Manuscript accepted on :22-01-2026
Published online on: 26-02-2026
Plagiarism Check: Yes
Reviewed by: Dr. Nasir Abdelrafie
Second Review by: Dr. Jerwin Prabu
Final Approval by: Dr. Prabhishek Singh
Maria Lawrensia Tampubolon1 . Edith Nadya Apriliani2
. Edith Nadya Apriliani2 . Daniel Joko Wahyono2
. Daniel Joko Wahyono2 . and Siti Maemun3,4*
. and Siti Maemun3,4*
1Department of Neurology, Sulianti Saroso Infectious Disease Hospital, Jakarta, Indonesia.
2Faculty of Biology, Jenderal Soedirman University, Purwekerto, Indonesia.
3Clinical Research Unit, Sulianti Saroso Infectious Disease Hospital, Jakarta, Indonesia.
4Indonesian College of Epidemiology, Indonesian Health College, Jakarta, Indonesia.
Corresponding Author E-mail: muntee83@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3355
Abstract
Meningitis is an infectious disease that attacks the central nervous system. Meningitis can be caused by pathogenic microorganisms. including bacteria. viruses. and fungi. The incidence of meningitis in Indonesia is high; thus, cerebrospinal fluid (CSF) analysis is needed to establish the diagnosis and identify the pathogen. This study used a cross-sectional design. The research used secondary data from medical records of patients with meningitis who underwent CSF examination at Sulianti Saroso Infectious Disease Hospital (SSIDH). The sampling method used was purposive sampling. The study population consisted of patients aged 18 years or older who were diagnosed with meningitis and underwent CSF examination. The result of this study shows that of the 17 cases of meningitis at SSIDH during 2019 - 2023. The causing pathogens were identified from 8 cases (47.06%). while the causing pathogens of the other 9 cases (52.94%) could not be identified. The identified pathogens are 4 cases of TB meningitis (23.53%) and 4 cases of cryptococcal meningitis (23.53%). Meanwhile. The cases with unidentified pathogens include 3 cases of suspected TB meningitis (17.65%) and 6 cases of suspected viral meningitis (35.29%). This occurs because of microbiological examinations. such as the GeneXpert or Molecular Rapid Test examination to detect the genetic material of TB bacteria, and the PCR virus panel test to detect the genetic material of the virus. were not performed in some cases.
Keywords
Examination; Liquor Cerebrospinal Fluid; Pathogen; Suspected; Viral Meningitis
Download this article as:| Copy the following to cite this article: Tampubolon M. L, Apriliani E. N, Wahyono D. J, Maemun S. Cerebrospinal Fluid Profile in Patients with Suspected Viral Meningitis at Tertiary Infectious Disease Hospital in Jakarta. Biomed Pharmacol J 2026;19(1). |
| Copy the following to cite this URL: Tampubolon M. L, Apriliani E. N, Wahyono D. J, Maemun S. Cerebrospinal Fluid Profile in Patients with Suspected Viral Meningitis at Tertiary Infectious Disease Hospital in Jakarta. Biomed Pharmacol J 2026;19(1). Available from: https://bit.ly/40uPDc3 |
Introduction
The global impact of viral meningitis (VM) remains poorly defined and remains a significant public health concern, particularly in low- and middle-income countries. VM is considered a serious condition due to its potential for rapid clinical deterioration, long-term complications, and risk of death. Although VM most frequently occurs in infants under one year of age, individuals of all ages may also be affected.1,2 Meningitis is an infectious disease that affects the meninges. Typical symptoms of meningitis include neck stiffness, fever, headache 2,3, fatigue, irritability, reduced appetite, diarrhea, vomiting, anorexia, skin rash, signs of meningeal involvement, and seizures.2
According to WHO data cited in a book published by the Indonesian Ministry of Health (2019), there were 19,135 suspected meningitis cases with 1,398 deaths reported across the meningitis belt up to October 2018. In Indonesia, there were 19,381 meningitis cases with 1,025 deaths recorded by 2010. Cipto Mangunkusumo Hospital in Jakarta reported 273 meningitis cases between 2005 and 2006, with 114 resulting in death. Meanwhile, Prof. Dr. R. D. Kandou General Hospital (RDK) in Manado reported 112 cases of meningitis between 2012 and 2016.4,5
Based on the causative pathogens, meningitis can be classified into bacterial meningitis (S. pneumoniae, N. meningitidis, H. influenzae, etc), TB meningitis (M. tuberculosis), cryptococcal meningitis (C. neoformans), and viral meningitis (HEV, HSV, EBV, VZV, CMV).6–11 TB meningitis and cryptococcal meningitis are influenced by risk factors, especially HIV status. People living with HIV/AIDS experience weakened immune systems, making them more susceptible to meningitis infections, particularly TB and cryptococcal meningitis.12 A study by Imran et al. (2018) reported that the prevalence of cryptococcal meningitis was higher among HIV-positive patients (9%) compared to non-HIV patients (1%) at RSCM Jakarta in 2015–2016.13 However, the same study stated that the prevalence of TB meningitis at RSCM during the same period was higher in non-HIV patients (47%) compared to HIV-positive patients (22%). Meanwhile, a study conducted by Tampubolon et al. (2019) found that the prevalence of TB meningitis in HIV-positive patients was 6.6% at Sulianti Saroso Infectious Disease Hospital (SSIDH) in Jakarta between 2016 and 2018.
Untreated or improperly managed meningitis infections can lead to long-term neurological complications (sequelae), including cognitive decline, hearing loss, vision impairment, paralysis, and even death. Therefore, accurate diagnosis and identification of the causative pathogen are crucial for timely and appropriate treatment.14 The diagnosis and identification of meningitis pathogens can be conducted through cerebrospinal fluid (CSF) examination, which includes physical, biochemical, microbiological, and parasitological assessments.10,15
In cases of viral meningitis, molecular microbiological testing of CSF using PCR techniques is required to detect the genetic material of the causative virus.16 However, not all healthcare facilities in Indonesia are equipped to perform molecular diagnostics for viral meningitis, and the cost is often high. These diagnostic challenges, along with the lack of studies on CSF profiles in patients with meningitis at SSIDH, prompted the authors to conduct this research.
Materials and Methods
Design Study
This study used a cross-sectional survey. The research used secondary data from medical records of patients with meningitis who underwent CSF (cerebrospinal fluid) examination at Sulianti Saroso Infectious Disease Hospital.
Population and Sample
The sampling method used was purposive sampling. The study population consisted of patients aged 18 years or older who were diagnosed with suspected meningitis and underwent CSF examination between 2019 and 2023. The samples obtained from medical records included those from patients diagnosed with meningitis or meningoencephalitis.
Variables
Variables or data on demographic characteristics (age, sex), meningitis symptoms (neck stiffness, fever, headache, seizures, decreased consciousness), CSF examination results (physical, chemical, microbiological, and parasitological tests), and HIV status.
Examination
Meningitis can be detected in patients through clinical assessment of signs and symptoms, along with supporting diagnostic examinations. One of the key supporting tests used to both detect meningitis and identify the causative pathogen is cerebrospinal fluid (CSF) analysis. CSF examination in patients with meningitis is performed by collecting cerebrospinal fluid via a lumbar puncture. The physical examination of CSF includes assessment of fluid color, clarity, and leukocyte count and differential. Normal CSF is colorless and clear, whereas in patients with meningitis, the CSF may appear yellow (xanthochromia) or red. In addition, normal CSF is clear, whereas CSF in patients with meningitis is often cloudy or turbid. Molecular microbiological examination of CSF using polymerase chain reaction (PCR) techniques is performed to detect viral genetic material responsible for meningitis.16 Various biomarkers have been suggested to distinguish bacterial meningitis from viral meningitis, including detection of bacterial antigens in cerebrospinal fluid (CSF) parameters—namely mononuclear (MN) or lymphocyte cell counts, protein concentration, and glucose levels.17,18
Analysis
Data were processed using SPSS software version 16. Descriptive data analysis was carried out using univariate statistics to determine the frequency distributions of demographic characteristics, meningitis symptoms, CSF examination types, and HIV status.
Ethics
This study received ethical approval from the Health Research Ethics Committee of Sulianti Saroso Infectious Disease Hospital (SSIDH) under approval number DP.04.03/D.XXXIX.14/42/2024.
Results
Based on the study criteria, a total of 21 samples were obtained from the period 2019 to 2023. Of these 21 samples, 3 records (14.29%) lacked CSF examination results, and 1 record (4.76%) involved a patient under 18 years of age; thus, these were excluded from further analysis. Accordingly, the final number of samples analyzed in this study was 17 (80.95%).
Based on the data analysis of meningitis cases at Sulianti Saroso Infectious Disease Hospital. cases were classified into those with an identified pathogen (47.06%) and those with an unidentified pathogen (52.94%) (Figure 1).
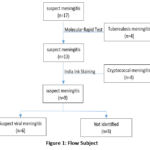 |
Figure 1: Flow Subject. |
The majority of meningitis cases occurred in the 18–27 age group (41.18%) and in males (70.59%) (Table 1).
Table 1. Frequency Distribution of Demographic Characteristics of Meningitis Patients
| Characteristics | M. TB(n=4) | Susp. M. TB (n=3) | M. Cryptococcal (n=4) | Susp. M. Viral(n=6) | Total(n=17) |
| Age | |||||
| 18 – 27 years | 2 (50) | 1 (33.33) | 1 (25) | 3 (50) | 7 (41.18) |
| 28 – 37 years | 0 (0) | 1 (33.33) | 2 (50) | 0 (0) | 3 (17.65) |
| 38 – 47 years | 1 (25) | 1 (33.33) | 1 (25) | 2 (33.33) | 5 (29.41) |
| 48 – 57 years | 1 (25) | 0 (0) | 0 (0) | 1 (16.67) | 2 (11.76) |
| Sex | |||||
| Male | 2 (50) | 3 (100) | 4 (100) | 3 (50) | 12 (70.59) |
| Female | 2 (50) | 0 (0) | 0 (0) | 3 (50) | 5 (29.41) |
The most commonly observed symptoms among meningitis cases were fever and headache (94.12%), followed by neck stiffness (58.82%). One case may have multiple symptoms (Table 2).
Table 2: Frequency Distribution of Symptoms in Meningitis Patients
| Characteristics | M. TB(n=4) | Susp. M. TB (n=3) | M. Cryptococcal (n=4) | Susp. M. Viral(n=6) | Total(n=17) |
| Symptom Triad | |||||
| Neck stiffness, yes | 3 (75) | 2 (66.67) | 2 (50) | 3 (50) | 10 (58.82) |
| Fever, yes | 4 (100) | 3 (100) | 4 (100) | 5 (83.33) | 16 (94.12) |
| Headache, yes | 4 (100) | 3 (100) | 4 (100) | 5 (83.33) | 16 (94.12) |
| Other Symptoms | |||||
| Seizures, yes | 1 (25) | 1 (33.33) | 1 (25) | 2 (33.33) | 5 (29.41) |
| Decreased consciousness, yes | 4 (100) | 2 (66.67) | 1 (25) | 4 (66.67) | 11 (64.71) |
The CSF color examination showed that most cases (82.35%) had colorless CSF, while yellow CSF was found in 17.65% of cases. In the clarity examination, clear CSF was observed in 82.35% of cases (Table 3).
Table 3: Frequency Distribution of CSF Physical Examination Findings
| Physical Examination | M. TB(n=4) | Susp. M. TB (n=3) | M. Cryptococcal (n=4) | Susp. M. Viral(n=6) | Total(n=17) |
| Color | |||||
| Colorless | 4 (100) | 2 (66.67) | 3 (75) | 5 (83.33) | 14 (82.35) |
| Yellow | 0 (0) | 1 (33.33) | 1 (25) | 1 (16.67) | 3 (17.65) |
| Red | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Clarity | |||||
| Clear | 4 (100) | 2 (66.67) | 2 (50) | 6 (100) | 14 (82.35) |
| Slightly turbid | 0 (0) | 1 (33.33) | 2 (50) | 0 (0) | 3 (17.65) |
| Turbid | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Leukocyte Count | |||||
| Normal | 0 (0) | 0 (0) | 0 (0) | 4 (66.67) | 4 (23.53) |
| Increased | 4 (100) | 3 (100) | 3 (75) | 2 (33.33) | 12 (70.59) |
| Markedly increased | 0 (0) | 0 (0) | 1 (25) | 0 (0) | 1 (5.88) |
| Percentage of PMN Cells | |||||
| 0 – 25% | 2 (50) | 2 (66.67) | 1 (25) | 5 (83.33) | 10 (58.82) |
| 26 – 50% | 2 (50) | 1 (33.33) | 2 (50) | 1 (16.67) | 6 (35.29) |
| 51 – 75% | 0 (0) | 0 (0) | 1 (25) | 0 (0) | 1 (5.88) |
| 76 – 100% | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Percentage of MN Cells | |||||
| 0 – 25% | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| 26 – 50% | 0 (0) | 0 (0) | 1 (25) | 1 (16.67) | 2 (11.76) |
| 51 – 75% | 2 (50) | 1 (33.33) | 2 (50) | 0 (0) | 5 (29.42) |
| 76 – 100% | 2 (50) | 2 (66.67) | 1 (25) | 5 (83.33) | 10 (58.82) |
Cases of Suspected Meningitis Viral were found with decreased protein levels (16.67%) and glucose levels (33.33%) (Table 4).
Table 4: Frequency Distribution of CSF Biochemical Examination Results
| Biochemical Examination | M. TB(n=4) | Susp. M. TB (n=3) | M. Cryptococcal (n=4) | Susp. M. Viral(n=6) | Total(n=17) |
| Protein Level | |||||
| Decreased | 0 (0) | 0 (0) | 0 (0) | 1 (16.67) | 1 (5.88) |
| Normal | 1 (25) | 1 (33.33) | 0 (0) | 1 (16.67) | 3 (17.65) |
| Increased | 0 (0) | 1 (33.33) | 2 (50) | 4 (66.67) | 7 (41.18) |
| Markedly increased | 3 (75) | 1 (33.33) | 2 (50) | 0 (0) | 6 (35.29) |
| Glucose Level | |||||
| Decreased | 1 (25) | 2 (66.67) | 4 (100) | 2 (33.33) | 9 (52.94) |
| Normal | 3 (75) | 1 (33.33) | 0 (0) | 4 (66.67) | 8 (47.06) |
| Increased | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
Gram staining and acid-fast bacilli (AFB) staining showed no bacterial detection in all cases (100%). However, the GeneXpert/MRT assay detected Mycobacterium tuberculosis genetic material in 23.53% of cases (4 out of 17) (Table 5).
Table 5: Frequency Distribution of CSF Microbiological Examination Results
| Characteristics | M. TB (n=4) |
Susp. M. TB (n=3) | M. Cryptococcal (n=4) | Susp. M. Viral (n=6) |
Total (n=17) |
| Gram Stain | |||||
| Gram-positive | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Gram-negative | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| No bacteria found | 4 (100) | 3 (100) | 4 (100) | 6 (100) | 17 (100) |
| AFB Staining | |||||
| Positive | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Negative | 4 (100) | 3 (100) | 4 (100) | 6 (100) | 17 (100) |
| Molecular Rapid Test | |||||
| Positive | 4 (100) | 0 (0) | 0 (0) | 0 (0) | 4 (23.53) |
| Negative | 0 (0) | 0 (0) | 4 (100) | 5 (83.33) | 9 (52.94) |
| Not performed | 0 (0) | 3 (100) | 0 (0) | 1 (16.67) | 4 (23.53) |
| Viral PCR Panel Test | |||||
| Positive | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Negative | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Not performed | 4 (100) | 3 (100) | 4 (100) | 6 (100) | 17 (100) |
CSF parasitological results by India ink staining revealed a positivity rate of 23.53% (Table 6).
Table 6: Frequency Distribution of CSF Parasitological Examination Results
| India Ink Staining | M. TB(n=4) | Susp. M. TB (n=3) | M. Cryptococcal (n=4) | Susp. M. Viral(n=6) | Total(n=17) |
| Positive | 0 (0) | 0 (0) | 4 (100) | 0 (0) | 4 (23.53) |
| Negative | 4 (100) | 2 (66.67) | 0 (0) | 3 (50) | 9 (52.94) |
| Not performed | 0 (0) | 1 (33.33) | 0 (0) | 3 (50) | 4 (23.53) |
Meningitis cases occur in HIV-negative patients (64.71%). Meningitis cases in HIV-positive patients consisted of 1 suspected TB meningitis case (33.33%), 4 cryptococcal meningitis cases (100%), and one suspected viral meningitis case (16.67%) (Table 7).
Table 7: Frequency Distribution of HIV Status in Meningitis Patients
| HIV Status | M. TB(n=4) | Susp. M. TB (n=3) | M. Cryptococcal (n=4) | Susp. M. Viral(n=6) | Total(n=17) |
| Positive | 0 (0) | 1 (33.33) | 4 (100) | 1 (16.67) | 6 (35.29) |
| Negative | 4 (100) | 2 (66.67) | 0 (0) | 5 (83.33) | 11 (64.71) |
Discussion
Previous studies have reported that TB meningitis cases are most common in individuals aged 30, while cryptococcal meningitis is most common among individuals aged 30-40, and viral meningitis is most common in individuals aged 27–49.8,19,20
The table also indicates that meningitis cases were more prevalent among male patients (70.59%) compared to female patients (29.41%). These findings are in line with previous research, which has shown that the majority of TB meningitis, cryptococcal meningitis, and viral meningitis cases occur in male patients 8,15,19
The results of this study are consistent with previous research, which has reported that fever and headache are common symptoms in cases of TB meningitis, cryptococcal meningitis, and viral meningitis.9,21,22 This study also aligns with earlier findings indicating that neck stiffness is only present in a subset of meningitis cases.23
Other symptoms found in cases of meningoencephalitis include decreased consciousness (64.71%) and seizures (29.41%). This finding supports previous research suggesting that only a small proportion of patients with meningitis experience seizures.24 Decreased consciousness was most frequently observed in TB meningitis cases (100%), followed by suspected TB meningitis (66.67%), suspected viral meningitis (66.67%), and cryptococcal meningitis (25%). These findings are in line with earlier studies indicating that decreased consciousness is common among patients with TB infections due to the spread of infection into the brain parenchyma.25,26 Previous studies have also noted that only some cases of viral meningitis and a small proportion of cases of cryptococcal meningitis present with decreased consciousness.9,27
The results of this study are consistent with previous research, indicating that most cases of meningitis are associated with clear, colorless cerebrospinal fluid (CSF).28,29 Changes in CSF color to yellow and turbidity are generally caused by elevated protein levels exceeding 1 g/L (100 mg/dL) or increased leukocyte counts.
In the leukocyte count examination, cases were found with normal leukocyte levels (23.53%), elevated levels (70.59%), and markedly elevated levels (5.88%). These findings align with previous studies indicating that leukocyte levels typically increase in cases of TB and cryptococcal meningitis.24,30 Meanwhile, patients with viral meningitis generally experience three phases, beginning with a phase of no leukocyte elevation, during which leukocyte levels remain within normal limits.31
Examination of CSF polymorphonuclear (PMN) and mononuclear (MN) cells can help identify the causative pathogen. For PMN cells, cases were found with PMN percentages of 0–25% (58.82%), 26–50% (35.29%), and 51–75% (5.88%). An elevated PMN count—particularly neutrophils—that exceeds MN cell counts in CSF is usually found in cases of bacterial meningitis. PMN cells, especially neutrophils, play a key role in innate immunity, primarily through phagocytosis of bacteria.32 No cases were found with PMN counts exceeding 75%, as such a high level is commonly associated with bacterial meningitis. Acid-fast bacilli (AFB) staining is performed to detect Mycobacterium tuberculosis, a bacterium that can survive in acidic environments. The GeneXpert or Xpert MTB/RIF assay is used to detect DNA from Mycobacterium tuberculosis, the causative agent of tuberculous meningitis, and to identify rifampicin (RIF) resistance in the bacterium.33
Regarding MN cell counts, cases were observed with MN percentages of 26–50% (11.76%), 51–75% (29.41%), and 76–100% (58.82%). An increased MN count—particularly of lymphocytes—surpassing PMN counts in CSF is typically found in cases of TB, cryptococcal, and viral meningitis. These findings are in line with previous research showing that patients with TB and cryptococcal meningitis exhibit elevated leukocyte counts, particularly in MN cells.30,34 Patients with viral meningitis typically go through three leukocyte phases: an initial phase with no leukocyte increase, a second phase with PMN-dominant leukocytosis, and a third phase marked by MN-dominant leukocytosis 31
The findings of this study are consistent with previous research, which reported that patients with TB meningitis and cryptococcal meningitis experienced elevated CSF protein levels exceeding 100 mg/dL.30,34 Meanwhile, earlier studies also noted that CSF protein levels in patients with viral meningitis typically remain normal or only slightly elevated, except in cases of VZV infection, which can lead to increased protein levels.35
In the biochemical analysis of CSF, decreased glucose levels (52.94%). The results regarding TB meningitis contrast with previous studies reporting a consistent decrease in CSF glucose levels among patients with TB meningitis.25 It is possible that TB meningitis and suspected TB meningitis patients who presented with normal CSF glucose levels had already received treatment, leading to a reduction in bacterial load and normalization of glucose levels.
On the other hand, findings regarding cryptococcal meningitis are consistent with previous studies reporting decreased glucose levels in patients with this infection.34,36 The reduction in glucose levels in patients with meningitis is due to blood-brain barrier disruption and to glucose utilization by microorganisms during metabolism. Patients with viral meningitis tend to have normal glucose levels, as viruses do not carry out metabolic activity.24 The findings of this study are consistent with previous research, which stated that the GeneXpert test has higher sensitivity than the AFB examination. False-negative results in AFB testing may occur when the concentration of M. tuberculosis in the CSF sample is too low.37
Therefore, TB meningitis cases that test positive on GeneXpert but negative on AFB examination are highly plausible. Meanwhile, GeneXpert testing was not performed in 4 out of 17 cases (23.53%), namely in all 3 suspected TB meningitis cases (100%) and in 1 of the 6 suspected viral meningitis cases (16.67%); thus, the causative pathogen could not be identified. The three suspected TB cases were classified based on a history of pulmonary TB, clinical findings, and head imaging results.
Regarding the viral PCR panel, no cases were tested. Viral PCR panel testing has not yet been implemented at SSIDH, which is why the causative pathogen in suspected viral meningitis cases could not be determined. Negative results in microbiological examinations (Gram staining, AFB staining, and GeneXpert or TCM) in suspected viral meningitis cases indicate that the infection was not caused by bacteria, TB, or fungi. Thus, viral PCR panel testing should be performed to determine whether the infection is viral in origin or caused by another pathogen. The findings of this study are consistent with previous research indicating that patients with cryptococcal meningitis typically show positive India ink staining results.8,27 A positive result on CSF parasitology examination indicates the presence of Cryptococcus neoformans in the cerebrospinal fluid, as detected by India ink staining. In microscopic examination of CSF samples from patients with cryptococcal meningitis, C. neoformans fungi can be observed as round, white-colored organisms against a dark background.
The findings of this study are consistent with previous research stating that all cryptococcal meningitis cases occurred in individuals with HIV-positive status 24 These findings also align with the CDC (2021), which categorizes cryptococcal meningitis caused by Cryptococcus neoformans as an AIDS-related opportunistic infection. In addition to cryptococcosis caused by C. neoformans, the CDC (2021) also identifies tuberculosis and cytomegalovirus infections as opportunistic infections. These meningitis-causing pathogens can easily infect individuals with weakened immune systems, such as those with HIV/AIDS.
Suspected viral meningitis cases accounted for only 6 of the 17 total cases (35.29%) identified at Sulianti Saroso Infectious Disease Hospital during the 2019–2023 period. These cases were classified as viral meningitis based on biochemical CSF examination results, including elevated mononuclear (MN) or lymphocyte cell counts, normal or slightly elevated CSF protein levels, and normal CSF glucose levels.17 Microbiological examination of CSF revealed negative results on Gram staining, acid-fast bacilli (AFB) tests, and GeneXpert, indicating no bacterial pathogens or Mycobacterium tuberculosis as causes of bacterial or TB meningitis. Furthermore, negative results from CSF parasitological examination using India ink staining ruled out cryptococcal meningitis. These findings suggest viral etiology in the six analyzed samples, underscoring the need for a viral PCR panel to identify the specific viral pathogen. CSF findings may also be influenced by prior empirical antibiotic therapy, a history of antibiotic use, HIV co-infection, and delayed referral to tertiary care due to limitations in healthcare facilities.
A major limitation of this study is incomplete CSF testing, which prevented the identification of the causative pathogens in some cases. The absence of GeneXpert or TCM testing and India ink staining in several samples, along with the lack of viral PCR panel testing across all samples, resulted in pathogens being undetermined in suspected TB and viral meningitis cases. To improve pathogen identification in meningitis cases, it is essential to establish a standardized CSF laboratory testing protocol that includes diagnostic methods for all major pathogen types, particularly viruses. This would ensure that no critical examinations are missed and that the causative pathogens—especially viruses—can be promptly identified. However, this data can support the need for viral testing in meningitis cases, especially in limited health facilities and areas with limited national insurance coverage.
Conclusion
Based on cerebrospinal fluid (CSF) examination results. The causative pathogens of meningitis may include Mycobacterium tuberculosis, Cryptococcus fungi, and suspected viruses. Suspected viral meningitis cases accounted for 6 out of 17 total cases (35.29%) based on physical examination (colour, clarity, and count of cells), elevated mononuclear (MN) or lymphocyte cell counts, and biochemical examination (normal or slightly elevated CSF protein levels, and normal CSF glucose levels)
Acknowledgement
The authors would like to express their sincere gratitude to the President Director and the Director of Human Resources of Sulianti Saroso Infectious Disease Hospital for their valuable support in implementing this research.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This study received ethical approval from the Health Research Ethics Committee of Sulianti Saroso Infectious Disease Hospital (SSIDH) under approval number DP.04.03/D.XXXIX.14/42/2024.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable.
Author contributions
- Maria Lawrensia Tampubolon: Conceptualization, Data Analysis, Methodology;
- Edith Nadya Apriliani: Data Collection, Data Analysis, Writing – Original Draft;
- Daniel Joko Wahyono: Supervision;
- Siti Maemun: Writing – Review & Editing.
References
- Saraiva M das GG, Santos ECS, Saraceni V, et al. Epidemiology of infectious meningitis in the State of Amazonas, Brazil. Rev Soc Bras Med Trop. 2015;48 Suppl 1:79-86. doi:10.1590/0037-8682-0116-2014
CrossRef - Mathew S, Al Khatib HA, Al Ansari K, et al. Epidemiology Profile of Viral Meningitis Infections Among Patients in Qatar (2015–2018). Front Med. 2021;8(June):1-11. doi:10.3389/fmed.2021.663694
CrossRef - Riasari N, Bintoro A. Profil Klinis Pasien Meningoencephalitis di Instalasi Rawat Intensif RSUP. Dr. Kariadi Semarang. CoMPHI J Community Med Public Heal Indones J. 2022;2:44-50. doi:10.37148/comphijournal.v2i2.44
CrossRef - Pangandaheng EASS, Mawuntu AHP, Karema W. Gambaran Tingkat Pengetahuan dan Perilaku Masyarakat Tentang Penyakit Meningitis di Kelurahan Soataloara II Kecamatan Tahuna Kabupaten Kepulauan Sangihe. e-CliniC. 2017;5(2).
CrossRef - Enggune CM, Pertiwi JM, Mawuntu AHP. Gambaran Fungsi Kognitif Pasien Infeksi Otak Di Rsup Prof. Dr. RD Kandou Manado. JKK (Jurnal Kedokt Klin. 2018;2(2):1-6.
- Nuraeni F, Dzulfikar DLH, Solek P. Feritin Cairan Serebrospinal sebagai Biomarker untuk Membedakan Meningitis Bakterialis dan Meningitis Tuberkulosis pada Anak. Sari Pediatr. 2020;21(4):246-252.
CrossRef - Iswari P, Widhiani N, Akelba S, et al. Aspek Patofisiologi Meningitis Tuberkulosis dan Tuberkuloma. Unram Med J. 2023;12:262-268. doi:10.29303/jk.v12i3.4448
CrossRef - Sutrisno P, Tumewah R, Mawuntu AHP, Bernadus J. LUARAN PASIEN AIDS DENGAN MENINGITIS KRIPTOKOKUS DEFINIT DI RSUP PROF RD KANDOU MANADO. JKK (Jurnal Kedokt Klin. 2017;1(2):22-28.
- Wami AA, Hundie GB, Ambachew R, et al. High rate of human enteroviruses among clinically suspected meningitis cases at selected Hospitals in Addis Ababa, Ethiopia. PLoS One. 2021;16(11):e0258652.
CrossRef - Arafa ZAA, Gabr MS, Kamel EM, ElMasry SA, Fahim NA. Cerebrospinal Fluid Lactate as a Differential Biomarker for Bacterial and Viral Meningitis. Egypt J Immunol. 2023;30(3):148-161. http://europepmc.org/abstract/MED/37440535
CrossRef - Taniguchi T, Tsuha S, Shiiki S, Narita M. Point-of-care cerebrospinal fluid Gram stain for the management of acute meningitis in adults: a retrospective observational study. Ann Clin Microbiol Antimicrob. 2020;19:1-11.
CrossRef - Centers for Disease Control and Prevention. About HIV. Published 2021. https://www.cdc.gov/hiv/about/index.html
- Imran D, Estiasari R, Maharani K, et al. Presentation, etiology, and outcome of brain infections in an Indonesian hospital: A cohort study. Neurol Clin Pract. 2018;8(5):379-388. doi:10.1212/CPJ.0000000000000517
CrossRef - Dezfouli SMM, Khosravi S. Impact of meningitis on cognitive skills and development in children. Indian J Forensic Med Toxicol. 2021;15(1):2674-2678. doi:10.37506/ijfmt.v15i1.13802
CrossRef - Sow MS, Diallo ST, Camara A. Microbiological Profile of Meningitis: Analyzes of Cerebrospinal Fluid at the Laboratory of the National Institute of Public Health in 2017. Microbiol Infect Dis. 2020;4(1):1-5.
CrossRef - McGill F, Griffiths MJ, Bonnett LJ, et al. Incidence, aetiology, and sequelae of viral meningitis in UK adults: a multicentre prospective observational cohort study. Lancet Infect Dis. 2018;18(9):992-1003.
CrossRef - Alnomasy SF, Alotaibi BS, Mujamammi AH, Hassan EA, Ali ME. Microbial aspects and potential markers for differentiation between bacterial and viral meningitis among adult patients. PLoS One. 2021;16(6):e0251518. https://doi.org/10.1371/journal.pone.0251518
CrossRef - Shahan B, Choi EY, Nieves G. Cerebrospinal Fluid Analysis. Am Fam Physician. 2021;103(7):422-428.
- Kuswanto RA, Rizal A, Koesoemadinata RRC. Association between Fever during Admission and Outcome of Treatment in Tuberculous Meningitis Patients in Hasan Sadikin General Hospital Bandung. J Med Heal. 2019;2(3). doi:10.28932/jmh.v2i3.1233
CrossRef - Jakobsen A, Skov MT, Larsen L, et al. Herpes Simplex Virus 2 Meningitis in Adults: A Prospective, Nationwide, Population-Based Cohort Study. Clin Infect Dis an Off Publ Infect Dis Soc Am. 2022;75(5):753-760. doi:10.1093/cid/ciab1071
CrossRef - Paruntu D, Mawuntu AHP, Ngantung D, Bernadus JBB. PERBANDINGAN PEMERIKSAAN LATERAL FLOW ASSAY CAIRAN SEREBROSPINAL DENGAN TINTA INDIA DALAM MENDETEKSI MENINGITIS KRIPTOKOKUS PADA PASIEN AIDS. JKK (Jurnal Kedokt Klin. 2017;1(2):13-21.
- Quayum S, Miah MT, Ferdous T, et al. Clinical and Diagnostic Findings Among Tubercular Meningitis Patients without Pulmonary Signs and Symptoms. SAS J Med. 2023;8:883-888.
CrossRef - Liu Y, Carlson SA, Watson KB, Xu F, Greenlund KJ. Trends in the Prevalence of Chronic Obstructive Pulmonary Disease Among Adults Aged ≥18 Years — United States, 2011–2021. MMWR Recomm Reports. 2023;72(46):1250-1256. doi:10.15585/mmwr.mm7246a1
CrossRef - de Blauw D, Bruning AHL, Wolthers KC, et al. Incidence of Childhood Meningoencephalitis in Children With a Suspected Meningoencephalitis in the Netherlands. Pediatr Infect Dis J. 2022;41(4):290-296. doi:10.1097/INF.0000000000003441
CrossRef - Prakoso AB, Dewi MM, Sugianli AK. Gambaran cairan serebrospinal pada pasien anak dengan infeksi susunan saraf pusat di rumah sakit rujukan Jawa Barat. Sari Pediatr. 2020;21(6):339-345.
CrossRef - Nataprawira HM, Gafar F, Risan NA, et al. Treatment outcomes of childhood tuberculous meningitis in a real-world retrospective cohort, Bandung, Indonesia. Emerg Infect Dis. 2022;28(3):660.
CrossRef - Gunawan P, Feliana F, Cucunawangsih C. GAMBARAN KLINIS DAN ANALISA CAIRAN SEREBROSPINAL PADA PASIEN MENINGITIS KRIPTOKOKUS DENGAN ATAU TANPA INFEKSI HUMAN IMMUNODEFICIENCY VIRUS DI RUMAH SAKIT SHLV. Medicinus. 2018;4. doi:10.19166/med.v4i8.1182
CrossRef - Riasari NS, Bintoro AC. Profil Klinis Pasien Meningoensefalitis di Instalasi Rawat Intensif RSUP. Dr. Kariadi Semarang. CoMPHI J Community Med Public Heal Indones J. 2021;2(2):44-50.
CrossRef - Yunita Y, Riastiti Y, Muhyi A. Karakteristik Analisis LCS Meningoensefalitis Anak di RSUD Abdoel Wahab Sjahranie Samarinda Tahun 2019-2022. J Kesehat Andalas. 2023;12(2):87-92.
CrossRef - García-Grimshaw M, Gutiérrez-Manjarrez FA, Navarro-Álvarez S, González-Duarte A. Clinical, Imaging, and Laboratory Characteristics of Adult Mexican Patients with Tuberculous Meningitis: A Retrospective Cohort Study. J Epidemiol Glob Health. 2020;10(1):59-64. doi:10.2991/jegh.k.191023.001
CrossRef - Lee KY, Seol JH, Yi CH, Lee WH. Cerebrospinal fluid type I interferon and cytokine profiles in enteroviral meningitis according to the presence or absence of pleocytosis. Pediatr Neonatol. 2021;62(3):305-311.
CrossRef - Widayanti A, Rizal A, Parwati I. Association Between Polymorphonuclear Leukocyte Cell Percentage in Cerebrospinal Fluid and Outcome of Tuberculous Meningitis Patients in a Tertiary Hospital in Indonesia. Maj Kedokt Bandung. 2020;52(4):220-226.
CrossRef - Marouane C, Smaoui S, Kammoun S, Slim L, Messadi-Akrout F. Evaluation of molecular detection of extrapulmonary tuberculosis and resistance to rifampicin with GeneXpert® MTB/RIF. Med Mal Infect. 2016;46(1):20-24. doi:10.1016/j.medmal.2015.10.012
CrossRef - Takahashi Y, Osawa R, Kubota Y, et al. Early diagnosis of Cryptococcus neoformans var. grubii meningitis using multiplex PCR assay in an immunocompetent patient. J Infect Chemother. 2021;27(12):1765-1768.
CrossRef - Cui S, Chen S, Li X, Liu S, Wang F. Prevalence of venous thromboembolism in patients with severe novel coronavirus pneumonia. J Thromb Haemost. 2020;18(6):1421-1424. doi:10.1111/jth.14830
CrossRef - Zhang C, Tan Z, Tian F. Impaired consciousness and decreased glucose concentration of CSF as prognostic factors in immunocompetent patients with cryptococcal meningitis. BMC Infect Dis. 2020;20:1-6.
CrossRef - Metcalf T, Soria J, Montano SM, et al. Evaluation of the GeneXpert MTB/RIF in patients with presumptive tuberculous meningitis. PLoS One. 2018;13(6):e0198695. doi:10.1371/journal.pone.0198695
CrossRef







