Manuscript accepted on :02-01-2026
Published online on: 27-01-2026
Plagiarism Check: Yes
Reviewed by: Dr. Daniel Oyeka and Dr. Meena Kumari K
Second Review by: Dr. Takkella Nagamma
Final Approval by: Dr. Anton R Keslav
Siti Nur Syahida Abdul Jalil1 , Ng Chin Theng2
, Ng Chin Theng2 , Fong Lai Yen3
, Fong Lai Yen3 , Yong Yoke Keong4
, Yong Yoke Keong4 , Muhammad Nazrul Hakim1,5*
, Muhammad Nazrul Hakim1,5* and Zuraini Ahmad1
and Zuraini Ahmad1
1Department of Biomedical Sciences, Faculty of Medicine and Health Sciences, Universiti Putra Malaysia, Serdang, Selangor, Malaysia
2Department of Physiology, Asian Institute of Medicine, Science and Technology, Kedah, Malaysia.
3Department of Pre-clinical Sciences, M. Kandiah Faculty of Medicine and Health
Sciences, Universiti Tunku Abdul Rahman, Kajang, Selangor, Malaysia
4Department of Human Anatomy, Faculty of Medicine and Health Sciences, Universiti Putra Malaysia, Serdang, Selangor, Malaysia.
5Laboratory of Halal Science Research, Halal Product Institute, Universiti Putra Malaysia, Serdang, Selangor, Malaysia.
Corresponding Author E-mail: nazrulh@upm.edu.my
DOI : https://dx.doi.org/10.13005/bpj/3391
Abstract
Inflammation is a protective physiological response that may cause tissue damage when prolonged. Copper–amino acid complexes, including Cu(L-alanine)₂, have been reported to possess various biological activities. However, their anti-inflammatory potential remains underexplored. This study evaluated the in vivo anti-inflammatory effects of Cu(L-ala)₂ using three experimental models in rats: acetic acid-induced peritoneal vascular permeability, carrageenan-induced paw edema, and nitric oxide (NO) production in paw tissue. Rats were divided into four groups (n = 6) and treated with vehicle control (1% DMSO), indomethacin (10 mg/kg), or Cu(L-ala)₂ at doses of 2 or 20 mg/kg. Acetic acid markedly increased vascular permeability, which was significantly inhibited by Cu(L-ala)₂ by 74.1% (2 mg/kg) and 81.23% (20 mg/kg). In the paw edema model, both doses significantly reduced inflammation compared with the control, with the 2 mg/kg dose showing the greatest inhibition (63.79%) at 1 hour. In contrast, Cu(L-ala)₂ produced only a moderate reduction in NO levels, reaching statistical significance only at 20 mg/kg (27.56%), while indomethacin showed a stronger effect (46.28%). Overall, Cu(L-ala)₂ demonstrated significant anti-inflammatory activity by reducing vascular permeability and edema formation. However, its limited effect on NO production suggests that its mechanism of action may be largely NO-independent. Further studies are required to clarify its molecular targets, dose–response behavior, and therapeutic potential.
Keywords
Animal models; Anti-inflammatory; Cu(L-Alanine)₂; Paw edema; Peritoneal vascular permeability
Download this article as:| Copy the following to cite this article: Jalil S. N. S. A, Theng N. G, Yen F. L, Keong Y. Y, Hakim M. N, Ahmad Z. Assessment of the Anti-Inflammatory Activity of Cu(L-Alanine)₂ in Acute Inflammation Animal Models. Biomed Pharmacol J 2026;19(1). |
| Copy the following to cite this URL: Jalil S. N. S. A, Theng N. G, Yen F. L, Keong Y. Y, Hakim M. N, Ahmad Z. Assessment of the Anti-Inflammatory Activity of Cu(L-Alanine)₂ in Acute Inflammation Animal Models. Biomed Pharmacol J 2026;19(1). Available from: https://bit.ly/46hFqTF |
Introduction
Inflammation underlies many major noncommunicable diseases (NCDs), including cardiovascular disease, diabetes, cancer, and autoimmune disorders, which together account for about 75% of global deaths. The global economic burden of NCDs is estimated to exceed US$30 trillion over recent decades, driven by direct healthcare costs and indirect losses from reduced productivity and disability. Consequently, inflammation-related diseases represent one of the largest and fastest-growing economic burdens worldwide, particularly in aging and low- to middle-income populations.1,2
The interaction of metal complexes with nucleic acids and their constituents has been of interest to bioinorganic chemistry, especially since the discovery of cisplatin and its analogs. These studies have contributed to the development and comprehension of new chemotherapeutic medications meant to treat a variety of diseases. Copper (Cu) complexes of non-steroidal anti-inflammatory drugs (NSAIDs) have been found to be more effective than their parent drugs in many recognized models of inflammation.3 Although these low-toxicity copper medications have not yet made it to the general public, they are of great interest because many anti-inflammatory drug treatments, such as those based on NSAIDs, are still either largely ineffective or have problematic side effects related to the kidneys, gastrointestinal tract, and heart. However, the source of Cu-NSAIDs’ anti-inflammatory and gastric-sparing properties is yet unknown.4 In order to better understand the anti-inflammatory characteristics of copper complexes with amino acids, namely the Cu(L-ala)2 molecule, in inflammation-induced experimental models with regard to enhanced permeability and edema production in rats, this study was conducted. The effect of Cu(L-ala)2 compound on the level of nitric oxide (NO); a permeability-regulating molecule, is also investigated.
Copper, an essential trace element required for enzymes and animal tissues in biological systems in both the +1 and +2 valence states, is found in redox metalloenzymes, oxygenation, and oxygen-carrying proteins. The binding of copper ions to specific sites may change the conformational configurations of proteins, polynucleotides, DNA, and biomembranes.5 In addition to its benefits, copper (II), Cu2+, has been linked to a number of illnesses, including Wilson’s disease, Menkes disease, Alzheimer’s disease, Parkinson’s disease, prion disease, and other neurodegenerative diseases. This is because the redox-active copper ions produce a toxin that causes reactive oxygen species (ROS) to be produced through a Fenton or Haber-Weiss mechanism.6 There are a number of studies that relate copper as being an important element during inflammation. The discovery that metallic Cu may dissolve in perspiration and be absorbed via the skin provided a possible scientific foundation for the anti-inflammatory Cu bracelet treatment. It is thought that copper has anti-inflammatory properties. The distribution of copper in the blood varies in patients with osteoarthritis and RA. For example, arthritis patients’ total serum copper levels have been shown to be higher than those of controls.4 According to Ji et al.,5 copper ions have a strong affinity for DNA and can change the conformational structures of proteins, polynucleotides, DNA, and biomembranes by adhering to certain sites. Recently we have reported that Cu(Phen)(C-dimethylglycine)NO3 significantly proliferation of colorectal carcinoma (HT-29) and ovarian carcinoma (A2780) cells.7 Thus, the general objective of this study is to study the anti-inflammatory effects of administrating Cu (L-ala)2 on inflammation induced in experimental rat models.
Materials and Methods
Chemicals and Reagents
Cu (L-ala)2 was kindly provided by Department of Chemistry, Faculty of Science, University of Malaya. Indomethacin and Griess reagent (Sigma-Aldrich, Malaysia), acetic acid (J.T. Baker, Thailand), chloroform (Scharlau Chemie S.A., European Union), ethyl alcohol (Fisher Scientific, Malaysia), Carrageenan powder, Evans blue dye and all other standard chemicals are from Sigma-Aldrich, Malaysia.
Experimental Animals
Male Sprague-Dawley rats weighed 180-200 g and housed at Animal House in Faculty of Medicine and Health Medicine and Health Sciences, UPM, prior to and during the experiments. The room temperature for rats were maintained at 27-30oC with 12 hours of light and dark cycles. Pelleted feed and tap water stored in water bottles were provided ad libitum throughout the study. The animals were acclimated for a week to the animal house condition. The animals were grouped prior to experiment with six rats per each group. The animals used in this study were approved under the reference number UPM/FPSK/PADS/BR-UUH/00455 by Animals Care and Use Committee (ACUC), Faculty of Medicine and Health Sciences, UPM.
24 rats were selected randomly and divided into 4 groups per experiment. All treatments were administered orally by using oral gavage needle. Negative controls received dosed vehicle (1% DMSO in DH2O), positive control received 10 mg/kg Indomethacin, treatment groups 2 and 20 mg/kg Cu(L-ala)2. New rats were used for each experiments of Acetic acid-induced Peritoneal Vascular Permeability, Carrageenan-induced Paw Edema and Nitric Oxide Level Measurements.
Anti-inflammatory Experiments
Three anti-inflammatory experiments were performed; acetic acid-induced peritoneal vascular permeability, carrageenan-induced paw inflammation and nitric oxide level measurement during carrageenan-induced inflammation.
Acetic acid-induced Peritoneal Vascular Permeability Assay
The acetic acid-induced peritoneal vascular permeability assay was employed to evaluate peripheral anti-inflammatory activity, as this model reflects the release of endogenous nociceptive mediators sensitive to non-steroidal anti-inflammatory drugs (NSAIDs).8,9 The procedure was adapted from Dantas et al.10 Rats were randomly assigned into four groups (n = 6 per group). One hour after oral administration of test compounds or vehicle, Evans Blue (1%, 0.05 mL/10 g body weight) was administered intravenously. After 10 minutes, acetic acid (0.7%, 0.05 mL/10 g body weight) was injected intraperitoneally. Thirty minutes later, animals were sacrificed, and the peritoneal cavity was washed with 10 mL of normal saline. The collected exudate was centrifuged at 3400 rpm for 15 minutes, and Evans Blue concentration in the supernatant was quantified spectrophotometrically at 590 nm using a microplate reader (Tecan Infinite M200, Switzerland). Results were expressed as absorbance (A590).
Carrageenan-induced Paw Edema
The carrageenan-induced paw edema model was used to assess in vivo anti-inflammatory activity, as it is sensitive to non-steroidal anti-inflammatory drugs and reflects both early and late inflammatory phases.11 The procedure was adapted from Huang et al.12 Twenty-four rats were randomly assigned to four groups (n = 6): vehicle control (1% DMSO), indomethacin (10 mg/kg), Cu(L-ala)₂ (2 mg/kg), and Cu(L-ala)₂ (20 mg/kg), administered orally. One hour later, inflammation was induced by subplantar injection of 1% carrageenan (0.1 mL) into the right hind paw. Paw volume was measured using a plethysmometer (Ugo Basile, Model 7140) immediately after carrageenan injection and at 1, 2, 3, 4, and 5 hours. Edema was calculated as the change in paw volume relative to baseline. The anti-inflammatory effect was expressed as a percentage inhibition of edema compared with the control group13.
Nitric oxide (NO) level from carrageenan-induced paw edema
Nitric oxide production during acute inflammation was assessed using the Griess reaction, as the L-arginine–NO pathway is involved in carrageenan-induced inflammatory responses. The procedure was adapted from Huang et al.12 Twenty-four rats were randomly assigned to four groups (n = 6) and pre-treated as described previously. One hour after oral administration, paw inflammation was induced by subplantar injection of 1% carrageenan (0.1 mL) into the right hind paw. After one hour, animals were sacrificed, and approximately 1.6 g of paw tissue was collected, homogenized in cold distilled water, and centrifuged at 8000 rpm for 10 minutes. Supernatant samples (50 μL) were mixed with an equal volume of Griess reagent in a 96-well plate, incubated for 10 minutes at room temperature, and absorbance was measured at 548 nm using a microplate reader (Tecan Infinite M200, Switzerland).
Statistical Analysis
All data in the figure and text were expressed as mean ± SEM. Statistical analysis was performed using One-way ANOVA and was further analyzed with Tukey post hoc test. p value lesser than 0.05 (p<0.05) was considered significant.
Results
Effects of Cu (L-Ala)2 on Acetic acid-induced Peritoneal Vascular Permeability
In this inflammatory model, increased of vascular permeability was caused by the inflammation induced by acetic-acid. The peritoneal exudates were collected and the results of dye leakage were recorded and showed in Table 1. Oral pretreatment of Cu(L-ala)2 compound at 2 and 20 mg/kg significantly reduced (p <0.05) the increased in peritoneal vascular permeability compared to the negative control group which was the non-treated group (16.645 μg/rat ± 2.263). Standard drug-treated group (Indomethacin) also showed a significant reduction (p < 0.05) in the increased in peritoneal vascular permeability when compared to the negative control group. When comparing between standard drug-treated group and compound treated group, the result showed there was no significant difference between them. There was also no significant difference when compared between the compound treated groups. Between the 2 and 20 mg/kg of Cu(L-ala)2 compound, the compound at 20 mg/kg showed the lowest dye leakage in peritoneal vascular permeability (3.128 μg/rat ±0.762) compared to 2 mg/kg of compound (4.312 μg/rat ±1.25). The percentage of inhibition of dye leakage of each treatment with their respective doses were shown in Table 1. 20 mg/kg of Cu(L-Ala)2 compound showed the highest percentage of inhibition (81.23 %) compared to 2 mg/kg of Cu(L-ala)2 (74.1%). Standard drug-treated group showed the lowest percentage of inhibition (64.46 %). There was no significant difference among the treated groups.
Table 1: Effects of Cu (L-ala)2 on increased peritoneal vascular permeability induced by acetic acid.
| Treatment | Dose (mg/kg) | Dye Leakage(μg/rat) | % Inhibition |
| Negative control | – | 16.65 ± 2.26a | – |
| Positive control (Indomethacin) | 10 | 5.92 ± 2.08b | 64.46 |
| Cu (L-ala)2 | 2 | 4.31 ± 1.25b | 74.1 |
| 20 | 3.13 ± 0.76b | 81.23 |
Values are mean ± SEM, n= 6
a-b: mean with different superscript differ significantly (p<0.05) between groups
Effects of Cu (L-ala)2 on Carrageenan-induced Paw Edema
The paw volume was evaluated six times at intervals of one hour in this inflammatory investigation. Figure 1 displays the mean paw volume of carrageenan-induced paw edema assessed at -1, 0, 1, 2, 3, 4, and 5 hours. Subplantar injection at the right paw of rats had led to a quadratic and time- dependent increase in paw volume. All the six times measurement there was significantly different in the mean of paw volume (P<0.05) in Cu(L-ala)2 compound treatment groups compared to the non-treated group. Indomethacin (positive control) also showed significant different to non-treated group. The treatment group of 2 mg/kg and 20 mg/kg Cu(L-ala)2 also show a significant difference (p<0.05). When analyzed for each hour, the paw volume was increased significantly (p<0.05) by the time of carrageenan injection (0 hour) and was maximal (0.19 ml ± 0.04) at the first hour after injection of carrageenan in the negative control group. Then the paw volume started to decrease slightly at hour 2 until fifth hour. In the drug-treated group (Indomethacin), the paw volume increased significantly (p<0.05) at 0 hour and started to decrease significantly from the first hour until the fourth hour. At the fifth hour, the paw volume did not show any significant difference compared to the prior treatment hour (-1). On the other hand, the compound-treated group, which was the 2mg/kg Cu(L-ala)2 did not show any significant increase starting from the prior treatment hour (-1) until the fifth hour, as with 20 mg/kg Cu(L-ala)2 group.
When compared between groups, at the first hour, the standard drug-treated group rats showed reduction of paw volume when compared to the compound-treated group of rats at both concentration (2 and 20mg/kg), but there was no significant different among these three groups. All the comparison only taken at the first hour due to the maximal edema induction was on the first hour. Following carrageenan injection into the hind paw, the percentage inhibition of edema in rats with carrageenan-induced paw edema was determined by calculating the mean increase in paw volume data, as illustrated in Figure 1. Edema was calculated as a percentage by deducting the control paw’s volume from the treated paw’s. The results acquired in the first hour following the carrageenan injection, when the maximum edema induction takes place, were used to compute the percentage of inhibition. Based on the Figure 2, the compound-treated group treated with 2mg/kg and 20mg/kg Cu(L-ala)2 showed significantly inhibition (p<0.05) which are 63.79% and 62.06% respectively compared with non-treated group of rats at the first hour. In addition, standard drug-treated (Indomethacin) group also showed significantly inhibition (p<0.05) which is 43.96% when comparing with non-treated group at the first hour. Between compound-treated group and standard drug-treated (Indomethacin) also showed significant (p<0.05) inhibition between these two groups. However, compound-treated group at 2mg/kg and 20mg/kg Cu(L-ala)2 did not show any significant different throughout the 5 hours.
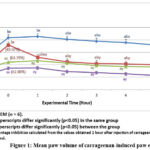 |
Figure 1: Mean paw volume of carrageenan-induced paw edema. |
Number in parenthesis are percentage inhibition calculated from the values obtained 1 hour after injection of carrageenan (maximal edema induction) when compared to negative control.
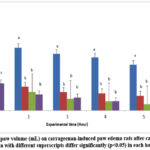 |
Figure 2: Changes of paw volume (mL) on carrageenan-induced paw edema rats after carrageenan injection. |
Effects of Cu (L-ala)2 on Measurement of Nitric Oxide Level
In this inflammation study, measurement of nitric oxide was measured by addition of Griess reagant. Nitric oxide (NO) was produced by the injection of 1% carrageenan (0.1ml/rat) into the rats paw. The paw tissue of the rats was obtained, homogenized and mixed with Griess Reagant to produce colour intensity. Based on result shown in Table 2, treatment group which were 2 mg/kg and 20 mg/kg Cu(L-ala)2 did lower NO level (33.9 µM/g ±4.29 and 29.04 µM/g ±2.71) but it was not significant compared to non-treated group (40.08 µM/g ± 2.5). On the other hand, standard drug-treated group (Indomethacin) showed significantly reduced (P < 0.05) the increased in NO level when compared with the negative control group. There was no significant difference between the 2 and 20 mg/kg of Cu (L-ala)2 in the reduction of NO level. However, NO level were significantly reduced when comparing 2 mg/kg Cu(L-ala)2 with standard drug-treated group (21.53 µM/g ± 2.49).
Between 2 and 20 mg/kg of Cu(L-ala)2, at 20 mg/kg showed the lowest concentration of NO (29.04 µM/g ±2.71) compared to 2 mg/kg (33.91 µM/g ±4.29). Percentage of inhibition on the NO level of paw tissue for each treatment with their respective dosages was shown in Table 2. The maximal percentage of inhibition was produced by Indomethacin which was 46.28% and it was significantly different compared to non-treated group. Meanwhile, between the 2 and 20 mg/kg of Cu(L-ala)2, 20 mg/kg showed the higher percentage of inhibition (27.56%) compared to 2 mg/kg (15.38%). However, both of this compound-treated group did not show significant reduction in NO level.
Table 2: Effect of the Cu (L-ala)2 on carrageenan-induced on the NO level of paw tissue
| Treatment | Dose(mg/kg) | NO level (µM/g) | % inhibition |
| Negative control | – | 40.08±2.5a | – |
| Positive control(Indomethacin) | 10 | 21.53± 2.4b | 46.28 |
| Cu(L-ala)2 compound | 2 | 33.91± 4.29a | 15.38 |
| 20 | 29.04±2.71ab | 27.56 |
Values are mean ± SEM, n=6
a-b: Mean with different superscript differ significantly (p<0.05) between groups
Discussion
Acetic acid–induced peritoneal vascular permeability is a well-established model of acute inflammation characterized by enhanced microvascular leakage and leukocyte migration, reflecting early inflammatory mediator release.14,15 The present findings confirm that acetic acid induces significant vascular permeability, consistent with previous reports describing concentration-dependent irritation and inflammatory exudation.16 Oral administration of Cu(L-ala)₂ at both 2 and 20 mg/kg significantly reduced Evans Blue dye extravasation, indicating suppression of vascular leakage during the acute inflammatory phase. This reduction suggests interference with mediator-driven increases in endothelial permeability, potentially involving prostanoids, kinins, and cytokines released by mast cells and macrophages (TNF-α, IL-1β) following acetic acid challenge.17 The comparable inhibitory effect of Cu(L-ala)₂ to indomethacin supports the possibility that this copper complex may modulate inflammatory signaling pathways linked to cyclooxygenase activity, as NSAIDs reduce vascular permeability primarily through COX inhibition.18,19 However, unlike classical NSAIDs, copper complexes have been reported to exert anti-inflammatory effects through additional redox-related mechanisms, including modulation of oxidative stress and inflammatory cell activation.20,21 This distinction is important, as NSAIDs are associated with gastrointestinal toxicity due to prostaglandin suppression,19 whereas copper-based complexes have been proposed as alternatives with potentially different safety profiles, though this remains controversial and requires further validation.
Carrageenan-induced paw edema is a biphasic inflammatory model22 involving early mediator release (histamine, serotonin, bradykinin) and a later phase dominated by prostaglandins, NO, cytokines, and neutrophil infiltration.12,23 The present study demonstrates that Cu(L-ala)₂ significantly reduced paw edema across the observation period, confirming its in vivo anti-inflammatory activity. Notably, the lower dose (2 mg/kg) exhibited stronger or earlier inhibition of edema than the higher dose (20 mg/kg) at certain time points. This apparent non-linear dose–response warrants careful interpretation. Biologically, such an effect may reflect hormesis, saturation of active binding sites, or differential redox behavior of copper complexes at higher concentrations, where excess copper may exert competing pro-oxidant effects. Similar non-monotonic dose–responses have been reported for metal-based anti-inflammatory compounds.24 Statistically, the small group size (n = 6) may also limit sensitivity in detecting subtle dose-dependent differences, emphasizing the need for larger cohorts and pharmacokinetic evaluation.
Compared with indomethacin, Cu(L-ala)₂ demonstrated a more sustained anti-edematous effect at later time points, aligning with findings by Agotegaray et al.24 that copper complexes maintain anti-inflammatory activity beyond the peak inflammatory phase. This supports the hypothesis that Cu(L-ala)₂ may act not only via COX-related pathways but also through suppression of oxidative and neutrophil-mediated mechanisms. Copper’s reported superoxide dismutase (SOD)-mimetic activity25 provides a plausible explanation, as scavenging of superoxide radicals may indirectly limit prostaglandin synthesis and leukocyte-driven edema progression. Nevertheless, the precise molecular targets remain unresolved, and the assumption of direct COX inhibition by Cu(L-ala)₂ remains speculative, despite similarities to NSAID responses.8,25
Nitric oxide is widely recognized as a mediator of inflammation, contributing to vasodilation, vascular permeability, and leukocyte recruitment during carrageenan-induced inflammation.26,27 Based on the established involvement of the L-arginine–NO pathway,10 it was hypothesized that Cu(L-ala)₂ would attenuate NO production. Contrary to this expectation, Cu(L-ala)₂ produced only modest and statistically insignificant reductions in NO levels, particularly at the lower dose, while indomethacin significantly suppressed NO production. This discrepancy highlights a key mechanistic limitation of the present findings: the anti-inflammatory effects of Cu(L-ala)₂ observed in vascular permeability and edema models do not appear to be primarily mediated through inhibition of NO synthesis. This finding is not entirely unexpected, as NO measurement in vivo is technically challenging due to its short half-life and rapid conversion into reactive nitrogen species.28 Moreover, previous studies have reported that copper complexes may exert anti-inflammatory effects independently of direct iNOS suppression, instead modulating oxidative balance or downstream signaling pathways.26 Thus, the minimal effect on NO observed here suggests that Cu(L-ala)₂ may act via NO-independent mechanisms, which contrasts with earlier mechanistic assumptions and warrants further investigation.29
Several limitations should be acknowledged. First, the study employed a single acute inflammation model for each assay, limiting generalizability to chronic or immune-mediated inflammation. Second, the small sample size reduces statistical power, particularly for dose-response comparisons. Third, mechanistic conclusions are constrained by the absence of molecular analyses (e.g., COX-2, iNOS, cytokine expression). Finally, toxicological and pharmacokinetic data were not assessed, precluding conclusions regarding safety or therapeutic applicability.
Conclusion
This study demonstrates that Cu(L-ala)₂ exerts significant anti-inflammatory effects in vivo by reducing acetic acid-induced vascular permeability and carrageenan-induced paw edema. These findings indicate that the compound effectively attenuates acute inflammatory responses, with efficacy comparable to indomethacin in certain parameters. However, the lack of significant nitric oxide suppression, particularly at lower doses which contradicts the initial mechanistic hypothesis that NO inhibition underlies its anti-inflammatory action.
The observation that the lower dose (2 mg/kg) produced equal or stronger edema inhibition than the higher dose suggests a non-linear dose–response, potentially driven by redox-related or pharmacodynamic factors rather than simple concentration dependence. Importantly, the data indicate that Cu(L-ala)₂ likely mediates its anti-inflammatory effects through NO-independent pathways, possibly involving modulation of oxidative stress or inflammatory mediator release.
While these findings support the potential of Cu(L-ala)₂ as a copper-based anti-inflammatory agent, they also underscore the need for mechanistic clarification, expanded dose–response analysis, and comprehensive safety evaluation. Future studies incorporating molecular targets and chronic inflammation models will be essential to define the therapeutic relevance and limitations of this compound.
Acknowledgment
This research was supported by the Faculty of Medicine and Health Sciences, Universiti Putra Malaysia.
Funding Source
This study was supported by the Faculty of Medicine and Health Sciences, Universiti Putra Malaysia. Final Year Project Funding (FYP 2020/01).
Conflicts of Interest
The author(s) do not have any conflict of interest
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
The animals used in this study were approved under the reference number UPM/FPSK/PADS/BR-UUH/00455 by Animals Care and Use Committee (ACUC), Faculty of Medicine and Health Sciences, UPM.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trials Registration
This research does not involve any clinical trials.
Permission to Reproduce Material from Other Sources
Not Applicable
Author Contributions
- Siti Nur Syahida Abdul Jalil performed the experiments in this study.
- Ng Chin Theng, Fong Lai Yen and Yong Yoke Keong assisted in the experiments and review the study design
- Muhammad Nazrul Hakim and Zuraini Ahmad drafted the manuscript and supervised the study.
References
- Alshadfan H, Qais J, Omar Fauzee MS, Zakaria ZA, Yong YK, et al. Non-steroidal anti-inflammatory drug-induced gastric ulcers: A review on some current issues. Trop J Pharmaceu Res. 2025; 24(6): https://dx.doi.org/10.4314/tjpr.v24i6.11
- Alhatim H, Alshadfan H, Omar Fauzee MS, Zakaria, ZA, Hakim MN. Analyzing NSAIDS research trends: A six-year bibliometric study (2018-2023) on directions, themes, and dimensions. Multidis Rev. 2026; 9(3): 2026130. https://doi.org/10.31893/multirev.2026130
CrossRef - Puranik R, Bao S, Bonin AM, et al. A novel class of copper(II)- and zinc(II)-bound non-steroidal anti-inflammatory drugs that inhibits acute inflammation in vivo. Cell Biosci. 2016; 6(9). https://doi.org/10.1186/s13578-016-0076-8
CrossRef - Malis G, Geromichalou E, Geromichalos GD, Hatzidimitriou AG, Psomas G. Copper(II) complexes with non-steroidal anti-inflammatory drugs: Structural characterization, in vitro and in silico biological profile. J Inorg Biochem. 2021; 224:111563. https://doi.org/10.1016/j.jinorgbio.2021.111563.
CrossRef - Ji P, Wang P, Chen H, Xu Y, Ge J, et al. Potential of Copper and Copper Compounds for Anticancer Applications. Pharmaceuticals. 2023; 16(2):234. https://doi.org/10.3390/ph16020234
CrossRef - Zhong G, Wang X, Li J, Xie Z, et al. Insights Into the Role of Copper in Neurodegenerative Diseases and the Therapeutic Potential of Natural Compounds. Curr Neuropharmacol. 2024; 22(10):1650-1671. https://doi.org/10.2174/1570159X22666231103085859
CrossRef - Zainudin N, Ng CT, Fong LY, Yong YK, Hakim MN, Zuraini A. (2025). Anti-proliferative effects of Cu(phen)(C-dimethylglycine)NO3 on HT-29 and A2780 cancer cell lines: A potential chemotherapeutic approach. Asian J Pharm Clin Res. 2025; 8(6): 76-80. http://dx.doi.org/10.22159/ajpcr.2025v18i6.54576.
CrossRef - Zakaria ZA, Mustapha S, Sulaiman MR, Mat Jais AM, Somchit MN, Abdullah FC. The antinociceptive action of aqueous extract from Muntingia calabura leaves: the role of opioid receptors. Med Princ Pract. 2007;16(2):130-6. https://doi.org/10.1159/000098366.
CrossRef - Somchit MN, Sulaiman MR, Zuraini A, et al. Antinociceptive and antiinflammatory effects of Centella asiatica. Ind J Pharmacol. 2004; 36(6): 377-380.
- Dantas LLSFR, Fonseca AG, Pereira JR, et al. Anti-inflammatory and antinociceptive effects of the isatin derivative (Z)-2-(5-chloro-2-oxoindolin-3-ylidene)-N-phenyl-hydrazinecarbothioamide in mice. Braz J Med Biol Res. 2020; 53(10):e10204. https://doi.org/10.1590/1414-431X202010204.
CrossRef - Jomova K, Raptova R, Alomar SY, et al. Reactive oxygen species, toxicity, oxidative stress, and antioxidants: chronic diseases and aging. Arch Toxicol. 2023; 97(10):2499-2574. https://doi.org/10.1007/s00204-023-03562-9.
CrossRef - Huang MH, Huang SS, Wang BS, et al. Antioxidant and anti-inflammatory properties of Cardiospernum halicacabum and its reference compounds ex vivo and in vivo. J Ethnopharmacol. 2011; 133: 43-750. https://doi.org/10.1016/j.jep.2010.11.005.
CrossRef - Somchit MN, Shukriyah MHN. Anti inflammatory property of ethanol and water extracts of Zingiber zerumbet. Ind J Pharmacol. 2003; 35(3): 181-182. https://doi.org/10.3923/ijp.2005.277.280
CrossRef - Chiong HS, Yong YK, Ahmad Z, Sulaiman MR, Zakaria ZA, Yuen KH, Hakim MN. Cytoprotective and enhanced anti-inflammatory activities of liposomal piroxicam formulation in lipopolysaccharide-stimulated RAW 264.7 macrophages. Int J Nanomedicine. 2013; 8:1245-55. https://doi.org/10.2147/IJN.S42801
CrossRef - Ampadu FA, Boakye-Gyasi E, Osafo N, Benneh CK, Ekuadzi E, et al. Antipleuritic and Vascular Permeability Inhibition of the Ethyl Acetate-Petroleum Ether Stem Bark Extract of Maerua angolensis DC (Capparaceae) in Murine. Int J Inflam. 2018:6123094. https://doi.org/10.1155/2018/6123094.
CrossRef - Yadav SK, Adhikary B, Chand S, Maity B, Bandyopadhyay SK, Chattopadhyay S. Molecular mechanism of indomethacin- induced gastropathy. Free Radical Bio & Med. 2012; 52, 1175-1187. https://doi.org/0.1016/j.freeradbiomed.2011.12.023
CrossRef - Longhi-Balbinot DT, Lanznaster D, Baggio CH, et al.Anti-inflammatory effect of triterpene 3β, 6β, 16β-trihydroxylup-20(29)-ene obtained from Combretum leprosum Mart & Eich in mice. J Ethnopharmacol. 2012; 142(1): 59- 64. https://doi.org/10.1016/j.jep.2012.04.013
CrossRef - Chiong HS, Yong YK, Hijaz MSM, Sulaiman MR, Yuen KH, Hakim MN. Improved Anti-nociceptive, Anti-pyretic and Anti-inflammatory Effects of Orally Administered Liposome-encapsulated Piroxicam. Biomed Pharmacol J. 2024;17(2). https://dx.doi.org/10.13005/bpj/2905
CrossRef - Zakaria ZA, Balan T, Azemi AK, Omar MH, Mohtarrudin N, Ahmad Z, et al. Mechanism(s) of action underlying the gastroprotective effect of ethyl acetate fraction obtained from the crude methanolic leaves extract of Muntingia calabura. BMC Complement Altern Med. 2016;16:78. https://doi.org/10.1186/s12906-016-1041-0
CrossRef - Hakim M, McCarthy EF. Heterotopic mesenteric ossification. AJR Am J Roentgenol. 2001;176(1):260-1. https://doi.org/10.2214/ajr.176.1.1760260.
CrossRef - Zakaria ZA, Somchit MN, Zaiton H, Mat Jais AM, Sulaiman MR, Farah WO, Nazaratulmawarina R, Fatimah CA. The in vitro Antibacterial Activity of Corchorus olitorius Extracts. Int. J. Pharmacol. 2006, 2(2), 213–215. https://doi.org/10.3923/ijp.2006.213.215
CrossRef - Kim KH, Im HW, Karmacharya MB, Kim S, Min BH, Park SR, Choi BH. Low-intensity ultrasound attenuates paw edema formation and decreases vascular permeability induced by carrageenan injection in rats. J Inflamm (Lond). 2020;17:7. https://doi.org/10.1186/s12950-020-0235-x.
CrossRef - Ramakrishnan G, Allan JJ, Goudar K, Amit A. Comparative evaluation of anti-inflammatory activity of different extracts of Boswellia serrata in Wistar Albino rats. Int J PharmTech Res. 2011; 3(1): 261-267. https://doi.org/10.18203/2320-6012.ijrms20161989
CrossRef - Agotegaray MA, Dennehy M, Boeris MA, Grela A, Burrow RA, Quinzani OV. Therapeutic properties, SOD and catecholase mimetic activities of novel ternary copper (II) complexes of the anti-inflammatory drug Fenoprofen with imidazole and caffeine. Polyhedron. 2012; 74-83. https://doi.org/10.1016/j.poly.2011.12.005
CrossRef - An Y, Li S, Huang X, Chen X, Shan H, Zhang M. The Role of Copper Homeostasis in Brain Disease. Int J Mol Sci. 2022; 23(22):13850. https://doi.org/10.3390/ijms232213850
CrossRef - Ng CT, Fong LY, Low YY, Ban J, Hakim MN, Ahmad Z. Nitric Oxide Participates in IFN-γ-Induced HUVECs Hyperpermeability. Physiol. Res. 2016; 65: 1053-1058, https://doi.org/10.33549/physiolres.933237
CrossRef - Wautier JL, Wautier MP. Vascular Permeability in Diseases. International Journal of Molecular Sciences. 2022; 23(7): 3645. https://doi.org/10.3390/ijms23073645
CrossRef - Man MQ, Wakefield JS, Mauro TM, Elias PM. Regulatory Role of Nitric Oxide in Cutaneous Inflammation. Inflammation. 2022, 45(3). https://doi.org/10.1007/s10753-021-01615-8
CrossRef - Alotaibi NO, Abdulhussein HA, Alamri SM, Hamza NA, Abo Nasria AH. Computational insights into the physico-chemical properties of pure and single-atom copper-indium sub-nanometre clusters: a DFT-genetic algorithm approach. RSC Adv. 2025 ;15(8):5856-5875. https://doi.org/10.1039/d4ra07404a.
CrossRef







