Manuscript accepted on :22-12-2025
Published online on: 06-02-2026
Plagiarism Check: Yes
Reviewed by: Dr. M Mohan Varma
Second Review by: Dr. Mohamad R. Abdullah
Final Approval by: Dr. Anton R Keslav
Sagar Bhayye1 , Rakhee Dangi2*
, Rakhee Dangi2* and Sagar Datir3
and Sagar Datir3
1Department of Bioinformatics, Bharati Vidyapeeth (Deemed to be University), Pune, India.
2Department of Cell and Molecular Biology, Bharati Vidyapeeth (Deemed to be University), Pune, India.
3Department of Plant taxonomy, Naoroji Godrej Centre for Plant Research, Pune, India.
Corresponding Author Email: rakhee.dangi@bharatividyapeeth.edu
DOI : https://dx.doi.org/10.13005/bpj/3351
Abstract
Ethnobotanically, Premna serratifolia has been used in folk medicine to treat a number of aliments including diabetes. However, there is no data on its antiglycation activity. The present study evaluated the alpha-glucosidase inhibition potential, antiglycation, cell protective and antioxidant potential of crude total methanol and fractionated hexane, ethyl acetate and methanol extracts prepared from leaves and stem bark of P. serratifolia. The stem ethyl acetate extract (S-EA) had the highest alpha-glucosidase inhibition potential, total phenolic and flavonoid content. The S-EA extract also displayed highest total antioxidant activity and highest reducing power. The total methanol extract of leaves (L-TM) displayed maximum free radical scavenging activity evaluated using 2,2-diphenyl-1-picrylhydrazyl (DPPH) and 2,2′-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid (ABTS). All aqueous extracts of leaves and stem exerted noticeable antiglycation potential evaluated using multistage glycation markers (fructosamines, protein carbonyls, thiols and β aggregation) with reduction in carbonyl levels and increase in free thiol groups (R=0.890; p<0.05) well correlated. However, the S-EA extract was more efficient in protecting erythrocytes from oxidative damage induced by glycated albumin. LC-MS-based metabolomic profiling of the S-EA extract, combined with in silico molecular docking studies, identified six compounds—6-hydroxy salvinolone, 10-O-trans-p-coumaroylcatalpol (10OCC), 10-O-trans-p-methoxycinnamoylcatalpol, Premnacorymboside B, 4-hydroxy-E-globularinin (B), and Premnaodoroside B—as putative novel inhibitors of α-glucosidase. These compounds warrant further purification and experimental validation to confirm their inhibitory activity.
Keywords
Anti-diabetic; Anti-glycation; Docking; Iridoid-glycosides; Stem
Download this article as:| Copy the following to cite this article: Bhayye S, Dangi R, Datir S. LC-MS Based Metabolomics, In vitro and In silico Approaches to Characterize Possible Inhibitors of Alpha-glucosidase and Protein Glycation from Premna serratifolia L: Implications in Controlling Diabetes. Biomed Pharmacol J 2026;19(1). |
| Copy the following to cite this URL: Bhayye S, Dangi R, Datir S. LC-MS Based Metabolomics, In vitro and In silico Approaches to Characterize Possible Inhibitors of Alpha-glucosidase and Protein Glycation from Premna serratifolia L: Implications in Controlling Diabetes. Biomed Pharmacol J 2026;19(1). Available from: https://bit.ly/4rBgVJv |
Introduction
In diabetes, inhibition of alpha-glucosidase is a primary approach to control postprandial hyperglycemia. This enzyme in the gastrointerstinal tract catalyses the hydrolysis of alpha-glucosidic linkages of oligosaccharides to generate monosaccharides that are absorbed in the small intestine. Inhibition of alpha-glucosidase activity can reduce glucose formation from carbohydrates leading to reduction of postprandial hyperglycemia in diabetes. Undesirable gastrointestinal side effects, poor absorption from gut, excretion in stool and low bioavailability of currently used alpha-glucosidase inhibitors impede their long term application.1 Therefore, there is still a need to identify novel inhibitors with minimal side effects.
The chronic hyperglycaemic conditions in diabetes leads to generation of ROS and glycation of structural and functional plasma proteins like albumin, fibrinogen, globulins and collagen.2 The oxidative stress triggered by ROS causes significant damage to cellular biomolecules while glycation of protein leads to formation of AGEs (advanced glycation end products). In glycation, Schiff base is formed due to the reaction of sugars with lysine or arginine of proteins. This compound undergoes rearrangements to form amadori product which is broken down by oxidation, dehydration, and other chemical processes resulting in formation of AGEs.3 Many studies have proved that AGEs generate oxidative stress. Moreover, by interacting with receptors for AGEs (RAGE) they can cause vascular dysfunction and inflammation leading to diabetic complications.4 Free radicals derived from glycation cause fragmentation of proteins and oxidative damage of biomolecules.5 Thus, AGEs are targets for the treatment of diabetic complications.4 Oxidative stress as well as AGEs contribute to the progression and complications associated with diabetes. Synthetic antioxidants (butylhydroxyanisole (BHA) and butylhydroxytoluene (BHT) pose health risks and toxicity.6 Chemically synthesized drugs with antiglycation activity also cannot be used in long term because of adverse reactions.3 Hence, natural compounds from medicinal plants that can release oxidative stress and target specific steps of glycation cascade have attracted increasing attention as lead compounds for drug development because of their minimal toxic effects. 7
Premna serratifolia L. (Verbenaceae, synonym P. integrifolia) is a small medicinal tree commonly found across the tropical and subtropical regions of Asia. It is used for treating cardiovascular disorder, cough, diabetes, inflammation, stomach disorders and rheumatism.8 Literature survey has revealed many pharmacological studies that confirmed the ethno medicinal use of P. serratifolia as an anti-diabetic plant.9,10 The plant is known to contain bioactive molecules with antihyperglycemic,11-13 anti-inflammatory,14, 15 and antioxidant activities.16-17
Increasing research has proved that one mechanism by which P. serratifolia exerts its antidiabetic activity is by inhibiting carbohydrate digesting enzymes and reducing postprandial blood glucose rise in diabetes.18-20 However, pharmacological profiling of P. serratifolia to identify bioactive molecules with potent alpha-glucosidase inhibition potential has been restricted to the identification of only few flavonoids, phenylethanoid glycosides (PhGs) and sesquiterpenoids from leaves.20 Although, flavonoids showed strong inhibition of alpha-glucosidase in vitro, most of them showed less or no in vivo actions due to poor water solubility and low GI absorption. 1 Moreover, no systematic study has investigated the protective role of P. serratifolia in glucose-induced albumin glycation and glycated albumin mediated toxicity to erythrocytes. Being an antidiabetic plant with high antioxidant potential, we hypothesized that P. serratifolia might contain unique phytochemistry to attenuate glycation. The objectives were (1) to evaluate the role of various extracts prepared from leaves and stem of P. serratifolia for alpha-glucosidase inhibition potential and antiglycation activity (2) to assess the extracts capacity to shield erythrocytes against glycated albumin-induced oxidative damage (3) to correlated the antioxidant potential and content of phenolics, and flavonoids with the protective properties of extracts (4) to identify potential bioactive metabolites in the extract with most promising alpha-glucosidase inhibition, antiglycation and cell protective potential using LC-MS (5) molecular docking of identified compounds with maltase-glucoamylase and isomaltase to understand their in silico molecular interactions and affinity.
Materials and Methods
Collection and authentication of P. serratifolia
P. serratifolia was collected from Western Ghats in flowering and fruiting period which occurs between October to December. The leaves and stem wood were separated from flowers and fruits and shade dried separately. Flowering and fruiting twigs were kept in blotters under pressure for drying. The blotters were changed every 24 to 48 hours to avoid any type of microbial infection till complete drying of the specimens. Later, the dried specimens were poisoned using the solution of HgCl2, formaldehyde, alcohol and water. After complete drying, the specimens were mounted on standard herbarium sheet. The identification process was conducted in collaboration with established taxonomists from Naroji Godrej Centre for Plant Research (NGCPR) ensuring accuracy and reliability. The herbarium sample (NGCPR09908) was formally submitted to the NGCPR, Shindewadi, Shriwal, Maharashtra, India for archival and reference purpose.
Preparation of Plant extracts
A dry grinder was used to grind the stem wood and shade-dried leaves into a fine powder. Ten grams of powder (stem and leaf) was extracted with methanol (100 ml) for 16 hours using Soxhlet. To get the crude total methanolic extract (TM) of the leaves and stem (L-TM and S-TM), rotary evaporator was used to evaporate methanol under reduced pressure. To prepare fractionated extracts, 25 grams of powder (stem and leaf) was extracted with hexane (H), ethyl acetate (EA) and methanol (M) for 16 hours. The crude H, EA and M extracts were concentrated and dried. For in vitro assays, the dried crude TM, H, EA and M extracts of leaves (L-TM, L-H, L-EA and L-M) and stem (S-TM, S-H, S-EA and S-M) were dissolved in water and methanol to prepare aqueous and methanol extract respectively (100 mg/ml).
Inhibition of alpha-glucosidase
The ability of aqueous extracts to inhibit enzyme alpha-glucosidase was carried out according to the method of Gaonkar and Dangi.3 Positive control was Acarbose. Percentage inhibition was determined as described by Gaonkar and Dangi.3 Using Microsoft Excel IC50 was recorded graphically.
Total phenolic contents (TPC)
TPC of leaves (L-TM, L-H, L-EA and L-M) and stem (S-TM, S-H, S-EA and S-M) extracts was done using the Folin-Ciocalteau reagent with minor modifications. 3 Plant extract (500 µl, 100 mg/ml) was mixed with 500 µl of diluted (10-fold dilution with deionised water) Folin-Ciocalteau reagent and 500 μl of 1 M Na2CO3. After an incubation (15 minutes, dark, 25° C), absorbance was recorded at 765 nm for all samples. Gallic acid (10 to 100 µg/ml, Y =0.0155x + 0.1119; R2 =0.9995) was used as standard. Findings were articulated as mg GAE/g DE by computing it with standard calibration curve.
Total flavonoid contents (TFC)
TFC of leaves (L-TM, L-H, L-EA and L-M) and stem (S-TM, S-H, S-EA and S-M) extracts was performed using spectrophotometric method described previously.3 Plant extract (0.4 – 4 mg/ml, 100 µl), methanol (400 µl), AlCl3 (10%, 100 µl) and CH3CO2K (1M, 100 µl) were mixed and incubated (30 minutes, dark, 25 °C). Absorbance was noted at 415 nm. Quercetin was the standard (10 to 100 µg/ml, Y =0.0093x + 0.148; R2 =0.9993). Results was expressed as mg QE/g DE.
Radical scavenging by DPPH
The scavenging action of methanolic extracts of leaves (L-TM, L-H, L-EA, and L-M) and stems (S-TM, S-H, S-EA, and S-M) towards DPPH was ascertained using ascorbic acid as standard (Y = 2.5533x + 3.7306; R2 = 0.9441).21 The reaction mixture containing 300 µl of DPPH (0.1 M) and 100 µl of extracts (5.0 – 60 µg/ml) was incubated (30 minutes, dark, 37° C). Absorbance at 517 nm was used to calculate DPPH scavenging activity and IC50 as described by Gaonkar and Dangi.3
Radical scavenging by ABTS
The assay was performed as reported by Re et al. 22 Stocks used were 2.4 mM potassium persulfate and 7 mM ABTS solutions in water. 500 µL of ABTS and 5.0 µL of potassium persulfate were combined to create the working solution. The working solution was left to incubate at 25° C for 12 to 16 hours in dark. Distilled water was added to dilute the solution until it at 734 nm it had an absorbance of 0.70 ± 0.02. 50 μl of methanolic plant extracts at different concentrations (0.5 to 25 µg/mL) and 250 μl of the freshly made ABTS solution were mixed, vortexed, and incubated (25° C, dark) for precisely six minutes. Gallic acid with concentrations of 0.2 to 1 µg/ ml was used as standard. Standard graph was obtained for Y =61.26x + 7.8909; R² =0.9972. The percentage scavenging activity and IC50 of each extract on ABTS was calculated using the formula stated above.
Total antioxidant activity (TAA)
TAA expressed as mg AAE/g DE of methanolic extracts was estimated using phosphomolybdenum method described by Gaonkar and Dangi 3 using ascorbic acid as standard (10 – 100 µg/ml, Y = 0.0371x+0.1773, R2 = 0.9976). All leaf and stem extracts (1 mg/ml) mixed with 1000 µl of reagent (0.6 M, H2SO4; 28 mM, Na3PO4 and 4 mM, (NH4)2MoO4) were incubated at 95° C for 90 min. Absorbance was measured at 695 nm against blank.
Ferric Reducing Antioxidant Power Assay (FRAP Assay)
FRAP assay was performed following the standard method.23 Phosphate buffer (0.2 M, pH 6.6, 2.5 ml), plant extracts (0.1 to 3 mg/ml), and potassium ferricyanide (1%, 2.5 ml) were mixed and incubated (50°C, 20 min). After cooling, trichloroacetic acid (10%, 2.5 ml) was added and the mixture was centrifuged at 3000 rpm for 10 min. Upper layer (2.5 ml) was mixed with distilled water (2.5 ml) and freshly prepared ferric chloride solution (0.1%, 0.5 ml). Absorbance was recorded at 700 nm. Ascorbic acid (50 to 300 μg/ml) was used as standard. RP was expressed as mg AAE/g DE.
Antiglycation Potential
Glycated samples (BSA + Glucose + sodium azide), glycated samples co-incubated with plant extracts (BSA + Glucose + sodium azide + 150 µg/ml aqueous plant extract) and positive control (BSA + Glucose + sodium azide + Quercetin final concentration 10 µg/ml) were prepared as described by Goankar and Dangi.3 Antiglycation activity was assessed by determining Fructosamines, 24 Protein carbonyls, 25 Protein thiols, 26 and amyloid β aggregation (Congo Red absorbance) as described previously.3
Impact of plant extracts on toxicity to erythrocytes by glycated albumin
Erythrocyte cell suspension
Fresh sheep blood was drawn into a heparin-containing tube, centrifuged for 15 minutes at 3000 rpm to separate plasma and washed three times with a 0.15M sodium chloride solution. A 4% cell suspension was created by suspending the pellet in PBS (pH 7.3).
Erythrocytes treated with glycated albumin samples
Erythrocyte suspension was incubated with equal amount of glycated albumin, glycated albumin samples co-incubated with extracts and quercetin along with controls (PBS containing 1% BSA and PBS containing 1% Triton X-100 (v/v)) for 4 h at 37 °C with intermittent shaking. In the treated erythrocytes, hemolysis, LPO (Lipid peroxidation) and GSH (reduced glutathione) were measured.
Erythrocytes hemolysis
After incubation 0.5 ml of the treated erythrocytes were centrifuged (3000 rpm, 15 minutes). The absorbance of supernatant was noted at 540 nm.27 The following formula was used to calculate the hemolysis percentage:
[ASample -AControl]/ [ATritonX100 – AControl] ×100
Where ASample, AControl and ATritonX100 represents the results of glycated samples, PBS control and positive control respectively
LPO
Oxidative damage to lipids was determined by measuring Malondialdehyde (MDA), the end product of LPO as reported by Placer et al. with minor modifications.28 To 200 µl of treated erythrocytes 0.8 ml of Tris Maleate buffer (0.2M, pH 6.0) was added. For deproteinization, 200 µl of solution was mixed with 1 ml of TCA (10%). The mixture was centrifuged at 3000 rpm for 10 minutes and to 1 ml of the supernatant, 2 ml of Thiobarbutaric acid (0.67% in 0.25 M HCL) was added in a separate tube. Tubes were incubated in a boiling water bath for 25 minutes. After incubation, the cooled tubes were centrifugation. The content of MDA (as nM of MDA per mg of protein) was recorded at 532 nm.
GSH
Ellman modified approach was used to estimate reduced GSH.26 50 µl of treated erythrocytes were mixed with precipitating solution (125 µl, 5% TCA in 1 mM EDTA). Samples were centrifuged (10 minutes, 2000 rpm) following protein precipitation. Phosphate buffer (625 µl, 0.1 M, pH 8.0) and 25 µl of DTNB (0.01% in PBS) were combined with the supernatant (25 µl). The absorbance at 412 nm was measured after 10 minutes of incubation and compared to a blank sample that was cell-free. Using a molar extinction coefficient value of 14150M-1 cm-1, the GSH concentration (nM of GSH per mg of protein) was determined.
Liquid Chromatography-Mass Spectrometry (LC-MS) studies
The tentative phyto compounds in the S-EA extract were analysed using LC-MS (Instrument: Agilent Q-ToF G6540B connected to Agilent 1260 Infinity II HPLC, Column: Agilent Eclipse XDB-C18, 3X150 mm, 3.5 micron). Full-scan mode from m/z 100 to 1700 was performed with a source temperature of 40°C. Solvents A (0.10% formic acid in water) and B (0.10% formic acid in acetonitrile) made up the mobile phase (gradient mode). The gradient flow was 0–2 minutes (5% B), 2–25 minutes (5% B), 25–28 minutes (95% B), and 28–30 minutes (5% B). 0.3 mL/min was the flow rate. Both positive and negative ion modes were used for the mass spectrometry analysis. With a gas flow rate of 11 L/min and a surrounding sheath gas temperature of 350 °C, the nebulizer pressure was adjusted at 35 psi. A flow rate of 8 L/min and a drying gas temperature of 300 °C were established. The voltage gradient for the nozzle, skimmer, capillary (VCap), and fragmentor was set to 1000 V, 65 V, and 175 V, respectively, using the default parameters. Using a MassHunter 6200 series TOF/6500 series Q-TOF B.05.01, LC-MS data was recorded. To enable column conditioning and assess repeatability, three replicate injections were used for MS acquisition. To process mass spectra, the METLIN Metabolited AM PCDI was utilized. The precise m/z and the molecular formula generation software (Agilent MassHunder Qualitative Analysis) were used to generate the right elemental composition for presumptive chemical identification along with a database of reported compounds from P. serratifolia.
Molecular docking
To identify novel inhibitors of Maltase-Glucoamylase, Isomaltase and RAGE from the extract of P. serratifolia, molecular docking of phyto compounds identified with the help of LC-MS and literature was performed. PubChem database (https://pubchem.ncbi.nlm.nih.gov/ (accessed on 3 April 2025)) was used to download the chemical structures of ligands identified by LC-MS. Crystal structures of human Maltase-Glucoamylase having Acarbose binding site at N-terminal (PDB ID: 2QMJ) and at C-terminal (PDB ID: 3TOP) were taken from protein data bank 20. Similarly, crystal structure of Isomaltase (PDB ID: 3A4A) belonging to Saccharomyces cerevisiae was also selected for molecular docking studies.20 All crystal structures were prepared for molecular docking by removing non-protein molecules (such as ligands, ions) followed by addition of missing hydrogen’s and Kollman charges in AutoDock tool. All the ligands in dataset were prepared for docking by adding Gasteiger charges. Grid for molecular docking was prepared by keeping co-crystal ligand (Acarbose) at the centre for N- and C-terminal of human maltase crystal structures, and for Isomaltase grid was prepared by keeping Maltose (co-crystal ligand) at the centre. Grid size was 40 x 40 x 40 Å considering variable size of ligands in dataset. Molecular docking performed using AutoDock Vina.29 Protein-ligand interactions were recorded using PyMOL and Desmond’s Maestro interface.30
ADMET studies
The pharmacokinetic properties of newly docked metabolites were calculated using admetSAR servers (https://lmmd.ecust.edu.cn/admetsar2).31 The ADMET properties studied for the selected putative alpha-glucosidase inhibitor metabolites include solubility, carcinogenicity, human intestinal absorption (HIA), toxicity and blood-brain barrier penetration. The obtained results were saved in tabular form.
Statistical analysis
All assays were done thrice (n= 3) and results were documented as mean and standard deviations. Pearson correlation coefficient ‘R’, ANOVA and post hoc analysis (Bonferroni test, p< 0.05) was done using Microsoft Excel
Results
Alpha-glucosidase inhibitory activity
Aqueous extracts prepared from crude leaf (L-TM, L-H, L-EA and L-M) and stem (S-TM, S-H, S-EA and S-M) extracts were tested for alpha-glucosidase inhibition. The standard graph of acarbose (5-60 µg/ml, Y= 0.397 + 27.06, R2 = 0.9733) showed alpha-glucosidase inhibitory activity with an IC50 value of 49.325 ± 5.83 µg/ml. All aqueous extracts inhibited alpha-glycosidase activity in concentration ranging from 10 to 50 µg/ml. A comparable level of inhibition of alpha-glycosidase was observed in L-H and S-H extracts. Significant difference was found in the TM, EA and M extracts of leaves and stem for inhibition of alpha-glucosidase enzyme (Table 1). Comparing the IC50 values, the L-TM, L-EA and L-M extracts were less effective than acarbose in inhibiting alpha-glucosidase enzyme. The S-TM, S-EA and S-M extracts had stronger inhibitory activity as compared to the extracts prepared from leaves (Table 1). The inhibition potencies of S-TM, S-H, S-EA and S-M extracts was also much stronger than the inhibitory effect obtained from acarbose. The S-EA extract showed the best enzyme inhibition activity (IC50= 1.941 ± 0.12 µg/ml).
Table 1: Alpha-glucosidase inhibition by extracts of P. serratifolia
|
Extract |
IC50 values of extracts prepared from leaves (µg/ml) |
IC50 values of extracts prepared from stem (µg/ml) |
|
Acarbose |
49.325 ± 5.838 |
|
|
Total methanol (TM) |
77.340 ± 2.310a |
7.165 ± 1.175b |
|
Hexane (H) |
6.615 ± 0.025c |
4.685 ± 0.965c |
|
Ethyl acetate (EA) |
78.03 ± 1.22d |
1.245 ± 0.165e |
|
Methanol (M) |
66.665 ± 1.965f |
8.505 ± 0.005g |
Means (n=3, mean ± standard error) in a row with different small case letters are significantly different (P< 0.05).
TPC and TFC
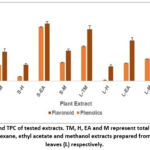 |
Figure 1: TFC and TPC of tested extracts. TM, H, EA and M represent total methanol and fractionated hexane, ethyl acetate and methanol extracts prepared from stem (S) and leaves (L) respectively.
|
The variation in TPC and TFC of all tested extracts is shown in Fig. 1. The S-EA extract has the highest TPC (576.666 ± 1.862 mg GAE/g DE) and TFC (87.2631 ± 4.184 mg QE/g DE). The fractionated H extract from both leaves and stem has the lowest values for TPC (49.275 ± 1.303 and 10.354 ± 2.234 mg GAE/g DE) and TFC (24.881 ± 2.581 and 22.436 ± 1.61 mg QE/g DE) respectively. A very good correlation was observed in TPC and TFC for extracts prepared from stem (R=0.995, p<0.01) as well as leaves (R= 0.968, p<0.05).
Antioxidant activity by DPPH and ABTS assays
The comparative evaluation of ABTS and DPPH radical scavenging activities in stem and leaf extracts as evidenced by their respective IC 50 values is displayed in Fig. 2 a & b respectively. All the extracts displayed concentration dependent DPPH and ABTS free radical scavenging activity. In both, DPPH and ABTS assays, the extracts prepared from leaves had a better radical scavenging activity as compared to extracts prepared from stem (Fig. 2a & b). In DPPH assay, the L-TM had the highest radical scavenging activity (IC50= 11.3044 ± 5.267 µg/ml) which was better than the standard (IC50= 17.32 ± 0.803 µg/ml). For the remaining extracts, the IC50 values were higher than that of standard. In agreement to DPPH assay, the L-TM had the highest ABTS radical scavenging activity (0.624 ± 0.02 µg/ml) which was again better than the standard gallic acid (IC 50= 0.704 ± 0.103 µg/ml). For the remaining extracts the IC50 values were higher than that of standard (Fig. 2a & b). Antioxidant activity tested by DPPH and ABTS were significantly correlated (R=0.61, p<0.001). These results imply that all extracts made from stems and leaves have a consistent correlation between the antioxidant potential measured by DPPH and ABTS assays.
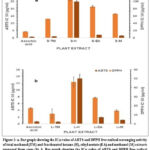 |
Figure 2 a: Bar graph showing the IC50 value of ABTS and DPPH free radical scavenging activity of total methanol (TM) and fractionated hexane (H), ethyl acetate (EA) and methanol (M) extracts prepared from stem (S).
|
TAA and RP
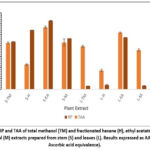 |
Figure 3: RP and TAA of total methanol (TM) and fractionated hexane (H), ethyl acetate (EA) and methanol (M) extracts prepared from stem (S) and leaves (L). Results expressed as AAE (mg of Ascorbic acid equivalence).
|
The plant part and solvent type displaced a noticeable variation in the antioxidant activity determined by phosphomolybdenum method (TAA) and FRAP assay (RP). The TM, H, EA and M extracts prepared from stem had higher TAA and RP when compared with TM, H, EA and M extracts prepared from leaves respectively (Fig. 3). Among the stem extracts, the S-EA extract had highest TAA (97.4582 ± 0.7242 mg AAE/g DE) and RP (1254.17 ± 3.009 mg AAE/g DE). Similarly, among the extracts prepared from leaves, the L-EA extract had the highest TAA (88.832 ± 1.753 mg AAE/g DE) and RP (948.3 ± 4.2 mg AAE/g DE).
Antiglycation activity
Extracts had inhibitory effects ranging from 36.885% to 55.17% on the production of fructosamines, an early glycation product (Fig. 4a). The S-EA extract exhibited the highest level of inhibition among the stem extracts. Likewise, the L-EA extract exhibited the highest level of inhibition among the leaf extracts. The levels of protein carbonyl were assessed after 28 days in order to determine the protein oxidation caused by the glycation process, as illustrated in Fig. 4b. Comparative analysis of aqueous extracts prepared from the crude TM, H, EA and M extracts of leaves and stem showed almost similar level of inhibition of reactive carbonyl compounds. Among the extracts tested, the EA and TM extracts from both leaves and stem were most potent inhibitors of carbonyl compounds. As shown in Fig. 4c, the DTNB reagent was used to evaluate the free thiol groups in BSA following glycation. Glycated samples co-incubation with standard quercetin as well as plant extracts notably elevated free thiol groups. The EA and TM extracts from stem as well as leaves showed higher percent protection of free thiol groups. In albumin, the degree of β-amyloid formation was estimated utilizing the amyloid-specific Congo red dye known for its specific affinity towards fibrous structures. Results from the present study are presented as percentage inhibition of amyloid aggregate formation and illustrated in Fig. 4d. Among the stem extracts the S-TM showed the highest decrease of inhibition of β-amyloid aggregation. Similarly, among the leaf extracts, the L-TM extract was a more potent inhibitor of β-amyloid aggregation. There was a strong positive correlation between decrease in carbonyl content and increase in free thiol groups in extracts prepared from leaves (R=0.991, p<0.05) as well as stem (R=0.940, p<0.05). The TM extracts from leaves as well as stem outperformed all extracts in inhibiting β-amyloid aggregation. Based on their additive ability to inhibit formation of fructosamines, carbonyl compounds, β-amyloid aggregation and protection of thiol group, the TM and EA extract from leaves and stem displayed a higher overall antiglycation potential (Fig. 4e).
 |
Figure 4: Anti glycation activity of plant extracts and standard (quercetin). a: Percent Fructosamine inhibition b. Percent carbonyl inhibition. c: Percent thiol protection
|
Toxicity of glycated albumin to erythrocytes
The stem extracts (S-TM, S-H, S-EA and S-M) were analysed for their protective effect in preventing erythrocyte hemolysis and maintaining antioxidant status (Table 2). Glycated albumin sample (positive control) showed 86% hemolysis indicating significant cellular oxidative damage to erythrocytes by glycated albumin as compared to BSA (negative control) alone. Erythrocyte hemolysis was reduced in the range of 36% to 72% in presence of stem extracts (Table 2). The S-EA extract showed maximum attenuation with a percentage inhibition of 36 % which was comparable to that obtained by standard (quercetin).
Table 2: The effect of plant extracts on erythrocytes toxicity measured during albumin glycation.
|
Sample |
Haemolysis (%) |
Lipid peroxidation ( nM MDA/mg protein) |
Reduced glutathione (nM/mg protein) |
|
BSA (negative control) |
1 ± 0.004*** |
37.06 ± 0.77 * |
40.34 ± 0* |
|
Glycated sample (Positive control) |
86 ±0.01 |
51.35 ± 0.84 |
20.64 ± 0.32 |
|
Quercetin (Standard) |
38 ± 0.005** |
38.83 ± 1.03* |
33.77 ± 0.14* |
|
S-TM |
67 ± 0.005* |
41.48 ± 0.19* |
22.06 ± 0.19 ns |
|
S-H |
72 ± 0.01* |
48.54 ± 0.00 ns |
27.26 ± 0.29* |
|
S-EA |
36 ± 0.15** |
38.15 ± 0.55* |
30.96 ± 0.44* |
|
S-M |
55 ± 00* |
37.06 ± 0.5 * |
27.19 ± 0.38* |
Mean (n=3, mean ± standard deviation) significantly different: *p<0.05, **p<0.01, ***p<0.001, ns: non-significant.
Erythrocytes antioxidant status
Lipid peroxidation (LPO measured in terms of MDA levels) and reduced glutathione (GSH) was measured to assess oxidative damage to erythrocytes by glycated albumin (Table 2). Damage to erythrocytes with glycated sample (positive control, 51.35 ± 0.84 nM MDA/mg protein) was significantly higher as compared to BSA alone (negative control 37.06 ± 0.77 nM MDA/mg protein). The presence of S-TM, S-EA and S-M extracts during glycation showed suppression in erythrocyte LPO. The S-H extract did not demonstrate any considerable effect on LPO levels. Treated erythrocytes incubated in the presence of glycated albumin (positive control) showed a significant reduction in GSH (20.64 ± 0.32 nM/mg protein) as compared with BSA alone (40.34 ± 0.51 nM/mg protein negative control table 1). Glycated samples co incubated with S-TM, S-H, S-EA and S-M showed higher GSH levels as compared to positive control with the S-EA extract displaying the highest GSH levels. Cumulatively, as compared to S-TM (with similar total antiglycation potential), the S-EA extract was most promising in protecting erythrocytes from glycated albumin induced erythrocytes dysfunction measured in terms of % hemolysis, LPO, and GSH level of erythrocytes.
LC−MS data and molecular docking
In order to explore compounds responsible for enzyme inhibition, antioxidant and antiglycation activity of S-EA extract, LC-MS was performed. Only high intensity peaks were annotated for m/z values. A tentative identification of twenty-four substances was made using the molecular formula and precise m/z (Table 3). Major peaks in the negative ion mode represented compounds that could also could be detected and analysed in the positive ion mode. The tentatively identified compounds were categorized into 7 groups as phenolics, alkaloids, flavonoids, phenylethanoid glycosides iridoid glycosides and terpenes (Table 3). These were studied by molecular docking for their possible role as alpha-glucosidase inhibitors. Acarbose, a well-known inhibitor of Maltase-Glucoamylase and Isomaltase was used to compare the binding free energy and molecular interactions of phyto compounds under investigation. Putative alpha-glucosidase inhibitory metabolites not reported previously and displaying better binding affinity than acarbose are presented in Table 4. To identify the interacting amino acid
Table 3: Putative compounds detected in S-EA fraction of P. serratifolia by LC-MS.
|
No.
|
Tentatively |
Retention |
Ion (m/z) |
Experimental |
Theoretical |
Mass Error (ppm) |
Molecular |
Metabolite class |
|
1 |
Casticin |
19.943 |
-ve |
374.0993 |
374.1002 |
-2.21 |
C19 H18 O8 |
flavone |
|
2 |
Centaureidin |
17.891 |
+ve |
360.0833 |
360.0845 |
-3.51 |
C18 H16 O8 |
Flavonoid |
|
3 |
Chrysin |
20.937 |
+ve |
254.0575 |
254.0579 |
-1.51 |
C15 H10 O4 |
Flavonoid |
|
4 |
Diosmin |
14.428 |
+ve |
608.1741 |
608.1741 |
-0.02 |
C28 H32 O15 |
Flavonoid |
|
5 |
Glycitein |
21.137 |
+ve |
284.0677 |
284.0685 |
-2.75 |
C16 H12 O5 |
Flavonoid |
|
6 |
isorhamnetin-3- |
13.684 |
-ve |
478.11 |
478.1111 |
-2.29 |
C22 H22 O12 |
Flavonoid glycoside |
|
7 |
Kaempferide |
19.589 |
+ve |
300.0625 |
300.0634 |
-2.81 |
C16 H12 O6 |
Flavonoid |
|
8 |
kaempferol 3,7-di- |
12.081 |
+ve |
578.1641 |
578.1636 |
0.9 |
C27 H30 O14 |
Flavonoid glycoside |
|
9 |
Luteolin |
18.273 |
+ve |
286.0467 |
286.0477 |
-3.63 |
C15 H10 O6 |
Flavonoid |
|
10 |
Pectolinaringenin |
21.486 |
+ve |
314.0781 |
314.079 |
-3.14 |
C17 H14 O6 |
Flavonoid |
|
11 |
Syringetin |
17.608 |
+ve |
346.0678 |
346.0689 |
-3.06 |
C17 H14 O8 |
Flavonoid |
|
12 |
Tricin |
17.391 |
+ve |
330.0732 |
330.074 |
-2.38 |
C17 H14 O7 |
Flavonoid |
|
13 |
10-O-trans-p- |
3.446 |
-ve |
508.1582 |
508.1581 |
0.19 |
C24 H28 O12 |
Iridoid glycoside |
|
14 |
10-O-trans-p- |
14.844 |
+ve |
522.1725 |
522.1737 |
-2.26 |
C25 H30 O12 |
Iridoid glycoside |
|
15 |
4-hydroxy-E- |
11.764 |
-ve |
526.1669 |
526.1686 |
-3.38 |
C24 H30 O13 |
Iridoid glycoside |
|
16 |
6-O-(3″-O-trans-p- (Premnacorymbo-side B) |
13.279 |
+ve |
654.2156 |
654.216 |
-0.52 |
C30 H38 O16 |
Iridoid glycoside |
|
17 |
Premnaod |
20.476 |
-ve |
890.4138 |
890.4147 |
-1.02 |
C42 H66 O20 |
Iridoid glycoside |
|
18 |
Premnazole |
2.476 |
+ve |
141.0421 |
141.0426 |
-3.33 |
C6 H7 N O3 |
Isoxazole alkaloid |
|
19 |
Caffiec acid |
2.326 |
+ve |
180.0423 |
180.0423 |
0.46 |
C9 H8 O4 |
Phenolics |
|
20 |
acteoside |
14.133 |
-ve |
624.2058 |
624.2054 |
0.6 |
C29 H36 O15 |
Phenylethyl alcohol glycoside |
|
21 |
6-hydroxy salvinolone |
24.97 |
-ve |
330.1827 |
330.1831 |
-1.18 |
C20 H26 O4 |
Terpene (diterpenoid) |
|
22 |
citronellol |
12.847 |
+ve |
154.1354 |
154.1358 |
-2.64 |
C10 H18 O |
Terpene (monoterpenoid) |
|
23 |
Clerodin |
20.587 |
+ve |
434.2298 |
434.2305 |
-1.46 |
C24 H34 O7 |
Terpenes (Diterpenoid) |
|
24 |
Clerodendrin A |
19.31 |
-ve |
606.2685 |
606.2676 |
1.43 |
C31 H42 O12 |
Terpenes (diterpenoid) |
Table 4: Molecular docking scores of compounds against C-terminal- and N-terminal Maltose-Glucoamylase (PDB ID: 3TOP and 2QMJ, repectively) and Isomaltase (PDB ID: 3A4A).
|
Compound |
PDB ID |
Binding Free Energy (kcal/mol) |
Hydrogen bond |
Pi interaction |
|
Acarbose |
3TOP |
-8.19 |
GLN 1372, SER 1366, ASP 1357, ASP 1281, ARG 1285 |
ILE 1587, TYR 1251 |
|
2QMJ |
-7.15 |
ASP 607, TYR 301, ASN 306, GLY 302, ASP 343, GLU 300, ARG 298, ASP 329 |
— |
|
|
3A4A |
-8.94 |
ASP 242, SER 241, TYR 158, ASP 69, HIS 112, ARG 213, ASP 352, GLU 277, GLU 411, HIS 280, ASP 307 |
PHE 303, TYR 158 |
|
|
6-hydroxy salvinolone |
3TOP |
-9.48 |
ASP 1420, ARG 1510, ASP 1157 |
TYR 1251, ASP 1526, TRP 1369, PHE 1560, TRP 1355, PRO 1159, PHE 1559 |
|
2QMJ |
-8.17 |
ASP 203, MET 444 |
ASP 542, TYR 299, PHE 575, TRP 406 |
|
|
3A4A |
-8.93 |
GLN 279, HIS 280, GLU 411 |
ARG 315, TYR 158, PHE 303 |
|
|
10-O-trans-p-coumaroylcatalpol |
3TOP |
-9. 35 |
GLN 1286 |
TYR 1251, TRP 1369 |
|
2QMJ |
-8.08 |
THR 204, THR 205 |
ASP 443 |
|
|
3A4A |
-9.62 |
ASN 415, GLU 422, ASP 307, SER 240, TYR 158 |
PHE 314, ALA 418, LYS 156, TYR 158 |
|
|
10-O-trans-p-methoxycinamoylcatalpol |
3TOP |
-8.65 |
THR 1586, GLY 1588, ARG 1377, TRP 1369, ASP 1420 |
TRP 1418, ILE 1315, PHE 1559, TYR 1251 |
|
2QMJ |
-7.31 |
ASP 443, MET 444, ASP 542 |
— |
|
|
3A4A |
-9.29 |
HIS 423, GLY 160, SER 240, GLU 411 |
ILE 419, PHE 314, TYR 158 |
|
|
4-hydroxy-E-globularinin (B) |
3TOP |
-9.27 |
GLN 1372, ARG 1377, GLY 1365 |
PHE 1559, TYR 1251 |
|
2QMJ |
-8.41 |
ASP 203, THR 205 |
PHE 450, TRP 406, TYR 299, PHE 575 |
|
|
3A4A |
-9.35 |
SER 240, LYS 156, ARG 315, HIS 280, ASP 215, TYR 158 |
VAL 216, ARG 315 |
|
|
Premnacorymbo-side B [6-O-[3-O-trans-p-coumaroyl]-alpha-Lrhamnopyronosylcatalpol |
3TOP |
-8.27 |
ASP 1357, ARG 1377, GLN 1372, TYR 1251, MET 1283, GLN 1254 |
TRP 1355 |
|
2QMJ |
-7.54 |
THR 205, ASP 542, ARG 526, GLN 603 |
TRP 406 |
|
|
3A4A |
-9.86 |
GLN 279, ASP 307, ARG 315, SER 157, LEU 313, LYS 156, ASN 415, SER 157, ILE 419 |
ALA 418 |
|
|
Premnaodoroside B [Bis[(1S)-1alpha-[(β-D-glucopyranosyl)oxy]-1,4aalpha,5,6,7,7aalpha-hexahydro-7alpha-hydroxy-7-methylcyclopenta[c]pyran-4-carboxylic acid]3,7-dimethyloctane-1,8-diyl ester] |
3TOP |
-9.27 |
ASP 1279, ASP 1420, ASP 1526, TRP 1369, MET 1283, GLN 1286, ARG 1377, MET 1421 |
PHE 1560, ILE 1587, TRP 1355 |
|
2QMJ |
-7.33 |
ARG 526, ASP 542, ASP 443, HIS 600, ASP 327, ARG 334 |
PHE 450 |
|
|
3A4A |
-9.0 |
GLN 353, ASP 352, HIS 280, THR 310, TYR 158 |
PHE 303, TYR 158 |
residues and predict the binding modes of the compounds with alpha-glucosidase, 3D and 2D interaction diagrams generated are shown in Supplementary Fig. 1.
Pharmacokinetic properties of selected hits
The solubility values for the putative alpha-glucosidase inhibitory metabolites identified in the present study was within the acceptable range of −6.0 to 0.5 indicating good aqueous solubility property (Table 5). All the compounds displayed human intestinal absorption (HIA) and are not carcinogenic. The calculated oral toxicity for all compounds except Premnaodoroside B fell in category III of acute oral toxicity (LD50 = >500 mg/kg to <5000 mg/kg), indicating that the compounds are non-toxic. 6-hydroxy salvinolone, 10-O-trans-p-methoxycinamoylcatalpol and Premnacorymbo-side B could not penetrate blood brain barrier (BBB) whereas 10-O-trans-p-coumaroylcatalpol, 4-hydroxy-E-globularinin and Premnaodoroside B could penetrate BBB.
Table 5: ADMET properties of selected putative lead compounds
|
Ligand |
Solubility |
HIA |
BBB |
Carcinogenicity |
Acute oral Toxicity category |
|
6-hydroxy salvinolone |
-4.1426 |
+ |
– |
– |
III |
|
10-O-trans-p-coumaroylcatalpol |
-2.276 |
+ |
+ |
– |
III |
|
4-hydroxy-E-globularinin |
-2.021 |
+ |
+ |
– |
III |
|
10-O-trans-p-methoxycinamoylcatalpol |
-2.188 |
+ |
– |
– |
III |
|
Premnaodoroside B |
-3.960 |
– |
+ |
– |
I |
|
Premnacorymbo-side B |
-2.284 |
+ |
– |
– |
III |
HI: human intestinal absorption, BBB: blood brain barrier
Discussion
In recent years, nutritional intervention using dietary plants which are free from adverse side effects are gaining increasing importance in controlling diabetes and its consequences. Hence, antidiabetic herbs used in traditional medicines are being evaluated for their antidiabetic (hypoglycemic, antioxidant, anti-inflammatory, antiglycation and protection from glycation induced cellar damage) potential. Many in vivo as well as in vitro studies have proved the effectiveness of P. serratifolia, an important nutraceutical food used in Indian Ayurveda, as an antidiabetic plant. 17 The present study, to the best of our knowledge, additionally provides the first scientific evidence of its antiglycation and cell protective potential. Further, we also report the plant metabolites which need further studies to assess their protective role against glycation and glycation induced cellular dysfunction.
Although few reports have demonstrated that leaf extracts of P. serratifolia showed inhibitory effect on alpha-glucosidase no study has comparatively reported this activity from the stem.18-20 Results of in vitro assays showed that the hexane extract prepared from leaves (L-H) and stem (S-H) exhibited a stronger inhibition of alpha-glucosidase as compared to the standard acarbose. Recently, GC/MS analysis of hexane leaf extract of P. serratifolia identified 9-octadecenoic acid, palmitic acid, phytol and stearic acid as major volatile organic compounds.32 Of these, stearic acid, palmitic acid and 9-octadecenoic acid, and are known potent inhibitors of alpha-glucosidase.33-35 The presence of these compounds in hexane extract may have been responsible for the observed activity. In agreement to previous reports, in the present study also extracts prepared from leaves of P. serratifolia (L-TM, L-EA and L-M) had a lesser inhibitory effect on alpha-glucosidase as compared to acarbose.19 However, the extracts prepared from stem had a more potent alpha-glucosidase inhibitory potential with the S-EA extract more potent than acarbose in inhibiting alpha-glucosidase.
In the present study, results indicate that in P. serratifolia, TPC and TFC differ significantly with the solvent and plant part used for extraction. The fractionated S-EA extract had a the highest TPC and TFC. Results agree with previous studies which have reported that in some medicinal plants like Amphimas pterocarpoides and Phyllanthus emblica extracts prepared from stem contained higher TPC and TFC as compared to those prepared from leaves. 36, 37
Phenolic and flavonoid are phyto compounds in which the aromatic ring bears at least one hydroxyl group responsible for free radical scavenging. Phenolics and flavonoids with more than one hydroxyl group on the aromatic ring are more active antioxidants than those with fewer hydroxyl groups. With both leaves and stem, the EA extracts had highest TAA and RP than TM, H and M extracts. Among the stem extracts, S-EA with highest TPC and TFC displayed the highest TAA and RP. Among the leaf extracts tested, the L-TM had the highest TPC and TFC but L-EA extract showed the highest TAA and RP. This indicates that although the L-EA extract had lesser TPC and TFC, it possibly contained phenolics and flavonoids with higher antioxidant potential. Thus, in P. serratifolia, ethyl acetate appears to be a more suitable solvent to recover potent antioxidants from leaves as well as stem wood. Previous studies have reported variation in TAA and RP with plant part and solvent used for extraction. 38
In biological cells, ROS are produced continuously. Antioxidant system of the cell can neutralize these free radicals. However, in many chronic disorders including diabetes mellitus the generation of antioxidants in cell is decreased leading to increase in ROS and resulting oxidative injury to biomolecules. 39 Natural dietary antioxidants found in medicinal plants have the ability to prevent or reduce the interaction of biological molecules with ROS. The DPPH and ABTS represent methods for assessing antioxidant activity of plant extracts based on the scavenging activity toward stable free radicals. These chemical assays are based on mixed tests and involve reactions which can be a hydrogen atom transfer (HAT) or a single electron transfer (SET). On the other hand, in vitro analytical methods like RP and TAA are SET based and relay on the capacity of the antioxidant to reduce compounds by transferring an electron.39 A high correlation (R=0.61, p<0.001) found between DPPH and ABTS in the present study is not surprising as both rely on the same mechanism, the mixed test. Interestingly, with both DPPH and ABTS, the extracts prepared from leaves (Fig. 2 a & b) were more potent in scavenging free radicals as compared to extracts prepared from stem which displayed higher TAA and RP (Fig. 3). The L-TM extract was the most potent with an IC50 value less than standard in both DPPH and ABTS. Results indicate that the S-EA extract (displaying highest TAA and RP) from P. serratiofolia was more suited to extract natural antioxidants that could reduce molecules by SET while the L-TM extract with obviously more phytochemicals contained antioxidants that acted using both HAT and SET. Many studies have reported variation in antioxidant activity values quantified through DPPH, RP, ABTS and FRAP depending on extraction solvent.40
Structural and functional modifications of proteins by glycation leads to diabetic complications. Increasing research has reported that plant metabolites with potent antioxidant potential exhibit strong antiglycating activity.3 Hence, in the present study antioxidant potential was tested by multiple assays. All the extracts prepared from leaves and stem exerted a noticeable total antiglycation potential (Fig. 4e). Interestingly, the EA extract from leaves as well as stem with higher TAA and RP were most potent in inhibition of fructosamine, reducing carbonyl levels and increasing free thiol groups. Results are in agreement to previous studies reporting RP (compared to DPPH, ABTS) as the assay with highest sensitivity to assess antioxidant potential of extracts.40 The highest inhibition of β-amyloid aggregations belonged to TM extracts of leaf and stem. Thus, the bioactive metabolites present in EA extract were more effective in attenuating glycation by blocking the early glycation product (fructosamine) with subsequent reduction in the generation of reactive carbonyl or dicarbonyl and restoring oxidatively modified amino acids. However, the TM demonstrated a strong prevention of amyloid aggregation, suggesting that it has a greater capacity to stop albumin structural changes during glycation.
Diabetes has been linked to glycated albumin-mediated erythrocyte dysfunction. The present study evaluated the protective effect of stem extracts from P. serratifolia in glycated albumin induced erythrocytes dysfunction. In the presence of glycated sample, erythrocytes exhibited increased haemolysis (86 ±0.001%) and oxidative damage (increased LPO and decrease in GSH levels) when compared to negative control (BSA, Table 2). During glycation in the presence of plant extracts, the toxicity as well as oxidative damage of glycated albumin to erythrocyte was reduced. The EA extract was most effective in reducing glycated albumin induced erythrocyte haemolysis Further, the EA extract was also effective in preventing erythrocyte oxidative damage measured in terms of reduction in LPO and concomitant increase in GSH levels (Table 2). Recently, Singh et al. 41 reported that EA extracts of P. serratifolia could significantly attenuated oxidative stress and antioxidant markers (LPO, GSH, catalase and superoxide dismutase enzymes). Although the TM and EA extracts were equally effective in attenuating glycation, the EA extract displayed a higher potential in improving viability and antioxidant status of erythrocytes damaged by glycated albumin. This may be attributed to its high antioxidant potential measured by TAA and RP as well as its highest TPC and TFC.
LC-MS based metabolomics of S-EA extract responsible for most promising alpha-glucosidase inhibition, antiglycation and cell protective activity from stem of P. serratifolia identified flavonoids, iridoid glycosides and terpenes as major metabolites in agreement to previous reports (Table 3).17 From in silico study, 6-hydroxy salvinolone and iridoid glycosides 10-O-trans-p-coumaroylcatalpol (10OCC), 10-O-trans-p-methoxycinamoylcatalpol, 6-O-(3-O-trans-p-coumaroyl)-alpha-Lrhamnopyranosylcatalpol (Premnacorymbo-side B), 4-hydroxy-E-globularinin (B), Premnaodoroside B, known to be present in stem bark of P. serraatifolia but not previously docked with Maltose-Glucoamylase and Isomaltase have been highlighted. 42-44 The complexes of C-terminal- and N-terminal Maltose-Glucoamylase with 6-hydroxy salvinolone had the best binding affinity. This anti-inflammatory and immunomodulatory compound isolated from roots of P. serratifolia and P. obtusifolia has been postulated as a lead molecule for the development of COX-LOX dual inhibitors.43 It also exhibits cytotoxic activities against cancerous cell line. No previous reports have predicted the possible role of 6-hydroxy salvinolone as an inhibitor of alpha-glucosidase by molecular docking. Based on binding energy, hydrogen bonds, hydrobhobic and Pi interactions, iridoid glycosides had more potential as alpha-glucosidase inhibitors compared to the control ligand (Table 4 and Supplementary Fig. 1). These compounds warrant further purification and experimental validation to confirm their inhibitory activity.
Conclusion
Traditionally, infusion or decoction in water prepared from P. serratifolia is used as antidiabetic food or drink additive. The possible presence of polar metabolites with alpha-glucosidase inhibitory potential identified in the present study supports the traditional use of P. serratifolia as an antibiabetic food. Further investigations involving the purification of observed metabolites, especially the polar iridoid glycosides, to test their ability in ameliorating the oxidative status, glycation and inhibit alpha-glucosidase are imperative and underway.
Acknowledgement
The authors are grateful to Smita Godrej Crishna, Director, and Dr. Kranti Yardi, Head, NGCPR for providing support.
Funding source
Funds were provided by Depart. of Biotechnology, Govt. of India, Ministry of Science and Technology vide file no. BT/INF/22/SP41297/2021 (DBT BUILDER).
Conflict of interest
The author(s) do not have any conflict of interest.
Data Availability
This statement does not apply to this article
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Author contributions
- Sagar Bhayye: In silico data Collection and analysis
- Rakhee Dangi: Conceptualization, Methodology, Data Collection, Analysis, Writing of MS, Funding Acquisition, Resources and Supervision.
- Sagar Datir: Collection and identification of plant material, LC-MS analysis.
References
- Dirir M, Daou M, Yousef F and Yousef F. A review of Alpha-Glucosidase Inhibitors from Plants as Potential Candidates for the Treatment of Type-2 Diabetes. Rev. 2022; 21(4): 1049-1079. doi: 10.1007/s11101-021-09773-1.
CrossRef - Salazar-García M and Corona C. The Use of Natural Compounds as a Strategy to Counteract Oxidative Stress in Animal Models of Diabetes Mellitus. J. Mol. Sci. 2021; 22(13): 7009. doi: 10.3390/ijms22137009.
CrossRef - Gaonkar N and Dangi R. Comparative Evaluation of Antioxidant, Antiglycation and alpha-glucosidase Inhibitory Potential of Some Indigenous Medicinal Trigonella Vegeto.2022; 36: 1337–1346.
CrossRef - Yamagishi S, Maeda S, Matsui T, Ueda S, Fukami K and Okuda S. Role of Advanced Glycation End Products (AGEs) and Oxidative Stress in Vascular Complications in Diabetes. Biophys. Acta. 2012; 5: 663–671. https://doi.org/10.1016/j.bbagen.2011.03.014.
CrossRef - Nowotny K, Jung T, Höhn A, Weber D and Grune T. Advanced Glycation End Products and Oxidative Stress in Type 2 Diabetes Mellitus. Biomolecules. 2015; 5(1): 194–222. doi: 10.3390/biom5010194.
CrossRef - Dudonné S, Vitrac X, Coutière P, Woillez M and Mérillon M. Comparative Study of Antioxidant Properties and Total Phenolic Content of 30 Plant Extracts of Industrial Interest using DPPH, ABTS, FRAP, SOD, and ORAC Assays. J Agric Food Chem. 2009; 57(5): 1768-74. doi: 10.1021/jf803011r.
CrossRef - Song Q, Liu J, Dong L, Wang X and Zhang X. Novel advances in inhibiting advanced glycation end product formation using natural compounds. Pharmacother. 2021; 140:111750. https:// doi: 10.1016/j.biopha.2021.111750.
CrossRef - Khalisha K, Febriyanti M and Muhaimin M. 2022. Phytochemistry and Pharmacological Potentials of Premna serratifolia: Traditional Medicinal Plant Used by Local People in Kalimantan. Indones. J. Biol. Pharm. 2022; 2(3):178. http://dx.doi.org/10.24198/ijbp.v2i3.43537
CrossRef - Mali Y. Pharmacological Potentials of Premna integrifolia Anc. Sci. Life. 2016; 35(3): 132–42. doi: 10.4103/0257-7941.179864.
CrossRef - Francis S. Gideon A, Britto J and Dessy A. Review on Two Endemic Species of Genus Premna and their Conservational Importance. Drug Delivery Ther. 2019; 9(4-s): 666-9. https://jddtonline.info/index.php/jddt/article/view/3330
CrossRef - Hutapea M, Sinaga R and Hutapea M. Antihyperglycemic Effect Premna serratifolia in Attenuating Postprandial Hyperglycemia in Healthy Subjects When Consumed Simultaneously with Glucose. Presented at 11th International Scholars Conference. 2018; 6(1): 93. https://doi.org/10.35974/isc.v6i1.1393
CrossRef - Majumdar R, Akter S and Badrul A. Antioxidant and Anti-Diabetic Activities of the Methanolic Extract of Premna integrifolia Adv. Biol. Res. 2014; 8 (1): 29–36.
- Dash K, Patro C and Maiti A. study on the anti-hyperglycaemic effect of Premna corymbosa Roots. J. Nat. Rem. 2005; 5: 31–34.
- Belinda A, Febriyanti R and Muhaimin. Chemical and Pharmacological Properties of Selected Medicinal Plant Species from Genus Premna and their Immunomodulatory Potentials. J. App. Pharm. 2022; 14(5): 24-31. https://doi.org/10.22159/ijap.2022.v14s5.30.
CrossRef - Gokani H, Lahiri K, Santani D and Shah B. Evaluation of Anti-inflammatory and Antioxidant Activity of Premna integrifolia J. Complement. Integr. Med. 2011; 8. https://www. degruyter.com/view/j/jcim.2011.8.
CrossRef - Harikrishnan R and Beevy S. Phytochemical Analysis and in vitro Antioxidant Activities of Three Species of Premna L. from Kerala. Asian J. Biol. Sci. 2023; 12. https://doi.org/10.5530/ajbls.2023.12.70.
CrossRef - Dianita R and Jantan I. Ethnomedicinal uses, Phytochemistry and Pharmacological Aspects of the genus Premna: A Review. Biol. 2017; 55(1):1715–1739. doi: 10.1080/13880209.2017.1323225.
CrossRef - Timotius K, Imamora A and Santoso W. Chemical Characteristics and In vitro Antidiabetic and Antioxidant Activities of Premna serratifolia Leaf Infusion and Decoction. Pharmacog. J. 2018; 18: 1114-1118. Doi.10.5530/pj.2018.6.189
CrossRef - Simamora A, Santoso W, Timotius H and Rahayu Antioxidant Activity, Enzyme Inhibition Potentials, and Phytochemical Profiling of Premna serratifolia L. Leaf Extracts. Int. J. Food Sci. 2020: 3436940. https://doi.org/10.1155/2020/3436940
CrossRef - Hadiarti D, Haryadi W, Matsjeh S, Swasono T and Awaliyah N. Profiling of alpha-Glucosidase Inhibitors from Ethyl Acetate Fraction of Buas-buas (Premna serratifolia) leaves using UHPLC-Q-orbitrap HRMS and Protein–Ligand Interaction with Molecular Docking. Appl. Pharm. Sci. 2023; 13(02): 089–098. https://doi.org/10.7324/JAPS.2023.130210
CrossRef - Brand-Williams W, Cuvelier E and Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT Food Sci. Technol. 1995; 28: 25–30
CrossRef - Re R, Pellegrini N, Proteggente A, Pannala A, Yang M and Rice-Evans C. Antioxidant Activity Applying an Improved ABTS Radical Cation Decolorization Assay. Free Biol. Med. 1999; 26: 1231-1237. doi: 10.1016/s0891-5849(98)00315-3. PMID: 10381194.
CrossRef - Benzie I and Strain J. The Ferric Reducing Ability of Plasma (FRAP) as a Measure of “Antioxidant Power”: The FRAP Assay. Biochem. 1996; 239: 70–76. https://doi.org/10.1006/abio.1996.0292.
CrossRef - Baker R, Zyzak V, Thorpe R and Baynes W. Chemistry of the Fructosamine Assay: D-glucosone is the Product of Oxidation of Amadori Compounds. Chem. 1994; 40: 1950–1955.
CrossRef - Uchida M, Kanematsu K and Sakai T. Protein-bound Macrolein: Potential Markers for Oxidative Stress (covalent modification of protein/antibody/atherosclerosis). Natl. Acad. Sci. 1998; 95: 4882–4887.
CrossRef - Ellman GL. Tissue sulfhydryl groups. Biochem. Biophys. 1959; 82: 70–77. doi: 10.1016/0003-9861(59)90090-6.
CrossRef - Luize S, Tiuman S, Morello G, et al. Effects of Medicinal Plant Extracts on Growth of Leishmania (l.) amazonensis and Trypanosoma cruzi. J. Pharm. Sci. 2005; 41: 85–94. https://doi.org/10.1590/S1516-93322005000100010.
CrossRef - Placer A, Cushman L and Johnson C. Estimation of Product of Lipid Peroxidation (malonyl dialdehyde) in Biochemical Systems. Biochem. 1966;16: 359–364. doi: 10.1016/0003-2697(66)90167-9.
CrossRef - Eberhardt J, Santos-Martins D, Tillack F and Forli S. AutoDock Vina 1.2.0: New Docking Methods, Expanded Force Field, and Python Bindings. Chem. Inf. Model. 2021; doi: 10.1021/acs.jcim.1c00203
CrossRef - Schrödinger L and DeLano W. PyMOL. 2020. Retrieved from http://www.pymol.org/pymol. (accessed on April 14, 2025).
- Daina A, Michielin O and Zoete V. SwissADME: a free web tool to evaluate Pharmacokinetics, Drug-likeness and Medicinal Chemistry Friendliness of Small Molecules. Rep. 2017; 7:42717. doi: 10.1038/srep42717 (accessed on April 14, 2025).
CrossRef - Rahman A, Siddiqui A, Altun M and Demirtas I. Premna integrifolia Linn: Phytochemical Profile and Evaluation of Antiproliferative Effects. select. 2023: 8(9), e202204854. https://doi.org/10.1002/slct.202204854
CrossRef - Cherigo L and Martínez-Luis S. Alpha-Glucosidase Inhibitor Isolated from Blechum pyramidatum. Nat Prod Commun. 2018; 13: 461-64. https://doi.org/10.1177%2F1934578X1801300421.
CrossRef - Nguyen H and Kim M. Alpha-Glucosidase Inhibitory Activities of Fatty Acids Purified from the Internal Organ of Sea Cucumber Stichopus japonicas. Food Sci. 2015, 8(4), H841-7. doi: 10.1111/1750-3841.12810.
CrossRef - Li X, Bai Y, Jin Z and Svensson B. Food-derived Non-phenolic alpha-amylase and alpha-glucosidase Inhibitors for Controlling Starch Digestion Rate and Guiding Diabetes-friendly Recipes. 2022, 153: 112455. https://doi.org/10.1016/j.lwt.2021.112455.
CrossRef - Asante-Kwatia E, Adjei S, Jibira Y, et al. Amphimas pterocarpoides: An Evaluation of Flavonoid and Phenolic Contents, Wound Healing, Anthelmintic and Antioxidant Activities of the Leaves and Stem Bark. Heliyon. 2021; 7: e08261. https://doi.org/10.1016/j.heliyon.2021.e08261.
CrossRef - Nurlaela F, Lia A, Yessi, F and Emi S. Correlation of Total Phenolic, Flavonoid and Carotenoid Content of Phyllanthus emblica Extract from Bandung with DPPH Scavenging Activities. J. 2018; 10(3): 447-452.
CrossRef - Shah M, Seelan S and Iqbal M. Phytochemical Investigation and Antioxidant Activities of Methanol Extract, Methanol Fractions and Essential Oil of Dillenia suffruticosa Arab J. Chem. 2020; 13(9): 7170-7182. https://doi.org/10.1016/j.arabjc.2020.07.022.
CrossRef - Siddeeg A, AlKehayez N, Abu-Hiamed H, Al-Sanea E, AL-Farga A. Mode of Action and Determination of Antioxidant Activity in the Dietary Sources: An overview. Saudi J. Bio. Sci. 2012; 28: 1633-1644. https://doi.org/10.1016/j.sjbs.2020.11.064.
CrossRef - Chaves N, Santiago A and Alías C. Quantification of the Antioxidant Activity of Plant Extracts: Analysis of Sensitivity and Hierarchization Based on the Method Used. 2020; 9(1): 76. doi: 10.3390/antiox9010076.
CrossRef - Singh C, Prakash C, Tiwari N, Mishra K and Kumar V. Premna integrifolia Ameliorates Cyclophosphamide-induced Hepatotoxicity by Modulation of Oxidative Stress and Apoptosis. pharmacothe. 2018; 107: 634-643 https://doi.org/10.1016/j.biopha.2018.08.039
CrossRef - Yadav D, Tiwari N and Gupta M. Diterpenoids from Premna integrifolia. Lett. 2010; 3: 143-147.
CrossRef - Azad R, Babu K, Gupta D and Reddanna P. Evaluation of Anti-inflammatory and Immunomodulatory Effects of Premna integrifolia Extracts and Assay-guided Isolation of a COX-2/5-LOX dual inhibitor. 2018; 131:189-199. doi: 10.1016/j.fitote.2018.10.016.
CrossRef - Yadav D and Gupta M. Isolation and HPTLC Analysis of Iridoids in Premna integrifolia, an Important Ingredient of Ayurvedic Drug Dashmool. JPC-J. Planar Chromat. 2013; 26: 260–266. https://doi.org/10.1556/JPC.26.2013.3.10.
CrossRef
Abbreviations List
ABTS: 2,2′-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid (ABTS)
AGEs: advanced glycation end products (AGEs).
DPPH: 2,2-diphenyl-1-picrylhydrazyl
FRAP: Ferric Reducing Antioxidant Power Assay
GSH: reduced glutathione
IC50: Concentration required for 50% enzyme inhibition
LC-MS: Liquid Chromatography-Mass Spectrometry
LPO: Lipid peroxidation
mg GAE/g DE: mg of gallic acid equivalents per gram of dried extract
mg QE/g DE: mg of quercetin equivalents per gram of dried extract
ROS: reactive oxygen species
RP: Reducing potential
TAA: Total antioxidant activity
TFC: Total flavonoid contents
TPC: Total phenolic contents







