Manuscript accepted on :02-02-2026
Published online on: 27-02-2026
Plagiarism Check: Yes
Reviewed by: Dr. Suaad Al-Majidi
Second Review by: Dr. Nagaraj Perumal
Final Approval by: Dr. Anton R Keslav
Khalid Mohammad. Abu Khadra1* , Mohamed Izzat. Bataineh1
, Mohamed Izzat. Bataineh1 , Rania Khalid. Abu-Khadra2
, Rania Khalid. Abu-Khadra2 and Jumana Saleh3
and Jumana Saleh3
1Department of Biological Sciences, Yarmouk University, Irbid, Jordan.
2Faculty of Medicine, Yarmouk University, Irbid, Jordan.
3Biochemistry Department, Sultan Qaboos University, Muscat, Oman
Corresponding Author E-mail:khalidmohd99@yahoo.com.
DOI : https://dx.doi.org/10.13005/bpj/3364
Abstract
The relationship between obesity, diabetes, and serum oxidative susceptibility remains incompletely understood. This study examined serum oxidizability across metabolically distinct groups to assess how obesity and diabetes interact to influence systemic oxidative potential. Copper-induced serum oxidation kinetics were analysed in four groups: non-diabetic non-obese (NN), non-diabetic obese (NOB), diabetic non-obese (DN), and diabetic obese (DOB). Oxidation was monitored at 245 nm, and total serum oxidizability was quantified as the area under the curve (AUC). Group differences were assessed by one-way ANOVA with post hoc analysis, supported by non-parametric testing where applicable. Correlations and predictors of oxidizability were determined by stepwise multiple regression analysis. Oxidation kinetics demonstrated a stepwise increase in oxidizability across NN → NOB → DN → DOB, indicating additive effects of obesity and diabetes. AUC_total values were significantly higher in NOB, DN, and DOB compared with NN (p < 0.001 for all). The difference between NOB and DOB was significant (p = 0.03), suggesting that diabetes superimposed on obesity further amplifies serum oxidative susceptibility. The largest oxidative gap was observed between NN and DOB (+78.6 AU). Correlation analyses showed that triglycerides (r = 0.27, p < 0.001) and fasting glucose (r = 0.28, p < 0.001) were positively associated with oxidizability, whereas HDL correlated inversely (r = –0.15, p = 0.002). Serum oxidizability rises progressively with metabolic deterioration, reflecting both independent and compounded effects of obesity and diabetes. These findings provide a basis for future longitudinal studies to explore oxidative susceptibility as an early marker and potential therapeutic target in cardiometabolic risk.
Keywords
AUC kinetics; Copper-induced oxidation; Obesity, Oxidative stress, Serum oxidizability, Type 2 diabetes
Download this article as:| Copy the following to cite this article: Khadra K. M. A, Bataineh M. I, Khadra R. K. A, Saleh J. Differential Oxidative Susceptibility Between Metabolically Healthy and Unhealthy Obesity. Biomed Pharmacol J 2026;19(1). |
| Copy the following to cite this URL: Khadra K. M. A, Bataineh M. I, Khadra R. K. A, Saleh J. Differential Oxidative Susceptibility Between Metabolically Healthy and Unhealthy Obesity. Biomed Pharmacol J 2026;19(1). Available from: https://bit.ly/3N6E0Fe |
Introduction
Overproduction and accumulation of reactive oxygen species (ROS) and other free radical species launch a dubious physiological riot called oxidative stress.1,2 This brings about tissue damage and creates pathological chaos.3,4 Oxidative stress is a state where ROS mediates various signalling pathways that underlie tissue damage and inflammation, leading to irreversible tissue damage in the vital organs and sometimes hormone-producing glands.5,6 The pathogenesis of chronic diseases was investigated thoroughly from an oxidative stress point of view.8 Therefore, pathophysiological conditions occur as a result of oxidative stress, and the buildup of ROS is linked to the pathogenesis of many chronic diseases.7,8 This is supported by high levels of oxidized biological molecules and the lipid peroxidation-related markers buildup in the damaged tissues of affected organs.6,8
Oxidative stress adversely affects insulin production and is linked to diabetic hyperglycaemia by interaction with insulin homeostasis.9,10 It serves as a unifying mechanism for Type 2 diabetes mellitus (T2DM) risk factors, underscoring its central role in T2DM.10 Increased oxidative stress has been observed in the plasma of T2DM patients and correlates directly with FBS and HbA1c levels, while inversely relating to insulin levels.11,12 Several case-control studies disclose the association between T2DM and oxidative stress.13,14 Research efforts rationalised many risk factors involved in the development of T2DM, including oxidative stress.15,16 However, a second opinion considers oxidative stress to be an aftermath of hyperglycaemia.15,16 Consequently, examining the biomarkers and the enzymes of oxidative stress in T2DM blood and comparing that to the obese group will clarify oxidative stress as an etiological factor associated with T2DM.1,6 Recent data suggest that the oxidizability of a biological sample indicates its susceptibility to oxidation, and assessing oxidizability is crucial for evaluating the kinetics of oxidative processes in LDL, blood serum, and other body fluids.17,18 This approach can be used to monitor oxidative stress across various diseases.19,20 Therefore, studying serum oxidisability and evaluating the serum antioxidant capacity for patients suffering from diabetes and obesity may elucidate how an increase in oxidative stress impacts insulin-producing cells and the pancreatic β-cells-related proteins.21,22
Our study was designed to analyse and assess oxidative stress biomarkers among T2DM and obese nondiabetic groups, namely serum oxidizability and serum antioxidant capacity in comparison with that of healthy individuals. The serum antioxidant capacity of T2DM patients and obese individuals was evaluated through the copper–induced 50–fold assessment method, which measures the increase in serum oxidizability and serum antioxidant capacity.
Materials and Methods
Ethical consideration
Approval for the research from the corresponding department of the Ministry of Health, Amman, Jordan, and the Institutional Review Board Council at Yarmouk University (Irbid, Jordan) IRB committee (YUIRB DSR 2025/177) were obtained. We informed all patients who participated in the research about the goals and importance of this study, and the information they provided will be used for research purposes, with access provided to the researchers alone. We assured them of their rights to privacy, safety and confidentiality.
Subjects
We designed a cross-sectional study by recruiting 401 adults (183 males and 218 females), with 198 T2DM patients (100 males and 98 females) and a group of 203 non-diabetic healthy individuals, from April to June 2018, aged between 35 – 53 years. We randomly selected the T2DM patients from a specialised diabetic care clinic, and the control group from volunteers of the Jordanian community. The control group was further separated into obese and healthy non-obese subgroups based on calculated body mass index (BMI) as follows: 16.5 or less to 25 as non-obese, 25 to 30 as obese. Written informed consent was obtained from every participant. Any pateints who is a smoker, pregnant, taking antioxidant medication or had exposed to X-rays in the past three months was excluded from the study.
Biochemical testing
All were fasted for 12 hours before blood collection. We collected 10 ml of peripheral blood from them, obtaining serum for oxidizability testing and biochemical analysis, which included fasting plasma glucose (FBS), cholesterol, HDL cholesterol, LDL cholesterol, and Triglycerides were measured by conventional standard enzymatic assays. HbA1c was determined by enzymatic immunoassay method.
Serum Oxidation measurement
Preparation of Phosphate-buffered saline (PBS) and Copper chloride CuCL2:
We prepared Phosphate buffered saline (PBS) by dissolving PBS tablets purchased from Gibco Company, REF 18912-014) in 500ml of double distilled water. 0.035 g of copper chloride (CuCL2 Sigma, MW:170.98) was dissolved in 50 ml PBS, and the pH of this solution was adjusted to 7.4. While we stirred the copper chloride solution with a magnetic stirrer, we took 0.5 ml and added it to 9.5 ml of PBS. Reaction solution (10ml) was mixed fresh for each run at room temperature for the copper-induced oxidation experiment.
Cu+2-Induced Oxidation of Serum
This experiment attempts to assess the susceptibility of serum to oxidation. Serum oxidizability was used to evaluate the susceptibility of lipids to oxidation in vitro using copper chloride to induce non fractionated lipids via nonenzymatic pathway to be oxidized. The method used is based on Schnitzer et al.23 The onset of serum oxidation induced by copper is monitored by the spectroscopic technique of oxidation products (23,24). Measurements were carried out in quartz UV plates (96 wells, No Lid purchased from CORNING). First, serum samples were thawed and immediately put in ice before they reached room temperature. In each well, 10µl of PBS was added, then 2µl of serum sample, followed by the addition of 100µl of copper chloride CuCL2. All determination was carried out in duplicate. Also, there is a negative control in each plate that contains PBS buffer and CuCL2 only. Right away, the plate was placed in the SYNERGY microplate reader. The oxidation reaction was measured at 25°C, continuously obtaining the absorbance over three hours every minute at a wavelength of 245 nm. 7-ketocholesterol and conjugated dienic hydroperoxide were produced, leading to the accumulation of dienes and ketodienes. We plotted the absorbance against time, obtaining a curve consisting of three phases: lag, propagation, and decomposition. The serum oxidizability parameters were calculated, including the BOS (Basal oxidation status), and the lag time of the start of oxidation rate.
Statistical analysis
All statistical analyses were performed using IBM SPSS Statistics (Version 28.0, IBM Corp., Armonk, NY, USA) [26]. Data were tested for normality using the Shapiro–Wilk test. Group differences in metabolic biochemical measures and serum oxidizability (AUC) were evaluated using one-way analysis of variance (ANOVA) followed by post hoc tests for pairwise comparisons. Where appropriate, non-parametric comparisons were conducted to confirm marginal differences. For kinetic oxidation curves, group means ± standard error of the mean (SEM) were plotted as time-dependent absorbance at 245 nm. The area under the curve (AUC) was calculated using the trapezoidal integration method applied to sequential optical density values (OD₁ to OD₃₀) to quantify total oxidizability for each sample. Statistical significance was set at p < 0.05 (two-tailed). Bivariate correlation (Pearson and Spearman) analysis was performed to examine the association between the lipid profile parameters in each group with oxidative measures in this study.
Results
Table 1: Descriptive anthropometric and metabolic measures among different groups
|
Variable |
NN (n=115) |
NOB (n=88) |
DN (n=90) |
DOB (n=108) |
|
Age (years) |
41.7 ± 6.0 |
45.9 ± 5.4 |
48.4 ± 5.1 |
49.5 ± 3.8 |
|
BMI (kg/m²) |
25.3 ± 2.7 |
34.3 ± 3.4§§ |
27.1 ± 2.6§§ |
34.8 ± 3.8§§ |
|
FBS (mmol/L) |
5.2 ± 0.5 |
5.6 ± 0.6 |
10.4 ± 4.6** |
9.9 ± 4.5** |
|
HbA1c (%) |
5.3 ± 0.4 |
5.4 ± 0.4 |
7.9 ± 1.8** |
7.8 ± 1.9** |
|
Cholesterol (mmol/L) |
4.6 ± 0.9 |
4.8 ± 0.97 |
4.6 ± 1.1 |
4.7 ± 1.1 |
|
Triglycerides (mmol/L) |
1.4 ± 0.9 |
2.2 ± 1.2** |
2.3 ± 1.6** |
2.3 ± 1.3** |
|
HDL (mmol/L) |
1.1 ± 0.2 |
1.07 ± 0.2 |
1.1 ± 0.3 |
1.1 ± 0.3 |
|
LDL (mmol/L) |
2.9 ± 1.2 |
2.7 ± 0.8 |
2.5 ± 0.96* |
2.6 ± 1.0* |
|
Albumin (g/L) |
41.79 ± 6.04 |
42.26 ± 4.71 |
42.48 ± 6.20 |
40.71 ± 5.40 |
|
Total Protein (g/L) |
71.63 ± 4.49 |
71.83 ± 4.53 |
71.87 ± 5.92 |
71.21 ± 5.01 |
Parameters and biochemical results are presented as mean ± SD.
*p< 0.05 values differ compared to group NN
**p< 0.001 compared to NN and NOB
§§ p < 0.001 compared to group NN
Our study recruited 185 males and 216 females aged between 35-53 years. The non-diabetic group was composed of 115 healthy (nonobese nondiabetic) (NN) volunteers as controls (50 of them males and 65 females). The non-diabetic obese group includes 88 obese (NOB), 35 males and 53 females. The diabetic group consisted of 198 patients, 100 males and 98 females; 90 diabetic nonobese (DN) and 108 diabetic obese (DOB). The anthropometric parameters and biochemical analysis of participants are presented in Table 1. The differences were determined by one-way ANOVA
Baseline Clinical and Biochemical Characteristics
Baseline characteristics of the study participants across the four groups — non-diabetic non-obese (NN), non-diabetic obese (NOB), diabetic non-obese (DN), and diabetic obese (DOB) — are summarized in Table 1. Group comparisons were performed using one-way ANOVA.
As expected, body mass index (BMI) values were significantly higher in the obese groups (NOB and DOB) compared with their non-obese counterparts (NN and DN). Fasting blood glucose (FBS) and glycated hemoglobin (HbA1c) showed the most prominent and statistically significant differences between groups, with both parameters markedly elevated in the diabetic groups relative to the non-diabetic participants. Triglyceride levels were also significantly higher among metabolically affected groups compared with healthy controls, reflecting the dyslipidemic component associated with both obesity and diabetes. Other biochemical parameters, including total cholesterol, LDL-C, HDL-C, albumin, and total protein, showed comparatively weak or no significant group differences. These findings collectively confirm appropriate metabolic grouping and demonstrate that the major discriminating variables across cohorts were glycemic and triglyceride-related measures.
Kinetics results of copper-induced 50–fold ‘serum oxidizability are described by the kinetic curves of the accumulation of dienes and ketodienes. The results are demonstrated as kinetic oxidation curves and calculated area under the curve as shown below:
Serum Oxidation Kinetics and Area Under the Curve (AUC)
Copper-induced oxidation kinetics of serum samples demonstrated different oxidative susceptibility patterns across metabolic groups (Figure 1A). The oxidation curves for the non-diabetic non-obese (NN), non-diabetic obese (NOB), diabetic non-obese (DN), and diabetic obese (DOB) groups exhibited a stepwise increase in oxidation absorbance at 245 nm over time. While NN sera showed a gradual and delayed rise in absorbance, indicative of lower oxidizability, both NOB and DN groups displayed faster oxidation kinetics, suggesting enhanced oxidative potential. The DOB group demonstrated the highest oxidation rate and final absorbance plateau, reflecting markedly greater oxidative susceptibility compared with all other groups.
 |
Figure 1A: Serum oxidizability kinetics in different metabolic groups.
|
Copper-induced serum oxidation curves (mean ± SEM, n = 401) showing time-dependent changes in absorbance at 245 nm across four metabolic groups: non-diabetic non-obese (NN), non-diabetic obese (NOB), diabetic non-obese (DN), and diabetic obese (DOB). The oxidation rate and final plateau increase progressively from NN → NOB → DN → DOB, indicating rising oxidative susceptibility.
The calculated area under the curve (AUC) values revealed significant group differences (Figure 1B). One-way ANOVA showed a robust overall effect of metabolic status on total oxidizability (F(3, 397) = 14.27, p < 0.001; η² = 0.097). Post hoc comparisons indicated that AUC values were significantly higher in NOB (347.7 ± 97.2 AU), DN (362.8 ± 96.3 AU), and DOB (374.7 ± 109.4 AU) groups compared to NN (296.1 ± 81.9 AU; p < 0.001 for all). The difference between NOB and DOB reached significance (p = 0.03), suggesting that diabetes superimposed on obesity further enhances serum oxidative susceptibility.
The mean AUC difference between NN and DOB was +78.6 AU, representing the largest oxidative gap among groups, followed by +67 AU (NN vs DN) and +52 AU (NN vs NOB). These findings indicate a progressive rise in serum oxidizability with increasing metabolic impairment, wherein both obesity and diabetes exert additive effects on oxidative potential. Notably, although obesity alone was associated with elevated oxidizability compared with the non-obese state, the presence of diabetes produced a clear increase in oxidation rate and magnitude, reflecting an additive metabolic burden.
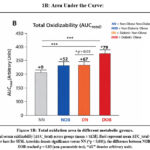 |
Figure 1B: Total oxidation area in different metabolic groups.
|
Total serum oxidizability (AUC_total) across groups (mean ± SEM). Bars represent mean AUC_total with error bars for SEM. Asterisks denote significance versus NN (*p < 0.001); the difference between NOB and DOB reached p = 0.03 (non-parametric test). “AU” denotes arbitrary units.
Correlation and Regression Analyses
To explore the determinants of serum oxidizability, a pooled analysis including all study participants (N = 401) was conducted. Spearman’s correlation analysis revealed that the total oxidizability (AUC) was positively correlated with (FBS; r = 0.281, p < 0.001), HbA1c; r = 0.216, p < 0.001), and (TG; r = 0.269, p < 0.001). Conversely, AUC exhibited a weak but significant negative correlation with HDL cholesterol (r = –0.151, p = 0.002), whereas total cholesterol and LDL cholesterol showed no significant associations.
To further identify independent predictors of oxidizability, a stepwise multiple regression analysis was performed with AUC as the dependent variable and biochemical and anthropometric parameters as predictors. The model identified TG as the strongest independent predictor of AUC (β = 0.245, p < 0.001), followed by (BMI; β = 0.135, p = 0.007) and (FBS; β = 0.105, p = 0.044). Together, these variables accounted for approximately 8.7% of the total variance in oxidizability (adjusted R² = 0.08, p < 0.001).
Discussion
The kinetics studies of serum oxidation by copper-induced oxidation are an accepted, validated, and reproducible method used to evaluate serum oxidizability and antioxidant capacity.5,23 This assay is based on copper-induced oxidation leading to dienes and ketodienes formation.24 Different methods and approaches were used to appraise and assess serum oxidizability and antioxidant capacity.25,26 Oxidative stress is associated with increased free radical generation and ROS formation in the blood, leading to oxidative modification of cellular protein, lipid, and DNA.27,28 Increased ROS production appears to impair endothelium-dependent vasorelaxation, leading to ischemia, provoking cell apoptosis and inflammation, leading to cellular dysfunction and deterioration.29 Only a few studies have addressed the issue of ROS accumulation and oxidative stress in diabetes from this point of view.30,31 Moreover, obesity was found to be involved in the development of many obesity-related diseases due to oxidative stress.13,31 Research studies disclose obesity to T2DM and atherosclerosis through increased free radical generation and ROS accumulation.18,32 Indirectly, obesity could trigger the release of inflammatory cytokines, which enhance free radical and ROS generation, thus endowing a vicious circle of oxidative stress.33,34 Our study investigates oxidative stress in metabolic disease by studying serum oxidizability and comparing the time course of copper-induced oxidation in the serum of obese and T2DM patients. The hypothesis linking oxidative stress and T2DM was based on the increase in ROS production in T2DM patients compared to non-diabetic controls. This may also establish groundwork for the significance of oxidative stress in exacerbating the progression of metabolic disturbance in T2DM, and may not only be the result of it.
This study demonstrates that serum oxidative susceptibility differs significantly between metabolically healthy and unhealthy obesity, revealing a graded increase in oxidative potential with worsening metabolic status. Obesity alone increased serum oxidizability compared with the non-obese state, reflecting greater lipid substrate availability and a more pro-oxidant milieu. However, the superimposition of diabetes on obesity produced an even higher oxidation rate and total oxidative burden, underscoring the synergistic effect of hyperglycemia and adiposity in promoting redox imbalance.
The observed hierarchy, NN < NOB < DN < DOB, suggests that oxidative susceptibility reflects both adiposity and metabolic dysregulation, rather than obesity alone. These findings support the concept that metabolically healthy obesity represents a transitional phenotype, which may progress toward the diabetic obese state as oxidative stress intensifies. This differential serum oxidation response provides mechanistic insight into how combined metabolic insults reshape the redox landscape, contributing to atherogenic and inflammatory risk profiles characteristic of diabetic obesity.
Our study shows a significant difference between the obese and T2DM groups compared to the control group, analysing the serum oxidizability parameters, the antioxidant enzyme activity and antioxidant capacity. The substantial increase in serum oxidizability among diabetic and obese groups was consistent with the high oxidative stress found in diabetes and obese groups.22,35 These results are consistent with the last decade’s extensive research regarding the association between oxidative stress and T2DM development.11,14 Our findings are consistent with research reporting that increased serum oxidizability and recurrent oxidative stress in T2DM reduce and deplete oxidative capacity and antioxidant enzymes.9,12
Higher MDA levels in diabetic and obese individuals compared to non-diabetic, non-obese groups were reported in our previously published data and are consistent with findings from similar studies examining the association between obesity and oxidative stress.16,31 Furthermore, the absence of a significant difference between the T2DM and obese groups in lipid peroxidation and antioxidant capacity probably explains the oxidative stress linked to the development of T2DM.12,16 The serum oxidisability parameters in our study are related to serum components such as LDL, albumin, uric acid, antioxidant enzymes, and intermediates of lipid peroxidation.16,36 Previous reports indicating that ultrafiltrate serum lacks large molecular weight molecules interrupted the ability of the ultrafiltrate to protect against oxidation by copper and promote the oxidation of LDL.35,37 The kinetics of LDL oxidation are subjected to different variables, including copper concentration.38,39 Copper chelation by serum antioxidant proteins would beneficially reduce the availability of copper to oxidize LDL.38 There is no significant difference in the levels of cholesterol, LDL and HDL, making it compatible with those of Bansal et al.41 and Kumawat et al40, but not compatible with the data of Steinberg and Lewis regarding LDL.42 However, a significant association between lipid peroxidation and serum oxidizability was reported.41,42
The correlation and regression analyses collectively demonstrate that both lipid and glycemic disturbances are key determinants of serum oxidative susceptibility. Triglycerides and glycemic indices (FBS, HbA1c) showed strong positive correlations with oxidizability, while HDL exhibited an inverse association, consistent with its well-established antioxidant and anti-atherogenic roles. These findings suggest that hypertriglyceridemia and hyperglycemia promote oxidative stress, likely via enhanced lipid peroxidation and glycoxidative mechanisms, whereas HDL may exert a counter-regulatory protective effect.
Regression modelling further underscored triglycerides as the principal determinant of serum oxidizability, with BMI entering the model before FBS, suggesting that adiposity potentiates oxidative vulnerability, especially in individuals with concomitant metabolic dysregulation. This pattern aligns with group-level observations showing that the diabetic obese (DOB) group had the highest oxidizability, emphasising the synergistic contribution of lipid load, adiposity, and glycemic stress. Overall, these results indicate that oxidative burden in obesity and diabetes reflects the degree of metabolic dysregulation rather than adiposity per se, reinforcing the central role of lipid and glucose metabolism in driving systemic redox imbalance.
Conclusion
These findings collectively suggest that both obesity and diabetes independently contribute to increased serum oxidizability; however, the compounded effect observed in the diabetic–obese (DOB) group indicates a synergistic interaction that markedly amplifies oxidative susceptibility. Elevated triglycerides, hyperglycemia, and adiposity appear to act as upstream metabolic triggers of oxidative stress–mediated pathways that promote vascular injury, inflammation, and metabolic disease progression. By identifying these metabolic drivers of oxidizability, the present study highlights potential mechanistic links between dysmetabolism and downstream complications associated with obesity and diabetes.
Nevertheless, it is important to acknowledge that, although the observed associations are statistically significant and biologically credible, the cross-sectional design limits causal interpretation. However, these findings provide a strong basis for hypothesis generation and open new avenues for longitudinal and mechanistic investigations to elucidate the causal pathways through which oxidative susceptibility contributes to metabolic and vascular disease progression.
Acknowledgement
The authors thank the Deanship of Scientific Research and Graduate Studies at Yarmouk University.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
Informed consent was confirmed by the IRB before being signed by the participants who agreed to participate. The authors declare that the procedures followed the regulations established by the Helsinki Declaration of the World Medical Association.
Clinical Trial Registration
This trial is registered at the Institutional Review Board Council of Yarmouk University (Irbid, Jordan) IRB committee (YUIRB DSR 2025/177). Informed consent was confirmed by the IRB before being signed by the participants who agreed to participate. The authors declare that the procedures followed the regulations established by the Helsinki Declaration of the World Medical Association.
Permission to reproduce material from other sources
Not Applicable
Author contributions
- Khalid Abu khadra: Conceptualization, Visualization, Supervision, Funding Acquisition.
- Rania Mohamed Bataineh: Data Collection, Analysis, Writing – Original Draft.
- Rania Khalid Abu khadra: Writing – Review & Editing.
- Jumana Saleh: Conceptualization, Methodology, Resources
References:
- Black HS. A Synopsis of the Associations of Oxidative Stress, ROS, and Antioxidants with Diabetes Mellitus. Antioxidants 2022; 11, 2003. https://doi.org/10.3390/antiox11102003
CrossRef - Andrés C, Pérez de la Lastra J, Juan C, Plou F, Pérez-Lebeña E, Chemistry of hydrogen peroxide formation and elimination in mammalian cells, and its role in various pathologies. Stresses 2022; 2, 256. https://doi.org/10.3390/stresses2030019.
CrossRef - Perry JJ, Shin DS, Getzoff ED, Tainer JA. The structural biochemistry of the superoxide dismutases. Biochim Biophys Acta.2010;1804, 245. doi: 10.1016/j.bbapap.2009.11.004.
CrossRef - Kıran T, Otlu O, Karabulut A, Oxidative stress and antioxidants in health and Disease. Journal of Laboratory Medicine 2023; 47 (1):1-11. https://doi.org/10.1515/labmed-2022-0108.
CrossRef - Savchenkova AP, Dudnik LB, Solov’eva NP, Azizova OA. Kinetic characteristics of Cu2+-induced lipid peroxidation in blood plasma and serum. Biomed Khim. 2003 Nov-Dec;49(6):576-87. Russian. PMID: 16119086.
- Evans J, Goldfine I, Maddux B, Grodsky G, Oxidative stress and stress-activated signaling pathways: A unifying hypothesis of type 2 diabetes. Endocrine Rev. . 2002; 23, 599. https://doi.org/10.1210/er.2001-0039
CrossRef - Madamanchi N, Vendrov A, Runge M, Oxidative stress and vascular disease. Arterioscler Thromb Vasc Biol 2005; 25,29. https://doi.org/10.1161/01.ATV.0000150649.39934.13
CrossRef - Vassalle C, Petrozzi L, Botto N, Andreassi MG, Zucchelli C, Oxidative stress and its association with coronary artery disease and different atherogenic risk factors. J Intern Med 2004; 256, 308. https://doi.org/10.1111/j.1365-2796.2004.01373.x
CrossRef - Pieme C, Tatangmo J, Simo G, Nya P, Moor V, Moukette B, Nzufo F, Nono B, Sobngwi E, Relationship between hyperglycemia, antioxidant capacity and some enzymatic and non-enzymatic antioxidants in African patients with type 2 diabetes. BMC Res Notes, 2017; 10, 141 DOI 10.1186/s13104-017-2463-6.
CrossRef - Bhatti J, Sehrawat A, Mishra J, Sidhu S, Navik U, Khullar N, Kumar S, Bhatti G, Reddy H, Oxidative stress in the pathophysiology of type 2 diabetes and related complications: Current therapeutics strategies and future perspectives. Free Radical Biology and Medicine, 2022; 184, 114. doi.org/10.1016/j.freeradbiomed.2022.03.019.
CrossRef - Tiwari K, Pandey B, Abidi B, Rizvi I, Markers of Oxidative Stress during Diabetes Mellitus. Journal of Biomarkers. 2013; 2013:378790. doi.org/10.1155/2013/378790.
CrossRef - Pandey K B, Mishra N, Rizvi SI. Protein oxidation biomarkers in plasma of type 2 diabetic patients. Clinical Biochemistry. 2010;43(4-5):508–511. doi: 10.1016/j.clinbiochem.2009.11.011.
CrossRef - Li H, Ren J, Li Y, Wu Q, and Wei J, Oxidative stress: The nexus of obesity and cognitive dysfunction in diabetes. Front. Endocrinol. 2023; 14:1134025. https://doi.org/10.3389/fendo.2023.1134025.
CrossRef - Matsuda M., and Shimomura I. Increased oxidative stress in obesity: implications for metabolic syndrome, diabetes, hypertension, dyslipidemia, atherosclerosis, and cancer. Obes Res Clin Pract. 2013; 7(5): e330-41. DOI: 10.1016/j.orcp.2013.05.004..
CrossRef - Tirosh A, Potashnik R, Bashan N, Rudich A, Oxidative stress disrupts insulin-induced cellular redistribution of insulin receptor substrate-1 and phosphatidylinositol 3-kinase in 3T3-L1 adipocytes. J. Biol. Chem. 1999; 274,10595. https://doi.org/10.1074/jbc.274.15.10595.
CrossRef - Abu Khadra K, Bataineh M, Khalil A, Salah J, Oxidative stress and type 2 diabetes: the development and the pathogenesis, Jordanian cross-sectional study. Eur J Med Res2024; 29, 370. https://doi.org/10.1186/s40001-024-01906-4.
CrossRef - Haffner S, Agil A, Mykkanen L, Stern M, Jialal I, Plasma oxidizability in subjects with normal glucose tolerance, impaired glucose tolerance, and NIDDM. Diabetes Care 1995; 18:646.
CrossRef - Friedman J, Peleg E, Kagan T, Shnizer S, Rosenthal T, Oxidative stress in hypertensive, diabetic, and diabetic hypertensive rats. Am J Hypertens. 2003;16,1049. https://doi.org/10.1016/j.amjhyper.2003.07.013.
CrossRef - Kontush A, Beisiegel U, Measurement of oxidizability of blood plasma. Methods Enzymol 1999;299,35. https://doi.org/10.1016/s0076-6879(99)99007-9.
CrossRef - Kregel K, Zhang H, An integrated view of oxidative stress in aging: basic mechanisms, functional effects, and pathological considerations. Am J Physiol Regul Integr Comp Physiol 2007;292,18. https://doi.org/10.1152/ajpregu.00327.2006.
CrossRef - Jakubiak G, Osadnik K, Lejawa M, Kasperczyk S, Osadnik T, Pawlas N, Oxidative stress in association with metabolic health and obesity in young adults. Oxidative Medicine and Cellular Longevity. 2021;26, 2021. https://doi.org/10.1155/2021/9987352.
CrossRef - Chrysohoou C, Panagiotakos DB, Pitsavos C, Skoumas I, Papademetriou L, Economou M, Stefanadis C. The implication of obesity on total antioxidant capacity in apparently healthy men and women: the ATTICA study. Nutr Metab Cardiovasc Dis. 2007;17,590 doi: 10.1016/j.numecd.2006.05.007.
CrossRef - Schnitzer E, Pinchuk I, Bor A, Fainaru M, Samuni AM, Lichtenberg D. Lipid oxidation in unfractionated serum and plasma. Chem Phys Lipids. 1998;92,151. doi: 10.1016/s0009-3084(98)00021-8. PMID: 9682469.
CrossRef - Pinchuk I, Shoval H, Bor A, Schnitzer E, Dotan Y, Lichtenberg D. Ranking antioxidants based on their effect on human serum lipids peroxidation. Chem Phys Lipids. 2011;164,42. doi: 10.1016/j.chemphyslip.2010.10.002.
CrossRef - Alberti A, Bolognini L, Macciantelli D, Caratelli M, The radical cation of N,N-diethyl-para-phenylendiamine: a possible indicator of oxidative stress in biological samples. Research on Chemical Intermediates. 2000;26,253. doi: 10.1163/156856700X00769.
CrossRef - Imam H, Chowdhury A, Mahbub NU, Hossain A, Karim MF, Uddin MB, Sarker MM. Oxidizability assay of unfractionated plasma of patients’ with different plasma profile: a methodological study. J Diabetes Metab Disord. 2014;1,13. doi: 10.1186/2251-6581-13-54. PMID: 24918093; PMCID: PMC4031925.
CrossRef - Cadet J, Douki T, Ravanat J, Oxidatively generated base damage to cellular DNA. Free Radic Biol Med. 2010;49,9. doi: 10.1016/j.freeradbiomed.2010.03.025.
CrossRef - Berlett B, Stadman E, Protein oxidation in aging, disease and oxidative stress. J Biol Chem. 1997;272,20313. https://doi.org/10.1074/jbc.272.33.20313.
CrossRef - Friedman J, Peleg E, Kagan T, Shnizer S, Rosenthal T, Oxidative stress in hypertensive, diabetic, and diabetic hypertensive rats. Am J Hypertens. 2003;16,1049. https://doi.org/10.1016/j.amjhyper.2003.07.013.
CrossRef - Sayar N, Terzi S, Yilmaz Y, Tangurek B, Bilsel T, Cakmak N, Orhan L, Emre A, Ciloglu F, Peker I, Yesilcimen K, Exercise-induced increase in lipid peroxidation in patients with chronic heart failure: relation to exercise intolerance. Cardiology. 2007;108;307. https://doi.org/10.1159/000099100.
CrossRef - Huang C, McAllister M., Slusher A, Webb H, J. Thomas Mock T, Acevedo E, Obesity-Related Oxidative Stress: the Impact of Physical Activity and Diet Manipulation. Sports Med, 2015;32, doi.org/10.1186/s40798-015-0031-y.
CrossRef - Bryan S, Baregzay B, Spicer D, Singal K., Khaper N, Redox-inflammatory synergy in the metabolic syndrome. Can. J. Physiol. Pharmacol. 2013;91,22. https://doi.org/10.1139/cjpp-2012-0295.
CrossRef - Dandona P, Aljada A, Chaudhuri A, Mohanty P, Garg R, Metabolic syndrome: A comprehensive perspective based on interactions between obesity, diabetes, and inflammation. Circulation. 2005;111,1448. https://doi.org/10.1161/01.cir.0000158483.13093.9d.
CrossRef - Crujeiras A, Díaz-Lagares A., Carreira M, Amil M., Casanueva F, Oxidative stress associated to dysfunctional adipose tissue: A potential link between obesity, type 2 diabetes mellitus and breast cancer. Free Radic. Res. 2013;47,243. https://doi.org/10.3109/10715762.2013.772604.
CrossRef - Smriti K, Pai K, Ravindranath V, Pentapati C, Role of salivary malondialdehyde in assessment of oxidative stress among diabetics. J Oral Biol Craniofac Res. 2016;6,41. doi: 10.1016/j.jobcr.2015.12.004.
CrossRef - Nyyssönen K, Porkkala-Sarataho E, Kaikkonen J, Salonen J, Ascorbate and urate are the strongest determinants of plasma antioxidative capacity and serum lipid resistance to oxidation in Finnish men. Atherosclerosis. 1997;130, 223. doi: 10.1016/s0021-9150(96)06064-9.
CrossRef - Roland A, Patterson R, Leake D, Measurement of copper-binding sites on low density lipoprotein. Arterioscler. Thromb. Vasc. Biol. 2001; 21, 594. doi: 10.1161/01.atv.21.4.594.
CrossRef - Davies K, Sevanian A, Muakkassahkelly S, and Hochstein P, Uric acid iron ion complexes – a new aspect of the antioxidant functions of uric acid. Biochem. J. 1986; 235, 747. doi: 10.1042/bj2350747.
CrossRef - Ziouzenkova O, Sevanian, Abuja A, Ramos P, Esterbauer H, Copper can promote oxidation of LDL by markedly different mechanisms. Free Radic. Biol. Med. 1998;24, 607. doi: 10.1016/s0891-5849(97)00324-9.
CrossRef - Kumawat M, Sharma TK, Singh I, Singh N, Ghalaut VS, Vardey SK, Shankar V. Antioxidant Enzymes and Lipid Peroxidation in Type 2 Diabetes Mellitus Patients with and without Nephropathy N, Am J Med Sci. 2013;5,213.. doi: 10.4103/1947-2714.109193.
CrossRef - Bansal S, Chawla D, Siddarth M, Banerjee BD, Madhu SV, Tripathi AK. A study on serum advanced glycation end products and its association with oxidative stress and paraoxonase activity in type 2 diabetic patients with vascular complications. Clin Biochem. 2013;46,109. doi: 10.1016/j.clinbiochem.2012.10.019.
CrossRef - Steinberg D. Lewis A. Conner Memorial Lecture. Oxidative modification of LDL and atherogenesis. Circulation. 1997;95(4):1062. doi: 10.1161/01.cir.95.4.1062. PMID: 9054771.
CrossRef







