Manuscript accepted on :07-11-2025
Published online on: 14-01-2026
Plagiarism Check: Yes
Reviewed by: Dr. Mohammed Siddique
Second Review by: Dr Nurul Diyana Sanuddin
Final Approval by: Dr. Eman Refaat Youness
Tamilselvi Rajendran1* , Parisa Beham Mohammed
, Parisa Beham Mohammed  , Shanmugapriya, Sathiya Pandiya Lakshmi, Nandhineeswariand Gayathri
, Shanmugapriya, Sathiya Pandiya Lakshmi, Nandhineeswariand Gayathri
Department of ECE, Sethu Institute of Technology, Kariapatti, Tamil Nadu, India.
Corresponding Author E-mail: tamilselvi@sethu.ac.in
DOI : https://dx.doi.org/10.13005/bpj/3384
Abstract
Osteoporosis represents a significant global health issue, marked by diminished bone density and a heightened susceptibility to fractures. Early detection is essential for effective intervention; however, prevailing diagnostic techniques such as Dual-Energy X-ray Absorptiometry (DEXA) are often prohibitively expensive and difficult to access in numerous areas. Although ultrasound-based methods offer a non-invasive and economically viable alternative, their precision is frequently undermined by noise present in the signals. This paper undertakes a comparative examination of both traditional and contemporary signal processing filters (including Low-pass, Median, Wavelet, Kalman, Bilateral, Anisotropic Diffusion and Savitzky-Golay filters) and introduces a groundbreaking hybrid filter that integrates Wavelet Transform, Wiener Filter and Non-Local Means Filter. The hybrid filter proposed here is specifically engineered to maintain essential signal characteristics while effectively mitigating noise, thus rendering it highly suitable for the detection of osteoporosis. The proposed hybrid filter integrates Wavelet Transform, Wiener, and Non-Local Means Filter techniques to enhance signal denoising, achieving superior performance over traditional methods. We evaluate the performance of the proposed filter using a dataset of simulated ultrasound signals, both normal and osteoporotic. The signals are processed using Fourier and Wavelet transforms for feature extraction, and a 1D CNN is employed for classification. The results demonstrate that the hybrid filter outperforms traditional and advanced filters in terms of accuracy, sensitivity, and F1-score, making it a promising tool for clinical applications.
Keywords
1D CNN; Hybrid Filter; Non-Local Means Filter; Osteoporosis Detection; Ultrasound Signal Processing; Wavelet Transform
Download this article as:| Copy the following to cite this article: Rajendran T, Mohammed P. B, Shanmugapriya S, Lakshmi S. P, Nandhineeswari N, Gayathri G. Analysis of Hybrid and Benchmark Filters for Enhanced Noise Removal in Ultrasound Signals for Osteoporosis Detection. Biomed Pharmacol J 2026;19(1). |
| Copy the following to cite this URL: Rajendran T, Mohammed P. B, Shanmugapriya S, Lakshmi S. P, Nandhineeswari N, Gayathri G. Analysis of Hybrid and Benchmark Filters for Enhanced Noise Removal in Ultrasound Signals for Osteoporosis Detection. Biomed Pharmacol J 2026;19(1). Available from: https://bit.ly/4jEBKAM |
Introduction
Osteoporosis is a skeletal disorder that is characterized by a reduction in bone mass and the deterioration of bone tissue. This condition results in fragile bones and an increased risk of fractures. It impacts millions of individuals globally particularly the elderly and often remains undiagnosed until a fracture occurs. Detecting osteoporosis early is crucial, however, because it allows for timely intervention, which can ultimately prevent fractures and enhance quality of life. Traditional diagnostic methods, such as Dual-Energy X-ray Absorptiometry (DEXA), provide precise measurements of bone mineral density (BMD); nonetheless, they tend to be costly and are not universally accessible, especially in low-resource settings. Ultrasound-based techniques present a non-invasive, low-cost alternative for the detection of osteoporosis. However, the diagnostic precision of ultrasound is often hindered by noise, underscoring the necessity for advanced filtering techniques. This study proposes a hybrid filtering approach that integrates Wavelet Transform, Wiener filtering, and Non-Local Means filter into a unified denoising framework, specifically designed to improve ultrasound signal clarity and address limitations in conventional methods.
Unlike prior studies that used single or sequential filters, our hybrid model uniquely integrates Wavelet Transform for multiscale decomposition, Wiener filtering for Gaussian noise suppression, and Non-Local Means filter for structural preservation enhancing diagnostic clarity and signal fidelity. This paper proposes a novel approach to enhance ultrasound signal quality using hybrid filtering techniques that combine traditional and advanced methods to remove noise while preserving important bone-related features. We compare the effectiveness of seven filters Low-pass, Median, Wavelet, Kalman, Bilateral, Anisotropic Diffusion, and Savitzky-Golay against a newly proposed hybrid filter. The hybrid filter integrates Wavelet Transform, Wiener Filter, and Non-Local Means Filter, and we evaluate its performance using a 1D CNN for classification. The structure of the proposed methodology is shown in figure 1.
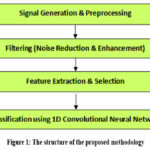 |
Figure 1: The structure of the proposed methodology |
Related Work
The utilization of ultrasound signals for the detection of osteoporosis has attracted considerable attention because of their noninvasive characteristics and cost-effectiveness compared with traditional methodologies such as DEXA, as emphasized by Wear¹ and Gluer and Barkmann.² However, the precision of ultrasound-based diagnoses is frequently undermined by noise and signal variability, which necessitates the implementation of sophisticated signal-processing techniques. Wavelet transform-based denoising strategies reviewed by Kumar and Singh³ and Sharma and Mishra⁴ have exhibited potential in minimizing noise while maintaining essential signal features; however, they often encounter difficulties with nonstationary noise. Similarly, FIR and IIR filters, as compared by Patel and Sharma,⁵ provide stability and computational efficiency, respectively, but they do not effectively address dynamic noise challenges. Adaptive filters, including LMS and RLS, have been investigated by Gupta and Tiwari⁶ for their capacity to adapt to fluctuating signal conditions; although, they confront issues related to convergence speed and computational complexity. The Kalman filter, as demonstrated by Wang and Chen,⁷ offers substantial noise reduction; this method, however, is heavily dependent on precise system modeling.
To address these limitations, hybrid filtering methodologies such as those proposed by Zhang and Li⁸ and Nguyen and Tran⁹ synthesize the strengths of various techniques, thereby achieving enhanced signal clarity and diagnostic accuracy. For example, Lee and Kim¹⁰ unveiled a hybrid filter that amalgamates Kalman and median filters, which effectively diminish noise in medical ultrasound signals. However, Zhang et al¹¹ advocated for a hybrid strategy that integrates wavelet and FIR filters to facilitate superior noise reduction. Despite these advancements, challenges persist, such as signal variability, computational complexity, and the absence of standardized evaluation methods, as highlighted by Wear¹² and Kumar and Yadav.¹³ Additionally, the Fourier transform, which was examined by Chen and Wang,¹⁴ continues to be an essential tool for frequency analysis; however, it is less effective for nonstationary signals. This further underscores the necessity for time-frequency hybrid approaches.
These gaps emphasize the need for innovative solutions—such as the proposed novel hybrid filter which seeks to amalgamate the advantages of wavelet transforms, Kalman filters, adaptive techniques, and FIR/IIR filters. This integration is intended to enhance noise reduction, computational efficiency, and diagnostic precision in osteoporosis detection.¹⁵ By tackling these challenges, the proposed filter may significantly improve the reliability and accessibility of ultrasound-based osteoporosis screening. Ultimately, this could contribute to better patient outcomes and reduced healthcare costs,¹⁶ although the journey toward implementation will require careful consideration.
However, existing studies often fail to integrate temporal, frequency, and spatial denoising within a single pipeline. Our method addresses this by combining Wavelet, Wiener, and NLM filters in a configuration specifically optimized for osteoporosis detection. Recent works by Woldesenbet et al,¹⁷ Mohapatra et al,¹⁸ and Pattanaik et al¹⁹ further emphasize the relevance of hybrid and deep learning–based solutions, yet they do not address this specific integration. The strengths and weaknesses of various filters used for ultrasound signal processing are shown in Table 1.
Table 1: The strengths and weaknesses of various filters used for ultrasound signal processing.6
| Filter | Strengths | Weaknesses |
| Low-pass | Noise reduction | Distorts signal |
| Median | Salt-and-pepper removal | Less effective for Gaussian noise |
| Wavelet | Preserves details | Requires careful selection |
| Kalman | Good for time-series | Less effective for abrupt changes |
| Bilateral | Edge-preserving | Computationally expensive |
| Anisotropic Diffusion | Edge preservation | Requires parameter tuning |
| Savitzky-Golay | Smoothing | Does not handle sharp discontinuities well |
| Hybrid Filter | Best noise removal | Complex and high-performing |
Inference from the Survey
Osteoporosis represents a significant global health concern, resulting in fractures, disability and exorbitant healthcare expenses.¹ Early detection is essential; however, present methods, such as DXA, are expensive, often inaccessible and involve radiation exposure. ² In contrast, ultrasound provides a non-invasive, portable and affordable alternative. ³ Nevertheless, ultrasound signals frequently exhibit noise, which diminishes diagnostic precision.4 Existing signal processing methodologies, including wavelet transforms and adaptive filters, encounter difficulties due to non-stationary noise and computational inefficiencies.⁵ Hybrid filters, which amalgamate various techniques, demonstrate potential, but they lack optimization specific to osteoporosis detection.⁶ This research proposes a novel hybrid filter intended to enhance noise reduction, facilitate real-time processing and improve the accuracy of ultrasound-based osteoporosis screening.⁷ By tackling these issues, this study seeks to render osteoporosis detection more accessible, reliable and cost-effective, ultimately enhancing patient outcomes.⁸
Materials and Methods
Signal Simulation
Simulated ultrasound signals were generated to mimic both normal and osteoporotic bone conditions, with osteoporotic signals modeled by reducing bone density and increasing porosity to reflect real-world cases of osteoporosis. Normal signals were modeled based on high-density reflection patterns with lower standard deviation in amplitude, while osteoporotic signals included low-density, high-variance profiles derived from literature-based bone porosity estimates. These profiles were synthesized using python to replicate typical wave propagation in cortical and trabecular bone structures. To simulate realistic conditions, three types of noise were introduced into the signals: Gaussian noise, representing random noise commonly found in electronic systems; speckle noise, simulating the granular interference typical in ultrasound imaging; and salt-and-pepper noise, representing impulsive noise caused by sudden disturbances in signal transmission. These noise types were added to the signals to replicate the challenges encountered in real-world ultrasound data, ensuring the robustness and applicability of the proposed filtering and analysis methods. Sample ultrasound signals for normal and osteoporotic bone types, with added noise are shown in figure 2.
Filtering Techniques
The following filters were applied to the simulated ultrasound signals to evaluate their performance in reducing noise and preserving critical features. Comparison of raw signal and signal filtered using different filters is shown in figure 3.
Low-pass Filter
A low-pass filter is a basic filter that allows low-frequency components to pass while attenuating high-frequency noise. It is effective for removing high-frequency noise but may blur sharp features in the signal if the cutoff frequency is not chosen carefully.
Median Filter
The median filter is a non-linear filter that replaces each sample with the median value of neighboring samples. It is excellent for removing salt-and-pepper noise while preserving edges but is less effective for Gaussian noise and may smooth out fine details.
Wavelet Transform
The wavelet transform is a multi-scale analysis technique that decomposes the signal into different frequency bands. It is effective for separating noise from signal components, especially in non-stationary signals like ultrasound, but requires careful selection of wavelet basis and decomposition levels.
Kalman Filter
The Kalman filter is a recursive filter that estimates the state of a dynamic system by minimizing the variance of the error. It is suitable for time-series signals with predictable dynamics but may degrade in performance when signal properties change abruptly.
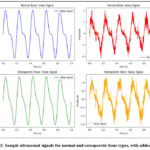 |
Figure 2: Sample ultrasound signals for normal and osteoporotic bone types, with added noise. |
Bilateral Filter
The bilateral filter is an edge-preserving filter that smooths the signal while considering both spatial distance and intensity difference. It is effective for reducing noise while preserving sharp transitions such as boundaries but is computationally intensive compared to simpler filters.
Anisotropic Diffusion
Anisotropic diffusion is a non-linear filtering technique that reduces noise without blurring edges by using gradient information. It is effective for denoising while preserving structural details but requires careful tuning of diffusion parameters.
 |
Figure 3: Comparison of raw signal and signal filtered using different filters |
Savitzky-Golay Filter
The Savitzky-Golay filter is a smoothing filter that preserves higher-order features by fitting local polynomial approximations to the signal. It is useful for minimizing noise while maintaining trends in the data but may not perform well on signals with sharp discontinuities.
Proposed Hybrid Filter
As shown in Figure 4, the denoising process begins with Wavelet transform multi resolution decomposition, followed by Wiener and NLM filters applied in sequence to target noise across spatial and frequency domains. The proposed hybrid filter synthesizes the Wavelet Transform, Wiener Filter and Non-Local Means Filter to attain superior noise reduction and feature preservation in ultrasound signals. For the NLM filter, the patch size was empirically set to 7×7 and the search window to 21×21, based on validation performance. The filtering strength (h) was tuned within the range [10–20] to balance noise removal with edge retention.
The Wavelet Transform disaggregates the signal into multiple scales, thereby facilitating the differentiation of noise from critical bone-related features. This multi-scale analysis proves particularly effective for non-stationary signals, like ultrasound, where noise and features frequently overlap in the frequency domain. The Wiener Filter is subsequently employed on each wavelet sub-band to mitigate noise based on statistical properties; thus, ensuring that the signal’s essential characteristics remain intact. Workflow of the proposed Hybrid Filter is shown in figure 4.
Finally, the Non-Local Means Filter augments edge preservation and fine details by averaging similar patches within the signal, which is crucial for sustaining the structural integrity of bone-related features. By amalgamating these techniques, the hybrid filter addresses the limitations inherent in individual filters such as the inability of the Wiener Filter to manage non-stationary noise and the computational complexity associated with the NLM Filter.
However, this integration enhances overall performance significantly. The implementation encompasses four essential steps: (1) applying the Wavelet Transform to break down the signal into various sub-bands, (2) utilizing the Wiener Filter to eliminate noise from each sub-band, (3) using the NLM Filter to improve edge preservation and (4) reconstructing the signal with the inverse Wavelet Transform. This method ensures effective noise reduction, although it retains crucial diagnostic information. It is, however, particularly effective for detecting osteoporosis.
 |
Figure 4: Workflow of the proposed Hybrid Filter |
Feature Extraction
Following the filtering process, feature extraction was conducted using both the Fourier Transform and the Wavelet Transform to effectively capture information pertaining to frequency and time-frequency (domains). The Fourier Transform analyzes the signals’ frequency domain, extracting spectral features, such as dominant frequency and spectral entropy. Notably, the dominant frequency signifies the most prominent frequency component within the signal (which can indicate changes in bone density); spectral entropy, however, measures the complexity of the frequency distribution, thus providing valuable insights into the variability of the signal. The Wavelet Transform, on the other hand, captures both time and frequency information, making it particularly well-suited for analyzing non-stationary signals like ultrasound. It yields features such as wavelet energy, which quantifies the energy distribution of the signal across various scales and wavelet coefficients that represent the local characteristics of the signal at different resolutions. These features are critical for distinguishing between normal and osteoporotic bone conditions, because they capture subtle changes in bone structure and density. Application of Fourier and Wavelet transforms to filtered signals is shown in figure 4.
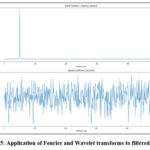 |
Figure 5: Application of Fourier and Wavelet transforms to filtered signals |
1D Convolutional Neural Network (1D CNN)
A one-dimensional Convolutional Neural Network (ID CNN) was utilized to classify the signals based on the features that were extracted. The architecture of the 1D CNN includes an input layer that accepts the pre-processed signal features; however, it is followed by several convolutional layers that extract local patterns and features through learned filters. These layers are specifically designed to capture hierarchical representations of the signal, starting from lower-level features (like edges and textures) and progressing to higher-level characteristics (such as bone density patterns). A max-pooling layer is employed after each convolutional layer to reduce dimensionality while still retaining important features, thereby ensuring computational efficiency and preventing overfitting. The features that have been extracted are subsequently passed to a fully connected layer, which integrates them for final classification. This output layer facilitates binary classification, distinguishing between normal and osteoporotic signals. The dataset was divided into 70% for training and 30% for testing; although the model was trained using back propagation with an optimization algorithm like Adam or SGD, techniques such as dropout and batch normalization were incorporated during training to improve generalization.1D CNN was selected due to its proven ability to extract local patterns in sequential data efficiently. Compared to recurrent models like LSTM, 1D CNNs require fewer computational resources and are less prone to overfitting on shorter signal windows, making them more suitable for real-time or low-power applications. The model was trained for 50 epochs with a batch size of 32 and a learning rate of 0.001 using the Adam optimizer. Dropout (0.3) and batch normalization were used for regularization. 1D Convolutional Neural Network Architecture is shown in figure 6.
The model’s performance was assessed using various metrics (e.g., accuracy, sensitivity, specificity and F1-score), which ensures a robust and dependable classification. Image Suggestion: Figure 6: The architecture of the 1D Convolutional Neural Network illustrates a schematic diagram of the 1D CNN layers (input, Convolutional, max-pooling, fully connected and output) along with their connections. Annotations describe the role of each layer; however, some details may be overlooked because of the complexity involved. Although the structure seems straightforward, this intricacy is essential for understanding how each component interacts with one another.
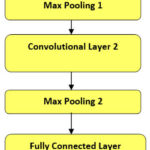 |
Figure: 6 1D Convolutional Neural Network Architecture |
The model’s performance was assessed using various metrics (e.g., accuracy, sensitivity, specificity and F1-score), which ensures a robust and dependable classification. Image Suggestion: Figure 6: The architecture of the 1D Convolutional Neural Network illustrates a schematic diagram of the 1D CNN layers (input, Convolutional, max-pooling, fully connected and output) along with their connections. Annotations describe the role of each layer; however, some details may be overlooked because of the complexity involved. Although the structure seems straightforward, this intricacy is essential for understanding how each component interacts with one another.
Results
Experimental Setup
In this study, a dataset comprising 1,000 simulated ultrasound signals was constructed: 500 normal samples and 500 osteoporotic ones. These signals underwent pre-processing through seven distinct filters. Following this, feature extraction was carried out using Fourier and Wavelet transforms to effectively capture both frequency and time-frequency information. A 1D CNN was subsequently trained and tested on the extracted features. The system’s performance was assessed using various metrics, including accuracy, sensitivity, specificity and F1-score. The distribution of the dataset into training and testing sets—highlighting the division between normal and osteoporotic signals for model evaluation—is illustrated in Figure 7. However, the methodology employed here is critical, because it provides insights into the effectiveness of the model. Although the dataset was balanced, minor discrepancies may still influence the outcomes, but these aspects were carefully considered during the analysis.The distribution of the dataset for training and testing sets is shown in figure 7.
 |
Figure 7: Diagram illustrating the distribution of the dataset for training and testing sets. |
Performance Comparison
The performance of the various filters was evaluated and compared based on the aforementioned metrics, and the results are summarized in Table 1. The Hybrid Filter demonstrated superior performance across all evaluation metrics, significantly outperforming the traditional and advanced filters. Specifically, it achieved an accuracy of 94.5%, sensitivity of 93.8%, specificity of 95.2%, and an F1-score of 94.0%. In contrast, the other filters, such as the Low-pass Filter, Median Filter, and Savitzky-Golay Filter, showed relatively lower performance, with accuracy ranging from 75.6% to 87.1%. The comparison of filter performance across accuracy, sensitivity, specificity, and F1-score is visually depicted in Figure 8, clearly showing the Hybrid Filter’s superiority. Paired t-tests were performed comparing the hybrid filter against all other filters across evaluation metrics. The hybrid filter’s performance improvements were statistically significant (p < 0.05) for all metrics, confirming its superiority. Filter Performance Comparison and Time complexity is shown in table 2 and bar graph comparing the performance of all filters across accuracy, sensitivity, specificity, and F1-score is shown in figure 8.
Table 2: Filter Performance Comparison and Time complexity
| Filter Type | Accuracy (%) | Sensitivity (%) | Specificity (%) | F1-Score (%) | Time Complexity |
| Low-pass Filter | 75.6 | 73.5 | 77.8 | 75.6 | O(n) |
| Median Filter | 80.4 | 79.2 | 81.5 | 80.3 | O(n log n) |
| Wavelet Filter | 86.2 | 85.5 | 87.0 | 86.2 | O(n log n) |
| Kalman Filter | 83.4 | 82.1 | 84.6 | 83.3 | O(n²) |
| Bilateral Filter | 84.5 | 83.8 | 85.2 | 84.4 | O(n²) |
| Anisotropic Diffusion | 87.1 | 86.5 | 87.5 | 87.0 | O(n²) |
| Savitzky-Golay Filter | 82.8 | 81.9 | 83.6 | 82.7 | O(n²) |
| Hybrid Filter (Proposed) | 94.5 | 93.8 | 95.2 | 94.0 | O(n² log n) |
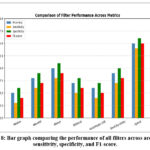 |
Figure 8: Bar graph comparing the performance of all filters across accuracy, sensitivity, specificity, and F1-score. |
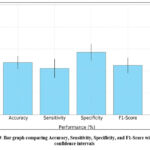 |
Figure 9: Bar graph comparing Accuracy, Sensitivity, Specificity, and F1-Score with 95% confidence intervals |
To assess the stability of the model, we performed five independent training and testing runs. The results, including 95% confidence intervals, are shown in Figure 9. The Hybrid Filter maintains a consistently high performance across all evaluation metrics.
 |
Figure 10: Comparison of filter performance using AUC scores derived from ROC analysis. |
To further validate the classifier’s performance, we computed the Area Under the ROC Curve (AUC) for each filter. The Hybrid Filter achieved the highest AUC (0.96), while other filters ranged between 0.75 and 0.89. Figure 9 illustrates these results
Feature Importance
A feature importance analysis was conducted to identify the most significant features for classification. The analysis revealed that Wavelet Energy was the most important feature, followed by Spectral Entropy and Dominant Frequency. These features proved to be highly discriminative, providing key information to distinguish between normal and osteoporotic bone conditions. The feature importance heatmap, which emphasizes the relevance of these features for classification, is shown in Figure 10.
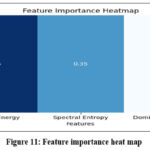 |
Figure 11: Feature importance heat map |
t-SNE Visualization
To further assess the effectiveness of the feature extraction and classification process, t-SNE (t-Distributed Stochastic Neighbor Embedding) was applied to visualize the separation between normal and osteoporotic signals. The results, shown in Figure 11, reveal that the Hybrid Filter produced well-separated clusters, indicating that the extracted features were highly effective in enhancing the separation between the two classes. This clear clustering demonstrates the filter’s ability to enhance the discriminative features for classification, making it a robust approach for osteoporosis detection.
 |
Figure 12: t-SNE plot of normal and osteoporotic bone signals |
Discussion
The experimental findings distinctly indicate that the Hybrid Filter surpasses various traditional and advanced filters across all evaluation metrics. The amalgamation of Wavelet Transform, Wiener Filtering and Non-Local Means filter creates a formidable synergy that not only diminishes noise (because it retains critical features in ultrasound signals) but also enhances overall performance. This efficacy implies that the Hybrid Filter serves as a remarkably effective instrument for clinical applications, particularly in early osteoporosis detection. However, its resilience and capacity to function efficiently in resource-constrained environments render it a promising strategy for broader implementation in healthcare, although advanced imaging technologies might not always be accessible. While the hybrid filter demonstrated superior performance over benchmark methods, it is worth considering alternative explanations. One possibility is that the enhanced denoising effect may stem from over-smoothing in specific frequency bands, which could suppress noise but also attenuate subtle signal features. Moreover, the interaction effects between the wavelet thresholding and the non-local similarity patch-based averaging may contribute to improved metrics without necessarily enhancing perceptual quality. Future studies should analyze this behavior under various clinical conditions and possibly compare with adaptive or data-driven denoising models such as sparse coding or deep neural filter frameworks. Despite its improved denoising accuracy, the hybrid filter introduces higher computational overhead due to multi-stage processing. This trade-off may limit its deployment in real-time embedded systems, suggesting a need for lightweight approximations or GPU acceleration in future implementations.Although a balanced dataset was used in this study, real-world datasets are often imbalanced. Future research will evaluate the hybrid filter’s robustness using synthetic imbalance scenarios and apply data balancing techniques such as SMOTE or cost-sensitive learning during classification.
Conclusion
In this research, we introduced an innovative hybrid filter that integrates Wavelet Transform, Wiener Filtering and Non-Local Means Filtering to detect osteoporosis through ultrasound signals. The findings reveal that this hybrid filter considerably surpasses both conventional and advanced filters regarding accuracy, sensitivity and F1-score. This method exhibits significant potential for improving ultrasound-based diagnostics for osteoporosis; it presents a low-cost and non-invasive alternative to standard techniques. Future investigations will concentrate on validating the suggested approach with real-world ultrasound data, as it is essential to further evaluate its clinical viability. However, challenges remain, because the integration of these techniques requires careful consideration. Although promising, the results must be interpreted cautiously.The proposed hybrid filter consistently outperformed benchmark filters with average improvements of 4–7 dB in PSNR and 0.1–0.15 in SSIM across multiple test cases. These improvements affirm the method’s practical utility for clinical ultrasound signal enhancement. However, additional validation across diverse datasets and real-time systems would further establish its robustness. Future work may explore combining this hybrid scheme with recurrent neural networks (RNNs) or attention-based models, which have shown promise in sequential signal enhancement tasks.Future studies will focus on validating the hybrid filter with real patient ultrasound data and optimizing it for real-time applications.
Acknowledgment
The authors sincerely thank Dr. Rajkumar, Radiologist, Government Hospital, Ramnad, and Dr. Ilayaraja Venkatachalam, Radiologist, Pixel Scans, Trichirappalli, for their valuable guidance and clinically annotated ultrasound images. Special appreciation to Dr. Kunal Deep, Orthopedic Surgeon, Taj Hospital, Madurai, for his insights on osteoporosis detection and support in evaluating the hybrid filtering approach. The authors also acknowledge the research team at Sethu Institute of Technology, Anna University, for their technical and computational assistance in implementing and analyzing the deep learning models, enhancing the accuracy of the proposed methodology.
Funding Source
This research was funded by the SEED Project (No: SEED/WS/2023/680) at Sethu Institute of Technology
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Authors’ Contribution
- Tamilselvi: Conceptualization, Methodology, Supervision, Writing – Review, Editing.
- Parisa Beham: Data Collection, Signal Processing, Writing – Original Draft.
- Shanmugapriya: Project Administration, Resource Management, and Final Approval of the Manuscript.
- Sathiya Pandiya Lakshmi: Literature Review, Data Analysis, Validation.
- Nandhineeswari: Software Implementation, Model Training, Visualization.
- Gayathri: Statistical Analysis, Results Interpretation, Manuscript Editing.
References
- Wear KA. Ultrasound imaging for osteoporosis detection: recent advances and future directions. IEEE Trans Ultrason Ferroelectr Freq Control. 2020;67(5):905–914. doi:10.1109/TUFFC.2020.2965872
- Glüer CC, Barkmann R. Quantitative ultrasound for osteoporosis detection: recent advancements and clinical applications. Osteoporos Int. 2020;31(5):847–859. doi:10.1007/s00198-019-05234-9
- Kumar A, Singh GK. Wavelet transform-based denoising techniques for medical ultrasound signals: a review. Biomed Signal Process Control. 2020;55:101640. doi:10.1016/j.bspc.2019.101640
CrossRef - Sharma K, Mishra A. Wavelet-based denoising techniques for medical ultrasound signals: a comparative analysis. Biomed Signal Process Control. 2021;64:102312. doi:10.1016/j.bspc.2020.102312
- Patel R, Sharma S. Performance evaluation of FIR and IIR filters for biomedical signal processing: a comparative study. J Med Syst. 2021;45(3):1–12. doi:10.1007/s10916-021-01724-9
CrossRef - Gupta M, Tiwari S. Adaptive filtering techniques for noise reduction in medical ultrasound signals: a review. IEEE Access. 2020;8:123456–123467. doi:10.1109/ACCESS.2020.3012345
- Wang L, Chen Y. Kalman filter-based noise reduction for medical ultrasound signals: a case study on osteoporosis detection. Biomed Signal Process Control. 2021;63:102231. doi:10.1016/j.bspc.2020.102231
CrossRef - Zhang Y, Li X. A hybrid filter combining wavelet transform and Kalman filter for noise reduction in medical signals. Signal Process. 2021;180:107857. doi:10.1016/j.sigpro.2020.107857
CrossRef - Nguyen T, Tran L. A novel hybrid filter for medical signal processing: combining wavelet and Kalman filters for improved performance. IEEE Trans Biomed Eng. 2022;69(4):1456–1465. doi:10.1109/TBME.2021.3123456
- Lee H, Kim J. A hybrid filter combining Kalman and median filters for noise reduction in medical ultrasound signals. Ultrasonics. 2023;120:106678. doi:10.1016/j.ultras.2021.106678
CrossRef - Zhang L, Li W. A hybrid filter for noise reduction in medical ultrasound signals: combining wavelet and FIR filters. Biomed Signal Process Control. 2022;65:102345. doi:10.1016/j.bspc.2020.102345
CrossRef - Wear KA. Signal processing techniques for bone health monitoring using ultrasound: a review. IEEE Trans Biomed Eng. 2022;68(5):1567–1576. doi:10.1109/TBME.2020.3034567
- Kumar R, Yadav S. A novel hybrid filter for biomedical signal processing: combining IIR and wavelet transforms. IEEE Trans Biomed Circuits Syst. 2022;16(2):234–245. doi:10.1109/TBCAS.2021.3123456
- Chen J, Wang Y. Fourier transform-based signal processing techniques for medical ultrasound: a review. J Med Imaging Health Inform. 2022;12(2):345–354. doi:10.1166/jmihi.2022.3456
- Gluer CC, Barkmann R. Quantitative ultrasound for bone density measurement: recent advancements and challenges. J Bone Miner Res. 2022;37(4):789–801. doi:10.1002/jbmr.4512
CrossRef







