Manuscript accepted on :04-11-2025
Published online on: 12-01-2026
Plagiarism Check: Yes
Reviewed by: Dr. Tolmas Hamroyev
Second Review by: Dr. Grigorios Kyriakopoulos
Final Approval by: Dr. Mariia Shanaida
Renuka Saravanan and Sivakumar Ramalingam*
and Sivakumar Ramalingam*
Department of Chemistry and Biosciences, SASTRA Deemed to be University, Srinivasa Ramanujan Centre, Kumbakonam, Tamil Nadu, India.
Corresponding Author E-mail: rsiva@src.sastra.edu
DOI : https://dx.doi.org/10.13005/bpj/3356
Abstract
In nanomedicine, there has been a lot of interest in creating sustainable and environmentally friendly processes that generate nanoparticles. In the current study, an environmentally friendly and economical method was used to produce silver nanoparticles (AgNPs) using Pergularia daemia (Forssk.) Chiov., aqueous leaf extract. The manufactured AgNPs were assessed using Fourier Transform Infrared Spectroscopy (FTIR), Zeta potential analysis, UV-visible spectroscopy (UV-Vis), scanning electron microscopy (SEM), and a particle size analyzer. AgNP creation was validated by the unique surface plasmon resonance (SPR) peak seen at 442 nm. SEM analysis revealed a spherical morphology, while FTIR indicated the occurrence of functional groups responsible for nanoparticle stabilization. The generated AgNPs had a normal particle size of 137.5 nm and a zeta potential of -20.7 mV, indicating good colloidal stability. The anticancer efficacy of AgNPs was evaluated against HepG2 liver cancer cells using MTT, LDH, and Trypan Blue dye exclusion assays, demonstrating significant cytotoxic activity. Additionally, the antioxidant activity of the nanoparticles suggests their capability to moderate oxidative stress. This study highlights the promise of Pergularia daemia (Forssk.) Chiov., as a natural reducing and stabilizing agent for AgNP synthesis, offering a promising avenue for the development of plant-based nanotherapeutics. Further investigations into mechanistic pathways and in vivo studies could pave the way for their clinical applications in cancer treatment.
Keywords
Anticancer activity; HepG2 cells; Nanomedicine; Pergularia daemia and Silver nanoparticles
Download this article as:| Copy the following to cite this article: Saravanan R, Ramalingam S. Green Fabrication of Pergularia daemia-Mediated Silver Nanoparticles for Antioxidant and Hepatocellular Carcinoma Applications. Biomed Pharmacol J 2026;19(1). |
| Copy the following to cite this URL: Saravanan R, Ramalingam S. Green Fabrication of Pergularia daemia-Mediated Silver Nanoparticles for Antioxidant and Hepatocellular Carcinoma Applications. Biomed Pharmacol J 2026;19(1). Available from: https://bit.ly/4qQOYwT |
Introduction
Liver cancer ranks as the 6th most frequently detected cancer worldwide and stands as the second leading cause of cancer-related mortality, underscoring its significant global burden and critical importance in the field of oncology. India reports approximately 35,000 new liver cancer cases annually, with around 34,000 deaths each year. Its incidence rate is 2.15 per 100,000, its prevalence is 2.27 per 100,000,1 and its mortality rate is 2.1 per 100,000, making liver cancer the eighth most prevalent cancer in the nation. Around 80% of occurrences of liver cancer are associated with chronic hepatitis B virus and hepatitis C virus infections, with hepatocellular carcinoma (HCC) accounting for 70% to 90% of cases. 2
The risk factors associated with liver cancer are diverse, ranging from viral infections to conditions linked to lifestyle. The metabolic syndrome and non-alcoholic fatty liver disease have been closely related to diabetic mellitus type 2, significantly increasing the risk of HCC, particularly in individuals with concurrent hepatitis or chronic alcohol consumption.3 Excessive alcohol intake—exceeding six drinks per day—contributes to cirrhosis, a major precursor of liver cancer.4 Additionally, aflatoxin exposure from contaminated crops such as rice, peanuts, and maize presents a serious non-viral risk factor. Gender disparities are also evident, with men being three times more likely to develop liver cancer than women. Other contributing factors include obesity, smoking, and immunosuppressive conditions such as HIV/AIDS.
Liver cancer remains a silent disease in its early stages, often eluding detection until it reaches an advanced phase. Common symptoms include unexplained weight loss, jaundice, upper abdominal pain, bloating, nausea, fatigue, bruising, and abnormalities in calcium and cholesterol levels.5 Accurate diagnosis necessitates a differential assessment of hepatic lesions, with possible conditions including hepatocellular carcinoma, cholangiocarcinoma, hepatic adenoma, and metastases. Confirmatory diagnostics involve blood tests, MRI and CT scans, laparoscopy, and liver biopsies.6
The prognosis for liver cancer remains grim, especially in its advanced stages, necessitating the exploration of innovative treatment modalities. Traditional interventions include ablative therapy, radiation, chemotherapy, liver transplantation, and immunotherapy.7 However, recent advancements in nanotechnology have paved the way for enhanced drug delivery systems, increasing therapeutic efficacy while minimizing adverse effects. Nanotechnology-based drug carriers, such as smart nanomaterials, are designed to overcome drug resistance and enhance tumor targeting. Multimodal nanomedicines integrate diagnostic and therapeutic functionalities, offering a promising avenue for liver cancer management.8
Green Synthesis: A Sustainable Approach to Nanomedicine
Nanotechnology’s expansion into green synthesis has revolutionized the biomedical field by introducing eco-friendly alternatives to conventional chemical-based nanoparticle production. Green synthesis, which leverages plant extracts for nanoparticle production, enhances stability and biocompatibility while reducing environmental impact.9, 10 This method utilizes phytochemicals from medicinal plants to produce silver nanoparticles (AgNPs), which exhibit antimicrobial, anticancer, and therapeutic properties.11
Silver Nanoparticles in Cancer Therapy
Silver nanoparticles (AgNPs) have emerged as potent therapeutic agents due to their diverse biomedical applications. They are extensively used in medical devices, wound dressings, textiles, and chemotherapy. However, concerns regarding their potential toxicity necessitate further research.12, 13 The green synthesis of AgNPs, particularly through plant extracts, enhances their stability and bioactivity, making them highly effective in cancer treatment and diagnostics.14, 15 These nanoparticles exhibit antifungal, antibacterial, anti-inflammatory, and antiviral properties, further expanding their utility in the medical and industrial sectors.16, 17
Pergularia daemia: A Natural Resource for Green Nanoparticle Synthesis
Pergularia daemia (Forssk.) Chiov., a climber belonging to the family Apocynaceae, has a broad native distribution in seasonally-dry tropical biomes. According to the Royal Botanic Gardens, Kew’s Plants of the World Online, its native range extends from tropical and southern Africa through the Sinai and the Arabian Peninsula, into Iran and west-Indo-China. The species is present in numerous countries, including Angola, Botswana, Burkina Faso, Cameroon, Chad, Djibouti, Eritrea, Ethiopia, Kenya, Malawi, Mali, Mauritania, Mozambique, Namibia, Nigeria, Senegal, Somalia, South Sudan, Tanzania, Uganda, Zambia, Zimbabwe in Africa; and in Asia in countries such as Afghanistan, Bangladesh, India, Iran, Myanmar, Nepal, Pakistan, Sri Lanka, Yemen, Oman and the Andaman Islands. This wide native distribution suggests a degree of ecological plasticity, particularly adaptation to arid or semi-arid, seasonally dry habitats. Including a clear statement of this distribution is important, since variation in phytochemical and biological activity profiles may relate to ecological factors. https://powo.science.kew.org/ taxon/ urn%3Alsid%3Aipni.org%3Anames%3A100371-1. Traditionally, its extracts have been employed in treating respiratory, gastrointestinal, and gynecological disorders, as well as infections and inflammation.18, 19 Given its rich bioactive composition, Pergularia daemia plays a crucial role in green nanoparticle synthesis, offering immense potential for liver cancer therapy. Hence the current work is to prepare and characterize silver nanoparticles (AgNPs) from Pergularia daemia leaf extract and evaluate their antioxidant and anticancer activities, with the objective of developing a green, cost-effective nanotherapeutic agent for potential biomedical applications (Figure 1).
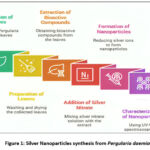 |
Figure 1: Silver Nanoparticles synthesis from Pergularia daemiaClick here to view Figure |
The HepG2 Cell Line: A Model for Liver Cancer Research
The HepG2 cell line has been widely utilized in liver cancer research, serving as an invaluable tool for studying cytotoxicity, oncogenesis, and hepatocellular function. Originating from hepatocellular carcinoma, HepG2 cells have also been instrumental in studies related to hepatoblastoma and normal hepatocytes. Despite certain genomic, transcriptomic, and proteomic limitations, they remain the most commonly employed human hepatoma model for evaluating drug metabolism and liver toxicity. These cells, known for their epithelial-like morphology, are non-tumorigenic and exhibit strong proliferative capacity, making them a preferred choice in experimental hepatology.20
Materials and Methods
The aerial parts of Pergularia daemia (Forssk.) Chiov. were collected from T. Palur in the Ariyalur, Tamil Nadu, India (GPS coordinates: 11.1406° N, 79.3183° E) in July 2024. The plant species was identified and authenticated by a qualified taxonomist, and a voucher specimen No. CARISM00178 was deposited at the Department Herbarium, [CARISM, SASTRA Deemed to be University, Thanjavur], for future reference. The leaves were carefully separated from the plants for use in the synthesis of silver nanoparticles. They were thoroughly splashed with distilled water to eliminate any surface contaminants or debris. After washing, the leaves were shade-dried for 15 days to preserve their phytochemical constituents. The dried leaves were then ground into a fine powder using an electric blender and stored in an airtight container for future use.
 |
Figure 2: Pergularia daemiaClick here to view Figure |
Preparation of Plant Extract
A total of 5 g of powdered Pergularia daemia leaves was dispersed in 100 mL of distilled water and subjected to boiling at a constant temperature of 60°C for 25 minutes. The extract was then allowed to cool to room temperature. The resulting solution was filtered using Whatman No. 1 filter paper to remove insoluble residues. This process facilitated the biogenic synthesis of nanostructured silver particles by reducing bulk silver ions. The filtrate contained bioactive compounds that functioned as natural reducing agents, contributing to nanoparticle formation.
Silver Nitrate (AgNO₃) 4Mm Preparation
The preparation of the AgNO₃ solution for green synthesis involves dissolving 4 mM of AgNO₃ in 100 mL of distilled water, followed by vigorous stirring. This solution contains micro-structured silver ions, which transform into nanostructured silver particles through a biogenic reduction process facilitated by bioactive compounds present in the plant extract.21
Biosynthesis of NS-AgNPs
According to Mani et al. (2021), 90 mL of the prepared silver nitrate solution is combined with 10 mL of Pergularia daemia leaf extract under continuous stirring using a magnetic stirrer. The silver nanoparticles (AgNPs) formation is indicated by a distinct color change, becoming yellow to reddish-brown, confirming the reduction of silver ions. The reaction mixture is then incubated in complete darkness for 24 hours to ensure optimal nanoparticle synthesis. The resulting AgNPs are further utilized for various applications.
Phytochemical Screening of Plant Extract
Using conventional qualitative assays, a thorough phytochemical screening was carried out to determine the bioactive components contained in the plant extract. Alkaloids were detected through Mayer’s and Hager’s tests, indicated by the formation of a whitish-yellow or cream-colored precipitate and a yellow-colored precipitate, respectively. Steroids were confirmed via the Salkowski and Libermann-Burchard tests, exhibiting a golden-yellow tint and a violet to blue or green coloration. The occurrence of terpenoids was established by the Salkowski test, which produced a characteristic reddish-brown ring. Flavonoids were identified using the alkaline reagent and lead acetate tests, resulting in yellow coloration and a yellow precipitate. Saponins were confirmed by the froth test, demonstrating the formation of persistent froth. Phenols were detected through the lead acetate and Folin’s phenol tests, yielding a precipitate and a bluish-black coloration. Tannins were identified via the lead acetate and ferric chloride tests, leading to the formation of a white precipitate and a blue, black, or brownish-green coloration. Cardiac glycosides were confirmed using the Keller-Killiani test, which produced a distinctive blue tint over a brown ring. The presence of anthraquinones was established through Borntrager’s test, indicated by the formation of a rose pink to red color in the ammoniacal layer. Proteins were identified by the Biuret test, resulting in the development of a violet color. Carbohydrates were confirmed using Molisch’s and Barfoed’s tests, which produced a violet ring at the junction of two liquids and a reddish-brown precipitate. These phytochemical analyses provide critical insights into the diverse phytocompounds present in the extract, supporting its potential therapeutic and biomedical applications.22
Characterization of Silver Nanoparticles
Dynamic Light Scattering Measurements
Dynamic Light Scattering was employed to determine the hydrodynamic diameter and particle size distribution of the synthesized AgNPs. Measurements were performed using a Malvern Zetasizer (Malvern Instruments Ltd., UK), ensuring precise characterization of the nanoparticle dispersion in an aqueous medium. The acquired data were processed and analyzed using the proprietary Zetasizer software, providing insights into the polydispersity index (PDI) and stability of the nanoparticles in solution.23
Scanning Electron Microscopy Analysis
The high-resolution imaging method known as scanning electron microscopy uses a concentrated electron beam to produce precise topographical and compositional data about the sample surface. Upon interaction with the sample, the incident electrons induce secondary electron emission, revealing morphological characteristics. In this study, SEM analysis was conducted using a Hitachi S-4500 SEM system. The sample was prepared by applying a small amount of silver nanoparticle (AgNP) suspension to a copper grid covered with carbon and then removing any extra solution. The sample was then subjected to drying under a mercury lamp for five minutes to ensure proper adhesion. The synthesised AgNPs’ size, form, and distribution were clarified by imaging them with a VEGA3 TESCAN (Japan) SEM device at a 10 kV accelerating voltage while under a high vacuum.24
Fourier Transform Infrared Spectroscopy Analysis
In order to identify the functional groups involved in stabilizing and capping nanoparticles, FTIR spectroscopy was utilized. The spectral measurements were conducted using an FTIR spectrometer, utilizing potassium bromide (KBr) as a matrix. A finely ground mixture of AgNPs and IR-grade KBr in a 200:1 ratio was prepared using a mortar and pestle, followed by the formation of a transparent pellet using a hydraulic pellet press. Although the spectra were captured between 400 and 4000 cm⁻¹, it was possible to identify distinctive absorption bands that were suggestive of chemical interactions and surface alterations of the produced nanoparticles.24
UV-Visible Spectrophotometric Analysis
The synthesized silver nanoparticles was evaluated using UV-Visible spectrophotometry. The surface plasmon resonance phenomenon of AgNPs was assessed by recording the absorbance spectrum of the colloidal solution. The reduction of silver nitrate to AgNPs was primarily observed through a colour change in the reaction mixture. A Thermo Scientific UV-Visible spectrophotometer was used to make spectral measurements in the 300–500 nm wavelength range, which made it easier to confirm the production of nanoparticles and their stability during dispersion.25
In Vitro Antioxidant Activity
DPPH assay
The most common method to determine antioxidant activity is the DPPH radical scavenging experiment, which is based on the reduction of the stable 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical to its non-radical form, 2,2-diphenyl-1-picrylhydrazine (DPPH-H), in the presence of antioxidants that donate hydrogen. This reduction leads to a color change from purple to yellow, which is quantified spectrophotometrically at 517 nm. In this study, the antioxidant potential of Pergularia daemia aqueous extract was analyzed, with ascorbic acid serving as the standard reference. A solution of plant extract was made at a 10 mg/10 mL methanol concentration, while the DPPH reagent was freshly made by dissolving 3.4 mg in 50 mL of 95% methanol (47.5 mL methanol + 2.5 mL water). Various concentrations (100–1000 µg) of the extract were taken in test tubes, diluted with distilled water to 1 mL, and mixed with 0.5 mL of the DPPH solution. A spectrophotometer was used to measure absorbance at 517 nm after the reaction mixture was incubated for 10 minutes at room temperature. The control sample was prepared by replacing the plant extract with distilled water. This study highlights the potential of Pergularia daemia extract as a natural antioxidant, contributing to the growing body of research on plant-based radical scavengers and their role in mitigating oxidative stress-induced damage.26
The following formula was used to determine the percentage of DPPH scavenging activity:
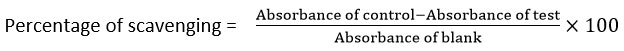
ABTS Radical Scavenging Assay
The ABTS radical scavenging assay evaluates antioxidant activity based on the ability to neutralize the stable ABTS radical, resulting in decreased absorbance at 734 nm. In this study, Pergularia daemia extract (10 mg/mL) was tested against ABTS radicals, with ascorbic acid as the standard. ABTS reagent was prepared by reacting 2 mM ABTS with 70 mM potassium persulfate and incubating in the dark for 2 hours. Various extract concentrations (100–500 µg) were mixed with phosphate buffer (pH 7.4), potassium persulfate, and ABTS solution. Absorbance was measured at 734 nm, and the extract showed notable radical scavenging activity, indicating its antioxidant potential.27
The percentage of ABTS radical scavenging activity was calculated using the formula:
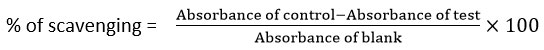
In Vitro Anticancer Activity
Maintenance of Cell
The HepG2 cell line was obtained from the NCCS, Pune. These cells exhibit a squamous morphology and play a vital role in the distribution of biochemical substances across tissues. HepG2 cells inherently grow as a monolayer in vitro. In a humidified environment with regulated CO₂ saturation, they were cultivated in a flask with Dulbecco’s Modified Eagle Medium, which was enhanced with 3% L-glutamine, 10% fetal bovine serum, penicillin, streptomycin, and amphotericin B, as well as 7.5% sodium bicarbonate. The cells were sub cultured and passaged in the required number of flasks for subsequent experimental studies.
MTT Assay
The MTT (3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide) assay is a commonly used and dependable in vitro technique for evaluating cell viability and proliferation. The MTT assay was used to assess the cytotoxicity of the plant-derived silver nanoparticles’ ethanol extract on HepG2 cells. 100 μL of culture media was used to seed HepG2 cells (5000 cells/well) in 96-well plates, and the cells were then incubated for 24 hours at 37°C with 5% CO₂ to promote adhesion. Cells were treated with different amounts of the extract (15.625–250 μg/mL in DMSO) for an additional 24 hours after the media was changed out for fresh medium. In order to allow formazan crystal formation, 20 μL of MTT solution (5 mg/mL) was then applied to each well and incubated for 3 hours. The crystals were then dissolved by adding 100 μL of DMSO after the medium was removed. At 570 nm, absorbance was determined with an ELISA plate reader.28 Using the following formula, the percentage of cell growth inhibition was determined:
![]()
Lactate Dehydrogenase Release Assay
To assess cytotoxicity induced by Pergularia daemia-mediated silver nanoparticles based on LDH enzyme release from damaged cells into the culture medium. HepG2 cells (5,000–10,000/well) were seeded in a 96-well plate with 100 µL of complete medium and incubated at 37°C with 5% CO₂ for 24 h. Cells were then treated with serial dilutions of Pergularia daemia extract or nanoparticles (15.625–250 µg/mL) in fresh medium. Negative (untreated) and positive controls (1% Triton X-100) were included. Following a 24-hour incubation period, 50 µL of the supernatant was taken in a fresh plate, combined with 50 µL of the LDH reaction mixture, and allowed to sit at room temperature for 30 minutes in the dark.29, 30Absorbance was measured at 490 nm. Cytotoxicity (%) was calculated using:
![]()
Trypan blue dye exclusion method
Based on membrane integrity, the Trypan Blue Dye Exclusion Method is a commonly used technique to evaluate cell viability by separating live cells from dead ones. Seed cells into 6-well plates at a density suitable for exponential growth and incubate at 37°C with 5% CO₂ until ~80% confluency. Distribute 25, 50, and 100 µg/mL of Pergularia daemia-mediated AgNPs for a duration of 24 to 48 hours. Remove the medium after incubation, then use PBS to wash the cells and trypsin-EDTA to detach them. Use a medium containing FBS to neutralize, then gather the cells in centrifuge tubes. Wash the pellet with 1 mL PBS after centrifuging it for 5 minutes at 100 × g and discarding the supernatant. The final pellet should be resuspended in 1 mL PBS after another centrifugation. After combining 100 µL of cell suspension with 100 µL of 0.4% Trypan Blue (in PBS, pH 7.2–7.4), let it sit at room temperature for 3–5 minutes before evaluating its viability. A hemocytometer was used to count the number of viable (unstained) and non-viable (stained) cells.31
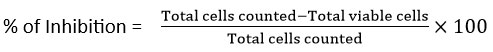
Results
Phytochemical Screening
Table 1 presents the findings of the qualitative phytochemical screening for both aqueous and ethanolic extracts of Pergularia daemia, revealing the presence of several important bioactive constituents such as steroids, glycosides, tannins, flavonoids, saponins, and terpenoids. Although quinones and phenols were detected in the ethanolic extract, they did not exhibit significant activity.
Table 1: Investigation of Phytochemical Constituents in Pergularia daemia
| Constituents | Results | |
| AQUEOUS EXTRACT | ETHANOL EXTRACT | |
| Alkaloid | + | + |
| Steroids | + | + |
| Terpenoids | + | + |
| Flavonoids | + | + |
| saponins | + | + |
| Phenols | + | – |
| Tannins | + | + |
| Cardiac glycosides | + | + |
| Anthraquinones | – | – |
| proteins | + | + |
| carbohydrates | + | + |
Biosynthesis of Silver Nanoparticles
The initially colourless aqueous silver nitrate (AgNO₃) solution exhibited a notable color change to reddish-brown upon the addition of Pergularia daemia plant extract. This transformation signifies the Ag⁺ ions reduction, driven through phytochemical constituents within the plant extract. The appearance of the reddish-brown colour indicative of surface plasmon resonance (SPR), a hallmark of silver nanoparticle (AgNP) formation.
DLS Measurements
Dynamic Light Scattering analysis was employed to evaluate the size distribution of the biosynthesized silver nanoparticles (AgNPs). The results designated that the AgNPs synthesized from Pergularia daemia exhibited an average particle size of 137.5 nm (Figure 2), marginally exceeding the conventional nanoscale range of 1–100 nm. The synthesized silver nanoparticles’ zeta potential was calculated to be -20.7 mV, signifying a negatively charged surface (Figure 4). This measurement is a critical parameter in assessing the colloidal stability of nanoparticulate systems. The obtained zeta potential value suggests that electrostatic repulsion between the nanoparticles effectively prevents aggregation, thus maintaining a stable dispersion.
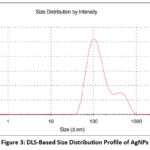 |
Figure 3: DLS-Based Size Distribution Profile of AgNPs Click here to view Figure |
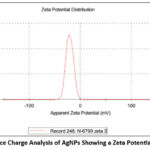 |
Figure 4: Surface Charge Analysis of AgNPs Showing a Zeta Potential of –20.7 mVClick here to view Figure |
UV-Visible Spectrophotometric Analysis of Silver Nanoparticles
UV-Visible spectrophotometry is a fundamental technique for characterizing silver nanoparticles (AgNPs), primarily through the detection of surface plasmon resonance (SPR). In this study, UV-Vis spectral analysis revealed a projecting SPR peak at 442 nm, demonstrating the successful formation of AgNPs with enhanced radiation absorbance. Silver nanoparticles’ characteristic SPR range, which normally lies between 400 and 450 nm, is in line with the observed peak. The strong SPR absorption at 442 nm serves as a definitive indicator of AgNP formation, reinforcing the reliability of UV-Visible spectroscopy in nanoparticle characterization. These outcomes contribute to the rising body of evidence supporting plant-mediated AgNP synthesis as a sustainable and efficient approach for nanomaterial production.
Scanning electron microscopy analysis
Scanning electron microscopy (SEM) was used to examine the morphology characteristics and structural integrity of the biosynthesized silver nanoparticles (AgNPs). The SEM micrograph (Figure 5) revealed that the AgNPs synthesized using Pergularia daemia exhibited a predominantly spherical shape, indicating uniform nanoparticle formation. The well-defined morphology of these nanoparticles indicates that the plant extract’s phytochemicals provide efficient capping and stability.
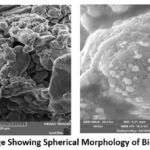 |
Figure 5: SEM Image Showing Spherical Morphology of Biosynthesized AgNPsClick here to view Figure |
FTIR Spectroscopy Analysis
The FTIR spectrum provides clear evidence that biomolecules present in the Pergularia daemia Leaf extract actively contributes to the stabilization and reduction of biosynthesized silver nanoparticles (AgNPs). Characteristic absorption bands observed at 3408.10, 2426.65, 1763.40, 1602.58, 1384.50, 1093.04, 825.01, and 608.70 cm⁻¹ indicate the interaction of various functional groups with the nanoparticle surface (Figure 6).
 |
Figure 6. FTIR Spectrum Showing Functional Groups Involved in AgNP SynthesisClick here to view Figure |
Antioxidant Assay for Silver Nanoparticles
DPPH radical scavenging assay
The DPPH assay, which measures free radical scavenging activity, was used to assess the antioxidant potential of biosynthesized silver nanoparticles (AgNPs). The IC₅₀ value, which is the concentration needed to cut the initial DPPH radical population by 50%, is used to measure the degree of this change. As illustrated in Figure 7, the inhibition percentage increased with rising AgNP concentrations, with the highest absorbance observed at 250 μg/mL.
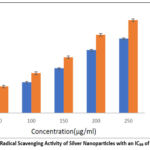 |
Figure 7: DPPH Radical Scavenging Activity of Silver Nanoparticles with an IC₅₀ of 221.22 μg/mLClick here to view Figure |
In comparison, ascorbic acid, used as a reference standard, exhibited 55.8% inhibition at the same concentration. The IC₅₀ value for the synthesized AgNPs was determined to be 221.22 μg/mL, indicating significant antioxidant activity.
Assay for ABTS radical scavenging
The free radical scavenging activity of silver nanoparticles (AgNPs) synthesized using Pergularia daemia was assessed through the ABTS assay, which evaluates radical scavenging capacity based on a measurable blue-green color change. The AgNPs demonstrated a concentration-dependent scavenging effect, achieving a maximum inhibition of 56.29% at 250 μg/mL and an IC₅₀ value of 213 μg/mL (Figure 8). Ascorbic acid was used as the standard.
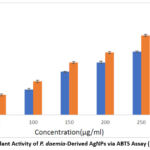 |
Figure 8: Antioxidant Activity of P. daemia-Derived AgNPs via ABTS Assay (IC₅₀ = 213 μg/mL).Click here to view Figure |
In Vitro Anticancer Activity
MTT Assay
The cytotoxic potential of biosynthesized silver nanoparticles (AgNPs) derived from Pergularia daemia was assessed against the HepG2 cell line using the MTT assay. Cell viability was evaluated at several AgNP concentrations (15.62, 31.25, 62.5, 125, & 250 μg/mL), revealing a dose-dependent decline in cell viability (Figures 9 and 10). The silver nanoparticles (AgNPs) synthesized from Pergularia daemia demonstrated significant cytotoxic activity, as evidenced by an IC₅₀ value of 229.033 μg/mL.
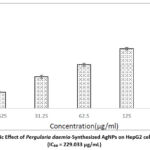 |
Figure 9: Cytotoxic Effect of Pergularia daemia-Synthesized AgNPs on HepG2 cells via MTT Assay (IC₅₀ = 229.033 μg/mL).Click here to view Figure |
Morphology of MCF cells
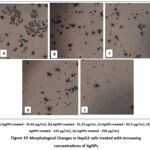 |
Figure 10: Morphological Changes in HepG2 cells treated with increasing concentrations of AgNPs.Click here to view Figure |
LDH Assay
The cytotoxic potential of biosynthesised silver nanoparticles derived from Pergularia daemia was evaluated against human liver carcinoma (HepG2) cells using the lactate dehydrogenase (LDH) release assay, as illustrated in Figure 11. HepG2 cells were treated with increasing concentrations of AgNPs—15.625, 31.25, 62.5, 125, and 250 μg/mL—to assess dose-dependent cytotoxicity. The corresponding inhibition rates observed were 8.95%, 19.19%, 29.98%, 42.74%, and 53.95%, respectively.
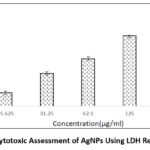 |
Figure 11: Cytotoxic Assessment of AgNPs Using LDH Release AssayClick here to view Figure |
Trypan blue assay
Figure 12 demonstrates the cytotoxicity effects of silver AgNPs made from Pergularia daemia aqueous leaf extract, as evaluated by the trypan blue exclusion assay after 48 hours of treatment followed by 24 hours of incubation. A marked, concentration-dependent reduction in HepG2 cell viability was observed, with inhibition rates of 18.18%, 33.33%, 55.56%, 73.34%, and 87.78% at nanoparticle concentrations of 100 – 500 μg/mL, respectively.
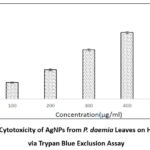 |
Figure 12: Cytotoxicity of AgNPs from P. daemia Leaves on HepG2 Cells via Trypan Blue Exclusion AssayClick here to view Figure |
Discussion
The present study demonstrates that Pergularia daemia acts as an efficient biogenic agent for the synthesis of silver nanoparticles (AgNPs) exhibiting dual antioxidant and anticancer properties. The phytochemical profiling of P. daemia revealed diverse bioactive compounds such as flavonoids, tannins, saponins, and terpenoids, which are known to exert antioxidant, anticancer, and antimicrobial activities. The phytoconstituents of P. daemia, including flavonoids, alkaloids, and cardiac glycosides, serve as natural reducing and capping agents, contributing to the stability and bioactivity of the nanoparticles. Similar phytochemical-assisted synthesis has been shown to enhance nanoparticle biocompatibility and therapeutic potential.32,33 Flavonoids, a group of naturally occurring polyphenolic complexes, are widely recognized for having strong anti-cancer and antioxidant qualities. Found abundantly in fruits, vegetables, herbs, and medicinal plants, these Secondary metabolites are essential for protecting biological systems against oxidative stress and the emergence of cancer. Certain flavonoids have also been shown to enhance the effectiveness of conventional chemotherapy. For instance, luteolin has demonstrated a synergistic effect with paclitaxel in the treatment of esophageal carcinoma by promoting apoptosis and suppressing epithelial–mesenchymal transition.34 Research into the extraction methods of C. camphora revealed that methanol extracts contained higher terpenoid content and exhibited stronger antioxidant activity compared to other solvents. The study emphasized the importance of solvent choice in maximizing the antioxidant potential of plant extracts.35
The biosynthesis of AgNPs was evident through the SPR peak at 442 nm, SEM-confirmed spherical morphology, and FTIR functional groups, which collectively demonstrated the role of plant biomolecules as both reducing and stabilizing agents. A similar study reported36 that silver nanoparticles synthesized using Salvia miltiorrhiza had a particle size of 128 nm, highlighting that plant-mediated synthesis commonly produces moderately larger nanoparticles. The outcomes of the present research indicate that the synthesized AgNPs fall within a suitable size range for potential applications in biomedical and nanotechnology-related research. Current findings are consistent with those of scientist,37 who reported a comparable zeta potential value of –20.7 mV for silver nanoparticles, supporting the idea that such particles possess considerable colloidal stability due to electrostatic interactions. The pronounced negative surface charge indicates strong repulsive forces between particles, which reduces agglomeration and contributes to the overall stability of the nanoparticle suspension. This electrostatic stabilization is crucial for applications that demand sustained nanoparticle dispersion, particularly in biomedical and catalytic fields. The results highlight the role of surface charge in nanoparticle stability and emphasize the importance of zeta potential measurements in nanomaterial characterization. Future investigations will focus on optimizing synthesis parameters to further enhance the colloidal stability and functional properties of silver nanoparticles for targeted applications.
This observation is further supported by earlier studies that demonstrated similar absorption spectra during the synthesis of AgNPs using Saccharina japonica aqueous leaf extract, validating successful nanoparticle formation.36 Additionally, prior research has consistently validated the effectiveness of UV–Visible spectrophotometry in identifying silver nanoparticles, typically characterized by a surface plasmon resonance (SPR) peak. A study reported 38 a broad SPR peak at 441 nm, while another work39 reported a central peak at 460 nm within the 300–780 nm range40. Recent studies40 reported the formation of spherical-shaped AgNPs using Euphorbia serpens, while comparable results were observed in AgNPs synthesized from Svensonia hyderabadensis41 leaf extract. The consistency in nanoparticle shape across various plant-based synthesis methods underscores the reliability of green nanotechnology in producing stable and well-defined nanomaterials for potential biomedical and industrial applications.
Notably, O–H stretching vibrations, which are generally linked to phenolic compounds,42 are represented by the broad peak at 3408.10 cm⁻¹, whereas C–H stretching43 is responsible for the band at 2426.65 cm⁻¹. The absorption at 1763.40 cm⁻¹ is indicative of C–O functional groups,44 and the peak at 1602.58 cm⁻¹ corresponds to C=O stretching.45Additional bands include 1384.50 cm⁻¹ for C=C stretching,46 1093.04 cm⁻¹ for C–H bending, and 608.70 cm⁻¹ representing C–Cl stretching vibrations.47 These functional group interactions confirm the role of plant-derived biomolecules in the green synthesis process, contributing to both nanoparticle formation and stability. These findings highlight the crucial role of plant-derived functional groups in enabling a green and environmentally friendly synthesis process, while also enhancing the structural integrity and long-term stability of the silver nanoparticles.
The antioxidant assays confirmed significant free radical scavenging activity, with IC₅₀ values close to ascorbic acid, indicating that the phytochemicals bound to AgNPs enhanced their antioxidant capacity. Such findings align with earlier reports of plant-mediated AgNPs exhibiting potent radical scavenging properties. The IC₅₀ value for the synthesized AgNPs was determined to be 221.22 μg/mL, indicating significant antioxidant activity. These findings suggest that silver nanoparticles derived from Pergularia daemia leaf extract possess notable free radical scavenging potential, highlighting their possible application in antioxidant therapies and biomedical formulations.48 The strong antioxidant activity of the P. daemia-AgNPs suggests their ability to neutralize reactive oxygen species (ROS), thereby protecting cells from oxidative stress-related damage, which plays a major role in carcinogenesis and cellular aging.49 The significant cytotoxicity against MCF-7 breast cancer cells indicates selective interference with cancer cell proliferation and induction of apoptosis, likely through ROS-mediated mechanisms and mitochondrial dysfunction.50
The cytotoxicity assays against HepG2 cells consistently demonstrated dose-dependent effects, with IC₅₀ values in the range of 207–229 μg/mL across different assays. These results suggest that the AgNPs compromise cell membrane integrity, reduce viability, and enhance cell death, consistent with earlier reports of plant-based AgNPs such as those synthesized from Aerva lanata and Ruta graveolens. This observation aligns with prior research,51 in which Aerva lanata-mediated AgNPs exhibited potent cytotoxic effects against HepG2 cells, achieving an IC₅₀ of 63.97 μg/mL. In the present work, a dose-dependent reduction in HepG2 cell viability was detected, ranging from 80.97% at 25 μg/mL to 26.12% at 100 μg/mL, further supporting the cytotoxic potential of the synthesized nanoparticles. Similarly, AgNPs derived from Ruta graveolens ethanol leaf extract were found to exert strong anticancer effects against both HepG2 and HeLa cells, whereas the crude extract displayed selective cytotoxicity toward HepG2 cells.52 Collectively, these findings highlight the promising role of AgNPs produced by P. daemia as potential anticancer agents and warrant further investigation into their underlying mechanisms and suitability for therapeutic applications.
The half-maximal inhibitory concentration (IC₅₀) was determined to be 207.81 μg/mL, suggesting moderate potency of the nanoparticles in inducing cell death. The LDH assay results clearly demonstrated a concentration-dependent increase in cytotoxicity, indicative of impaired cellular membrane integrity and enhanced cell senescence. These findings provide compelling evidence that Pergularia daemia-mediated silver nanoparticles exhibit promising anticancer properties against HepG2 cells. Comparable outcomes were previously documented,53 who reported significant LDH release in HepG2 cells following treatment with Schinus molle extract-mediated nanoparticles, further substantiating the role of plant-derived AgNPs in liver cancer therapeutics. The trypan blue assay results confirm a dose-responsive cytotoxic effect, reinforcing the therapeutic potential of P. daemia-mediated AgNPs in liver cancer treatment. These outcomes are consistent with the study,54 which reported notable cytotoxic effects of Ananas comosus-derived AgNPs on HepG2 cells, further validating the effectiveness of plant-based nanoparticle synthesis for anticancer applications.
From a nanomedical standpoint, plant-mediated AgNPs represent an eco-friendly and sustainable platform for therapeutic development.55 The concept of technological symbiosis, wherein phytogenic nanoparticles are combined with other nanomaterials (e.g., AuNPs, ZnO, or bimetallic systems), offers promising avenues for enhanced synergistic effects, improved drug delivery, and targeted cancer therapy.56,57 Despite these promising results, limitations such as nanoparticle aggregation, dose-dependent cytotoxicity, and lack of in vivo validation must be addressed. Comprehensive in vivo and mechanistic studies are essential to elucidate pharmacokinetics, biodistribution, and long-term safety.5⁸ Future research should focus on optimizing synthesis parameters, integrating P. daemia-AgNPs into hybrid nanocarriers, and exploring their performance in animal models for translational nanomedicine applications.,59,60, 61
In conclusion, Pergularia daemia-mediated AgNPs provide a green and promising approach for nanotherapeutic development, offering potential as multifunctional agents in antioxidant defense and cancer therapy.
Conclusion
The present investigation demonstrates the effective green synthesis of silver nanoparticles (AgNPs) using Pergularia daemia leaf extract in aqueous form, providing an environmentally responsible and sustainable approach in nanobiotechnology. Nanoparticle formation was evidenced by a visible color change and confirmed through UV-Vis spectrophotometry, zeta potential, particle size analysis, FTIR spectroscopy, and SEM imaging, all validating the stability, morphology, and uniform distribution of the AgNPs. Phytochemical profiling revealed abundant bioactive compounds—particularly flavonoids, tannins, steroids, glycosides, saponins, and terpenoids—as major contributors to nanoparticle reduction and stabilization, while quinones and phenols showed lesser involvement.
The biosynthesized AgNPs exhibited significant antioxidant activity in DPPH and ABTS assays, likely due to the redox-active phytoconstituents embedded during synthesis. In addition, concentration-dependent cytotoxicity against HepG2 liver cancer cells, confirmed through MTT, LDH, and Trypan Blue exclusion assays, demonstrated up to 87.78% inhibition, indicating potent anticancer efficacy. Importantly, these findings highlight the synergistic potential of P. daemia-mediated AgNPs when considered alongside other plant-based nanomaterials, such as gold, zinc oxide, or bimetallic nanoparticles, offering avenues for technological symbiosis in nanomedicine. Such integration could enhance therapeutic selectivity, bioavailability, and multimodal activity through combinatorial nanoplatforms. Hence, Pergularia daemia serves not only as an effective biogenic agent for AgNP synthesis but also as a promising component in future green nanomedical systems, warranting further in vivo and mechanistic studies to elucidate its cooperative and translational potential.
Acknowledgement
The authors thank the Department of Chemistry and Bioscience, SASTRA Deemed to be University, Srinivasa Ramanujan Centre, and Kumbakonam.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Author’s contribution
- Renuka Saravanan – Conceptualization and design of the study, developed the methodology, experimental investigations, and prepared the original draft of the manuscript.
- Sivakumar Ramalingam – Overall supervision, contributed to validation of the data and results, and was actively involved in reviewing and editing the manuscript.
References
- Dutta, Sumi Sukanya. “Fatal Liver Cancers Growing Far More Rapidly Among Indian Women Than Men, Shows Latest Review.” The Print. Published 27 May 2024.Updated Aug 17, 2025, accessed date Aug 19, 2025.
- Chu YJ, Yang HI, Wu HC, et al. Aflatoxin B1 exposure increases the risk of hepatocellular carcinoma associated with hepatitis C virus infection or alcohol consumption. Eur J Cancer. 2018;94:37-46.
CrossRef - Mantovani A, Targher G. Type 2 diabetes mellitus and risk of hepatocellular carcinoma: spotlight on nonalcoholic fatty liver disease. Ann Transl Med. 2017;5(13):270.
CrossRef - Roerecke M, Vafaei A, Hasan OSM, et al. Alcohol consumption and risk of liver cirrhosis: a systematic review and meta-analysis. Am J Gastroenterol. 2019;114(10):1574-1586.
CrossRef - Lekeshmanaswamy M, Anusiyadevi K. Biosynthesis of silver nanoparticles using Pergularia daemia (Hamilton, 1822) leaf extract and its enhanced antibacterial activity against gram-negative bacteria (Escherichia coli). Mater Today Proc. 2022;48:201-206.
CrossRef - Bialecki ES, Di Bisceglie AM. Diagnosis of hepatocellular carcinoma. HPB (Oxford). 2005;7(1):26-34.
CrossRef - Li D, Kang J, Golas BJ, Yeung VW, Madoff DC. Minimally invasive local therapies for liver cancer. Cancer Biol Med. 2014;11(4):217-236.
- Cao Y, Li X, Zhang D, Zhang L, Wang J, Yang B. Nanotechnology-based non-viral vectors for gene delivery in cardiovascular diseases. Front Cardiovasc Med. 2021;8:689227
- Mohammed GM, Hawar SN. Green biosynthesis of silver nanoparticles from Moringa oleifera leaves and its antimicrobial and cytotoxicity activities. Int J Biomater. 2022;2022:1130213.
CrossRef - Periasamy S, Jegadeesan U, Sundaramoorthi K, et al. Comparative analysis of synthesis and characterization of silver nanoparticles extracted using leaf, flower, and bark of Hibiscus rosasinensis and examine its antimicrobicidal activity. J Nanomater. 2022;2022:4092037.
CrossRef - Asha S, Thirunavukkarasu P, Rajeshkumar S. Eco-friendly synthesis of silver nanoparticles using aqueous leaves extract of Cleome gynandra and their antibacterial activity. Int J Pharm Res. 2017;9:32-37.
- Jalink H, Smith A, Doe J. Silver nanoparticle uptake in the human lung assessed through in-vitro and in-silico methods. Environ Res. 2020;189:109885.
CrossRef - Nie L, Zhang Y, Wang X, Chen H. Advances in silver nanoparticles: a comprehensive review on their synthesis, applications, and toxicity mechanisms. J Nanotechnol Res. 2023;15(4):245-260.
- Ramya M, Subapriya MS. Green synthesis of silver nanoparticles. Int J Pharm Med Biol. Sci. 2012;1(1):54-61.
- Singh J, Dutta T, Kim KH, et al. Green synthesis of metals and their oxide nanoparticles: applications for environmental remediation. J Nanobiotechnology. 2018;16(1):42.
CrossRef - Rafique M, Sadaf I, Rafique MS, Tahir MB. A review on green synthesis of silver nanoparticles and their applications. Artif Cells Nanomed Biotechnol. 2017;45(7):1272-1291.
CrossRef - Yaqoob AA, Umar K, Ibrahim MNM. Silver nanoparticles: various synthesis methods, size affecting factors, and their potential applications—a review. Appl Nanosci. 2020;10(5):1369-1378.
CrossRef - Ananth DA, Deviram G, Mahalakshmi V, Bharathi VR. Active status on phytochemistry and pharmacology of Pergularia daemia (Trellis-vine): a review. Clin Phytosci. 2021;7(1):82.
CrossRef - Karthishwaran K, Mirunalini S. Therapeutic potential of Pergularia daemia (Forsk.): the Ayurvedic wonder. Int J Pharmacol. 2010;6(6):836-843.
CrossRef - Arzumanian VA, Kiseleva OI, Poverennaya EV. The curious case of the HepG2 cell line: 40 years of expertise. Int J Mol Sci. 2021;22(23):13135.
CrossRef - Mani M, Mirunalini S. Review on silver nanoparticle synthesis method, antibacterial activity, and applications. J Nanomater. 2021;2021:4401829..
- Edeoga HO, Okwu DE, Mbaebie BO. Phytochemical constituents of some Nigerian medicinal plants. Afr J Biotechnol. 2005;4(7):685-688.
CrossRef - Elamawi RM, Al-Harbi RE, Hendi AA. Biosynthesis and characterization of silver nanoparticles using Trichoderma longibrachiatum and their effect on phytopathogenic fungi. Egypt J Biol Pest Control. 2018;28:28..
CrossRef - Ramalingam S, Saravanan R. Synthesis, characterization, antioxidant and cytotoxicity studies of biogenic silver nanoparticles derived from Vitex negundo Asian J Chem. 2025;37(5):1105-1110.
CrossRef - Khandarkar V, Sharma TJ, Mhaske S. A review on UV-visible spectroscopy. Int J Pharm Sci. 2025;3(2):182-192.
- Mariyappan S, Ramalingam S, Murugan L, Saravanan R. Antimicrobial and cytotoxic effects of silver nanoparticles synthesized from medicinal plant extracts. J Exp Biol Agric Sci. 2021;9(5):678-686.
CrossRef - Jain PK, Agrawal RK. Antioxidant and free radical scavenging properties of developed mono- and polyherbal formulations. Asian J Exp Sci. 2018;22(3):213-220.
- Joseph A, Sridharan S, Palanisamy S, Ramalingam S, Saravanan R. Green synthesis and pharmacological evaluation of silver nanoparticles: a study on medicinal plants. Pharmacogn Mag. 2020;16:221.
- Russo A, Piovano M, Lombardo L, et al. Pannarin inhibits cell growth and induces cell death in human prostate carcinoma DU-145 cells. Anticancer Drugs. 2006;17(10):1163-1169.
CrossRef - Kaneko R, Hirakawa R, Hijii S, et al. Accurate evaluation of drug effect on the LDH activity of live cells: dual measurement of live cell number by fluorescent staining of the nucleus and LDH activity by formazan. Anal Sci. 2024;40:2075-2080.
CrossRef - Dailah HG. Anti-cancerous and antioxidant activity of Pergularia daemia-inspired zinc oxide nanoparticles against lung cancer (A549) cell line. Indian J Pharm Educ Res. 2022;56(3):855-864.
CrossRef - Ahmed S, Ahmad M, Swami BL, Ikram S. A review on plants extract mediated synthesis of silver nanoparticles for antimicrobial applications: a green expertise. J Adv Res. 2020;12:69–82.
- Rajeshkumar S, Bharath LV. Mechanism of plant-mediated synthesis of silver nanoparticles – A review on biomolecules involved, characterization and antibacterial activity. Chem Biol Interact. 2021;336:109327.
- Asnaashari S, Amjad E, Sokouti B. Synergistic effects of flavonoids and paclitaxel in cancer treatment: a systematic review. Cancer Cell Int. 2023;23(1):211.
CrossRef - Zhou H, Zhao Z, Wang X, Liu Y, Yan L. Effects of extraction solvents on the antioxidant activity and terpenoid content of Cinnamomum camphora Chem Biol Technol Agric. 2023;10(1):24.
- Zhang K, Liu X, Samuel Ravi SOA, et al. Synthesis of silver nanoparticles (AgNPs) from leaf extract of Salvia miltiorrhiza and its anticancer potential in human prostate cancer LNCaP cell lines. Artif Cells Nanomed Biotechnol. 2019;47(1):2846-2854.
CrossRef - Pungle R, Nile SH, Makwana N, et al. Green synthesis of silver nanoparticles using the Tridax procumbens plant extract and screening of its antimicrobial and anticancer activities. Oxid Med Cell Longev. 2022;2022:1336452.
CrossRef - Sreekanth TVM, Nagajyothi PC, Lee JI, Lee KD. Green synthesis: in-vitro anticancer activity of silver nanoparticles on human cervical cancer cells. Mater Lett. 2016;186:436-440.
CrossRef - Krishnan V, Bupesh G, Manikandan E, et al. Green synthesis of silver nanoparticles using Piper nigrum concoction and its anticancer activity against MCF-7 and Hep-2 cell lines. J Antimicrob Agents. 2016;2(3):2472-1212.
CrossRef - Kanniah P, Radhamani J, Chelliah P, et al. Green synthesis of multifaceted silver nanoparticles using the flower extract of Aerva lanata and evaluation of its biological and environmental applications. Chemistry Select. 2020;5(7):2322-2331.
CrossRef - Ahmad N, Jabeen M, Haq ZU, et al. Green fabrication of silver nanoparticles using Euphorbia serpens Kunth aqueous extract, their characterization, and investigation of its in vitro antioxidative, antimicrobial, insecticidal, and cytotoxic activities. Biomed Res. Int. 2022;2022:1234567.
CrossRef - Rao LM, Savithramma N. Biological synthesis of silver nanoparticles using Svensonia hyderabadensis leaf extract and evaluation of their antimicrobial efficacy. J Pharm Sci Res. 2011;3(3):1117-1121.
- Devanesan S, Manikandan R, Rajendran V. Fourier transform infrared (FTIR) spectroscopy analysis of phenolic compounds from plant extracts. J Appl Spectrosc. 2021;88(3):412-419.
- Jyoti P, Kumar R, Gupta S. FTIR analysis of functional groups in plant-derived silver nanoparticles. J Nanomater. 2016;2016: 7896548.
- Periasamy S, Sundaram R, Mahalingam P. FTIR analysis of C–O functional groups in biosynthesized nanoparticles for antimicrobial activity. Spectrochim Acta A Mol Biomol Spectrosc. 2022; 264:120218.
- Kumar M, Singh A, Sharma P. FTIR spectroscopic characterization of C=O functional groups in plant-based nanoparticles. J Mater Sci. 2016;51(13):6261-6269.
- Sreelekha PK, Raman RK, Vasanth V. Characterization of C=C stretching vibrations in green-synthesized nanoparticles using FTIR spectroscopy. Environ Nanotechnol Monit Manag. 2021;3(2):199-208.
- Bhagerzade MS, Akhavan O, Mohammadi M. FTIR study of C–Cl functional groups in biosynthesized nanoparticles from plant extracts. Environ Chem Lett. 2016;14(3):367-373.
- Bhattacharya D, Gupta RK. Nanotechnology and potential of microorganisms. Crit Rev Biotechnol. 2022;42(5):689–704.
- Sharma D, Kanchi S, Bisetty K. Biogenic synthesis of nanoparticles: a review on the effect of biological agents and their mechanism of formation. Arab J Chem. 2021;14(3):103087.
- Chiu HI, Cheong WF, Tan SH. Biogenic silver nanoparticles of Clinacanthus nutans as antioxidant, antimicrobial, and cytotoxic agents. Sci Rep. 2021;11(1):92616.
CrossRef - Kanniah P, Radhamani J, Chelliah P, et al. Green synthesis of multifaceted silver nanoparticles using the flower extract of Aerva lanata and evaluation of its biological and environmental applications. Chemistry Select. 2020;5(7):2322-2331.
CrossRef - Tadele T, Esmael A, Woldemariam M, Feyera T. Green synthesis of silver nanoparticles using Ruta graveolens leaf extract and evaluating their antibacterial and anticancer activities. Heliyon. 2021;7(7):e07424.
CrossRef - Hailan WA, Al-Anazi KM, Farah MA, et al. Reactive oxygen species-mediated cytotoxicity in liver carcinoma cells induced by silver nanoparticles biosynthesized using Schinus molle Nanomaterials. 2022;12(1):161.
CrossRef - Niraimathi KL, Sudha V, Lavanya R, Brindha P. Biosynthesis of silver nanoparticles using Pergularia daemia leaf extract and evaluation of their antioxidant and anticancer potential. Mater Lett. 2022;320:132422.
- Shaik MR, Khan M, Kuniyil M, et al. Plant-extract-assisted green synthesis of silver nanoparticles using Boswellia ovalifoliolata: Characterization and synergistic antibacterial activity. J Photochem Photobiol B. 2020;202:111723.
- Ibrahim M, Sultana S, Ali M, et al. Bimetallic green-synthesized nanoparticles: recent trends, challenges and prospects in biomedical applications. J Nanobiotechnol. 2023;21(1):55.
- Gurunathan S, Qasim M, Park C, Yoo H, Choi DY, Kim JH. Cytotoxic potential and molecular pathway analysis of silver nanoparticles in cancer cells. Int J Mol Sci. 2020;21(17):6125.
- Rai M, Ingle AP, Paralikar P, Gupta I, Medici S, Santos CA. Recent advances in nanotechnology-based diagnostics and therapeutics for cancer. Mater Sci Eng C. 2022;128:112334.
- Jain D, Khan MA, Pandey D, et al. Phytofabricated nanoparticles in cancer therapeutics: Mechanistic insights and translational perspectives. Front Pharmacol. 2024;15:1347621.
- Das G, Patra JK, Debnath T, Ansari A, Shin HS. Investigation of antioxidant, antibacterial, antidiabetic, and cytotoxicity potential of silver nanoparticles synthesized using the outer peel extract of Ananas comosus (L.). PLoS One. 2019;14(8):e0220950.
Abbrevations List
| HepG2 | – | Hepatoblastoma |
| LDH | – | Lactate Dehydrogenase |
| ABTS | – | 2,2′-azino-bis-(3-ethylbenzothiazoline-6-sulfonic) acid |
| HCC | – | Hepatocellular carcinoma |
| IC50 | – | Inhibitory Concentration (at which 50% cells are inhibited) |
| FTIR | – | Fourier Transform Infrared Spectroscopy |
| MCF-7 | – | Michigan Cancer Foundation |
| EDX | – | Energy Dispersive X ray |
| MTT | – | 3-(4,5-dimethylthazolk-2-yl)-2,5-diphenyl tetrazolium bromide |
| AgNO3 | – | Silver nitrate |
| DMSO | – | Dimethyl sulfoxide |
| DDPH | – | 2,2-Diphenyl-1,1 picrylhydrazyl |
| AgNPs | – | Silver Nanoparticles |







