Manuscript accepted on :16-03-2026
Published online on: 24-03-2026
Plagiarism Check: Yes
Reviewed by: Dr. Abdulrahman Rasheed Mahmood and Dr. Murali Krishna Prasad Vallabhaneni
Second Review by: Dr. Hassan Shora
Final Approval by: Dr. Patorn Piromchai
Rania Kadhim1 , Emad Yousif1*
, Emad Yousif1* , Suhad Ibrahim1
, Suhad Ibrahim1 and Muna Bufaroosha2
and Muna Bufaroosha2
1Department of Chemistry, College of Science, Al-Nahrain University, Baghdad, Iraq
2Department of Chemistry, College of Science, United Arab Emirates University, Al-Ain, United Arab Emirates
Corresponding Author E-mail: yousif@nahrainuniv.edu.iq
DOI : https://dx.doi.org/10.13005/bpj/3346
Abstract
Breast cancer is the second leading cause of cancer-related deaths worldwide after lung cancer, making early diagnosis and continuous treatment crucial. This study evaluated Lactate dehydrogenase (LDH) enzyme levels and other biochemical parameters in post-operative chemotherapy in Iraqi women diagnosed with breast cancer. This study highlights the potential use of lactate dehydrogenase (LDH) as s biomarker for monitoring treatment response, rather than being limited to its traditional diagnostic role. In this investigation, a total of 150 serum samples were analyzed. To assess the prognostic value of LDH along with other biochemical markers including ALP, ALT, AST, electrolytes (Na⁺, K⁺, Cl⁻), kidney functions (urea, creatinine), and prolactin hormone. A cross-sectional study was conducted on a total of 150 serum samples collected from 100 female patients diagnosed with breast cancer (after surgery and during chemotherapy) and 50 age-matched healthy women as a control group, aged between 25–60 years. All biochemical parameters (LDH, ALP, ALT, AST, Na⁺, K⁺, Cl⁻, urea, creatinine, and prolactin) were measured in the laboratory using standard automated biochemical analyzers and commercially available diagnostic kits. LDH activity was significantly elevated in breast cancer patients, highlighting its potential as an early prognostic marker for disease progression or recurrence. Renal function tests (urea and creatinine) and liver enzymes (ALT and AST) were significantly higher in patients compared to controls (p < 0.05). Electrolytes, specifically K⁺ and Cl⁻, were elevated (p < 0.05), while Na⁺ showed no significant difference (p > 0.05). ALP levels were not significantly different between patients and controls (p > 0.05). Prolactin hormone levels were also significantly higher in patients (p < 0.05). These findings underscore LDH as the central prognostic marker, with other biochemical parameters providing supportive information on patient status. Furthermore, the results indicate that LDH activity in breast cancer patients was markedly higher than that observed in healthy controls, which may be attributed to increased cellular proliferation and metabolic activity in cancer cells. Cancer cells rely heavily on aerobic glycolysis to meet their energy demands, and LDH plays a critical role in this metabolic pathway. Elevated LDH levels may therefore serve as an early prognostic biomarker for metastasis or disease recurrence and may also be useful for monitoring disease progression and therapeutic response during chemotherapy.
Keywords
Breast Cancer; Chemotherapy Monitoring; Electrolyte Imbalanc; Lactate Dehydrogenase; Liver Enzymes; Prognostic Biomarker; Prolactin Hormone; Renal Function
Download this article as:| Copy the following to cite this article: Kadhim R, Yousif E, Ibrahim S, Bufaroosha M. Lactate Dehydrogenase and Biochemical Markers as Indicators of Treatment Response in Breast Cancer. Biomed Pharmacol J 2026;19(1). |
| Copy the following to cite this URL: Kadhim R, Yousif E, Ibrahim S, Bufaroosha M. Lactate Dehydrogenase and Biochemical Markers as Indicators of Treatment Response in Breast Cancer. Biomed Pharmacol J 2026;19(1). Available from: https://bit.ly/3NYwug2 |
Introduction
The human breast is a complex glandular organ composed mainly of ductal and lobular structures embedded within adipose and connective tissues, along with vascular and lymphatic components. These structures are supported by stromal tissue and epithelial cell populations that form the functional units of the breast.¹–²
Breast development begins at puberty and continues through adolescence and adulthood with progressive lobular differentiation and glandular maturation, particularly during pregnancy. Hormonal changes, including progesterone and human chorionic gonadotropin, play an important role in promoting glandular growth and functional development of the mammary gland. ³–⁵
Cancer is characterized by uncontrolled cell growth leading to the formation of tumors, which may be benign—localized and slow-growing—or malignant, with the ability to invade and spread.6 Breast cancer is among the most common malignant tumors worldwide, with over two million new cases annually.7 Several risk factors, including obesity, physical inactivity, and alcohol consumption, lobal economic growth and the rising participation of women in the workforce have contributed to notable shifts in lifestyle patterns, social structures, and environmental exposures.8,9 These transformations have influenced the distribution of established breast cancer risk factors across populations. Consequently, the gap in breast cancer incidence between high-income and low- to middle-income countries is gradually narrowing. Reproductive patterns such as postponing childbirth or having fewer children, combined with sedentary behavior and increased rates of overweight and obesity, have been linked to a higher likelihood of disease development.10,11
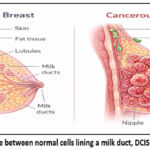 |
Figure 1: The difference between normal cells lining a milk duct, DCIS and invasive cancer.12 |
BC is characterized by a long doubling time of cancer cells (100–300 days), which provides a valuable opportunity for early detection and treatment. The classification of BC Staging is determined by evaluating the dimensions of the primary tumor, the extent of regional lymph node involvement, and whether distant metastatic spread has occurred.12
Stage 0: The tumor is confined to the site of origin. Stage 1: The tumor spreads into the fatty tissue of the breast. Stage 2: The tumor increases in size and may extend further within the breast. Stage 3: The tumor involves nearby lymph nodes and the chest wall. Stage 4 (Metastatic stage): The tumor spreads to organs beyond the primary site, such as bones, hepatic and pulmonary sites and brain.13,14
Early diagnosis, adjuvant chemotherapy, hormonal therapy, and radiotherapy play a major role in the treatment of BC and in reducing disease-related risks.15 Both estrogen (ER) and progesterone (PR) receptors are considered predictive factors in BC. Radical mastectomy was applied in the early twentieth century but proved less efficient compared to less severe surgical approaches. Chemotherapy, endocrine therapy, and anti-HER2 targeted therapy have demonstrated high efficacy in reducing BC risks and improving survival.16 BC is a heterogeneous disease, and in cases of small tumors, locoregional treatment may be sufficient, whereas cancer that has spread to distant sites requires additional therapy. The main goals of treatment are surgical resection, sampling, axillary lymph node removal, followed by radiation.17
Breast cancer represents a growing health and economic burden on healthcare systems, particularly in developing countries, where limited access to early screening programs and insufficient public awareness often lead to diagnosis at advanced stages. This situation negatively affects survival rates and patients’ quality of life while substantially increasing the cost and duration of medical care. In this context, blood-based biomarkers have emerged as promising tools for the diagnosis and monitoring of breastcancer, 2 as they are non-invasive, cost-effective, and easily repeatable compared to tissue biopsies. These biomarkers provide valuable real-time information about the functional and metabolic status of tumor cells, thereby facilitating dynamic assessment of disease progression and treatment response. Recent studies have demonstrated that metabolic reprogramming is a hallmark of cancer cells, enabling them to rely on alternative biochemical pathways to fulfill their elevated bioenergetic and anabolic requirements. Such metabolic adaptations enhance tumor cell survival under hypoxic and oxidative stress conditions and contribute to increased tumor aggressiveness and metastatic potential. Accordingly The current investigation was designed to highlight biomarkers belonging to selected in breast cancer and to correlate them with patients’ clinical characteristics, supporting their use as complementary tools for diagnosis, monitoring, and evaluation of therapeutic response.6
To better understand disease progression and treatment response, biomarker analysis has become an essential tool. Histological grade, obtained via core needle biopsy (CNB), axillary status, and tumor size are important parameters in BC. ER-alpha is a key predictive factor for advanced-stage patients responding to hormone therapy, which is non-toxic and suitable for long-term use.18 Cancer antigen CA15.3 is elevated pre-surgically and serves as an important marker for evaluating response after chemotherapy.19
Among emerging biomarkers, lactate dehydrogenase (LDH) has gained attention due to its strong association with BC progression and early detection.20 LDH regulates the dynamic equilibrium between pyruvate and lactatem, playing a crucial role in anaerobic metabolism and maintaining NAD+ levels for glycolysis.21 It is found in heart and muscle tissues and exhibits high activity and stability, making it a central enzyme in the glycolytic pathway and a potential therapeutic target.22 Elevated LDH levels are observed in a variety of disorders, including cardiac, hepatic, skeletal muscle, and renal diseases, as well as leukemia.23-25 In cancer, high LDH levels reflect altered metabolism that supports tumor growth and metastasis, establishing its value as a diagnostic and prognostic biomarker.26 Lactate dehydrogenase (LDH) plays a central role in cellular metabolism by regulating the reversible conversion between pyruvate and lactate., playing a key role in maintaining NAD+ levels for glycolysis. In cancer cells, LDH contributes to the “Warburg effect,” a metabolic shift where glucose is preferentially metabolized to lactate even in the presence of oxygen, supporting rapid tumor growth and survival.27-31. Mitochondrial dysfunction in tumor cells leads to increased membrane permeability and the release of intracellular enzymes, including LDH, which correlates with abnormal apoptotic signaling.32,33 Elevated LDH levels in serum thus reflect altered cellular metabolism and tumor progression, highlighting its value as a diagnostic and prognostic biomarker in breast cancer.hypertension, ischemic heart disease, thyroid disorders, as well as smokers or alcohol consumers, were excluded. Verbal Participants gave their informed consent in advance of sample collection. The present study focused on evaluating biochemical parameters including LDH activity, liver enzymes, renal function markers, and serum electrolytes in breast cancer patients in order to assess metabolic and physiological alterations associated with the disease. Therefore, tumor markers such as CA15-3 were not included within the scope of the current investigation.
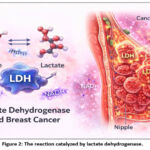 |
Figure 2: The reaction catalyzed by lactate dehydrogenase. |
Experimental Materials and Procedures
Clinical Study Population
This cross-sectional investigation enrolled 150 Iraqi women aged 25–60 years who were categorized into two groups. Blood samples were collected in Baghdad city from patients attending Al-Amal Hospital. The sample collection was conducted over a period of eight months. The first group included 50 women with no history of cancer or other chronic diseases, serving as the control group. The second group included 100 women diagnosed with breast cancer who were enrolled at the beginning of their chemotherapy and before undergoing radiotherapy. The diagnosis was confirmed through medical record review. Ethical approval for the study was obtained from the Ethics Committee of the University. Patients with chronic diseases such as diabetes mellitus were excluded from the study.
Specimen Procurement and Preparation
A volume of 5 mL of peripheral blood was obtained from each individual collected from each participant and placed in gel tubes. Samples from breast cancer patients were collected at different time points relative to chemotherapy including before starting chemotherapy, after the first session, or after two to three sessions. Samples were kept at standard laboratory temperature until blood clotting occurred, after which they were centrifuged at 3000 rpm for 15 minutes. The resulting sera were transferred into Eppendorf tubes and stored at −20 °C until biochemical analysis was performed.
All biochemical analyses, including LDH, ALT, AST, ALP, prolactin, electrolytes, and kidney function tests, were performed using automated analyzers available in the laboratory. Data distribution was tested using Cobas C111 tests, and multifactorial analysis was used to control for potential confounding variables such as age, disease stage, and treatment type. Statistical analyses were interpreted using a significance level set at p < 0.05.
Analytical Approach
All statistical evaluations were carried out using GraphPad Prism (v8.0.2). Prior to further analysis, data normality was examined using both the Shapiro–Wilk and Kolmogorov–Smirnov tests. Numerical variables are presented as mean values accompanied by their standard deviations unpaired t-test was used to compare normally distributed data between study groups. Multifactorial analysis was applied to control for potential confounding variables, including age, disease stage, and treatment type. A p-value below 0.05 was regarded as statistically significant
Findings and Discussion
Serum Lactate Dehydrogenase Evaluation (LDH)
LDH enzyme has a crucial role in the glycolysis by converting the pyruvate to lactate in the end of this process. The release of high levels of LDH into the blood is related with several diseases such as cancers causes destruction the tissue.34.35 The LDH levels in postoperative patients suffering from breast cancer as well as the healthy individuals are shown in Summary of Study Variables (1) and figure (3) which explained that the LDH levels in the patients, were significantly high (346.73±74.13) with a p-value (<0.0001) when comparing it with that for the control (121.90±28.92)). The LDH activity in the patients with breast cancer was higher compared with that in the controls and this may be because of the increasing in the cell growth and its migration comparing with that in normal cells. Additionally, cancer cells have high energy demands and rely on aerobic glycolysis (Warburg effect), mitochondrial dysfunction, increased cellular permeability, and chemotherapy-induced inflammation, all of which contribute to elevated LDH levels in the serum. The cancer cells require excess of energy so the LDH can be considered an alternative to meet the metabolic and the aerobic glycolysis needs of cancer cells. In patients with breast cancer, the high levels in LDH activity can give a prognostic indicator for the early warning of metastasis or recurrence it.36, 37
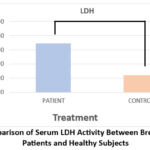 |
Figure 3: Comparison of Serum LDH Activity Between Breast Carcinoma Patients and Healthy Subjects |
Table 1: Serum LDH levels for breast cancer patients and healthy control
| GROUP | MEAN ±SD (U/L) | P value |
| CONTROL | 121.90±28.92 | <0.0001 |
| PATIENT | 346.73±74.13 |
Serum LDH levels were markedly elevated in breast cancer patients (346.73 ± 74.13 U/L) compared with the healthy control group (121.90 ± 28.92 U/L). Statistical analysis revealed a highly significant difference between the two groups (p < 0.0001). The increased LDH activity observed in patients may be related to enhanced metabolic processes and rapid proliferation of cancer cells.
Evaluation of serum Urea and Creatinine Levels:
The urea cycle reactions are important for removing free ammonia by hepatic detoxification and expelling it via the urethra, so urea levels are particularly important in patients exposed to diseases or treatments that can impair kidney function.38 In this work, both the urea and creatinine levels were evaluated for the breast cancer patients, and the results are shown in table (2) and figure (4), illustrating significantly higher levels of urea in breast cancer patients (89.91±24.66) compared with controls (29.81±9.53p < 0.0001). Creatinine levels were also elevated (2.195±0.60414vs. 0.7002±0.1399 in controls, p < 0.0001).
Elevated urea levels in breast cancer patients may be attributed to not only tissue destruction and nucleic acid turnover but also to chemotherapy-induced renal stress, inflammation, and altered metabolism, which can affect nitrogen balance. The levels of creatinine in serum play an important role in evaluating glomerular filtration rate and kidney function efficiency. Chemotherapeutic drugs can impair kidney function directly by damaging renal cells or indirectly through vascular and tubular effects, contributing to elevated serum urea and creatinine levels in these patients.39–41It is important to note that all patients included in this study had no previous history of kidney disease or any known renal disorders prior to cancer diagnosis or treatment.
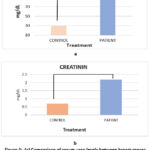 |
Figure 4: (a) Comparison of serum urea levels between breast cancer patients and healthy controls; (b) Comparison of serum creatinine levels between breast cancer patients and healthy controls |
Table 2: Assessment of urea and creatinine concentrations in the breast cancer cohort relative to the reference group
| GROUP | MEAN± SD UREA (mg/dL) | P value | GROUP | MEAN± SD CREATININ (mg/dL) | P value |
| CONTROL | 29.81± 9.53 | <0.0001 | CONTROL | 0.7002± 0.1399 | <0.0001 |
| PATIENT | 89.91 ±24.66 | PATIENT | 2.195 ±0.60414 |
As presented in Table 2, breast cancer patients exhibited markedly elevated serum urea and creatinine levels compared with the healthy control group (p < 0.0001). This increase reflects metabolic disturbances and alterations in renal function associated with breast cancer progression.
Evaluation of Serum Liver Enzymes (S.AST, S.ALT and ALP)
The levels of AST, ALT, and ALP were assessed among individuals diagnosed with breast cancer and contrasted with those of healthy subjects. The results are summarized in Table 3 and illustrated in the accompanying figure (5). ALP is a nonspecific enzyme involved in hydrolyzing phosphate in alkaline medium, with activity reflecting isoenzymes present in bone, liver, kidney, and intestinal lining.42 ALT and AST are liver-related enzymes, and their serum levels increase in various diseases, including liver damage, viral hepatitis, myocardial infarction, bile duct obstruction, acute pancreatitis, and anemia.43
The levels of ALP in patients (165.03±8.957, p = 0.0752) did not show a statistically significant difference compared with controls (92.33±19.48), suggesting that early-stage breast cancer or non-hepatic factors may not markedly affect ALP levels. ALP increases are generally observed in advanced stages of breast cancer and may serve to distinguish between stages or indicate metastasis.44,45
ALT activity was significantly higher in breast cancer patients (58.13±11.05) compared with controls(25.63±7.62 < 0.0012), and AST was also elevated (25.89±7.44vs. 40.59±29.6
, p = 0.008). ALT and AST catalyze the conversion of alanine to pyruvate and aspartate to oxaloacetate, respectively, and elevated levels reflect liver stress or damage, which may be secondary to tumor invasion, chemotherapy effects, or systemic inflammation. Previous studies report higher ALT and AST in malignant versus benign breast cancer patients, supporting their use as indicators of liver and renal function compromise in cancer progression.
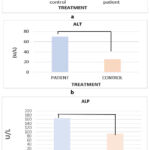 |
Figure 5: (a) Comparison of serum AST levels between breast cancer patients and healthy controls; (b) Comparison of serum ALT levels between breast cancer patients and healthy controls; (c) Comparison of serum ALP levels between breast cancer patients and healthy controls |
Table 3: Comparison of serum ALP, AST, and ALT levels between breast cancer patients and healthy controls.
| Laboratory measurements | Mean values accompanied by SD | ||
| Control | Patients | p value | |
| ALP U/L | 92.33±19.48 | 165.03±8.957 | 0.0752 |
| S.AST U/L | 25.89±7.44 | 40.59±29.6 | 0.008 |
| S.ALT U/L | 25.63 ±7.62 | 58.13±11.05 | <0.0012 |
According to the data presented in Table 3, serum ALT and AST activities were significantly elevated in breast cancer patients compared with healthy individuals (p < 0.0012 and p = 0.008, respectively). Although ALP values were higher in the patient group, the difference did not reach statistical significance (p = 0.0752). This may be related to the non-specific nature of ALP, which can originate from multiple tissues such as the liver and bone. Overall, these findings indicate changes in liver enzyme activity among patients with breast cancer.
Evaluation of Serum Electrolyte Levels (Na+, K+, Cl–)
Kidney dysfunction is widely common in patients with malignant cancer because chemotherapy and other treatments can impair renal function, cause electrolyte disturbances, and induce immune deficiency.46.47 However, many factors affect electrolyte levels, so identifying the causes is not straightforward.48 Electrolyte measurements in breast cancer patients were examined against values obtained from healthy controls.(Table 4, Figure 6). Sodium levels did not differ significantly in patients (143.1± 6.87, p = 0.0656) compared with controls (140.6± 4.43), suggesting that homeostatic mechanisms may maintain serum sodium despite renal stress or chemotherapy.48
On the other hand, chloride levels were significantly higher in patients (112.8±4.72, p < 0.00014) compared with controls (109.3±2.87). Potassium, which plays a key role in muscle excitability and nerve function, was also elevated in patients (4.40 ±0.49, p = 0.0219) compared with controls (4.71± 0.24). This may result from the release of intracellular contents from damaged tumor cells, chemotherapy-induced renal impairment, or altered hormonal regulation associated with cancer.
Serum electrolyte imbalances are clinically important for monitoring breast cancer patients, helping assess renal injury, nephrotoxicity, and overall prognosis. They may also reflect systemic effects of tumor metabolism and treatment-related stress.49-51.
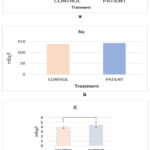 |
Figure 6: (a) Comparison of serum chloride levels between breast cancer patients and healthy controls; (b) Comparison of serum sodium levels between breast cancer patients and healthy controls; (c) Comparison of serum potassium levels between breast cancer patients and healthy controls |
Table 4: Comparison of serum electrolyte levels between breast cancer patients and healthy controls
| Laboratory measurements | Mean ± SD | |||
| Control | Patients | p value | ||
| Na+ (mEq/l) | 140.6± 4.43 | 143.1± 6.87 | 0.0656 | |
| K+ (mEq/l) | 4.71± 0.24 | 4.40 ±0.49 | 0.0219 | |
| Cl– (mEq/l) | 109.3±2.87 | 112.8±4.72 | < 0.00014 | |
Table 4 presents the comparison of serum electrolyte levels between breast cancer patients and healthy controls. Sodium levels showed no significant difference between the two groups (p = 0.0656). In contrast, potassium levels were significantly lower in patients compared with controls (p = 0.0219), while chloride levels were significantly higher in the patient group (p < 0.00014). These findings suggest alterations in electrolyte balance among breast cancer patients.
Evaluation of Serum Prolactin Levels
Imbalances in hormone levels are associated with the development of various diseases. Recently, studies have focused on serum prolactin in breast cancer prognosis. Many studies have shown a possible relationship between breast cancer and prolactin hormone. PRL has cytokine-like properties; its receptor binds to the cell surface and cytoplasm, activating the Jak2/Stat5 signaling pathway.51
Table (5) and figure (9) show that breast cancer patients had significantly higher PRL levels (32.16±8.83, p < 0.0001) compared with controls (7.25±2.47). Elevated PRL may act as a contributing factor in breast carcinogenesis, potentially by promoting cell division, reducing apoptosis, and contributing to resistance to cytotoxic drugs, although the exact mechanism remains unclear.It is also important to note that prolactin levels show only minimal variations during the menstrual cycle; therefore, the timing of sample collection in relation to the menstrual cycle was not expected to significantly influence the study outcomes.
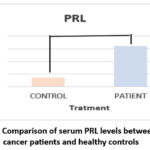 |
Figure 7: Comparison of serum PRL levels between breast cancer patients and healthy controls |
Table 5: Comparison of serum prolactin (PRL) levels between breast cancer patients and healthy controls.
| GROUP | MEAN ±SD (ng/ml) | P value |
| CONTROL | 7.25±2.47 | <0.0001 |
| PATIENT | 32.16±8.83 |
Table 5 shows that serum prolactin (PRL) levels were markedly higher in breast cancer patients (32.16 ± 8.83 ng/mL) compared with healthy controls (7.25 ± 2.47 ng/mL), with a highly significant difference (p < 0.0001). This elevation suggests a possible association between increased prolactin levels and the development or progression of breast cancer.
Conclusion
The present study investigated the serum activity of lactate dehydrogenase (LDH) in breast cancer patients undergoing chemotherapy and compared the results with those obtained from healthy controls. The findings indicated that LDH levels were generally higher in patients than in the control group. This elevation may reflect the increased metabolic demands of tumor cells, which rely heavily on aerobic glycolysis to sustain rapid proliferation and cellular activity.
The observed increase in LDH activity suggests that this enzyme may have potential clinical relevance in reflecting tumor metabolic activity during disease progression. Elevated LDH levels could therefore provide supportive information for assessing disease status and monitoring changes that occur during chemotherapy.
Overall, the results indicate an association between increased serum LDH activity and breast cancer. While these findings highlight the possible value of LDH as a biochemical indicator for monitoring disease progression and treatment response, further studies with expanded statistical validation and larger patient cohorts are recommended to confirm its prognostic and clinical utility in breast cancer management.
This study evaluated the levels of LDH in patients with breast cancer at different stages of chemotherapy, which shows that LDH activity in the patients was higher compared with that in the controls. This may be due to increased cell growth and migration in cancer cells compared with normal cells. Cancer cells require excess energy, so LDH serves as an alternative to meet the metabolic and aerobic glycolysis needs of cancer cells. In patients with breast cancer, Elevated LDH activity may serve as an early prognostic biomarker for metastasis or disease recurrence. Additionally, it can be used for monitoring cancer progression and response to treatment.
In conclusion, this study demonstrates that elevated serum LDH levels are significantly associated with breast cancer and its progression. LDH can represent a valuable marker of prognosis, early-stage diagnosis of metastasis, and therapeutic monitoring therapeutic response, highlighting its potential role in clinical management of breast cancer patients.
Acknowledgement
The authors would like to express their sincere gratitude to the Department of Chemistry, College of Science, Al-Nahrain University for providing the facilities and academic support necessary to complete this research. The authors also appreciate the assistance of all supporting institutions and staff members who contributed to the successful completion of this work.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article
Conflict of Interest
The author(s) do not have any conflict of interest
Data Availability Statement
This statement does not apply to this article.
Ethics statement
Ethical approval for the study was obtained from the Ethics Committee of the University. Patients with chronic diseases such as diabetes mellitus were excluded from the study.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not applicable
Author Contributions
- Rania Kadhim performed the experimental work and wrote the first draft of the manuscript;
- Suhad Ibrahim revised the draft; and Emad Yousif supervised the study and approved the final version of the manuscript
References
- Sinha T. Tumors: Benign and Malignant. Cancer Therapy & Oncology International Journal 2018;10(3):1-3.
CrossRef - Gomes CC. Recurrent driver mutations in benign tumors. Mutation Research/Reviews in Mutation Research 2022;789:108412.
CrossRef - Patel A. Benign vs Malignant Tumors. JAMA Oncology 2020;6(9):1488.
CrossRef - Lee Y, Jee WH, Whang YS, et al. Benign versus Malignant Soft-Tissue Tumors: Differentiation with 3T Magnetic Resonance Image Textural Analysis Including Diffusion-Weighted Imaging. Investigative Magnetic Resonance Imaging 2021;25(1):118-128.
CrossRef - Liu Y, Li H, Zhu Z, et al. RSDCNet: an efficient and lightweight deep learning model for benign and malignant pathology detection in breast cancer. Digital Health 2025;11:1-11.
CrossRef - Satpathi S, Gaurkar SS, Potdukhe A, Wanjari MB. Unveiling the Role of Hormonal Imbalance in Breast Cancer Development: A Comprehensive Review. Cureus 2023;15(7):e41737.
CrossRef - Giaquinto AN, Sung H, Miller KD, et al. Breast Cancer Statistics, 2022. CA: A Cancer Journal for Clinicians 2022;72(1):524-541.
CrossRef - Lina L. Signs and Symptoms of Breast Cancer. European Journal of Clinical Oncology 2021;3(2):1.
- Prusty RK, Begum S, Patil A, et al. Knowledge of symptoms and risk factors of breast cancer among women. BMC Women’s Health 2020;20:106.
CrossRef - Ikhuoria EB, Bach C. Introduction to Breast Carcinogenesis Symptoms, Risk Factors, Treatment and Management. European Journal of Engineering Research and Science 2018;3(7):58-66.
CrossRef - Hessari PM, Montazeri A. Health related quality of life in breast cancer patients. Health and Quality of Life Outcomes 2020;18(1):1-25.
CrossRef - Lips EH, Kumar T, Megalios A, et al. Genomic analysis defines clonal relationships of ductal carcinoma in situ and recurrent invasive breast cancer. Nature Genetics 2022;54(1):850-860.
CrossRef - Balala K, Ancheta M, Arriola G, Aban JL. Different stages, grades and types of cancer: A systematic review. 2024.
- Li Y, Liu F, Cai Q, et al. Invasion and metastasis in cancer: molecular insights and therapeutic targets. Signal Transduction and Targeted Therapy 2025;10:57.
CrossRef - Xiong X, Zheng LW, Ding Y, et al. Breast cancer: pathogenesis and treatments. Signal Transduction and Targeted Therapy 2025;10(1):49.
CrossRef - Anampa J, Makower D, Sparano JA. Progress in adjuvant chemotherapy for breast cancer: an overview. BMC Medicine 2015;13:195.
CrossRef - Waks AG, Winer EP. Breast Cancer Treatment: A Review. JAMA 2019;321(3):288-300.
CrossRef - Colomer R, Lopez IA, Albanell J, et al. Biomarkers in breast cancer: A consensus statement. Clinical & Translational Oncology 2017:1-12.
- Patani N, Martin LA, Dowsett M. Biomarkers for the clinical management of breast cancer. International Journal of Cancer 2013;133(1):1-13.
CrossRef - Adnan SF, Al-Abady ZN. Investigation the Role of Lactate Dehydrogenase in Breast Cancer Patients. Journal of Biomedicine and Biochemistry 2024;3(4).
- Erdem A, Kaye S, Caligiore F, et al. Lactate dehydrogenase A-coupled NAD regeneration is critical for acute myeloid leukemia cell survival. bioRxiv 2025.
CrossRef - Chaari A, Al-Ali D, Roach J. Purification and identification of an unknown lactate dehydrogenase isoenzyme. Biochemistry and Molecular Biology Education 2020;48(1):369-380.
CrossRef - Kumar P, Nagarajan A, Uchil PD. Analysis of Cell Viability by the Lactate Dehydrogenase Assay. Cold Spring Harbor Protocols 2018:465-468.
CrossRef - Klein R, Nagy O, Tothova C, et al. Clinical and Diagnostic Significance of Lactate Dehydrogenase and Its Isoenzymes in Animals. Veterinary Medicine International 2020:1-11.
CrossRef - Cai X, Wang T, Xie L. Lactate dehydrogenase is associated with flow mediated dilation in hypertensive patients. Scientific Reports 2023;13:768.
CrossRef - Valvona CJ, Fillmore HL, Nunn PB, et al. The Regulation and Function of Lactate Dehydrogenase A. Brain Pathology 2016;26:3-17.
CrossRef - Kocianova E, Piatrikova V, Golias T. Revisiting the Warburg Effect with Focus on Lactate. Cancers 2022;14(24):6028.
CrossRef - Mathew M, Nguyen NT, Bhutia YD, et al. Metabolic Signature of Warburg Effect in Cancer. Cancers 2024;16(3):504.
CrossRef - Chen J, Huang Z, Chen Y, et al. Lactate and lactylation in cancer. Signal Transduction and Targeted Therapy 2025;10:88.
CrossRef - Fukushi A, Kim HD, Chang YC, Kim CH. Revisited Metabolic Control and Reprogramming Cancers. International Journal of Molecular Sciences 2022;23(1):1-21.
CrossRef - Huang Y, Zhang W, Hao J, Zhu R. The application value of lactate dehydrogenase in gynecological malignant tumors. Frontiers in Oncology 2025;15:1731187.
CrossRef - Zaib S, Hayyat A, Ali N, et al. Role of Mitochondrial Membrane Potential and Lactate Dehydrogenase A in Apoptosis. 2022;22(11):2048-2062.
CrossRef - Glover T, Mitchell K. An introduction to biostatistics. Waveland Press 2008.
- Liu D, Wang D, Wu C, et al. Prognostic significance of serum lactate dehydrogenase in patients with breast cancer. Cancer Management and Research 2019;11:3611-3619.
CrossRef - Kasht GJ, Abed BA, Farhan LO, et al. Comparative Study to Determine LDH Enzyme Levels. Journal of Medicinal and Chemical Sciences 2023;6(1):883-890.
- Basnyat AS, Jha A, Pathak R, et al. Study of Serum Lactate Dehydrogenase in Breast Cancer Patients. Journal of Tropical Life Science 2017;7(2):128-132.
CrossRef - Jakhar D, Sarin SK, Kaur S. Gut microbiota and ammonia metabolism in liver disease. npj Gut and Liver 2024;1:11.
CrossRef - Kebede T, Melak T, Ibn-Sina A, et al. Assessment of Serum Uric Acid, Urea, and Glucose Levels. Ethiopian Journal of Health Sciences 2022;32(6):1183-1192.
- Prabhu SN, Mukhopadhyay SC, Gooneratne C, et al. Interdigital sensing system for detection of creatinine levels. Springer 2021;36(1):267-309.
CrossRef - Parker AL, Toulabi L, Oike T, et al. Creatine riboside is a cancer cell–derived metabolite. The Journal of Clinical Investigation 2022;132(14):1-17.
CrossRef - Jiang C, Hu F, Xia X, Guo X. Prognostic value of alkaline phosphatase in breast cancer. The International Journal of Biological Markers 2023;38(1):25-36.
CrossRef - AL-Mashhadani ZI, Mukhlis AJ, AL-Faraji AS. Estimation of ALP, GPT and GOT Activities in Iraqi Patients. Ibn Al-Haitham Journal for Pure and Applied Science 2012;25(1):1-10.
- Long ZQ, Hua X, Zhang W, et al. Prognostic impact of albumin to alkaline phosphatase ratio. Cancer Management and Research 2019;11:4809-4814.
CrossRef - Singh AK, Pandey A, Tewari M, et al. Advanced stage of breast cancer hoist alkaline phosphatase activity. Biotech 2013;3(1):517-520.
CrossRef - Yadav CP, Kaushal V, Beniwal P. Evaluation of serum biochemical profile of breast cancer patients. International Journal of Medical Research & Health Sciences 2016;5(7):1-7.
- Abdulaziz O, Almehmadi M. Dysregulation of serum electrolytes in untreated cancer patients. Annals of Cancer Research and Therapy 2021;29(2):126-130.
CrossRef - Alem A, Edae CK, Wabalo EK, et al. Factors influencing electrolyte disorders in cancer patients. SAGE Open Medicine 2021;9(1):1-7.
CrossRef - Obadipe JA, Adeyemi OA, Adebayo OA, et al. Chemotherapy-induced electrolyte disorder and nephrotoxicity in cancer patients undergoing chemotherapy. Journal of Oncology Pharmacy Practice 2022;28(6):1350-1358.
- Abbasi B, Khan S, Ahmad S, et al. Serum protein and electrolyte imbalances are associated with chemotherapy-induced neutropenia in cancer patients. Heliyon 2022;8:e10763.
CrossRef - Alrfaei BM, Alsharif KF, Alzahrani K, et al. Electrolytes play a role in detecting cisplatin-induced renal toxicity in cancer patients. Medicina 2023;59:890.
CrossRef - Chen C, Zhang Y, Wang L, et al. Nephrotoxicity in cancer treatment: an update. Advances in Clinical Chemistry 2022;107:1-35.







