Manuscript accepted on :28-11-2025
Published online on: 23-12-2025
Plagiarism Check: Yes
Reviewed by: Dr. Aparna Gupta
Second Review by: Dr. Dunya Abdal-Malik
Final Approval by: Dr. Mariia Shanaida
Sri Wahjuni , I Gusti Agung Gede Bawa, Anak Agung Istri Agung Mayun Laksmiwati*
, I Gusti Agung Gede Bawa, Anak Agung Istri Agung Mayun Laksmiwati* , Ida Ayu Raka Astiti Asih
, Ida Ayu Raka Astiti Asih , Ni Made Nonik Sawitri
, Ni Made Nonik Sawitri
Department of Chemistry Faculty of Mathematics and Natural Sciences Udayana University, Jalan Raya Kampus Unud, Jimbaran, Badung, Bali, Indonesia
Corresponding Author E-mail:sriwahjunimanuaba@unud.ac.id
DOI : https://dx.doi.org/10.13005/bpj/3302
Abstract
The red galangal rhizomes (Alpinia purpurata K. Schum) plant has natural product potential depending on the amount of active compound extracted and the extraction method. This study aimed to determine the effect of the administration of ethanol red galanga rhizomes of suplementation to histopathological changes in breast cancer and identified the coumpound content of red galangal rhizomes (Alpinia purpurata K. Schum). Use of phytochemical screening methods, identification using LC-MS/MS instruments, histopathological examination of breast glandular tissue, benzo(a)pyrene,Vacum rotary evaporator, set of tools LC-MS/MS AC QUITY UPLC®H- Class System (Waters, USA). The red galangal rhizome (Alpinia purpurata K.Schum) was obtained from Badung Regency, Bali, Indonesia, and determined at the LIPI-UPT Center for Plant Conservation of the Bali “Eka Karya” Botanical Garden. The red galanga rhizomes reported that the higher the ethanol concentration. Red galanga rhizomes contain perhexiline as an anti-breast cancer agent. Research shows growing evidence concerning toxic effects induced by benzo(a)pyrene substance. This xenobiotic is metabolized by cytochrome P450 (CYP P450) to carcinogenic metabolite: 7β,8α-dihydroxy-9α,10α-epoxy-7,8,9,10-tetrahydrobenzo[a]pyrene (BPDE), which creates DNA adducts, causing mutations and malignant transformations. CYP P450 is strongly involved in B[a]P metabolism, and it is simultaneously expressed as a result of the association of B[a]P with aromatic hydrocarbon receptor (AhR), playing an essential role in the carcinogenic potential of various xenobiotics. In turn, polymorphism of CYP P450 genes determines the sensitivity of the organism to B[a]P. Ethanol extract galangal rhizomes contains alkaloid, flavonoid, polifenol, steroid, triterpenoid and saponin compounds. Contained Perhexiline has the anticancer activity of red galanga rhizomes with benzo(a)pyrene induction at various doses the ethanol extract of red galangal rhizomes had an effect in improving the histopathological picture of mouse breast cancer cells at doses of 70 mg/kgBW/day and 90 mg/kgBW/day.
Keywords
Anticancer; Breast cancer; Benzo(a)pyrene; Histopathology; Red galanga rhizomes
Download this article as:| Copy the following to cite this article: Wahjuni S, Bawa I. G. A. G, Laksmiwati A. A. I. A. M, Asih I. A. R. A, Sawitri N. M. N. Phytochemical Screening and Evaluation of the Effect of Ethanol Extract Red Galanga Rhizomes Extract for Histopathology of Breast Cancer Cells in Mice. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Wahjuni S, Bawa I. G. A. G, Laksmiwati A. A. I. A. M, Asih I. A. R. A, Sawitri N. M. N. Phytochemical Screening and Evaluation of the Effect of Ethanol Extract Red Galanga Rhizomes Extract for Histopathology of Breast Cancer Cells in Mice. Biomed Pharmacol J 2025;18(4). Available from: https://bit.ly/49bYKm1 |
Introduction
Cancer is a disease associated with abnormal cell growth and division. Cancer is the second biggest cause of death in the world after heart or cardiovascular disease.1 According to the research, in 2020 there were around 18.1 million cases of cancer worldwide and it is estimated that this will increase in 2040 to 29.4 million cases.2 This disease is known to cause 1 in 6 people to die.3 Meanwhile in Indonesia, according to the Basic Health Research Agency (RISKESDAS), it was reported that there were 1.4 % of cancer cases in 2013 and this increased in 2018 to around 1.8 %.4
According to previous research, forecast status reports regarding events and cancer deaths, breast cancer is the second most frequently diagnosed malignancy, accounting for more than 11.6% of all cancers in women and causing 6.6 % of all cancer deaths worldwide. Breast cancer remains the main cause of cancer-related morbidity and death throughout the world. So studying the development of breast cancer epidemiology is very valuable because prevention can be carried out through a combination of primary and secondary prevention strategies.5
One of the factor contributing to the increasing prevalence of cancer is the uncontrolled presence of carcinogenic substances. Thousands of toxic chemicals are commonly found in the environment and can lead to contamination. Benzo[a]pyrene (B[a]P), a primary representative of polycyclic aromatic hydrocarbons (PAHs), is frequently detected in the air, surface water, soil, and sediments. It is also present in cigarette smoke and a variety of food products, particularly those that are smoked and grilled, making human exposure to B[a]P very common. Research has shown growing evidence of the toxic effects induced by this compound.6 PAHs, including B[a]P are a class of environmental pollutants affecting both humans and experimental animals. Exposure to PAHs typically occurs through air pollution, vehicle emissions, cigarette smoke, diesel fuel, and the consumption of grilled or smoked foods.7
This compound has hydrophobic and lipophilic properties, making it easily deposited within the food chain system and acts as carcinogenic whose activity depends on the presence of certain enzymes.8 As the number of cancer cases increases, the costs and duration of treatment also increase.9 In recent years, cancer sufferers have begun to combine medical and non-medical (herbal) treatments from natural ingredients. Drug combinations that include dietary supplements and natural products have been postulated to achieve similar effects to conventional chemotherapy drugs but with fewer side effects. This natural (herbal) treatment is supported by Indonesia’s potential as a country with millions of biodiversity.10 Research on the potential content of these metabolites is very much needed to guarantee the efficacy and level of safety of herbal plants that have this potential.11
One of the extensively studied herbal plants is red galanga (Alpinia purpurata K. Schum) whose rhizomes possess various pharmacological activities, such as antiviral, antifungal, antioxidant, antibacterial, anti-inflammatory and anticancer.12 The content of flavonoid derivatives (galangin) and phenol derivatives (ACA(1-Acetoxychavicol Acetate) in red galangal rhizomes (Alpinia purpurata K. Schum) is thought to have anticancer potential.13 According to research the content of flavonoid compounds in galangal methanol extract 16.76 µg/mL is able to inhibit murine leukemia P388 cancer cells by 50% (P <0.05) at very low extract concentrations. Inhibition is influenced by amide -CONH2), hydroxyl (-OH) and dilactone groups.14
Considering the potential of ethanol extract of red galangal rhizomes against breast cancer based on a literature review15, it is important to carry out further research as an innovation in preventing breast cancer. This study also identified the compound content in red galangal rhizomes.
Materials and Methods
Collection and Extraction of Red galangal rhizomes
Material
Red galangal rhizomes was purchased from Badung regency, Bali, Indonesia. The following chemicals were used: 96% ethanol (Sigma Aldrich), distilled water, benzo(a)pyrene, method of extracting active components from natural ingredients, male Wistar rats aged 2-3 months and weighing 150 – 200 g, were used this experiment breast cancer.
Equipment
Laboratorium glasswear, rotary vacuum evaporator, histopathological picture of mouse breast cancer, LC-MS/MS Xevo type G2TOF, pythochemistry test.
Preparation and Extraction
Samples of red galanga rhizomes were cut into smaller sizes. Red galangal is then dried by air-drying, without being exposed to direct sunlight. Samples of dried red galangal rhizomes were then ground again using a blender and weighed on an analytical balance. Dry red galanga rhizomes (Alpinia purpurata K. Schum) Rhizomes as much as 1 kg with water content of 5,26% were extracted with ethanol 96% at room temperature for 48h,16 the solvent was evaporated at 450C. Ethanol extract of red galanga rhizome (Alpinia purpurata K. Schum) was produced and then determination of Water Content of Red Galangal Rhizome Powder. This extract was also analyzed using LC-MS/MS.
Phytochemical screening
Phytochemical tests were conducted using thick extracts of red galangal rhizomes added with chloroform and water to form two phases. This method uses various chemical detection reagents for secondary metabolite compounds such as alkaloids, flavonoids, tannins, terpenoids, saponins, and others. The chemical changes that occur indicate the types of compounds contained in the extract.17 This analysis specifically aims to determine the biosynthesis mechanism, chemical structure, distribution of compounds, isolation and chemical composition of a compound.18
Haematoxylin-eosin staining
The examination uses the Haematoxylin-Eosin staining method. Tissue staining is based on the interaction of the chemicals used with cell and tissue components.19 The dyes used are able to interact and repel specific components which are influenced by their chemical properties. Haematoxylin is a chemical compound that is basic (basophilic), this compound can interact or bind to cells and tissues (such as DNA and RNA) which are acidic and produce a blue color change. Meanwhile, Eosin is a chemical compound that is acidic (eosinophilic), this compound is able to interact or bind to cells and tissues (such as cytoplasmic proteins) which are alkaline and produce a color change to red or pink.20
Histopathological slides were deparaffinized in xylene (I and II) for 2 minutes each and then rehydrated by immersing the samples in a series of graded alcohols (absolute alcohol, 95% alcohol, 90% alcohol, and 80% alcohol) for 1 minute each, followed by rinsing under running water for 1 minute. The slides were then immersed in Mayer’s hematoxylin solution for 8 minutes and washed under running water for 30 seconds. Afterward, they were dipped in lithium carbonate solution for 15-30 seconds and rinsed again under running water for 2 minutes. The slides were subsequently counterstained with eosin solution for 2-3 minutes and washed under running water for 30-60 seconds. Rehydration and clearing were performed by dipping the slides in 95% alcohol and absolute alcohol, followed by xylene (absolute I for 2 minutes, xylol I for 1 minute, and xylol II for 2 minutes). After staining is complete, the slide is dripped with Etellan and covered with a cover glass. The slide is then observed under a light microscope.21
Identification of active compounds through LC-MS/MS
The active compounds of the ethanol extract of red galangan rhizomes (Alpinia purpurata K. Schum) Rhizomes were identified through phytochemical test and LC-MS/MS. The red galangal sample was prepared using the Solid Phase Extraction (SPE) method, a solid-phase separation technique based on chromatography, utilizing a Waters HLB (Hydrophilic-Lipophilic-Balanced) stationary phase composed of N-vinylpyrrolidone (hydrophilic) and divinylbenzene (lipophilic). The mobile phase consisted of methanol for polar compounds and dichloromethane for non-polar compounds. The sample, dissolved in methanol, was introduced into a Waters column cartridge containing the stationary phase, then eluted with methanol, and the eluate was collected. The collected fraction was subsequently injected into the LC-MS/MS instrument for analysis. The results of LC-MS/MS data analysis will produce a chromatogram in the form of a peak height plot and the molecular weight of the compounds contained in the extract will be obtained so that you can know the number of compounds contained in each sample.22 The resulting chromatogram was processed using MassLynx V4.1 software to determine the mass spectra.
Results
Ethanol extract Red galangal rhizomes (Alpinia purpurata K. Schum)
The water content of red galangal rhizomes (Alpinia purpurata K. Schum) is shown in Table 1.
Table 1: Results of water content analysis
|
Test |
Wa (g) |
Wb (g) |
Water contant (%) |
|
I |
6,52 |
6,45 |
1,40 |
|
II |
6,51 |
6,44 |
1,39 |
|
III |
6,51 |
6,44 |
1,40 |
|
% Average water content: 1,39 % |
|||
Phytochemical Screening of Ethanol Extract of Red Galangal Rhizomes
The phytochemical screening test result of ethanol extract of red galangal rhizomes showed positive reaction for all secondary metabolites, as shown in Table 2
Table 2: Results of phytochemical screening of ethanol extract of red galanga rhizomes
|
Phytochemical test |
Reagen |
Result |
Description |
|
Alkaloid |
Mayer and Wagner reagent |
A cloudy precipitate appears |
+ |
|
Flavonoid |
Mg powder and HCl concentrated |
Red solution |
+ |
|
Polifenol |
FeCl3 |
Blackish brown solution |
+ |
|
Steroid and triterpenoid |
Asam asetat Anhidrat. (CH2CO)2O+H2SO4 Concetred |
A green solution and a red brown solution |
+ |
|
Saponin |
HCl |
Constant foam |
+ |
Compound Identification
Compound identification was carried out using the LC-MS/MS method. Analysis was carried out by comparing the mass spectrum of each peak with the database to obtain the metabolite compounds contained in the ethanol extract of red galanga (Alpinia purpurata K. Schum) rhizomes. The chromatogram result of the ethanol fraction of red galangal rhizomes are shown in Figure 1.
 |
Figure 1: Chromatogram of Ethanol Fraction of Red Galanga Rhizomes
|
The results of compound identification in samples of thick red galanga rhizome extract are shown in table 3. The differences in retention times for the same compound may be caused by factors such as an unbalanced prior to injection or imperfect sample preparation. Spectrum of ethanol extract of red galangal rhizomes at low and high energy positive ionization showing fragment ions. Table 3 shows that there are 4 suspected compounds found in the thick ethanol extract of red galangal rhizome. One of the compounds was perhexiline (C19H36N), which was identified at a retention time of 16.75 with a positive ionized m/z of 278.
Table 3: Results of compound identifikasi of red galanga rhizomes with LC-MS/MS
|
Retention time |
Precusor ion [M] |
Molecular formula |
Suspected compound |
|
12,60 |
318 |
C18 H39 NO3 |
Phytosphingosine |
|
12,75 |
519 |
C26 H38N12 |
N-[2-(Dimethylamino)ethyl]-N′-[4-(4-methyl-1-piperazinyl)phenyl]-6-[4-(2-pyrimidinyl)-1-piperazinyl]-1,3,5-triazine-2,4-diamine |
|
12,99 |
519 |
C26H38N12
|
N-[2-(Dimethylamino)ethyl]-N′-[4-(4-methyl-1-piperazinyl)phenyl]-6-[4-(2-pyrimidinyl)-1-piperazinyl]-1,3,5-triazine-2,4-diamine |
|
13,87 |
522 |
– |
– |
|
15,34 |
426 |
– |
– |
|
16,26 |
426 |
– |
– |
|
16,75 |
278 |
C19H36N |
Perhexiline |
|
17,23 |
514 |
– |
– |
Image of histopathological changes in breast cancer
The results of histopathological examination of breast tissue in mice are shown in Figure 4.
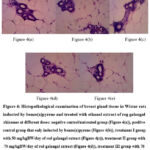 |
Figure 4: Histopathological examination of breast gland tissue in Wistar rats inducted by benzo(a)pyrene and treated with ethanol extract of reg galangal rhizomes at different doses:
|
Disscussion
Ethanol extract Red galangal rhizomes (Alpinia purpurata K. Schum)
The low water content of red galangal rhizomes indicates good drying quality and stability of simplicia, which is important for preventing microbial growth and degradation of bioactive compounds. The standard water content value set to maintain the quality of simplicia is ≤ 10%23. This moisture level meets estabilished quality standards, ensuring that the extraction process yields reliable and reproducible results.
Compound Identification
Phytochemical screening confirmed that the ethanol extract of red galangal rhizomes contains diverse secondary metabolites, including alkaloids, flavonoids, polyphenols, steroids/triterpenoids, and saponins. These classes of compounds are widely reported to possses antioxidant, anti-inflammatory, and anticancer activities. The presence of multiple bioactive groups suggests a synergistic effect that may contribute to the observed anticancer potential of the extract. LC-MS/MS analysis further supported the phytochemical findings by identifying specific compounds within the extract. The ethanol extract of red galangal rhizomes was found to contain alkaloid compound (Perhexiline) and polyphenols [N-[2-(Dimethylamino)ethyl]-N′-[4-(4-methyl-1-piperazinyl)phenyl]-6-[4-(2-pyrimidinyl)-1-piperazinyl]-1,3,5-triazine-2,4-diamine], both of which exhibit potential anticancer activity. Perhexiline also was known to be an anti-breast cancer agent that is able to inhibit the growth of breast cancer cell.24
Animal Treatment
Considering that testing of the anticancer activity of red galanga rhizomes (Alpinia purpurata K. Schum) used various variations in doses tested on Wistar rats induced by benzo(a)pyrene. So that data results were obtained regarding the effect of giving ethanol extract of red galangal rhizomes. Variations in doses used were the negative control group was not given any treatment, the positive control was only induced by benzo(a)pyrene and was not given ethanol extract of red galangal rhizome, the first treatment group was given ethanol extract of red galangal rhizome orally with a dose of 50 mg/kgBW/day, the second treatment group was given ethanol extract of red galangal rhizome orally at a dose of 70 mg/kgBW/day, and the third group was given ethanol extract of galangal rhizome red orally at a dose of 90 mg/kgBW/day. Test results at a dose of 90 mg/kgBW/day can effectively prevent cancer cell proliferation.
This dihydrodiol epoxide compound covalently bound the purine ecocyclic amino group on DNA with the deoxyadenosine (dA) or deoxyguanosine (dG) exocyclic amino group on DNA to form a stable adduct. Adducts formed on nucleotide bases result in a mutation process caused by errors in DNA repair or replication in lesions. Meanwhile, the formation of radical cations is influenced by the removal of one electron due to oxidation in the π electron system of the molecule. Cell cycle abnormalities due to induction of this compound will result in uncontrolled cell division and result in apoptosis of lymphoma cells. So, based on this mechanism, cancer cells will form in mouse breasts.25
According to previous studies, benzopyrene is a compound belonging to the same class as benzo[a]pyrene, namely polycyclic aromatic hydrocarbons (PAHs) was used to induce breast cancer in mice. The induction was carried out for 14 days in group B (treatment I: induction and breast excision) and group C (treatment II: induction and administration of red galangal extract at dose of 4.5 mg/head/day). This treatment caused cuboidal epithelial hyperplasia in the lactiferous ducts, resulting in thickened ductal walls composed of more than four layers of epithelial cells. Furthermore, induction with benzo[a]pyrene resulted in the death of three mice, which was attributed to improper administration during the gavage process, leading the compound to enter an incorrect physiological pathway.26
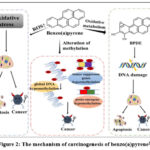 |
Figure 2: The mechanism of carcinogenesis of benzo(a)pyrene27
|
The toxicity of benzo(a)pyrene (BaP) to cells is mainly caused by DNA damage and oxidative stress from an increase in reactive oxygen species (ROS) production. BaP requires metabolic activation before reaction with DNA, and its metabolite benzo(a)pyrene-trans-7,8-dihydrodiol-9,10-epoxide (BPDE) induced genotoxicity by forming BPDE-DNA adduct, which showed mutagenic and carcinogenic potential in cells.28 Concurrently, BaP generates reactive oxygen species (ROS), causing oxidative stress and further DNA damage, which can trigger apoptosis or malignant transformation. Importantly, BaP also induces epigenetic changes, particularly aberrant DNA methylation.29 It reduces genome-wide DNA methylation by inhibiting DNA methyltransferases (DNMTs), while also causing promoter hypermethylation of tumor suppressor genes and hypomethylation of proto-oncogenes. These methylation changes disrupt normal gene expression, promote oncogene activation, and silence genes involved in DNA repair, cell cycle regulation, and apoptosis.30 This dual impact direct DNA damage and epigenetic dysregulation underlies BaP’s role in initiating and promoting various cancers, particularly in the respiratory, digestive, and reproductive systems.31
After 2 weeks, the mice in the treatment groups were administered a concentrated extract ofred galangal rhizomes for another two weeks, according to the specified dose for each treatment as part of the anticancer regimen. The mechanism of this phenolic compound as an anticancer is shown by the inhibitory effect of this compound on Nrf2 (Nuclear factor erythroid 2-related factor 2). Inhibition of Nrf2 by phenolic compounds leads to increased sensitivity of cancer cells to conventional anticancer therapy, reducing tumor growth and cancer cell death. Nrf2 has an important role in cellular defense against oxidative stress and exogenous toxicants.32 So Nrf2 has emerged as a therapeutic target for cancer prevention and therapy.33 However, because Nrf2 has a paradoxical role in cancer biology, it is necessary to understand the molecular pathways leading to the tumor suppression or oncogenic effects of Nrf2 for the development of drugs with highly specific and limited side effects. Natural products, including phenolic compounds, mediate the Nrf2/ARE pathway and can act as chemopreventive or chemotherapeutic agents.34
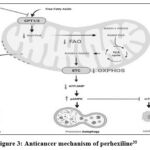 |
Figure 3: Anticancer mechanism of perhexiline35
|
Perhexiline’s inhibition of carnitine palmitoyltransferase 1 (CPT1) and CPT2 limits the entry of free fatty acids into the mitochondrial matrix, thereby inhibiting fatty acid oxidation (FAO).35 This can limit the production of the electron transport chain (ETC) coenzymes nicotinamide adenine dinucleotide (NADH) and flavin adenine dinucleotide (FADH2), which will inhibit oxidative phosphorylation (OXPHOS) and the formation of adenosine triphosphate (ATP).36 A decrease in the ratio of ATP to adenosine monophosphate (AMP) activates AMP-activated protein kinase (AMPK) through phosphorylation (pAMPK).37 pAMPK triggers autophagy, and inhibits cell proliferation by inhibiting mammalian target of rapamycin complex 1 (mTORC1).38 Additionally, perhexiline increases reactive oxygen species (ROS) levels, which compromise the integrity of mitochondrial membranes, leading to the release of cytochrome C and activation of caspases that initiate apoptosi.39
Image of histopathological changes in breast cancer
Based on these results (Figure 4), it can be concluded that benzo(a)pyrene induction is able to cause cell proliferation which is an early sign of the formation of cancer cells. Meanwhile, administration of red galangal rhizome extract to Wistar rats after benzo(a)pyrene induction at a dose of 70 mg/kgBW/day (Figure 4.d) and 90 mg/kgBW/day (Figure 4.e) resulted in suppression of cuboidal epithelial cell hyperplasia so that cancer cells did not form. However, the dose of 50 mg/kgBW/day (Figure 4.c) in treatment was not optimal in preventing cell proliferation caused by the benzo(a)pyrene compound. Cancer is excessive and uncontrolled cell proliferation, causing gene mutations which are a factor in the occurrence of cancer. Cells that undergo abnormal division will accumulate and from tumors. Tumor cells are divided into benign and malignant tumor cells. These malignant tumor cells are cancer cells.
Flauire of DNA repair processes leads to the accumulation of mutation in genes that regulate the cell cycle, such as the conversion of proto-oncogenes to oncogenes, resulting in uncontrolled cell proliferation. Excessive cell proliferation coupled with uncontrolled genetic changes can lead to cell transformation into cancer. This process, known as carcinogenesis, consists of several stages, including initiation or initial mutation, proliferation, and progression, characterized by invasive cancer growth. Overtime, continued proliferation increases the likelihood of additional mutations contributing to the development of malignant tumors.
Conclusion
Ethanol extract of red galangal rhizome (Alpinia purpurata K. Schum) contains alkaloid, flavonoid, polifenol, steroid, triterpenoid and saponin compounds. The doses of 70 mg/kgBW/day and 90 mg/kgBW/day affects the histopathology of Wistar rat breast cancer cells by preventing the proliferation of cuboidal epithelial cells, indicating its potential as an anticancer agent due to the presence of the secondary metabolite compound Perhexiline (cinnamaldehyde), a polyphenol compound contained in the rhizome.
Acknowledgment
Gratitude is expressed to all parties who helped and contributed to this research, thanks to the Dean of the Faculty of Mathematics and Natural Sciences, Udayana University for the opportunity given to the author.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
In this study, test animals were used, so the author validated the animal ethics approval certificate by the animal ethics committee of the Faculty of Veterinary Medicine, Udayana University with certificate number No:B/180/UN14.2.9/PT.01.04/2024 dated October, 2024. In this study, no clinical trials were conducted on humans.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Author’s Contribution
- Sri Wahjuni : First Author, Conceptualization, Methodology, Writing– Original Draft.
- I Gusti Agung Gede Bawa : Data Collection, Analysis, Writing – Review & Editing.
- A.I.A.Mayun Laksmiwati : Analysis, Writing – Review & Editing.
- Ida Ayu Raka Astiti Asih : Analysis, Writing – Review & Editing.
- Ni Made Nonik Sawitri : Analysis, Editor
Reference
- Saraswati, M., Sriatmi. A., and Jati, S.P. Analisis Implementasi Program Deteksi Dini Kanker Serviks Melalui Metode Inspeksi Visual Asam Asetat (IVA) di Puskesmas Kota Semarang. Jurnal Kesehatan Masyarakat. 2017;5(4): 85-94
- Report on Cancer Setting Priorities, Investing Wisely and Providing Care for All. Geneva: World Health Organization, 2020
- Cancer. Geneva: World Health Organization, 2022 [Cited 2025 Oct 19. Available from: : https://www.who. int/health-topics/cancer#tab=tab_1.
- Kementerian Kesehatan Republik Indonesia. 2018. Hasil Utama RISKESDAS.
- Huang, J., Chan, PS, Lok, V., Chen, X., Ding, H., Jin, Y., & Wong, MC. Insiden Global dan Kematian Akibat Kanker Payudara: Analisis Tren. Penuaan (Albany NY). 2021;13(4): 5748
CrossRef - Bukowska, B., Mokra, K., & Michałowicz, J. Benzo [a] pyrene—Environmental occurrence, human exposure, and mechanisms of toxicity. International Journal Of Molecular Sciences. 2022;23(11): 6348
CrossRef - Ledet, M. M., Oswald, M., Anderson, R., & Van de Walle, G. R. Differential signaling pathway activation in 7, 12-dimethylbenz [a] anthracene (DMBA)-treated mammary stem/progenitor cells from species with varying mammary cancer incidence. Oncotarget. 2018;9(67): 32761
CrossRef - Abbass, M., Chen, Y., Arlt, V. M., & Stürzenbaum, S. R. Benzo[a] pyrene and Caenorhabditis Elegans: Defining the Genotoxic Potential in an Organism Lacking the Classical CYP1A1 Pathway. Archives of toxicology. 2021;95 (1): 1055-1069
CrossRef - Arini, H.D., Medina, B.F., and Yuliawati, A. Analisis dan Faktor yang Mempengaruhi Biaya Kemoterapi Pasien Kanker Payudara. Journal of Pharmacy and Science. 2023;7(1): 46-53
CrossRef - Cahyaningsih, R., Brehm, J. M., & Maxted, N. Setting the priority medicinal plants for conservation in Indonesia. Genet Resour Crop Evol. 2021;68:2019–2050
CrossRef - Pangestuti, D., Harris, B., & Rahman, T. K. Comparison of the Effectiveness between Red Galangal Rhizome’s (Alpinia purpurata schum) Extract and Ketoconazol 2% to Inhibit Growth of Dermatophyte Fungi In Vitro. Buletin Farmatera. 2020;5(1):156-162
CrossRef - Liu, D., You, P., Luo, Y., Yang, M., & Liu, Y. Galangin Induces Apoptosis In MCF-7 Human Breast Cancer Cells Through Mitochondrial Pathway and Phosphatidylinositol 3-Kinase/Akt Inhibition. Pharmacology. 2018;102(1-2):58-66
CrossRef - Pramushinta, I. A. K., & Ajiningrum, P. S. Uji Aktivitas Sel Kanker dengan menggunakan senyawa Flavonoid dari Lengkuas (Alpinia Galanga). STIGMA: Jurnal Matematika dan Ilmu Pengetahuan Alam UNIPA. 2017;10(2):89 – 91
CrossRef - Khairullah, A. R., Solikhah, T. I., Ansori, A. N. M., Fadholly, A., Ramandinianto, S. C., Ansharieta, R., & Anshori, A. A Review of an Important Medicinal Plant: Alpinia galanga (L.) willd. Syst Rev Pharm. 2020;11(10):387-395
- Wendersteyt, N.V., Wewengkang, D.S., and Abdullah, S.S. Uji Aktivitas Antimikroba dari Ekstrak dan Fraksi Ascidian Herdmania momus dari Perairan Pulau Bangka Likupang Terhadap Pertumbuhan Mikroba Staphylococcus aureus, Salmonella typhimurium dan Candida albicans. Pharmacon. 2021;10(1):706-712
CrossRef - Putri, D. M., and Lubis, S. S. Skrining Fitokimia Ekstrak Etil Asetat Daun Kalayu (Erioglossum rubiginosum (Roxb.) Blum). Amina. 2020;2(3):120-125
CrossRef - Agustina, S., Ruslan, and Wiraningtyas, A. Skrining Fitokimia Tanaman Obat di Kabupaten Bima. Cakra Kimia. 2016;4(1):71-75
- Soesilawati, P. Histologi Kedokteran Dasar. Surabaya: Airlangga University Press, 2020
- Digambiro, R. A., and Parwanto, E. Panduan Prosesing dan Pewarnaan Jaringan dalam Histopatologi. Klaten: Penerbit Lakeisha, 2024
- Mamay, Mutmaina, G. N., dan Nurahma, I. A. Penggunaan Sabun Pencuci Piring Sebagai Pengganti Xilol Dalam Proses Deparafinasi Pewarnaan Hematoksilin-Eosin. Medicra (Journal of Medical Laboratory Science Technology). 2022;5(1):47 – 55
CrossRef - Mangurana, W. O. I., Yusnaini, Y., & Sahidin, S. Analisis LC-MS/MS (Liquid Crhomatogaph Mass Spectrometry) dan Metabolit Sekunder Serta Potensi Antibakteri Ekstrak n-heksana Spons Callyspongia aerizusa yang Diambil pada Kondisi Tutupan Terumbu Karang Yang Berbeda di Perairan Teluk Staring. Jurnal biologi tropis. 2019;19(2):131-141
CrossRef - Willian, N., dan Pardi, H. Buku Ajar Pemisahan Kimia. Tanjungpinang: UMRAH Press, 2022
- Wijaya, A., and Noviana, N. Penetapan Kadar Air Simplisia Daun Kemangi (Ocimum basilicum) Berdasarkan Perbedaan Metode Pengeringan. Jurnal Riset Kefarmasian Indonesia. 2022;4(2): 185-194
- Dhakal, B., Li, C. M. Y., Li, R., Yeo, K., Wright, J. A., Gieniec, K. A., & Fenix, K. 2022. The Antianginal Drug Perhexiline Displays Cytotoxicity Against Colorectal Cancer Cells In Vitro: A Potential For Drug Repurposing. Cancers. 2022;14(4):1043
CrossRef - Wuyung, P. E. Induksi DMBA dalam Karsinogenesis Kelenjar Payudara. Pratista Patologi. 2016;5(1):44-51
- Liangan, R., Kairupan, C., & Durry, M. Pengaruh Pemberian Ekstrak Lengkuas (Alpinia galanga) Terhadap Gambaran Histologik Payudara Mencit (Mus musculus) yang Diinduksi Benzo(a)pyrene. eBiomedik. 2015;3(1): 480-484
CrossRef - Wang, H., Liu, B., Chen, H., Xu, P., Xue, H., & Yuan, J. Dynamic changes of DNA methylation induced by benzo(a)pyrene in cancer. Genes and Environment. 2023;45(1):21
CrossRef - Shiizaki, K., Kawanishi, M., and Yagi, T. Modulation of benzo[a]pyrene–DNA adduct formation by CYP1 inducer and inhibitor. Genes and Environment. 2017;39(14):1-8
CrossRef - Pandelides, Z., Sturgis, M.C., Thornton, C., Aluru, N., Willet, K.L. Benzo[a]pyrene-induced multigenerational changes in gene expression, behavior, and DNA methylation are primarily influenced by paternal exposure. Toxicology and Applied Pharmacology. 2023;469:116545
CrossRef - Geissler, F., Nesic, K., Kondrashova, O., Dobrovic, A., Swisher, E.M., Scott, C.L., and Wakefield, M.J. The role of aberrant DNA methylation in cancer initiation and clinical impacts. Therapeutic Advances in Medical Oncology. 2024;16:1-23
CrossRef - Mondal A, Sarkar A, Das D, Sengupta A, Kabiraj A, Mondal P, Nag R, Mukherjee S, Das C. Epigenetic orchestration of the DNA damage response: Insights into the regulatory mechanisms. Int Rev Cell Mol Biol. 2024;387:99-141.
CrossRef - Ngo, V. And Duennwald, M.L. Nrf2 and Oxidative Stress: A General Overview of Mechanisms and Implications in Human Disease. 2022;11:1-27
CrossRef - Lin L, Wu Q, Lu F, Lei J, Zhou Y, Liu Y, Zhu N, Yu Y, Ning Z, She T, Hu M. Nrf2 signaling pathway: current status and potential therapeutic targetable role in human cancers. Front Oncol. 2023;13:1184079.
CrossRef - Sharifi-Rad, J., Seidel, V., Izabela, M., Monserrat-Mequida, M., Sureda, A., Ormazabal, V., & Cho, W. C. Phenolic compounds as Nrf2 inhibitors: potential applications in cancer therapy. Cell Communication and Signaling. 2023;21(1):1-18
CrossRef - Dhakal, B., Tomita, Y., Drew, P., Price, T., Maddern, G., Smith, E., & Fenix, K. Perhexiline: Old Drug, New Tricks? A Summary of Its Anti-Cancer Effects. Molecules. 2023;28(8):1-19
CrossRef - Amjad, S., Nisar, S., Bhat, A.A., Shah, A.R., Frenneaux, M.P., Fakhro, K., Haris, M., Reddy, R., Patay, Z., Baur, J., and Bagga, P. Role of NADþ in regulating cellular and metabolic signaling pathways. Moolecular Metabolism. 2021;49:1-17
CrossRef - Yan, Y., Zhou, E., Xu, H.E., and Melcher, K. Structure and Physiological Regulation of AMPK. Int J Mol Sci. 2018;19(11):1-15
CrossRef - Mandic, M., Marjanovic, M.M., Janjetovic, K., Bosnjak, M., Trajkovic, L.H., Trajkovic, V., and Vucicevic, L. Multifaceted role of AMPK in autophagy: more than a simple trigger?. American Journal Of Physiology Cell Physiology. 2025;329:1380-1397
CrossRef - Maureen, R. D., Diana, A. A. B. Activation of apoptosis signalling pathways by reactive oxygen species. Biochimica et Biophysica Acta (BBA) – Molecular Cell Research.2016;1863(12):2977-2992
CrossRef







