Manuscript accepted on :26-09-2025
Published online on: 14-10-2025
Plagiarism Check: Yes
Reviewed by: Dr. Mohini Kuchekar
Second Review by: Dr. Fariha Jasin Mansur
Final Approval by: Dr. Prabhishek Singh
Jayshree Dawane1* , Priti Dhande1
, Priti Dhande1 and Bharati Dalal2
and Bharati Dalal2
1Department of Pharmacology, Bharati Vidyapeeth DU Medical College, Pune, Maharashtra, India.
2Department of Microbiology, Bharati Vidyapeeth DU Medical College, Pune, Maharashtra, India.
Corresponding Author E-mail: priti.dhande@bharatividyapeeth.edu
DOI : https://dx.doi.org/10.13005/bpj/3294
Abstract
The study aims to evaluate the haemostatic and in vitro antimicrobial activity of Feracrylum gel Revalum ®, a novel topical formulation designed for wound management. An assessment was done to determine the haemostatic efficacy of the Feracrylum gel through standardized clotting assays, measuring its ability to promote bleeding control in both in-vitro and in-vivo models. The antimicrobial efficacy of the gel was assessed by testing its activity against a range of bacterial strains, encompassing both Gram-positive and Gram-negative bacteria, using the disk diffusion technique. The Feracrylum gel (Revalum) ® demonstrated significant hemostatic properties, with a marked reduction in bleeding time (p<0.05) and clotting time (p<0.001) compared to control treatments. In antimicrobial assays, the gel exhibited moderate antimicrobial activity against Pseudomonas aeruginosa, Escherichia coli and Staphylococcus aureus with 12mm, 10mm and 5mm zone of inhibition respectively. Feracrylum gel (Revalum) ® shows promising potential as an effective hemostatic agent and the gel demonstrates significant antimicrobial activity, indicating its potential for effective use in clinical wound care. Additional research is needed to evaluate its long-term efficacy and safety across various wound management scenarios.
Keywords
Antimicrobial; Feracrylum; Haemostatic; In-vitro; Wound
Download this article as:| Copy the following to cite this article: Dawane J, Dhande P, Dalal B. Evaluation of Hemostatic and In vitro Antimicrobial Activity of (Revalum®) Feracrylum Gel. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Dawane J, Dhande P, Dalal B. Evaluation of Hemostatic and In vitro Antimicrobial Activity of (Revalum®) Feracrylum Gel. Biomed Pharmacol J 2025;18(4). Available from: https://bit.ly/4oip1ov |
Introduction
Hemostasis is the physiological process that halts bleeding1 and is a critical first step in wound management and surgical interventions. Rapid control of bleeding is not only essential to prevent hemorrhagic shock and minimize blood loss, but also plays a pivotal role in reducing the risk of subsequent infections and promoting optimal wound healing.2 The inability to achieve timely hemostasis can significantly complicate surgical outcomes, increase morbidity, and prolong hospitalization, thereby elevating overall healthcare costs.
To address these challenges, effective topical hemostatic agents have become indispensable tools in both clinical and emergency settings. These agents aid in the rapid formation of stable clots, create physical barriers over wounds and help maintain a clean environment conducive to tissue regeneration.3 In recent years, the need for hemostatic products with dual functionalities like both hemostatic and antimicrobial activity, has intensified, given the high incidence of wound-related infections that complicate the healing process and may lead to chronic wounds, sepsis, or even amputation in severe cases.
Feracrylum, a synthetic active molecule, water-soluble polymer, originally developed for use in surgical and traumatic wound management, has gained attention for its excellent hemostatic efficacy. When formulated as a gel, it acts by forming a rapid, protective barrier over the site of injury, facilitating clot formation and providing mechanical support to the underlying tissue.4 Beyond its hemostatic properties, Feracrylum also exhibits antimicrobial activity, which is highly advantageous in preventing wound colonization by pathogenic microorganisms.5 This antimicrobial feature is critical, considering that wound infections remain a major cause of delayed healing and increased healthcare burden worldwide.6
The newly introduced Feracrylum gel (Revalum®) by Unireva Healthcare represents a modified and improved formulation of the conventional Feracrylum. In this novel approach, the active pharmaceutical ingredient (API) is synthesized through an enhanced process involving activated charcoal and sodium chloride (NaCl) for purification. This method ensures that the final product contains no more than 10.0% unreacted protein content, ensuring greater purity and demonstrates a minimum biological activity of 90.0%, confirming its potent efficacy. Additionally, the pH of a 1.0% w/v aqueous solution of the API ranges between 2.9 and 4.0, optimizing its stability and compatibility with biological tissues. The use of water for injection-grade purified water done during the manufacturing process further underscores the pharmaceutical quality of the preparation.
Given the potential advantages of this improved formulation, the study was undertaken to rigorously assess the hemostatic effectiveness, antimicrobial activity and overall wound healing potential of Feracrylum gel (Revalum®). Through this investigation, we aim to establish its role as an advanced wound care solution capable of offering rapid bleeding control, infection prevention, and enhanced healing outcomes.
Present study undertaken to assess these properties and to establish the potential of Feracrylum gel as a wound care solution.
Materials and Methods
The present study was conducted following the approval of the Institutional Animal Ethics Committee, with permission number BVDUMC/2216/2024/2/19.
In Vitro Antimicrobial Activity
The antimicrobial activity of feracrylum was assessed using the agar diffusion method against standard laboratory strains of Staphylococcus aureus, Escherichia coli and Pseudomonas aeroginosa. The bacterial strains were first introduced into nutrient broth and incubated at 37°C for 3 hours. The turbidity of each culture was then adjusted to the 0.5 McFarland standard to standardize the bacterial concentration.
Using sterile cotton swabs, the bacterial suspension was evenly spread onto Muller Hinton agar plates employing the lawn culture technique. Sterile discs impregnated with 1% Feracrylum gel was gently applied to the surface of the inoculated agar plates, which were then incubated at 37°C for 24 hours. After incubation, the antimicrobial activity was assessed by measuring the diameter of the inhibition zones around the discs. All experiments were conducted in duplicate to ensure consistent and reliable results.
In Vivo Study: Experimental Design
A total of 24 healthy Wistar rats of either sex were used for the study. The rats were housed under standard laboratory conditions, four rats per polypropylene cage with autoclaved rice husk bedding, kept at a constant temperature of 25 ± 3°C, with a relative humidity of 60 ± 5%, and exposed to a 12-hour light/dark cycle. A continuous 10% air exhaust system was in operation within the air-conditioning unit. Rats were provided with a commercially available rodent diet (Pranav Agro Industries) and aqua-guarded drinking water ad libitum. All The experimental procedures were conducted in accordance with the guidelines set by the Committee for the Control and Supervision of Experiments on Animals (CCSEA).
The test material, feracrylum ointment, was kindly provided by Unireva Healthcare.
Evaluation of Hemostatic Potential
Clotting Time Measurement
Rats were anesthetized with an intraperitoneal injection of ketamine. Blood samples were obtained from the retro-orbital plexus and collected into tubes containing 3.2% sodium citrate as an anticoagulant. The collected blood was gently mixed with the anticoagulant by slow inversion to prevent premature clot formation.
Each sample was divided into two aliquots. In the test aliquot, 0.2 ml of 1% feracrylum gel was added to 2 ml of citrated blood, whereas the control aliquot contained only citrated blood. Clotting time was recorded for each sample using a digital stopwatch, noting the interval between the addition of feracrylum and the visible formation of a fibrin clot.
Subsequently, the blood samples were centrifuged at 1500–2000 × g for 15 minutes at 4°C to separate the plasma. The resulting plasma was then used to determine Prothrombin Time (PT) and Activated Partial Thromboplastin Time (aPTT). For PT estimation, thromboplastin reagent was added to the plasma sample, and the time to clot formation at 37°C was recorded. For aPTT assessment, a partial thromboplastin reagent and an activator were added, followed by calcium chloride, and the clotting time was noted. All coagulation assays were performed using standard protocols.
Incision Wound Model for Hemostasis
Prior to surgery, the dorsal surface of each rat was shaved and disinfected using 70% ethanol. Under ketamine anesthesia, a standardized incision approximately 2 cm in length and a 2 mm in deep incision was made on the dorsal region of each rat using a sterile surgical scalpel. The rats were randomly divided into two groups: the control group, which received only the gel base application and the experimental group, which was treated with feracrylum gel.
Immediately after creating the incision, the respective treatments were applied to the wounds. The time taken to achieve complete hemostasis was recorded using a stopwatch from the moment of incision to the cessation of bleeding. After hemostasis was achieved, wounds were sutured, and postoperative care was provided. Animals were closely monitored during recovery for any signs of pain, infection, or wound dehiscence, with appropriate analgesics administered as needed.
Statistical Analysis
Statistical analysis was performed to compare the hemostatic and antimicrobial effectiveness of Feracrylum ointment with the control agent. Various statistical tests (such as ANOVA followed by unpaired t-test) were applied to evaluate the significance of the observed differences. The results are presented as mean ± standard deviation (SD), with p-values indicating statistical significance.
Results
The antimicrobial activity of 1% Feracrylum gel was evaluated against Staphylococcus aureus, Escherichia coli and Pseudomonas aeruginosa using the agar diffusion technique are as below:
Table 1: Effect of Feracrylum gel (Revalum®) on various microorganisms in Disk Diffusion
| Microorganism | Standard Control Zone of Inhibition (mm) | Feracrylum Gel (1%) Zone of Inhibition (mm) | Interpretation |
| Staphylococcus aureus | 20 | 5 | Intermediate |
| Escherichia coli | 22 | 10 | Intermediate |
| Pseudomonas aeruginosa | 19 | 12 | Intermediate |
The disk diffusion assay revealed that the standard drug produced a statistically significantly larger zone of inhibition against Staphylococcus aureus (p < 0.001), Escherichia coli, and Pseudomonas aeruginosa in comparison with Feracrylum. Zone of inhibition around Feracrylum disk was maximum for Pseudomonas aeruginosa. All tests were performed in duplicate, and results were consistent across replicates. No bacterial growth was observed within the zones of inhibition in standard treatment group. (Fig. 1).
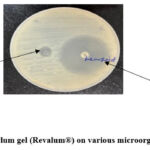 |
Figure 1: Effect of Feracrylum gel (Revalum®) on various microorganisms in Disk Diffusion. |
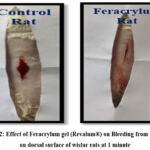 |
Figure 2: Effect of Feracrylum gel (Revalum®) on Bleeding from incision on dorsal surface of wistar rats at 1 minute |
The haemostatic efficacy of feracrylum gel was assessed by measuring the time required to achieve complete cessation of bleeding following a standardized incision (Fig. 2) on the dorsal surface of rats.
Table 2: Effect of Feracrylum gel (Revalum®) on Bleeding Time, Clotting time, Prothrombin time and activated partial thromboplastin time
| Groups (n=6) | Bleeding Time (minutes) | Clotting Time (minutes) | PT (sec) | aPTT (sec) |
| Control | 3.88 ± 0.91 | 5.23± 0.67 | 14.25± 0.90 | 27.19± 0.99 |
| Feracrylum Gel | 1.5± 0.36 *** | 2.30± 0.56 *** | 9.76± 0.37 *** | 22.53± 1.04 *** |
PT: Prothrombin time, aPTT: activated partial thromboplastin time
*** p<0.001 as compared to control group using unpaired t-test
The application of feracrylum gel led to a notable reduction in bleeding time compared to the control group (p < 0.05), indicating enhanced hemostatic activity (Table 2). No adverse reactions, wound infections or complications were observed during the postoperative recovery period in either group. All animals tolerated the procedure well with proper analgesic support.
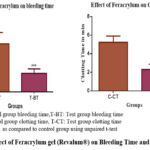 |
Figure 3: Effect of Feracrylum gel (Revalum®) on Bleeding Time and Clotting time |
Statistical analysis revealed that the reduction in clotting time with feracrylum treatment (Fig. 3) was highly significant (p < 0.001). No clotting abnormalities, hemolysis, or other adverse reactions were observed in either group during the study.
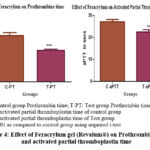 |
Figure 4: Effect of Feracrylum gel (Revalum®) on Prothrombin time and activated partial thromboplastin time |
The effect of feracrylum gel on the coagulation cascade was assessed by measuring Prothrombin Time (PT) and Activated Partial Thromboplastin Time (aPTT) in plasma samples. The reduction in both PT and aPTT values (Fig. 4) in the feracrylum-treated samples was found to be statistically significant (p < 0.001). No abnormalities such as hypercoagulability or clotting defects were observed during the study.
Discussion
Study evaluates the in vitro antibacterial activity, haemostatic efficacy and impact on coagulation parameters of 1% Feracrylum gel (Revalum®). The findings demonstrate that Feracrylum possesses moderate antimicrobial activity and significantly enhances haemostasis both at the site of incision wounds and in blood coagulation assays.
In the antibacterial assay, Feracrylum gel (Revalum®) produced measurable zones of inhibition against all three organisms, although the zones were smaller compared to those produced by standard antibiotics. (Table 1 & Figure 1) These results are consistent with previous studies, such as those conducted by Bhagwat AM et al.,7 who reported that Feracrylum exhibited bacteriostatic effects against Gram-positive and Gram-negative organisms but with lower potency compared to standard antibiotics. Shenoy et al.8 documented a rare post-tonsillectomy oronasopharyngeal stricture linked to secondary infection, where Feracrylum could have played a preventive role by reducing microbial colonization. The intermediate response observed in the present study indicates that while Feracrylum gel has antimicrobial activity, its primary role in wound management may be more attributable to its haemostatic and barrier-forming capabilities rather than direct bactericidal activity. This antibacterial activity may be attributed to the gel’s modified chemical composition and its interaction with bacterial cell wall or cellular processes.9 This intermediate antibacterial activity could be due to lower concentration of Feracrylum gel (1%) and further studies with higher concentration of Feracrylum (2% – 5%) are needed to understand the level of antibacterial activity with such increased concentrations.
The incision wound model provided clear evidence of Feracrylum’s (Revalum®) haemostatic potential. (Figure 2) The mean time to achieve complete cessation of bleeding was significantly shorter in the Feracrylum-treated group compared to control. The results demonstrated that Feracrylum gel (Revalum®) effectively promoted hemostasis as evidenced by a statistically significant reduction (Table 2 & Figure 3) in bleeding time and blood loss compared to control group. Jain et al. highlighted the need for safe agents in dental procedures for anticoagulated patients. Feracrylum, with its bacteriostatic ferric ions, meets this need by offering both infection control and hemostasis by topical application, making it ideal for use in high-risk patients and compromised surgical fields.10 Mahardawi et al. reviewed various hemostatic agents used post-tooth extraction, noting the common use of gelatin sponges and oxidized cellulose. While Feracrylum was not directly assessed, it offers potential advantages such as faster onset, easy application, and biocompatibility.11 The gel’s hemostatic properties can be attributed to its ability to enhance the clotting process by interacting with blood components, facilitating rapid clot formation, and stabilizing the clot.1,2 These results are consistent with previous studies highlighting Feracrylum’s efficacy in other formulations. 12,13,14 The observed hemostatic benefits suggest that Feracrylum gel could be a valuable asset in surgical settings and trauma care, where rapid control of bleeding is crucial.15 Tsamarah et al. showed that 4% Feracrylum is more effective than 1% in achieving rapid hemostasis following gingival incisions, confirming its dose-dependent efficacy.16 The gel forms a mechanical barrier that promotes clot formation without impeding healing. Madhu et al. evaluated hemocoagulase, but Feracrylum holds superiority in terms of chemical stability, low immunogenicity, and added antibacterial effects.17
These findings are consistent with earlier research by Chauhan et al.,18 who demonstrated that Feracrylum promoted rapid haemostasis when applied topically to surgical wounds, attributed its ability to concentrate clotting factors at the wound site and form a mechanical barrier that stabilizes the clot.
In addition to promoting external haemostasis, Feracrylum (Revalum®) was found to significantly reduce clotting time, PT and aPTT in vitro (Figure 4). This suggests that Feracrylum (Revalum®) not only facilitates localized clot formation but may also enhance intrinsic and extrinsic coagulation pathways. Similar observations were made by Mingarro-de-León A et al.19 who noted a reduction in PT and aPTT values upon exposure to Feracrylum, proposing that the polymeric network of Feracrylum might interact with plasma proteins, promoting thrombin generation and accelerating fibrin formation. Girotra et al.20 and Jian et al.21 emphasized the importance of local hemostatics in patients on antiplatelet or anticoagulant therapy, suggesting that Feracrylum could serve as a non-invasive first-line option due to its topical action and lack of systemic absorption.
Importantly, no adverse effects such as hypercoagulability, thrombosis, or tissue irritation were noted during the study, aligning with previous safety evaluations (Laddha et al.),22 which concluded that Feracrylum was well tolerated in both clinical and experimental settings. The modified preparation used in the current study was also devoid of adverse effects like local irritation and allergic reactions. Collectively, the present findings suggest that Feracrylum gel (Revalum®) can serve as a valuable adjunct in surgical and wound care, especially in scenarios requiring rapid bleeding control and infection prevention. However, its moderate antibacterial action indicates that it should not be considered a substitute for systemic antibiotic therapy when infection risk is high.
Further studies are required to explore potential synergistic effects when Feracrylum is used alongside antimicrobial agents or growth-promoting wound dressings. The dual functionality of Feracrylum gel makes it a promising candidate for application in clinical settings where both bleeding control and infection prevention are required.17 In wound care, for instance, the gel could be used to manage bleeding while simultaneously mitigating the risk of infection, improving patient outcomes.
Conclusion
Feracrylum gel (Revalum®) has shown promising results as a strong hemostatic agent with moderate antibacterial activity. In surgical procedures, its application could simplify the management of bleeding and reduce the need for additional antibacterial treatments.
Acknowledgement-
We sincerely thank Unireva Healthcare for providing the Feracrylum gel (Revalum®), seed money and essential technical support. We appreciate the efforts of the animal house staff in ensuring the success of this research.
Funding Sources
The study was financially supported by Unireva Healthcare LLP, Moshi, Pune-412105, India, with grant number Uni/72629273/2024/01.
Conflict of Interest
The author(s) do not have any conflict of interest
Data Availability
This statement does not apply to this article
Ethics Statement
For the animal study, ethics approval was taken from the Institutional Animal Ethics Committee, with permission number BVDUMC/2216/2024/2/19.
Informed consent statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not applicable
Authors’ contribution-
- Jayshree Dawane: Conceptualization, Methodology, Funding Acquisition, Writing the original draft.
- Priti Dhande: Funding Acquisition, Reviewing & Editing the final draft.
- Bharati Dalal: In-vitro experiment and writing the original draft.
References
- Hickman DA, Pawlowski CL, Sekhon UD, Marks J, Gupta AS. Biomaterials and advanced technologies for hemostatic management of bleeding. Adv Mater. 2018;30(4):1700859.
CrossRef - Guo B, Dong R, Liang Y, Li M. Haemostatic materials for wound healing applications. Nat Rev Chem. 2021;5(11):773–91.
CrossRef - Hassanin MZ, Abdallah RA, Ragab SM. Topical blood stopper agents during adenoid surgery in young children: a prospective randomized controlled trial. Eur Arch Otorhinolaryngol. 2018;275(5):1157–63.
CrossRef - Valse D, Hosalli Kumaraswamy N. To evaluate the role of Feracrylum (1%) as hemostatic agent in Tonsillectomy. Indian J Otolaryngol Head Neck Surg. 2021;73(2):240–5.
CrossRef - Sen CK. Human wounds and its burden: an updated compendium of estimates. Adv Wound Care (New Rochelle). 2019;8(2):39–48.
CrossRef - Jacobsen S. Topical wound treatments and wound-care products. In: Equine wound management. 3rd ed. Wiley Blackwell; 2016. p.75–103.
CrossRef - Bhagwat AM, Save S, Burli S, Karki SG. A study to evaluate the antimicrobial activity of Feracrylum and its comparison with povidone-iodine. Indian J Pathol Microbiol. 2001;44(4):431–3.
- Shenoy VS, Samanth R, Parvathareddy N, Apoorva KV. Pediatric Oronasopharyngeal Stricture–A Rare Surgical Complication of Adeno-Tonsillectomy Abstract. Indian Journal of Otolaryngology and Head & Neck Surgery. 2023 Sep;75(3):2352-4.
CrossRef - Rao AM, Patel R. Drug evaluation: Feracrylum 1% gel in the local management of wound. Indian Med Gaz. 2004;84:518–21.
- Jain P, Jain AK, Patidar M, Banthia R, Batham PR. Dental considerations in patients on antiplatelet and anticoagulant drugs–clearing the fog: a case series. Journal of Datta Meghe Institute of Medical Sciences University. 2023 Jan 1;18(1):114-9.
CrossRef - Mahardawi B, Jiaranuchart S, Rochanavibhata S, Arunjaroensuk S, Mattheos N, Pimkhaokham A. The role of hemostatic agents after tooth extractions: A systematic review and meta-analysis. The Journal of the American Dental Association. 2023 Aug 1;154(8):742-52.
CrossRef - Lahoti BK, Aggarwal G, Diwaker A, Sharma SS, Laddha A. Hemostasis during hypospadias surgery via topical application of feracrylum citrate: a randomized prospective study. J Indian Assoc Pediatr Surg. 2010;15(3):87–9.
CrossRef - Ebrahimi F, Mahmoudi J, Torbati M, Karimi P, Valizadeh H. Hemostatic activity of aqueous extract of Myrtus communis L. leaf in topical formulation: in vivo and in vitro evaluations. J Ethnopharmacol. 2020;249:112398.
CrossRef - Janardhan K, Parikh H. Feracrylum—An Unexplored Aspect of Local Drug Delivery in Treating Chronic Periodontitis: An Original Research. Journal of Pharmacy and Bioallied Sciences. 2024 Jul 1;16(Suppl 3):S2655-7.
CrossRef - Rai S, Rattan V. Efficacy of feracrylum as a topical hemostatic agent in therapeutically anticoagulated patients undergoing dental extraction: a comparative study. J Maxillofac Oral Surg. 2019;18(4):579–583.
CrossRef - Tsamarah A, Rahman EF, Praptiningsih RS. Comparison of The Effectiveness of Feracrylum 1% And Feracrylum 4% In Stoping Blooding After Gingiva Incision. Jurnal Medali. 2022;4(2):163-9.
CrossRef - Madhu CP, Naik D, Vishak Nair TU. Effect of topical hemocoagulase therapy in wound healing. Int J Surg Sci. 2019 Oct 1;3:151-6.
CrossRef - Chauhan MK, Kumari M. Feracrylum: An effective and safe topical haemostatic agent. World J Pharm Res. 2017;6(10):319–25.
CrossRef - Mingarro-de-León A, Chaveli-López B, Gavaldá-Esteve C. Dental management of patients receiving anticoagulant and/or antiplatelet treatment. J Clin Exp Dent. 2014;6(2):e155.
CrossRef - Girotra C, Padhye M, Mandlik G, et al. Assessment of the risk of haemorrhage and its control following minor oral surgical procedures in patients on anti-platelet therapy: a prospective study. International journal of oral and maxillofacial surgery. 2014 Jan 1;43(1):99-106.
CrossRef - Jian P, Yang X, Jihong Z, et al. Experts’ consensus on perioperative management of tooth extractions in patients receiving oral antithrombotic treatment. West China Journal of Stomatology. 2022 Jun 1;40(3).
- Laddha AK, Mulla M, Sharma SS, Lahoti BK, Mathur R. A prospective comparison of topical feracrylum citrate versus adrenaline as haemostatic agent in hypospadias surgery in children. Afr J Paediatr Surg. 2014;11(3):215–8.
CrossRef







