Manuscript accepted on :20-08-2025
Published online on: 18-09-2025
Plagiarism Check: Yes
Reviewed by: Dr. Anjaneyulu Vinukonda
Second Review by: Dr. Nishu Raina
Final Approval by: Dr. Kamal Upreti
Jayasri Bantaram , Shakthi Harikrishnan
, Shakthi Harikrishnan , Sandhya Ravishankar
, Sandhya Ravishankar and Renukadevi Jeyavelkumaran*
and Renukadevi Jeyavelkumaran*
Department of Pharmaceutics, Saveetha College of Pharmacy, Saveetha Institute of Medical and Technical Sciences, Chennai, India.
Corresponding Author E-mail: renukadevij.scop@saveetha.com
DOI : https://dx.doi.org/10.13005/bpj/3405
Abstract
Parkinson’s disease (PD) is a progressive neurodegenerative illness characterized by the buildup of misfolded alpha-synuclein in Lewy bodies and the loss of dopaminergic neurons in the substantia nigra. The unfolded protein response (UPR) and endoplasmic reticulum (ER) stress have been implicated in the pathophysiology of Parkinson’s disease (PD) in recent research.BiP/GRP78, an ER-resident chaperone, serves as a central regulator of the UPR by sensing misfolded proteins and modulating three primary stress sensors: PERK, IRE1, and ATF6. While BiP initially acts protectively by restoring ER homeostasis, its sustained activation may lead to maladaptive UPR signaling and neuronal apoptosis. Experimental models have demonstrated that agents such as salubrinal can modulate BiP levels and UPR activity, offering neuroprotection. Moreover, small molecules targeting BiP interactions or its ATPase activity are emerging as potential therapeutics. However, challenges such as blood–brain barrier penetration, tissue-specific effects, and long-term safety must be addressed. Future strategies may include targeted delivery systems, combination therapies involving autophagy modulation, and personalized treatment guided by BiP expression or genomic profiling. This review outlines the dual role of BiP in PD and discusses its therapeutic implications, highlighting the need for precise modulation to maximize clinical benefits while minimizing risks.
Keywords
Alpha-synuclein; Apoptosis; BiP (Binding Immunoglobulin Protein); Endoplasmic reticulum stress; Protein aggregation; Unfolded protein response
| Copy the following to cite this article: Bantaram J, Harikrishnan S, Ravishankar S, Jeyavelkumaran R. BiP/GRP78 and Unfolded Protein Response in Parkinson’s Disease: Mechanisms, Therapeutic Potential, and Future Directions. Biomed Pharmacol J 2025;18(October Spl Edition). |
| Copy the following to cite this URL: Bantaram J, Harikrishnan S, Ravishankar S, Jeyavelkumaran R. BiP/GRP78 and Unfolded Protein Response in Parkinson’s Disease: Mechanisms, Therapeutic Potential, and Future Directions. Biomed Pharmacol J 2025;18(October Spl Edition). Available from: https://bit.ly/4nIJanM |
Introduction
The motor system is the primary target of Parkinson’s disease (PD), a progressive neurodegenerative illness characterized by bradykinesia, postural instability, muscle rigidity, and resting tremors1. A variety of non-motor symptoms, such as mood problems, autonomic dysfunction, and cognitive loss, are commonly linked to Parkinson’s disease (PD) in addition to motor dysfunction.2 According to epidemiological data, those 60 years of age and older account for almost 50% of PD cases, highlighting age as a major risk factor.3 PD is typified at the cellular level by the development of intracellular inclusions called Lewy bodies, which are mainly made up of misfolded alpha-synuclein protein, and the specific degradation of dopaminergic neurons in the substantia nigra pars compacta.4 Although the etiology of Parkinson’s disease (PD) is complex and includes genetic, environmental, and aging-related factors, new research suggests that endoplasmic reticulum (ER) stress and unfolded protein response (UPR) activation are important pathways that contribute to the pathophysiology of the disease.5 The ER is an essential organelle involved in protein folding, calcium homeostasis, and lipid metabolism.6 The UPR, a cellular adaptation mechanism intended to re-establish proteostasis, is triggered by disruptions in ER function, namely the buildup of misfolded or unfolded proteins. This response involves the increase of misfolded protein breakdown pathways, the activation of ER chaperones, and the temporary inhibition of global protein translation. On the other hand, persistent or unresolved ER stress might cause the UPR to change from its protective to pro-apoptotic function, which will ultimately encourage the death of neurons.7 The ER-resident chaperone BiP/GRP78, a crucial sensor of protein misfolding, is a major mediator of the UPR. BiP binds to and suppresses the UPR transducers IRE1, ATF6, and PERK in non-stressful situations.Upon accumulation of misfolded proteins, BiP dissociates from these sensors to engage aberrant proteins, thereby permitting activation of the UPR signaling pathways. Understanding the molecular mechanisms by which BiP and the UPR contribute to PD pathophysiology may open new avenues for pharmacological intervention aimed at enhancing neuronal resilience and mitigating disease progression.8 This review aims to explore the emerging role of ER stress and the UPR in Parkinson’s disease, with a particular focus on the regulatory function of BiP/GRP78 and its therapeutic potential in modulating neurodegenerative processes.
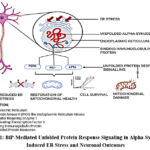 |
Figure 1: BiP-Mediated Unfolded Protein Response Signaling in Alpha-Synuclein-Induced ER Stress and Neuronal Outcomes |
BiP and the Unfolded Protein Response in Parkinson’s Disease
One important member of the heat shock protein 70 (HSP70) family that is found in the endoplasmic reticulum (ER) is binding immunoglobulin protein (BiP), often referred to as glucose-regulated protein 78 (GRP78). BiP has a substrate-binding domain (SBD) that interacts with unfolded or misfolded proteins and an N-terminal nucleotide-binding domain (NBD) that hydrolyzes ATP.9 BiP is a key regulator of the unfolded protein response (UPR), which is triggered to restore proteostasis in the setting of ER stress. BiP interacts to the luminal domains of the three primary UPR sensors—activating transcription factor 6 (ATF6), inositol-requiring enzyme 1 (IRE1), and PKR-like ER kinase (PERK)—under homeostatic conditions, keeping them dormant.10 Upon ER stress, BiP dissociates from these sensors to bind accumulating misfolded proteins, leading to activation of UPR pathways: phosphorylation of eIF2α via PERK, splicing of XBP1 mRNA via IRE1, and translocation of ATF6 to the Golgi for proteolytic activation. These events collectively reduce global protein synthesis, increase folding capacity, and enhance degradation of misfolded proteins.11 In Parkinson’s disease (PD), chronic ER stress—largely due to intracellular accumulation of misfolded alpha-synuclein—can result in persistent UPR activation, tipping the balance from adaptive to apoptotic signaling, and contributing to neuronal death. The regulation of BiP is thus crucial: while early activation may help mitigate initial stress, prolonged or excessive BiP expression can exacerbate pathological ER stress.12 Dissociation of BiP from UPR transducers was shown to initiate essential signaling pathways required for maintaining ER homeostasis. Alpha-synuclein aggregates were found to sustain BiP activation, leading to persistent ER stress and subsequent neuronal apoptosis.Their findings indicate that while transient BiP upregulation supports neuronal survival, prolonged overexpression can intensify ER dysfunction and accelerate neurodegeneration as depicted in FIG1.13 Hence, fine-tuning BiP activity represents a promising therapeutic target in PD.
Recent Breakthroughs: BiP Modulation in Experimental Parkinson’s Models
Salubrinal and BiP Downregulation
Salubrinal, a selective inhibitor of eIF2α phosphatase complexes, prevents the dephosphorylation of eIF2α, maintaining suppression of global protein synthesis and alleviating ER stress.In experimental PD models, salubrinal has shown neuroprotective effects by reducing protein load and preserving dopaminergic neurons.14 Interestingly, salubrinal has also been observed to modulate BiP expression, suggesting a dual role in UPR regulation.Though BiP assists in protein folding, sustained overexpression may prolong UPR signaling detrimentally.Salubrinal appears to initially elevate BiP expression but subsequently downregulates it, potentially helping to resolve persistent ER stress.15 BiP levels were observed to increase at 9 and 24 hours after chemical hypoxia in primary cortical neurons, but to decrease by 48 hours following salubrinal treatment16 In mammalian cell lines, salubrinal-induced eIF2α phosphorylation was found to reduce protein synthesis and ER stress, thereby supporting cell survival16. In an LPS-induced rat model of Parkinson’s disease, salubrinal administration was shown to reduce inflammation, preserve dopaminergic neurons, and inhibit caspase-3 and -9 activation, underscoring its neuroprotective potentia17 In α-synuclein-overexpressing mice, salubrinal treatment was observed to mitigate motor deficits and downregulate ER stress markers such as BiP and CHOP18 These findings collectively support salubrinal’s potential to restore UPR balance and reduce neurodegeneration in PD and is collectively depicted in FIG 2.
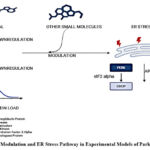 |
Figure 2: BiP Modulation and ER Stress Pathway in Experimental Models of Parkinson’s Disease |
Other Small Molecules and BiP Regulators
Compounds like as thapsigargin, an inhibitor of ER calcium ATPase, and tunicamycin, an N-linked glycosylation inhibitor, are frequently employed in experimental models to investigate BiP and UPR regulation under regulated ER stress. They are useful for analyzing BiP’s function in UPR signaling, despite not being appropriate for clinical usage. Recent pharmacological efforts aim to develop small molecules that either enhance or inhibit BiP’s chaperone activity or modulate its client protein interactions.19 Such targeted modulation may help restore ER homeostasis during early disease stages or limit detrimental effects during prolonged stress.20 Supporting this approach, In HeLa cells, both tunicamycin and thapsigargin were shown to increase BiP expression and activate the unfolded protein response, providing mechanistic insight into ER stress induction.21 In hematopoietic stem cells, tunicamycin was found to induce apoptosis through PERK–ATF4–CHOP signaling, highlighting its utility in modeling early UPR-mediated decisions.22Chemical modulators of HSP70 family chaperones, including BiP, were identified to act through ATPase domain interactions, thereby laying the groundwork for drug development targeting ER stress..23 Tunicamycin was also shown to enhance ER stress and apoptosis in drug-resistant gastric cancer cells, reinforcing its relevance in studying chronic stress and BiP dynamics..24 All of these investigations highlight how important it is to precisely regulate BiP activity in both healthy and diseased settings. Parkinson’s disease and other neurodegenerative diseases may benefit from new therapeutic approaches that involve modifying BiP and the UPR.
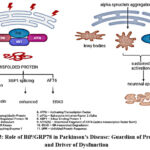 |
Figure 3: Role of BiP/GRP78 in Parkinson’s Disease: Guardian of Proteostasis and Driver of Dysfunction |
The PERK–eIF2α–CHOP Pathway and BiP’s Role in Cell Fate Regulation
The PERK–eIF2α–CHOP signaling axis, which is essential for determining cellular fate under chronic ER stress, is regulated by BiP/GRP78. Prolonged production of the pro-apoptotic transcription factor C/EBP homologous protein (CHOP) in this pathway results in programmed cell death. By stabilizing upstream components, BiP helps control this pathway, delaying or reducing CHOP induction and averting premature apoptosis.25 Beyond its role in ER stress, BiP also interacts with mitochondrial apoptotic machinery. It has been shown to associate with Bcl-2 family proteins and suppress caspase activation, particularly caspase-3. Elevated BiP levels inhibit caspase-3 activation, protecting neurons in PD models. In contrast, reduced BiP expression sensitizes cells to apoptotic signals. However, BiP’s protective functions are context-dependent. Persistent overexpression can maintain chronic UPR activation, leading to heightened neuronal vulnerability and enhanced apoptotic signaling. This has been clearly illustrated in FIG 3.26 Therefore, therapeutic strategies involving BiP must account for dosage and timing to avoid detrimental effects. BiP was similarly shown to attenuate mitochondrial apoptosis by modulating Bcl-2 and caspase activity, thereby preventing dopaminergic neuron loss in Parkinson’s disease models 27 They advocate for precision in both the timing and extent of BiP modulation for neuroprotection.28
Therapeutic Potential and Challenges
Given its dual role in promoting ER homeostasis and neuronal survival, BiP is a promising therapeutic target for Parkinson’s disease. Modulating its activity could reduce ER stress, restore proteostasis, and potentially slow or prevent neurodegeneration. However, several key challenges must be addressed.One major limitation is the difficulty of delivering BiP-modulating agents across the blood–brain barrier (BBB). Effective therapeutic strategies require delivery systems—such as targeted gene therapies or BBB-permeable small molecules—that ensure adequate drug concentration in affected brain regions.29Moreover, combining BiP activation with other therapeutic targets, such as alpha-synuclein aggregates or mitochondrial dysfunction, could enhance treatment efficacy. Still, systemic UPR modulation may inadvertently disrupt ER functions in peripheral tissues. Chronic or non-specific BiP activation may lead to unintended side effects, including UPR dysregulation in organs such as the liver or pancreas.30 Therefore, therapeutic approaches must be tightly regulated in both spatial and temporal dimensions. BiP has shown promise in maintaining ER integrity and supporting neuron viability.Yet, delivery remains a significant hurdle. Gene therapy and rationally designed molecules that penetrate the BBB are currently under investigation as viable delivery strategies.31 While boosting BiP could support alpha-synuclein clearance and mitochondrial health, caution is warranted to avoid off-target UPR activation. Thus, targeted, brain-specific delivery systems are essential for maximizing benefit while minimizing risk.
Future Directions
Future therapeutic strategies for Parkinson’s disease may increasingly rely on compounds that selectively modulate endoplasmic reticulum (ER) chaperones such as BiP, with the ability to cross the blood–brain barrier and act specifically at sites of pathology. One promising avenue involves modifying BiP’s interaction with unfolded protein response (UPR) sensors including PERK, IRE1, and ATF6—to fine-tune cellular stress signaling. Combining BiP-targeted interventions with autophagy enhancers could further promote α-synuclein clearance and restore proteostasis. A multi-target therapeutic approach has been proposed in the literature, suggesting that simultaneous modulation of UPR and autophagy pathways may synergistically enhance neuronal survival. Beyond therapeutic intervention, BiP expression itself could serve as a biomarker for patient stratification and monitoring of treatment response, aligning with precision medicine goals. Incorporating genomic profiling into clinical practice further strengthens this strategy by identifying individuals most likely to benefit from BiP-modulating therapies. It has been emphasized that integrating genomic data into clinical decision-making enables the development of personalized UPR-targeting strategies tailored to each patient’s ER stress profile and BiP expression patterns. Collectively, these insights underscore the emergence of precision medicine frameworks that combine molecular diagnostics with targeted interventions to achieve improved therapeutic outcomes in neurodegenerative diseases.
Conclusion
The ER stress response, particularly the UPR, plays a fundamental role in Parkinson’s disease pathogenesis, with BiP/GRP78 at the center of this process. BiP initiates UPR signaling and governs protein folding, determining whether neurons adapt or undergo apoptosis. While its early upregulation offers neuroprotection, prolonged elevation can become maladaptive.BiP modulation holds considerable therapeutic promise, but its application requires careful consideration of dosage, delivery, and systemic impact. To translate preclinical findings into clinical success, advancements are needed in small molecule design, gene therapy vectors, and brain-targeted delivery technologies. With continued research and optimization, BiP-targeted interventions may become a cornerstone of personalized treatment strategies for Parkinson’s disease.
Acknowledgement
The authors would like to express their sincere gratitude to Saveetha college of Pharmacy for providing the necessary resources and support for this review.
Funding source
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest
The author(s) do not have any conflict of interest
Data Availability
This statement does not apply to this article
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Authors contribution
- Renuka Devi J: Writing-Original draft Conceptualization, Supervision.
- Jayasri Bantaram:Writing-Review & Editing
- Shakthi Harikrishnan: Writing-Review & Editing
- Sandhya Ravishankar: Writing – Review & Editing.
References:
- K.Pavithra P, Rhea A, M.Dhanraj D, Pravinya P P. Prosthodontic management of a patient with Parkinson’s disease- A case report. J Young Pharm. 2018;10(3):377-379.
CrossRef - Lakshmipriya T, Gopinath SCB, Chen Y, Sasidharan S, Salim ET, Fakhri MA. Neurological inflammation in parkinsonism: Current prognosticative diagnostics and pitfalls. Curr Top Med Chem. Published online June 4, 2025. doi:10.2174/0115680266365134250530063901
CrossRef - Kouli A, John van Geest Centre for Brain Repair, Department of Clinical Neurosciences, University of Cambridge, UK, Torsney KM, et al. Parkinson’s Disease: Etiology, Neuropathology, and Pathogenesis. In: Parkinson’s Disease: Pathogenesis and Clinical Aspects. Codon Publications; 2018:3-26.
CrossRef - Srinivasan E, Chandrasekhar G, Chandrasekar P, et al. Alpha-synuclein aggregation in Parkinson’s disease. Front Med (Lausanne). 2021;8:736978.
CrossRef - Ranxhi B, Bangash ZR, Chbihi ZM, et al. Regulation of polyamine interconversion enzymes affects α-Synuclein levels and toxicity in a Drosophila model of Parkinson’s Disease. NPJ Parkinsons Dis. 2025;11(1):231.
CrossRef - Thangaraj S, Sun J. Ocean warming and acidification affect the transitional C:N:P ratio and macromolecular accumulation in the harmful raphidophyte Heterosigma akashiwo. Commun Biol. 2023;6(1):151.
CrossRef - Almanza A, Carlesso A, Chintha C, et al. Endoplasmic reticulum stress signalling – from basic mechanisms to clinical applications. FEBS J. 2019;286(2):241-278.
CrossRef - Lee AS. The ER chaperone and signaling regulator GRP78/BiP as a monitor of endoplasmic reticulum stress. Methods. 2005;35(4):373-381.
CrossRef - Pobre KFR, Poet GJ, Hendershot LM. The endoplasmic reticulum (ER) chaperone BiP is a master regulator of ER functions: Getting by with a little help from ERdj friends. J Biol Chem. 2019;294(6):2098-2108.
CrossRef - Bravo R, Parra V, Gatica D, et al. Endoplasmic reticulum and the unfolded protein response: dynamics and metabolic integration. Int Rev Cell Mol Biol. 2013;301:215-290.
CrossRef - Oslowski CM, Urano F. Measuring ER stress and the unfolded protein response using mammalian tissue culture system. Methods Enzymol. 2011;490:71-92.
CrossRef - Costa CA da, Manaa WE, Duplan E, Checler F. The endoplasmic reticulum stress/unfolded protein response and their contributions to Parkinson’s disease physiopathology. Cells. 2020;9(11):2495.
CrossRef - Ren H, Zhai W, Lu X, Wang G. The cross-links of endoplasmic reticulum stress, autophagy, and neurodegeneration in Parkinson’s disease. Front Aging Neurosci. 2021;13:691881.
CrossRef - Matsuoka M, Komoike Y. Experimental evidence shows salubrinal, an eIF2α dephosphorylation inhibitor, reduces xenotoxicant-induced cellular damage. Int J Mol Sci. 2015;16(7):16275-16287.
CrossRef - Hosoi T, Nomura J, Ozawa K, Nishi A, Nomura Y. Possible involvement of endoplasmic reticulum stress in the pathogenesis of Alzheimer’s disease. Endoplasmic Reticulum Stress Dis. 2015;2(1). doi:10.1515/ersc-2015-0008
CrossRef - López-Hernández B, Ceña V, Posadas I. The endoplasmic reticulum stress and the HIF-1 signalling pathways are involved in the neuronal damage caused by chemical hypoxia. Br J Pharmacol. 2015;172(11):2838-2851.
CrossRef - Sidrauski C, McGeachy AM, Ingolia NT, Walter P. The small molecule ISRIB reverses the effects of eIF2α phosphorylation on translation and stress granule assembly. Elife. 2015;4. doi:10.7554/eLife.05033
CrossRef - Shi M, Chai Y, Zhang J, Chen X. Endoplasmic reticulum stress-associated neuronal death and innate immune response in neurological diseases. Front Immunol. 2021;12:794580.
CrossRef - Abdullahi A, Stanojcic M, Parousis A, Patsouris D, Jeschke MG. Modeling acute ER stress in vivo and in vitro. Shock. 2017;47(4):506-513.
CrossRef - Shenkman M, Eiger H, Lederkremer GZ. Genesis of ER stress in Huntington’s disease. Endoplasmic Reticulum Stress Dis. 2015;2(1). doi:10.1515/ersc-2015-0007
CrossRef - Dibdiakova K, Saksonova S, Pilchova I, Klacanova K, Tatarkova Z, Racay P. Both thapsigargin- and tunicamycin-induced endoplasmic reticulum stress increases expression of Hrd1 in IRE1-dependent fashion. Neurol Res. 2019;41(2):177-188.
CrossRef - van Galen P, Kreso A, Mbong N, et al. The unfolded protein response governs integrity of the haematopoietic stem-cell pool during stress. Nature. 2014;510(7504):268-272.
CrossRef - Radons J. The human HSP70 family of chaperones: where do we stand? Cell Stress Chaperones. 2016;21(3):379-404.
CrossRef - Wu J, Chen S, Liu H, et al. Tunicamycin specifically aggravates ER stress and overcomes chemoresistance in multidrug-resistant gastric cancer cells by inhibiting N-glycosylation. J Exp Clin Cancer Res. 2018;37(1):272.
CrossRef - Rozpedek W, Pytel D, Mucha B, Leszczynska H, Diehl JA, Majsterek I. The role of the PERK/eIF2α/ATF4/CHOP signaling pathway in tumor progression during Endoplasmic Reticulum stress. Curr Mol Med. 2016;16(6):533-544.
CrossRef - Pihán P, Carreras-Sureda A, Hetz C. BCL-2 family: integrating stress responses at the ER to control cell demise. Cell Death Differ. 2017;24(9):1478-1487.
CrossRef - Oyadomari S, Koizumi A, Takeda K, et al. Targeted disruption of the Chop gene delays endoplasmic reticulum stress–mediated diabetes. J Clin Invest. 2002;109(4):525-532.
CrossRef - Hetz C, Mollereau B. Disturbance of endoplasmic reticulum proteostasis in neurodegenerative diseases. Nat Rev Neurosci. 2014;15(4):233-249.
CrossRef - Wang D, Qu S, Zhang Z, et al. Strategies targeting endoplasmic reticulum stress to improve Parkinson’s disease. Front Pharmacol. 2023;14:1288894.
CrossRef - Risiglione P, Zinghirino F, Di Rosa MC, Magrì A, Messina A. Alpha-Synuclein and mitochondrial dysfunction in Parkinson’s disease: The emerging role of VDAC. Biomolecules. 2021;11(5):718.
CrossRef - Ma C, Liu Y, Fu Z. Implications of endoplasmic reticulum stress and autophagy in aging and cardiovascular diseases. Front Pharmacol. 2024;15:1413853.
CrossRef







