to play a role in regulating IGF-1 receptor expression and vascular anabolic metabolism,9 so deficiencies in both may impair vascular reparative capacity. This study strengthens previous observational evidence linking low vitamin D levels to hypertension 41 and low IGF-1 to increased blood pressure.13 The strength of this study is the simultaneous evaluation of both biomarkers in an elderly population, reflecting the real-world biology of aging.
Observational evidence supports these associations; however, findings from interventional trial particularly those involving vitamin D supplementation remain heterogeneous, with antihypertensive effects depending on baseline status and specific population characteristics.42 Therefore, current clinical emphasis is placed on identifying and correcting vitamin D deficiency in at-risk older adults and promoting lifestyle interventions (adequate nutrition, physical activity) that may help maintain IGF-1 levels and vascular health. More targeted RCTs and longitudinal studies are still needed to clarify causal pathways and determine the clinical benefits of such interventions for hypertension prevention. 43,44
The primary approach centers on addressing nutritional deficiencies (vitamin D and IGF-1) and implementing healthy lifestyle interventions to improve vascular function and stabilize blood pressure. Accordingly, monitoring vitamin D and IGF-1 status may provide an effective means of early risk screening in older populations. Should these factors be established as hypertension risk determinants, early recognition and coordinated interventions may help reduce hypertension-associated complications and support healthy aging outcomes.
Conclusion
In conclusion, the findings of this study indicate a significant association between reduced vitamin D and IGF-1 levels and the presence of hypertension among older adults. Participants with low vitamin D levels demonstrated a 4.000-fold increased risk of hypertension, whereas those with decreased IGF-1 levels exhibited a 5.289-fold higher risk compared with the control group with normal vitamin D and IGF-1 levels. There is a relationship between vitamin D and IGF-1 levels, where subjects with low vitamin D levels have a four-fold risk of having low IGF-1 levels as well, and both are independent predictors of hypertension in the elderly. These findings have important clinical implications, as low vitamin D and IGF-1 levels can be considered as additional risk markers in stratifying hypertension in the elderly. Identification of high-risk groups based on biomarkers can facilitate a more personalized preventive approach. However, because a case-control design cannot confirm a causal relationship, longitudinal studies are needed to assess whether correcting deficiencies in both biomarkers can reduce the incidence of hypertension.
Acknowledgement
The researchers express their gratitude to the Head of Selat Village, Abiansemal, Badung, Bali, as well as the Head and staff of Puskesmas Abiansemal IV, and the older adult participants of the posyandu lansia for their facilitation and cooperation throughout this research. Their support was essential in ensuring that the study could be conducted smoothly.
Funding sources
The author(s) received no financial support for the research, authorship, and/or publication of this article
Conflict of interest
The author(s) do not have any conflict of interest
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
The study received ethical approval from the Ethics Commission of the Faculty of Medicine at Udayana University, Denpasar-Bali, Indonesia No. 2808/UN.14.2.2.VII.14/LT/2025 with protocol number 2025.02.1.1250
Informed Consent Statement
This study involved human participants, and therefore, informed consent was required, the participants had signed the informed consent.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources – Not Applicable
Author contributions
- Luh Putu Ratna Sundari: Conceptualization, Methodology, Writing – Original Draft.
- Dewa Ayu Inten Dwi Primayanti: Analysis, Writing – Review & Editing, Funding Acquisition
- I Ketut Tunas: Visualization, Supervision, Collecting Data, Analysis.
- Made Alyashanti Radya Bulandari: Collecting Data, Writing and Editing, Project Administration.
References
- Ghebre YT., Yakubov E., Wong WT., Krishnamurthy P., Sayed N., Sikora AG., Bonnen MD. Vascular Aging: Implications for Cardiovascular Disease and Therapy. Transl Med (Sunnyvale). 2016 Dec;6(4):183. doi: 10.4172/2161-1025.1000183.
CrossRef - Jensen, N.S., Wehland, M., Wise, P.M., Grimm, D. Latest Knowledge on the Role of Vitamin D in Hypertension. Int J Mol Sci. 2023 Feb 28;24(5):4679. doi: 10.3390/ijms24054679.
CrossRef - Sundari L. P. R, Arsani N. L. K. A. Regular Physical Exercise Increase of Growth Hormone (GH) and Insulin-Like Growth Factor-1 (IGF-1) Activity in Elderly Improve the Aging Process and Quality of Life: A Mini Review. Biomed Pharmacol J. 2022;15(2).
CrossRef - Higashi, Y., Quevedo, H.C., Tiwari, S., Sukhanov, S., Shai, S.Y., Anwar, A., et.al. Interaction between insulin-like growth factor-1 and atherosclerosis and vascular aging. Front Horm Res. 2014; 43:107-24. doi: 10.1159/000360571.
CrossRef - Vicinanza, R., Frizza, A., Pollard, J.A., Mazza, V., De Martino, M.U., Imbimbo, G., et.al. Aging and IGF-1: relationships with vitamin D and body composition. A mediation analysis. 2025; June 24. doi: 10.3389/fnut.2025.1585696.
CrossRef - Sundari L. P. R, Purnawati S, Tunas I. K, Weta I. W. Low 25 Hydroxyvitamin D and High Leptin Level as Risk Factors of Metabolic Syndrome in Obese Women. Curr Res Nutr Food Sci 2022; 10(3). doi : http://dx.doi.org/10.12944/
CrossRef - Giustina, A., Bouillon, R., Dawson-Hughes, B., Ebeling, P.R., Lazaretti-Castro, M., Lips, P, et. al. Vitamin D in the older population: a consensus statement. Endocrine. 2023 Jan;79(1):31-44. doi: 10.1007/s12020-022-03208-3.
CrossRef - Conover, C.A., Oxvig, C. The IGF System and Aging. Endocrine Reviews. 2025; 46(2): 214-22. https://doi.org/10.1210/endrev/bnae029.
CrossRef - Ameri, P., Giusti, A., Boschetti, M., Murialdo, G., Minuto, F., Ferone, D. Interactions between vitamin D and IGF-I: from physiology to clinical practice. Clinical Endocrinology. 2013; 79 (4): 457-463. https://doi.org/10.1111/cen.12268
CrossRef - Carrara D, Bruno RM, Bacca A, Taddei S, Duranti E, Ghiadoni L, Bernini G. Cholecalciferol treatment downregulates renin-angiotensin system and improves endothelial function in essential hypertensive patients with hypovitaminosid D. J Hypertens. 2016 Nov;34(11):2199-205. doi: 10.1097/HJH.0000000000001072.
CrossRef - Tarantini S., Valcarcel-Ares, N.M., Yabluchanskiy, A., Springo, Z., Fulop, G.A., Ashpole, N., et.al. Insulin-like growth factor 1 deficiency exacerbates hypertension-induced cerebral microhemorrhages in mice, mimicking the aging phenotype. Aging Cell. 2017 Jun;16(3):469-479. doi: 10.1111/acel.12583.
CrossRef - Chakhtoura, M., Alam, S. Vitamin D and hypertension in the elderly; closer to implementation! Am Heart J Plus. 2025; 56:100570. doi: 10.1016/j.ahjo.2025.100570.
CrossRef - Leng, L., Xing, Y., Liang, Y., Wang, C., Ma,H. Relationship between circulating insulin-like growth factor-1 and blood pressure in adults: A systematic review and meta-analysis of observational studies. Growth Hormone & IGF Research. 2021; 60-61. ISSN 1096-6374. https://doi.org/10.1016/j.ghir.2021.101416.
CrossRef - Mokhtari, E., Hajhashemy, Z., Saneei, P. Serum Vitamin D Levels in Relation to Hypertension and Pre-hypertension in Adults: A Systematic Review and Dose-Response Meta-Analysis of Epidemiologic Studies. Front Nutr. 2022 Mar 10;9:829307. doi: 10.3389/fnut.2022.829307.
CrossRef - Hermawan, D and Andoko. Hubungan Antara Kadar Vitamin D Dalam Darah Dengan Tekanan Darah Usia Lanjut Di Natar Lampung Selatan. Jurnal Keperawatan Sriwijaya, 2019; 6 (1), Januari 2019, ISSN No 2355 5459
- Ferawaty, Sunardi, D., Dwimartutie, N., Chandra, D. N., Mudjihartini, N., & Manikam, N. R. M. Effect of vitamin D supplementation on serum 25(OH) D levels and blood pressure among the elderly in a nursing house: A double-blind, randomised placebo-controlled trial. Malaysian Journal of Nutrition. 2024; 30(2): 153-165. https://doi.org/10.31246/mjn-2023-0136.
CrossRef - Meshkini, F., Abdollahi, S., Clark, C.C.T., Soltani, S. The effect of vitamin D supplementation on insulin-like growth factor-1: A systematic review and meta-analysis of randomized controlled trials. Complementary Therapies in Medicine. 2020; 50. ISSN 0965-2299. https://doi.org/10.1016/j.ctim.2020.102300.
CrossRef - Hernandez-Vila E. A review of the JNC 8 Blood Pressure Guideline. Tex Heart Inst J. 2015 Jun 1;42(3):226-8. doi: 10.14503/THIJ-15-5067. PMID: 26175633; PMCID: PMC4473614.
CrossRef - Kaur J, Khare S, Sizar O, Givler A. Vitamin D Deficiency. 2025 Feb 15. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 30335299.
- Braverman E, Oscar-Berman M, Lohmann R, Kennedy R, Kerner M, Dushaj K, Blum K. Low and Normal IGF-1 Levels in Patients with Chronic Medical Disorders (CMD) is Independent of Anterior Pituitary Hormone Deficiencies: Implications for Treating IGF-1 Abnormal Deficiencies with CMD. J Genet Syndr Gene Ther. 2013 Feb 9;4(123):1000123. doi: 10.4172/2157-7412.1000123.
CrossRef - Kasiulevicius, V., Sapoka, V. and Filipaviciūte, R. Sample Size Calculation in Epidemiological Studies. 2006. Vilnius University, Institute of Experimental and Clinical Medicine at Vilnius University.
- Karadeniz Y., Özpamuk-Karadeniz F., Ahbab S., Ataoğlu E., Can G. Vitamin D Deficiency Is a Potential Risk for Blood Pressure Elevation and the Development of Hypertension. Medicina.2021;57:1297. doi: 10.3390/medicina57121297.
CrossRef - McMullan C.J., Borgi L., Curhan G.C., Fisher N., Forman J.P. The effect of vitamin D on renin-angiotensin system activation and blood pressure: A randomized control trial. J. Hypertens. 2017;35:822–829. doi: 10.1097/HJH.0000000000001220.
CrossRef - Li YC, Qiao G, Uskokovic M, Xiang W, Zheng W, Kong J. Vitamin D: a negative endocrine regulator of the renin-angiotensin system and blood pressure. J Steroid Biochem Mol Biol. 2004 May;89-90(1-5):387-92. doi: 10.1016/j.jsbmb.2004.03.004. PMID: 15225806.
CrossRef - Han L, Xu XJ, Zhang JS, Liu HM. Association between Vitamin D Deficiency and Levels of Renin and Angiotensin in Essential Hypertension. Int J Clin Pract. 2022 Jun 10;2022:8975396. doi: 10.1155/2022/8975396. PMID: 35814306; PMCID: PMC9205726.
CrossRef - Andrukhova O., Slavic S., Zeitz U., Riesen SC., Heppelmann MS., Ambrisko TD., Markovic M., Kuebler WM., Erben RG.. Vitamin D is a regulator of endothelial nitric oxide synthase and arterial stiffness in mice. Mol Endocrinol. 2014 Jan;28(1):53-64. doi: 10.1210/me.2013-1252. Epub 2013 Jan 1. PMID: 24284821; PMCID: PMC5426652.
CrossRef - Wee CL., Azemi AK., Mokhtar SS., Yahaya S., Yaacob NS., Rasool AHG. Vitamin D deficiency enhances vascular oxidative stress, inflammation, and angiotensin II levels in the microcirculation of diabetic patients, Microvascular Research, Volume 150, 2023, 104574, ISSN 0026-2862, https://doi.org/10.1016/j.mvr.2023.104574.
CrossRef - Ann Bugeja, Gregory L Hundemer, Vitamin D and Hypertension: An Uncertain Relationship at Best, American Journal of Hypertension, Volume 37, Issue 12, December 2024, Pages 945–947, https://doi.org/10.1093/ajh/hpae114
CrossRef. - Ajabshir, S., Asif, A., Nayer, A. The effects of vitamin D on the renin-angiotensin system. Journal of Nephropathology. 2014;3(2):41. doi: 10.12860/jnp.2014.09.
- Kim, D.H., Meza, C.A., Clarke, H., Kim, J.S., Hickner, R.C. Vitamin D and endothelial function. Nutrients. 2020;12(2):575. doi: 10.3390/nu12020575.
CrossRef - Soleimani M. Insulin resistance and hypertension: new insights. Kidney Int. (2015) 87:497–9. doi: 10.1038/ki.2014.392
CrossRef - Borissova AM, Tankova T, Kirilov G, Dakovska L, Kovacheva R. The effect of vitamin D3 on insulin secretion and peripheral insulin sensitivity in type 2 diabetic patients. Int J Clin Pract. (2003) 57:258−61.
CrossRef - Erlandsson, M.C., Lyngfelt, L., Åberg, N.D. et al.Low serum IGF1 is associated with hypertension and predicts early cardiovascular events in women with rheumatoid arthritis. BMC Med 17, 141 (2019). https://doi.org/10.1186/s12916-019-1374-x
CrossRef - Zhang L, Curhan GC, Forman JP. Plasma insulin-like growth factor-1 level and risk of incident hypertension in nondiabetic women. J Hypertens. 2011 Feb;29(2):229-35. doi: 10.1097/HJH.0b013e32834103bf.
CrossRef - Conti E, Carrozza C, Capoluongo E, Volpe M, Crea F, Zuppi C, Andreotti F. Insulin-like growth factor-1 as a vascular protective factor. 2004 Oct 12;110(15):2260-5. doi: 10.1161/01.CIR.0000144309.87183.FB.
CrossRef - Ungvari Z, Csiszar A. The emerging role of IGF-1 deficiency in cardiovascular aging: recent advances. J Gerontol A Biol Sci Med Sci. 2012;67(6):599–610. doi: 10.1093/gerona/gls072.
CrossRef - Denti L, Annoni V, Cattadori E, Salvagnini MA, Visioli S, Merli MF, Corradi F, Ceresini G, Valenti G, Hoffman AR, Ceda GP. Insulin-like growth factor 1 as a predictor of ischemic stroke outcome in the elderly. Am J Med. 2004 Sep 1;117(5):312-7. doi: 10.1016/j.amjmed.2004.02.049. .
CrossRef - Sukhanov S, Higashi Y, Shai SY, Vaughn C, Mohler J, Li Y, Song YH, Titterington J, Delafontaine P. IGF-1 reduces inflammatory responses, suppresses oxidative stress, and decreases atherosclerosis progression in ApoE-deficient mice. Arterioscler Thromb Vasc Biol. 2007 Dec;27(12):2684-90. doi: 10.1161/ATVBAHA.107.156257. Epub 2007 Oct 4. PMID: 17916769.
CrossRef - Juul A, Scheike T, Davidsen M, Gyllenborg J, Jørgensen T. Low serum insulin-like growth factor I is associated with increased risk of ischemic heart disease: a population-based case-control study. Circulation. 2002 Aug 20;106(8):939-44. doi: 10.1161/01.cir.0000027563.44593.cc. PMID: 12186797.
CrossRef - Colao, A., Di Somma, C., Spiezia, S., Savastano, S., Rota, F., Savanelli, M. C., & Lombardi, G. (2008). Growth hormone treatment on atherosclerosis: Results of a 5-year open, prospective, controlled study in male patients with severe growth hormone deficiency. Journal of Clinical Endocrinology and Metabolism, 93(9), 3416–3424. https://doi.org/10.1210/jc.2007-2810
CrossRef - Kunutsor SK, Apekey TA, Steur M. Vitamin D and risk of future hypertension: meta-analysis of 283,537 participants. Eur J Epidemiol. 2013 Mar;28(3):205-21. doi: 10.1007/s10654-013-9790-2. Epub 2013 Mar 2. PMID: 23456138.
CrossRef - Rahme, M., Al-Shaar, L., Tamim, H., El-Hajj Fuleihan, G. Blood pressure decreases in overweight elderly individuals on vitamin D: a randomized trial. Journal of the Endocrine Society. 2024; 8(12) doi: 10.1210/jendso/bvae168.
CrossRef - Stankovics, L., Ungvari, A., Fekete, M., Nyul-Toth, A., Mukli, P., Patai R., et.al. The vasoprotective role of IGF-1 signaling in the cerebral microcirculation: prevention of cerebral microhemorrhages in aging. Geroscience. 2025 Feb;47(1):445-455. doi: 10.1007/s11357-024-01343-5. Epub 2024 Sep 14.
CrossRef - Carey R.M., Wright J.T Jr, Taler S.J., Whelton P.K. Guideline-Driven Management of Hypertension: An Evidence-Based Update. Circ Res. 2021 Apr 2;128(7):827-846. doi: 10.1161/CIRCRESAHA.121.318083.
CrossRef







