Manuscript accepted on :06-10-2025
Published online on: 27-10-2025
Plagiarism Check: Yes
Reviewed by: Dr. Karthikeyan
Second Review by: Dr. Anton Sumarpo
Final Approval by: Dr. Kapil Joshi
Bhawna Chhabra1![]() , Malvika Kandpal5
, Malvika Kandpal5 , Prince Raj2
, Prince Raj2 , Arkan2
, Arkan2 , Komal Kumari2
, Komal Kumari2 , Pushpanjali Kumari2
, Pushpanjali Kumari2 , Pankaj Pant4
, Pankaj Pant4 , Vikash Jakhmola3
, Vikash Jakhmola3 and Mansi Butola2
and Mansi Butola2
1Department of Pharmacology, Amity Institute of Pharmacy, Amity University, Noida, Uttar Pradesh, India
2Department of Pharmaceutics, Uttaranchal Institute of Pharmaceutical Sciences, Uttaranchal University, Dehradun, Uttarakhand, India
3Department of Pharmaceutical Chemistry, Uttaranchal Institute of Pharmaceutical Sciences, Uttaranchal University, Dehradun, Uttarakhand, India
4Department of Pharmacology, Faculty of Pharmacy, DIT University, Dehradun, Uttarakhand, India
5Department of Education, Shri Guru Ram Rai University, Dehradun, Uttarakhand, India
Corresponding Author E-mail:mansibutola1995@gmail.com
Abstract
The field of computer science known as artificial intelligence (AI) is concerned with simulating human intellect through the use of machines that can replicate the human brain's capacity for problem-solving and decision-making. The scientific study of the brain's structure and cognitive processes is known as neuroscience. AI and neurology have a reciprocal interaction. These two fields benefit from one other's advancements. The notion of neuroscience has brought to a number of novel advancements in the realm of artificial intelligence. Complex deep neural network architectures have been developed because of the biological neural network, which are used to build a variety of applications, including object detection, speech recognition, and text processing. The validation of the current AI-based models is also aided by neuroscience. Artificial systems can now autonomously perform complex tasks because to the development of algorithms that mimic the idea of reinforcement learning, which has been seen in both humans and animals. These algorithms have been applied in various advanced technologies, including surgical robotics, self-driving cars, and intelligent gaming systems. At the same time, AI has proven highly effective in processing complex neurological data by identifying underlying patterns. Large-scale AI simulations offer valuable support to neuroscientists by allowing them to test scientific theories. Through brain-machine interfaces, AI can decode brain signals and convert them into actions, such as controlling robotic limbs to assist individuals with motor impairments. Moreover, AI significantly contributes to the interpretation of neuroimaging data, reducing the burden on radiologists and enhancing diagnostic capabilities. Early identification of brain-related problems is greatly aided by neuroscience, and AI is also being utilized more and more to predict and identify these conditions. This review explores the interconnected roles of AI and neuroscience, highlighting their collaborative potential in diagnosing and predicting neurological diseases, also the opportunities and limitations of these technologies.
Keywords
Algorithms; Artificial intelligence; Human intelligence; Neurosciences; Surgical robotics
| Copy the following to cite this article: Chhabra B, Kandpal M, Raj P, Arkan A, Kumari K, Kumari P, Pant P, Jakhmola V, Butola M. A Scoping Review on the Future of Neurological Disorder Diagnosis: Artificial Intelligence and Machine Learning. Biomed Pharmacol J 2025;18(October Spl Edition). |
| Copy the following to cite this URL: Chhabra B, Kandpal M, Raj P, Arkan A, Kumari K, Kumari P, Pant P, Jakhmola V, Butola M. A Scoping Review on the Future of Neurological Disorder Diagnosis: Artificial Intelligence and Machine Learning. Biomed Pharmacol J 2025;18(October Spl Edition). Available from: https://bit.ly/47klv7P |
Introduction
Neurological diseases are the primary cause of disability and the second greatest cause of death globally. As neurological diseases’ fatalities and impairments are recognized as a global public health concern, the burden of these conditions will increase over the ensuing decades. Over the past 30 years, there has been a 39% increase in the total number of deaths and a 15% increase in disability-adjusted life years, despite a drop in communicable neurological disorders. LMICs, or low- and middle-income countries, are disproportionately affected. As to the latest estimates, neurological illnesses that are part of the Global Burden of Disease (GBD) Study account for 3% of the global disease burden. These conditions include headache disorders such migraine, tension-type headache [TTH], and medication-overuse headache [MOH], as well as Parkinson’s disease, multiple sclerosis, epilepsy, Alzheimer’s, and other dementias. Despite the relatively low total percentage, the top 50 causes of disability-adjusted life years (DALYs) include dementia, epilepsy, migraine, and stroke.1
The five most prevalent neurological conditions were idiopathic epilepsy (0.4 [95% UI, 0.3-0.6] million people), AD and other dementias (2.9 [95% UI, 2.6-3.2] million people), stroke (7.8 [95% UI, 7.4-8.2] million people), TTH (121.6 [95% UI, 110-133] million people), and migraine (68.5 [95% UI, 64-73] million people). According to DALYs, the most incapacitating illnesses were stroke (3.6 [95% UI, 3.3-3.9] million people), AD and other dementias (2.6 [95% UI, 2.4 [2.7] million people), migraine (2.4 [95% UI, 1.5-3.4] million people), and Parkinson’s disease (0.4 [95% UI, 0.3-0.4] million people). The leading causes of mortality were AD and other dementias (258 600 [95% UI, 254 000-263 000], stroke (172 000 [95% UI, 166 000-178 000]), Parkinson’s disease (30 000 [95% UI, 24 000-31 000]), multiple sclerosis (4000 [95% UI, 3000-4000]), and MND (8400 [95% UI, 8000-9000]). Following AD and other dementias (0.48 [0.47-0.57] million cases annually), migraine (5.0 [95% UI, 4.6-5.5] million cases annually), TBI (0.96 [95% UI, 0.8-1.2] million cases annually), and stroke (0.60 [95% UI, 0.55-0.65] million cases annually), the highest incidence was for new-onset TTH (44.5 [95% UI, 40.0-48.8] million cases annually). Despite historical awareness of the need to include sex as a variable in studies—emphasized by the US National Institute of Mental Health in 2011 and previously by the Institute of Medicine in 2001—the gaps remain considerable in appreciating sex differences across neurological diseases. These exist in epidemiology, clinical presentation, and outcome from treatment, especially in epilepsy, multiple sclerosis, migraine, and stroke. Critical issues involve a lack of sex-specific data, sparse longitudinal and interdisciplinary research, and underrepresentation of men in certain trials. Such a dearth of evidence compromises successful clinical management, particularly in situations such as pregnancy and hormonal interactions where women are at greater risk. Recommendations and registries exist but are fragmented or incomplete, and there is a critical need for thorough, sex-informed research to enhance neurological treatment of both sexes.2
Historically, neurological disorder diagnosis has depended considerably on clinical examination, consisting of an extensive patient history and a thorough neurological examination. Doctors evaluate signs and symptoms such as headaches, seizures, numbness, weakness, cognitive change, or movement abnormalities. Diagnostic techniques like electroencephalography (EEG), electromyography (EMG), and imaging tests like computed tomography (CT) scans and magnetic resonance imaging (MRI) are also used to identify structural or functional problems in the brain and nervous system. The diagnosis of infection, inflammatory disease, or metabolic disease can be made using blood tests and lumbar puncture study of cerebrospinal fluid. Even with these instruments, standard diagnostic procedures have not always taken sex differences into consideration, which may result in misdiagnosis or delayed diagnosis in some groups.3 Conventional management of neurological illnesses tends to be oriented towards alleviation of symptoms and deceleration of disease progression, mostly with the help of pharmacological treatments. Antiepileptics, precursors to dopamine for Parkinson’s disease, corticosteroids in multiple sclerosis, and analgesics for migraines are common forms of treatment. Occasionally, surgical techniques are employed, such as deep brain stimulation for Parkinson’s illness or epilepsy surgery. Rehabilitation treatments such as physical, occupational, and speech therapy are also important in treating chronic neurological diseases. Treatment interventions have in the past been guided by generalized clinical trials that did not consider sex-specific responses to therapies, resulting in less personalized and possibly less effective treatment for both men and women. Conventional diagnosis and treatment of neurological disease are commonly dependent on symptomatic and imaging features, which will not pick up early or subtle changes. They can result in delayed or misleading diagnoses. Treatment strategies are often generalized and are not individualized according to unique patient biology. They might not target the fundamental causes of intricate neurological diseases.
Fundamentals of AI and ML in Healthcare
Artificial intelligence (AI) aims to replicate human cognitive processes. It is causing a paradigm shift in the medical industry. Recent successful applications of AI in the health industry have been made possible by the abundance of health-related data and the rapid advancement of big data analytical tools. When a relevant clinical query is in mind, powerful AI approaches can reveal clinically significant information concealed in the vast volume of data. Clinical decision-making may then benefit from this,4 AI may “learn” characteristics from enormous volumes of medical data using complex algorithms, then apply what it has discovered to support clinical practice. It can also be set up with learning and self-correcting features to increase its accuracy based on experience. An AI system can help doctors by giving them the most recent medical data from clinical procedures, textbooks, and journals so they can deliver the right patient treatment. Furthermore, an AI system might lessen the inevitable therapeutic and diagnostic errors that occur during human healthcare procedures. Additionally, an AI system can collect valuable information from a sizable patient base to support real-time findings for health risk warnings and outcome prediction.5
Because AI and ML enable faster, more accurate, and data-driven decision-making in healthcare, they are revolutionizing disease detection. AI and ML can examine vast amounts of medical data, such as genetic data, imaging scans, and patient records, to identify patterns and abnormalities that may be suggestive of certain diseases. AI-based solutions increasingly find application in radiology, pathology, and dermatology to aid physicians in identifying diseases like cancer, diabetes, and cardiovascular diseases at an earlier stage. ML algorithms become more refined as they gain more data, with enhanced predictive power and assisting in tailored treatment planning. In general, AI and ML are transforming disease diagnosis by maximizing efficiency, minimizing human error, and enabling early intervention. Particularly in the diagnostic phase, a sizable amount of the AI literature examines data from genetic testing, electrodiagnosis, and diagnostic imaging. For example, while analyzing diagnostic images with a lot of data, Jha and Topol urged radiologists to adopt AI technologies. The use of aberrant long non-coding RNA genetic expression in the diagnosis of gastric cancer was investigated by Li et al.6 AI gadgets come in two primary varieties. The first group consists of machine learning (ML) methods that work with structured data, including genomic, imaging, and EP data. Machine learning algorithms are used in medical applications to classify patient characteristics or determine the likelihood of illness consequences.7 The second-class extracts information from unstructured clinical notes and medical publications using natural language processing (NLP) approaches to support and enhance organized medical data. NLP processes aim to convert texts into machine-readable, structured data so that machine learning algorithms can process them. Despite the expanding literature on AI in healthcare, the study primarily focuses on a few disease types: cardiovascular diseases, nervous system problems, and cancer. In a double-blind validation research, Somashekhar et al. showed that IBM Watson for oncology would be a trustworthy AI tool to aid in cancer identification.8 Esteva et al. identified subgroups of skin cancer by analyzing clinical pictures.9 An AI system was created by Bouton et al. to help quadriplegics regain control over their movements.10 Farina et al. evaluated the effectiveness of an offline man-machine interface for controlling upper-limb prostheses using the spinal motor neurons’ discharge timings.11 The Arterys Cardio DL application, which uses artificial intelligence (AI) to automatically generate editable ventricular segmentations from conventional cardiac MRI images, has been approved for sale by the US Food and Drug Administration (FDA).12 In therapy, AI and ML are employed to create individualized treatment plans by examining unique patient information, forecasting treatment outcomes, and recommending the most advantageous therapies. They also aid in drug research, robotic surgery, and continual monitoring of patient health with wearable technology. With AI and ML combined, healthcare is becoming increasingly specific, effective, and customized to meet individual requirements, ultimately enhancing patient results, and minimizing costs. Table 1 highlights the application of AI in healthcare.
Table 1: AI Applications in Healthcare
| MEDICAL FIELD | AI APPLICATION | EXAMPLES | REF. |
| Radiology | Image recognition Emergency alert systems |
Intracranial bleeding detection (MaxQ-AI) Vertebral fracture detection (Zebra Medical Vision) |
13 |
| Oncology | Cancer detection & staging treatment recommendation | Watson achieved 93% concordance with tumor board in breast cancer treatment | 14 |
| Pathology | Automated slide analysis, Cancer cell identification | Faster analysis with high accuracy compared to pathologists | 15 |
| Cardiology | ECG & echo analysis, Heart failure prediction Procedural decision support |
Early detection of heart failure via EMR patterns, better prediction of treatment outcome for angina | 16 |
| Gastroenterology | Polyp & cancer detection during endoscopy | CADe/CADx systems: 94% detection rate for colonic polyps; 96.3% precision in early gastric cancer detection | 17 |
| Ophthalmology | Detection of diabetic retinopathy from retinal images | Comparable performance to ophthalmologists using deep learning | 18 |
| Neurology | Diagnosis support from RNA-protein expression data | Identification of RNA-binding proteins in ALS (IBM Watson) | 19 |
| General Medicine (EMR) | Diagnosis aid from patient records Pattern recognition |
Hyperparathyroidism detection with 97% accuracy from lab data General enhancement in diagnosis and treatment via integrated EMR analytics |
20 |
| Dermatology | Skin lesion classification from images | AI matched expert dermatologists in detecting melanomas and keratoses | 21 |
| Surgery | workflow optimization Surgical assistance Instrument tracking |
Potential for autonomous assistance Systems like OR.NET for integrated device communication |
22 |
| Anesthesiology | Monitoring during sedation Oxygenation and ventilation support |
Systems like SEDASYS for improved anesthesia management | 23 |
| Cloud-Based AI | Remote algorithm access Continuous updates across specialties |
Zebra Medical, Arterys, VIDA Diagnostics offering cloud-based diagnostics for lung, heart, liver, and bone health | 24 |
Current AI and ML Applications in Neurological Disorder Diagnosis
The recursive link between AI and neurology has been the subject of some reviews during the past few years. the authors carried out a review that focused on how neuroscience advances AI research and how AI may be used to advance neuroscience. 25 The authors talked about how neuroscience could inspire new kinds of algorithms and be used to test AI techniques. The investigators of,26, discussed the history of the connection between AI and neurology as well as the most recent advancements in four fields: computational psychiatry, AI big neuroscience data analysis, AI working memory models, and AI processing of visual information. In,27, The authors discussed the sharing relationship between AI and neuroscience by emphasizing the links between biological vision and AI vision networks. Fig 1 provides representation of AI in neurological disease diagnosis.
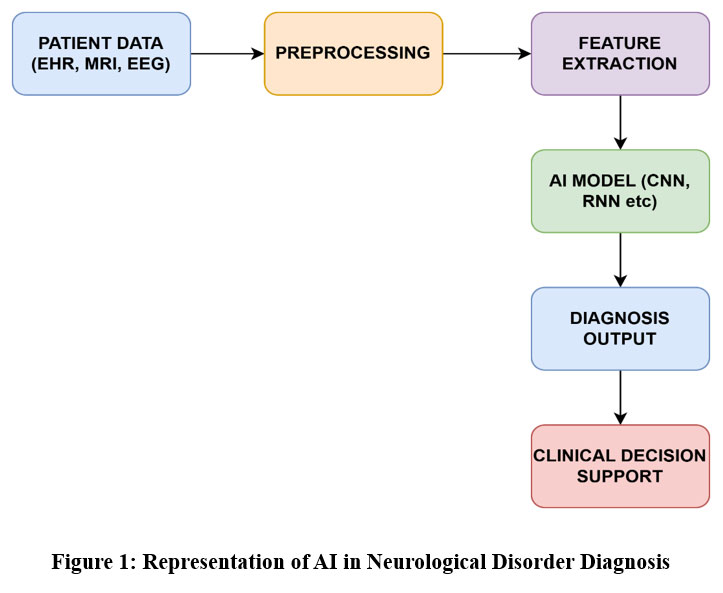 |
Figure 1: Representation of AI in Neurological Disorder DiagnosisClick here to view Figure |
Deep learning allows machines to directly process raw data, which facilitates the automatic identification of key characteristics for tasks such as speech recognition and object detection. Artificial intelligence (AI) and neuroscience have a particularly beneficial relationship. Two main ways that neuroscience contributes significantly to AI are by guiding the creation of various artificial neural network (ANN) topologies and associated learning algorithms. Secondly, by offering a framework for validating previously established AI models. The identification of an algorithm that is known to be used by the human brain gives credibility to its potential role in the development of more generalizable and comprehensive AI systems. The following sections look deeper into the impact of neuroscience on the development of AI. These models have been explained in Table 2.
Artificial neural networks (ANNs)
Computational models known as artificial neural networks these are modelled after the composition and operations of the human brain. Like Hebbian learning, they are made up of interconnected artificial neurons that send signals via weighted connections that are modified during learning to store knowledge. Frank Rosenblatt developed the perceptron in the 1950s with the help of the microscope, which made it easier to comprehend the architecture of neurons. To generate a binary output, a perceptron applies an activation function, also known as the binary step function, multiplies binary inputs by matching weights, and adds a bias. This is like how organic neurons fire. However, sigmoid neurons with continuous input values and a sigmoid activation function are utilized for smoother transitions to prevent sudden changes in output. To successfully classify inputs, the perceptron is trained by varying weights and biases until its output resembles the desired outcome. Fig 2 explains perceptron classifier inspired by neuroscience.
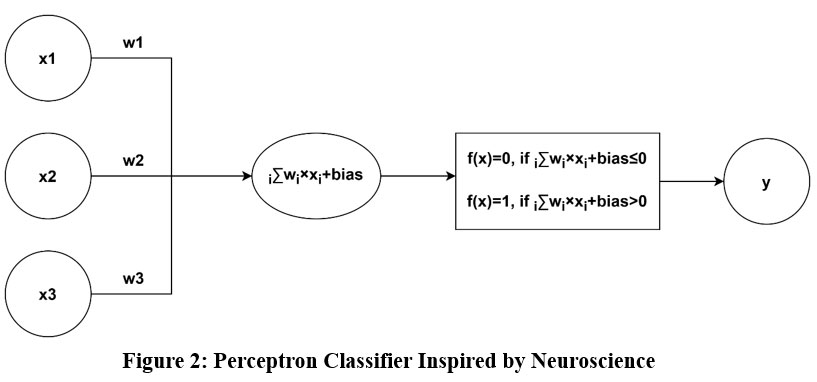 |
Figure 2: Perceptron Classifier Inspired by NeuroscienceClick here to view Figure |
Multilayer Perceptron (MLP)
MLP, which is made up of several hidden layers of neurons, was developed in response to the single-layer perceptron’s limitations in addressing non-linearly separable problems like the XOR function. The output of one layer in an MLP is used as the input for the subsequent layer, and activation functions are employed to process the final outputs to get the desired results. Using gradient descent and the chain rule, the backpropagation learning algorithm is used to train MLPs. It computes outputs in a forward pass and calculates and propagates error for weight modification in a backward pass. This approach mimics the biological learning processes seen in cortical networks while simplifying training because it does not require fixed settings beyond inputs. The hidden layers need domain-specific setup to satisfy application requirements, although the input and output layer designs are simple.
Recurrent Neural Network (RNN)
Recurrent neural networks, or RNNs, are inspired by the human brain’s working memory, namely the constant neuronal activity in regions like the hippocampus, neocortex, and prefrontal cortex. Unlike feed-forward networks, which only use current inputs, RNNs process sequential data and produce context-aware predictions by using feedback loops to retain prior outputs in an internal memory. Standard RNNs, however, are only capable of short-term memory. Long Short-Term Memory (LSTM) networks were created to solve this problem. These networks use specialized memory cells and three different kinds of gates—input, output, and forget—that control the flow, retention, and disposal of data. Because of these improvements, LSTMs can capture long-term dependencies, which makes them useful for tasks like text summarization, video classification, and speech recognition. The midbrain attention network circuit architecture of the brain serves as an inspiration for the construction of RNNs and LSTMs. Recurrent neural network for future prediction with memory of previous output is explained in Fig 3.
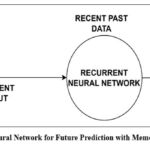 |
Figure 3: Recurrent Neural Network for Future Prediction with Memory of Previous Output.Click here to view Figure |
Convolutional Neural Network (CNNs)
With the development of GPUs and software, CNNs, a second-generation deep learning model, came into being. The ventral visual stream of the brain, which interprets visual information hierarchically—from basic elements like edges to complex objects—is the source of their inspiration. Convolutional, pooling, and fully linked are the three primary layers that CNNs use to imitate this structure. The convolutional layer uses elementwise multiplications throughout the image to apply filters (kernels) to extract features. By combining features using techniques like max or average pooling, the pooling layer lowers dimensionality. Lastly, for tasks like detection or classification, the fully linked layer polishes the data. In a manner like how the human brain processes visual stimuli from simple shapes to entire things, CNNs use layers of increasing complexity to convert raw visual inputs into high-level representations.
Reinforcement Learning (RL)
It draws inspiration from behavioural psychology and neuroscience, teaches an agent to make decisions by interacting with its surroundings and learning from mistakes. The agent improves its behaviour over time to optimize cumulative rewards by receiving rewards for right actions and penalties for errors. This learning is like biological processes where learning is driven by prediction errors between predicted and actual results, such Pavlovian and instrumental conditioning. RL in artificial intelligence uses a learning algorithm and a policy, which is a mapping from states to actions. The algorithm is updated by observed states, actions, and rewards. RL has been used in fields like as control systems, gaming, and robotics. Distributed reinforcement learning and other sophisticated RL models have been impacted by the dopaminergic neurons in the brain, which encode reward prediction mistakes. Fig 4 represents the AI reinforcement learning via trial and error.
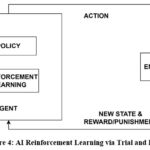 |
Figure 4: AI Reinforcement Learning via Trial and ErrorClick here to view Figure |
Deep Reinforcement Learning (DNNs)
Basic look-up tables can be used to express policies in RL for simple contexts. Such tables, however, are impracticable in complex settings, such as video games, and deep neural networks are used to model policies, allowing for gradient-descent-based nonlinear mappings between environmental conditions and potential actions. The Complementary Learning Systems (CLS) theory, which describes how the human brain integrates two learning processes—specific event-based learning by the hippocampus and generalization by the neocortex—is incorporated into contemporary deep reinforcement learning. The hippocampus stores quick experiences in episodic memory, while the neocortex subsequently incorporates them into long-term memory. Like this, deep RL models in AI combine instance-based and generalized learning to maximize decision-making by using neural networks to learn from successful instances and generalize from data.
Spiking Neural Network (SNN)
Although DNNs have made great strides in text, voice, and image recognition possible, their high processing overhead makes them less appropriate for situations with limited resources, such as edge devices. SNNs, which are modelled after biological neurons that only send messages when their membrane potential over a specific threshold, are being developed as a remedy. Like this, neurons in SNNs only “spike” or fire when they cross this threshold, as opposed to standard DNNs, which transmit signals continuously. SNNs are a possible substitute for modeling sophisticated information processing in energy-constrained settings because of their event-driven mechanism, which also makes them more energy-efficient and appropriate for low-power applications. Fig 5 represents the energy efficient spiking neural network.
 |
Figure 5: AI Spiking Neural Network with Low Energy Consumption Click here to view Figure |
Table 2: Various computational models used in AI
| MODEL | KEY FEATURE | REF. |
| Artificial neural networks | An ANN is made up of many interconnected components, or artificial neurons, that work together. Messages can be sent along the connections between many artificial neurons. | 28 |
| Multilayer Perceptron (MLP) | Each layer’s output in MLP serves as the input for the layer after it. This continues up to the buried layer and, at last, the final layer, where the appropriate activation function alters the values attained to produce the intended results. MLP is taught via the backpropagation learning approach, which necessitates two rounds through MLP. | 29 |
| Recurrent Neural Network (RNN) | RNNs, which store recent output in a memory structure, were developed in response to the human brain’s working memory. Only in respect to the current inputs can pre-RNN designs (such feed-forward networks) produce outputs. | 30 |
| Convolutional Neural Network | The advancement of high-performance computing hardware, graphics processing units (GPU), and software technologies enabled the second generation of deep neural networks, also known as convolutional neural networks (CNNs). | 31 |
| Reinforcement Learning (RL) | The agent learns to behave in the environment by acting in it during reinforcement learning. By using this technique, the agent itself learns to repeat tasks that have rewards and avoid those that have penalties. | 32 |
| Deep Reinforcement Learning | Current deep RL frequently uses the Complementary Learning Systems (CLS) hypothesis, which analogously predicts how the human brain reconciles generalization (through the neocortex) and learning through instances (through the hippocampus). | 33 |
| Spiking Neural Network (SNN) | Taking inspiration from biological neurons, SNNs fire only when a neuron’s membrane potential exceeds a threshold, as opposed to DNNs that send signals continuously. | 34 |
AI for Neuroscience Development
The ability of AI to examine vast volumes of intricate data and uncover hidden patterns within it is its main strength. Artificial intelligence is the most efficient means of gleaning patterns and conclusions from the complex inputs coming from the brain. High-performing artificial intelligence systems have been developed using the models that inform theories about how the brain works, as mentioned in. 35 By creating extensive models of the neurological processes that produce intelligence, artificial intelligence essentially makes it easier to research cognition processes. For instance, an IBM research team used the IBM Blue Gene CPU to simulate 8 × 106 neurons with 6400 synapses apiece. This could be a useful tool for neuroscience research. Before spending enormous quantities of money on actual animal research, neuroscientists can use this study tool to test their hypotheses and examine simulation outcomes.
BCI, or brain-computer interface, benefits from AI.
The Brain–Computer Interface (BCI) helps people with neuromuscular conditions like ALS, cerebral palsy, or spinal cord injuries by allowing direct connection between the brain and external equipment, including computers or robotic limbs. BCIs function by gathering brain signals using semi-invasive (like electrocorticography) or non-invasive (like EEG, MEG) techniques. These signals undergo preprocessing for feature extraction, amplification, digitization, and noise reduction. After the features are categorized by AI algorithms, external devices are controlled by the commands that are produced. Applications include AI-based prosthesis control and cursor control for typing, as demonstrated by the Brain Gate system. Patients with motor disabilities benefit greatly from the help of BCIs in regaining their ability to move and communicate.36
AI supports genetically based neuron analysis and stimulation studies
Finding cellular phenotypes that cause neurodegenerative disorders requires measuring gene expression in certain cell types. AI helps by enabling genomic-level neuron analysis and building simulated models, which provide deeper understanding of how impulses originate and travel through the brain. This makes it easier to identify the cellular phenotypes that cause disease. This strategy is demonstrated by initiatives such as the Blue Brain Project, which uses AI-powered simulations as virtual research instruments to investigate brain properties and their connections to cognitive processes. These initiatives promote the development of focused therapies and increase our understanding of brain illnesses.37,38
Artificial Intelligence Aids in Connectome Research
A connectome is an extensive map of the brain’s neuronal connections that has been examined using methods such as DMRI and fMRI. Connectome data are network-based, with brain areas represented as nodes connected by weighted edges that indicate structural or functional correlations, in contrast to conventional medical pictures with grid layouts. To properly assess the data due to its network nature, specific machine learning methods are needed. AI-powered connectome analysis has shown promise in the early identification of conditions such as autism, Parkinson’s, Alzheimer’s, and developmental abnormalities in premature babies. Additionally, it helps identify important brain connections and subnetworks involved in the course of disease and permits differential diagnosis of mild cognitive impairment.39
AI aids in the analysis of neuroimaging
By learning hierarchical feature compositions, artificial neural networks (ANNs) effectively analyze complicated, high-dimensional sensory data, simulating human cognitive processes. Deep neural networks have become extremely effective tools for automated image interpretation because of the radiological imaging data’s exponential increase beyond the number of radiologists accessible. These networks improve upstream processes like noise reduction, image quality improvement, and quicker image capture while matching radiologist-level accuracy in tasks like segmentation, identification, and diagnosis. Applications of AI in neuroimaging include increasing the speed of MRI scans using compressed sensing, optimizing radiation exposure in CT imaging, improving signal-to-noise ratios, and improving image reconstruction and registration accuracy. AI also makes it easier to create synthetic CT scans from MRIs, translate EEG data dynamically, evaluate MRI quality automatically, and use explainable AI to make clear decisions in neuroimaging.40 The demand for automated AI-based solutions in neuroimaging processes stems from the inefficiency and irregularity of manual processing due to the complexity and volume of MRI data. While deep learning models may directly analyze raw EEG time-series data for analytical purposes, artificial intelligence (AI) supports multistep processes including spatial realignment, coregistration, normalization, and smoothing of fMRI images. Deep learning is used by software programs like Brainvoyager, FreeSurfer, and sophisticated pipelines like Fastsurfer,41 and Fast-AID Brain to automate structural MRI analysis, resulting in precise, repeatable measurements and quick brain region segmentation. Large-scale research and clinical applications require accurate, scalable neuroimaging analysis, which these advancements make possible.
AI in the Research of Aging Brains
By examining structural MRI scans, CNNs can determine the age of a human brain; cerebrospinal fluid cavities have been found to be important characteristics that affect the prediction. One of the most important biomarkers for determining the risk of neurodegenerative illnesses is brain aging. When notable differences are discovered between the calculated biological brain age and the actual chronological age, AI models that create precise anatomical brain maps to capture certain aging patterns can assist in identifying those who are more likely to develop Alzheimer’s disease.42
Key Neurological Disorders and AL Based Diagnostics
The AI has been applied in the various neurological disorders diagnosis which have been discussed below:
AI in tumors
An MRI scan is the conventional procedure to find a brain tumor. Tumor detection, segmentation, and grade estimate are three crucial uses for machine and deep learning models applied to MRI data. The survey in,43, found four different categories of techniques for classifying and segmenting brain tumors: CNN-based methods, vision transformer-based methods, capsule neural network-based methods, and conventional machine learning methods. The texture information from MRI images has been extracted using a variety of feature extraction techniques, including first-order statistical feature extraction, histogram-oriented gradient, and gray-level co-occurrence matrix. Various machine learning techniques then use the texture information to identify tumors.44 A CNN-based multigrading classification system for brain tumors is introduced.45 Several deep CNNs with user-defined hyperactive values for brain tumor classification are shown in.46 The use of 3D CNNs for multiscale grade estimate is demonstrated.47 CNN is quite good at analyzing brain tumors, but its accuracy depends on picture rotation. Additionally, CNN required massive amounts of training data. These two constraints were avoided while using capsule neural network based techniques.48 Additionally, CNNs cannot extract long-range information that is image-sequence-dependent about time because of their lower kernel size, but vision transformer (ViT) neural networks can handle the sequences with ease.49 Fig 6 represents the AI in brain tumor diagnosis.
 |
Figure 6: AI for Brain Tumor DiagnosisClick here to view Figure |
Seizures with AI
Because of their unpredictable patterns, epileptic seizures are difficult to detect. They arise from an abrupt, aberrant spike in electrical activity in the brain. Machine learning was utilized for processing recordings of electroencephalography recordings for automatic seizure detection.50 In,51, Features were extracted from EEG data using one-dimensional local gradient pattern and local neighbor descriptive pattern approaches. An ANN was then used to process the derived features to identify seizures. Additionally, automatic seizure detection is discussed. 52,53 Unlike the procedures mentioned above,52,53 which use single-channel EEG waves to extract characteristics in,54, Using a two-dimensional deep convolution autoencoder in conjunction with a neural network, automatic feature extraction was carried out to categorize the recovered features and detect seizures in children.
AI and Developmental and Intellectual Disabilities
Attention-deficit/hyperactivity disorder, cerebral palsy, Down syndrome, autism spectrum disorders, fragile X syndrome, and other intellectual and developmental issues start to show themselves during a child’s maturation, usually before the age of 18. Multimodal data, including genetic, genomic, neuroimaging, electronic health record, clinical, and behaviour data (obtained by various approaches), are used as inputs in ID and DD analysis. A DNN processes the neuroimaging data to determine if children have ID or DD. By examining the pattern of functional connectivity in fMRI data, a DNN has identified the presence of schizophrenia (SCZ). To detect autism, the fMRI data was processed using an autoencoder-based DNN.55 In,56, EEG data was processed using an SVM algorithm to detect children with attention-deficit/hyperactivity disorders. Other medical records, such as those pertaining to the circulatory, endocrine, digestive, and genitourinary systems, are associated with fragile X syndrome. Considering this, screening techniques based on artificial intelligence have been created to examine individuals’ electronic medical information to detect fragile X syndrome.57 In,58, Based on sensitivity and specificity criteria, Random Forest fared better than K-nearest neighbor, SVM, backpropagation, and deep learning in the classification of autism spectrum disorders (ASDs) in children and adolescents. Targeted screenings for high-risk children are made possible by early ASD detection. Machine learning uses a toddler’s eye movements to detect the existence of ASDs. In addition, the diagnosis of ASDs using maternal auto-antibody-based biomarkers and blood is another use of machine learning.
Neurodegenerative Diseases and AI
The most common neurodegenerative diseases Motor neuron disease, AD, and PD provide significant challenges since symptoms do not appear until a significant number of neurons have been destroyed.59 Thus, early detection is difficult. Machine learning techniques can be used to analyze MRI pictures to detect the disorders early. In,60, To identify AD, a 3D neural network architecture was employed. People who suffer from moderate cognitive impairment (MCI) are particularly vulnerable to AD. In,61, To predict the change of MCI to AD one to three years before a clinical diagnosis, MRI data were analyzed using random forest. Huntington’s disease (HD), Parkinson’s disease (PD), and motor neuron disease (MND) are all linked to motor dysfunction. Early detection of Parkinson’s disease can be achieved with basic writing and line-drawing exercises.62 In,63, For the first time, the authors used machine learning algorithms with basic line sketching to help diagnose Parkinson’s disease. As explained,64 Individual features that were created using SVM weights as a significance map (p-map) allowed for an improvement in dementia classification. Neurodegenerative disorders like Parkinson’s disease (PD), Huntington’s disease (HD), and amyotrophic lateral sclerosis (ALS) are diagnosed by looking for irregularities in gait. Although gait disturbances can be analyzed using classic machine learning techniques like SVM, nearest neighbor, and naïve Bayesian algorithms, they are not very effective at capturing time-related information concerning gait disturbances. LSTM has proven to be a useful tool for analyzing gait problems.65 Fig 7 represents the role of AI in Alzheimer disease diagnosis.
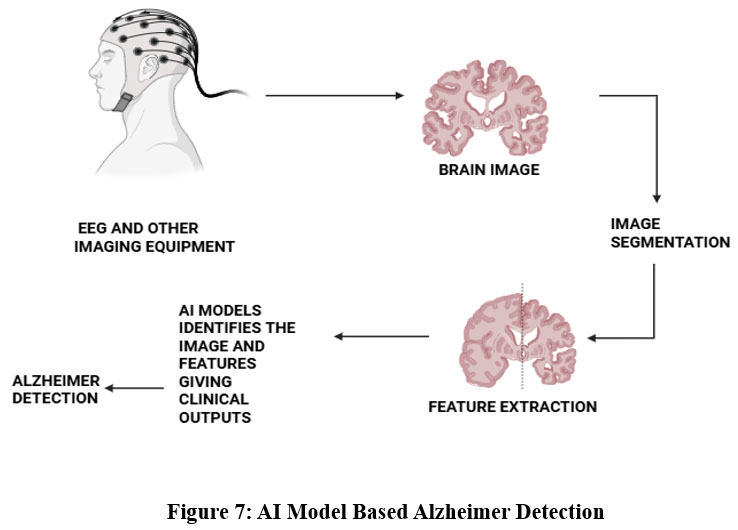 |
Figure 7: AI Model Based Alzheimer DetectionClick here to view Figure |
AI in Headaches
AI is being utilized to detect migraines.66 The participants’ self-report data was analyzed and classified into several headache categories, such as migraine, epicranial, tension type headache (TTH), trigeminal autonomic cephalalgia (TAC), and thunderclap headaches, using a four-layer XG Boost classifier.67 As mentioned,68 the classification of headaches is being done using both deep learning and machine learning techniques. Doctors, who usually have a busy schedule, should pay close attention to the symptoms and experiences of patients while diagnosing headaches. The study and categorization of headaches can greatly benefit from the application of machine learning and natural language processing, as demonstrated.69
Cerebrovascular Accidents and AI
A cerebrovascular accident, often known as a stroke, happens when there is a reduction or interruption in blood flow to the brain. The brain will not receive oxygen due to a diminished or disrupted blood flow, and brain cells will fall dormant. If treatment is not received within 3 to 4.5 hours of the beginning of symptoms, the affected persons may die or suffer lasting disability.70 Making an early diagnosis during an occurrence is essential. There are three forms of strokes: subarachnoid hemorrhage, hemorrhagic stroke, and ischemic stroke. Even experienced doctors can diagnose stroke incorrectly 20–45% of the time. Strokes can be categorized using a three-layer feed-forward ANN with a backpropagation error technique. It was discovered that AI methods for analysing stroke images were promising.71 By employing a basic support vector machine (SVM) technique to analyze MRI images, the study,72 was able to predict acute ischemic stroke with 95% accuracy. A random forest algorithm has been used in to determine who is most likely to develop malignant cerebral edema, the primary outcome of an ischemic stroke.73 In,74 ANN, SVM, random forest, and other machine learning techniques were used to determine the stroke’s subtype. In,75 It was discussed how to determine the severity of strokes using machine learning techniques.
AI in Neurological Infections
AI and ML have both shown promise in improving the identification and management of CNS disorders. AI-based techniques examine several indicators, including CSF and blood characteristics, in contrast to conventional methods that depend on a single variable. This allows them to more precisely distinguish between illnesses like bacterial and viral meningitis. By employing non-invasive data for early disease identification, AI can also lessen the necessity for intrusive procedures. For example, metabolomics in conjunction with machine learning approaches has been used to detect newborn sepsis with meningoencephalitis, and artificial neural networks (ANNs) have achieved high diagnostic accuracy using blood-based data. Applications of AI go beyond simple diagnoses. Deep learning models are used to analyze complex imaging data in diseases including autoimmune encephalitis and brain cancers to aid in prognosis and therapy planning. Moreover, machine learning algorithms aid in the interpretation of various patient data and the identification of risk factors for hospital-acquired infections in critical care settings. Because they provide quantitative insights into tumor properties that standard imaging cannot, radiomics and radiogenomics technologies have improved neuro-oncology.76 Notwithstanding advances in technology, brain infections still pose problems due to a lack of effective preventative measures, uncommon organisms that go undetected, and a lack of available treatment choices. More advanced, AI-driven diagnostic tools are desperately needed, as seen by the rise of novel infectious diseases brought on by population increase, environmental changes, and international travel. These technologies are essential in contemporary neurology since they not only increase early identification and lower mortality but also aid in preventing long-term neurological problems.
Numerous instances demonstrate how AI can be used practically to diagnose viral brain illnesses. For example, a study that employed an ANN to identify meningitis based on blood and temperature characteristics had a 96.67% accuracy rate, potentially providing a substitute for invasive lumbar punctures. AI has been used to analyze metabolomics data in neonatal care to differentiate between sepsis cases with and without meningoencephalitis, improving early management. Using CT and MRI characteristics, deep convolutional neural networks (CNNs) have been used to distinguish between cystic gliomas and bacterial brain abscesses. Furthermore, by identifying important risk variables for hospital-acquired infections in neurological intensive care units, ensemble models such as XGBoost have improved patient monitoring and outcomes.77 These illustrations highlight AI’s adaptability and efficiency in handling the intricacies of CNS illness diagnosis.
Challenges and Limitations
A promising basis for the development of intelligent applications that can anticipate and diagnose neurological illnesses has been established by the fusion of artificial intelligence and neuroscience. However, putting such applications into practice has been linked to several difficulties, which also influence future research avenues.
Problems with Creating Interconnected Datasets Because of Isolated Teams’ Working Culture
Although neuroscience has enormous potential to improve computing, medicine, and our knowledge of cognition, it needs to move from solitary, small-scale endeavors to extensive, interdisciplinary partnerships. Now, distinct study groups concentrate on tasks while working alone with different animals, including mice, rats, cats, primates, and humans. Developing integrated datasets that provide a comprehensive understanding of brain architecture and functioning is the primary problem. These interconnected datasets can be produced by combining AI, neurology, and systems biology through sizable, varied teams. Future advancements in neuroscience will be fueled by this interdisciplinary approach, which will make predictive modeling and big data simulations possible.
Difficulties with Deep Understanding in Neuro-Inspired AI
While neuro-inspired AI concentrates on tasks where people perform better than computers, neuroscience aims to employ large-scale, high-resolution data to identify disease loci for possible remedies. Experts in AI usually create computational solutions without completely comprehending the underlying biological mechanisms. This makes figuring out the right level of comprehension extremely difficult for AI-inspired neuroscience. Many of the methods in use today rely on high-level abstractions, including black-box deep neural networks, which can work well for implementation but are challenging to understand and evaluate.
Challenges in Deciphering and Assessing AI-Powered Solutions
Even if AI-based solutions have enormous potential, they must pass stringent testing before they can be used in real-world scenarios. Where the current solutions fall short, the adopted solutions must demonstrate their results. The idea of explainable AI is developing in this area. Explainable A-based interpretations for the prediction-making process can be adopted by the AI-based solutions. AI models can be coupled with certain software tools. Doctors and neuroscience specialists will have more faith in AI-based solutions because of this empowerment.
Issues with Regulations and Standards
The lack of regulatory standards to evaluate the effectiveness and safety of these systems is one of the biggest obstacles to putting AI-based solutions into practice. The U.S. FDA has started working on guidelines for evaluating AI technologies to solve this. Furthermore, data sharing—which is necessary for creating reliable and complete AI systems—is hampered by the current healthcare environment. Notwithstanding these obstacles, a revolution in healthcare that aims to promote data sharing is gathering steam in the US, indicating a move toward more data-driven and collaborative innovation.
Challenges in Methodology and Ethics
Across a variety of data types, AI-based algorithms exhibit enormous potential in identifying early indicators of neurological illnesses; yet, there are substantial obstacles to its clinical implementation. The possible failure or unpredictability of AI systems is one methodological and epistemic worry; problems like over-fitting or over-optimization might provide biased results. There are also ethical conundrums: it is still unclear if AI should make decisions for doctors or work independently. Additionally, even while these technologies might help those with cognitive and motor impairments live better lives, their independence may limit their users’ cognitive freedom.78 Regardless matter how sensitive the results are, transparency in AI outputs presents additional ethical issues, particularly when it comes to high-risk forecasts. Furthermore, the training datasets must be representative; a lack of diversity can lead to “neurodiscrimination,” in which people who are not included in the data may be given incorrect or less accurate diagnoses. These difficulties show how urgently strong regulation, inclusive training data, and well-defined ethical frameworks are needed for implementing AI in neuroscience.
Difficulties with Neuroimaging Methods
There are several practical and technological difficulties with multimodal neuroimaging, which integrates methods such as fMRI and EEG to capitalize on their complimentary advantages. EEG recordings may be hampered by the magnetic environment of the MRI, making it challenging to get clear, useful signals. Furthermore, it is difficult to align and register images when integrating data from several modalities because of variations in acquisition techniques and image attributes. Variability in tissue appearance and the absence of standardized correspondence rules further impede image registration, which is crucial for comparing scans acquired at various times or with different techniques. An additional layer of complexity is introduced by patient-related problems, such as mobility, collaboration, and medical conditions, especially in paediatric neuroimaging, which necessitate the use of specialized teams to provide precise and superior diagnostic pictures.79,80
Issues with Privacy and Data Availability
The fact that many algorithms’ performance indicators do not match the demands of actual clinical applications presents a major obstacle to the integration of AI into clinical neuroscience. Most AI models are verified using historical data, which is not representative of patient populations or clinical contexts today. These models must be verified prospectively, that is, tested on live patients in real-time situations, to be clinically applicable. The discrepancy between clinical efficacy and AI accuracy cannot be closed without access to current medical data. Therefore, data sharing among interdisciplinary teams is necessary for true innovation. Standardized privacy and data-sharing laws, such the US’s Health Insurance Portability and Accountability Act (HIPAA), which permits the exchange of completely anonymised or deidentified data, must be accepted and followed by healthcare companies. Finding a balance between patient privacy and data accessibility is necessary to enable AI systems for clinical use that are both effective and ethical.81
Interpretation Difficulties
Numerous factors can lead to diagnostic errors in neuroimaging interpretation, such as failing to refer to previous research, inadequate or limited imaging protocols, an incomplete patient history, failing to notice abnormalities outside the focused region, making a diagnosis too quickly after discovering the first anomaly (“satisfaction of search”), and misinterpreting normal variants. Neurologists and radiologists working together can help reduce these mistakes. Additional obstacles to the trustworthy application of AI in neuroscience include reproducibility problems, algorithmic biases, demographic inequalities, methodological errors, and inadequate data quality. Future research must address these issues in a number of ways, such as: developing interconnected datasets through extensive interdisciplinary collaborations across biology, neuroscience, and computing; curating high-quality clinical data; standardizing and regulating data sharing practices; validating AI models with prospective, real-time clinical data; coordinating clinical utility with performance metrics; improving multimodal neuroimaging integration methods; developing data fusion software; establishing training data repositories that are large and easily accessible; and putting interoperability standards in place across organizations in the neurological healthcare ecosystem.82
Future Directions and Emerging Trends
Going forward, the development of extensive, multidisciplinary partnerships that can produce and preserving interconnected, superior datasets must be the core goal of neuro-AI research. To standardize, integrate, and make interoperable data from diverse sources—such as mouse genomic studies, rat neurocircuitry data, and human cognitive imaging—initiatives should seek to bring together neuroscientists, AI specialists, clinicians, and bioinformaticians to collaborate on common platforms. Initiatives like the UK Biobank and the Allen Brain Atlas provide examples of how to harmonize and make large datasets accessible across domains. In addition to implementing interoperability standards and legislative frameworks that guarantee data privacy and ethical compliance, it will be essential to establish strong data-sharing policies and safe, anonymised data repositories.
Simultaneously, research must focus on creating clinically validated, explainable AI systems that meet medical standards and can be utilized with confidence in actual healthcare settings. This entails developing AI models that are prospectively evaluated on a range of patient demographics and integrating interpretation aids like attention maps and SHAP straight into therapeutic procedures. Furthermore, to enable systems to consistently combine and analyze data from sources such as fMRI, EEG, and PET scans, future work should concentrate on developing multimodal neuroimaging integration algorithms. It is imperative to address ethical issues like algorithmic prejudice and cognitive autonomy as AI models grow more transparent and linked with neuroscience. In the end, fostering trust in AI by openness, uniformity, inclusivity, and cooperative creativity is essential to future advancement. Creating cutting-edge software tools and platforms especially for multimodal data fusion and analysis is another important future direction. In addition to having features for picture registration, normalization, and real-time processing, these tools ought to make it easier to integrate neuroimaging data from other modalities (such as fMRI, EEG, MEG, and PET). Future software may, for instance, automatically align brain images from several scans and adjust for distortions or motion aberrations using AI-driven algorithms. Furthermore, democratizing access to cutting-edge neuroinformatics will need the development of user-friendly systems that enable researchers and clinicians to visualize, analyze, and communicate complicated brain data without requiring extensive technical knowledge. In addition to improving diagnostic precision and effectiveness, these developments will pave the way for personalized therapy in psychiatry and neurology, where therapies can be customized to each patient’s particular brain profile.
Conclusion
The diagnosis, prognosis, and treatment of neurological disorders could all be revolutionized by the application of artificial intelligence (AI) and machine learning (ML) in neuroscience. The utilisation of AI models, from deep learning techniques for identifying autism and Alzheimer’s disease to convolutional neural networks for brain tumor detection, shows a notable improvement in speed and accuracy over conventional diagnostic instruments, as this paper examines. However, there are several drawbacks to these technological developments, including issues with interpretability, ethics, and regulatory compliance; data fragmentation brought on by separate research cultures; and integration of multimodal neuroimaging data. Going forward, overcoming these obstacles calls for a multipronged approach that emphasizes developing interdisciplinary cooperation, guaranteeing the accessibility and quality of data, putting strong rules and laws in place, and enhancing AI models’ clinical usefulness and interpretability. To close current gaps, projects like the Human Brain Project and the creation of extensive, explicable, and prospectively validated AI systems are essential. AI and ML have the potential to completely transform neurological healthcare by facilitating earlier diagnosis, more individualized treatment, and eventually better patient outcomes worldwide—but only with sustained innovation, ethical supervision, and cooperative research.
Acknowledgement
The authors expressed their gratitude to Mr. Jitender Joshi, President, Prof. (Dr.) Dharam Buddhi, Vice Chancellor of Uttaranchal University Dehradun, and Prof. (Dr.) Vikash Jakhmola, Director, Uttaranchal Institute of Pharmaceutical Sciences, for their encouragement and guidance in publishing this review work.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Authors contribution
- Bhawna Chhabra, Prince Raj, Arkan, Pushpanjali Kumari, and Komal Kumari: Data collection
- Pankaj Pant and Malvika Kandpal: Manuscript Draft
- Mansi Butola and Vikash Jakhmola: Manuscript Draft and Reviewed
References
- Iorio-Morin C, Villeneuve SA, Masson-Côté L, Mathieu D. Neurological Disorders. Med Radiol. 2016;Part F1243:181–190.
CrossRef - A spotlight on sex differences in neurological disorders. The Lancet Neurology. 2019;18(4):319–330.
CrossRef - National Academies of Sciences, Engineering, and Medicine. Techniques for Neurological Disorders. Adv Diagn Eval Disabling Phys Health Cond. 2023;1:1–12.
- Kolker E, Özdemir V, Kolker E. How Healthcare Can Refocus on Its Super-Customers (Patients, n =1) and Customers (Doctors and Nurses) by Leveraging Lessons from Amazon, Uber, and Watson. OMICS. 2016;20(6):329–333.
CrossRef - Jiang F, Jiang Y, Zhi H, Dong Y, Li H, Ma S, et al. Artificial intelligence in healthcare: past, present and future. Stroke Vasc Neurol. 2017;2(4):230–240.
CrossRef - Integrated analysis of long non-coding RNA competing interactions reveals the potential role in progression of human gastric cancer. Int J Oncol. 2016;49(4):1651–1663.
- Darcy AM, Louie AK, Roberts LW. Machine Learning and the Profession of Medicine. JAMA. 2016;315(6):551–552.
CrossRef - Somashekhar S, Kumar C, Rauthan A, Arun K, Patil P, Ramya Y. Double blinded validation study to assess performance of IBM artificial intelligence platform, Watson for oncology in comparison with Manipal multidisciplinary tumour board – First study of 638 breast cancer cases. Cancer Res. 2017;77(4 Suppl):S6-07.
CrossRef - Esteva A, Kuprel B, Novoa RA, Ko J, Swetter SM, Blau HM, et al. Dermatologist-level classification of skin cancer with deep neural networks. Nature. 2017;542(7639):115–118.
CrossRef - Bouton CE, Shaikhouni A, Annetta NV, Bockbrader MA, Friedenberg DA, Nielson DM, et al. Restoring cortical control of functional movement in a human with quadriplegia. Nature. 2016;533(7602):247–250.
CrossRef - Farina D, Vujaklija I, Sartori M, Kapelner T, Negro F, Jiang N, et al. Man/machine interface based on the discharge timings of spinal motor neurons after targeted muscle reinnervation. Nat Biomed Eng. 2017;1(2):1–12.
CrossRef - Marr B. First FDA approval for clinical cloud-based deep learning in healthcare. Forbes. 2017;1:1–2.
- Food and Drug Administration. FDA permits marketing of clinical decision support software for alerting providers of a potential stroke in patients. FDA News Release. 2018;1:1–2.
- Somashekhar SP, Sepúlveda MJ, Puglielli S, Norden AD, Shortliffe EH, Rohit Kumar C, et al. Watson for Oncology and breast cancer treatment recommendations: Agreement with an expert multidisciplinary tumor board. Ann Oncol. 2018;29(2):418–423.
CrossRef - Shafi S, Parwani AV. Artificial intelligence in diagnostic pathology. Diagn Pathol. 2023;18(1):1–12.
CrossRef - Slomka PJ, Dey D, Sitek A, Motwani M, Berman DS, Germano G. Cardiac imaging: working towards fully-automated machine analysis and interpretation. Expert Rev Med Devices. 2017;14(3):197–212.
CrossRef - Misawa M, Kudo SE, Mori Y, Cho T, Kataoka S, Yamauchi A, et al. Artificial intelligence-assisted polyp detection for colonoscopy: initial experience. Gastroenterology. 2018;154(8):2027–2029.
CrossRef - American Academy of Ophthalmology. Diabetic retinopathy preferred practice pattern. Ophthalmology. 2024;131(5):1012–1024.
- Kalani M, Anjankar A. Revolutionizing neurology: the role of artificial intelligence in advancing diagnosis and treatment. Cureus. 2024;16(6):e61706.
CrossRef - Lee S, Kim HS. Prospect of artificial intelligence based on electronic medical record. J Lipid Atheroscler. 2021;10(3):282–289.
CrossRef - Pai VV, Pai RB, Pai VV, Pai RB. Artificial intelligence in dermatology and healthcare: an overview. Indian J Dermatol Venereol Leprol. 2021;87(4):457–67.
CrossRef - Soroush H, Goldman J. Toward a safe and secure medical Internet of Things. IIC J Innov. 2021;1:1–12.
- Singh M, Nath G. Artificial intelligence and anesthesia: a narrative review. Saudi J Anaesth. 2022;16(1):86.
CrossRef - Molnar MJ, Molnar V. AI-based tools for the diagnosis and treatment of rare neurological disorders. Nat Rev Neurol. 2023 Aug 1;19(8):455–456.
CrossRef - Hassabis D, Kumaran D, Summerfield C, Botvinick M. Neuroscience-inspired artificial intelligence. Neuron. 2017;95(2):245–58.
CrossRef - Macpherson T, Churchland A, Sejnowski T, DiCarlo J, Kamitani Y, Takahashi H, et al. Natural and artificial intelligence: a brief introduction to the interplay between AI and neuroscience research. Neural Netw. 2021;144:603–13.
CrossRef - Zhaoping L. Artificial and natural intelligence: from invention to discovery. Neuron. 2020;105(3):413–5.
CrossRef - Nwadiugwu MC. Neural networks, artificial intelligence and the computational brain. J Comput Sci. 2020;1(1):1–12.
- Whittington J, Bogacz R. An approximation of the error backpropagation algorithm in a predictive coding network with local Hebbian synaptic plasticity. Neural Comput. 2017;29(5):1229–62.
CrossRef - Xie Y, Liu YH, Constantinidis C, Zhou X. Neural mechanisms of working memory accuracy revealed by recurrent neural networks. Front Syst Neurosci. 2022;16:1–19.
CrossRef - Taye MM. Theoretical understanding of convolutional neural network: concepts, architectures, applications, future directions. Computation. 2023;11(3):52.
CrossRef - Blakeman S, Mareschal D. A complementary learning systems approach to temporal difference learning. Neural Netw. 2020;122:218–30.
CrossRef - Blakeman S, Mareschal D. Generating explanations from deep reinforcement learning using episodic memories. CEUR Workshop Proc. 2022;3227:53–8.
- Kim T, Hu S, Kim J, Kwak JY, Park J, Lee S, et al. Spiking neural network (SNN) with memristor synapses having non-linear weight update. Front Comput Neurosci. 2021;15:1–15.
CrossRef - Healtheuropa. The role of artificial intelligence in neuroscience. Health Europa. 2025;1:1–5.
CrossRef - Zhang X, Ma Z, Zheng H, Li T, Chen K, Wang X, et al. The combination of brain-computer interfaces and artificial intelligence: applications and challenges. Ann Transl Med. 2020;8(11):712.
CrossRef - Almeida JE, Oliveira E. Applications of artificial intelligence in neuroscience research: an overview. Kriativ Tech J. 2025;1:1–10.
CrossRef - Dias R, Torkamani A. Artificial intelligence in clinical and genomic diagnostics. Genome Med. 2019;11(1):1–14.
CrossRef - Brown CJ, Hamarneh G. Machine learning on human connectome data from MRI. arXiv. 2016;1–15.
- Zhu G, Jiang B, Tong L, Xie Y, Zaharchuk G, Wintermark M. Applications of deep learning to neuro-imaging techniques. Front Neurol. 2019;10:869.
CrossRef - Henschel L, Conjeti S, Estrada S, Diers K, Fischl B, Reuter M. FastSurfer – a fast and accurate deep learning based neuroimaging pipeline. Neuroimage. 2020;219:117012.
CrossRef - Levakov G, Rosenthal G, Shelef I, Raviv TR, Avidan G. From a deep learning model back to the brain—identifying regional predictors and their relation to aging. Hum Brain Mapp. 2020;41(12):3235–52.
CrossRef - Akinyelu AA, Zaccagna F, Grist JT, Castelli M, Rundo L. Brain tumor diagnosis using machine learning, convolutional neural networks, capsule neural networks and vision transformers, applied to MRI: a survey. J Imaging. 2022;8(8):1–24.
CrossRef - Jena B, Nayak GK, Saxena S. An empirical study of different machine learning techniques for brain tumor classification and subsequent segmentation using hybrid texture feature. Mach Vis Appl. 2022;33(1):1–17.
CrossRef - Sajjad M, Khan S, Muhammad K, Wu W, Ullah A, Baik SW. Multi-grade brain tumor classification using deep CNN with extensive data augmentation. J Comput Sci. 2019;30:174–82.
CrossRef - El Kader IA, Xu G, Shuai Z, Saminu S, Javaid I, Ahmad IS. Differential deep convolutional neural network model for brain tumor classification. Brain Sci. 2021;11(3):1–14.
CrossRef - Mzoughi H, Njeh I, Wali A, Slima MB, BenHamida A, Mhiri C, et al. Deep multi-scale 3D convolutional neural network (CNN) for MRI gliomas brain tumor classification. J Digit Imaging. 2020;33(4):903–15.
CrossRef - Kwabena PM, Felix AA, Abra MA, Edward BY. Capsule networks – a survey. J King Saud Univ Comput Inf Sci. 2022;34(1):1295–310
CrossRef - Dosovitskiy A, Beyer L, Kolesnikov A, Weissenborn D, Zhai X, Unterthiner T, et al. AN IMAGE IS WORTH 16X16 WORDS: TRANSFORMERS FOR IMAGE RECOGNITION AT SCALE. ICLR 2021 – 9th International Conference on Learning Representations. 2021;
- Application of machine learning to epileptic seizure onset detection and treatment. 2025 May 21
- Jaiswal A, Banka H. Local pattern transformation based feature extraction techniques for classification of epileptic EEG signals. Biomed Signal Process Control. 2017;34:81–92.
CrossRef - Hossain MS, Amin SU, Alsulaiman M, Muhammad G. Applying deep learning for epilepsy seizure detection and brain mapping visualization. ACM Trans Multimed Comput Commun Appl. 2019;15(1s):1–19.
CrossRef - Hu W, Cao J, Lai X, Liu J. Mean amplitude spectrum based epileptic state classification for seizure prediction using convolutional neural networks. J Ambient Intell Humaniz Comput. 2023;14(11):15485–95.
CrossRef - Abdelhameed A, Bayoumi M. A deep learning approach for automatic seizure detection in children with epilepsy. Front Comput Neurosci. 2021;15:650050.
CrossRef - Heinsfeld AS, Franco AR, Craddock RC, Buchweitz A, Meneguzzi F. Identification of autism spectrum disorder using deep learning and the ABIDE dataset. Neuroimage Clin. 2018;17:16–23.
CrossRef - Chen H, Song Y, Li X. Use of deep learning to detect personalized spatial-frequency abnormalities in EEGs of children with ADHD. J Neural Eng. 2019;16(6):066021.
CrossRef - Movaghar A, Page D, Brilliant M, Mailick M. Advancing artificial intelligence-assisted pre-screening for fragile X syndrome. BMC Med Inform Decis Mak. 2022;22(1):1–11.
CrossRef - Zhang P, Wang Z, Qiu H, Zhou W, Wang M, Cheng G. Machine learning applied to serum and cerebrospinal fluid metabolomes revealed altered arginine metabolism in neonatal sepsis with meningoencephalitis. Comput Struct Biotechnol J. 2021;19:3284–92.
CrossRef - Michel PP, Hirsch EC, Hunot S. Understanding dopaminergic cell death pathways in Parkinson disease. Neuron. 2016;90(4):675–91.
CrossRef - Korolev S, Safiullin A, Belyaev M, Dodonova Y. Residual and plain convolutional neural networks for 3D brain MRI classification. Proc Int Symp Biomed Imaging. 2017;835–8.
CrossRef - Moradi E, Pepe A, Gaser C, Huttunen H, Tohka J. Machine learning framework for early MRI-based Alzheimer’s conversion prediction in MCI subjects. Neuroimage. 2015;104:398–412.
CrossRef - Alty J, Cosgrove J, Thorpe D, Kempster P. How to use pen and paper tasks to aid tremor diagnosis in the clinic. Pract Neurol. 2017;17(6):456–463
CrossRef - Kotsavasiloglou C, Kostikis N, Hristu-Varsakelis D, Arnaoutoglou M. Machine learning-based classification of simple drawing movements in Parkinson’s disease. Biomed Signal Process Control. 2017;31:174–180.
CrossRef - Bron EE, Smits M, Niessen WJ, Klein S. Feature selection based on the SVM weight vector for classification of dementia. IEEE J Biomed Health Inform. 2015;19(5):1617–1626.
CrossRef - Zhao A, Qi L, Dong J, Yu H. Dual channel LSTM based multi-feature extraction in gait for diagnosis of neurodegenerative diseases. Knowl Based Syst. 2018;145:91–97.
CrossRef - Ferroni P, Zanzotto FM, Scarpato N, Spila A, Fofi L, Egeo G, et al. Machine learning approach to predict medication overuse in migraine patients. Comput Struct Biotechnol J. 2020;18:1487–1496.
CrossRef - Kwon J, Lee H, Cho S, Chung C, Lee M, Park H, et al. Machine learning-based automated classification of headache disorders using patient-reported questionnaires. Sci Rep. 2020;10(1):14062.
CrossRef - Menon B, Pillai A, et al. Artificial intelligence–assisted headache classification: a review. Elsevier. 2022.
CrossRef - Vandenbussche N, Van Hee C, Hoste V, Paemeleire K. Using natural language processing to automatically classify written self-reported narratives by patients with migraine or cluster headache. J Headache Pain. 2022;23(1):129.
CrossRef - Demaerschalk BM, Cheng NT, Kim AS. Intravenous thrombolysis for acute ischemic stroke within 3 hours versus between 3 and 4.5 hours of symptom onset. Neurohospitalist. 2015;5(3):101–109.
CrossRef - Lee EJ, Kim YH, Kim N, Kang DW. Deep into the brain: Artificial intelligence in stroke imaging. J Stroke. 2017;19(3):277–285.
CrossRef - Giacalone M, Rasti P, Debs N, Frindel C, Cho TH, Grenier E, et al. Local spatio-temporal encoding of raw perfusion MRI for the prediction of final lesion in stroke. Med Image Anal. 2018;50:117–126.
CrossRef - Chen Y, Dhar R, Heitsch L, Ford A, Fernandez-Cadenas I, Carrera C, et al. Automated quantification of cerebral edema following hemispheric infarction: application of a machine-learning algorithm to evaluate CSF shifts on serial head CTs. Neuroimage Clin. 2016;12:673–680.
CrossRef - Ni Y, Alwell K, Moomaw CJ, Woo D, Adeoye O, Flaherty ML, et al. Towards phenotyping stroke: Leveraging data from a large-scale epidemiological study to detect stroke diagnosis. PLoS One. 2018;13(2):e0192586.
CrossRef - Hayden DT, Hannon N, Callaly E, Ní Chróinín D, Horgan G, Kyne L, et al. Rates and determinants of 5-year outcomes after atrial fibrillation–related stroke: a population study. Stroke. 2015;46(12):3488–3493.
CrossRef - Aneja S, Chang E, Omuro A. Applications of artificial intelligence in neuro-oncology. Curr Opin Neurol. 2019;32(6):850–856.
CrossRef - Savin I, Ershova K, Kurdyumova N, Ershova O, Khomenko O, Danilov G, et al. Healthcare-associated ventriculitis and meningitis in a neuro-ICU: incidence and risk factors selected by machine learning approach. J Crit Care. 2018;45:95–104.
CrossRef - Ienca M, Ignatiadis K. Artificial intelligence in clinical neuroscience: methodological and ethical challenges. AJOB Neurosci. 2020;11(2):77–87.
CrossRef - Tong L. Evaluation of different brain imaging technologies. Proc Int Conf Public Art Hum Dev. 2022.
CrossRef - Barkovich MJ, Li Y, Desikan RS, Barkovich AJ, Xu D. Challenges in pediatric neuroimaging. Neuroimage. 2019;185:793–801.
CrossRef - White T, Blok E, Calhoun VD. Data sharing and privacy issues in neuroimaging research: opportunities, obstacles, challenges, and monsters under the bed. Hum Brain Mapp. 2022;43(1):278–291.
CrossRef - Samper-González J, Burgos N, Bottani S, Fontanella S, Lu P, Marcoux A, et al. Reproducible evaluation of classification methods in Alzheimer’s disease: framework and application to MRI and PET data. Neuroimage. 2018;183:504–521.
CrossRef
Abbrevations List
AI; Artificial Intelligence, ML; Machine Learning, ANNB; Artificial Neural Network, CNN; Convolutional Neural Network, RNN; Recurrent Neural Network, MLP; Multilayer Perceptron, LSTM; Long Short-Term Memory, SNN; Spiking Neural Network, RL; Reinforcement Learning, DNN; Deep Neural Network, NLP; Natural Language Processing, BCI; Brain-Computer Interface, EMR; Electronic Medical Record, FDA; Food and Drug Administration, MRI; Magnetic Resonance Imaging, CT; Computed Tomography, EEG; Electroencephalography, EMG; Electromyography, PET; Positron Emission Tomography, DMRI; Diffusion Magnetic Resonance Imaging, fMRI; Functional Magnetic Resonance Imaging, MEG; Magnetoencephalography, AD; Alzheimer’s Disease, PD; Parkinson’s Disease, ALS; Amyotrophic Lateral Sclerosis, HD; Huntington’s Disease, MCI; Mild Cognitive Impairment, MND; Motor Neuron Disease, SCZ; Schizophrenia, ASD; Autism Spectrum Disorders, TTH; Tension-Type Headache, MOH; Medication-Overuse Headache, TAC; Trigeminal Autonomic Cephalalgia, TBI; Traumatic Brain Injury, ID; Intellectual Disability, DD; Developmental Disability, UI; Uncertainty Interval, DALYs; Disability-Adjusted Life Years, GBD; Global Burden of Disease, CLS; Complementary Learning Systems, CADe; Computer-Aided Detection, CADx; Computer-Aided Diagnosis, SHAP; SHapley Additive exPlanations, SVM; Support Vector Machine, RF; Random Forest, XGBoost; Extreme Gradient Boosting, ViT; Vision Transformer







