Manuscript accepted on :11-11-2025
Published online on: 03-12-2025
Plagiarism Check: Yes
Reviewed by: Dr. Arif Ansori
Second Review by: Dr. Nasir Abdelrafie
Final Approval by: Dr. Eman Refaat Youness
Mohamed Nabil Ibrahim1,2* , Eman Fawzy El Azab1
, Eman Fawzy El Azab1 and Mohamed Eid Anwar3
and Mohamed Eid Anwar3
1Department of Clinical Laboratory Sciences, College of Applied Medical Sciences -Qurrayat, Jouf University, Al-Qurayyat, Saudi Arabia
2Department of Microbiology, Faculty of Science, Ain Shams University, Cairo, Egypt.
3Department of Hematology, Shebeen El-Kom Hospital, El-Monofia, Egypt
Corresponding Author E-mail: Mnmicro2010@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3293
Abstract
Herpes simplex virus type 2 (HSV-2) is still a common sexually transmitted disease (STD) linked to obstetric complications, such as miscarriage, premature birth, and herpes in newborns. Despite its prevalence worldwide, little information is known about pregnant Egyptian women. This cross-sectional hospital-based study aimed to evaluate the seroprevalence of HSV-2-specific IgG and IgM antibodies and examine their sociodemographic and clinical correlations by enrolling 185 pregnant Egyptian women aged 22 to 48. Serum samples were tested using the enzyme-linked immunosorbent assay (ELISA) technique. Statistical analyses were conducted using the chi-square test, with p < 0.05 considered statistically significant. 67% of the participants were positive for HSV-2 IgG, reflecting past infection, while 19.5% were positive for HSV-2 IgM, indicating recent or active infections. IgG positivity was strongly related to low socioeconomic status (p = 0.001), illiteracy (p = 0.001), and first-trimester pregnancy (p = 0.001). However, IgM positivity was clearly correlated with the gestational stage (p = 0.001). The high seroprevalence of HSV-2 among pregnant Egyptian women signposts the importance of both prevention strategies and routine prenatal screening. Improved healthcare accessibility and greater health literacy among women may play a critical role in reducing the adverse obstetric outcomes related to HSV-2 infection.
Keywords
Herpes simplex virus type 2 (HSV-2); HSV-2 IgG; HSV-2 IgM; Maternal infection; Pregnancy; Prenatal screening; Seroprevalence
Download this article as:| Copy the following to cite this article: Ibrahim M. N, El-Azab E. F, Anwar M. E. HSV-2 Seroprevalence and Adverse Pregnancy Outcomes among Egyptian Women: A Cross-Sectional Study. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Ibrahim M. N, El-Azab E. F, Anwar M. E. HSV-2 Seroprevalence and Adverse Pregnancy Outcomes among Egyptian Women: A Cross-Sectional Study. Biomed Pharmacol J 2025;18(4). Available from: https://bit.ly/4apZ9Dw |
Introduction
Herpes simplex virus type 2 (HSV-2) is a widespread sexually transmitted alpha-herpesvirus that affects nearly 15–25% of the global population, with noticeably higher infection rates observed in developing countries and among groups exhibiting high-risk sexual behaviors.¹ HSV-2 represents the primary cause of genital herpes and is linked to substantial obstetric challenges, as well as neonatal herpes a rare but life-threatening condition associated with severe morbidity and mortality.² Structurally, HSV-2 consists of a double-stranded DNA genome enclosed within a protein capsid and enveloped by a tegument having immunogenic glycoproteins that facilitate viral entry, replication, and immune evasion.³ Following initial infection, the virus establishes latency in the sensory ganglia and may reactivate periodically, leading to recurrent genital lesions or asymptomatic viral shedding, thereby sustaining transmission potential.⁴
Transmission occurs mainly through direct sexual contact and exposure to infected mucosal secretions such as vaginal fluid, semen, or saliva.⁵ Vertical transmission from mother to infant during childbirth constitutes a major clinical concern, particularly when primary infection or active genital lesions are present in late pregnancy.⁶ Various demographic and behavioral factors including age, sexual activity, socioeconomic status, and healthcare access—strongly influence HSV-2 seroprevalence across populations.⁷,⁸ Although congenital and perinatal HSV-2 infections occur less frequently than those caused by other viruses such as cytomegalovirus (CMV), their clinical consequences are profound, often resulting in neurological impairment, developmental delay, or neonatal death.⁹ The likelihood of neonatal transmission may reach 30–50% when primary maternal infection occurs during the third trimester and vaginal delivery takes place in the presence of active lesions.¹⁰ Because asymptomatic viral shedding complicates prevention and diagnosis, routine serological testing of pregnant women becomes essential to identify those at increased risk.¹¹ Assessing HSV-2 seroprevalence and its associated determinants among pregnant women is therefore crucial for guiding national prevention programs and obstetric counseling. This cross-sectional study aimed to determine the prevalence of HSV-2-specific IgG and IgM antibodies in Egyptian pregnant women and to explore their relationship with sociodemographic and obstetric characteristics to inform targeted screening and preventive interventions.
Materials and Methods
Ethical Approval
On March 24, 2023, the Ethics Committee of Shebeen El-Kom Educational Hospital in Egypt examined and accepted the research protocol. Prior to their involvement, all participants were briefed on the goals and methods of the study, and written consent was acquired in compliance with the institutional ethical guidelines.
Study Design and Population
This study evaluated the seroprevalence of Herpes simplex virus type 2 (HSV-2) in pregnant women using a cross-sectional methodology. In all, 185 volunteers between the ages of 22 and 48 were chosen from the obstetrics and gynecology departments of El-Menya General Hospital and Shebeen El-Kom Hospital in Egypt. Regardless of parity or gestational stage, recruitment focused on women who attended regular prenatal clinics during the study period and gave their voluntary agreement to participate.
Sampling Technique
Venous blood samples were collected under aseptic conditions by qualified medical personnel. Serums were separated from the samples by centrifugation and kept at -20°C until they were analyzed in a lab. Through structured interviews utilizing a validated questionnaire created specifically for this study, sociodemographic and obstetric data such as age, educational attainment, socioeconomic status, and gestational age were acquired.
Sample Size Determination
In accordance with an estimated 20% HSV-2 seroprevalence among pregnant women in the area, a 95% confidence level, and a 5% margin of error, and the formula for sample size calculation N= [Z2 * P(1 – P)] / E2. In this equation, N= required sample size – Z = Z-value (1.96 for 95% confidence) – P = estimated prevalence (50%) – E = margin of error (5%), the minimum required sample size was approximately 185. For reliable prevalence estimation and subgroup analysis, this provided sufficient statistical strength.
Laboratory Investigations
HSV-2-specific IgG and IgM antibodies have been evaluated serologically using the enzyme-linked immunosorbent assay (ELISA) method. The HSV-2 IgM ELISA Kit (Cat. No. H2048M, Calbiotech Inc., USA) and the HSV-2 IgG ELISA Kit (Cat. No. H2047M, Calbiotech Inc., USA) were commercially available kits that were utilized.
Each method fulfilled the manufacturer’s instructions to precisely. The optical density (OD) at the specified wavelength was measured using a calibrated microplate reader, and antibody positivity was determined using the cutoff values specified in the kit instructions.
Statistical Analysis
All data were analyzed using SPSS version 26.0 (IBM Corp., USA). Descriptive statistics (frequency, percentage, mean ± SD) were used to summarize the participants’ demographic and clinical profiles. The prevalence of HSV-2 IgG and IgM antibodies was calculated, and associations between seropositivity and categorical variables were assessed using the chi-square test (χ²). A p-value < 0.05 was considered statistically significant in all analyses.
Results
185 pregnant women between the ages of 22 and 48 were enrolled in the study. Table 1 illustrated that 138 (74.5%) of these were categorized as having a poor socioeconomic position. Furthermore, 81% of participants only had an elementary education or were illiterate. Most were in the first trimester of pregnancy (125 out of 185, or 67%). Consistent with serological testing, 36 participants (19.5%) tested positive for HSV-2 IgM antibodies, revealing current or active infection, while 124 out of 185 participants (67%) tested positive for HSV-2 IgG antibodies, suggesting past or latent infection (Figure 1).
Table 1: Clinical and Demographic Data of Pregnant Women.
| No. of Cases | % | ||
| Age group | 22-28 years | 56 | 30% |
| 29-35 years | 81 | 44% | |
| 36-48 years | 48 | 26% | |
| Life style | Low | 128 | 69% |
| High | 57 | 31% | |
| Education | Illiterate | 102 | 55% |
| Primary | 46 | 25% | |
| University | 37 | 20% | |
| Gestation | 1st trimester | 125 | 67% |
| 2nd trimester | 29 | 16% | |
| 3rd trimester | 31 | 17% | |
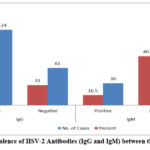 |
Figure 1: Prevalence of HSV-2 Antibodies (IgG and IgM) between the Study Cases. |
Prevalence of HSV-2 -IgG and IgM According to Lifestyle of the Study Cases
Among participants with a high socioeconomic status (n=57), 25 (44%) were positive for HSV-2 IgG, and 10 (17.5%) were positive for HSV-2 IgM. In contrast, among those with low socioeconomic status (n=128), 99 (77%) tested positive for HSV-2 IgG, and 26 (20%) for HSV-2 IgM (Figure 2). The association between HSV-2 IgG seropositivity and low socioeconomic status was statistically significant (p=0.001), whereas HSV-2 IgM seropositivity showed no significant association with lifestyle (p>0.05).
Prevalence of HSV-2-IgG and IgM According to Education of the Study Cases:
Among illiterate participants (n=102), 84 (82%) were positive for HSV-2 IgG, and 14 (14%) for HSV-2 IgM. In those with primary education (n=46), 28 (61%) were positive for HSV-2 IgG, and 13 (28%) for HSV-2 IgM. Among university-educated participants (n=37), 12 (32%) tested positive for HSV-2 IgG, and 9 (24%) for HSV-2 IgM (Figure 2). The association between HSV-2 IgG seropositivity and education level was statistically significant (p=0.001), with higher seroprevalence among illiterate participants. No significant association was observed for HSV-2 IgM seropositivity across education levels (p>0.05).
Prevalence of HSV-2-IgG and IgM According to Gestation Stage of the Study Cases
From the 125 individuals in the first trimester, 15 (12%) and 97 (78%) showed a positive results for HSV-2 IgM and HSV-2 IgG, respectively. In the second trimester (n=29), 10 (34%) were positive for HSV-2 IgG, and 10 (34%) for HSV-2 IgM. In the third trimester (n=31), 17 (55%) displayed a positive for HSV-2 IgG, and 11 (35%) for HSV-2 IgM (Figure 2). Both HSV-2 IgG and IgM seropositivity were noticeably related to the gestational stage (p=0.001), with the highest prevalence noticed in the first trimester. The prevalence of HSV-2-IgG was statistically noteworthy to lifestyle (Table 2), while the prevalence of HSV-2-IgG and IgM was statistically relevant to the gestation stage (Table 2, 3).
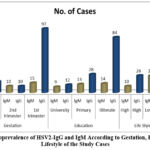 |
Figure 2: Seroprevalence of HSV2-IgG and IgM According to Gestation, Education and Lifestyle of the Study Cases. |
HSV-2 IgG Seropositivity
The relationship between HSV-2 IgG seropositivity and demographic/clinical variables is summarized in Table 2. Significant associations were observed with lifestyle (p=0.001), education level (p=0.001), and gestational stage (p=0.001), indicating that low socioeconomic status, illiteracy, and first-trimester gestation were associated with higher HSV-2 IgG prevalence. No significant association was found with age group (p=0.3).
Table 2: Relation of Positive HSV-2-IgG with Age Group, Lifestyle, Education Level and Gestation in Egyptian Pregnant Women
| Parameter | N (%) | |
| Age group | 22-28 years | 40 (32.5%) |
| 29-35 years | 45 (36%) | |
| 36-48 years | 39 (31.5%) | |
| Total | 124 | |
| Life Style | Low | 99 (80%) |
| High | 25 (20%) | |
| Total | 124 | |
| Education | Illiterate | 84 (67.7%) |
| Primary | 28 (22.6%) | |
| University | 12 (9.7%) | |
| Total | 124 | |
| Gestation | 1st trimester | 97 (78.2%) |
| 2nd trimester | 10 (8.1%) | |
| 3rd trimester | 17 (13.7%) | |
| Total | 124 | |
HSV-2 IgM Seropositivity
The relationship between HSV-2 IgM seropositivity and demographic/clinical variables is presented in Table 3. A significant association was observed with the gestational stage (p=0.001), with a higher prevalence in the second and third trimesters. No significant associations were found with age group, lifestyle, or education level (p>0.05 for all).
Table 3: Relation of Positive HSV-2-IgM with Age Group, Lifestyle, Education Level and Gestation in Egyptian Pregnant Women
| Parameter | N (%) | P-value | |
| Age group | 22-28 years | 11 (30.5%) | >0.05 |
| 29-35 years | 9 (25.1%) | ||
| 36-48 years | 16 (44.4%) | ||
| Total | 36 | ||
| Life Style | Low | 26 (72.2%) | >0.05 |
| High | 10 (27.8%) | ||
| Total | 36 | ||
| Education | Illiterate | 14 (38.9%) | >0.05 |
| Primary | 13 (36.1%) | ||
| University | 9 (25%) | ||
| Total | 36 | ||
| Gestation | 1st trimester | 15 (41.7%) | 0.001 |
| 2nd trimester | 10 (27.8%) | ||
| 3rd trimester | 11 (30.5%) | ||
| Total | 36 | ||
Discussion
Herpes simplex virus type 2 (HSV-2) seroprevalence among pregnant women in Egypt was examined in the current research, which exhibited an overall positivity rate of 19.5% for HSV-2 IgM and 67% for HSV-2 IgG. The results revealed that this community has a noteworthy burden of latent and recent HSV-2 infections. Our results are in line with other reports of HSV-2 IgG seroprevalence rates in pregnant women that ranged from 50% to 80% in different Middle Eastern and African regions.⁸,¹²
A previous study conducted in Sudan reported that HSV-2 IgG prevalence of 65.2%, attributed to socioeconomic disparities and variations in sexual behavior.¹²,¹³ Likewise, research conducted in Egypt stated HSV-2 IgG seropositivity of approximately 70% among women of reproductive age, supporting the notion of widespread endemicity.¹³ The detected IgM seroprevalence of 19.5% in the current study was relatively higher than figures reported in Iran (10.5%) and Turkey (8%), yet lower than in several African countries, where prevalence exceeded 25%.⁶,¹⁴ These interregional differences may be due to variations in diagnostic tools, healthcare accessibility, cultural norms, and awareness of sexually transmitted infections (STIs).
A statistically significant relationship was found between HSV-2 IgG positivity and low socioeconomic status, illiteracy, and first-trimester gestation (p = 0.001 for all). These associations highlight the pivotal role of socioeconomic and educational inequalities in increasing vulnerability to HSV-2 infection. Poorer women often have limited access to antenatal care, restricted health literacy, and fewer opportunities for preventive education, all of which heighten their risk of infection.¹⁵ This aligns with global literature showing that HSV-2 prevalence tends to be higher in populations with constrained economic conditions and limited education.¹⁶,²¹ The finding that 78% of first-trimester women were HSV-2 IgG-positive compared to 34% and 55% in the second and third trimesters respectively suggests increased viral reactivation or recent infection during early pregnancy. This observation supports earlier research indicating that hormonal and immunological changes in the early gestational period may facilitate viral shedding and reactivation.¹⁷ The significant association between HSV-2 IgM seropositivity and gestational stage (p = 0.001), with higher rates during the second (34%) and third trimesters (35%), could indicate increased rates of recent infections or reactivations later in pregnancy. Such reactivations elevate the risk of vertical transmission to the fetus or newborn.¹⁸ In contrast, no significant correlation was found between HSV-2 seropositivity and age group (p = 0.3), suggesting that exposure risk is relatively uniform across reproductive ages. This observation contrasts with reports from other countries where HSV-2 seroprevalence tends to rise with age due to cumulative lifetime exposure.¹⁹,²³
The high HSV-2 IgG prevalence of 67% emphasizes the necessity of routine antenatal screening, especially for high-risk groups such as women of lower socioeconomic background or limited education. Early identification and management of HSV-2 infections are crucial to preventing neonatal herpes a condition that, if untreated, carries a mortality rate of up to 60%.²¹ The elevated IgM seropositivity (19.5%) observed in this study underscores the persistence of active or recent infections among expectant mothers. Furthermore, the notable link between HSV-2 IgG positivity and first-trimester pregnancy suggests a potential association between infection and adverse pregnancy outcomes such as spontaneous abortion or preterm delivery. 22 This finding aligns with earlier evidence implicating HSV-2 infection as a contributing factor to negative obstetric outcomes.23,24 However, establishing a causal relationship requires prospective longitudinal studies and larger sample sizes to differentiate between primary infection, reactivation, and reinfection mechanisms.
Collectively, the findings of the current study position HSV-2 as an under recognized yet important maternal health concern in Egypt. Integrating HSV-2 serological screening into routine antenatal care, alongside targeted health education initiatives that enhance women’s awareness of sexually transmitted infections, may substantially reduce the risk of vertical transmission and associated neonatal complications. Expanding healthcare accessibility, improving women’s health literacy, and addressing socioeconomic inequities should be prioritized as part of a comprehensive national strategy to mitigate the burden of HSV-2 infection among pregnant women.
This study has some limitations. Its cross-sectional design prevents establishing causality between HSV-2 infection and pregnancy outcomes. The sample size and single-hospital setting may also limit the generalizability of results, and the use of ELISA alone without molecular confirmation could not distinguish between primary and recurrent infections.
Future research should include larger, multicenter longitudinal studies using molecular diagnostic tools to better define infection dynamics and clarify causal relationships. Broader assessments of behavioral and socioeconomic factors are also recommended to support targeted prevention and antenatal screening strategies.
Conclusion
This study revealed a high seroprevalence of HSV-2 infection among pregnant Egyptian women, with 67% testing positive for HSV-2 IgG antibodies and 19.5% for HSV-2 IgM. The highest infection rates were noticed between women in their first trimester, those of low socioeconomic status, and those with limited educational attainment. Our findings highlight the substantial public health burden of HSV-2 in this population and emphasize the urgent need for targeted screening and preventive strategies, particularly for high-risk groups. Routine serological testing, mainly for IgM antibodies, may aid in early detection and timely intervention to prevent adverse pregnancy outcomes and neonatal herpes. Public health efforts should prioritize improving women’s health literacy, expanding access to healthcare services, and addressing socioeconomic disparities to reduce HSV-2 transmission and its associated complications during pregnancy.
Acknowledgment
The authors express sincere gratitude to Jouf University, Saudi Arabia, for its support and encouragement throughout this work and to the managers of the hospitals reported in this study for their permission to collect blood samples.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
The study was approved by the Review Board of Shebeen El-Kom Educational Hospital with an approval date of March 24, 2023.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study. Participation was entirely voluntary, and all participants were informed about the objectives, procedures, and potential implications of the research prior to their inclusion.
Clinical Trial Registration
This work does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Author Contributions
- Mohamed Nabil Mohamed: Conceptualization, Writing – original draft.
- Mohamed Eid Anwar: Investigation.
- Mohamed Nabil Mohamed & Eman Fawzy El Azab.: Writing – Review, Editing& supervision
References
- Naffeti B. S, Ayoub H. H, Abu-Raddad L. J. Quantifying population-level sexual risk behavior through HSV-2 transmission dynamics in the United States, 1950–2020. Rep., 2025; 15: 34521.
CrossRef - Patton M, Bernstein K, Liu G. Seroprevalence of herpes simplex virus types 1 and 2 among pregnant women and sexually active, nonpregnant women in the United States. Clin Infect Dis., 2018;67(10):1535-1542. PMID: 29668856.
CrossRef - McQuillan G, Kruszon-Moran D, Flagg E, Paulose-Ram R. Prevalence of herpes simplex virus type 1 and type 2 in persons aged 14–49: United States, 2015–2016. NCHS Data Brief., 2018;(304):1-8. PMID: 29442994.
- Hammad WAB, Konje JC. Herpes simplex virus infection in pregnancy: An update. Eur J Obstet Gynecol Reprod Biol., 2021;259:38-45. PMID: 33581405.
CrossRef - Sari K, Gianopulos K, Nicola A. Glycoprotein C of herpes simplex virus 1 shields glycoprotein B from antibody neutralization. J Virol., 2020; 94(5):e01703-19. PMID: 31826995.
CrossRef - Subramaniam A, Britt WJ. Herpesviridae infection: Prevention, screening, and management. Clin Obstet Gynecol., 2018; 61(1):157-176. PMID: 29283902.
CrossRef - Alareeki A, Osman A. M. M, Khandakji M. N, Looker K. J, Harfouche M, Abu-Raddad L. J. Epidemiology of herpes simplex virus type 2 in Europe: systematic review, meta-analyses, and meta-regressions. Lancet Infect. Dis., 2023; 25: 100558.
CrossRef - De Rose DU, Auriti C, Purcaro V, et al. Neonatal herpes simplex virus infection: From the maternal–fetal interface to the infant. J Med Virol. 2023;95(10):e29024.
CrossRef - AlMukdad S, Farooqui U. S, Harfouche M, et al. Epidemiology of herpes simplex virus type 2 in Canada, Australia, and New Zealand: systematic review, meta-analyses, and meta-regressions. Sex. Transm. Dis., 2022; 49: 403–413.
CrossRef - American College of Obstetricians and Gynecologists. Management of genital herpes in pregnancy: ACOG practice bulletin, number 220. Obstet Gynecol., 2020;135(5):e193-e202. PMID: 32332414.
CrossRef - Looker KJ, Johnston C, Welton NJ, et al. The global and regional burden of genital ulcer disease due to herpes simplex virus: A natural history modelling study. BMJ Glob Health. 2020;5(3):e001875. PMID: 32201620.
CrossRef - Centers for Disease Control and Prevention. Genital herpes – CDC fact sheet (detailed). 2021. Accessed April 29, 2025. https://www.cdc.gov/std/herpes/stdfact-herpes-detailed.htm
- El-Ansary MR. Seroprevalence of herpes simplex virus type 1 and type 2 among Egyptian children aged from one to 15 years old: A comparative study. Afro-Egypt J Infect Endem Dis., 2022;12(4):383-389. https://aeji.journals.ekb.eg/article_270734.html
CrossRef - Rodriguez H. Can herpes affect your fertility? Natural Fertility Info. 2021. Accessed April 29, 2025. https://natural-fertility-info.com/herpes-fertility.html
- Rodriguez H. Can herpes affect your fertility? Natural Fertility Info. 2021 [cited 2025 Apr 29]. Available from: https://natural-fertility-info.com/herpes-fertility.html
- World Health Organization. Herpes simplex virus. 2022. Accessed April 29, 2025. https://www.who.int/news-room/fact-sheets/detail/herpes-simplex-virus
- Felker AM, Nguyen P, Kaushic C. Primary HSV-2 infection in early pregnancy results in transplacental viral transmission and dose-dependent adverse pregnancy outcomes in a novel mouse model. Viruses, 2021;13(10):1929. PMID: 34696359.
CrossRef - Silhol R, Coupland H, Baggaley R. F, et al. What is the burden of heterosexually acquired HIV due to HSV-2? Global and regional model-based estimates of the proportion and number of HIV infections attributable to HSV-2 infection. Acquir. Immune Defic. Syndr., 2021; 88: 19–30.
CrossRef - Huai P, Li F, Li Z, et al. Seroprevalence and associated factors of HSV-2 infection among general population in Shandong Province, China. BMC Infect Dis., 2019; 19:382. https://doi.org/10.1186/s12879-019-3995-2
CrossRef - Pavia G, Licata F, Marascio N, et al. Seroprevalence and age-related susceptibility of TORCH infections in childbearing age women: A 5-year cross-sectional retrospective study and a literature review. Infect. Public Health, 2024; 17(10): 102537.
CrossRef - Kilbas EPK, Ciftci IH, Kilbas I, Toptan H. Seroprevalence of TORCH viral agents in pregnant women in Turkey: Systematic review and meta-analysis. Pathogens, 2025; 14(1):37. PMID: 39860998.
CrossRef - Khameneh Z, Sepehrvand N, Mohammadian M. Herpes simplex virus type 2 seroprevalence in pregnant women in Urmia, Northwest of Iran, during 2014–2015. Iran Biomed J., 2020;24(2):136-139. PMID: 31677608.
CrossRef - Patel R, Moran B, Clarke E, Geretti A. M, Lautenschlager S, Green J, Donders G, Gomberg M, Samraj S, Tiplica G. S, Foley E. 2024 European guidelines for the management of genital herpes. J. Eur. Acad. Dermatol. Venereol., 2024; 39(4): 742–758. doi:10.1111/jdv.20450.
CrossRef - Muñiz-Salgado J. C, Juárez-De la Cruz G, Vergara-Ortega D. N, et al. Seroprevalence and vaginal shedding of herpes simplex virus type 2 in pregnant adolescents and young women from Morelos, Mexico. Viruses, 2023; 15(5): 1122. doi:1
CrossRef







