Manuscript accepted on :12-12-2025
Published online on: 23-12-2025
Plagiarism Check: Yes
Reviewed by: Dr. Md. Sarwar Hossain
Second Review by: Dr. Sumayah Faruq Kasim
Final Approval by: Dr. Patorn Piromchai
Desak Putu Oki Lestari1* , Analysa2
, Analysa2 , Ni Wayan Armerinayanti3
, Ni Wayan Armerinayanti3 , Ni Putu Diah Witari4
, Ni Putu Diah Witari4 , Indah Budiapsari5
, Indah Budiapsari5 and I Nyoman Putu Riasa6,7
and I Nyoman Putu Riasa6,7
*Department of Pathological Anatomy, Faculty of Medicine and Health Sciences, Universitas Warmadewa, Bali, Indonesia.
2Department of Pathological Anatomy, Tabanan Hospital, Bali, Indonesia.
3Department of Pathological Anatomy, Faculty of Medicine and Health Sciences, Universitas Warmadewa, Bali, Indonesia
4Department of Histology, Faculty of Medicine and Health Sciences, Universitas Warmadewa, Bali, Indonesia.
5Department of Parasitology, Faculty of Medicine and Health Sciences, Universitas Warmadewa, Bali, Indonesia.
6Department of Plastic Surgery, Faculty of Medicine, Universitas Udayana, Bali, Indonesia.
7Department of Plastic Surgery, Prof.dr.I.G.N.G Ngoerah General Hospital, Bali, Indonesia.
Corresponding Author E-mail: oki.lestari@warmadewa.ac.id
DOI : https://dx.doi.org/10.13005/bpj/3313
Abstract
Persistent infection of high risk human papilloma virus (HPV) is a recognized driver oncogenesis in penile cancer, often characterized by upregulation of p16 and immune evasion via programmed death-ligand 1 (PD-L1) expression. This study aimed to assess the prognostic significance of p16 and PD-L1 in advanced penile cancer. Methods: A total of 80 cases of advanced penile cancer were retrospectively evaluated. Expression of p16 and PD-L1 was assessed by immunohistochemistry. Various clinicopathological parameters, such as patient age, tumor dimensions, histopathological grading, evidence of vascular invasion, and presence of lymphatic metastasis, were systematically evaluated in relation to PD-L1 overexpression. PD-L1 overexpression was observed in 37 (46.25%) of cases. PD-L1 positivity was significantly higher in p16 positive tumors (57.5%) than in p16 negative cases (35%) (p=0.044). Elevated PD-L1 expression strongly correlated with increased tumor dimension (p=0.002), advanced histological grade (p=0.000), the presence of vascular invasion (p=0.001), and lymphatic node involvement (p=0.000). PD-L1 overexpression was not significantly associated with the age of patients (p=0.932). PD-L1 and p16 are associated with clinicopathological features in advanced penile squamous cell carcinoma and may serve as prognostic biomarkers. Further multi-institutional studies are needed to clarify their predictive value for tailoring immunotherapy in penile cancer.
Keywords
HPV; P16; PDL-1; penile cancer; SCC
Download this article as:| Copy the following to cite this article: Lestari D. P. O, Analysa A, Armerinayanti N. W, Witari N. P. D, Budiapsari I, Riasa I. N. P. Prognostic Role of Programmed Death Ligand-1 and P16 in Advanced Penile Cancer. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Lestari D. P. O, Analysa A, Armerinayanti N. W, Witari N. P. D, Budiapsari I, Riasa I. N. P. Prognostic Role of Programmed Death Ligand-1 and P16 in Advanced Penile Cancer. Biomed Pharmacol J 2025;18(4). Available from: https://bit.ly/4aqyMgU |
Introduction
Globally, penile carcinoma represents a rare type of malignancy,1 however, it poses a significant burden in Asia, accounting for 57% of global cases and over 8,650 deaths annually.2 Indonesia, particularly Bali, exhibits a notably high prevalence of penile cancer,3 where patients often present in advanced stages.4 Despite standard treatments like surgery and chemotherapy, survival rates remain poor, particularly in patients with metastases.5 This indicates the necessity for alternative therapies involving Programed Death-Ligand 1 (PDL-1).
Targeting PDL-1 has become a cornerstone in cancer immunotherapy, with compelling evidence supporting its effectiveness across a broad spectrum of tumor types.6 The blockade of the PD-1/PD-L1 axis has been shown to elicit robust anti-tumor immune responses while minimizing systemic toxicity, a critical advantage over traditional therapies such as chemotherapy and radiation.7,8 Immunotherapeutic approaches, particularly immune checkpoint inhibitors, now play a leading role in cancer treatment protocols.9–11 Among patients with advanced penile cancer, elevated PD-L1 expression is observed in 32.9% of cases and is linked to more aggressive pathological characteristics and unfavorable clinical outcomes.12 This therapeutic innovation appears to improved patient management and may contributes to better survival rates.
Persistent HPV infection acts as a critical driver in the pathogenesis of several cancers, primarily via the induction of an immunosuppressive tumor microenvironment. HPV’s E5 oncoprotein has been reported to stimulate epidermal growth factor receptor (EGFR) activation, subsequently resulting in increased PD-L1 expression on malignant cells. This process permits tumor cells to evade immune detection by inducing apoptosis in T-cells, consequently undermining the robustness of the anti-tumor immune response and diminishing the host’s immunological control over malignant cells.13 Elevated PD-L1 levels are frequently observed in HPV-positive squamous cell carcinomas and are closely linked with HPV related malignancies.14 High risk HPV varieties are recognized carcinogens for penile cancer, and strong p16 immunostaining is a dependable indicator of active infection with HPV.15,16 Furthermore, p16 overexpression stems from the disruption of retinoblastoma (Rb) protein function by the HPV E7 oncoprotein, leading to altered cell cycle regulation and facilitating tumorigenesis.17
Research on the correlation between PDL-1 expression and P16 in penile cancer is still small and shows results that vary widely in different countries. Further investigations are required to elucidate the molecular pathways via which HPV affects PD-L1 expression in penile cancer. Comprehending these pathways may uncover prospective therapeutic targets and enhance treatment approaches. The prevalence of HPV-related penile cancer varies significantly across different regions, such as the high rates observed in Bali, Indonesia. More research is needed to understand how regional factors influence HPV prevalence, PD-L1 expression, and therapy effect. This work seeks to elucidate the relationship between P16 overexpression and clinical outcomes associated with PDL-1 in PeSCC.
Materials and Methods
Research Design
A retrospective cohort design utilizing purposive sampling was applied in this study. The relationships between P16 expression and clinicopathological parameters, including patient age, tumor dimension, histological tumor grade, vascular invasion, lymph node involvement, and PD-L1 expression, were analyzed using the chi-square test. Statistical significance was determined at a threshold of p < 0.05.
Location and Time of Research
The research was carried out for 6 months, from February to August 2024, at the Biochemical Lab of the Faculty of”Medicine and Health Sciences, Warmadewa University, and the Tabanan Regional General Hospital.
Population and Sample
Penile cancer tissue specimens, primarily obtained from primary tumors from radical penectomy or penectomy procedures, were collected from several pathology anatomy laboratories in Bali between 2015 and 2024. All patients included were of Balinese ethnicity. Primary tumor samples were preserved using formalin fixation and embedded in paraffin (FFPE technique). From a larger group of 160 patients diagnosed with penile squamous cell carcinoma, 80 cases were chosen. Selection criteria required paraffin blocks to contain malignant tissue and meet standards for specimen integrity.
Research Procedure
Clinical Data: Age, tumor size, grade, vascular invasion, and nodal metastasis was identified from clinical data.
Immunohistochemistry: For immunohistochemical procedures, tumor blocks preserved in formalin and embedded in paraffin underwent sectioning at a thickness of 2–3 μm and were positioned on charged slides. The sections were processed to remove paraffin using xylene and then gradually rehydrated with alcohol sequences and distilled water. Antigens were retrieved by warming the specimens in citrate buffer (pH 6.0) for 20 minutes in a pressure cooker. Endogenous peroxidase was inhibited by treating with 3% hydrogen peroxide at ambient temperature for 10 minutes. The slides were subsequently rinsed in phosphate-buffered saline and incubated for 60 minutes with specific antibodies: p16 (Ventana, E6H4 clone, ready-to-use) and PD-L1 (Biocare Medical, CAL10 clone, diluted 1:100). Afterwards, the EnVision+ polymer detection system (Dako) was applied for 30 minutes. Visualization was accomplished using 3,3′-diaminobenzidine (DAB), with hematoxylin counterstaining, then slides were dehydrated, cleared, and finally mounted for microscopic review. Immunohistochemistry evaluation criteria were then applied to assess results. Immunohistochemistry evaluation criteria:
P16 Expression: p16 Immunoreactivity was interpreted as positive when tumor cell nuclei and cytoplasm both demonstrated staining. Following WHO recommendations for HPV-associated squamous cell cancers, positivity was defined as strong, diffuse (block-type) staining in at least 75% of malignant cells.18
The expression of PD-L1: PD-L1 expression was assessed by analyzing membrane staining patterns in tumor cells. In line with recognized standards for assessing solid tumors, a case was designated as PD-L1 positive if at least 1% of cancer cells displayed distinct membranous staining by immunohistochemistry.19
Each staining batch incorporated suitable positive and negative controls. Two skilled pathologists, blinded to patient information, independently assessed all slides, and any differences in interpretation were addressed through collaborative discussion and agreement.
Results
This study demonstrates strong p16 immunohistochemical staining within penile squamous cell carcinoma (PeSCC) tumor cells, indicating p16 overexpression. Elevated expression of p16 immunostaining is widely used as an indicator of high-risk HPV involvement, implying a significant etiological role for HPV in these tumors. The presence of strong p16 staining in Figure 1.A supports the HPV-driven pathogenesis outlined, where HPV infection leads to increased p16 overexpression. The p16 results highlight the strong correlation between HPV and penile cancer.
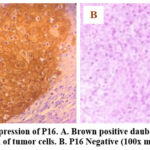 |
Figure 1: Overexpression of P16. A. Brown positive daubed in the nucleus and cytoplasm of tumor cells. B. P16 Negative (100x magnification). |
This study investigated the relationship between PD-L1 overexpression and various clinicopathological characteristics in a cohort of 80 penile cancer patients. Our findings reveal significant associations between PD-L1 overexpression and several key factors (Table 1). A large portion of patients in this study presented locally advanced penile cancer, which is characterized by larger tumor size, higher grade, intravasal invasion, and lymph node involvement.
Table 1: Correlation of PDL-1 overexpression with P16 and clinicopathology
| Characteristic | Sample n=80
n (%) |
Overexpression of PDL-1 n=80
n (%) |
p value |
| Expression P16 | |||
| PositiveNegative | 40 (50%)40 (50%) | 23 (28.75%)14 (17.5%) | 0.044 |
| Age (years) | |||
| <45 | 10 (12.5%) | 4 (5%) | 0.932 |
| >=45 | 70 (87.5%) | 33 (41%) | |
| Tumor Size | |||
| <2 cm | 22 (27.5%) | 4 (5%) | 0.002 |
| >=2 cm | 58 (72.5%) | 33 (41%) | |
| Grading | |||
| Grade 1Grade 2
Grade 3 |
19(23.75%)33(41.25%)
28 (35.0%) |
4 (5%)11 (13.8%)
22 (27.5%) |
0.000 |
| Intravasa invasion | |||
| Negative | 37 (46.25%) | 10 (12.5%) | 0.001 |
| Positive | 43 (53.75%) | 27 (33.75%) | |
| Lymph Node Metastasis | |||
| NegativePositive | 37 (46.25%)43 (53.75%) | 9 (11.25%)28 (35%) | 0.000 |
PD-L1 overexpression in penile cancer (Figure 2) correlates significantly with several indicators of aggressive tumor behavior. Increased tumor size (>=2 cm, p = 0.002) and higher tumor grade (p < 0.001), particularly in Grade 3 tumors, were substantially correlated with heightened PD-L1 expression. This suggests that PD-L1 may be upregulated as tumors grow and dedifferentiate, contributing to a more aggressive phenotype. Furthermore, PD-L1 overexpression was significantly more prevalent in tumors exhibiting intravasal invasion (p = 0.001) and lymph node metastasis (p < 0.001), underscoring its potential role in promoting tumor spread.
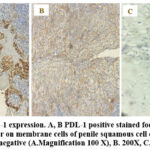 |
Figure 2: PDL-1 expression. A, B PDL-1 positive stained focal and diffuse brownish color on membrane cells of penile squamous cell carcinoma, C. PDL-1 negative (A.Magnification 100 X), B. 200X, C. 400 X). |
The data revealed an equal distribution between p16 positive and negative samples, with 40 cases in the groups (50%). Among p16 positive cases, 23 (57.5%) exhibited PDL-1 overexpression. While only 14 (35%) of p16 negative cases showed PDL-1 overexpression. When accounted for entire sample population, this corresponded to 28.75% and 17.5%, respectively. The difference in PDL-1 overexpression between two groups was statistically significant (p = 0.044). These findings demonstrate that PDL-1 overexpression is much more frequent in samples expressing p16, suggesting a notable association between these molecular markers.
Discussion
This study presents compelling evidence regarding the clinical features of patients diagnosed with locally advanced penile cancer. The present analysis revealed that metastatic involvement of inguinal of pelvic lymph nodes occurred in 53.75% of cases, a proportion notably higher than international averages, which typically report metastasis in approximately one-third of cases,20 This difference may reflect regional disease burden or healthcare access limitation specific to studied Balinese population. Urological studies validated our findings of substantial intravasal invasion rates and lymph node metastases.21 The investigation corroborated our findings that larger tumor size and higher grade forecasted intravasa involvement are significant. The overall pattern of aggressive characteristics linked to lymph node metastasis is consistent across multiple investigations, underscoring the critical importance of early detection and intervention in penile cancer. These findings highlight the need for improved screening and public awareness to facilitate earlier diagnosis and potentially improve outcomes in penile cancer patients.
A crussial aspect highlighted from meta-analyses is the heterogeneity in p16 expression among PeSCC cases world wide. The current study found p16 positivity in approximately half of the cases, reinforcing reports of variable of p16 prevalence by region and HPV epidemiology. Data suggest that p16 immunorectivity continues to serve as a surrogate for high risk HPV exposure and often correlates with better diseases specific survival, thought its prognostic value may be modulated by additional host and tumor factors.22 The effectiveness of p16 as an indicator for HPV infection in PeSCC continues to be a subject of discussion, as its utility varies across studies. Some research has demonstrated a strong association between p16 immunopositivity and the presence of HPV 16 DNA, reinforcing its value as a marker for HPV driven neoplasms in certain contexts.23 Conversely, other research reported that up to 50% of HPV positive penile cancer cases (by molecular assay) were negative for p16 immunohistochemical expression, indicating potential limitations in its use as a standalone marker.24 These discrepancies underscore the need for combined approaches, such as using p16 immunohistochemistry alongside HPV genotyping, to more accurately determine HPV status in penile cancer cases and its prognostic implications.
Our findings further demonstrated a strong association between high PDL-1 expression and adverse pathological features, including larger tumor size, high histological grade, and presence of lymph node metastasis (Table 1). This result is corroborated by recent international cohort meta-analytic studies showing that PD-L1 expression is consistently linked with poor prognosis marker and increased risk of regional and distant metastasis.25 The studies from Brazil confirmed that up to 69.2% of lymph node metastases in penile cancer were PDL-1 positive, supporting its utility of PDL-1 as both a prognostic biomarker and therapeutic target.26 Studies have documented substantial PD-L1 expression in both initial and metastatic lesions of PeSCC, implying a potential contribution to tumor aggressiveness.27 In a cohort of 37 individuals diagnosed with penile carcinoma, increased PD-L1 expression showed a statistically significant link to metastasis involving regional lymph nodes ( ), as well as poorer cancer specific survival outcomes ( ).28 The consistent results from much research highlight the potential of PD-L1 as a biomarker for aggressive disease behavior and its putative involvement in tumor growth and metastasis in penile cancer.
A complex relationship between p16 expression and PD-L1 overexpression in penile cancer is found in this study, 57.5% of p16 positive tumors exhibited PD-L1 overexpression, compared to 35% of p16 negative tumors (p = 0.044). This suggest that PD-L1 overexpression is substantially more common within HPV associated (p16 positive) penile cancer in our cohort. Recent studies from endemic regions confirm this association, indicated a strong correlation between PD-L1 and p16 expression (p = 0.002 (p = 0.002),26 supporting the biologic plausibility HPV driven carcinogenesis upregulates immune evasion mechanism via PDL-1. Furthermore, we observed a higher overall prevalence of PD-L1 overexpression (46.25% across all samples) compared to some previous reports, such as a study in Biomarkers Research that reported PD-L1 expression in only 18% of cases.27 Nevertheless, conflicting evidence exist, with some report indicating PD-L1 expression with HPV negative tumor,29 highlighting the context dependency and possibly region specific pathways involved. This finding may have implications for immunotherapeutic eligibility, as HPV mediated of upregulation of PD-L1 could identify patients most likely to benefit from PD-1/PD-L1 check point inhibition.
Our research corroborates and extends the current literature emphasizing the function of PD-L1 in facilitating tumor advancement and immune evasion across diverse cancers. Elevated PD-L1 expression is associated with advanced disease stages and unfavorable prognoses in many malignancies.6 In penile cancer, PD-L1 overexpression was substantially correlated with elevated tumor grade, increased tumor size, intravascular invasion, and lymph node metastases, all of which are markers of aggressive illness (Figure 3). Recent meta-analyses and multicenter studies indicate that elevated PD-L1 expression in penile squamous cell carcinoma correlates with diminished cancer-specific survival and more advanced clinicopathological characteristics.6 These findings underscore PD-L1’s potential as a predictive biomarker and therapeutic target in penile carcinoma. The pronounced correlation identified in our work between PD-L1 expression and lymph node metastasis indicates a role for PD-L1 in promoting tumor dispersion and regional spread, hence reinforcing its significance for risk classification and therapeutic planning in this condition.
 |
Figure 3: Tumor progression in advance penile cancer. |
In this analysis, age did not exhibit a statistically significant association with PD-L1 overexpression ( ). The absence of correlation in our sample might be attributed to the predominance of older individuals presenting with advanced-stage disease, potentially obscuring any age-dependent variation in PD-L1 status. Additionally, the retrospective approach and modest sample size represent notable study constraints. Prospective, multicenter studies with larger and more diverse cohorts are needed to validated these results and further explore the interplay between HPV status, p16 expression, and immune check point activation in different populations. Recent recommendations emphasize the need for integrated biomarker panels to more accurately inform immunotherapeutic indications and prognostication.
Conclusion
PD-L1 upregulation in penile carcinoma was significant associated with adverse pathological characteristics, such as increased tumor size, higher histological grade, vascular invasion, and lymphatic spread. Notably, A higher prevalence of PD-L1 positivity was observed in p16-positive tumors (57.5%) compared to p16-negative cases (35%), indicating a link between HPV-driven pathogenesis and immune checkpoint activation. The findings support PD-L1 and p16 as potensial prognostic biomarkers for advanced penile cancer. Larger, prospective studies are required to define the predictive value of these biomarkers for response to anti-PD-L1 therapy and personalize treatment strategies for different patient populations.
Acknowledgement
The authors gratefully acknowledge the financial assistance provided by the Research and Community Service Unit at Warmadewa University in support of this project. Appreciation is also extended to fellow scientists whose contributions were invaluable throughout the course of this research
Funding Source
Grant Fund from Research and Community Service Unit of the Faculty of Medicine and Health Sciences, Universitas Warmadewa (Award No. 2038/Unwar/PD-10/2020).
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research was reviewed and approved by the Health Research Ethics Committee Faculty of Medicine and Health Sciences, Universitas Warmadewa (Number: 2/Unwar/FKIK/EC-KEPK/III/2024). The research was conducted with respect for the rights and privacy of research subjects, and all requirements for ethical approval were fulfilled.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Author Contributions
Desak Putu Oki Lestari: Conceptualization, Methodology, Writing, Original draft
Analysa: Analysis, Visualization
Ni Wayan Armerinayanti: Data collection
Ni Putu Diah Witari: Data collection
Indah Budiapsari: Editing
I Nyoman Putu Riasa: Supervision
Reference
- Thomas A, Necchi A, Muneer A, et al. Penile cancer. J Nat Rev. 2021; 7: 1-24.
CrossRef - Globocan. Penile cancer. Global Cancer Observatory Website. International Agency for Reseach on Cancer. 2020. Accessed December 19,2025. https://gco.iarc.who.int/media/globocan/factsheets/cancers/26-penis-fact-sheet.
- Lestari DPO, Riasa INP, Niramayah PKI, et al. The burden of penile cancer in bali compared to other provinces in indonesia. Open Access Maced J Med Sci. 2021;9(E):1273-1278.
CrossRef - Suharta IGNAI, Yudiana IW, Oka AAG. Clinico-Pathological Characteristics of Penile Cancer Patients at Sanglah General Hospital Denpasar, Bali between 2013-2020. JBN (Jurnal Bedah Nasional). 2023;8(1):29.
CrossRef - Pandey A, Noronha V, Joshi A, Tongaonkar H, Bakshi G, Prabhash K. Resistant metastatic penile carcinoma and response to biochemotherapy with paclitaxel and epidermal growth factor receptor monoclonal antibody, nimotuzumab. Indian J Med Paediatr Oncol. 2013;34(1):24-27.
CrossRef - Tong C, Wu Y, Wu R. New dimensions of PD-1 / PD-L1 inhibitor combination therapy in cancer treatment : current advances and future perspectives. Front Immunol. 2025;1-13.
CrossRef - Parvez A, Choudhary F, Mudgal P, et al. PD-1 and PD-L1: architects of immune symphony and immunotherapy breakthroughs in cancer treatment. Front Immunol. 2023;14:1-16.
CrossRef - Gutic B, Bozanovic T, Mandic A, et al. Programmed cell death-1 and its ligands: Current knowledge and possibilities in immunotherapy. Clinics. 2023;78:0-6.
CrossRef - Shimada Y, Matsubayashi J, Kudo Y, et al. Serum-derived exosomal PD-L1 expression to predict anti-PD-1 response and in patients with non-small cell lung cancer. Sci Rep. 2021;11(1):1-10.
CrossRef - Jiang Y, Chen M, Nie H, Yuan Y. PD-1 and PD-L1 in cancer immunotherapy: clinical implications and future considerations. Hum Vaccines Immunother. 2019;15(5):1111-1122.
CrossRef - Gong J, Chehrazi-Raffle A, Reddi S, Salgia R. Development of PD-1 and PD-L1 inhibitors as a form of cancer immunotherapy: A comprehensive review of registration trials and future considerations. J Immunother Cancer. 2018;6(1):1-18.
CrossRef - Haritsyah Warli M, Prapiska FF, Siregar GP, Sihombing B, Warli SM. PD-L1 expression as predictor of immunotherapy eligibility in penile squamous cell carcinoma patients. F1000Research. 2022;11:1-12.
CrossRef - Allouch S, Malki A, Allouch A, Gupta I, Vranic S, Al Moustafa AE. High-Risk HPV Oncoproteins and PD-1/PD-L1 Interplay in Human Cervical Cancer: Recent Evidence and Future Directions. Front Oncol. 2020;10:1-9.
CrossRef - Jeong JY, Park TI, Ahn D. Comprehensive analysis and clinical implication of PD-l1 expression considering HPV status in oropharyngeal squamous cell carcinoma. Anticancer Res. 2020;40(7):4001-4010.
CrossRef - El-Daly MM, Faizo AA, Madkhali SA, et al. Study of the Prevalence of Human Papillomavirus Genotypes in Jeddah, Saudi Arabia. J Epidemiol Glob Health. 2025;15(1):1.
CrossRef - Ardhaputri KADL, Lestari DPO, Armerinayanti NW, et al. The Proportion of p16 Expression in Penile Squamous Cell Carcinoma Based on Immunohistochemistry Examination in the Balinese Population. Indones J Cancer. 2022;16(2):94.
CrossRef - Uddin S, Singh A, Mishra V, Agrawal N, Gooi Z, Izumchenko E. Molecular drivers of oral cavity squamous cell carcinoma in non-smoking and non-drinking patients: what do we know so far? Oncol Rev. 2022;16(1).
CrossRef - Kaushik V, Pai K, Rao A, Sharma S. Clinicopathological features and reclassification of penile squamous cell carcinoma according to WHO classification 2022 for penile carcinoma with p16 immunohistochemical expression and its prognostic impact. Published online 2025.
CrossRef - Taghizadeh H, Fajkovic H. Immunotherapy in the Management of Penile Cancer—A Systematic Review. Cancers (Basel). 2025;17(5):1-15.
CrossRef - Emerson RE, Ulbright TM, Eble JN, Geary WA, Eckert GJ, Cheng L. Predicting cancer progression in patients with penile squamous cell carcinoma: The importance of depth of invasion and vascular invasion. Mod Pathol. 2001;14(10):963-968.
CrossRef - Deng S, Xuan Z, Feng J, et al. Global research trends in penile cancer: Bibliometric and visualized analysis. Front Oncol. 2023;12(January):1-14. doi:10.3389/fonc.2022.1091816
CrossRef - Parza K, Mustasam A, Ionescu F, et al. The Prognostic Role of Human Papillomavirus and p16 Status in Penile Squamous Cell Carcinoma—A Systematic Review. Cancers (Basel). 2023;15(14):1-13.
CrossRef - Mustasam A, Parza K, Ionescu F, et al. The Prognostic Role of HPV or p16INK4a Status in Penile Squamous Cell Carcinoma: A Meta-Analysis. JNCCN J Natl Compr Cancer Netw. 2025;23(2):1-9.
CrossRef - Shetty M, Adiga DSA, Chaithra G V. Study of Expression of P16 in Premalignant and Malignant Lesions of Penis and Their Significance. Iran J Pathol. 2024;19(1):50-58.
CrossRef - Lu Y, Wang Y, Su H, Li H. PD-L1 is associated with the prognosis of penile cancer: A systematic review and meta-analysis. Front Oncol. 2022;12(November):1-11.
CrossRef - De Bacco MW, Carvalhal GF, MacGregor B, et al. PD-L1 and p16 Expression in Penile Squamous Cell Carcinoma From an Endemic Region. Clin Genitourin Cancer. 2020;18(3):e254-e259.
CrossRef - Sangkhamanon S, Kotano N, Sirithanaphol W, et al. Programmed death-ligand 1 expression in tumor cells and tumor-infiltrating lymphocytes are associated with depth of tumor invasion in penile cancer. Biomed Reports. 2023;19(1):1-8. doi:10.3892/br.2023.1627
CrossRef - Udager AM. Frequent PD-L1 expression in primary and metastatic penile squamous cell carcinoma: Potential opportunities for immunotherapeutic approaches. Ann Oncol. 2016;27(9):1706-1712.
CrossRef - Hrudka J, Hojný J, Prouzová Z, et al. High tumour mutational burden is associated with strong PD-L1 expression, HPV negativity, and worse survival in penile squamous cell carcinoma: an analysis of 165 cases. Pathology. 2024;56(3):357-366.
CrossRef







