Manuscript accepted on :13-11-2025
Published online on: 10-12-2025
Plagiarism Check: Yes
Reviewed by: Dr. Sharath BS
Second Review by: Dr. Subhasis Chakraborty
Final Approval by: Dr. Mariia Shanaida
I Made Sukadana1* , Sri Rahayu Santi1
, Sri Rahayu Santi1 , Ni Nyoman Rupiasih2
, Ni Nyoman Rupiasih2 , Luh Gede Sri Surya Heryani3
, Luh Gede Sri Surya Heryani3 , Lilik Parwati1and Nadya Pamesthi Annisa Ramadhani1
, Lilik Parwati1and Nadya Pamesthi Annisa Ramadhani1
1Chemistry Department, Faculty Mathematics and Natural Sciences Udayana University, Badung, Indonesia.
2Physic Department, Faculty Mathematics and Natural Sciences Udayana University, Badung, Indonesia
3Faculty of Veterinary Medicine Udayana University, Badung, Indonesia.
Corresponding Author E-mail: sr_santi@unud.ac.id
DOI : https://dx.doi.org/10.13005/bpj/3318
Abstract
Preclinical studies indicate that gayam seeds (Inocarpus fagiferus Fosb) at a dose of 50 mg/kg body weight exhibit anti- atherosclerotic effects, as evidenced by changes in biomarkers including SOD activity, lipid profile, blood MDA, SOD-3, SOD-2, ICAM-1, IL-6, and TNF-α in aortic endothelial cells. While optimized solid and granule formulations have been developed, the formation of nanoparticles (1–100 nanometers in size) may further enhance the bioactivity of the extract. Nanoparticles provide improved solubility, bioavailability, stability, targeted delivery, increased extract efficacy, reduced dosages, and minimized side effects. This study aimed to prepare and characterize ethanol extract nanoparticles of gayam seeds for anti-atherosclerotic drug development. Nanoparticles were synthesized using the quality by design approach (Fractional Factorial Design, FFD), with the optimal formulation comprising 1.5 mL of 0.1% extract, 10 mL of 1.754% chitosan, and 5 mL of 0.1% NaTPP, stirred at 500 rpm for 15 minutes. The resulting nanoparticles demonstrated 100% encapsulation efficiency, 44.89% transmittance, a polydispersity index (PDI) of 0.3489, a zeta potential of 34.07 mV, and a particle size of 250.63 nm, meeting established nanoparticle criteria.
Keywords
Antioxidant; Atherosclerosis; Drug formulation; Inocarpus fagiferus Fosb; Nanoparticles
Download this article as:| Copy the following to cite this article: Sukadana I. M, Santi S. R, Rupiasih N. N, Heryani L. G. S. S, Parwati L, Ramadhani N. P. A. Formulation and Characterization of Ethanol Extract Nanoparticles of Gayam Inocarpus fagifer (Parkinson) Fosberg Seeds for Anti-Atherosclerotic Applications. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Sukadana I. M, Santi S. R, Rupiasih N. N, Heryani L. G. S. S, Parwati L, Ramadhani N. P. A. Formulation and Characterization of Ethanol Extract Nanoparticles of Gayam Inocarpus fagifer (Parkinson) Fosberg Seeds for Anti-Atherosclerotic Applications. Biomed Pharmacol J 2025;18(4). Available from: https://bit.ly/48tMUEL |
Introduction
Inocarpus fagifer (Parkinson) Fosberg seeds, also known as gayam or gatep in Bali,1,2 demonstrate potential for atherosclerosis prevention, as supported by preclinical studies. The ethanol extract exhibits DPPH radical inhibition with an IC50 of 280 ppm, enhances SOD activity in Wistar rat plasma, and improves lipid profiles. These antioxidant effects are attributed to the presence of linoleic acid, ethyl linoleate, ethyl oleate, and homopterocarpine.3 The extract upregulates SOD-3 expression in aortic endothelial cells and reduces MDA levels in rat plasma,4 supporting its role in atherosclerosis prevention. It may also stimulate endogenous antioxidants, particularly SOD-2, which serves as a biomarker for anti-atherosclerotic activity. 5,6,7,8,9,10 SOD-2 expression influences ICAM-1 levels, positioning ICAM-1 as an early biomarker for atherosclerosis. Reduced plasma MDA is associated with decreased inflammation, as indicated by lower TNF-α and IL-6 expression in aortic endothelial cells.11 Preclinical biomarker and toxicity assessments have identified 50 mg/kg body weight as an effective anti-atherosclerotic dose. Standardizing this dose in nanoparticle form may further enhance its therapeutic efficacy.12
Nanoparticles are 1-100 nanometers in size (one nanometer is one-billionth of a meter) and offer advantages such as improving dissolution of difficult substances, enhancing bioavailability,13,14 modifying drug delivery, increasing stability, and boosting absorption.15,16 These features make nanoparticle-based extracts more efficient for developing anti-atherosclerosis drugs.17 Nanoparticles also penetrate spaces between cells that can be accessed by colloidal particles. The formation of nanoparticles is influenced by the characteristics of both the polymer and the drug. The aim of this research is to formulate and determine the characteristics of nanoparticles including organoleptic properties, particle size and distribution, morphology, zeta potential, and percent transmittance.
Material and Methods
Material
Inocarpus fagifer (Parkinson) Fosberg seeds sourced from Tabanan, Bali, served as the plant material. The chemicals utilized included 96% ethanol (EMSURE), sodium carboxymethylcellulose, chitosan, 1% glacial acetic acid, sodium tripolyphosphate (NaTPP), and demineralized water.
The equipment comprised standard laboratory glassware, a blender, sieve, extractor, rotary vacuum evaporator, centrifuge, analytical balance, pipettes, flasks, freezer, air cooler, oven, mortar, particle size analyzer (PSA), transmission electron microscope (TEM), water bath, plethysmometer, timer, filter paper, aluminum foil, and gloves.
Methods
Preparation of ethanol extract of gayam (Inocarpus fagifer (Parkinson) Fosberg ) seeds and preparation of nanoparticles
Six kilograms of dried gayam (Inocarpus fagifer (Parkinson) Fosberg ) seed powder were macerated with ethanol in a dark extractor at room temperature for 24 hours. The extract was filtered, and the remaining solids were re-macerated with ethanol until complete extraction was achieved. The combined filtrates were concentrated using a rotary vacuum evaporator and subsequently weighed. Nanoparticle optimization was conducted using the quality by design approach (Fractional Factorial Design, FFD), as outlined in Table 1.
Table 1: Fractional Factorial Design method
|
Level |
Factors |
||||
|
Numerical factors |
|||||
|
1 |
2 |
3 |
4 |
5 |
|
|
Extract Concentration 1.5 mL (%) |
Chitosan Concentration 10 mL (%) |
Concentration of Na TPP 5 mL (%) |
Mixing Speed (RPM) |
Mixing Time (Minutes) |
|
|
Lower level (-1) |
0.1 |
1.75 |
0.1 |
500 |
15 |
|
Upper level (+1) |
0.2 |
2 |
0.2 |
1000 |
60 |
According to Table 1, the extract was dissolved in 96% ethanol, chitosan in 1% acetic acid, and NaTPP in water. The extract and chitosan solutions were combined in a beaker and stirred at 500 rpm for 5 minutes. NaTPP was then added while maintaining the specified stirring speed and duration until the mixture became cloudy. The percentage of entrapment encapsulation (EE) and transmittance for each formulation were measured. The resulting nanoparticle precipitate was stored in a freezer at 3°C for two days, dried using air cooling, and ground with a mortar prior to characterization.
Characterization of nanoparticles
Following identification of the optimal formula using FFD, formulations F1, F2, and F3 were prepared in triplicate to assess their characteristics. Nanoparticles were evaluated for particle size and distribution using a particle size analyzer (PSA), morphology via transmission electron microscopy (TEM), as well as zeta potential and percent transmittance.
Results
All data presented in the Results section were generated from research conducted in the Natural Product Chemistry Laboratory, Chemistry and Physical Sciences Departments, Udayana University.
Preparation of ethanol extract of gayam (Inocarpus fagifer (Parkinson) Fosberg ) seeds and preparation optimum of nanoparticles
Maceration of 6 kg of dry Inocarpus fagifer (Parkinson) Fosberg seed powder produced approximately 30 g of concentrated extract. The total flavonoid content of the extract is presented in Table 2.
Table 2: Results of total flavonoid content test of gayam extract
|
Sample |
Replication |
Flavonoid content (quercetin equivalent) in 1 g extract (ug) |
Average (ug) |
Flavonoid Content in Gayam Seeds (%) |
|
extract test solution |
1 |
307.778 |
307.778 |
0.03 % |
|
2 |
309.259 |
|||
|
3 |
306.296 |
Following determination of flavonoid content, optimization of gayam (Inocarpus fagifer (Parkinson) Fosberg) seed extract nanoparticles was performed using the quality by design approach (Fractional Factorial Design, FFD), with results summarized in Table 3.
Table 3: The results of the optimization of nanoparticle production with the response of % Entrapment Encapsulation (EE) and % Transmittance
|
|
|
Factor 1 |
Factor 2 |
Factor 3 |
Factor 4 |
Factor 5 |
Response 1 |
Response 2 |
|
Std |
Run |
A: Extract Concentration 1.5 mL |
B: Chitosan Concentration 10 mL |
C: Concentration of Na TPP 5 mL |
D: Stirring speed |
E: Stirring Time |
EE |
Transmitance |
|
|
|
% |
% |
% |
RPM |
Menit |
% |
% |
|
6 |
1 |
0.1 |
1.75 |
0.2 |
1000 |
60 |
100 |
32.43 |
|
15 |
2 |
0.2 |
1.75 |
0.1 |
1000 |
60 |
100 |
12.06 |
|
1 |
3 |
0.1 |
1.75 |
0.1 |
500 |
60 |
100 |
41.31 |
|
11 |
4 |
0.2 |
2 |
0.1 |
500 |
60 |
100 |
18.08 |
|
7 |
5 |
0.2 |
2 |
0.2 |
500 |
15 |
100 |
22.01 |
|
14 |
6 |
0.2 |
1.75 |
0.1 |
500 |
15 |
100 |
22.73 |
|
16 |
7 |
0.2 |
1.75 |
0.2 |
1000 |
15 |
100 |
14.81 |
|
5 |
8 |
0.1 |
1.75 |
0.1 |
500 |
15 |
100 |
44.81 |
|
8 |
9 |
0.2 |
1.75 |
0.2 |
500 |
60 |
100 |
19.21 |
|
12 |
10 |
0.1 |
1.75 |
0.2 |
500 |
15 |
100 |
36.42 |
|
4 |
11 |
0.2 |
2 |
0.2 |
1000 |
60 |
100 |
11.1 |
|
13 |
12 |
0.2 |
2 |
0.1 |
1000 |
15 |
100 |
16.26 |
|
3 |
13 |
0.1 |
2 |
0.2 |
1000 |
15 |
100 |
32.25 |
|
10 |
14 |
0.1 |
2 |
0.2 |
500 |
60 |
100 |
36.45 |
|
9 |
15 |
0.1 |
2 |
0.1 |
1000 |
60 |
100 |
26.12 |
|
2 |
16 |
0.1 |
2 |
0.1 |
500 |
15 |
100 |
42.98 |
Characterization of Optimal Formula Nanoparticles
The characterization results for the optimal nanoparticle formulations, specifically F1, F2, and F3, are presented in Table 4.
Table 4: Results of optimal formula verification for F1, F2 and F3
|
Formula |
Test Type |
Target |
Formula Repetition |
Observation results |
Average observation results |
Deviation (%) |
Condition |
Results |
|
1 |
% Entrapment Encapsulation |
100 |
F1 |
100 |
100 |
0 |
Maximize |
Qualify |
|
F2 |
100 |
|||||||
|
F3 |
100 |
|||||||
|
2 |
% Transmitance |
44.579 |
F1 |
45.35 |
44.89 |
0.698 % |
Maximize |
Qualify |
|
F2 |
44.98 |
|||||||
|
F3 |
44.35 |
|||||||
|
3 |
PDI |
– |
F1 |
0.3450 |
0.3489 |
– |
< 0.500 |
Qualify |
|
F2 |
0.3511 |
|||||||
|
F3 |
0.3506 |
|||||||
|
4 |
Zeta Potential (mV) |
– |
F1 |
34.29 |
34.07 |
– |
> ± 30 mV |
Qualify |
|
F2 |
33.84 |
|||||||
|
F3 |
34.09 |
|||||||
|
5 |
Particle size (nm) |
– |
F1 |
296.9 |
250.63 |
– |
<1000 nm |
Qualify |
|
F2 |
248.4 |
|||||||
|
F3 |
206.6 |
TEM analysis revealed that the synthesized nanoparticles exhibited semi-spherical or oval morphologies, as illustrated in Figure 1.
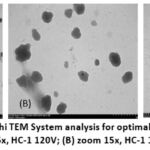 |
Figure 1: The results of Hitachi TEM System analysis for optimal formula nanoparticles (one of the formulas is F1): (A) zoom 5x, HC-1 120V; (B) zoom 15x, HC-1 120V; (C) zoom 40x, HC-1 120V
|
Discussion
Table 2 indicates that gayam (Inocarpus fagifer (Parkinson) Fosberg) seed extract contains 0.03% flavonoids (quercetin equivalent), confirming the presence of bioactive flavonoid compounds in relatively low concentrations. Despite this, these flavonoids contribute to the pharmacological potential of gayam seeds, including antioxidant, anti-inflammatory, and other biological activities characteristic of flavonoids.
Given the presence of flavonoids (0.03%) in gayam seeds, nanoparticle development was pursued to enhance the stability and bioavailability of these active compounds. The Fractional Factorial Design (FFD) within the Quality by Design framework was employed to efficiently evaluate and optimize key factors affecting nanoparticle characteristics: extract concentration, chitosan concentration, NaTPP (crosslinker) concentration, stirring speed, and stirring time. The primary responses measured were entrapment efficiency (EE), representing the percentage of flavonoids encapsulated within the nanoparticle matrix, and transmittance, reflecting the clarity and homogeneity of the nanoparticle solution. Higher transmittance values indicate well- formed, uniformly dispersed nanoparticles. Entrapment efficiency was most strongly influenced by chitosan and NaTPP concentrations via the ionotropic gelation mechanism. Excessive extract concentration reduced encapsulation efficiency, while appropriate stirring speed and duration were critical for achieving homogeneous particles with high transmittance. The optimal combination of these parameters produced stable, clear, and effective nanoparticles.
Preparation of gayam extract nanoparticles according to the FFD design (Table 3) resulted in 100% entrapment efficiency across all formulations, with transmittance values ranging from 11.1% to 44.81%. The optimal formula in Table 3 is highlighted prior to conducting analysis of variance (ANOVA) and establishing the criteria for independent and dependent variables. The optimal formula, as determined by ANOVA and subsequent software analysis, was finalized based on the criteria presented in Tables 5 and 6.
Table 5: The results of the analysis of variance (ANOVA)
|
Respon |
F value |
P value |
R2 |
|
% Entrapment Encapsulation (EE) |
0 |
0 |
0 |
|
% Transmitance |
28.44 |
< 0.0001 (significant) |
0.9614 |
Table 6: Criteria of Independent and Dependent Variables
|
Independent and Dependent Variables |
Criteria |
|
Extract Concentration 1.5 mL (A) |
In range |
|
Chitosan Concentration 10 mL (B) |
In range |
|
Concentration of Na TPP 5 mL (C) |
In range |
|
Stirring speed (D) |
In range |
|
Stirring Time (E) |
In range |
|
% EE (Y1) |
Maximize |
|
% Transmitance (Y2) |
Maximize |
The results for the optimal formula, determined through the FFD design (Table 3), ANOVA analysis (Table 5), and established criteria (Table 6), are presented in Table 7.
Table 7: Optimal formula
|
Factor 1 |
Factor 2 |
Factor 3 |
Factor 4 |
Factor 5 |
Response 1 |
Response 2 |
Desirability |
|
A: Extract Concentration 1.5 mL |
B: Chitosan Concentration 10 mL |
C: Concentration of Na TPP 5 mL |
D: Stirring speed |
E: Stirring Time |
EE |
Transmitance |
|
|
% |
% |
% |
RPM |
Minute |
% |
% |
|
|
0.100 |
1.754 |
0.100 |
500 |
15 |
100 |
44.579 |
0.993 |
Three formulations (F1, F2, and F3) were prepared in replicate, as determined by the optimal formula in Table 7, to characterize the nanoparticle formulation. The results are provided in Table 4. Gayam seed extract nanoparticles were synthesized using the ionic gelation method, which involves the interaction of positively charged chitosan with negatively charged sodium tripolyphosphate (NaTPP). This approach is straightforward, avoids the use of high temperatures, and allows for precise control.18,19 Chitosan, a non-toxic cationic polymer, serves as an effective drug carrier due to its high biodegradability and biocompatibility.20,21 Furthermore, chitosan can enhance drug efficacy without inducing side effects.22 During ionic gelation, chitosan dissolves in dilute acid, generating cationic amine groups that cross-link with NaTPP anions to form stable chitosan nanoparticles.23,24 NaTPP dissociates in water into sodium and tripolyphosphate ions, which interact with chitosan NH⁺ groups and function as a non-toxic crosslinker.25,26
The preparation of nanochitosan-tripolyphosphate-gayam seed extract involves the interaction between the positively charged amine groups in chitosan and the negatively charged tripolyphosphate. The resulting bond facilitates the absorption of the extract.27 Bioactive components from gayam seed extract can associate with the surface of nanochitosan-tripolyphosphate.28 This interaction enables the storage of bioactive substances within the nanoparticle system. The polymer and matrix system encapsulates these substances in the core, embedding the molecules within the polymer matrix.29 Nanoparticles are fine solids with nanometer-scale dimensions formed through these interactions.30 The dropwise addition of NaTPP under constant magnetic stirring prevents solidification, reduces aggregation, and stabilizes nanoparticle formation.31,32 Stirring speed influences both the stability of the emulsion and the size of the nanoparticles. Higher stirring speeds decrease interfacial tension and enhance emulsion stability. The formation scheme for nanochitosan-tripolyphosphate-gayam seed extract, which encapsulates active substances from gayam seed extract via the ionic gelation method,33 is illustrated in Figure 2.
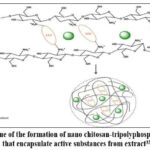 |
Figure 2. Scheme of the formation of nano chitosan-tripolyphosphate cross-links that encapsulate active substances from extract33
|
(Adapted with permission from Detsi, A., Kavetsou, E., Kostopoulou, I., Pitterou, I., Pontillo, A. R. N., Tzani, A., and Oumpoulakis, P. Nanosystems for the encapsulation of natural products: The case of chitosan biopolymer as a matrix. Pharmaceutics, 2020; 12(7): 669.DOI: Not applicable.)
Particle size measurements for 1.754% chitosan combined with 0.1% extract yielded an average size of 250.63 nm. The mean polydispersity index (PI) was 0.3489, and the zeta potential (ZP) measured 34.07 mV, as detailed in Table 3. Figure 3 and Figure 4 present the particle size and zeta potential data for the optimal nanoparticle formulation.
 |
Figure 3: The results of particle size measurements for optimal formula nanoparticles (F1, F2, and F3).
|
 |
Figure 4: The results of zeta potential measurements for optimal formula nanoparticles (F1, F2, and F3).
|
According to the data in Table 4, nanoparticle formulations are considered stable when the zeta potential exceeds ±30 mV.34 Nanoparticles are defined as materials with sizes ranging from 1 to 1000 nm, and the results confirm that the particles produced fall within this range35. The polydispersity index (PI) indicates the distribution width of particle sizes. A low PI value (less than 0.5) corresponds to a narrow size distribution and stable suspension, whereas a high PI value (greater than 0.7) indicates a broad distribution, increased non-uniformity, faster agglomeration, and reduced stability. High non-uniformity accelerates flocculation and coalescence, while uniform particle size supports suspension stability by minimizing size deviation.36,37 Therefore, a low PI value demonstrates the effectiveness of the stabilizer in preventing particle agglomeration,38,39 while a high PI value reflects a broad size distribution. In this study, the polydispersity index was 0.3489 and the particle size was 250.63 nm, indicating that the nanoparticles are monodisperse and meet the criteria for nanoparticle classification.
Zeta potential reflects the surface charge of particles and determines their tendency to aggregate or remain dispersed.40 A zeta potential above +30 mV or below -30 mV indicates a stable suspension, as high values promote electrostatic repulsion and prevent particle aggregation.41,42 The ethanol extract nanoparticles of gayam seeds exhibited an average zeta potential of +34.07 mV, suggesting good stability, although some aggregation may still occur over time. Zeta potential also represents the overall charge in the suspension, which is a key predictor of nanoparticle stability.43 The observed value of +34.07 mV indicates favorable storage stability. Further enhancement of stability can be achieved by incorporating surfactants, which create a protective barrier around the particles and inhibit aggregation.44,45
Transmission electron microscopy (TEM) analysis (Figure 1) revealed that the ethanol extract nanoparticles of gayam seeds are predominantly spherical with smooth surfaces. The observed particle size ranged from 100 to 200 nm, consistent with the average size of 206 nm measured by dynamic light scattering (DLS), which accounts for the hydration layer. The moderate variation in size distribution aligns with the polydispersity index (PDI) value of 0.3489. Although some aggregation was observed, the majority of nanoparticles remained discrete and uniform in shape, indicating a stable system as further supported by the zeta potential of +34.07 mV.
Conclusion
The optimal nanoparticles of anti-atherosclerosis gayam seed ethanol extract, prepared by the Fractional Factorial Design method (0.1% extract, 1.754% chitosan, 0.1% NaTPP, 500 rpm, 15 min), demonstrated characteristics as follows 100% encapsulation efficiency, 44.89% transmittance, a PDI of 0.3489, zeta potential of +34.07 mV, and particle size of 250.63 nm. TEM analysis confirmed the spherical morphology and uniform distribution, indicating their suitability as a stable nanoparticle formulation for potential anti-atherosclerosis therapy.
Acknowledgement
On this occasion, the author would like to thank Dr. Ni Ketut Widnyani from the Pharmacy Department, Faculty of Mathematics and Natural Sciences, Udayana University, who helped formulate the nanoparticles and several friends at the National Research and Innovation Agency (BRIN) for characterizing the nanoparticles made.
Funding Sources
The author would like to express his gratitude to Udayana University for providing PNBP funding through the PUU (Penelitian Unggulan Udayana or Udayana Superior Research) research grant No: B/255.268/UN14.4.A/PT.01.03/2024. This research funding has helped the author complete his student’s thesis.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement-
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Authors contributions
- I Made Sukadana: conceived the research idea and grant winner
- Sri Rahayu Santi: wrote the draft research methodology and is the corresponding author.
- Ni Nyoman Rupiasih: formulated the nanoparticles.
- Luh Gede Sri Surya Heryani: prepared the materials for characterization at BRIN.
- Lilik Parwati: assisted with the research in the laboratory.
- Nadya Pamesthi Annisa Ramadhani: assisted in editing the manuscript.
References
- Segatri, P. Gatep. In: Segatri, P. Taru Premana: The Efficacy of Plants for Traditional Medicine. Denpasar: Upada Sastra; 1995: 9.
- Heyne, K. Gayam (Inocarpus fagifer). In: Heyne, K. Useful Plants of Indonesia. Vol. II. 3rd ed. Jakarta: Forestry Research and Development Agency, Sarana Wana Jaya Foundation; 1987:1188–1189.
- Sukadana,M., Putra Manuaba, I.B., Wita, I.W., Sutirta Yasa, I.W.P., and Santi, S.R. Antioxidant Compounds of Gayam Seed (Inocarpus fagiferus Fosb) to Prevent Atherosclerosis through Increases of SOD Activity and Improvement of Lipid Profile on Wistar Rat. J. Biol. Chem. Research. 2015; 32 (1): 28-35.
- Sukadana, I.M. and Santi, S.R. Antioxidant Compounds of Gayam Seed (Inocarpus fagiferus Fosb) to Prevent Atherosclerosis in Wistar Rat With High Cholesterol Diet, Biomedical and Pharmacology Journal, 2015; 8(2):547-553. DOI: 13005/bpj/797
CrossRef - Fukai, T., Siegfried, M.R., Ushio-Fukai, M., Griendling, K.K., Harrison, G. Modulation of Extracellular Superoxide Dismutase Expression by Angiotensin II and Hypertension. Circ Res, 1999; 85: 23–8. DOI: 10.1161/01.res.85.1.23
CrossRef - Landmesser, U., Merten, R., Speikermann, S., Buttner, K., Drexler, H., Hornig, B. Vascular Extracellular Superoxide Dismutase Activity in Patients with Coronary Artery Disease. Relation to Endothelium-dependent Vasodilation. Circulation, 2000; 101: 2264 –70. DOI: 10.1161/01.cir.101.19.2264
CrossRef - Chan, P.H. Reactive Oxygen Radicals in Signaling and Damage in the Ischemic Brain. J Cerebral Blood Flow Metabol. 2001; 21: 2-14. DOI: 10.1097/00004647-200101000-00002
CrossRef - Stralin, P., Marklund, S.L. Vasoactive Factors and Growth Factors Alter Vascular Smooth Muscle Cell EC-SOD Expression. Am J Physiol, 2001; 281: 111621-9. DOI: 10.1152/ajpheart.2001.281.4.H1621
CrossRef - Fukai, T., Folz, R.J., Landmesser, U., Harrison, D.G. Extracellular Superoxide Dismutase and Cardiovascular Disease. Cardiovasc Res, 2002; 55: 239 –49. DOI: 10.1016/S0008-6363(02)00328-0
CrossRef - Zelko, I.N., Mariani, T.J., Folz, R.J. Superoxide Dismutase Multigene Family: A Comparison of the CnZn-SOD (SOD1), Mn-SOD (SOD2), and EC-SOD (SOD3) Gene Structures, Evolution, and Expression. Free Radical Biol Med, 2002; 33: 337-49. DOI: 10.1016/S0891-5849(02)00905-X
CrossRef - Sukadana , I.M. and Santi, S.R. The Effect of Antioxidant Compounds in Ethanol Extract Inocarpus Fagiferus Fosb Seed to Expression TNF-a And IL-6 Liver Cells In Hypercholesterolemia Wistar Rat. Journal of Pharmaceutical Sciences and Research. 2017; 9(5): 690-694. DOI: Not applicable
- Sukadana, M., Santi, S.R., and I Made Jawi. Expression of ICAM-1, SOD-2, TNF-α, AND IL-6 AORTA Endhotel Cells Hypercholesterolemia Wistar Rats when provided ethanol extracts of Inocarpus fagiferus Fosb seeds. Journal of Pharmaceutical Sciences and Research . 2020; 12(7): 858-863. DOI: Not applicable
- Müller, R. H., & Keck, C. M. Nanosuspensions as Advanced Colloidal Drug Delivery Systems to Improve Solubility of Poorly Soluble Drugs. European Journal of Pharmaceutics and Biopharmaceutics, 2023; 183: 123–135. https://doi.org/…
- Agarwal, G. PLGA‑PVA Nanoparticle Encapsulation Enhances Bioavailability of Curcumin. International Journal of Nanomedicine, 2021; 16: 4501–4512. https://doi.org/…
- Caparros‑Gonzalez, R. A. Nanostructured Lipid Carriers and Polymeric Nanoparticles: Enhancing Delivery of Phytocompounds. Frontiers in Chemistry, 2022; 10: https://doi.org/10.3389/fchem.2022.850757
CrossRef - Gigliobianco, M. R., Casadidio, C., Censi, R., and Di Martino, P. Nanocrystals of Poorly Soluble Drugs: Drug Bioavailability and Physicochemical Stability. Pharmaceutics, 2020; 12(3): 288. https://doi.org/10.3390/pharmaceutics12030234
CrossRef - Zhuo, Y., Zhao, Y.-G., and Zhang, Y. Enhancing Drug Solubility, Bioavailability, and Targeted Therapeutic Applications through Magnetic Nanoparticles. Molecules, 2024; 29(20): 4854. https://doi.org/10.3390/molecules29204854
CrossRef - Algharib, S. A., Dawood, A., Zhou, K., Chen, D., Li, C., Meng, K. Preparation of Chitosan Nanoparticles by Ionotropic Gelation Technique: Effects of Formulation Parameters and in Vitro Characterization. Journal of Molecular Structure, 2022; 1252: 132129. https://doi.org/10.1016/j.molstruc.2022.132129
CrossRef - Van Bavel, N., Issler, T., Pang, L., Anikovskiy, M., and Prenner, E. J. A Simple Method for Synthesis of Chitosan Nanoparticles with Ionic Gelation and Homogenization. Molecules, 2023; 28(11): 4328. https://doi.org/10.3390/molecules28114328
CrossRef - Desai, N., Rana, D., Salave, S., Gupta, R., Patel, P., Karunakaran, B., Sharma, A., Giri, J., Benival, D., and Kommineni, N. Chitosan: A Potential Biopolymer in Drug Delivery and Biomedical Applications. Pharmaceutics, 2023; 15(4): 1313. https://doi.org/10.3390/pharmaceutics15041313
CrossRef - Li, X., Zhang, Y., Liu, R., and Wang, L. Recent Advances of Chitosan-based Nanoparticles for Biomedical and Biotechnological Applications. International Journal of Biological Macromolecules, 2023; 203:, 379–388. https://doi.org/10.1016/j.ijbiomac.2022.01.162
CrossRef - Sangnim, T., Dheer, D., Jangra, N., Huanbutta, K., Puri, V., and Sharma, A. Chitosan in Oral Drug Delivery Formulations: A Review. Pharmaceutics, 2023; 15(9): https://doi.org/10.3390/pharmaceutics15092361
CrossRef - Amri, Y., Fajri, R., Fitriani, P., Novitasari, P., and Zulfajri, M. The Effect of Tripolyphosphate (TPP) Volumes on the Synthesis of Chitosan Nanoparticles Using Ionic Gelation Method. Advances in Social Science, Education and Humanities Research, 2021; 11: 213–216. https://doi.org/10.2991/assehr.k.210909.048
CrossRef - Pan, C., Qian, J., Zhao, C., Yang, H., Zhao, X., and Guo, H. Study on the Relationship Between Crosslinking Degree and Properties of TPP Crosslinked Chitosan N Carbohydrate Polymers, 2020; 241: 116349. https://doi.org/10.1016/j.carbpol.2020.116349
CrossRef - Liu, J., Wang, S., Xu, K., and Fan, Z., Fabrication of Double Crosslinked Chitosan/Gelatin Membranes with Na⁺ and pH Dual‑Responsive Controlled Permeability. Carbohydrate Polymers, 2020; 236: https://doi.org/10.1016/j.carbpol.2020.115963
CrossRef - Bhumkar and Pokharkar. Synthesis and Characterization of Chitosan‑modified Membrane for Urea Slow‑release F Heliyon. 2024; https://doi.org/10.1016/j.heliyon.2024.e11012
CrossRef - Di Santo, M. C., D’Antoni, C. L., Domínguez Rubio, A. P., Alaimo, A., and Pérez, O. E. Chitosan–tripolyphosphate Nanoparticles Designed to Encapsulate Polyphenolic Compounds for Biomedical and Pharmaceutical Applications – A review. Biomedicine & Pharmacotherapy, 2021; 142: https://doi.org/10.1016/j.biopha.2021.111970
CrossRef - Andrade, D., Maldonado‑Bravo, F., Alburquerque, A., Pérez, C., Gamboa, A., Caro, N., Díaz‑Dosque, M., Gotelland, M., Abugoch, L., and Tapia, C. Nanoencapsulation of Maqui (Aristotelia chilensis) Extract in Chitosan–Tripolyphosphate and Chenopodin‑Based Systems. Antioxidants, 2024; 13(3), 273. https://doi.org/10.3390/antiox13030273
CrossRef - Di Santo, M. C., D’Antoni, C. L., Domínguez Rubio, A. P., Alaimo, A., and Pérez, O. E. Chitosan–tripolyphosphate Nanoparticles Designed to Encapsulate Polyphenolic Compounds for Biomedical and Pharmaceutical Applications – A review. Biomedicine & Pharmacotherapy, 2021; 142: 111970. https://doi.org/10.1016/j.biopha.2021.111970
CrossRef - Joudeh, N., and Linke, D. Nanoparticle Classification, Physicochemical Properties, Characterization, and Applications: a Comprehensive Review for Biologists. Journal of Nanobiotechnology, 2022; 20(1): https://doi.org/10.1186/s12951-022-01477-8
CrossRef - Saeed, R. M., Dmour, I., and Taha, M. O. Stable Chitosan‑based Nanoparticles Using Polyphosphoric Acid or Hexametaphosphate for Tandem Ionotropic/Covalent Crosslinking and Subsequent Investigation as Novel Vehicles for Drug Delivery. Frontiers in Bioengineering and Biotechnology, 2020; 8, 4: https://doi.org/10.3389/fbioe.2020.00004
CrossRef - Bangun, Y., Hutagalung, S. O., and Simanjuntak, P. Preparation and Evaluation of Chitosan‑tripolyphosphate Nanoparticles Suspension by the Ionic Gelation MJournal of Applied Pharmaceutical Science, 2018; 8(12): 147–156. DOI : Not applicable
CrossRef - Detsi, A., Kavetsou, E., Kostopoulou, I., Pitterou, I., Pontillo, A. R. N., Tzani, A., and Oumpoulakis, Nanosystems for the Encapsulation of Natural Products: The Case of Chitosan Biopolymer as a Matrix. Pharmaceutics, 2020; 12(7): 669.DOI: Not applicable
CrossRef - Bhattacharjee, S. DLS and Zeta Potential – What They are and What They are not. Journal of Controlled Release, 2016; 235: 337–351. https://doi.org/10.1016/j.jconrel.2016.06.049
CrossRef - Mota, W. S., Severino, P., Kadian, V., Rao, R., Zielińska, A., Silva, A. M., Mahant, S., and Souto, E. B. Nanometrology: Particle Sizing and Influence on the Toxicological P Frontiers in Nanotechnology, 2025; 7: 1479464. https://doi.org/10.3389/fnano.2025.1479464
CrossRef - Madni, A., Smith, B., and Lee, C.-H. Application of Ensemble Machine Learning Approach to Assess the Stability of Nanoparticle Formulations. Scientific Reports, 2023; 13: 12345. https://doi.org/10.1038/s41598-023-43689-4
CrossRef - Gul, R., Khan, A., and Ullah, S. The Impact of Particle Size and Polydispersity Index on the Clinical Efficacy of Drug Delivery Systems. International Journal of Pharmaceutics, 2017; 525(1): 212–223. https://doi.org/10.1016/j.ijpharm.2017.04.053
CrossRef - Singh, R., and Lohan, S. Significance of Polydispersity Index in Nanoparticles: Correlation with Stability and Performance. Journal of Colloid and Interface Science, 2021; 606(1): 78–89. https://doi.org/10.1016/j.jcis.2021.05.012
CrossRef - Mohan, A., and Patel, P. Role of Polydispersity on the Aggregate Formation and Sedimentation in Solid Lipid Nanoparticles. Food Research International, 2018; 113: 123–131. https://doi.org/10.1016/j.foodres.2018.06.027
CrossRef - Mozafari, M. R. Unlocking the Power of Artificial Intelligence: Accurate Zeta Potential Prediction of N Nanomaterials, 2022; 13(7): 1209. https://doi.org/10.3390/nano13071209
CrossRef - Bhattacharjee, S. Zeta Potential – an Introduction in 30 Minutes. 2016; Malvern Instruments Ltd. DOI: Not applicable
- Bordonaba-Juste, I.,. Gold Nanoparticle Shape Dependence of Colloidal Stability D Nanoscale Advances, 2023; 5: Article d2na00809b. https://doi.org/10.1039/d2na00809b
CrossRef - Skalko–Basnet, N., Quality by Design–Driven Zeta Potential Optimisation Study of Liposomal F Pharmaceutics, 2022; 14(10): 2190. https://doi.org/10.3390/pharmaceutics14102190
CrossRef - Kumar, A., Influence of Preparation Characteristics on Stability, Properties, and Performance of Mono- and Hybrid Nanofluids: Current and Future Perspective. Processes, 2020; 11(1): https://doi.org/10.3390/pr11010112
CrossRef - Rahman, M., Advances in Stabilization of Metallic Nanoparticle with Biosurfactants – a Review on Current Trends. Heliyon, 2024; 10(9): https://doi.org/10.1016/j.heliyon.2024.e29773
CrossRef
|
Abbreviation |
Full Term |
|
MDA |
Malondialdehyde |
|
SOD |
Superoxide Dismutase |
|
SOD-2 |
Superoxide Dismutase 2 (Manganese-dependent, mitochondrial isoform) |
|
SOD-3 |
Superoxide Dismutase 3 (Extracellular isoform, EC-SOD) |
|
ICAM-1 |
Intercellular Adhesion Molecule-1 |
|
IL-6 |
Interleukin-6 |
|
TNF-α |
Tumor Necrosis Factor-alpha |
|
NaTPP |
Sodium Tripolyphosphate |
|
DPPH |
2,2-Diphenyl-1-picrylhydrazyl |
|
IC50 |
Half maximal inhibitory concentration |
|
LDL |
Low-Density Lipoprotein |
|
HDL |
High-Density Lipoprotein |







