Manuscript accepted on :09-10-2025
Published online on: 27-10-2025
Plagiarism Check: Yes
Reviewed by: Dr. Omobolanle Omoteso
Second Review by: Dr. Anjali Patadiya
Final Approval by: Dr. Kamal Upreti
Ranajit Nath and Rajesh Ephraim Jesudasan*
and Rajesh Ephraim Jesudasan*
School of Pharmacy, The Assam Kaziranga University, Jorhat, India.
Corresponding Author E-mail: rajeshjesudasan4@gmail.com
Abstract
Neurological disorders (NDs) remain a major global health burden, and conventional therapies often fail to achieve satisfactory outcomes due to limited brain bioavailability, systemic toxicity, and poor patient compliance. Nanotechnology offers novel opportunities to overcome these challenges, particularly by enhancing drug stability, bioavailability, and targeted delivery across the blood–brain barrier (BBB). A wide range of nanosystems—including lipid-based carriers, polymeric nanoparticles, metallic nanoparticles, and nanomicelles—have been explored for their ability to improve the therapeutic potential of both synthetic and plant-derived compounds. Increasing evidence demonstrates that nanoformulations of phytoconstituents such as curcumin, quercetin, Ginkgo biloba, and Nigella sativa exhibit neuroprotective effects with superior efficacy compared to their free forms. This review critically evaluates the current state of nano-phytomedicine in the management of NDs, highlighting key strengths, limitations, and translational challenges. Special emphasis is given to regulatory hurdles, safety concerns, and the gaps that must be addressed before clinical application. A structured literature search strategy was employed to identify recent advances and ongoing debates in this field. Finally, we propose novel perspectives on how rational design of phytochemical-based nanocarriers, in combination with advanced imaging and molecular tools, could accelerate the development of safe and effective neurotherapeutics.
Keywords
Curcumin; Herbal extracts; Neurodegenerative diseases; Nanoparticles; Nanotechnology
| Copy the following to cite this article: Nath R, Jesudasan R. E. Nanoparticles of Herbal Extracts in Treatment of Neurodegenerative Disorders. Biomed Pharmacol J 2025;18(October Spl Edition). |
| Copy the following to cite this URL: Nath R, Jesudasan R. E. Nanoparticles of Herbal Extracts in Treatment of Neurodegenerative Disorders. Biomed Pharmacol J 2025;18(October Spl Edition). Available from: https://bit.ly/3WWXyNz |
Introduction
Nanotechnology has emerged as a transformative force across multiple fields of science and technology, offering new perspectives for therapeutic interventions.¹ Among its promising applications is the integration of nanotechnology with herbal medicine, where bioactive plant extracts and dietary compounds are reformulated into advanced nanosystems such as polymeric nanoparticles, nanocapsules, nanoemulsions, transferosomes, and ethosomes.² These nanoscale formulations provide significant advantages over conventional herbal preparations, including enhanced solubility, bioavailability, stability, sustained release, targeted tissue delivery, protection from degradation, and improved pharmacological activity.¹˒² In addition, nanotechnology enables the co-delivery of multiple therapeutic agents and the use of imaging tools to optimize the distribution of poorly water-soluble phytoconstituents across biological barriers.³
Neurological sciences, in particular, are expected to benefit substantially from these innovations.⁴ Nanoparticles, with dimensions comparable to proteins, offer unique opportunities to interact with biological systems at the molecular level, allowing precise targeting of cells and tissues while minimizing systemic side effects.⁵ Conventional pharmacological approaches are often limited by the blood–brain barrier (BBB), a formidable obstacle that restricts the delivery of many therapeutic compounds into the central nervous system (CNS). Nanotechnology-based carriers, however, hold potential to overcome this challenge, making them a valuable strategy in addressing neurodegenerative diseases (NDs).
NDs represent a heterogeneous group of chronic, progressive disorders that involve the gradual loss of neuronal structure or function.⁶ They impair cognition, movement, memory, and reasoning, and include conditions such as Alzheimer’s disease (AD), Parkinson’s disease (PD), Huntington’s disease (HD), amyotrophic lateral sclerosis (ALS), and multiple sclerosis (MS).⁷ AD alone contributes to nearly 60% of dementia cases worldwide, affecting approximately 24 million individuals, and is characterized by neuronal loss, hippocampal atrophy, extracellular plaques, and neurofibrillary tangles.⁸˒⁹ Despite extensive knowledge of AD pathology, its etiology remains unclear, and current therapeutic strategies only manage symptoms rather than alter disease progression.¹⁰ Similarly, PD, a prevalent neurodegenerative disorder, arises from progressive dopaminergic neuronal loss in the substantia nigra, leading to hallmark symptoms such as tremors, rigidity, and bradykinesia.¹¹˒¹² Distinguishing PD from other parkinsonian syndromes remains challenging, as overlapping symptoms complicate diagnosis.¹³˒¹⁴ Current therapies such as levodopa alleviate symptoms but fail to halt disease progression, while alternative strategies like neuronal transplantation and deep brain stimulation remain experimental.¹⁴˒¹⁵
Given the rising global life expectancy, the prevalence and societal burden of NDs are projected to increase dramatically.¹⁷ Dementia and age-related neurodegenerative conditions now rank among the leading causes of mortality, with the World Health Organization reporting a sharp rise in dementia-related deaths between 2012 and 2021.¹⁸˒¹⁹ This escalating health challenge underscores the urgent need for innovative, effective, and safe therapeutic approaches. Current pharmacological agents for NDs are associated with limited efficacy and adverse effects, reinforcing the importance of alternative strategies.²⁰
Natural products are increasingly recognized as promising candidates for neuroprotection due to their pleiotropic mechanisms of action. However, their poor solubility, instability, and inability to cross the BBB significantly restrict their therapeutic potential.²¹ Nanotechnology-based drug delivery systems provide a solution by enhancing the transport, stability, and bioavailability of phytochemicals.²² Through nanoscale engineering, herbal bioactives can be reformulated into stable, targeted delivery systems that improve CNS penetration and therapeutic efficacy. This review therefore examines recent advances in nanoformulations of herbal medicines for the treatment of NDs, highlighting their potential to address current therapeutic gaps while also discussing challenges in translation, regulation, and clinical application.²³,²⁵
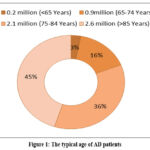 |
Figure 1: The typical age of AD patients. |
Biology of the BBB
Because it is so sensitive to contaminants in the blood, the brain needs a unique microenvironment to perform at its best.26 Tight connections between the brain capillary endothelial cells that make up the BBB prohibit chemicals from moving between the cells as they form a selective barrier. Additionally, the endothelial cells’ P-glycoprotein (P-gp) pumps aggressively efflux cytotoxic chemicals unidirectionally through the apical membrane then into the luminal region, getting rid of foreign substances that bypass the BBB.27 Microglia, pericytes, and astrocytes that surround the endothelial tube further strengthen the barrier. While tiny, lipophilic substances and gases can diffuse across the BBB down a concentration gradient, large and aqueous components require the utilization of transporters. The BBB has three different transport mechanisms: adsorptive-mediated transcytosis (AMT), receptor-mediated transcytosis (RMT), and carrier-mediated transport (CMT) (Fig. 2).28 Protein carriers are mostly used by CMT to transport relatively small particles and nutrients including ascorbic acid, amino acids, and glucose. In contrast, RMT and AMT, endocytosis and transport bigger proteins and molecules over the BBB using vesicles. AMT is less selective because it depends on interactions between cationic compounds as well as the opposite charges sulfated proteoglycans on the endothelium plasma membrane, whereas RMT is extremely selective since it needs to recognize the receptor and its ligand.29 Due to RMT’s specificity and AMT’s flexibility, which respectively allow for preferred medication localization to the brain plus independence from membrane receptors, have been combined to provide nanoparticle delivery. Among the most successful options to treat neurodegenerative illness is the noninvasive delivery of nano-medicine that can cross the BBB.30
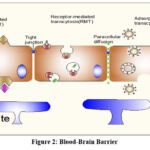 |
Figure 2: Blood-Brain Barrier. |
Drug Clearance
When exposed to the circulatory system, many medications, including nano-medicine, swiftly disintegrate. The reticuloendothelial system, also known as the mononuclear phagocyte system (MPS) (RES), is made up of immune cells that identify and eliminate medicines within a few hours of delivery.30 The main players in the MPS are macrophages, which remove nanoparticles from the liver or spleen when the blood passes past them. Drugs cannot elude clearance simply by being encapsulated in nanoparticles; however various surface changes applied to nanoparticles are very successful at extending stability and circulation time. Nearly every type of nanotechnology listed below can benefit from these surface alterations. Polyethylene glycol (PEG), the most effective modification, increases many nanoparticles’ endurance and biological effectiveness. PEG has a large excluded volume, or the volume created by the physical existence of PEG together with steric hindrance, which prevents other molecules from passing through it. It also has infinite large conformational entropy, solubility in water, and conformational entropy. In doing so, the core of the nanoparticle is effectively shielded, which slows down disintegration and lengthens circulation time. These characteristics have given rise to several suggestions about how PEG aids nanoparticles avoid macrophage engulfment, together with a steric obstruction, a decrease the nanoparticle’s ability to bind to certain proteins and participate in protein adsorption are two examples of this (Fig. 3). Although mice develop an immune response to repeated doses of PEG, certain studies have shown that PEGylated nanoparticle circulation times are worsened as a result. This is true even though PEG is tremendously effective in practically every form of nanomedicine.31
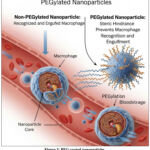 |
Figure 3: PEG-coated nanoparticles |
Neurodegenerative diseases
Before now, CNS illnesses were mostly classified as cognitive, motor, or mixed deficits based on the symptoms of the patient. This arrangement drew a lot of criticism because many symptoms did not fit into any one category and because certain symptoms were shared by many groups. Today, it is established that NDs are caused by anomalies in specific proteins such as alpha-synuclein, tau, and the amyloid precursor protein (APP). Currently, the amount of protein anomalies is being used to reclassify CNS diseases.32
Parkinson’s disease (PD)
Lewy body formation as well as dopaminergic neuron loss in the substantia nigra both lead to neurodegeneration in Parkinson’s disease (PD). In the neuron’s cell body and mechanisms, Lewy bodies, which are protein clumps of α-synuclein, can be found. Tremors, the rigidity of the body, slowness of voluntary movement, unsteady posture, and other motor dysfunctions are all brought on gradually by PD.33 The considerable loss of dopaminergic neurons cannot be reversed despite the efforts of current drugs to address the symptoms. Numerous genes have been linked to PD, supporting the theory that abnormal disorder might possibly be accelerated by abnormal protein folding and ubiquitin-proteasome pathway dysfunction. Furthermore, neurons from PD patients frequently have mitochondrial abnormalities and oxidative stress. Treatment for PD is particularly challenging due to the disease’s diverse pathology and underlying causes. Pharmacological intervention may be used to treat certain molecular diseases including mitochondrial dysfunction and aggregated alpha-synuclein. Subthalamic nuclei deep brain stimulation and cell transplanting are surgical procedures that have been studied.34
Alzheimer’s disease (AD)
The furthermost shared neurological disease in persons completed 65 is Alzheimer’s disease and it has attracted enormous financial and scientific attention. Memory loss, poor verdict, linguistic impairment, in addition to additional intellectual utilities is among the symptoms .33 Although the underlying molecular mechanisms are still unclear, two theories have gained prominence: the accumulation of amyloid-beta (Aß) and the development of neurofibrillary tangles (NFTs) (Fig. 4). When hyperphosphorylated tau protein separates from destabilized microtubules, it forms neurofibrillary tangles (NFTs), whereas amyloid precursor protein (APP) fragments that build up in Alzheimer’s patients cause Aß commemorations to form. NFTs develop inside the cell, whereas Aß signs are set down external of neurons; both reduce synaptic motioning and ultimately encourage neuronal demise. Enormous neuronal demise results in a considerable reduction in brain volume, mainly in the telencephalon, and this factor, together with the sharp drop in cognitive function, is a major factor in dementia. Along with synaptic dysfunction, the uneven mitochondrial activity causes oxidative stress, an accumulation of reactive oxygen species (ROS), a drop in the concentration of mitochondrial adenosine triphosphate, and an increase in intracellular calcium levels. Current treatment approaches aim to decrease ROS in the brain and target the destruction of Aß plaques and NFTs. Even though there are currently There are six FDA-approved Alzheimer’s therapies, but none of them are effective and individual success contrasts substantially.34, 35
Amyotrophic lateral sclerosis (ALS)
ALS patients experience severe paralysis, swallowing, speaking, and breathing failure symptoms due to spinal cord, brainstem, including motor cortex motor neuron deterioration. The usual duration from beginning to death without a cure is 20 to 48 months. Because although dangers associated with more than 40 genes have been found, the exact cause of ALS is still largely unknown. According to ongoing research, protein aggregation, degradation, and instability, particularly of RNA- and DNA-binding proteins, may contribute to the molecular pathophysiology of ALS . The pathogenesis of the disease is also influenced by the dysfunction of non-neuronal cells and the neuronal cytoskeleton. Particularly, co-culture experiments have shown that astrocytes motor neurons given ALS sufferers can be harmful, demonstrating the importance of intercellular communication in the degeneration of motor neurons.36 Additionally, since the in vivo environment is predominantly toxic, attempts to repair motor neurons are complicated by this. However, due to a lack of effective medication therapies, Motor neuron replacement using stem cells as well as pharmacological measures toward lessen the sternness of the microenvironment is being investigated. Since it aims to replace cells as well as change the environment in which transferred cells are placed, this dual strategy is particularly conducive approach mix stem cell treatment as well as nanotechnology.32, 34-36
Huntington’s Disease (HD)
An autosomal dominant monogenic neurological condition is Huntington’s disease. Patients and their families find the disease state traumatizing because of its autosomal dominant mode of transmission, progressive nature, and a combination of psychological, intellectual, and motor impairments. HD is a pathological condition caused by an enlarged tri-nucleotide CAG repeat in the gene HTT5 on chromosome 4, which codes for the aberrant pathogenic Huntington protein. The enlarged CAG repeat in the mutant protein’s unique poly-glutamine pattern is known to be hazardous and causes the death or malfunction of neuronal cells. The striatum region’s neurons are predominantly vulnerable regarding this mutated polypeptide, but HD has been shown to affect the entire brain and body. Huntington’s protein abnormality causes neuronal death in several ways, including direct effects on axonal transport, protein homeostasis, and mitochondrial function from the mutant protein’s exon 1 and its propensity to aggregate. The other theory linking HD’s neuronal damage to the loss of brain-derived neurotrophic factors, glutamate ex-cytotoxicity, and toxic effects of repeat-associated non-ATG translation.37
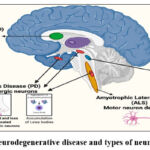 |
Figure 4: Neurodegenerative disease and types of neurons affected |
Real Problem of Neurodegenerative Disease
Age is a risk factor for ALS, Huntington’s disease, Parkinson’s disease, and Alzheimer’s disease is only a few examples of neurological illnesses. As the population ages, these neurodegenerative illnesses are significant social problems in many countries. As seen in Figure 4, the accumulation of summed and misfolded proteins in the brain is a typical neurotic aggregation in these diseases. The link between toxic protein aggregation and neurodegenerative illnesses is supported by a growing body of evidence. Because of a better understanding of the etiology of these infections, revelatory efforts for disease-changing therapies have recently greatly increased. Currently, the most prevalent and incapacitating illnesses affecting people are neurodegenerative diseases. As the fourth most common cause of mortality, they recently overtook cancer, myocardial infarction (cardiovascular illnesses), and stroke. Thinking, small-motor skills, feelings, cognitive actions, and memories are all disrupted by neurodegenerative diseases, which results in both short- and long-term problems .38
Therapeutic Approaches, Neurodegenerative Disease Care, Obstacles, and Constraints
Over the past two decades, major progress has been made in understanding neurological disorders (NDs), supported by advances in genetics and drug development.36 Several classes of drugs, including dopaminergic agents for Parkinson’s disease (PD), cholinesterase inhibitors for memory loss, analgesics for neuropathic pain, anti-inflammatory agents, and antipsychotics for dementia, are routinely used.39 Immunotherapies, both active and passive, also hold promise in Alzheimer’s disease (AD), although adverse immune responses remain a concern.39 Complementary approaches such as integrative medicine, which combines conventional treatments with traditional practices, have also gained attention. For example, Traditional Chinese Medicine (TCM) has shown benefits in improving non-motor symptoms of PD and reducing behavioral and cognitive deficits in AD.40,41 Similarly, Ayurveda has been reported to manage PD with fewer side effects, while Persian herbal medicine has demonstrated potential in alleviating ND symptoms. Exercise-based programs, such as balance training, have also improved postural stability in PD patients.
Despite these advances, several limitations and challenges remain. A major barrier is the difficulty in predicting pathological changes and identifying reliable biomarkers. In sporadic PD, for instance, non-specific symptoms make disease progression difficult to categorize, as many patients with dysentery never develop PD.42 Reliable biomarkers are urgently needed for diagnosis, patient stratification, and monitoring therapeutic response in both preclinical and clinical studies.
Another persistent challenge lies in systemic drug delivery to the central nervous system (CNS). Drugs targeting NDs often exhibit poor brain penetration due to the blood–brain barrier (BBB), first-pass metabolism, short half-lives, and off-target effects in peripheral tissues. The BBB, meninges, blood–cerebrospinal fluid barrier, choroid plexus, and circumventricular organs collectively limit CNS access.43 Thus, the development of more efficient systemic delivery strategies is critical.
In this context, nano-phytomedicines have emerged as promising alternatives; however, their translation from laboratory to clinic faces significant hurdles. Regulatory frameworks specific to plant-derived nanocarriers are lacking. Conventional nanomedicines follow existing pharmaceutical guidelines, but phytochemical-based systems present additional complexity due to variability in plant sources, batch-to-batch consistency, and stability of bioactive compounds. These issues complicate regulatory approval.
Toxicity and long-term safety also remain unresolved. The biological behavior of nanoparticles depends on their size, surface chemistry, and composition, which may lead to unpredictable effects in humans. Concerns such as tissue accumulation, unintended immune responses, and long-term neurotoxicity require systematic safety evaluation. The absence of harmonized international protocols for such testing further slows progress.
From a translational perspective, scalability and reproducibility are major obstacles. Many nano-formulations that perform well in laboratory settings lose their stability or efficacy when produced at scale. Cost-effective manufacturing methods that comply with Good Manufacturing Practices (GMP) are still underdeveloped for phytochemical-based nanosystems. Additionally, despite promising preclinical findings, only a few nano-phytomedicines have advanced to clinical trials, partly due to regulatory hesitation and the limited understanding of their performance in heterogeneous patient populations with comorbidities and polypharmacy.
Finally, issues of intellectual property and ethical commercialization need careful attention. Since many phytochemicals originate from traditional medicinal knowledge, disputes regarding ownership, patentability, and benefit-sharing may arise. Addressing these barriers will require establishing clear regulatory pathways, harmonizing safety standards, and fostering collaboration between researchers, clinicians, industry, and policymakers.
Techniques for Preparation of Nanoparticles and Nano-capsules
Nano-precipitation
The nano-precipitation method was developed by Fessi et al44 may be used to combine active chemicals with colloidal drug delivery systems. Controlled release, precise dispersion, and improved stability in biological fluids are some advantages of the produced nanoparticles. You can therefore anticipate low toxicity and moderate side effects. While the element is formed spending a non-solvent, the preparation of nanoparticles by nano-precipitation needs a strong solvent, usually an organic one like ethanol, isopropanol, or acetone (such as water). To ensure that all of the starting components are completely soluble, nano-precipitation combines an organic phase with a non-solvent phase, which is repeatedly mentioned toward as the water step. The active chemicals in the organic phase may be dissolved in an organic solvent, a combination of organic solvents, and a low HLB surfactant.44
One of the constraints limiting the amount of medication that can be placed onto the particle is the active molecule’s solubility in the solvent. The presence of water-soluble stabilizing chemicals facilitates physical stability and particulate production in the non-solvent stage. It has been reported that some particles lack stabilizing substances. Isoprenoid chains can be connected to the active molecule in this scenario due to their amphiphilic properties. When a sudden mixture of the organic component and the water step takes place, nanoparticles are created. The reliable procedure of nano-precipitation has no impact on particle size and polydispersity index. Instead, it appears that parameters related to the formulation are what control the characteristics of the nanosized system.45
This may be connected to the recommended techniques for creating nano-precipitation particles. Nano- and submicron-sized particles can only be created using particular polymer/solvent/non-solvent ratios. The Gibbs-Marangoni effect, which results from the mutual miscibility of the solvent and non-solvent and their different interfacial tensions, causes interfacial turbulence and thermal disparities and causes the organic phase to be converted into drops inside the liquid media during mechanical mixing. The substance that produces particle precipitation becomes insoluble as submicron-sized droplets of organic solvent drift away. The “ouzo effect,” This has also been put forth and is dependent on the chemical instabilities of the systems as a potential process.46
Particle-forming molecules become supersaturated in this scenario when the aqueous in addition to organic phases come together; enabling the formation of “proto particles” that adheres to the standard nucleation-and-growth process. Working situations must be provided in the direction of permit the endogenous development of submicron or nano-scale particle sizes with the lowest polydispersity indices. Due to the difficulty in standardizing nano-precipitation, polymer aggregation produces a wide variety of asymmetric particle sizes. A high organic phase ratio and a saturated organic layer are two overt indicators of polymer aggregates, as are inadequate stage interaction and low stabilizing reagent quantities.45-47
Emulsification-Diffusion Method
A process is known as “spontaneous emulsion diffusion” involves liquefying a polymer in a combination of water miscible in addition to water immiscible solvents to create biodegradable nanoparticles (such as dichloromethane). When this solution is added to water, the miscible solvent dissolves quickly and creates a nano-emulsion. When an immiscible liquid evaporates, nanoparticles are created. There is no true diffusion stage in the creation of nano-emulsions; instead, turbulence produced during solvent displacement may be used to manufacture them. This process, which combines solvent displacement with solvent evaporation, is a hybrid. The improved impulsive emulsification solvent method is a solvent displacement system because it only employs miscible solvents .48
A crucial factor in the emulsion diffusion strategy is the method employed towards persuades distribution. Low-solids dispersions are created as a result of the addition of dilution water. With this knowledge in hand, it was recommended to separate the solvent (since it has a low boiling point) as well as distill it immediately into the dispersed stage. To achieve maximum solid intensive dispersions (up to 30%) out of diverse materials, like polymers (for example, poly (D, L-lactic acid), poly (L-caprolactone), in addition to additional resources, the continuous aqueous phase must be saturated before. As a result of its quick evacuation, an anti-solvent media will be created by a free flux of the solvent globules. To cover PS80 with an emulsification solvent, Researcher created an emulsification solvent dispersion method that increased acetylcholinesterase levels while decreasing SOD1 levels and immobility in Piperine-SLNs made using Piperine. Additionally, histological analysis showed fewer plaques and tangles.49
Double Emulsion Technique
The single-emulsion method and the double-emulsion method are the most often used methods for removing solvent from emulsions. To produce the NP, this method employs an alcoholic stage that has been stabilized or emulsified utilizing surfactants in an aqueous media. Contrary towards single emulsion solvent method, which only employs two phases, the double emulsion solvent approach employs a three stage procedure. The primary drawback of these two approaches is shear stress during homogenization, which leads to ineffective protein encapsulation.50 The nano-precipitation method could be able to solve this problem. The layer-by-layer method for creating NPs is achieved by adding the organic solvent containing PLGA dropwise to the aqueous media. This causes the miscible solvent to diffuse quickly.51
The method for creating vesicular particles was developed by reported studied whichever the washing after the polymer solution or the introduction of a soluble solvent to reduce the solubility of the polymer, which can be used to apply adsorbed polymer layers to a colloidal template. Heparin, gelatin, polylysine, protamine sulfurate, chitosan, and polylysine and protamine sulfurate are a few instances of typical polymers that can polyanionic or polycationic .52
 |
Figure 5: Method of preparation of Nanoparticles |
Green Nanoparticle Production Method
Materials science in addition to technology research and improvement are moving into a new stage during which “green synthesis” approaches and technologies are gaining popularity. Regulation, control, cleanup, and remediation of the production process will enhance one of the key benefits of environmentally friendly synthesis of materials and nanomaterials. The creation of dependable, durable, and environmentally friendly synthesis processes is required for “green synthesis,” which aims to stop the production of undesired or dangerous by-products. It is essential to use the proper solvent systems and natural resources to achieve this goal. It is well known that metallic nanoparticles can be used in the environmentally friendly production of biological components like microorganisms, fungi, algae, as well as plant excerpts). The use of plant extracts to make metal and metal oxide nanoparticles is one of the more straightforward green fabrication techniques that is currently accessible, especially when compared to approaches mediated by microorganisms. These substances are grouped as “biogenic nanoparticles.53”
Solvent, temperature, pressure, and pH conditions are only a few of the reaction factors that have an impact on green methods for the synthesis biologically based components like acidic, basic, or neutral. Ketones, aldehydes, flavonoids, amides, terpenoids, carboxylic acids, phenols, and ascorbic acids are just a few examples of the potent phytochemicals found in many plant materials, particularly those made from leaves. These substances can be used to make metal and metal oxide nanoparticles (Fig.5).54
Phytomedicines Existing for Management
Acorus calamus
Acorus calamus, also known as the sweet flag and a member of the Acoraceae family, is a rejuvenator of favorable impacts on the brain and through nervous system improvements in remembrance, learning performance, and enactment. Acorus calamus is mostly composed of ɑ- and ß-asarone and ß-asarone prevents beta-amyloid-induced neuronal death from occurring in the hippocampus by downregulating Bcl-w and Bcl-2, which in turn activates caspase-3 and phosphorylates c-Jun N-terminal kinase (JNK). In addition to the previously indicated advantages, the Furthermore exhibited an inhibiting impact was Acorus calamus on AChE with an IC50 value of 188µg/mL. It has also demonstrated success in enhancing dopaminergic nerve function, acting as a neuroprotective for Parkinson’s disease.54
Allium sativum
Garlic, or Allium sativum, is a member of the Amaryllidaceae family and has been utilized for its therapeutic benefits since ancient times. It was used to treat and prevent cardiovascular disease as well as other metabolic illnesses like cancer, hypertension, diabetes, dementia, atherosclerosis, and thrombosis. One of aged garlic extract’s key ingredients (AGE), s-allyl cysteine (SAC), has undergone substantial research. Direct and indirect antioxidant action is present in SAC. It also results in a decrease in oxidation and nitration, in addition to lowering DNA fragmentation and lipid peroxidation. SAC protects dopamine levels, inhibits oxidative damage, and induces peroxidation of lipids in the Parkinsonian models 1-methyl-4-phenyl pyridinium (MPP) and 6-hydroxydopamine (6-OHDA).55
Bacopa monnieri
Bacopa monnieri (Linn), sometimes known as “Brahmi” and belonging to the Scrophulariaceae family is a tropical herb that can be found in places like India. The primary active substances in Bacopa monnieri are saponins and steroidal bacosides (A and B). Memories in addition to learning are improved by these energetic ingredients. Bacopa saponins F, E, and D, flavonoids, phytosterols, and alkaloids are other components. Bacoside A increases the activities of glutathione reductase (GSR), superoxide SOD, CAT, and GPx. Glutathione levels are consequently markedly upregulated in the brain. By altering the action of brain enzymes including Hsp 70 in addition to cytochrome P450, bacoside A prevents lipid peroxidation. Additionally, it maintains ionic homeostasis, enhances adenosine triphosphate (ATPase) activity, and replenishes the levels of zinc and selenium in the brain. Bacopa monnieri was also found to diminish the accumulation of the alpha-synuclein protein, according to studies.56
Curcumin (Cur)
Curcumin is among the most very well important natural polyphenols found in the plant Curcuma longa L. Cur is sensitive to severe effects because of its unique chemical makeup. Cur has an impact on a variety of biological and pharmaceutical targets, including genes, cytokines, growth factors, and transcription factors. Tumor necrosis factor-alpha (TNF-a), cyclooxygenase-2 (COX-2), and interleukin 8 are a few pro- and/or anti-inflammatory mediators that can be suppressed by cur to control the inflammation process (IL-8). Additionally, Cur has reportedly been shown to be efficient against Ab as well as other protein accumulation, which makes it advantageous to enhance NDs, particularly AD. Cur had inadequate pharmacokinetics, or inadequate fascination, minimal bioavailability, and rapid elimination from the body, despite its outstanding therapeutic qualities. Numerous NPS, nanocapsules, nano micelles, and nanoliposomes have been established to enhance the pharmacokinetics in addition to bioavailability of curcumin towards address these deficiencies.57
Glycyrrhiza glabra
Glycyrrhiza glabra, also known as licorice or Yashti-madhuh, is a member of the Leguminosae family. Its main component is a flavonoid known as Glabridin, which has a variety of pharmacological properties including anticancer, antiviral, anti-ulcer, antioxidant, anti-diabetic, immunomodulator, anti-inflammatory, antibacterial, and anticonvulsant action. Liquorice greatly improved learning and memory; however, studies show that it enhances general intelligence rather than short-term memory when used. Glabridin reduces glutathione levels while raising superoxide dismutase and decreasing MDA levels in the brain. According to one study, G. glabra treatment reversed the brain’s lowered levels of dopamine and glutamate as well as AChE activity.58
Benefits of Nanoparticles in ND Treatment
The biggest barrier to NDs treatment plans is the presence of BBB. To date, significant efforts have been made to solve this issue using various nano-technological techniques. A higher loading content, an extended circulatory half-life, and a strong ability to protect the medication from degradation are all characteristics of nanoparticles (PNPs), amongst the engineered drug delivery importers, have attracted particular interest. This is because they provide ligands with a variety of surface handling options for crossing the BBB. There are currently assertions that medication delivery methods based on nanoparticles can more efficiently increase drug transit even speed up the brain’s ability to absorb drugs through into the BBB. The key benefits of using nanomaterials for such medicinal objectives are reportedly their biodegradability in addition to lower poisonousness towards peripheral organs.59
To summarize, NPs laden with herbal extracts had favorable consequences on NDs through increasing the drug’s biodegradability in addition to biocompatibility, increasing its effectiveness as a treatment, eliminating pharmacokinetic constraints, minimizing its negative effects, regulating its release, and focusing on specific sites. Additionally, some nanoparticle materials have the additional prospective to improve the effectiveness of the treatment, including lowering ROS levels, having strong antioxidant characteristics, and even preventing the aggregation of Aß. Although nano-carriers can more easily pass through the BBB and are effective instruments for delivering specific substances to the brain, several issues still need to be solved. A smaller size could result in hemolysis, blood clots, and route dislocation, which would then lead to platelet aggregation. The potential hazards associated with an unbalanced dissemination of NPs in the brain are clear. The metabolism of these NPs is obscured by the inorganic components of nanomaterials, including cerium oxide, silica, iron, as well as gold particles. These substances can cause neurotoxicity when they build up in the brain because they affect mitochondrial function and interfere with processes including autophagy, apoptosis, and neuronal inflammatory processes.60
Management of Neurogenic Diseases with Nanoparticles
Nanoparticles of metal
Metal nanoparticles have drawn a great deal of interest because of their capacity to breach the BBB and improve brain imaging. For the delivery of drugs into the CNS, they can also be coated with a variety of ligands, such as proteins or antibodies. Size, surface area, stability, and structure are further characteristics that can be changed through synthesis to modify nanoparticle function.59, 60
Nanoparticles from Gold metal
Gold nanoparticles are one of the most well researched substances for CNS diagnostics and therapy. The gold core is perfect for imaging applications because of its distinctive optical characteristics, known as plasmonic features. The oscillating electromagnetic wave produced when light strikes a particle causes surface plasmon resonance (SPR), which is the resonation of surface conduction electrons. This phenomenon is specifically present in gold. This oscillation results in the formation of an oscillating dipole along the axis of light radiation, and an ionic core. The oscillation of electron charge is extremely restricted in nanomaterials and decays quickly the further away it is from the dielectric surface, with a spatial resolution related to nanoparticle size. SPR is also influenced by the nanoparticle’s surroundings, composition, shape, and structure. Through the use of SPR, light is absorbed by nanoparticles and either dispersed, radiated, used to extinguish adjacent fluorescence, or converted to heat. These optical characteristics can be used for micro-CT scanning or X-rays for in vivo imaging. Compared to conventional CT contrast agents like iodine, AuNPs absorb and decrease X-rays more effectively, providing improved contrast and more precision in the observation of nanoparticle location.60, 61
Silver nanoparticles (AgNPs)
It has been looked into if silver nanoparticles can penetrate the BBB besides trigger an immunological rejoinder in the brain. The hippocampus, a crucial organ in the treatment of neurodegenerative diseases, can be reached via the intraperitoneal injection of AgNPs. AgNPs as expected possess antibacterial properties that make them intriguing in certain situations, however at a concentration of 5 g/mL in mouse neural cells, they cause inflammatory and neurodegenerative gene expression responses. However, new research has also demonstrated that this immune response enhances the capacity of the brain’s immune cells, microglia, to express enzymes that have a general anti-inflammatory outcome and lower reactive oxygen species (ROS). Regrettably, non-targeted delivery of AgNPs to the brain frequently results in cytotoxicity, particularly to neurons, and the inert silver can add up quickly. AgNPs must either be carefully targeted to particular brain cell types or coated with ligands that lessen their cytotoxicity to be useful in treating neurodegenerative illnesses. AgNPs should not be discounted, either, since their improved capacity to traverse the BBB could result in intriguing options for immunotherapy or drug administration.62
Lipid-based nanoparticles
Because they are more biocompatible than inorganic materials, lipids and other organic compounds found in cells naturally provide effective tools in nanomedicine. Lipid nanocarriers are the best medication carriers for both nanoparticle therapy and co-treatment with stem cell therapy because they also evade efflux after entering the brain.63
PLGA nanoparticles
The FDA has cleared the synthetic polymer PLGA for use in several biological applications. In-vivo hydrolysis of PLGA into glycolic acid and lactic acid results in great biocompatibility. Through conjugation with different surface ligands, it is very helpful for prolonged drug release and brain-specific targeting. In one recent work, encapsulated in PLGA followed by a coating of the NP with Tet1 peptide, which has a high affinity for neurons and encourages retrograde transport, improved the administration of the hydrophilic medication, natto-kinase, over the BBB. To treat Alzheimer’s, these PLGA NPs successfully reduced amyloid aggregation and increased the stability of the nattokinase protein.64
Polymeric nanoparticles
Polymeric nanoparticles are advantageous drug carriers because of their flexible physical characteristics, easy production, and quick in vivo breakdown. They can lessen the cargo’s immunogenicity and enhance the encapsulated proteins’ pharmacokinetic characteristics. Poly (lactic acid), poly (D, L-lactic-co-glycolic acid), poly (aspartic acid), poly (glycolic acid), and poly (butyl cyanoacrylate) is the most widely utilized polymeric polymers (PBCA). These polymers are therefore promising compounds for treating neurodegeneration because they have been widely used in other regeneration disciplines. 64
Multiple production methods, high steadiness, as well as bioavailability, long-term movement of encapsulated pharmaceuticals, little immunological reactivity, and malleable one of the commonalities of polymer NPs that makes them effective drug transporters is their physical characteristics. Size, shape, zeta potential, rate of degradation, and efficiency of entrapment of particles are physical characteristics that should be taken into account while choosing and synthesizing polymeric nanoparticles. There are only a few examples that demonstrate the potential of NPs to cure neurological disorders illnesses will be provided in this study due to the enormous number of research that has been conducted employing each of these polymers.65
Green Synthesized Nanoparticles
Ecological Chemistry eco-friendly nanotechnology is currently being promoted as a purpose for green technology. Metal nanoparticles, however, have been demonstrated to be preferable to microbes in the synthesis of nanoparticles for herbal extracts. Due to the presence of polyphenols, the organic components might break down into biodegradable material. An ayurvedic plant called shankhapushpi has antihypertensive, immunomodulator, and anticonvulsant properties. Convolvulus pluricaulis, an ayurvedic herb, has antioxidant properties that have been found to enhance memory in nanoparticles produced using iron oxide as a precursor. The molecular mechanism underlying these microscopic particles is still a mystery to scientists. The Pulicaria undulata plant is widely used in traditional Chinese medicine as an anti-epileptic, insect repellent, and antioxidant. Silver nanoparticles (AgNPs) are found in higher concentrations in the brain, and this interaction with proteins limits the production of fibrils by reducing protein conformation and self-association in Pulicaria undulata silver nanoparticles (AgNPs).66
Liposomes-Based Drug Delivery Systems
The blood-brain barrier liposome transport mechanisms are still in their infancy.67 Negative charges on the BBB and cationic liposomal drug delivery methods cause cell internalization through an absorption mechanism through electrostatic interactions. By adding glucose and GSH to liposomes as nutrients, the BBB can be crossed.68 Because receptor-mediated transcytosis, which can connect particular ligands to a variety of BBB receptors, has therapeutic potential? The BBB bridging is improved by using nano-liposomes that have been dual functionalized with the HIV TAT peptide and curcumin. The two most frequently mentioned pathways for the uptake of TAT were endocytosis and micropinocytosis. TAT and nanoliposomes are covalently joined via a thiolmaleimide reaction. TAT-CurcNL was measured using HPLC-MS/MS and has a size range of 196.5 to 3.2nm.69 After TAT functionalization in human brain capillary endothelial cells, mass spectrometry, confocal microscopy, and a radioactivity assay employing [3H]-sphingomyelin all demonstrated a threefold increase in nano-liposome absorption (hCMEC and D3).70
Green-Extract Nanoparticles
Ginkgo biloba
An ancient Chinese tree known as Ginkgo biloba (Ginkgoaceae) is widely cultivated for both traditional and medical uses. Flavonol glycosides, bilobalide, terpene trilactones, various ginkgolides, and ginkgolic acid are all present in G. Biloba extract. The standardized G. Biloba extract is widely used in Europe to enhance the therapeutic state of dementia patients with various kinds of dementia.71 Niosome formulation of G. Biloba extract was discovered to be able to prolong the release period of flavonoid glycosides with better oral bioavailability and pharmacokinetic characteristics, making it a suitable method for delivering G. Biloba extract to the brain. Comparatively to animals in the control group, nanosized G. Biloba extract particles encouraged the release of the neurotransmitter acetylcholine from specific regions of the brain. G. Biloba extract with nanosized particles demonstrated enhanced tolerability and good absorption characteristics.72, 75
Pomegranate Seed Oil
Punica granatum, a fruit considered sacred, has large amounts of polyphenolic chemicals as well as punicic acid (PA). Pomegranate seed oil nano-droplets were developed in 2013 to treat Creutzfeldt-Jakob disease (CJD). The study’s findings showed that although the accumulation of the scrapie isoform of the prion protein (PrPSc) did not change significantly, neuronal loss and lipid oxidation did, which suggests that pomegranate seed oil has neuroprotective properties. Pomegranate seed oil in nano droplet form lowered the severity of the disease more than free pomegranate seed oil did in a mouse model of multiple sclerosis .73, 75
Thymoquinone (TQ)
TQ is the primary active ingredient in Nigella Sativa (Ranunculaceae) seed. TQ is a lipophilic substance that exhibits a variety of pharmacological properties in immunomodulation, neurodegeneration, and cognitive impairments. TQ delivery to the brain is difficult, though. The nanoemulsion of TQ rich fraction (TQRF) and TQ alleviated memory impairments and raised the total antioxidant status in rats on a high-fat, high-cholesterol diet, while drastically reducing Aß expression. The activity of g- and b-secretase enzymes was controlled in a similar situation by TQRF nanoemulsion and TQ nanoemulsion, which increased the breakdown of Aß and its removal from the brain. N. sativa oil (NSO) and plasmid DNA were co-encapsulated, which showed that NSO might be employed as a reliable gene delivery carrier for treating NDs, particularly in AD patients. TQ-encapsulated chitosan nanoparticles (NPs) were examined in a study for the nose-to-brain targeting strategy. Targeting from the nose to the brain can lessen the negative effects of TQ on the whole body. The study’s findings supported the validity of TQ when compared to earlier approaches.74, 75
Curcumin
To protect SKN-SH dopaminergic cells from rotenone-induced neurotoxicity, a model that imitates PD symptoms, curcumin-loaded lactoferrin NPs were created. Along with prolonged retention, Cur’s intracellular absorption and concentration rose, strengthening its neuroprotective properties. A lipid-polyethyleneglycol-polylactic acid (PEG)-Cur derivative greatly decreased the aggregation of Aß in vitro. In post-mortem samples from AD patients, it was claimed that Cur-derivative liposomes and Cur-derivative anti-transferrin antibody liposomes enhanced the drug’s brain penetration. Nanoliposomes of Cur or Cur derivatives can regulate or inhibit the development of Aß oligomers or fibrils in vitro.72-74
A Cur derivative in a modified HIV trans-activating Transcriptional Activator (TAT) peptide (TATCur-NL) double-functionalized nanoliposome demonstrated an excellent affinity for Aß peptides and was able to traverse the BBB in vitro. Apolipoprotein E3-mediated poly (butyl) cyanoacrylate NPs with Cur (ApoE3-C-PBCA) improved the photostability and cellular absorption of Cur due to a sustained drug release in another in vitro investigation. Due to their inhibitory effects on the synthesis of Aß and the resulting inhibition of oxidation and the development of free radicals, PLGA coated-Cur NPs conjugated with Tet-1 peptide have been proven to be effective for treating AD. Additionally, nano-liposomes coated with Cur exhibited a phenomenal affinity for the Aß1-42 fibril. The fibrillation of Aß42 fibrils was more successfully suppressed by Cur conjugated to a zwitterion polymer (carboxy betaine methacrylate)-NPs than by free Cur. In a different study, Cur-PLGA-NPs increased the expression of genes involved in neuronal differentiation and cell proliferation to induce neurogenesis in neural stem cells.74
Rats with 3-nitropropionic acid (3-NP)-induced HD were helped by curcumin-encapsulated solid lipid nanoparticles (CSLNs). The antioxidant enzyme activities (SOD and glutathione, for example) were significantly increased in the CSLNs-treated mice, whereas ROS, protein carbonyls, and lipid peroxidation were significantly reduced. In comparison to animals just treated with selenium-NPs, Cur-selenium-PLGA nanospheres were demonstrated to be more effective in AD mice. A dual drug-loaded lipid-based nanoformulation (Cur and PIP) was successful in treating PD in different research. In comparison to non-formulated medicines, this impact was associated with the suppression of a-synuclein aggregation, improvement of Cur bioavailability, reduction of oxidative stress, more effective clearance of faulty proteins, and acceleration of anti-apoptotic processes.74,75
In a different study, Cur was loaded onto a lactoferrin-modified low-density lipoprotein (LDL)-mimic nanostructured lipid carrier (Lf-mNLC). When Lf-mNLC was given to AD mice, the concentration of Cur in the brain increased and its bioavailability increased dramatically. This suggests that Lf-mNLC significantly regulated the symptoms and progression of AD. According to a study, a specific kind of nano-Cur proved effective in correcting the expression patterns of dysregulated miRNAs in MS patients.75
Quercetin (QC)
Known for its neuroprotective and anti-inflammatory properties, quercetin is a bioflavonoid that can be found in a variety of fruits, vegetables, and oils with herbal origins. Additionally, QC has a significant amount of ROS scavenging power. Despite having positive effects, its clinical applications were hampered by weak solubility and low absorption. As a result, other QC formulations, including nanocapsules, nanogels, liposomes, nanosuspensions, and microspheres, have been suggested to address such restrictions, with QC-nanoencapsulation having been demonstrated to be the most appropriate form. A study found that QC-loaded nano lipidic carriers (NLCs) increased the antioxidant activity of QC while also improving its absorption and distribution to the brain.41, 75
By loading QC into PLGA nanocapsules with dodecyl triphenylphosphonium bromide (TPPC) as one of the matrix components (N1QC) structure in the cerebral ischemia reperfusion-induced model, the mitochondrial delivery of QC was boosted. After cerebral ischemia-reperfusion, N1QC showed increased brain uptake, substantial bioavailability, and mitochondrial localization. 39, 75
Conclusion
Nanotechnology offers considerable promise in advancing therapeutic strategies for neurological disorders (NDs). By enabling nano-enhanced delivery systems, it contributes not only to the protection of neurons but also to the development of innovative approaches in neurophysiology and neuropathology. With the increasing global prevalence of NDs, driven partly by rising life expectancy, there is an urgent need for novel interventions that improve both symptom management and quality of life.
The blood–brain barrier (BBB) remains the major obstacle to effective central nervous system (CNS) drug delivery. Although multiple delivery platforms have been investigated, phytochemical-based nanocarriers have emerged as particularly attractive options due to their safety, eco-friendliness, lower toxicity, cost-effectiveness, and scalability, alongside their tunable particle size and morphology. Such systems can enhance the pharmacokinetic and bioavailability profiles of plant-derived therapeutics, facilitate deeper brain penetration, and potentially disrupt or prevent the aggregation of pathological proteins associated with neurodegeneration.
Preclinical studies have already demonstrated encouraging outcomes with nanoparticle-mediated phytochemicals; however, unresolved challenges persist. Long-term safety, biodistribution, and potential neurotoxicity of nanosystems remain important concerns. Moreover, despite promising laboratory results, translation into clinical practice is limited, highlighting a significant gap between experimental advances and patient applications. To move the field forward, more rigorous clinical trials and the development of disease-specific and cell-targeted nanoformulations are essential.
In summary, nano-phytomedicine represents a rapidly evolving and promising field that not only addresses limitations of conventional therapies but also offers novel avenues for precision medicine in neurological care. Its continued advancement will depend on balancing therapeutic innovation with thorough safety evaluations and regulatory considerations.
Acknowledgement
The authors would like to express their sincere gratitude to The Assam Kaziranga University, Jorhat, Assam, for providing the necessary support and academic environment to carry out this review work. We are especially thankful to the faculty and staff of the School of Pharmacy for their guidance, encouragement, and access to relevant resources throughout the preparation of this manuscript.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability
This statement does not apply to this article
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Authors’ Contribution
- Ranajit Nath helped to organize the literature, compiled references, and wrote the article, manuscript preparation, revision, and writing—original draft;
- Rajesh E Jesudasan contributed to the conception of the review and polished the language, table and picture layout.
Reference
- Bensadoun JC, Almeida LP, Fine EG, Tseng JL, Déglon N, Aebischer P. Comparative study of GDNF delivery systems for the CNS: Polymer rods, encapsulated cells, and lentiviral vectors. J Control Release. 2003;87(1-3):107-115.
CrossRef - Kishima H, Poyot T, Bloch J, et al. Encapsulated GDNF-producing C2C12 cells for Parkinson’s disease: A pre-clinical study in chronic MPTP-treated baboons. Neurobiol Dis. 2004;16(3):428-439.
CrossRef - McArthur JC. HIV dementia: An evolving disease. J Neuroimmunol. 2004;157(1-2):3-10.
CrossRef - Menei P, Montero-Menei C, Venier MC, Benoit JP. Drug delivery into the brain using poly(lactide-co-glycolide) microspheres. Expert Opin Drug Deliv. 2005;2(2):363-376.
CrossRef - Flachenecker P. Epidemiology of neuroimmunological diseases. J Neurol. 2006;253(Suppl 5):V2–V6.
CrossRef - Kabanov AV, Gendelman HE. Nanomedicine in the diagnosis and therapy of neurodegenerative disorders. Prog Polym Sci. 2007;32(8-9):1054-1082.
CrossRef - Ferri CP, Prince M, Brayne C, et al. Global prevalence of dementia: A Delphi consensus study. Lancet. 2005;366(9503):2112-2117.
CrossRef - Popovic N, Brundin P. Therapeutic potential of controlled drug delivery systems in neurodegenerative diseases. Int J Pharm. 2006;314(2):120-126.
CrossRef - Singh N, Pillay V, Choonara YE. Advances in the treatment of Parkinson’s disease. Prog Neurobiol. 2007;81(1):29-44.
CrossRef - Tysnes OB, Storstein A. Epidemiology of Parkinson’s disease. J Neural Transm (Vienna). 2017;124(8):901-905.
CrossRef - Freed CR, Greene PE, Breeze RE, et al. Transplantation of embryonic dopamine neurons for severe Parkinson’s disease. N Engl J Med. 2001;344(10):710-719.
CrossRef - Pollak P, Fraix V, Krack P, et al. Treatment results: Parkinson’s disease. Mov Disord. 2002;17(Suppl 3):S75-S83.
CrossRef - Johnson WG. Late-onset neurodegenerative diseases—The role of protein insolubility. J Anat. 2000;196(4):609-616.
CrossRef - Dickson DW. Misfolded, protease-resistant proteins in animal models and human neurodegenerative disease. J Clin Invest. 2002;110(10):1403-1405.
CrossRef - Woodruff BK, Graff-Radford NR, Ferman TJ, et al. Family history of dementia is a risk factor for Lewy body disease. Neurology. 2006;66(12):1949-1950.
CrossRef - Walia V, Kaushik D, Mittal V, et al. Delineation of Neuroprotective Effects and Possible Benefits of Antioxidants Therapy for the Treatment of Alzheimer’s Diseases by Targeting Mitochondrial-Derived Reactive Oxygen Species: Bench to Bedside. Mol Neurobiol. 2021.
CrossRef - Nischwitz V, Berthele A, Michalke B. Speciation analysis of selected metals and determination of their total contents in paired serum and cerebrospinal fluid samples: An approach to investigate the permeability of the human blood-cerebrospinal fluid-barrier. Anal Chim Acta. 2008;627(2):258-269.
CrossRef - Gendelman HE, Ding S, Gong N, et al. Monocyte chemotactic protein-1 regulates voltage-gated K+ channels and macrophage transmigration. J Neuroimmune Pharmacol. 2009;4(1):47-59.
CrossRef - Modi G, Pillay V, Choonara YE, et al. Nanotechnological applications for the treatment of neurodegenerative disorders. Prog Neurobiol. 2009;88(4):272-285.
CrossRef - Page MJ, McKenzie JE, Bossuyt PM, et al. Updating guidance for reporting systematic reviews: Development of the PRISMA 2020 statement. J Clin Epidemiol. 2021;134:103-112.
CrossRef - Alzheimer’s Association. 2016 Alzheimer’s disease facts and figures. Alzheimers Dement. 2016;12(4):459-509.
CrossRef - Tam VH, et al. Nanomedicine as a non-invasive strategy for drug delivery across the blood brain barrier. Int J Pharm. 2016;515(1-2):331-342.
CrossRef - Wen MM, et al. Nanotechnology-based drug delivery systems for Alzheimer’s disease management: Technical, industrial, and clinical challenges. J Control Release. 2017;245:95-107.
CrossRef - Sabelstrom H, et al. Resident neural stem cells restrict tissue damage and neuronal loss after spinal cord injury in mice. Science. 2013;342(6158):637-640.
CrossRef - Zhang B, et al. Nanomaterials in neural-stem-cell-mediated regenerative medicine: Imaging and treatment of neurological diseases. Adv Mater. 2018;30(1):1705694.
CrossRef - Lindvall O, Kokaia Z. Stem cells for the treatment of neurological disorders. Nature. 2006;441(7097):1094-1096.
CrossRef - Ahmad J, et al. Nanotechnology based theranostic approaches in Alzheimer’s disease management: Current status and future perspective. Curr Alzheimer Res. 2017;14(11):1164-1181.
CrossRef - Abbott NJ, et al. Structure and function of the blood-brain barrier. Neurobiol Dis. 2010;37(1):13-25.
CrossRef - Azad TD, et al. Therapeutic strategies to improve drug delivery across the blood brain barrier. Neurosurg Focus. 2015;38(3):E9.
CrossRef - Mathiisen TM, et al. The perivascular astroglial sheath provides a complete covering of the brain microvessels: An electron microscopic 3D reconstruction. Glia. 2010;58(9):1094-1103.
CrossRef - Reddy PH, et al. Abnormal mitochondrial dynamics and synaptic degeneration as early events in Alzheimer’s disease: Implications to mitochondria-targeted antioxidant therapeutics. Biochim Biophys Acta. 2012;1822(5):639-649.
CrossRef - AlDakheel A, Kalia LV, Lang AE. Pathogenesis-targeted, disease-modifying therapies in Parkinson disease. Neurotherapeutics. 2014;11(1):6-23.
CrossRef - Kalia LV, Lang AE. Parkinson’s disease. Lancet. 2015;386(9996):896-912.
CrossRef - Bjorklund A, Kordower JH. Cell therapy for Parkinson’s disease: What next? Mov Disord. 2013;28(1):110-115.
CrossRef - Chio A, et al. Prognostic factors in ALS: A critical review. Amyotroph Lateral Scler. 2009;10(5-6):310-323.
CrossRef - Peters OM, Ghasemi M, Brown RH Jr. Emerging mechanisms of molecular pathology in ALS. J Clin Invest. 2015;125(6):2548.
CrossRef - Silani V, et al. Stem-cell therapy for amyotrophic lateral sclerosis. Lancet. 2004;364(9429):200-202.
CrossRef - Huang Y, Mucke L. Alzheimer mechanisms and therapeutic strategies. Cell. 2012;148(6):1204-1222.
CrossRef - Sintov AC, et al. Metal nanoparticles as targeted carriers circumventing the blood brain barrier. Int Rev Neurobiol. 2016;130:199-227.
CrossRef - Velasco-Aguirre C, et al. Peptides and proteins used to enhance gold nanoparticle delivery to the brain: Preclinical approaches. Int J Nanomedicine. 2015;10:4919-4936.
CrossRef - Pujals S, et al. Mechanistic aspects of CPP-mediated intracellular drug delivery: Relevance of CPP self-assembly. Biochim Biophys Acta. 2006;1758(3):264-279.
CrossRef - Allen TM, et al. Uptake of liposomes by cultured mouse bone marrow macrophages: Influence of liposome composition and size. Biochim Biophys Acta. 1991;1061(1):56-64.
CrossRef - Lee RJ, Low PS. Delivery of liposomes into cultured KB cells via folate receptor mediated endocytosis. J Biol Chem. 1994;269(5):3198-3204.
CrossRef - Fessi H, Puisieux F, Devissaguet JP, Ammoury N, Benita S. Nanocapsule formation by interfacial polymer deposition following solvent displacement. Int J Pharm. 1989;55:R1-R4.
CrossRef - Mora-Huertas CE, Fessi H, Elaissari A. Influence of process and formulation parameters on the formation of submicron particles by solvent displacement and emulsification-diffusion methods: Critical comparison. Adv Colloid Interface Sci. 2011;163(1):90-122.
CrossRef - Cavalu S, Bisboaca S, Mates IM, et al. Novel formulation based on chitosan-arabic gum nanoparticles entrapping propolis extract: Production, physico-chemical and structural characterization. Rev Chim. 2018;69(12):3756-3760.
CrossRef - Erdogar N, Akkin S, Bilensoy E. Nanocapsules for drug delivery: An updated review of the last decade. Recent Pat Drug Deliv Formul. 2018;12(4):252-266.
CrossRef - Niwa T, Takeuchi H, Hino T, Kunou N, Kawashima Y. Preparations of biodegradable nanospheres of water-soluble and insoluble drugs with D,L-lactide/glycolide copolymer by a novel spontaneous emulsification solvent diffusion method, and the drug release behavior. J Control Release. 1993;25(1-2):89-98.
CrossRef - Murakami H, Kobayashi M, Takeuchi H, Kawashima Y. Preparation of poly(dl-lactide-co-glycolide) nanoparticles by modified spontaneous emulsification solvent diffusion method. Int J Pharm. 1999;187(2):143-152.
CrossRef - Quintanar-Guerrero D, Allemann E, Fessi H, Doelker E. Pseudolatex preparation using a novel emulsion-diffusion process involving direct displacement of partially water-miscible solvents by distillation. Int J Pharm. 1999;188(2):155-164.
CrossRef - Yusuf M, Khan M, Khan RA, Ahmed B. Preparation, characterization, in vivo and biochemical evaluation of brain-targeted piperine solid lipid nanoparticles in an experimentally induced Alzheimer’s disease model. J Drug Target. 2013;21(4):300-311.
CrossRef - McCall RL, Sirianni RW. PLGA nanoparticles formed by single- or double-emulsion with vitamin E-TPGS. J Vis Exp. 2013;(82):51015.
CrossRef - Huang W, Zhang C. Tuning the size of poly(lactic-co-glycolic acid) (PLGA) nanoparticles fabricated by nanoprecipitation. Biotechnol J. 2018;13(2):1700203.
CrossRef - Bandelow B, Michaelis S, Wedekind D. Treatment of anxiety disorders. Dialogues Clin Neurosci. 2017;19(2):93-107.
CrossRef - Powell CR, Dillon KM, Matson JB. A review of hydrogen sulfide (H2S) donors: Chemistry and potential therapeutic applications. Biochem Pharmacol. 2018;149:110-123.
CrossRef - Shang A, Cao SY, Xu XY, et al. Bioactive compounds and biological functions of garlic (Allium sativum L.). Foods. 2019;8(7):246.
CrossRef - Chen X, Pan W. The treatment strategies for neurodegenerative diseases by integrative medicine. Integr Med Int. 2014;1(4):223-225.
CrossRef - Roy S, Awasthi H. Herbal medicines as neuroprotective agent: A mechanistic approach. Int J Pharm Pharm Sci. 2017;9(6):1-7.
CrossRef - Chopra H, Dey PS, Das D, et al. Curcumin nanoparticles as promising therapeutic agents for drug targets. Molecules. 2021;26(16):4998.
CrossRef - Cavalu S, Prokisch J, Laslo V, Vicas S. Preparation, structural characterisation and release study of novel hybrid microspheres entrapping nanoselenium, produced by green synthesis. IET Nanobiotechnol. 2017;11(4):426-432.
CrossRef - Tarhini M, Greige-Gerges H, Elaissari A. Protein-based nanoparticles: From preparation to encapsulation of active molecules. Int J Pharm. 2017;522(1-2):172-197.
CrossRef - Nogueira E, et al. Design of liposomal formulations for cell targeting. Colloids Surf B. 2015;136:514-526.
CrossRef - Needham D, Kim DH. PEG-covered lipid surfaces: Bilayers and monolayers. Colloids Surf B. 2000;18(3-4):183-195.
CrossRef - Ahl PL, et al. Enhancement of the in vivo circulation lifetime of L-alpha-distearoylphosphatidylcholine liposomes: Importance of liposomal aggregation versus complement opsonization. Biochim Biophys Acta. 1997;1329(2):370-382.
CrossRef - Vert M, Domurado D. Poly(ethylene glycol): Protein-repulsive or albumin incompatible? J Biomater Sci Polym Ed. 2000;11(12):1307-1315.
CrossRef - Ishida T, Kiwada H. Accelerated blood clearance (ABC) phenomenon upon repeated injection of PEGylated liposomes. Int J Pharm. 2008;354(1-2):56-62.
CrossRef - Joshi S, Singh-Moon RP, Ellis JA, et al. Cerebral hypoperfusion-assisted intra-arterial deposition of liposomes in normal and glioma-bearing rats. Neurosurgery. 2015;76(1):92-100.
CrossRef - Noble GT, Stefanick JF, Ashley JD, Kiziltepe T, Bilgicer B. Ligand-targeted liposome design: Challenges and fundamental considerations. Trends Biotechnol. 2014;32(1):32-45.
CrossRef - Sancini G, Gregori M, Salvati E, et al. Functionalization with TAT-peptide enhances blood-brain barrier crossing in vitro of nanoliposomes carrying a curcumin derivative to bind amyloid-peptide. J Nanomed Nanotechnol. 2013;4(3):171.
CrossRef - Mourtas S, Lazar AN, Markoutsa E, et al. Multifunctional nanoliposomes with curcumin-lipid derivative and brain targeting functionality with potential applications for Alzheimer disease. Eur J Med Chem. 2014;80:175-183.
CrossRef - Boroushaki MT, Mollazadeh H, Afshari RA. Pomegranate seed oil: A comprehensive review on its therapeutic effects. Int J Pharm Sci Res. 2016;7:430.
- Alam S, Khan ZI, Mustafa G, et al. Development and evaluation of thymoquinone-encapsulated chitosan nanoparticles for nose-to-brain targeting: A pharmacoscintigraphic study. Int J Nanomedicine. 2012;7:5705-5718.
CrossRef - Dolati S, Babaloo Z, Ayromlou H, et al. Nanocurcumin improves regulatory T-cell frequency and function in patients with multiple sclerosis. J Neuroimmunol. 2019;327:15-21.
CrossRef - Brahmkhatri VP, Sharma N, Sunanda P, et al. Curcumin nanoconjugate inhibits aggregation of N-terminal region (Ab-16) of an amyloid beta peptide. New J Chem. 2018;42(24):19881-19892.
CrossRef - Moradi SZ, Momtaz S, Bayrami Z, Farzaei MH, Abdollahi M. Nanoformulations of herbal extracts in treatment of neurodegenerative disorders. Front Bioeng Biotechnol. 2020;8:238.
CrossRef







