Manuscript accepted on :24-12-2025
Published online on: 27-01-2026
Plagiarism Check: Yes
Reviewed by: Dr. Niharika Kondepudi
Second Review by: Dr. Surendra Kumar Swarnkar
Final Approval by: Dr. Mariia Shanaida
Tasnia Binte Bari Kabbo1 , Md. Sohel Rana1,5*
, Md. Sohel Rana1,5* , Sukanta Deb2
, Sukanta Deb2 , Farjana Afrin3
, Farjana Afrin3 , Md. Shajalal Reza4, Shahriar Mohammad Shohan3
, Md. Shajalal Reza4, Shahriar Mohammad Shohan3 and Pritesh Ranjan Dash1,5*
and Pritesh Ranjan Dash1,5*
1Department of Pharmacy, Jahangirnagar University, Savar, Dhaka, Bangladesh
2Department of Pharmacy, University of Science and Technology Chittagong (USTC) Chittagong, Bangladesh
3Department of Pharmacy, Primeasia University, Banani, Dhaka, Bangladesh
4Department of Pharmacy, Hamdard University Bangladesh, Gazaria, Munshigonj, Bangladesh
3Department of Pharmacy, ASA University Bangladesh, Shyamoli, Dhaka, Bangladesh
Corresponding Author E-mail: pritesh.ju@gmail.com and sohelrana.ju@juniv.edu
DOI : https://dx.doi.org/10.13005/bpj/3389
Abstract
Annona reticulata Linn. was investigated for sedative, anxiolytic, and antidepressant properties. Tail suspension and forced swimming experiments were utilized for assessing antidepressant potentials of methanol and aqueous extracts; test fractions notably lowered immobility time in both tests; demonstrating strong antidepressant activities (p<0.001) at 400 mg/kg bodyweight dose. Through hole board test, methanolic and aqueous leaf fractions also demonstrated significant anxiolytic effects (p<0.001); the obtained outcomes for test extracts at 400 mg/kg dose were even higher than the outcomes of standard drug diazepam. Furthermore, thiopental sodium-induced sleeping time and hole cross tests were utilized to assess sedative effectiveness of test fractions at 200 and 400 mg/kg dosages. In hole cross test, after 120 minutes of drug administration, observed hole crossing values for 400 mg/kg dose of methanol and aqueous extracts were much higher than the outcome of standard drug diazepam. Additionally, in TPS induced sleeping time assay, the obtained onset and duration of sleep values (p<0.05) from the mice groups treated with 400 mg/kg dose of methanol and aqueous extracts were less significant in comparison to the outcomes (p<0.001) of standard drug diazepam. Moreover, GC-MS analysis was performed to identify probable compounds causing these actions.
Keywords
Annona reticulata Linn.; Anxiolytic; Antidepressant; Extract; Sedative87
Download this article as:| Copy the following to cite this article: Kabbo T. B. B, Rana M. S, Deb S, Afrin F, Reza M. S, Shohan S. M, Dash P. R. Evaluation of Neuropharmacological Activities of Different Fractions of Annona reticulata Linn. Leaves. Biomed Pharmacol J 2026;19(1). |
| Copy the following to cite this URL: Kabbo T. B. B, Rana M. S, Deb S, Afrin F, Reza M. S, Shohan S. M, Dash P. R. Evaluation of Neuropharmacological Activities of Different Fractions of Annona reticulata Linn. Leaves. Biomed Pharmacol J 2026;19(1). Available from: https://bit.ly/4bUpI4x |
Introduction
For many years, GC-MS has been utilized to analyse biological samples for identifying probable components.1 A combination of genetic, physiological, environmental, and psychological factors has been shown to produce depression. The fact that some but not all forms of depression are inherited emphasizes the interplay between gene – gene and gene environment. By 2020, though, it will rank as the second most common cause of death.2-3Anxiety is a psychological disease that is defined as an unpleasant emotional state that has an unclear explanation or is thought to be uncontrollable or inevitable. Phobia, or persistent fear, has been linked to anxiety disorders. About 40% of patients with disorders of CNS worldwide experience anxiety and sleeplessness.4 Statistics demonstrate that 10% of individuals suffer from various forms of anxiety, while 30% of adults from insomnia around the globe.5 Psychotropic medications known as antidepressants are prescribed to treat mental illnesses exhibiting significant signs of emotional depression.6 Drugs known as sedatives and hypnotics can lower anxiety and have a soothing impact by promoting sleep and extending the length of sleep.7 Prolonged use of conventional sedative-hypnotic, anxiolytic and anti-depressant treatments, can have major adverse consequences that range from immunological, digestive, and respiratory system malfunction to decline in cognitive function, tolerance, and physical dependence. The formulation of novel medications with less adverse effects has thus been proposed as a viable strategy to treat various mental illnesses. In order to achieve superior therapeutic outcomes and minimal side effects, look for natural alternatives to conventional medicines, such as herbal treatments, which have been used for centuries and are now known to have safe profile.8-9Annona reticulata Linn., a member of the Annonaceae family, is also referred to as Sitaphala, Sarifa, and Custard-Apple. Annona reticulata Linn. is a deciduous tree and fruit-eating plant that is widely grown in Bangladesh. It is also grown in India and Pakistan butis indigenous to South America and the West Indies. The plant Annona reticulata Linn. is widely used in the Ayurvedic medical system to treat a wide range of illnesses. Epilepsy, diarrhoea, heart issues, worm infestation, constipation, bleeding, antibacterial infection, dysuria, fever, and ulcers have all historically been treated with the plant. It also possesses abortifacient, antitumor, and antifertility potentials.10-11In this study, the plant’s sedative, anxiolytic, and antidepressant properties were evaluated.
Materials and Methods
Preparation of Plant Extracts
Annona reticulata Linn. leaves were obtained from Savar, Dhaka. Dr. Khandakar Kamrul Islam, Senior Scientific Officer of Bangladesh National Herbarium, then identified the leaves. The sample was kept at Bangladesh National Herbarium (Voucher number: DACB 114865). The plant material (leaves) was sun-dried seperately before being dried in a Gallenkamp air oven (Size 1) at a lower temperature (no more than 50°C) until it was ready to be grinded. The plant components were then grinded into coarse powders using a large-capacity grinding mill. Then, 12.6 litres of methanol and 10.76 liters of water were used to seal 630 g and 538 g of prepared dry plant material in dark-coloured flasks. 24 hours later, filtration of infusions was performed. The process was repeated 48 hours later. Extraction was considered to be complete when it was determined that the plant materials had been exhausted. Extracts underwent fresh cotton bed filtering process. Resultant filtrates were dried at 40±2°C to create sticky crude extract. After being extracted, the materials were stored in labelled sterile containers at 4°C.12
GC-MS Analysis of Methanolic and Aqueous Leaf Extracts
Shimadzu GCMS-TQ8040 was used to perform GC-MS analysis. Helium (carrier gas) was used to successfully isolate and identify the analytes. According to the GC parameters, column oven’s temperature was 50°C, ramped up to 200°C, and eventually reached 300°C. The hold times were 1, 2, and 7 minutes, respectively. The injection was carried out at 250°C with sample duration of one minute, flow control mode set to pressure at 53.5 kPa, and total flow of 11 ml/min. Under MS circumstances, 230°C and 250°C were temperatures of ion source and surface. This configuration provides excellent sensitivity and specificity for identifying and quantifying volatile and semi-volatile chemicals in complicated plant extracts by comparing them to library compounds.13
Experimental Animals
Swiss albino mice (25 and 30 g) were gathered from department’s own animal house. Methanolic and aqueous leaf extracts of Annona reticulata Linn. were studied pharmacologically in the Jahangirnagar University Pharmacology Laboratory. Mice were placed under standard laboratory settings in cages made of polypropylene. Mice were fed pelletised mouse feed from International Centre for Diarrhoeal Disease Research, Bangladesh (ICDDR,B), and allowed unlimited access to water. Every action that involved working with animals was done in compliance with rules set forth by the university’s animal ethics committee [Ref No: BBEC, JU/M2024/11 (137)].
Assessment of In-vivo Antidepressant Effect
Tail Suspension Test
As reported by Steru L. et al., antidepressant potentials of methanol and aqueous extracts made from Annona reticulata Linn. leaves were examined using tail suspension method. To begin with, six groups of five mice were assembled. After weighing the mice, labelled each one with a tail number. Mice of group I received an oral dose of distilled water and group II administered Fluoxetine hydrochloride (20 mg/kg; via oral route), based on their body weight. Each mouse in groups III and IV was administered 200 and 400 mg/kg of methanolic leaf fraction orally. Mice of groups V and VI received aqueous leaf extract at 200 and 400 mg/kg dosages. With the aid of sticky tape, mice were hung from the top of the wooden frame, which was 50 cm above the floor. Over the course of 5 minutes, total amount of immobility brought on by tail suspension was noted. After 1 hour of the dose administration, the immobility duration and proportion of antidepressant activity for each group were measured.14-15
Forced Swimming Test
Antidepressant properties of methanolic and aqueous leaf fractions from Annona reticulata Linn. were assessed utilizing forced swimming test, according to Porsolt et al. Six groups, each consisting of five mice, were first put together and given a tail number on each mouse once they have been weighed. Glass cylinder (20 cm high; 10 cm in diameter), contained 10 cm depth of water (25°C). Mice were taken out of water five minutes later and dried up before putting back in their own cages after five minutes. After 24 hours, mice were treated with following treatments according to their groups. Distilled water and imipramine hydrochloride (15 mg/kg) were administered via oral route to mice of group I and II. 200 and 400 mg/kg oral doses of methanolic leaf fraction were given to each mouse in groups III and IV. Aqueous extract was given at 200 and 400 mg/kg dosages via oral route to mice of group V and VI. 1 hour after administration of treatments, the subjects were once more placed inside the cylinder. Length of immobility time was observed for five minutes after first one-minute’s acclimatization phase. Percentages of antidepressant activities were computed utilizing established formula.14-15
Evaluation of In-vivo Anxiolytic Activity
Hole Board Test
Hole board device consisted of 40 × 40 × 25 cm³ wooden chamber with 16 randomly placed, 3 cm diameter holes on the floor. For the mice to be able to peek through the holes, the device was raised 25 cm off the ground. Six groups of five mice each were first assembled. Each mouse was then given a tail number and weighed. Each mouse in control groups I and II received distilled water and diazepam (1 mg/kg) via oral route, respectively. The doses for mice in group III and IV were 200 and 400 mg/kg of methanolic fraction. Mice in group V and VI received 200 and 400 mg/kg doses of aqueous extract. Test animals were placed in the centre of hole board and allowed to freely inspect the apparatus for five minutes; 30 minutes later. On the board, quantity of head dipping and line crossings were recorded.5,16
Evaluation of In-vivo Sedative Activity
Hole Cross Test
This experiment made use of wooden equipment that measured 30 × 20 × 14 cm3 and in the centre of the equipment was a partition with a 3 cm diameter hole that was 7.5 cm high. Six sets of five mice each were first taken. Each mouse was then given a tail number and weighed. Mice in control groups I and II received oral distilled water and diazepam (2 mg/kg), respectively. Mice in group III and IV were administered methanolic fraction at 200 and 400 mg/kg doses via oral route. Aqueous extract was given to mice of group V and VI at 200 and 400 mg/kg dosages, orally. Following that, the animals were placed inside the apparatus, and at 0, 30, 60, 90, and 120 minutes, the number of times they passed through the hole from one chamber to another was computed carefully for three minutes. After that, percentage inhibition of movements was measured.17-18
Thiopental Sodium Induced Sleeping Time Assay
Current study used methodology outlined by Turner (1965). Initially, six groups of five mice each were put together. After that, each mouse was weighed and assigned a tail number. Each mouse in group I and II received distilled water and diazepam (2 mg/kg). Methanolic leaf fraction at 200 and 400 mg/kg dosages were administered to mice in group III and IV. Mice of group V and VI received 200 and 400 mg/kg doses of aqueous leaf fraction. 30 minutes after that, thiopental sodium (40 mg/kg) was administered intraperitoneally to each test animal for induction of sleep. Animals’ latent period and sleep duration were monitored and percentage of effect was then computed.19-20
Obtained data was analysed using SPSS 27. The experimental findings were presented using mean±SEM. ANOVA was used to compare the significance to the control at each P value threshold (*p<0.05, **p<0.01, ***p<0.001) for ascertaining statistical significance.
Results
GC-MS Analysis Reports of Methanolic and Aqueous Fractions of Annona reticulata Linn. Leaves
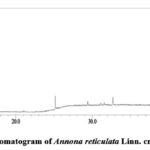 |
Figure 1: Total ionic chromatogram of Annona reticulata Linn. crude methanolic fraction
|
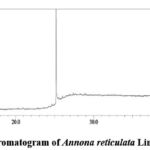 |
Figure 2: Total ionic chromatogram of Annona reticulata Linn.crude aqueous extract
|
The library search report indicated that there were 43 and 77 components in crude methanolic and aqueous fractions from Annona reticulata Linn. leaves, respectively. The identified compounds of methanolic leaf extract are: Hexadecanoic acid, methyl ester; Tridecanoic acid, 12-methyl-, methyl ester; Tridecanoic acid, methyl ester; Decanoic acid, methyl ester; Methyl tetradecanoate; 9,12-Octadecadienal; Dichloroacetic acid, dodec-9-ynyl; E,E-1,9,17-Docasatriene; 9-Dodecyn-1-ol; 3-Tetradecyne; 10-Undecyn-1-ol; 13-Tetradece-11-yn-1-ol; E-1,6-Undecadiene; 11,14,17-Eicosatrienoic acid, methyl ester; cis,cis,cis-7,10,13-Hexadecatrienal; 5-Pentadecen-7-yne, (Z)-; 3-Heptadecen-5-yne, (Z)-; 1-Tetradecen-3-yne; Phytol; 3,4-Dimethylcyclohexanol; 1,7-Octadien-3-ol, 2,6-dimethyl-; Oxirane, decyl-; Methyl stearate; Eicosanoic acid, methyl ester; Octadecanoic acid, 17-methyl-, methyl ester; Methyl tetradecanoate; 13-Docosenamide, (Z)-; 9-Octadecenamide, (Z)-; 8-Methyl-6-nonenamide; 3-Isopropoxy-1,1,1,5,5,5-hexamethyl-3-(trimethyl); Thymol, TMS derivative; 1,2-Bis(trimethylsilyl)benzene; 1-Isopropoxy-5-propyl-2,3-bis-trimethylsilyl-1; 2-(N,N’,N’-Trimethylhydrazino)-1,3-benzothiazole; 1-hexadecanesulfonamide, N-(3-aminopropyl)-; 3,5-bis(trimethylsilyl)cyclohepta-2,4,6-trien-1-one; Trisiloxane, 1,1,1,5,5,5-hexamethyl-3,3-bis(trimethylsilyl)oxy-; Trimethylsilyl-di(timethylsiloxy)-silane; Pterin-6-carboxylic acid; 1,4-Bis(trimethylsilyl)benzene; Cholesterol 3-O-[[2-acetoxy]ethyl]-; Androstane-11,17-dione, 3-[(trimethylsilyl)oxy; 1,5,6,7-Tetramethylbicyclo[3.2.0]hept-6-en-3-yl. The detected compounds from aqueous leaf extract are: Cyclopropyl carbinol; Thiophene-3-ol, tetrahydro-, 1,1-dioxide; Octodrine; 1-(5-Bicyclo[2.2.1]heptyl)ethylamine; 1-Methyldecylamine; 2-Heptanamine, 5-methyl-; Benzeneethanamine, 2,5-difluoro-.beta.,3,4-trihexyl-; Cystine; Acetic acid, [(aminocarbonyl)amino]oxo-; 4-Fluorohistamine; Cyclobutanol; 1-Methyldecylamine; Pentanal; D-Alanine; Eicosanoic acid, methyl ester; Triacontanoic acid, methyl ester; Docosanoic acid, methyl ester; Tridecanoic acid, 12-methyl-, methyl ester; Octadecanoic acid, 17-methyl-, methyl ester; 1,2-Ethanediamine, N-(2-aminoethyl)-; 2-Methylaminomethyl-1,3-dioxolane; Alanine; 1,2-Ethanediamine, N-methyl-; Propanamide; 2-Propanamine, 1-methoxy-; dl-Alanine; Hydroxyurea; 1-Propanol, 2-amino-, (.+/-.)-; 2-Hexanamine, 4-methyl-; Ethanol, 2-(methylamino)-; 1,3-Dioxolane-4-methanol; N-dl-Alanylglycine; 1-Octadecanamine, N-methyl-; dl-Alanyl-dl-norleucine; (S)-(+)-1-Cyclohexylethylamine; 1-Methyldecylamine; Benzeneethanamine, 2-fluoro-.beta.,3-dihydroxy-; Propanamide, N-methyl-2-amino-; 2-Methylaminomethyl-1,3-dioxolane; Formamide, N,N-dimethyl-; Propanediamide, 2-amino-; L-Alanine, TMS derivative; 1,3-Dioxolane-4-methanol; 2-(Methylamino)ethanol, O-trimethylsilyl-; 13-Docosenamide, (Z)-; 3-Ethoxy-1,1,1,5,5,5-hexamethyl-3-(trimethylsilyl); Trisiloxane, 1,1,1,5,5,5-hexamethyl-3,3-bis[(trimethylsilyl)oxy]-; Heptasiloxane, 1,1,3,3,5,5,7,7,9,9,11,11,13,13-hexadecamethyl-; 2,4,6-Cycloheptatrien-1-one, 3,5-bis-trimethylsilyl; Hexestrol, 2TMS derivative; 3-Butoxy-1,1,1,5,5,5-hexamethyl-3-(trimethylsilyl); Methyltris(trimethylsiloxy)silane; 1-Isopropoxy-5-propyl-2,3-bis-trimethylsilyl-1; Thymol, TMS derivative; Silane, trimethyl[5-methyl-2-(1-methylethyl)phenoxy]silane; Trimethylsilyl-di(timethylsiloxy)-silane; 3-Isopropoxy-1,1,1,5,5,5-hexamethyl-3-(trimethyl)-; 1,4-Bis(trimethylsilyl)benzene; Ethyl homovanillate, TMS derivative; 1,2-Bis(trimethylsilyl)benzene; 3-Ethoxy-1,1,1,5,5,5-hexamethyl-3-(trimethylsilyl); Trisiloxane, 1,1,1,5,5,5-hexamethyl-3,3-bis[(trimethylsilyl)oxy]-;1-Isopropoxy-5-propyl-2,3-bis-trimethylsilyl-1,2-dihydroborinine; 3-Isopropoxy-1,1,1,5,5,5-hexamethyl-3-(trimethylsiloxy)trisiloxane; Methyltris(trimethylsiloxy)silane; Tetrasiloxane, decamethyl-; 4-Hydroxyphenyllactic acid, ethyl ester, di-TM; Benzoic acid, 4-methyl-2-trimethylsilyloxy-; Hexasiloxane, 1,1,3,3,5,5,7,7,9,9,11,11-dodeca; 7,7,9,9,11,11-Hexamethyl-3,6,8,10,12,15-hexa; (R)-(-)-Phenylephrine, bis(trimethylsilyl) ether; Silicic acid, diethyl bis(trimethylsilyl) ester; 3-Methylsalicylic acid, 2TMS derivative; Octasiloxane, 1,1,3,3,5,5,7,7,9,9,11,11,13,13,15,15-hexadecamethyl-; Methyltris(trimethylsiloxy)silane; 7,7,9,9,11,11-Hexamethyl-3,6,8,10,12,15-hexa; Tetrasiloxane, decamethyl-.
Assessment of In-vivo Antidepressant Effect
Tail Suspension Test
During tail suspension test, the lengths of time animals were immobile,were carefully monitored and measured. It acted as gauge for test compounds’ antidepressant potential. Immobility time of standard group was significantly shorter, numerically 79.25±4.59 seconds (p<0.001) than that of the negative control group, which had an immobility time of 158.25±5.38 seconds. The animals’ reduced immobility indicated that fluoxetine hydrochloride, has potent antidepressant effect. With an immobility time value of 72.00±3.76 seconds (p<0.001), test animals that were administered 400 mg/kg of methanolic fraction illustrated strong performance. Additionally, even at dosage of 200 mg/kg, methanolic leaf fraction showed good antidepressant effect, 98.25±2.56 seconds was measured immobility time value (p<0.001).At dosage of 400 mg/kg, aqueous leaf fraction demonstrated strong activity; this group’s observed immobility time was 63.00±3.54 seconds (p<0.001). Furthermore, aqueous extract demonstrated good antidepressant effect even at 200 mg/kg dose, the immobility time value for this dosage was 86.75±4.66 seconds (p<0.001) (Table 1). Consequently, both fractions demonstrated dose-dependent antidepressant effects.The test samples’ antidepressant potentialsdeclined in the tail suspension test in the following order:
AEAR 400> MEAR 400> Standard> AEAR 200> MEAR 200
Table 1: Effect of Negative control, Standard, MEAR and AEAR in Tail Suspension Test
|
Group |
Time in Immobile Phase (Second) (Mean ± SEM) |
Percentage (%) of Antidepressant Activity |
|
Negative Control |
158.25±5.38 |
_ |
|
Standard |
79.25±4.59*** |
49.92% |
|
MEAR 200 |
98.25±2.56*** |
37.91% |
|
MEAR 400 |
72.00±3.76*** |
54.51% |
|
AEAR 200 |
86.75±4.66*** |
45.18% |
|
AEAR 400 |
63.00±3.54*** |
60.19% |
*p<0.05 (Significance); **p<0.01 (Highly significance); ***p<0.001 (Very highly significance) against control; Standard is Fluoxetine hydrochloride.Here, MEAR = Methanol extract of Annona reticulata Linn. AEAR = Aqueous extract of Annona reticulata Linn.
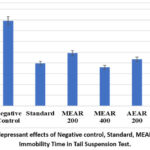 |
Figure 3: Antidepressant effects of Negative control, Standard, MEAR and AEAR on Immobility Time in Tail Suspension Test.
|
Forced Swimming Test
During forced swimming test, amount of time that the animals remained still without moving or trying to escape from the water was recorded. The immobility time count for the imipramine hydrochloride-treated standard group was 83.75±4.13 seconds (p<0.001), roughly half of the negative control group’s immobility time of 164.25±3.07 seconds. Methanolic fraction also demonstrated good antidepressant efficacy at 400 mg/kg with 74.50±3.50 seconds immobility time value (p<0.001). 200 mg/kg dose of methanolic fraction exhibited good antidepressant effect; the immobility time value was 94.50±5.14 seconds (p<0.001). Additionally, at dose of 400 mg/kg, aqueous leaf fraction demonstrated significantactivity; this group’s immobility time value was 64.25±4.21 seconds (p<0.001), which is even less than that of the standard group. The immobility time value for 200 mg/kg dosage of aqueous fraction was 85.00±2.89 seconds (p<0.001) (Table 2). Methanolic and aqueous leaf fractions thus depicted dose-dependent antidepressant effectiveness in this study. Therefore, antidepressant potentials of the test samples decreased in the following order in this study:
AEAR 400> MEAR 400> Standard> AEAR 200> MEAR 200
Table 2: Effect of Negative control, Standard, MEAR and AEAR in Forced Swimming Test.
|
Group |
Time in Immobile Phase (Second) (Mean ± SEM) |
Percentage (%) of Antidepressant Activity |
|
Negative Control |
164.25±3.07 |
_ |
|
Standard |
83.75±4.13*** |
49.01% |
|
MEAR 200 |
94.50±5.14*** |
42.47% |
|
MEAR 400 |
74.50±3.50*** |
54.64% |
|
AEAR 200 |
85.00±2.89*** |
48.25% |
|
AEAR 400 |
64.25±4.21*** |
60.88% |
*p<0.05 (Significance); **p<0.01 (Highly significance); ***p<0.001 (Very highly significance) against control; Standard is Imipramine hydrochloride. Here, MEAR = Methanol extract of Annona reticulata Linn. AEAR = Aqueous extract of Annona reticulata Linn.
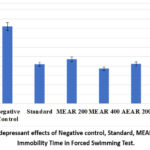 |
Figure 4: Antidepressant effects of Negative control, Standard, MEAR and AEAR on Immobility Time in Forced Swimming Test.
|
Evaluation of In-vivo Anxiolytic Activity
Hole Board Test
Two indicators in this investigation—head dipping into the holes and line crossing on the board—were meticulously observed in order to assess the test samples’ anxiolytic activities. In negative control group, mice dipped their heads 19.25±0.85 times and crossed 61.25±0.85 designated lines on the board. In contrast, head dipping number of diazepam-treated standard group was 39.75±1.38 (p<0.001), which was more than twice that of the negative control group. Additionally, standard group also had almost twice as many line crossings (118.75±1.11; p<0.001) as negative control group. Moreover, animals in group-IV demonstrated head dipping and line crossing numbers of 41.25±0.85 and 117.25±1.72 (p<0.001), which are greater than those of standard group, indicating 400 mg/kg dosage of methanol extract’s good anxiolytic efficacy. In addition, methanol leaf extract exhibited anxiolytic effecteven at 200 mg/kg. Animals given dose of 200 mg/kg of methanol extract dipped their heads 30.00±0.91 times and crossed 78.25±1.93 lines (p<0.001) on the board. Mice group which administered 400 mg/kg dose of aqueous extract; dipped their head 42.25±0.75 times (p<0.001); crossed 120.25±1.32 lines (p<0.001) on the board; values obtained from this group are higher than that of the standard group. Furthermore, head dipping value was 31.75±1.11 and line crossing value was 82.25±1.65 (p<0.001) in the group that received 200 mg/kg dose of aqueous extract (Table 3). Therefore, both leaf fractions showed dose-dependent anxiolytic properties in this study. The test samples’ anxiolytic activities declined in the following sequence in the hole board test:
AEAR 400> MEAR 400> Standard> AEAR 200> MEAR 200
Table 3: Effect of Negative control, Standard, MEAR and AEAR in Hole Board Test.
|
Group |
No. of Head Dipping (Mean ± SEM) |
No. of Line Crossing (Mean ± SEM) |
|
Negative Control |
19.25±0.85 |
61.25±0.85 |
|
Standard |
39.75±1.38*** |
118.75±1.11*** |
|
MEAR 200 |
30.00±0.91*** |
78.25±1.93*** |
|
MEAR 400 |
41.25±0.85*** |
117.25±1.72*** |
|
AEAR 200 |
31.75±1.11*** |
82.25±1.65*** |
|
AEAR 400 |
42.25±0.75*** |
120.25±1.32*** |
*p<0.05 (Significance); **p<0.01 (Highly significance); ***p<0.001 (Very highly significance) against control; Standard is Diazepam. Here, MEAR = Methanol extract of Annona reticulata Linn.AEAR = Aqueous extract of Annona reticulata Linn.
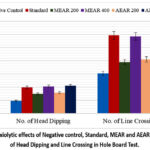 |
Figure 5: Anxiolyticeffects of Negative control, Standard, MEAR and AEAR on Number of Head Dipping and Line Crossing in Hole Board Test.
|
Assessment of In-vivo Sedative Potential
Hole Cross Test
In this investigation, sedative potentials of test samples were ascertained by counting the number of times the animals passed the test apparatus’s hole during experimental period at predetermined intervals. The animals of negative control group crossed the hole 13.00±0.91, 11.50±0.65, 12.50±0.65, 13.00±0.71 and 13.50±0.65 times at 0, 30, 60, 90 and 120 minutes. The standard group’s mice that were given diazepam, however, crossed the hole 11.50±0.65, 7.25±0.48, 5.00±0.41, 3.50±0.29, and 2.25±0.48 times (p<0.001) at 0, 30, 60, 90, and 120 minutes. A clear indication of sedative potential of diazepam was the reduction in hole crossing number seen in the animals of standard group over time following administration. Animals given 400 mg/kg of methanol extract crossed the hole 12.50±0.65, 10.50±0.48, 9.50±0.48(p<0.05), 8.50±0.41(p<0.001), and 8.00±0.29 times (p<0.001) at 0, 30, 60, 90, and 120 minutes; the values obtained at 60, 90 and 120 min are quite significant in comparison to negative control group. In addition, mice that administered 200 mg/kg of methanolic leaf fraction at 0, 30, 60, 90, and 120 minutes displayed hole crossing counts of 11.75±0.85, 11.25±0.29, 10.50±0.65, 9.00±0.41(p<0.05), and 8.75±0.29 (p<0.001). On the other hand, mice that were administered 400 mg/kg of aqueous extract traversed the hole 12.50±0.65, 10.75±0.48, 9.75±0.48 (p<0.05), 8.75±0.29 (p<0.01), and 8.25±0.25 (p<0.001) times at 0, 30, 60, 90, and 120 minutes; the values obtained after 60, 90 and 120 min of aqueous extract administration are considerably lower than those of negative control group. Additionally, at 0, 30, 60, 90, and 120 minutes, the mice that were administered 200 mg/kg dosage of aqueous fraction had the following hole crossing counts: 12.50±0.65, 11.50±0.65, 10.75±0.29, 9.50±0.29 (p<0.05), and 9.00±0.25 (p<0.01) (Table 4). By comparing with the outcomes given by standard drug it can be said that both extracts are found to possess mild sedative activities; but the sedative actions of the test extracts can be seen after 60 min of administration in case of 400 mg/kg dosage and after 90 min of administration in case of 200 mg/kg dosage. According to the results, methanolic and aqueous leaf fractions in this investigation demonstrated dose-dependent sedative effects. The sedative activities of the test samples declined in the hole cross test in the following order:
Standard>MEAR 400>AEAR 400>MEAR 200>AEAR 200
Table 4: Effect of Negative control, standard, MEAR and AEAR in Hole Cross Test.
|
Group |
Number of Hole Cross (Mean ± SEM) |
||||
|
0 min |
+30 min |
+60 min |
+90 min |
+120 min |
|
|
Negative Control |
13.00±0.91 |
11.50±0.65 |
12.50±0.65 |
13.00±0.71 |
13.50±0.65 |
|
Standard |
11.50±0.65 |
7.25±0.48*** |
5.00±0.41*** |
3.50±0.29*** |
2.25±0.48*** |
|
MEAR 200 |
11.75±0.85 |
11.25±0.29 |
10.50±0.65 |
9.00±0.41* |
8.75±0.29** |
|
MEAR 400 |
12.50±0.65 |
10.50±0.48 |
9.50±0.48* |
8.50±0.41*** |
8.00±0.29*** |
|
AEAR 200 |
12.50±0.65 |
11.50±0.65 |
10.75±0.29 |
9.50±0.29* |
9.00±0.25** |
|
AEAR 400 |
12.50±0.65 |
10.75±0.48 |
9.75±0.48* |
8.75±0.29** |
8.25±0.25*** |
*p<0.05 (Significance); **p<0.01 (Highly significance); ***p<0.001 (Very highly significance) against control; Standard is Diazepam.Here, MEAR = Methanol extract of Annona reticulata Linn.AEAR = Aqueous extract of Annona reticulata Linn.
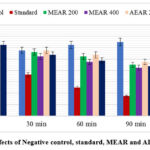 |
Figure 6: SedativeEffects of Negative control, standard, MEAR and AEAR in hole cross test.
|
Thiopental Sodium Induced Sleeping Time Assay
To ascertain test samples’ sedative action, the onset and duration of sleep after thiopental sodium administration were carefully monitored in the assay. The negative control group fell asleep 2.75±0.32 minutes after receiving thiopental sodium and slept for 63.25±0.75 minutes. In contrast, standard group that took Diazepam started to sleep 1.38±0.24 minutes after thiopental sodium was administered (p<0.01) and slept for 168.00±1.78 minutes (p<0.001). Standard group slept three times as long as the negative control group. When methanolic fraction (400 mg/kg) was administered to test animals, their sleeping began at 1.63±0.32 minute (p<0.05) and lasted for 107.75±2.56 minutes (p<0.05) following thiopental sodium administration. In addition, mice that administered 200 mg/kg dose of test fraction fell asleep 1.87±0.13 minutes following administration of thiopental sodium; mice that received methanol extract (200 mg/kg) slept for 90.50±2.10 minutes. On the other hand, mice that were administered 400 mg/kg of aqueous extract began to sleep 1.50±0.21 minutes (p<0.05) after thiopental sodium was administered, and they slept for 103.25±1.75 minutes (p<0.05). After taking 200 mg/kg dosage of aqueous extract, mice began to sleep 1.75±0.32 minutes after thiopental sodium administration and slept for 87.75±1.49 minutes (Table 5). From the outcomes of the investigation, it can be said that, both methanol and aqueous extracts exhibited mild sedative potential at high dose (400 mg/kg) but in low dose the observed sedative activity was insignificant. Therefore, methanolic and aqueous leaf fractions demonstrated dose-dependent mild sedative effects in this study. Test samples’ sedative activities declined in the following order in the thiopental sodium-induced sleeping time assay:
Standard>MEAR 400>AEAR 400>MEAR 200>AEAR 200
Table 5: Effect of Negative control, Standard, MEAR and AEAR on Onset and Duration of Sleeping in Thiopental Sodium Induced Sleeping Time Test
|
Group |
Onset of Sleeping (Minutes) (Mean ± SEM) |
Duration of Sleeping (Minutes) (Mean ± SEM) |
|
Negative Control |
2.75±0.32 |
63.25±0.75 |
|
Standard |
1.38±0.24** |
168.00±1.78*** |
|
MEAR 200 |
1.87±0.13 |
90.50±2.10 |
|
MEAR 400 |
1.63±0.32* |
107.75±2.56* |
|
AEAR 200 |
1.75±0.32 |
87.75±1.49 |
|
AEAR 400 |
1.50±0.21* |
103.25±1.75* |
*p<0.05 (Significance); **p<0.01 (Highly significance); ***p<0.001 (Very highly significance) against control; Standard is Diazepam.Here, MEAR = Methanol extract of Annona reticulata Linn.AEAR = Aqueous extract of Annona reticulata Linn.
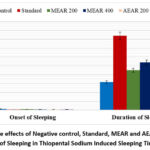 |
Figure 7: Sedative effects of Negative control, Standard, MEAR and AEAR on Onset and Duration of Sleeping in Thiopental Sodium Induced Sleeping Time Assay.
|
Discussion
GC-MS Analysis of Methanolic and Aqueous Fractions of Annona reticulata Linn. Leaves:
Among the compounds of methanolic extract that were identified by GC-MS analysis, 9-octadecenamide, (z)-, and cholesterol derivative, have antidepressant and anxiolytic properties;21-229-Octadecenamide, (z)- is reported to possess good sedative activity.21Among the identified components of aqueous extract, Thiophene derivative, is reported to possess antidepressant, anxiolytic and sedative activities.23-25In addition, propenamide derivative have anxiolytic activity.26
Assessment of In-vivo Antidepressant Effect
Methanolic and aqueous leaf extracts demonstrated strong antidepressant activities in both assays. GC-MS analysis revealed that test fractions contained compounds with antidepressant properties. It was discovered that the methanol extract included 9-octadecenamide, (z)-, and cholesterol derivative, both of which have antidepressant properties;21-22whereas aqueous extract contained thiophene derivative, that reportedly has antidepressant properties.23Some researches have suggested that 9-octadecenamide, sometimes referred to as oleamide, has antidepressant properties. This is probably because it interacts with the endocannabinoid system and other biochemical pathways. The complex network of receptors and neurotransmitters known as the endocannabinoid system, which is associated with mood regulation and other physiological functions, is known to be impacted by oleamide. It has a lower affinity than certain other cannabinoids for binding to the cannabinoid receptor CB1. Neurotransmitter release and signalling may be influenced by this interaction, which may have mood-modulating consequences. According to some research, oleamide may have an impact on the amounts or activity of monoamine neurotransmitters, such as dopamine and serotonin, which are important for mood regulation and are the focus of many antidepressants.21Through a variety of pathways, such as lipid raft dynamics, neurotrophic factor signalling, and neurotransmitter system modification, cholesterol derivatives can affect antidepressant action. In particular, it has been demonstrated that some cholesterol derivatives affect the GABAergic, serotonin, and dopamine systems, which may be a factor in the antidepressant efficacies. Furthermore, cholesterol derivatives may have an impact on mood modulation through their involvement in brain-derived neurotrophic factor (BDNF) signalling, which is essential for neuronal survival and function.22Incorporating thiophene derivatives into drug molecules can improve brain penetration, boost interactions with antidepressant targets (SERT, NET, DAT, MAO, glutamate receptors), and occasionally lead to the development of new mechanisms.23Researchers assessed antidepressant property of Annona vepretorum, Annona coriacea, and a few other Annonaceae plants.33-34This suggests strongly that the plant may have antidepressant property. Therefore, for the first time, the antidepressant potentials of Annona reticulata Linn’s leaf extracts were evaluated via this investigation. The obtained antidepressant activities of leaf extracts from Annona reticulata Linn. were similar to the activities observed from other Annonaceae plants.
Evaluation of In-vivo Anxiolytic Activity
Methanolic and aqueous leaf extracts demonstrated good anxiolytic properties in both experiments. The GC-MS analytical reports revealed that, the methanol extract included 9-octadecenamide, (z)-, and cholesterol derivative, which have anxiolytic properties;21-22aqueous extract included thiophene derivative that have the ability to reduce anxiety.24Oleamide, or 9-Octadecenamide, (Z)-, has shown anxiolytic efficacy, most likely as a result of its interaction with the GABAergic system and possibly additional neurotransmitter pathways. It has been demonstrated to increase GABA-mediated chloride currents in particular, which may have an impact on lowering anxiety via modifying neural inhibition. The GABAergic system, one of the brain’s main inhibitory systems, is known to interact with oleamide. GABA is a neurotransmitter that lowers neural excitability. Oleamide can help people feel more relaxed by raising GABAergic tone. Although the GABAergic system is the main target, interactions with other neurotransmitters including serotonin and dopamine may also play a role in oleamide’s calming effects.21,27Because they affect neurotransmitters and receptor activation in the brain, cholesterol derivatives can have anxiolytic (anxiety-reducing) effects. Certain derivatives of cholesterol have the ability to alter the activity of receptors that are involved in mood and anxiety regulation, such as GABA and serotonin receptors. Furthermore, cholesterol is essential for preserving nerve cell shape and membrane fluidity, both of which can affect neurotransmission and general brain function.28 The effects of GABA can be amplified by some propanamide derivatives that bind to the GABA-A receptor. As a result, they can promote relaxation and lessen anxiety by increasing the flow of chloride ions into neurons.26 Thiophene derivatives interact with particular neurotransmitter systems in the brain, mostly by modifying the activation of GABA receptors, but they may also affect serotonergic and dopaminergic systems, which might result in anxiolytic (anti-anxiety) effects.29Annona vepretorum, Annona coriacea, and a few other Annonaceaeplants were evaluated by researchers for their anxiolytic potential.33-34This strongly implies that the herb might possess anxiolytic property. Therefore, anxiolytic properties of Annona reticulata Linn. leaf extracts were evaluated for the first time in this study. Our study outcomes regarding Annona reticulataLinn.’s anxiolytic properties were comparable with the outcomes obtained from other Annonaceaeplants.
Assessment of In-vivo Sedative Potential
Methanol and aqueous leaf extracts from Annona reticulata Linn. demonstrated mild sedative activities in both assays. 9-Octadecenamide (z)- was detected in methanol extract, and thiophene derivative was found in aqueous extract according to the GC-MS analytical reports. These substances are reported to possess good sedative properties.21,25So, these compounds can be responsible for the light tranquilizing action of both extracts. The endogenous fatty acid amide 9-octadecenamide, sometimes referred to as oleamide, has been demonstrated to have sedative properties. In animals that are sleep deprived, it builds up in the cerebrospinal fluid and functions as an endogenous sleep-inducing agent.21By interfering with the brain’s neurotransmitter systems, including the GABA and adenosine systems, which control relaxation and sleep, thiophene derivatives can have sedative effects. The sedative effects of certain thiophene-based compounds may also be attributed to their potential to function as antagonists of particular enzymes or receptors.30 Some antidepressants, known as sedative antidepressants, have sedative properties and may be useful in treating insomnia, especially when depression is also present. Tricyclic antidepressants (TCAs) such as amitriptyline, doxepin, and trimipramine are among them, along with other antidepressants like mirtazapine and trazodone. The sedative effects of mirtazapine are particularly well-known.31 Many of these drugs work by inhibiting histamine receptors, especially H1 receptors, which are implicated in the regulation of sleep. Some also have the ability to contribute to sedation via acting on serotonin receptors, including 5-HT2 receptors.32 These drugs further establish the fact that, antidepressant components, molecules and extracts can also possess sedative potential. Diniz et al. evaluated the sedative potential of Annona vepretorum in mice model and found that the plant’s essential oil possessed notable sedative activity.33 A few scientists have already assessed the sedative potentials of Annona reticulata Linn. bark extracts.35 But this is the first study to assess the plant’s methanolic and aqueous leaf fractions and it was found that the bark extracts possessed greater sedative potentials than the leaf extracts (by comparing our study’s outcomes with the results obtained from bark extracts).
Conclusion
Good antidepressant and anxiolytic properties were demonstrated by methanolic and aqueous leaf extracts of Annona reticulata Linn. in in-vivo studies in mice; though both extracts exhibited mild sedative potentials. Comprehensive molecular study is necessary to further understand the mechanism of actions and specific molecules involved, even if animal models appeared to show potential outcomes for the leaf extracts. By isolating specific molecules that were identified to play crucial role in achieving these bioactivities, and then testing them via in-vitro and in-vivo methods can lead to firm conclusions regarding the exact mechanisms by which these activities were obtained.
Acknowledgment
The authors express gratitude to the Department of Pharmacy, Jahangirnagar University for laboratory and logistical support.
Funding Source
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest-
The author(s) do not have any conflict of interest
Data Availability Statement
The data that support the findings of this study are available on request from the corresponding author. All data and materials are contained and described within the manuscript. The plant’s materials for the study were identified and voucher specimens are deposited at Bangladesh National Herbarium DACB No. 114865
Ethics Statement
Every action that involved working with animals was done in compliance with rules set forth by the university’s animal ethics committee [Ref No: BBEC, JU/M2024/11 (137)].
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable.
Author Contributions
- Tasnia Binte Bari Kabbo: data curation (lead), formal analysis (lead), funding acquisition (equal), investigation (lead), methodology (lead), project administration (equal), resources (lead), software (lead), validation (equal), visualization (lead), writing – original draft (lead), writing – review and editing (equal).
- Sohel Rana: supervision (lead), investigation (lead), conceptualization (lead), formal analysis (supporting), funding acquisition (equal), writing – review and editing (equal).
- Sukanta Deb: data curation (equal),formal analysis (equal),
- Farjana Afrin: data curation (equal),formal analysis (equal),
- Shajalal Reza: data curation (equal),formal analysis (equal),
- Shahriar Mohammad Shohan: data curation (equal), formal analysis (equal).
- Pritesh Ranjan Dash: supervision (equal), formal analysis (supporting), investigation (equal), data curation (equal), writing – review and editing (equal).
References
- Rockwood L, Kushnir M, Clarke J. Principles and Applications of Clinical Mass Spectrometry: Small Molecules, Peptides, and Pathogens. Mass Spectrometry. 2018:33–65.
CrossRef - Uttu J, Waziri M, Dauda A, Bida M. Extract Accompanied by Potential Antidepressant Activity. Journal of Chemical Reviews. 2021;4(3):307-319.
- Fedorova S, Kulpin V, Kotova V, Denisova V, Beregovykh V. Study of the antidepressant properties of some plants. AIP Conference Proceedings. 2021;2419(1):050001.
CrossRef - Khan A, Akram M, Thiruvengadam M, et al. Anti-anxiety properties of selected medicinal plants. Current pharmaceutical biotechnology. 2022;23(8):1041-1060.
CrossRef - Abubakar R, Haque M. Medicinal plants with reported anxiolytic and sedative activities in Nigeria: A systematic review. İstanbul Journal of Pharmacy. 2019;49(2):92-104.
CrossRef - Zheng Y, Chang X, Huang Y, He D. The application of antidepressant drugs in cancer treatment. Biomedicine & Pharmacotherapy. 2023;157:113985.
CrossRef - Bahmani F, Kazemeini H, Hoseinzadeh-Chahkandak F, Farkhondeh T, Sedaghat M. Sedation with medicinal plants: A review of medicinal plants with sedative properties in Iranian ethnoblotanical documents. Plant Biotechnol Persa. 2019;1(1):13-24.
CrossRef - Salako A, Akindele J, Balogun O, Adeyemi O. Investigation of antidepressant, anxiolytic and sedative activities of the aqueous leaf extract of Musa sapientumLinn.(Banana; Musaceae). Drug research. 2019;69(03):136-143.
CrossRef - Faizi M, Khazaee R, Mojab F, Jahani R. Psychopharmacological assessment of antidepressant-like, anxiolytic, and sedative-hypnotic effects of Tilia platyphyllos Scop. extract using experimental animal models: Antidepressant-like, anxiolytic, and sedative-hypnotic properties of Tilia platyphyllos. Iranian Journal of Pharmaceutical Sciences. 2022;18(2):116-127.
- Jayaprakash A. Phytochemicals, antimicrobial and antioxidant properties of Annona reticulata Linn. JAIR. 2017;6(6):90-95.
- Ngbolua T, Inkoto L, Bongo N, et al. Phytochemistry and bioactivity of Annona reticulata L.(Annonaceae): A Mini-review. South Asian Research Journal of Natural Products. 2018;1(1):28-38.
- Rudraswamy S, Godhi B, Shankar P, Kenganora M, Sumana N. Detailed understanding of different extraction methods for the research on medicinal plants. Indian Journal of Oral Health and Research. 2021;7(1):14-20.
CrossRef - Agarwal A, Prajapati R, Raza K, Thakur K. GC-MS analysis and antibacterial activity of aerial parts of Quisqualis indica plant extracts. Indian Journal of Pharmaceutical Education and Research. 2017;51(2):329-336.
CrossRef - Yousuf S, Haq M, Rasool A, et al. Evaluation of antidepressant activity of methanolic and hydroalcoholic extracts of Acorus calamus L. rhizome through tail suspension test and forced swimming test of mice. Journal of Traditional Chinese Medical Sciences. 2020;7(3):301-307.
CrossRef - Sibgatullah M. Evaluation of Antidepressant Activity of Ethanolic Extract of AlangiumSalviifolium (LF) Wangerin in Swiss Albino Mice. Biomedical and Pharmacology Journal. 2017;10(1):427-433.
CrossRef - Shahed M, Lina M. Evaluation of sedative and anxiolytic activities of methanol extract of leaves of Persicaria hydropiper in mice. Clinical Phytoscience. 2017;3(1):20.
CrossRef - Begum A, Biswas P, Shahed M. Methanol extract of SpathodeacampanulataP.(Beauv.) leaves demonstrate sedative and anxiolytic like actions on swiss albino mice. Clinical Phytoscience. 2020;6(1):41.
CrossRef - Hossain L, Sultana S. In-vivo assessment of neuropharmacological activity of methanol bark extract of Mimosa pudica in mice. Advances in Pharmacology and Pharmacy. 2019;7(2):33-37.
CrossRef - Rahman M, Islam F, Parvez A, et al. Citrus limon L.(lemon) seed extract shows neuro-modulatory activity in an in vivo thiopental-sodium sleep model by reducing the sleep onset and enhancing the sleep duration. Journal of Integrative Neuroscience. 2022;21(1):42.
CrossRef - Akter A, Islam F, Bepary S, et al. CNS depressant activities of Averrhoa carambola leaves extract in thiopental-sodium model of Swiss albino mice: implication for neuro-modulatory properties. Biologia. 2022;77(5):1337-1346.
CrossRef - Li M, Jiang E, Song Y, Quan S, Yu L. Antidepressant and anxiolytic-like behavioral effects of erucamide, a bioactive fatty acid amide, involving the hypothalamus–pituitary–adrenal axis in mice. Neuroscience letters. 2017;640:6-12.
CrossRef - Yin Y, Liu X, Liu J, et al. The effect of beta-sitosterol and its derivatives on depression by the modification of 5-HT, DA and GABA-ergic systems in mice. RSC advances. 2018;8(2):671-680.
CrossRef - Kumar A, Kumar P, Suhasini S, et al. In-silico approach towards thiophene incorporated 1, 3-oxazepine using 5-HTT and 5-HT2AR receptors for antidepressant activity. Journal of Applied Pharmaceutical Science. 2023;13(12):190-200.
CrossRef - Shah R, Verma K. Therapeutic importance of synthetic thiophene. Chemistry Central Journal. 2018;12(1):137.
CrossRef - Chawla S, Sharma S, Kashid S, Verma K, Sapra A. Therapeutic potential of thiophene compounds: a mini-review. Mini reviews in medicinal chemistry. 2023;23(15):1514-1534.
CrossRef - Pastore V, Wasowski C, Martin P, Enrique A, Higgs J, Bruno-Blanch LE, Milesi V, Marder M. N-propyl-2, 2-diphenyl-2-hydroxyacetamide, a novel α-hydroxyamide with anticonvulsant, anxiolytic and antidepressant-like effects that inhibits voltage-gated sodium channels. European journal of pharmacology. 2018;819:270-280.
CrossRef - Hossen A, Reza S, Amin B, et al. Bioactive metabolites of Blumealacera attenuate anxiety and depression in rodents and computer‐aided model. Food science & nutrition. 2021;9(7):3836-3851.
CrossRef - Cheon Y. Impaired cholesterol metabolism, neurons, and neuropsychiatric disorders. Experimental neurobiology. 2023;32(2):57.
CrossRef - Sonal N, Suraj M, Vaishnavi S, Jnardan K, Mahesh B, Mahesh K. Synthesis of thiophene and Their Pharmacological Activity. International Journal of Pharmaceutical Research and Applications. 2025;10(2):871-875.
CrossRef - Alsayari A, Bin A, Asiri I, Venkatesan K, Mabkhot N. Synthesis and Biological Screening of Thiophene Derivatives. Polycyclic Aromatic Compounds. 2022;42(9):6720-6731.
CrossRef - Riemann D, Espie A, Altena E, et al. The European Insomnia Guideline: An update on the diagnosis and treatment of insomnia 2023. Journal of sleep research. 2023;32(6):e14035.
- Vargas I, Perlis L. Insomnia and depression: clinical associations and possible mechanistic links. Current opinion in psychology. 2020;34:95-99.
CrossRef - Diniz C, Júnior G, Medeiros A, et al. Anticonvulsant, sedative, anxiolytic and antidepressant activities of the essential oil of Annona vepretorum in mice: Involvement of GABAergic and serotonergic systems. Biomedicine & Pharmacotherapy. 2019;111:1074–1087.
CrossRef - Monteiro B, Rodrigues C, Nascimento E, et al. Anxiolytic and antidepressant-like effects of Annona coriacea (Mart.) and caffeic acid in mice. Food and Chemical Toxicology. 2020;136:111049.
CrossRef - Bhalke D, Chavan J. Analgesic and CNS depressant activities of extracts of Annona reticulata Linn. bark. Phytopharmacology. 2011;1(5):160-165.







