Manuscript accepted on :25-09-2025
Published online on: 03-11-2025
Plagiarism Check: Yes
Reviewed by: Dr. Emmanuel Dike
Second Review by: Dr. Satya Namani
Final Approval by: Dr. Patorn Piromchai
Sireesha Kalva1* , Sandeep Nayani2, Kandregula Keerthika1
, Sandeep Nayani2, Kandregula Keerthika1 , Kavati Supriya1
, Kavati Supriya1 , Tanzeel Fathima1
, Tanzeel Fathima1 and Syeda Nusrath Fathima1
and Syeda Nusrath Fathima1 .
.
1Department of Pharm D, Sri Venkateshwara College of Pharmacy, Osmania University, Madhapur, Hyderabad, India.
2Department of Neurology, Apollo Hospital, Hyderabad, India.
Corresponding Author E-mail:sireeshakalva@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3305
Abstract
Epilepsy, ranking as the second leading neurological cause of reduced disability-adjusted life years, underscores the necessity for effective long-term anti-epileptic drug therapy. Levetiracetam (Livipil), a newer anti-seizure medication, has gained prominence for its broad-spectrum efficacy in treating symptomatic and idiopathic epilepsy, offering pharmacokinetic advantages and a wide therapeutic range. Despite its efficacy, levetiracetam usage has been associated with behavioural side effects, including irritability, agitation, and aggression. Our study aims to conduct a comprehensive evaluation of the safety profile of levetiracetam in adults undergoing epilepsy therapy, focusing on the prevalence of adverse events, behavioural alterations, cognition abnormalities, and dose-dependent effects. A comprehensive evaluation was undertaken at Apollo Hospital, involving 100 patients receiving levetiracetam therapy, to assess psychotic adverse events associated with the medication. This rigorous methodology ensured a comprehensive understanding of the potential psychological effects of levetiracetam treatment in the patient cohort. Our study of 100 patients on levetiracetam highlighted several significant adverse effects, including changes in social behavior, depression, somnolence, anxiety, and cognitive impairment. Notably, 76% of patients showed changes in social behavior, while 78% reported depression. Somnolence affected all participants, impairing daily activities. Anxiety was present in 20% of patients, and 79% experienced varying degrees of cognitive impairment. We can conclude that despite of some consistency with existing literature, further research is needed to understand the underlying mechanisms and develop strategies to mitigate these adverse effects.
Keywords
Adverse event; Epilepsy; Levetiracetam; Neurological Cause; Psychological effects; Somnolence
Download this article as:| Copy the following to cite this article: Kalva S, Nayani S, Keerthika K, Supriya K, Fathima T, Fathima S. N. Incidence of Psychiatric Adverse Events with Levetiracetam in Epilepsy Patients in India. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Kalva S, Nayani S, Keerthika K, Supriya K, Fathima T, Fathima S. N. Incidence of Psychiatric Adverse Events with Levetiracetam in Epilepsy Patients in India. Biomed Pharmacol J 2025;18(4). Available from: https://bit.ly/4oP4Q1O |
Introduction
Epilepsy is a long-standing neurologic ailment with a series of events involving different kinds of seizures, syndromes and comorbidities.1 It is regarded as one of most common serious brain conditions, affecting more than 70 million individuals worldwide. Its occurrence is bimodally distributed, with infants and children at highest risk.2 Seizures are normally classified as generalized seizures, focal seizures and epileptic spasms. Generalized seizures originate in bilateral distributed neuronal networks whereas focal seizures originate in neuronal networks limited to one part of cerebral hemisphere.3 Epileptic seizures occur in particular brain regions and can either be localized (“focal” or “partial”) or spread throughout entire cerebral hemispheres (“generalized” seizures).4 Additionally, physical traumas like fractures and psychological disorders like depression, anxiety, etc. can be brought on by epilepsy.5 Epilepsy impacts development of learning as well as cognitive functions through numerous factors such as etiology, age of onset, seizure type, duration and severity, interictal epileptic form discharges and drug treatment.6 Primary treatment for epileptic patients is antiepileptic drugs (AEDs). It has been estimated that while AEDs control roughly two-thirds of epileptic episodes, about 30% of cases of epilepsy remain resistant to them. Levetiracetam, one of most commonly utilized second-generation anti-epileptic drugs, received FDA approval in 1999 for treatment of adult focal epilepsy.7 A novel antiepileptic drug named levetiracetam has been employed for treating myoclonic, partial, and tonic-clonic seizures. It serves as an attractive first-line or adjuvant treatment for epileptic seizures since it is more effective in focal seizures, had better safety profile, fewer drug interactions, fewer adverse drug reactions, and doesn’t require serum level monitoring.8 Levetiracetam acts by binding itself to unique synaptic vesicle protein 2A (SV2A), component of secretory vesicle membranes that facilitates release of vesicular neurotransmitters that are dependent on calcium. It seems that levetiracetam’s binding to SV2A slows down rate at which vesicles release.9 Levetiracetam has demonstrated additional modulating effects on AMPA (α-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid) receptor. Despite its effectiveness, treatment with levetiracetam has been linked to the potential development of behavioral adverse events, prompting discontinuation of the drug. Critically ill patients with neurological injuries face an elevated risk of experiencing these events due to their distinctive pathophysiology and the introduction of levetiracetam may potentially intensify this risk. Behavioral adverse effects were reported more often in pediatric than adult case series.10 However, Levetiracetam is not associated with serious systemic adverse effects.11
Materials and Methods
A comprehensive evaluation was undertaken at Apollo Hospital, involving 100 patients receiving levetiracetam therapy, to assess psychotic adverse events associated with the medication. Utilizing a structured questionnaire, meticulously designed to capture relevant data, each patient underwent a thorough assessment. This rigorous methodology ensured a comprehensive understanding of the potential psychological effects of levetiracetam treatment in the patient cohort. The study was conducted with strict adherence to ethical guidelines and medical standards, aiming to contribute valuable insights into the safety profile of this widely used anti-epileptic medication.
Informed Consent form: Data is collected from the patients after signing the consent forms.
Results
Our study emphasised the psychiatric side effects of Levetiracetam when administered as monotherapy. With levetiracetam therapy, we observed the development of psychotic adverse effects like difference in social behaviour, depression and somnolence.
 |
Figure 1: Distribution of patients based on their social behaviour. |
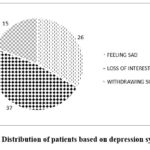 |
Figure 2: Distribution of patients based on depression symptoms. |
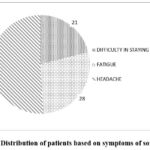 |
Figure 3: Distribution of patients based on symptoms of somnolence. |
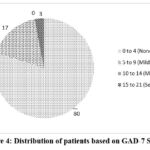 |
Figure 4: Distribution of patients based on GAD-7 Scores. |
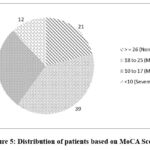 |
Figure 5: Distribution of patients based on MoCA Scores. |
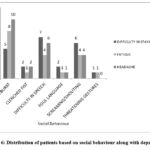 |
Figure 6: Distribution of patients based on social behaviour along with depression. |
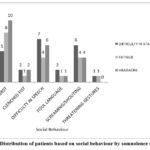 |
Figure 7: Distribution of patients based on social behaviour by somnolence symptoms. |
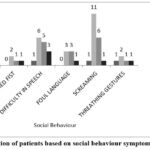 |
Figure 8: Distribution of patients based on social behaviour symptoms by MoCA Scores. |
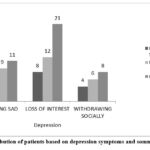 |
Figure 9: Distribution of patients based on depression symptoms and somnolence symptoms. |
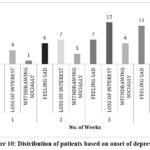 |
Figure 10: Distribution of patients based on onset of depression. |
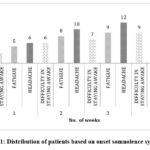 |
Figure 11: Distribution of patients based on onset somnolence symptoms. |
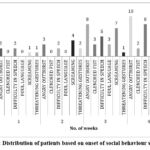 |
Figure 12: Distribution of patients based on onset of social behaviour symptoms. |
The Figure 13 represents the Corelation between levipil usage vs GAD-7 and MoCA Scores.
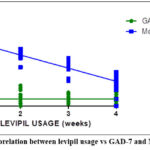 |
Figure 13: Corelation between levipil usage vs GAD-7 and MoCA Scores. |
Discussion
Among the various types of psychotic disorders in epilepsy, antiepileptic drug (AED)-induced psychotic disorder (AIPD) represents an iatrogenic, adverse drug reaction. Prevalence of AIPD has been reported to range from 1.0% to 8.4% in clinical trials of AEDs.12 Despite the favorable outcomes of LEV in Epilepsy, several studies have suggested that this drug has negative psychotropic effects, including irritability, aggressiveness, suicidality, mood disorders, and other psychiatric symptoms in both children and adult patients. 13-21
Following are the obsevations drawn from our study.
The data in fig 1 displays the prevalence of various changes in social behaviour pattern like angry outburst, screaming/shouting, difficulty in speech, foul language, threatening gestures and clenched fists reported by the patient. 76% of the subjects experienced changes in social behaviour. The highest were angry outbursts reported by 24% of patients and the lowest were clenched fists represented by 4%. These changes can severely affect a patient’s social support system and contribute to overall distress.
The prevalence of various signs of depression like loss of interest, feeling sad and withdrawing socially. The Figure 2 displays 78% of them experienced symptoms of depression, indicating that there is high chance of getting depression for patients undergoing levetiracetam therapy. This highlights the need for careful monitoring and timely intervention to manage mental health issues that may arise during treatment.
Central nerve system (CNS)-related adverse events such as somnolence, lethargy, dizziness, headache, and tiredness were common, but also transient and dose-related.22-23 Somnolence is a state of drowsiness or extreme sleepiness. The most frequently reported symptom was headache with 51%. Figure 3 highlights the importance of balancing therapeutic efficacy with quality of life considerations in prescribing levetiracetam.
These patients concomitantly complained of CNS-related adverse events due to LEV, such as anxiety, asthenia, and memory problems, and had experienced a recent extreme stress. The “Generalized Anxiety Disorder” (GAD-7) Assessment Scale is among most trusted and considered score for determining anxiety. The Figure 4 displays the scores of the patients which analyse the levels of anxiety experienced by the patients. The patients scoring between 0 – 4 experience no anxiety, 5 – 9 mild anxiety, 10 -14 moderate anxiety as well as 15 – 21 severe anxiety. 17% experience mild anxiety and only 3% of them experience severe anxiety. Patients experiencing heightened anxiety may struggle with increased stress and agitation, which can further exacerbate other adverse effects such as depression and cognitive impairment.
Similarly, cognitive impairment has been reported to be related to psychosis in patients with epilepsy. The level of intelligence is assessed by a person’s cognitive ability. The decline in cognitive skills is measured using MoCA score. The Figure 5 represents the scores of the patients which analyse the levels of cognitive impairment experienced by the patients. The patient’s scoring >=26 experience no cognition,18 – 25 mild cognitive impairment, 10 – 17 moderate cognitive impairment as well as 10 severe cognitive impairment. 39% patients experience mild cognitive and 12% patients experience severe cognitive impairment. Levetiracetam monotherapy patients are found to develop cognitive impairment of various degrees in 79% of patients which should be given a greater attention. Cognitive impairment poses challenges in adhering to treatment regimens and participating in therapeutic activities.
The Figure 6 displays the association between social behaviour and depression symptoms. The findings from the figure suggest that social behaviour and depression associates with each other in patients with levetiracetam therapy.
The Figure 7 represents the association between social behaviour symptoms and somnolence symptoms. Both the symptoms of social behavioural changes and somnolence were equally observed in patients treated with levetiracetam. Hence association exists between social behaviour and somnolence.
The Figure 8 represents the association between changes in social behaviour and cognitive impairment. The data suggests that levetiracetam affects the social behaviour differently on cognitive status (mild, moderate, severe). Mild to moderate levels of cognition loss is seen associated with social behaviour.
The Figure 9 represents the association between depressive symptoms and somnolence symptoms experienced by the patients. The findings suggest that levetiracetam can cause depressive symptoms along with somnolence, indicating a need for careful monitoring to manage these side effects.
In our study, while observing depression symptoms in patients undergoing epileptic therapy with levetiracetam, it was noted that depressive symptoms became more prominent over time. The Figure 10 represents the distribution of patients based on onset of depression. The data suggests a significant correlation between prolonged (4weeks) levetiracetam use and the onset of depressive symptoms.
In our study, while observing somnolence symptoms in patients undergoing epileptic therapy with levetiracetam, it was noted that somnolence symptoms became more prominent over time. The Figure 11 represents the distribution of patients based on onset of somnolence symptoms. The data suggests that, there exists a significant correlation between prolonged levetiracetam use and the onset of somnolence symptoms.
In our study, while observing social behaviour changes in patients undergoing epileptic therapy with levetiracetam, it was noted that changes in social behaviour pattern, became more prominent over time. The Figure 12 represents the distribution of patients based on onset of social behaviour symptoms. The graph provides a clear indication that social behaviour symptoms among the patients intensify over the four-week period. The data underscores the need to address the growing severity of social behaviour symptoms as they become worse with use of levetiracetam for long period.
Conclusion
Despite its efficacy, levetiracetam usage has been associated with behavioural side effects, including irritability, agitation, and aggression. These central nervous system adverse drug reactions occur in up to 16% of patients, often necessitating discontinuation. Personalized medicine approaches hold promise in identifying individuals at low risk for adverse effects, thus improving treatment outcomes and patient safety.
Our study of 100 patients receiving levetiracetam revealed significant adverse effects, including changes in social behaviour, depression, somnolence, anxiety, and cognitive impairment. These findings highlight a concerning impact on patient quality of life, underscoring the need for careful monitoring and management of these side effects in clinical practice. Our study investigating the adverse effects of levetiracetam in patients has provided valuable insights into the spectrum and severity of side effects associated with this antiepileptic drug. The findings highlight several significant issues that warrant careful consideration and further research.
Future research directions should focus on larger-scale studies to validate and extend our findings. Investigations into the specific mechanisms through which levetiracetam affects mood and cognition could lead to improved understanding and management of these adverse effects.
The identified adverse effects emphasize significance of balanced treatment plan that takes into account both therapeutic benefits as well as potential risks. By advancing our understanding of these effects and refining management strategies, we can improve patient outcomes and ensure more effective and compassionate care.
However, detailed analysis of the clinical profile of the psychotic episodes was lacking in these studies, which tend to rely on screening questionnaires to ascertain psychiatric symptoms with few patients undergoing structured interview by psychiatrist.
Acknowledgement
We are thankful to our college principal late Sri Bhagavan Raju for his support through out the project.
Funding Source
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
Data is collected from the patients after signing the consent forms.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Author Contributions:
- Sireesha Kalva: Supervision & Project Supervision.
- Sandeep Naini: Coceptulisation & Project administration.
- K.Keerthika: Data Collection & Methodology & Analysis.
- Tanzeel Fatima: Data Collection & Analysis.
- Nusrat Fatima: Witing the original draft.
- Supriya: Review and editing.
References
- Chandrarathna N, Parida A, Manju V and Adiga U.S. Drug Utilization Study in Epilepsy in a Tertiary Care Hospital. Biomed Pharmacol J. 2019; 12(2): 697-701.
CrossRef - Thijs R.D, Surges R, Brien T.J and Sander J.W. Epilepsy in adults. Lancet. 2019; 393(10172): 689-701.
CrossRef - Stafstrom C.E and Carmant L. Seizures and epilepsy: An overview for neuroscientists. Cold Spring Harb Perspect Med. 2015; 5(6): 224-26.
CrossRef - Bozzi Y and Borrelli E. The role of dopamine signaling in epileptogenesis. i Neurol. 2013; 7(157): 1-12.
CrossRef - Dwajani S, Ravi L, Bindu C.M and Prema R. Effect of Antiepileptic Drugs on Serum Vitamin B12 and Folic Acid Among Children with Epilepsy in Bangalore. Biomed Pharmacol J. 2024; 17(1): 263-269.
CrossRef - Zaki M.M, Moustafa R.S.I, Shady M.M.A and Youness E.R. Evaluation of Acetylcholinesterase and Acetylcholine Levels in Children with Idiopathic Epilepsy. Biomed Pharmacol J. 2023; 16(2): 715-719.
CrossRef - Hegazy M.I, Asaad A.M, Rashad L.A and Ahmed H.H. Combined Treatment of Levetiracetam and Mesenchymal Stem Cells Reverses Behavioural and Biochemical Aberrations in the Acute Phase of Experimental Epilepsy. Biomed Pharmacol J. 2022; 15(1): 91-115.
CrossRef - Praveen A.N, Panchaksharimath P and Nagaraj K. A Comparative Study to Evaluate the Efficacy and Safety of Levetiracetam as an Add-on to Carbamazepine and Phenytoin in Focal Seizures at a Tertiary Care Hospital. Biomed Pharmacol J. 2020; 13(1): 383-390.
CrossRef - Sills G.J and Rogawski M.A. Mechanisms of action of currently used antiseizure drugs. Neuropharmacology. 2020; 16(8):107966.
CrossRef - Micheal S, John P.H.B, Spiro S, Carley M, Sulaiman A and Gretchen M.B. Levetiracetam-associated behavioural adverse events in neurocritical care patients. 2023; 43(2).122-128.
CrossRef - Abou-Khalil B. Levetiracetam in the treatment of epilepsy. Neuropsychiatr Dis Treat. 2008; 4(3): 507-23.
CrossRef - Piedad J, Rickards H, Besag FM, Cavanna AE. Beneficial and adverse psychotropic effects of antiepileptic drugs in patients with epilepsy: a summary of prevalence, underlying mechanisms and data limitations. CNS Drugs 2012; 26: 319–35
CrossRef - Hurtado B, Koepp MJ, Sander JW, Thompson PJ. The impact of levetiracetam on challenging behavior. Epilepsy Behav 2006; 8:588-592.
CrossRef - Weintraub D, Buchsbaum R, Resor SR Jr, Hirsch LJ. Psychiatric and behavioral side effects of the newer antiepileptic drugs in adults with epilepsy. Epilepsy Behav 2007;10:105-110.
CrossRef - Bootsma HP, Ricker L, Diepman L, Gehring J, Hulsman J, Lambrechts D, et al. Levetiracetam in clinical practice: long-term experience in patients with refractory epilepsy referred to a tertiary epilepsy center. Epilepsy Behav 2007; 10:296-303.
CrossRef - Mula M, Sander JW. Suicidal ideation in epilepsy and levetiracetam therapy. Epilepsy Behav 2007; 11:130-132.
CrossRef - Mula M, Trimble MR, Sander JW. Are psychiatric adverse events of antiepileptic drugs a unique entity? A study on topiramate and levetiracetam. Epilepsia 2007; 48:2322-2326.
CrossRef - Helmstaedter C, Fritz NE, Kockelmann E, Kosanetzky N, Elger CE. Positive and negative psychotropic effects of levetiracetam. Epilepsy Behav 2008; 13:535-541.
CrossRef - Labiner DM, Ettinger AB, Fakhoury TA, Chung SS, Shneker B, Tatum Iv WO, et al. Effects of lamotrigine compared with levetiracetam on anger, hostility, and total mood in patients with partial epilepsy. Epilepsia 2009; 50:434-442.
CrossRef - De la Loge C, Hunter SJ, Schiemann J, Yang H. Assessment of behavioral and emotional functioning using standardized instruments in children and adolescents with partial-onset seizures treated with adjunctive levetiracetam in a randomized, placebo-controlled trial. Epilepsy Behav 2010; 18:291-298.
CrossRef - Andersohn F, Schade R, Willich SN, Garbe E. Use of antiepileptic drugs in epilepsy and the risk of self-harm or suicidal behavior. Neurology 2010; 75:335-340.
CrossRef - Cereghino JJ, Biton V, Abou-Khalil B, Dreifuss F, Gauer LJ, Leppik I. Levetiracetam for partial seizures: results of a double-blind, randomized clinical trial. Neurology 2000;55:236-242.
CrossRef - Heo K, Lee BI, Yi SD, Huh K, Kim JM, Lee SA, et al. Efficacy and safety of levetiracetam as adjunctive treatment of refractory partial seizures in a multicenter open-label single-arm trial in Korean patients. Seizure 2007;16: 402-409.
CrossRef







