Manuscript accepted on :11-11-2025
Published online on: 03-12-2025
Plagiarism Check: Yes
Reviewed by: Dr. Emmanuel Dike
Second Review by: Dr. Nina Mariana
Final Approval by: Dr. Anton R Keslav
DA WA1.2 , Dejidmaa Buyantogtokh 3*
, Dejidmaa Buyantogtokh 3* , Erdenechimeg Chuluunbaatar4
, Erdenechimeg Chuluunbaatar4 , Chimedragchaa Chimedragchaa3
, Chimedragchaa Chimedragchaa3 and Tsend-Ayush Damba5
and Tsend-Ayush Damba5
1Department of Internal Medicine, International school of Mongolian medicine, Mongolian National University of Medical Sciences, Ulaanbaatar, Mongolia,
2Department of Gal zasal, Inner Mongolia International Mongolian Hospital, Hohhot, China
3Department of Pharmacology, Research Center, Institute of Traditional Medicine and Technology, Ulaanbaatar, Mongolia,
4Department of Chemistry and Technology, Research Center, Institute of Traditional Medicine and Technology, Ulaanbaatar, Mongolia,
5Department of Mongolia Medicine Study, Institute of Traditional Medicine and Technology of Mongolia, Ulaanbaatar, Mongolia
Corresponding Author E-mail: b.dejidmaa@yahoo.com
DOI : https://dx.doi.org/10.13005/bpj/3329
Abstract
In Mongolian traditional medicine, Sapparin tablets are used to treat blood disorders and liver diseases. These tablets consist of four main ingredients: C. jubata, C. sappan, Z. officinale, and H.rhamnoides. This study evaluates the antihyperglycemic and anticoagulant effects of Sapparin tablets in a T2DM induced by streptozotocin and a high-fat food. Forty rats were divided into 4 groups (n = 10): normal control, diabetic control and two groups of Sapparin. Wistar rats were fed a high-fat diet at a rate of 40 grams per day for 28 days. On the 29th day, after fasting for 18 hours, the rats were intraperitoneally injected with streptozotocin at a dose of 35 mg/kg. Following this, two different doses of Sapparin (89 mg/kg and 178 mg/kg) were administered to the T2DM (type 2 diabetes mellitus) rats via oral gavage. The experimental results indicated that Sapparin significantly reduced fasting glucose levels, fasting blood insulin, triglycerides, cholesterol, and HDL. We demonstrated that Sapparin effectively alleviates diabetic symptoms by lowering fasting glucose and insulin levels while increasing GLUT4 levels. Additionally, Sapparin protects against insulin resistance, enhances superoxide dismutase activity, and improves lipid profiles in rats with type 2 diabetes mellitus
Keywords
Antidiabetic; C.Jubata; C.Sappan; Z.Officinale; H.rhamnoides; T2DM
Download this article as:| Copy the following to cite this article: WA D. A, Buyantogtokh D, Chuluunbaatar E, Chimedragchaa C, Damba T. A. Evaluation of the Antihyperglycemic and Anticoagulant Effects of Sapparin Tablets in Streptozotocin+High Fat Diet Induced Diabetic Rats. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: WA D. A, Buyantogtokh D, Chuluunbaatar E, Chimedragchaa C, Damba T. A. Evaluation of the Antihyperglycemic and Anticoagulant Effects of Sapparin Tablets in Streptozotocin+High Fat Diet Induced Diabetic Rats. Biomed Pharmacol J 2025;18(4). Available from: https://bit.ly/4phbUVK |
Introduction
Diabetes is a progressive condition that can contribute to a range of cardiovascular complications. It is important to note that metabolic changes and alterations in the blood coagulation system are interconnected, potentially leading to disruptions in the function of blood platelets.1.2
In a 1999 diabetes prevalence survey conducted in Mongolia, it was found that the diabetes prevalence among individuals over the age of 35 was 3.1%, while the rate of impaired glucose tolerance was 9.2%.3.4
The treatment of diabetes involves several approaches, including non-drug interventions such as dietary changes, regular exercise, and health education. Drug therapy: This consists of the use of glucose-lowering medications and insulin injections to manage blood sugar levels. Diabetes Management: This focuses on monitoring treatment results, preventing complications, and detecting any issues early. Emergency Care: This is crucial for managing hypoglycemia. Treatment of Chronic Complications: This includes addressing issues related to both large and small blood vessel complications, such as retinopathy, neuropathy, leg ulcers, necrosis, kidney complications, dyslipidemia, arterial hypertension, heart attacks, and strokes. Each component plays a vital role in effectively managing diabetes and ensuring overall health.1-4
In addition to the standard treatments available, a variety of natural remedies are also being explored for the management of diabetes and the potential reduction of related complications.5-6 Sapparin tablets, which are derived from Mongolian traditional medicine, are one such remedy.
Sapparin tablets are composed of 4 medicinal ingredients, including Caragana jubata (Pall.) Poiret, Zingiber officinale (Roscoe), Hippophae rhamnoides (L.), and Caesalpinia sappan (L.).7.8
C.jubata contains polyphenolic compounds, particularly flavonoids, which were identified in the freeze-dried aqueous extract of the plant. These flavonoids include mono- and diglycosides of primarily O-hydroxylated flavonols such as myricetin, quercetin, and kaempferol, as well as O-methylated flavonols like isorhamnetin, laricitrin, and syringetin. A new pterocarpan glycoside, named maackiain, has been isolated from the plant Caragana jubata (Pall.). C. Jubata is recognized for its anti-inflammatory, antibacterial, antithrombotic, and antitumor properties, as well as its benefits in ischemic stroke and antidiabetic effects.9-11
C.sappan contains several biologically active substances, including brazilin, chalcone, xanthones, protosappanin, flavones, homoisoflavonoids, diterpenes, and phenolic compounds. C.Sappan has been studied for its antiplatelet activity, treatment of hypercholesterolemia, and management of type 2 diabetes, as well as its antioxidant, wound healing, antibacterial, and antitumor properties.12-14
Ginger is known for its variety of valuable active compounds, particularly phenolic and terpene compounds. The phenolic compounds found in ginger include gingerols, shogaols, and paradols, as well as quercetin, zingerone, gingerenone-A and 6-dehydrogingerdione. Additionally, ginger is rich in terpene components, such as β-bisabolene, which are recognized as the primary constituents of ginger essential oils.15-17
H.rhamnoides, commonly known as sea buckthorn, is consumed globally as both a food and health supplement due to its rich nutritional and medicinal properties. Phytochemical studies have identified a diverse array of phytonutrients, which include essential dietary components such as proteins, minerals, and vitamins, as well as functional components like flavonoids, lignans, volatile oils, tannins, terpenoids, steroids, organic acids, and alkaloids. H.rhamnoides has antidiabetic properties, which are attributed to the flavonoids it contains.18-20
Sapparin tablets are utilized in treating blood disorders, specifically for addressing thickened blood or poor blood quality, as well as liver diseases, according to Mongolian Traditional Medicine. Previous studies have indicated that Sapparin possesses a total phenolic content of 5.33 ± 0.0005%, flavonoids at 12.95 ± 2.21%, and carotenoids at 4.31 ± 0.96%. Notably, significant increases were observed across all tests, with the exception of fibrinogen, PT, TT and aPTT, which are recognised for their roles in anticoagulation. Furthermore, treatment with Sapparin has been associated with a reduction in serum factor X levels and von Willebrand factor in rat subjects, suggesting potential beneficial effects that warrant further exploration.21
This study evaluates the antihyperglycemic and anticoagulant effects of Sapparin tablets in a diabetic model induced by streptozotocin and a high-fat diet.
Materials and Methods
Ethics statement
The Research Ethics Committee at the National University of Medical Sciences has granted ethical approval for this research.The approval was granted under protocol number 2024/3-06. This study did not involve human participants, and therefore, informed consent was not required.
Reagent
In our experiment, we utilised Streptozotocin (CAS Number: 18883-66-4), obtained from Sigma-Aldrich, along with a citrate buffer at pH 4.5 (C2488). The dietary composition included a high fat diet (HFD) with 20% carbohydrates, 20% protein, and 60% fat, as specified by Batch No. SYHF45.
All experimental animals were sourced from the Institute of Traditional Medicine Technology’s animal house. The rats were maintained in a box at room temperature, with a controlled 12-hour light/dark cycle to ensure optimal living conditions. Forty Wistar rats weighing between 170 and 180 grams were randomly divided into four groups (n=10). They were labelled as follows: diabetic control (DC), normal control (NC), and Sapparin-treated (S) at dosages of 89 mg/kg and 178 mg/kg. Blood samples were collected at 14 and 21 days, with a total of n=5 rats. This sample size was determined to achieve statistical significance in the study.
Induction of the type 2 diabetes mellitus (T2DM)
40 wistar rats were fed a high-fat diet consisting of 20% carbohydrates, 20% protein, and 60% fat, administered at a rate of 40 grams per day for 28 days. On the 29th day of the study, following an 18-hour fast, the rats received an intraperitoneal injection of streptozotocin (STZ) at a dose of 35 mg/kg.
To maintain the stability and effectiveness of the study, STZ was dissolved in freshly prepared ice-cold citrate buffer, and the pH was adjusted to 4.5. After a 72-hour observation period following the injection, rats exhibiting fasting glucose levels of 11.1 mmol/L or higher—indicative of hyperglycemia—were selected for the study.5 Following the administration of streptozotocin, Sapparin was orally received to the diabetic rats for a duration of 21 days at doses of 89 mg/kg and 178 mg/kg.
Blood samples, ranging from 3 to 4 ml, were collected from both the diabetic and normal control groups using cardiac puncture with 1:9 citrate and heparin tubes at the 14-day and 21-day marks. A biochemical analysis was performed employing a DR 7007 analyser to assess levels of triglycerides, cholesterol, and HDL across all experimental groups. Furthermore, serum concentrations of insulin, GLUT4, superoxide dismutase (SOD), plasminogen, the antithrombin complex (TAT) were determined using an ELISA assay at 14 and 21 days, in accordance with the manufacturer’s protocol.
Fasting glucose and body weight
Fasting blood glucose levels were measured using a glucometer (GluNEO Lite, South Korea) by collecting a tail-drop blood sample with the glucose oxidase method. These measurements were taken once a week for three weeks, particularly after the plasma glucose level reached or exceeded 11.1 mmol/L. Additionally, body weight was recorded using an electronic scale to ensure accurate and consistent measurements throughout the study.
Oral glucose tolerance test (OGTT)
On day 18, rats were fasted overnight and then given 2 g/kg of oral glucose at 9 a.m. the following morning for the oral glucose tolerance test. Glucose levels were measured at 0, 30, 60, and 120 minutes.5.22
Fasting serum insulin levels (FINS) and homeostasis model assessment of insulin resistance (HOMA-IR) analysis
The fasting serum insulin level (FINS) is an essential marker for assessing metabolic health, particularly in evaluating insulin resistance. FINS is measured utilising an enzyme-linked immunosorbent assay (ELISA) insulin assay kit from MLBIO Biotechnology Co., Ltd, based in Shanghai, China. To quantify insulin resistance, the Homeostasis Model Assessment of Insulin Resistance (HOMA-IR) is employed. This assessment is calculated using the formula: HOMA
score = fasting serum insulin (𝜇U/mL) × fasting glucose (mmol/L)/22.5
Assay of glycogen
Glycogen in muscle tissue is determined using Anthron’s reagent22
Histopathological examination
At the end of the experiment, euthanasia was performed on the rats using an overdose of pentobarbital, and their pancreatic tissues were collected for histopathological analysis. The collected tissues were fixed in 10% buffered formalin for 24 hours. After fixation, the samples were rinsed with running water and then processed through a series of incubations in increasing concentrations of ethanol, xylene, and paraffin. The tissues were embedded in paraffin blocks and sectioned to a thickness of 2-5 μm using a Yamato Kohki sledge microtome. The resulting sections were stained with hematoxylin and eosin, prepared as microslides, and examined morphologically under a Nikon Eclipse Ci microscope.
Statistical analysis
The data are presented as means ± standard deviations (SD). Statistical analysis was conducted using SPSS version 22.0 (SPSS Inc., Chicago, IL, USA). Differences between the study groups were evaluated using the Kruskal-Wallis test, with a p-value of less than 0.05 considered statistically significant.
Results
On the 14th day of the experiment, the diabetic control group exhibited a significant increase in glucose levels, rising 1.35-fold compared to the normal control group (p = 0.001). This finding confirmed the successful establishment of the diabetic model.
In contrast, administering Sapparin yielded promising results. The group that received 89 mg/kg of Sapparin experienced a 33% reduction in glucose levels. Meanwhile, the group that received 178 mg/kg of Sapparin showed a 22.5% reduction compared to the diabetic control group (p = 0.05).
Table 1: Effect of Sapparin on fasting glucose levels in a T2DM
| Time of treatment | Group | Glucose (mmol/l) |
| 2 week | Normal control | 6.04±1.3 |
| DC (HFD+STZ) | 14.2±1.4*** | |
| DC+S-89 mg/kg | 9.54±1.2# | |
| DC+S-178 mg/kg | 11.0±1.7# | |
| 3 week | Normal control | 6.04±1.3 |
| DC (HFD+STZ) | 16.4±1.8*** | |
| DC+S-89 mg/kg | 11.4±1.6# | |
| DC+S-178 mg/kg | 11.5±1.2# |
***p < 0.001 compared to normal controls; #p < 0.04 compared to diabetic controls.
By the 21st day of observation, the diabetic control group displayed an even greater increase in glucose levels, with a 1.72-fold rise compared to the normal control (p=0.001). However, the groups receiving 89 mg/kg and 178 mg/kg of Sapparin demonstrated notable reductions in glucose levels by 30% when compared to the diabetic model (p<0.04). These findings suggest a potential therapeutic effect of Sapparin in regulating glucose levels in diabetic conditions.
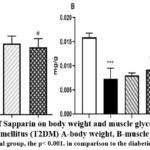 |
Figure 1: The effects of Sapparin on body weight and muscle glycogen in rats with type 2 diabetes mellitus (T2DM) A-body weight, B-muscle glycogen |
The findings revealed that the diabetic control group exhibited a 45.3% increase in body weight when compared to the control group on a regular diet (p=0.001). Conversely, administration of Sapparin at doses of 89 mg/kg and 178 mg/kg resulted in a trend of body weight reduction, with losses ranging from 9.04% to 13.6% relative to the diabetic control group (p=0.05). The results demonstrate that glycogen levels in the diabetic control group were significantly reduced compared to those in the normal control group (p=0.001). In the diabetic group administered Sapparin at a dosage of 178 mg/kg, there was a statistically significant increase in glycogen levels (p=0.05).
Table 2: The impact of Sapparin on blood glucose levels: a glucose tolerance test in rats with T2DM
| Glucose (mmol/l) | Time (min) | Normal control |
DC (HFD+STZ) | DC+S-89 mg/kg | DC+S-178 mg/kg |
| 0 | 5.9±1.1 | 7.1±0.6* | 9.3±1.3# | 5.8±0.3# | |
| 30 | 6.7±0.3 | 11.3±1.6*** | 10.2±1.9 | 7.9±2.8 | |
| 60 | 6.6±0.8 | 8.5±1.0** | 8.8±1.8 | 7.6±1.3# | |
| 120 | 5.7±0.5 | 10.4±1.0*** | 7.3±1.8# | 6.7±0.8# |
Compared to normal control, significance levels are indicated as follows: *p<0.05, **p<0.001, and ***p<0.000. When comparing to the diabetic control group, statistical significance is denoted as #p<0.04.
At the end of the study, glucose levels in normal control rats were measured to be between 5.9 and 6.7 mmol/L during the glucose tolerance test. In diabetic rats, blood glucose levels increased significantly to 20.3% at 0 minutes, 68.6% at 30 minutes, 15.1% at 60 minutes, and 82.4% at 120 minutes (p< 0.001).
In a study examining the effects of Sapparin on glucose levels, the group treated with 89 mg/kg exhibited a 31% increase in glucose concentration at the initial measurement compared to the diabetic control group. However, this trend was reversed over time, as glucose levels decreased by 10% at the 30-minute mark and further declined by 30% at the 120-minute measurement. The administration of Sapparin at a dosage of 178 mg/kg resulted in significantly lower glucose levels at 0, 30, 60, and 120 minutes when compared to the diabetic control group. The administration of Sapparin at a dosage of 178 mg/kg appeared to have a positive impact, significantly lowering glucose levels at 0, 30, 60, and 120 minutes compared with the diabetic control. The percentage reductions observed were 18.3%, 30.1%, 10.6%, and 35.6%, respectively (p<0.04).
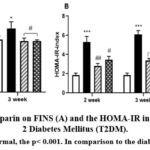 |
Figure 2: Activity of Sapparin on FINS (A) and the HOMA-IR index (B) in rats with Type 2 Diabetes Mellitus (T2DM). |
When comparing the normal, the p< 0.001. In comparison to the diabetic control, the p< 0.05.
The outcomes of this investigation elaborated on the changes in serum insulin concentrations among T2DM rat models. Data demonstrated that post-14 days, serum insulin levels in the T2DM exhibited a significantly increase of 20.5% relative to normal controls, escalating to 21% by the end of the 21-day period (p<0.05). Conversely, administration of 89 mg/kg of Sapparin resulted in a notable reduction in insulin levels, with declines of 20% at 14 days and 19.8% at 21 days in comparison to the T2DM group. Additionally, the group subjected to a higher dose of 178 mg/kg of Sapparin showed decreases of 15% and 20% in insulin levels, which were statistically analogous to those observed in the normal control cohort (p<0.05). These findings suggest that Sapparin may play a role in modulating insulin levels in diabetic conditions. The results indicated a significant increase in HOMA-IR, a measure of insulin resistance, in rats with Type 2 Diabetes Mellitus (T2DM) compared to normal controls. This increase was observed in a time-dependent manner, with a 1.85-fold elevation at the two-week mark and a 2.28-fold elevation at three weeks (p=0.001). In diabetic rats administered doses of 89 and 178 mg/kg of Sapparin, there was a statistically significant time-dependent reduction of 42-46% in HOMA-IR (p < 0.05).
GLUT4 (Glucose Transporter Type 4) is a protein responsible for the transport of glucose, a hydrophilic sugar, into cells. GLUT4 plays a crucial role in glucose homeostasis by allowing cells to uptake glucose in response to insulin signaling, thereby regulating blood sugar levels. In the context of insulin resistance associated with type 2 diabetes, GLUT4 translocation to the plasma membrane is impaired. This inhibition reduces cells’ ability to take up glucose, leading to elevated blood sugar levels.
Table 3: Effects of Sapparin on serum GLUT4 and lipid parameters in T2DM
| Time of treatment | Group | GLUT4(ng/ml) | Triglyceride (mmol/l) | Cholesterol (mmol/l) | HDL(mmol/l) |
| 2 week | Normal control | 6.5±0.64 | 1.0±0.2 | 2.5±0.6 | 1.35±0.1 |
| DC (HFD+STZ) | 4.42±0.9** | 1.60±0.3** | 3.3±0.2* | 0.4±0.06** | |
| DC+S-89 mg/kg | 6.35±0.5# | 1.30±0.4# | 2.3±0.2# | 1.15±0.3## | |
| DC+S-178 mg/kg | 4.91±0.4 | 1.40±0.3 | 2.2±0.4# | 1.1±0.5## | |
| 3 week | Normal control | 6.5±0.64 | 1.0±0.2 | 2.5±0.6 | 1.35±0.1 |
| DC (HFD+STZ) | 3.77±0.6** | 2.5±0.5*** | 4.2±0.9** | 0.4±0.02** | |
| DC+S-89 mg/kg | 6.28±0.3## | 1.90±0.3 | 3.2±0.7 | 1.5±0.05## | |
| DC+S-178 mg/kg | 4.95±0.3# | 1.2±0.2## | 3.3±0.1 | 1.4±0.04## |
***p <0.01 when compared to the normal control group; #p < 0.05, ##p < 0.02 when compared to the diabetic control group
Results were analyzed to assess the impact of Sapparin on GLUT4 levels in diabetic rats, compared with normal controls. The diabetic control group exhibited significantly lower GLUT4 levels, decreasing by 32% by day 14 and by 42% by day 21 (p<0.01).
Sapparin, administered at a dose of 89 mg/kg, significantly increased GLUT4 levels, showing improvements of 44% on day 14 and 66.6% on day 21. In contrast, the group receiving a higher dose of 178 mg/kg of Sapparin exhibited smaller increases, with only 11.1% on day 14 and 31.3% on day 21 (p<0.02). These findings indicate that Sapparin positively affects GLUT4 expression in diabetes.
In the diabetic control group, triglyceride levels increased significantly at both 14 and 21 days, by 60% and 1.5-fold, respectively, compared to normal controls (p=0.000). Administration of the 89 mg/kg dose of Sapparin resulted in a reduction of triglyceride levels by 19% and 24% on days 14 and 21, respectively, relative to the diabetic control group. Similarly, the 178 mg/kg dose of Sapparin induced a decrease of 12.5% and 52% on days 14 and 21, respectively (p<0.002).
The results indicated a substantial increase in cholesterol levels, rising by 32% and 68% after 14 and 21 days, respectively, compared to normal controls (p<0.001).
When treated with Sapparin at a dose of 89 mg/kg, cholesterol levels decreased by 30.3% after 14 days and by 24% after 21 days. Additionally, Sapparin 178 mg/kg resulted in even greater reductions, with decreases of 33.3% and 21.4% at the respective time points (p<0.04).
The diabetic control group exhibited a significant decrease in high-density lipoprotein (HDL) levels, which were 70.4% lower at both 14 and 21 days when compared to the normal control group. In contrast, treatment with Sapparin at 89 mg/kg increased HDL levels by 1.87-fold at 14 days and 2.75-fold at 21 days.
Likewise, a higher dose of 178 mg/kg of Sapparin also yielded significant gains, with HDL levels rising by 1.75-fold at 14 days and 2.5-fold at 21 days. These findings were statistically significant, with a p-value of 0.002, suggesting the potential efficacy of Sapparin in enhancing HDL levels in diabetic conditions.
Superoxide Dismutase (SOD) is an antioxidant enzyme found in the human body and in all living cells. Research underscores a significant relationship between SOD levels and diabetes, particularly type 2 diabetes (T2DM). This association is believed to arise from the role of SOD in mitigating oxidative stress, which is a contributing factor to the pathophysiology of T2DM.
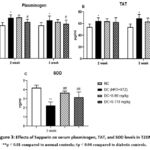 |
Figure 3: Effects of Sapparin on serum plasminogen, TAT, and SOD levels in T2DM. |
**p < 0.01 compared to normal controls; #p < 0.04 compared to diabetic controls.
This experiment aimed to investigate the effects of the drug Sapparin on coagulation parameters, with a particular emphasis on plasminogen levels. In the diabetic model group, plasminogen levels increased by 22.1% on day 14 and by 14.6% on day 21, compared to established normal reference values. However, administration of Sapparin at a dose of 178 mg/kg significantly reduced plasminogen levels, resulting in a decrease of 10.2% on day 14 and 8.5% on day 21 (p=0.05).
The Thrombin–Antithrombin Complex (TAT complex) is a vital marker of the blood coagulation system. It is formed by the interaction between thrombin and antithrombin III and serves as an essential indicator of blood activation for clot formation. In diabetes, elevated blood glucose levels lead to hyperglycemia, which contributes to oxidative stress, inflammation, and endothelial cell damage in blood vessels. This cascade of events activates the blood coagulation system, leading to increased thrombin production. As thrombin levels rise, antithrombin (AT III) binds to thrombin, forming a complex known as thrombin-antithrombin (TAT) that effectively inactivates thrombin. Consequently, elevated TAT levels serve as a direct indicator of active thrombin formation within the bloodstream.
The thrombin-antithrombin complex (TAT) levels in the diabetic model demonstrated an increase of 18.3% after 14 days and 28% after 21 days compared to the normal control. Furthermore, in the group that received a dose of 178 mg/kg of Sapparin, TAT levels were 18% lower than those in the diabetic control group on day 21, and this difference was statistically significant (p=0.05). After 21 days of observation, superoxide dismutase (SOD) levels in the diabetic control group were significantly decreased by 46.2% compared to the normal group (p< 0.01).
Conversely, the group receiving a dosage of 89 mg/kg experienced an increase in SOD levels of 61.5%, while the group administered 178 mg/kg displayed a 40% increase (p <0.02). These findings indicate a noteworthy response to the administered dosages in contrast to the diabetic control conditions.
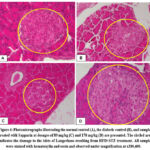 |
Figure 4: Photomicrographs illustrating the normal control (A), the diabetic control (B), and samples treated with Sapparin at dosages of 89 mg/kg (C) and 178 mg/kg (D) are presented. |
In healthy rats or normal control, the ratio of the exocrine and endocrine components of the pancreas is considered normal. The endocrine system’s central element, the islets of Langerhans, contains a substantial population of beta cells as well as various other secretory cells. Furthermore, the structure of the exocrine component, represented by the acinar gland, is also characterized as normal.
The analysis of pancreatic tissue from the diabetic control group indicated a greater occurrence of inflammation and degenerative changes in comparison to the other experimental groups. Notably, there was a significant reduction in both the number and size of beta cells within the pancreatic islets of Langerhans. The pancreatic tissue of rats in the 89 mg/kg Sapparin group demonstrated significantly fewer pathological changes, reduced levels of necrosis and inflammation, and a greater density of cells within the islets of Langerhans when compared to the other experimental groups. In the experimental group of animals administered Sapparin at a dosage of 178 mg/kg, there was a noticeable presentation of slight swelling and necrosis within the pancreatic tissue. Additionally, a slight decrease in the islet cell population was noted. In comparison to the diabetic control groups, the experimental groups showed significantly fewer pancreatic changes at the Sapparin 89 mg/kg dosage.
Discussion
Type 2 diabetes is one of the most prevalent metabolic disorders globally, representing 90-95% of all diabetes cases. The prevalence of this disease is increasing significantly in both developed and developing countries, which calls for our attention and collective action. The pathogenesis of this condition is primarily rooted in insulin resistance and the dysfunction of pancreatic beta cells. Consequently, therapeutic strategies are designed to target and mitigate these underlying factors, focusing on enhancing insulin sensitivity and restoring beta cell function.1-4 This dual approach is critical for effectively managing the condition and improving patient outcomes. Recent research efforts have increasingly focused on developing a range of natural medicines to supplement standard treatments for type 2 diabetes, as well as to prevent its associated complications. This includes rigorous validation of traditional knowledge and recipes to establish their effectiveness in diabetes management. In this study, we used a rat model of insulin resistance and hyperlipidemia, a condition known as type 2 diabetes mellitus (T2DM). This model was induced by administering a high-fat diet (45% HFD) along with a low dose of streptozotocin (STZ).
The therapeutic efficacy was subsequently evaluated by administering two different doses of Sapparin. In our study employing a streptozotocin-induced model of type 2 diabetes, rats fed a high-fat diet exhibited consistently elevated blood glucose levels. Notable insulin resistance was observed, characterised by increased fasting insulin levels and a heightened insulin resistance index. These findings are similar to the biochemical and pathological alterations seen in human type 2 diabetes. Furthermore, significant changes were documented in specific coagulation markers, with elevated levels of plasminogen and thrombin-antithrombin complex (TAT) noted.
Sapparin was administered orally to rats with type 2 diabetes mellitus (T2DM) at doses of 89 mg/kg and 178 mg/kg over 21 days.
Evaluations conducted on days 14 and 21 demonstrated significant favorable outcomes. These results included a reduction in blood glucose levels, decreased insulin levels, enhanced insulin sensitivity, and a lower HOMA-IR index. Furthermore, there were an increase in the expression of glucose transporter type 4 (GLUT4) and an elevation in glycogen content within muscle tissue. Additionally, a decrease was observed in specific lipid metabolism markers, notably triglycerides and cholesterol, accompanied by an increase in high-density lipoprotein (HDL) levels. Studies have indicated that Sapparin exerts a corrective influence on coagulation abnormalities associated with type 2 diabetes mellitus (T2DM). Notably, Sapparin reduces plasminogen and thrombin-antithrombin (TAT) levels at a dose of 178 mg/kg. Additionally, it has been shown to increase serum superoxide dismutase (SOD) levels in a T2DM model at doses of 89 and 178 mg/kg.
The effects of Sapparin in treating diabetes may relate to its biologically active compounds. Flavonoids extracted from the plant C. jubata enhance glucose uptake and improve the expression of GLUT4. This highlights the important role of Ca²⁺ release in the regulation of GLUT4. Additionally, these flavonoids primarily activate the PKC pathway, with minimal involvement of the Akt pathway.
C. jubata extract significantly lowered fasting glucose levels, enhanced glucose tolerance, reduced insulin resistance, and corrected lipid metabolism abnormalities in mice with type 2 diabetes. Additionally, the extract enhanced tissue insulin sensitivity through the upregulation of GLUT4 expression and mitigated damage to both the liver and pancreas.10 The chloroform extract of C. Sappan demonstrated a significant ability to lower serum glucose levels and modulate cholesterol and triglyceride levels in alloxan-induced diabetes models, particularly at doses of 200- 400 mg/kg. In a recent study involving rats fed a high-fat diet, treatment with combined extracts of C. sappan and Z. officinale at three varying doses over a two-week period resulted in a significant reduction in platelet activation markers, namely P-selectin, RANTES, and PCSK9. Moreover, the results indicate that bleeding time increases in a dose-dependent manner compared with the negative control group.12.13.23 A substantial body of research has investigated the antidiabetic properties of ginger, a widely recognized component in dietary interventions for type 2 diabetes. The mechanism by which ginger exerts its effects involves the activation of HMG-CoA reductase, which subsequently stimulates lipid and carbohydrate metabolism. This activation leads to a reduction in low-density lipoprotein (LDL) levels and is associated with significant antioxidant activity. It has been indicated that it enhances insulin sensitivity, protects pancreatic β cells, decreases the accumulation of fat, reduces oxidative stress, and improves glucose utilisation in muscle tissue.
The findings from these studies are consistent with the results of our experiments, thereby providing further support for the hypothesis that Sapparin is effective in the treatment of type 2 diabetes.
A limitation of our study is that we did not evaluate clinical outcomes associated with Sapparin administration in human subjects. Therefore, future clinical studies are necessary to investigate its effects on type 2 diabetes.
Conclusion
In this study, we demonstrated that Sapparin effectively alleviates diabetic symptoms by lowering fasting glucose and insulin levels while increasing GLUT4 levels. Furthermore, Sapparin protects against insulin resistance, raises superoxide dismutase levels and improves abnormal lipid profiles in rats with type 2 diabetes mellitus.
Acknowledgment
We thank the Mongolian Academy of Science and the Mongolian Foundation for Science and Technology. We also want to express our respect for the research workers in the Institute of Traditional Medicine and Technology for their assistance in executing this study.
Funding source
The Mongolian Foundation for Science and Technology funded this project. The agreement number of the project was S24/46
Conflict of Interest
The author(s) do not have any conflict of interest
Data Availability Statement
This statement does not apply to this article.
Ethics statement
This study was approved by the Research Ethics Committee of the National University of Medical Sciences of Mongolia. (Approval No 2024/3-06).
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Author Contributions
- DA WA: Data collection, Analysis, Writing- Original Draft
- Dejidmaa Buyantogtokh: Data collection, Analysis, review and & Editing, and Project
- Erdenechimeg Chuluunbaatar: Data collection, Analysis, Review& Editing
- Chimedragchaa Chimedtseren: Funding Acquisition, Resources, Supervision
- Tsend-Ayush Damba: Visualization, Supervision
References
- Hess K. The vulnerable blood. Coagulation and clot structure in diabetes mellitus. Hamostaseologie. 2015;35(1):25-33.
CrossRef - Pretorius L, Thomson GJA, Adams RCM, Nell TA, Laubscher WA, Pretorius E. Platelet activity and hypercoagulation in type 2 diabetes. Cardiovasc Diabetol. 2018;17(1):141.
CrossRef - Suvd J, Gerel B, Otgooloi H, Purevsuren D, Zolzaya H, Roglic G, King H. Glucose intolerance and associated factors in Mongolia: results of a national survey. Diabet Med. 2002;19(6):502-8.
CrossRef - Dayan A, Erkhembayar R, Luvsandavaajav O, Mukhtar Y, Enkhtuvshin B, Tumenbayar B. Prevalence of Type 2 Diabetes in Mongolia: Results from Population-Based Survey Compared with 1999 Study. Diabetes Metab Syndr Obes. 2023; 21(16):1833-1846.
CrossRef - Duan B, Zhao Z, Lin L, Jin J, Zhang L, Xiong H, Ta N, Gao T, Mei Z. Antidiabetic Effect of Tibetan Medicine Tang-Kang-Fu-San on High-Fat Diet and Streptozotocin-Induced Type 2 Diabetic Rats. Evid Based Complement Alternat Med. 2017;(2017):7302965.
CrossRef - Chen F, Xiong H, Wang J, Ding X, Shu G, Mei Z. Antidiabetic effect of total flavonoids from Sanguis draxonis in type 2 diabetic rats. J 2013;149(3):729-36.
CrossRef - Juru dosil. State Publishing House. 1991;13-16.
- Yuteg Youndongombo. The for medical tantra. State Publishing House. 1991;14-18.
- Zhao D, Chen X, Wang R, Pang H, Wang J, Liu L. Determining the chemical profile of Caragana jubata (Pall.) Poir. by UPLC-QTOF-MS analysis and evaluating its anti-ischemic stroke effects. J Ethnopharmacol. 2023; 12(309):116275.
CrossRef - Zhao P, Zhong S, Liao J, Tao J, Yao Y, Song P, Yang X. Caragana jubata ethanol extract ameliorates the symptoms of STZ-HFD-induced T2DM mice by PKC/GLUT4 pathway. J Ethnopharmacol. 2025; (13)339:119171.
CrossRef - He CR, Guo LN, Zhang Y, Shen D, Yang XD. [Screening of active fractions with antithrombotic effect from Caragana jubata]. Zhongguo Zhong Yao Za Zhi. 2016;41(13):2473-2480.
- Puteri MU, Afifah N, Mathriul AQ, Wicaksono FM, Sugiarti MT, Izzatinisa R, Kato M, Saputri FC. Antiplatelet Effects of a Combination of Sappan Wood (Caesalpinia sappan) and Red Ginger (Zingiber officinale var. Rubrum) Extracts in a High-Fat Diet-Induced Rat Model. Adv Pharmacol Pharm Sci. 2024;(20):5543717.
CrossRef - Lee MJ, Lee HS, Jung HJ, Lee CS, Kim JE, Moon HI, Park WH. Caesalpinia sappan L. ameliorates hypercholesterolemia in C57BL/6 mice and suppresses inflammatory responses in human umbilical vein endothelial cells (HUVECs) by antioxidant mechanism. Immunopharmacol Immunotoxicol. 2010;32(4):671-9.
CrossRef - Adnan M, Jeon BB, Chowdhury MHU, Oh KK, Das T, Chy MNU, Cho DH. Network Pharmacology Study to Reveal the Potentiality of a Methanol Extract of Caesalpinia sappan Wood against Type-2 Diabetes Mellitus. Life (Basel). 2022;12(2):277.
CrossRef - Mao QQ, Xu XY, Cao SY, Gan RY, Corke H, Beta T, Li HB. Bioactive Compounds and Bioactivities of Ginger (Zingiber officinaleRoscoe). 2019;8(6):185.
CrossRef - Liu Y, Liu J, Zhang Y. Research Progress on Chemical Constituents of Zingiber officinale Biomed Res Int. 2019;20(2019):5370823.
CrossRef - Rahojo S.J, Sari D.R.T, Wijayanti E. D. Chemical Compostion and Bioacttive Constituents of Standardized Ginger (Zingiber officnale Rosc) Simplica. Biomed Pharmacol J 2025; 18(3).
CrossRef - Pundir S, Garg P, Dviwedi A, Ali A, Kapoor VK, Kapoor D, Kulshrestha S, Lal UR, Negi P. Ethnomedicinal uses, phytochemistry and dermatological effects of Hippophae rhamnoides L.: A review. J Ethnopharmacol. 2021;10(266):113434.
CrossRef - Yang X, Liu Y, Tang Z, Song Z, Liu C, Wang C. Total flavonoids of Hippophae rhamnoides L. improves type 2 diabetes symptoms in rats through down-regulating of the DAG/PRKCA/MAPK10/p65/TNF-α signalling pathway. J Ethnopharmacol. 2024; 10(318):116962.
CrossRef - Dupak R, Hrnkova J, Simonova N, Kovac J, Ivanisova E, Kalafova A, Schneidgenova M, Prnova MS, Brindza J, Tokarova K, Capcarova M. The consumption of sea buckthorn (Hippophae rhamnoides L.) effectively alleviates type 2 diabetes symptoms in spontaneous diabetic rats. Res Vet Sci. 2022; 20(152):261-269.
CrossRef - Erdenechimeg Chuluunbaatar, Dejidmaa Buyantogtokh and Dagvatseren Begzsuren .Total Phenolic, Flavonoid and Carotinoid Content and Anticoagulation Activities of Sapparin Tablet. Journal of Pharmaceutical Research International. 2021; 33(44B): 268–278.
CrossRef - Manjusha Choudhary, Neha Aggarwal, Nitesh Choudhary. Effect of aqueous and alcoholic extract of Sesbania sesban (Linn) Merr. root on glycemic control in streptozotocin-induced diabetic mice. Drug Development and Therapeutics. 2014;5(2):116-122
CrossRef - Chinnala, K. M., Elsani, M. M., Nalla, M. K., & Chinnala, K. M. Anti diabetic activity of methanolic extract of Caesalpinia sappan Linn. on alloxan induced diabetes mellitus in rats. J. Exp. Pharmacol. 2015; (5): 65-69.
- Akash MS, Rehman K, Tariq M, Chen S. Zingiber officinale and Type 2 Diabetes Mellitus: Evidence from Experimental Studies. Crit Rev Eukaryot Gene Expr. 2015;25(2):91-112.
CrossRef - Governa P, Baini G, Borgonetti V, Cettolin G, Giachetti D, Magnano AR, Miraldi E, Biagi M. Phytotherapy in the Management of Diabetes: A Review. 2018;23(1):105.
CrossRef







