Manuscript accepted on :22-10-2025
Published online on: 20-11-2025
Plagiarism Check: Yes
Reviewed by: Dr. Huzef U
Second Review by: Dr. Zena Sideeq Tawfeek
Final Approval by: Dr. Prabhishek Singh
Ramaiyan Velmurugan1 , Yokesh Shanmugam1
, Yokesh Shanmugam1 , Patibandla Jahnavi2
, Patibandla Jahnavi2 , Rajeshwar Vodeti3
, Rajeshwar Vodeti3 , Elias Joel Mart4
, Elias Joel Mart4 , Konatham Teja Kumar Reddy5
, Konatham Teja Kumar Reddy5 , Balaji Pandiyan6
, Balaji Pandiyan6 , Soniya Rani7
, Soniya Rani7 , Prem Shankar Gupta8
, Prem Shankar Gupta8 , Lokeshvar Ravikumar1*
, Lokeshvar Ravikumar1* and Rahul Ambati1
and Rahul Ambati1
1Department of Pharmacology, Saveetha College of Pharmacy, Saveetha Institute of Medical and Technical Sciences, Chennai, Tamil Nadu, India.
2Wishmen Lifesciences Pvt Ltd, Banjara Hills, Hyderabad, Khairatabad, Telangana, India
3Department of Pharmaceutics, School of Pharmacy, Anurag University, Hyderabad, Telangana, India
4Department of Pharmacology, Vels institute of science, Technology and Advanced studies (VISTAS), PV Vaithiyalingam Rd, Velan Nagar, Krishnapuram, Pallavaram, Chennai, Tamil Nadu, India
5Department of Pharmaceutical Analysis, Malla Reddy Institute of Pharmaceutical Sciences, Malla Reddy Vishwavidyapeeth (Deemed to be University), Secunderabad, Telangana, India.
6Department of Pharmacology, School of Pharmaceutical Sciences, Vels Institute of Science, Technology and Advanced Studies, Pallavaram, Chennai, India.
7Department of Pharmacology, GITAM School of Pharmacy, GITAM (Deemed to be University), campus Hyderabad, Telangana, India
8Department of Pharmaceutics, Teerthankar Mahaveer College of Pharmacy, Teerthankar Mahaveer University, Moradabad (U.P), India.
Corresponding Author E-mail:lokeshvarr.scop@saveetha.com
DOI : https://dx.doi.org/10.13005/bpj/3276
Abstract
Liver cancer, particularly hepatocellular carcinoma (HCC), has a poor prognosis and a high fatality rate, making it a serious global health issue that is primarily caused by late-stage finding. Despite the widespread use of alpha-fetoprotein (AFP) and other conventional biomarkers, their low specificity and sensitivity, especially in the initial stages of disease, highlight the urgent need for improved diagnostic methods. In recent years, the development of novel protein (e.g., glypican-3, des-gamma-carboxy prothrombin) and genetic (e.g., microRNAs, circulating tumor DNA, long non-coding RNAs) biomarkers has significantly enhanced the prospects for HCC surveillance and early detection. Because of their great sensitivity, quick reaction time, and potential for point-of-care use, electrochemical biosensors have demonstrated remarkable promise among developing technologies. Significant advances in detection limits, frequently approaching femtogram levels and dynamic ranges, have been made possible by developments in nanomaterials, surface modification methods, and biorecognition components like aptamers and molecularly imprinted polymers. These innovations have enabled multiplexed and label-free sensing approaches that integrate with microfluidic platforms for more efficient, cost-effective diagnostics. Despite considerable progress, challenges such as reproducibility in complex biological matrices, standardization, and clinical validation remain. Future directions include the incorporation of artificial intelligence for signal processing, development of intelligent materials, and scalable fabrication methods for clinical translation. Overall, electrochemical biosensing represents a transformative approach in liver cancer diagnostics, providing fresh approaches to better patient outcomes, individualized care, and early identification.
Keywords
Alpha-fetoprotein (AFP); Electrochemical biosensors; Hepatocellular carcinoma (HCC); Liver cancer biomarkers; Nanomaterials
Download this article as:| Copy the following to cite this article: Velmurugan R, Shanmugam Y, Jahnavi P, Vodeti R, Mart E. J, Reddy K. T. K, Pandiyan B, Rani S, Gupta P. S, Ravikumar L. Ambati R. Electrochemical Biosensors in Hepatocellular Carcinoma: Diagnostic Advances and Pharmacological Perspectives. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Velmurugan R, Shanmugam Y, Jahnavi P, Vodeti R, Mart E. J, Reddy K. T. K, Pandiyan B, Rani S, Gupta P. S, Ravikumar L. Ambati R. Electrochemical Biosensors in Hepatocellular Carcinoma: Diagnostic Advances and Pharmacological Perspectives. Biomed Pharmacol J 2025;18(4). Available from: https://bit.ly/3K7BGw9 |
Introduction
About 90% of liver cancer cases are hepatocellular carcinoma, making it among the most prevalent and deadly cancers in the world .1,2 Chronic viral hepatitis infections, alcohol abuse, and the growing prevalence of non-alcoholic fatty liver disease are among the factors leading to the global rise in liver cancer.3,4 Due mostly to the disease’s late discovery, when there are few curative alternatives, the prognosis for cancer of the liver is still poor despite major advancements in therapeutic techniques.5 This case emphasizes the need for screening techniques; the fact may identify carcinoma of the liver when it becomes more treatable. In the endeavor to enhance liver disease examinations, biometrics are emerging as essential tools for prompt identification, future outcomes, and monitoring of reaction to therapy.6 Objective, visible indicators of biological states or activities are known as biomarkers.7,8 Antibody particles, genetic substances, or metabolites produced by cells of tumors or our bodies in reaction to illness can all be diagnostic for liver tumors.9,10 Numerous exciting potentials have emerged in recent years as a result of extensive study into the validation and identification of sensitive and specific indications for tumors of the liver. The identification of relevant biomarkers for liver cancer might transform the field’s approach to diagnosis and treatment. Because they enable early diagnosis and monitoring, biomarkers have the potential to enhance patient outcomes and lower death rates. To enhance our understanding and management of this devastating condition, further research is needed to identify potential biomarkers and use them in clinical settings.
For a long time, the most widely used biomarker for hepatocellular carcinoma has been alpha-fetoprotein (AFP). However, the search for new biomarkers that might increase diagnostic accuracy has been fuelled by its low sensitivity and specificity, especially for early-stage malignancies. Earlier investigations have identified additional amino acid indicators, including glypican-3 (GPC-3), des-gamma-carboxy prothrombin (DCP).11-13 Furthermore, circulating tumor DNA and microRNAs are examples of genetic markers that may increase the accuracy of diagnosis.14 There are new opportunities to develop more effective detection methods in the rapidly evolving field of liver cancer biomarkers.15,16 Amongst the many analytical techniques available for biomarker identification, electrical approaches have garnered plenty of interest due to their unique benefits.17,18 Strong sensibility, quick response instances, and the capacity to be compacted and incorporated into lightweight devices are some advantages of bio sensor electrodes. Because of these characteristics, they are especially well-suited for point-of-care diagnostics, which is crucial in environments with little money and restricted utilization of contemporary laboratory technology.19,20 The development of the electrolytic biosensors for liver cancer biomarkers holds potential for improving patient outcomes by revolutionizing early identification and surveillance.21,22
In contexts with limited resources, the inclusion of electrochemical biosensors into clinical practice may provide a reliable, effective, and cost-efficient method for detecting liver cancer. Researchers and medical professionals may attempt to develop more effective liver cancer detection tools by using the capabilities of electrochemical biosensors.23,24 A schematic overview of the basic components and working principle of an electrochemical biosensor is presented in Figure 1, illustrating the key elements involved in signal generation and transduction.
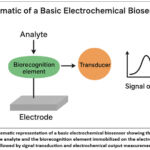 |
Figure 1: Schematic representation of a basic electrochemical biosensor showing the interaction between the analyte and the biorecognition element immobilized on the electrode surface, followed by signal transduction and electrochemical output measurement. |
The observation of electricity generated by redox processes or modifications in electrical characteristics at the electrode-solution contact is the foundation of electrostatic detection techniques.25 Accurate quantitative analysis is made possible by the direct correlation between these signals and the concentration of certain biomarkers.26,27 Many indicators, ranging from tiny particles to big enzymes and even genes, can be found thanks to the adaptability of electrochemical approaches.28 The last few years have seen a major advancement in the development of electrochemical biosensors for liver cancer metrics, which hold promises for early detection and surveillance.29,30 The detection sensitivity and specificity of electrochemical biosensors have significantly increased due to developments in the composition of electrodes, surface-modifying strategies, and amplifier technologies.31 Nanomaterials have greatly enhanced the efficacy of these biosensors by providing bigger surface areas for the immobilization of biorecognition components and permitting faster electron transfer kinetics.32 Additionally, multiplexing signature testing and treatment of samples on only one system have been made possible by the combination of electrochemical detection with microfluidic methods, opening the door for deeper and efficient detecting biomarker.33,34 This technology integration has the potential to significantly improve liver cancer diagnoses and advance electrochemical biosensing.
Even with great advancements, turning galvanic sensing concepts in the research setting into useful commercial tests is still quite difficult.35,36 Performance, stability, and repeatability in intricate biological matrices are crucial factors to take into account. Significant clinical research is needed to show how beneficial these devices are in actual situations.37,38 The latest developments in electrochemical detection techniques for tumor-related physiological indicators are thoroughly described in this article. We go over the state of biomarkers today, the fundamentals of electrochemical biosensing, and the latest developments in signal amplification and sensor design. The investigation also looks at scientific outcomes, application in healthcare advancements, and the difficulties in using these developments at the site.
This work provides a unique contribution by comprehensively evaluating electrochemical sensing modalities, such as label-free technologies and microfluidic integration, with a focus on clinical translation potential. By integrating studies on established protein biomarkers (AFP, DCP,) and new metrics of genetics (lncRNAs), it provides a comprehensive viewpoint on liver cancer diagnosis. Along with discussing practical answers and difficulties in converting these technologies, the report also discusses matrix effects, standardization, and production scalability.39
Materials and Methods
The present review analyses in a thorough manner peer-reviewed literature that focuses on applying electrochemical biosensors to detect and diagnose hepatocellular carcinoma (HCC). Relevant studies were retrieved from scientific databases which include PubMed, Scopus, Web of Science, and Google Scholar because keywords like “HCC biomarkers,” “electrochemical biosensors,” “alpha-fetoprotein,” “Glypican-3,” “nanomaterials in biosensing,” “miRNA detection,” and “point-of-care diagnostics” were used for this purpose. We considered English articles released from 2015 to 2025 using inclusion criteria for original research using electrochemical sensor technologies measuring liver cancer biomarkers such as AFP, DCP, GPC-3, miRNAs, circulating tumor DNA (ctDNA), and long non-coding RNAs (lncRNAs). Studies that were unrelated to biosensing or to liver cancer as well as non-electrochemical methods were excluded. Also excluded from that list were all of those lacking in experimental validation. The selected literature was critically reviewed along with being synthesized into thematic categories that included biomarker discovery, sensor design using nanomaterials, biorecognition strategies, signal amplification mechanisms, and clinical implementation. This systematic, structured, updated overview allowed recent advancements, current challenges, as well as translational prospects of electrochemical biosensors in liver cancer diagnostics.
Biomarkers for liver cancer
The necessity for prompt detection and superior results for patients has made the discovery of useful liver disease genes a top scientific goal in the last decade. Molecular markers known as biomarkers can be used to detect biological processes, pathogenic diseases, or treatment results. Diagnostic criteria are crucial for the tumors detection, evaluation, prediction, and treatment effectiveness tracking, particularly for hepatic cell carcinoma (HCC). Because of their proven methods for identification and relative abundance in physiological hydration, amino acid biomarkers continue to play a significant role in establishing the presence of disease of the liver. Alpha-fetoprotein (AFP), a glycoprotein produced throughout embryonic growth, has been utilized as the preferred method for detecting HCC (see Fig. 2). Even while high blood AFP levels are linked to HCC, their usefulness is constrained by their poor sensitivity and specificity, especially in the early stages of the illness.40 AFP is nevertheless often utilized in clinical practice despite these drawbacks, usually in conjunction with imaging modalities for the monitoring of HCC in high-risk individuals. To get around AFP’s drawbacks, researchers have created a number of other biological indicators of proteins with encouraging clinical value. A fucosylated form of AFP labelled AFP-L3 has shown more specificity for HCC than complete AFP.41 HCC can be distinguished from benign liver illnesses using the percentage ratio of AFP-L3 to total AFP. Des-gamma-carboxy the prothrombin (DCP), sometimes referred to as protein induced by vitamin K absence or antagonist-II (PIVKA-II), is another extensively researched protein biomarker for HCC.42 DCP, an aberrant form of this protein generated by malignant hepatocytes, has demonstrated more thoroughly analytical utility whether paired with AFP.
 |
Figure 2: Protein Structure of AFP106 |
Higher diagnosis accuracy and a better knowledge of liver cancer may arise from the use of many biomarkers, including AFP, AFP-L3, and DCP. More research is needed to demonstrate the therapeutic use of these biomarkers and to examine how they may affect treatment decisions and improve patient outcomes.
Glypican-3 is a hepatocellular carcinoma (HCC) biomarker that is incredibly selective. While it is rarely observed in normal liver or benign liver lesions, this cell-surface heparan sulfate proteoglycan is significantly overexpressed in HCC tissues.43 GPC-3, which promotes tumor growth and metastasis, offers potential as both a therapeutic target and a diagnostic marker (structure shown in Fig. 3). Because GPC-3 is overexpressed in HCC tissues, it is a promising target for developing novel diagnostic and therapeutic techniques. The use of other protein biomarkers, such as GPC-3, in detecting and understanding liver cancer is being investigated. These include midkine, osteopontin, and Golgi protein 73, each of which offers a unique viewpoint on the molecular pathways that drive hepatocarcinogenesis.44 These biomarkers might provide valuable insights into the underlying mechanisms that influence the development of liver cancer, perhaps resulting in the creation of better therapeutic and diagnostic alternatives.45 For people with liver cancer, the development and validation of biomarkers like GPC-3, among others, may improve therapeutic outcomes and diagnostic accuracy. To completely comprehend the therapeutic importance of these biomarkers and create efficient diagnostic and treatment strategies that specifically target these molecules, more study is required.
 |
Figure 3: Protein Structure of GPC-3106 |
Looking for innovative genetic indicators for tumors in the liver has been made much easier by the development of high-throughput genomic and proteomic tools.46 Because of their outstanding durability in circulatory and crucial function in regulating the activity of genes, scholars are becoming interested in microRNAs (miRNAs) as possible sensors. c. Certain miRNAs, such as miR-223 , and miR-122,47,48 were found to be irregular in hepatic carcinoma (HCC), and the degrees of their expression have been linked to the disease’s morbidity and advancement. The most well-studied miRNAs that alter a range of cell phenotypes and cause HCC are shown in Figure 4. MiRNA biomarkers have the benefit of offering a more thorough understanding of the underlying molecular changes in cancer cells. Examining the expression levels of certain miRNAs can help researchers learn a lot about the complicated molecular pathways that drive the start and progression of liver cancer.49 This information can help to develop more effective HCC therapy and diagnostic approaches. The use of miRNAs as biomarkers offers immense promise for improving our understanding and treatment of liver cancer. More research is needed to demonstrate the therapeutic use of these biomarkers and to examine how they may affect treatment decisions and improve patient outcomes.
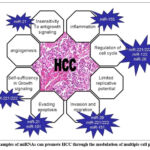 |
Figure 4: Examples of miRNAs can promote HCC through the modulation of multiple cell phenotypes106 |
Circulating tumor DNA (ctDNA) is a promising class of genetic markers with great potential for non-invasive cancer monitoring and detection because DNA fragments released into the bloodstream by dying cancer cells carry tumor-specific genetic and epigenetic alterations.50 Clinicians can use real-time data from ctDNA analysis to monitor the progression of the illness and change treatment approaches.51 chromosomal isolated malignancies of the liver is being found to carry alterations in proteins like CTNNB1, and the TERT promoter, which may result in non-invasive molecular tumor screening and individualized treatment choices. It has also been shown that long non-coding RNAs (lncRNAs) may serve as indicators of liver cancer. With a length of around 200 nucleic acids, these transcriptions of RNA are crucial for regulating genes and cell function, and they impact different facets of cancer development.52 Hepatocellular carcinoma (HCC) has been discovered to have abnormal expression of several lncRNAs, including HULC, MALAT1, and HOTAIR, and their levels are linked to tumor formation and patient outcomes.53 LncRNAs are promising biomarker candidates because of their tissue and cancer-type specificity, which might lead to more precise diagnosis and prognosis. The use of ctDNA and lncRNAs as biomarkers in liver cancer therapy has the potential to totally transform the field, allowing for earlier detection, more accurate treatment, and improved patient outcomes. More research is needed to prove the clinical efficacy of these biomarkers, as well as their potential for guiding therapy decisions and developing novel therapeutic methods.
Techniques for electrochemical biosensing
Electrochemical biosensing is now a viable analytical method for locating liver cancer biomarkers due to its high sensitivity, fast reaction times, and shrinking potential. Seeing electricity produced by reactive processes or modifications in electron density at the surface where the electrode and solution interact is the fundamental concept of electrolytic detecting. The quantity and availability of specific antigens may be directly correlated with these commands, providing the numerical information required for tracking and diagnostics. The interaction between the analyte of interest and the electrode surface is a crucial element of electrophysiological testing.54 This connection is often achieved in the field of liver cancer indicators by biorecognition components such as aptamers or antibodies that are bound on the electrode. As the intended analyte binds onto these recognizing places, the body’s electric characteristics alter. These alterations could show up as variations in flow, possible, the resistance, depending on the measuring method used.
Numerous approaches, including electrochemical impedance spectroscopy, voltammetry, and amperometry, can be employed to precisely quantify changes in electrochemical properties. Researchers can properly identify and track liver cancer by analyzing these changes to determine the presence and amount of certain biomarkers. Electrochemical biosensing’s high sensitivity and specificity make it a promising technique for developing novel liver cancer diagnostic tools.55
The electrode material used to make electrochemical biosensors has a significant impact on their performance. Because of their superior conductivity and electrochemical stability, traditional materials like carbon, gold, and platinum are frequently utilized. Gold electrodes are extensively employed because to their ease of surface modification and biocompatibility.56 One benefit of carbon-based electrodes is their affordability and versatility.57-59 The design of electrodes for biomarker detection has been completely transformed by recent developments in nanomaterials. The kinds of nanotechnology conductors that provide better transmission of electrons dynamics and greater dimension for blocking biorecognition pieces include graphene ,60 carbon nanotubes (CNT)61, and gold nanoparticles (AuNPs).62 A medical device with low thresholds for detection and an extensive linear range for DCP the identification was developed by Fu et al.63 employing AuNPs-functionalized nanotubes with multiple walls and core-shell Pd@PtCu nanostructures.
By boosting sensitivity and surpassing detection limitations, these nanoparticles make it possible to create innovative signal amplification techniques. By taking use of nanomaterials’ unique properties, researchers can create more powerful electrochemical biosensors for biomarker detection.
Surface modification strategies are critical for regulating electrode surface functionalization and optimizing biosensor performance. Self-assembled monolayers (SAMs) provide fine-grained control over the orientation and organization of recognition components, especially SAMs based on thiols on gold surfaces.64 While maintaining biological activity, mixed SAMs can alter the accessibility of biomolecules. In this setting, Giannetto et al.65 used a nanostructured matter metal platform customized with PAMAM dendrimers that were connected by SAMs to create an amperometry immunosensor for AFP measurement (Fig. 5). This method improved responsiveness and increased the conventional sensing area. Some instances of polymer paints that can improve resilience and offer the ideal circumstances for biological molecules entrapment are conductive chemicals like polypyrrole (PPy) and polyaniline (PANI).
 |
Figure 5: Amperometric immunosensor enhanced by PAMAM-dendrimers linked via SAMs for AFP detection106 |
Dendrimer designs are utilized in sophisticated ways to enhance biomolecule adhesion.66 Nanocomposite materials have also been shown to have the ability to provide synergistic effects that improve biosensor performance.67,68 For instance, Liu et al. used multidimensional macroporous the PANI (Fig. 6) as the base material to create a galvanic immunosensor to AFP detection with a modest limit of detection and a wide straight distance.
 |
Figure 6: Fabrication process of the AFP immunosensor based on macroporous PANI106 |
Choosing the right biorecognition ingredients is essential for biosensors with electrodes to achieve excellent accuracy as well as sensitivity. Determinants are utilized extensively due to their excellent specificity and affinity for antigens being targeted.69,70 But making them might be expensive and time-consuming. Aptamer synthetic oligonucleotides have shown tremendous promise as antibody substitutes due to their stability, ease of production, and regeneration potential.71,72 Another kind of synthetic recognition component that is gaining popularity in electrochemical biosensors because to its low cost and great durability is molecularly imprinted polymers (MIPs).73,74 A MIP electrochemical sensor that uses graphene-gold composite and polysulfide cordial to improve signal detection was created by Liu and Liu75 in order to detect AFP. The sensor’s broad linear range and low detection limit revealed high AFP selectivity and excellent separation from interfering compounds. MIPs and aptamers serve as adaptable recognition components for liver cancer indicators, delivering quicker binding kinetics and increased surface coverage.
Some nanomaterials are selected for biosensor development because of their special qualities and capacity to solve analytical problems. For instance, Fu et al. used core-shell Pd@PtCu nano layered on WS2 miniature sheets for DCP discovering utilizing the PtCu shell’s catalytic capacity as well as the lead core’s resistivity. The WS2 nanosheets reduced nanoparticle aggregation while providing a large surface area. In electrode modification, AuNPs and MWCNTs collaborate to enhance their complementary benefits. While AuNPs offer anchoring sites for immobilizing biomolecules and improving electron transport, MWCNTs form a conductive network, increasing the effective surface area. A modest DCP threshold of 71.6 fg/mL was obtained with this mix. Multifaceted macroporous PANI was chosen by Liu et al. because of its elevated conductance, oxidative activity, and numerous beneficial groups for monoclonal interaction. The macroporous creation enhanced susceptibility by optimizing its circumference while preserving efficient weight transportation, resulting in a detection limit of 3.7 fg/mL for AFP. By merging advanced battery materials, sensing techniques, and biorecognition elements, very sensitive and accurate biological sensors for tumor-related symptoms are currently developed. Future studies will concentrate on multi-modal sensing devices that combine electrochemical detection with other sensing methods to detect a broad variety of biomarkers.
Current advancements in liver cancer markers using electrochemical biosensors;
Thanks to developments in material research, nanotechnologies, and biology, the study of bio sensor electrodes for cancer-related antigens has advanced significantly. One potential method for enhancing the identification of biomarker while preserving substantial specificity and sensitivity is the use of label-free galvanic immunity sensors.76 These sensors detect electrical changes on the electrode surface caused by antigen-antibody binding events.77 In one instance, a label-free biological sensor for GPC-3 monitoring was developed by modifying a light-addressable potentiometric sensor (LAPS) with rGO/PEI/AuNPs and a GPC-3 aptamer (Fig. 7).
 |
Figure 7: Scheme of LAPS biosensors modified with rGO/PEI/AuNPs and aptamer for GPC-3 determination106 |
The sensor’s limits for detection is 40.0 ng/mL, while it features a broad reading variety, strong preference, with no adverse effects.78 A alternative technique used immunosensors based on field-effect transistors (FETs), which regulated conductivity by exploiting differences in surface charge distribution after antigen adsorption. A graphene FET (G-FET) biosensor was developed by Kim et al.79 to identify AFP in HCC patients’ plasma. Anti-AFP antibodies were rendered immobile by functionalizing the graphene surface with PBASE (Fig. 8). When assessing AFP in individual blood specimens at levels as minimal as 0.1 ng/mL, the G-FET showed outstanding specificity and discrimination. The G-FET is firmly bound via both AFP & anti-AFP (dissociation constant = 4.64 × 10^-11 M).
 |
Figure 8: Scheme of AFP detection using G-FET and modification process106 |
Electrochemical aptasensors have become more popular for identifying markers of liver cancer since aptamers function better than conventional antibodies. Aptamers’ security, adaptability, and capacity for regrowth make them appropriate for long-lasting and recyclable bio sensors. Li et al.80 created an advantageous electrostatic aptasensor for Granular amino acids 73 (GP73) identification using rGO-ferrocene-polyaniline nanocomposites. The sensor created a single-stranded configuration on the exterior of the electrode by using a GP73 aptamer beads to connect with an additional DNA segment.
Ferrocene’s oxidation current increased due to a competitive binding process that took place when GP73 was present (Fig. 9). The aptasensor demonstrated outstanding resilience and applicability with a narrow threshold for identification of 0.15 pg/mL and a broad proportional measurement bandwidth. The relative errors for the aptasensor and ELISA findings on human blood samples varied from 0.11% to 6.89%. The aptasensor’s performance is impressive, and Table 1 presents the most recent research on techniques based on electrochemistry to find liver cancer biomarkers.
 |
Figure 9: Scheme of GP73 competitive electrochemical aptasensor based on rGO-Fc-PANi nanocomposites106 |
Table 1: Electrochemical Biosensors for Hepatocellular Carcinoma (HCC) Biomarker Detection
| Electrode Material & Sensor Design | Target Biomarker | Linear Detection Range (LDR) | Limit of Detection (LOD) | Tested Sample Type | Reference |
| Metal-organic framework (MOF), carbon nanotubes (CNTs), and gold nanoparticles (GNPs) with primary antibody (Ab1) | Alpha-fetoprotein (AFP) | 1–200 nanograms per milliliter (ng/mL) | 0.3 ng/mL | Human serum (clinical sample) | [91] |
| Glassy carbon electrode (GCE) modified with bovine serum albumin (BSA), silver nanoparticles (AgNPs), secondary antibody (Ab2), and gold nanoparticles (AuNPs) | AFP | 0.1 picograms/mL (pg/mL) – 200 ng/mL | 0.035 pg/mL | Human serum | [88] |
| Screen-printed graphene electrode (SPGE) functionalized with BSA, aptamer (Apt), platinum nanoparticles (PtNPs), and carboxylated graphene oxide (GO-COOH) | AFP | 3–30 ng/mL | 1.22 ng/mL | Human serum | [86] |
| Conducting polymer (polyaniline, PANI), peptide-modified electrode with aptamer (Apt) on screen-printed electrode (SPE) | AFP | 0.1 pg/mL – 100 ng/mL | 0.03 pg/mL | Human serum | [87] |
| Reduced graphene oxide (rGO), hemin, palladium nanoparticles (PdNPs) with dual GPC3-specific aptamers and polydopamine-AuNPs composite | Glypican-3 (GPC3) | 0.001–10.0 ng/mL | 0.13 pg/mL | Human serum | [97] |
| Iron oxide (Fe₃O₄) decorated with carboxylated multi-walled carbon nanotubes (MWCNTs), gold nanoparticles (AuNPs), and anti-AFP antibody | AFP | 1 pg/mL – 10 micrograms per milliliter (μg/mL) | 1.09 pg/mL | Clinical sample | [60] |
| Indium tin oxide (ITO) coated with ordered vertically mesoporous silica film (O-VMSF) and BSA-blocked aptamer | AFP | 0.001–1000 ng/mL | 0.31 pg/mL | Human serum | [84] |
| Nickel hydroxide/carbon nanoboxes (Ni(OH)₂@N-C) with aptamer and BSA-modified screen-printed carbon electrode (SPCE) | AFP | 1 femtogram/mL (fg/mL) – 100 ng/mL | 0.3 fg/mL | Human serum | [89] |
| GCE coated with graphene oxide (GO), gold nanoparticles (AuNPs), anti-AFP antibody, and chitosan (CS) | AFP | 0.1–100 ng/mL | 0.041 ng/mL | Not specified | [83] |
| BSA/aptamer-modified polyethyleneimine-coated gold nanoparticles (PEI-AuNPs) on SPE | AFP | 10–50,000 pg/mL | 9.5 pg/mL | Human serum | [71] |
| GPC3-specific aptamer on reduced graphene oxide (RGO)–Cu₂O–gold nanoparticles-modified SPE | GPC3 | 0.1–500.0 ng/mL | 0.064 ng/mL | Serum from HCC patients | [98] |
| Bismuth vanadate (BiVO₄) and bismuth oxyiodide (BiOI)-MWCNT composite modified with chitosan, glutaraldehyde, and AFP antibodies | AFP | 0.1 pg/mL – 100 ng/mL | 0.014 pg/mL | Human blood | [90] |
| Carboxylated carbon black nanoparticles (CBNPs) on GCE with palladium nanoparticles (PdNPs), anti-AFP antibody, and BSA | AFP | 0.005–1000 ng/mL | 0.0131 ng/mL | Human serum | [81] |
| Silver nanoparticles (AgNPs) linked to anti-AFP antibody via nitrocellulose immunoelectrode (NIE) | AFP | 5 pg/mL – 500 ng/mL | 5 pg/mL | HCC patient serum | [82] |
| Reduced graphene oxide (RGO) with chitosan–ferrocene (CS-Fc) and platinum–palladium bimetallic nanoparticles (Pt-Pd BNPs) | GPC3 | 0.001–10.0 μg/mL | 3.67 ng/mL | Human serum | [96] |
| Gold electrode (AuE) modified with molecularly imprinted polymer (MIP) and gold nanoparticles | AFP | 0.1 pg/mL – 1.0 μg/mL | 0.075 pg/mL | Human serum | [85] |
| RGO–hemin–AuNP hybrid with GPC3 aptamer on SPE | GPC3 | 0.001–10 μg/mL | 2.86 ng/mL | Human serum | [93] |
| SPCE modified with AuNPs and 3-mercaptopropionic acid (MPA), using tyrosine–ferrocene conjugate and aptamer/antibody HRP sandwich | GPC3 | 0.1 ng/mL – 1 μg/mL | 0.087 ng/mL | Human serum | [94] |
| Aptamer-linked hollow gold nanospheres (HGNs) and AgNPs assembled on AuNP-SPCE | GPC3 | 10–100 μg/mL | 3.16 μg/mL | Human serum | [95] |
| Diketopyrrolopyrrole (DCP) sandwiched between Ab1/AuNP@MWCNTs/BSA and Ab2 for DCP sensing | Des-gamma-carboxy prothrombin (DCP) | 100 fg/mL – 100 ng/mL | 71.6 fg/mL | Human serum | [63] |
| Titanium dioxide (TiO₂)-decorated gold nanoparticles (Au@TiO₂) functionalized with SMPSF, cysteine (Cys), and antibody (Ab) | Protein caused by antagonist-II (PIVKA-II) or lack of vitamin K | 1–10,000 pg/mL | 0.77 pg/mL | Fetal and human serum (HCC samples) | [92] |
| AgNPs–hydrogen-reduced graphene oxide (H-rGO)–PdNPs–GPC3 aptamer combined with AuNP@rGO composite | GPC3 | 10–100 μg/mL | 3.30 ng/mL | Human serum | [99] |
Clinical applications and challenges
An essential beginning in enhancing healthcare for patients is the transition of biosensors using electrodes for liver tumor indicators through scientific designs to usage in clinical settings. Much effort has been put into developing very sensitive and specific detection systems, but many problems remain before they can be used successfully in clinical settings. The overall diagnostic workflow using electrochemical biosensors for hepatocellular carcinoma (HCC) detection is illustrated in Figure 10, highlighting the stepwise process from sample collection to clinical interpretation.
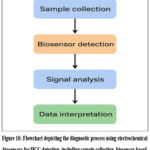 |
Figure 10: Flowchart depicting the diagnostic process using electrochemical biosensors for HCC detection, including sample collection, biosensor-based biomarker recognition, signal analysis, and clinical interpretation leading to treatment decision-making. |
Sensitivity and detection threshold are two analytical performance metrics that have a significant impact on the therapeutic usefulness of electrochemical biosensors. Achieving clinically significant detection limits is crucial, particularly for the early diagnosis of disease. As contrasted to conventional antibody tests, novel electrostatic biosensors for antigens such as AFP and DCP have demonstrated remarkable sensitivity in the picogram to femtogram per millimeter level.89 It is important to remember, too, that clinical samples might not be able to reach the incredibly low detection limits attained in a laboratory setting. The dynamic range of electrochemical biosensors is crucial since the concentrations of liver cancer biomarkers change greatly based on the patient’s features and the disease’s stage. Despite additional measurements or diluting the sample, sensor technology have to be capable to precisely assess the amount of biomarker spans different orders of range. Several recent innovations have created impressive straight static levels ranging from from four to five powers of severity, and the size levels of numerous liver cancer indicators are clinically significant.94 By resolving these issues and enhancing analytical performance, electrochemical biosensors may improve the diagnosis of liver cancer and patient treatment.
Specimen matrix consequences are a major obstacle when translating bio sensor technologies from research laboratories to hospital evaluation of samples. Sensing efficiency can be significantly affected by the complex makeup that constitutes living liquid like plasma, blood, or urination. Target biomarker signals can be obscured and electrochemical characteristics changed by the non-specific adsorption of proteins, lipids, and other macromolecules on sensor surfaces. Sample viscosity, pH, and ionic strength fluctuations can affect electron transfer and biorecognition processes. Scientists looked at the collection of samples and compound outcome reduction strategies for tackling these issues. Inhibition chemicals like BSA or polyethylene glycol (PEG) can be used to reduce unspecific adsorption on sensitive devices.100,101 Magnetic substances covered with specific aptamers or nanoparticles can be used to pre-concentrate and accurately collect specified molecules.102,103 This approach minimizes sample volume while separating the target biomarker from distracting molecules, potentially improving detection sensitivity. The creation of biosensors with electrochemical activity for malignancy diagnostics is being driven by point-of-care (POC) experiments, which enables quick, on-site testing in medical offices, enabling quicker evaluation and curative selection.104 Electrochemical biosensors are now closer to realizing this promise because to recent developments in downsizing, sample preparation step integration, and user-friendly interfaces.105 Researchers can improve the feasibility and accessibility of biosensors that use the field of electrochemistry for the detection of liver cancer by addressing sample matrix effects and developing proof-of-concept devices.
The next steps in electrical methods for searching for malignancy signals appear to have numerous interesting directions. The creation of integrated multiplexed detection techniques that can simultaneously analyze several biomarkers with various molecular properties is one crucial area. This would yield more thorough diagnostic data in addition to requiring creative electrode array designs and signal processing techniques. The advancement of machine learning and artificial intelligence provides new opportunities for increasing the performance of electrochemical biosensors. These computer approaches may allow for more precise pattern discovery in complicated biological data, as well as improved signal processing and noise reduction. Furthermore, machine learning algorithms may be able to detect minute connections between different biomarker data, resulting in a more accurate early diagnosis. Innovations in material science will continue to be important, especially in the areas of hybrid composites and new nanomaterials. The development of intelligent materials that can react to various stimuli or change their characteristics depending on the surroundings should be the main goal of future research. Combining electrochemical detection systems with microfluidic technology is another exciting avenue. This makes it possible for whole sample-to-answer methods to process medicinal data with little assistance from users. This legalization may enable true point-of-care applications, such as target biomarker concentration, separation, and sample processing on-chip.
Conclusion
The results of the study show that either electrochemical detection methods for liver tumor indicators have advanced significantly and could revolutionize early detection and surveillance. Highly potent and selective the devices for important biomarkers including DCP, AFP, and GPC-3 have been developed thanks to the use of nanotechnology, surface enhancement procedures, and amplifiers of signal processes. Detector effectiveness has improved due to the introduction of tiny particles, enabling limitations on detection in the region of femtograms per milliliter. Aptamer-based sensors and label-free techniques offer greater stability and regeneration potential. Quick real-time evaluation in healthcare environments may be made possible by the advancement of point-of-care techniques stability of complex biological matrices, reproducibility and comprehensive confirmation tests are some of the challenges that must be overcome before laboratory prototypes may be used as diagnostic tools in clinical settings. Resolving these problems and developing multiplexed detection systems that can assess numerous biomarkers at once should be the main goals of future research. By facilitating early detection and customized treatment regimens, electrochemical biosensors have the potential to greatly enhance carcinoma of the liver prognosis and outcome for patients as the field advances.
Acknowledgement
The authors express their thanks to Saveetha College of Pharmacy – SIMATS for providing the necessary facilities to carry out this research work.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Author’s Contribution
- Ramaiyan Velmurugan: Conceptualization, Methodology, Writing – Original Draft.
- Yokesh: Formal Analysis, Validation, Writing – Review & Editing
- Patibandla Jahnavi: Data Curation, Investigation
- Visualization. Rajeshwar Vodeti: Software, Validation, Data Analysis.
- Joel Mart: Literature Review, Data Interpretation.
- Konatham Teja Kumar Reddy: Methodology, Formal Analysis.
- Balaji: Writing – Review & Editing, Visualization.
- Soniya Rani: Investigation, Literature Search.
- Prem Shankar Gupta: Funding Acquisition, Resources, Supervision.
- Lokeshvar Ravikumar: Supervision, Project Administration
- Rahul Ambati: Writing – Review & Editing
References
- Huang L, Chen J, Yu Z, Tang D. Self-powered temperature sensor with seebeck effect transduction for photothermal–thermoelectric coupled immunoassay. Analytical chemistry. 2020 Jan 15;92(3):2809-14.
CrossRef - Ren R, Cai G, Yu Z, Zeng Y, Tang D. Metal-polydopamine framework: an innovative signal-generation tag for colorimetric immunoassay. Analytical Chemistry. 2018 Aug 23;90(18):11099-105.
CrossRef - Anil BC, Dayananda P, Nethravathi B, Mahesh SR. Efficient local cloud-based solution for liver cancer detection using deep learning. International Journal of Cloud Applications and Computing (IJCAC). 2022 Jan 1;12(1):1-3.
CrossRef - Ahmadian E, Janas D, Eftekhari A, Zare N. Application of carbon nanotubes in sensing/monitoring of pancreas and liver cancer. 2022 Sep 1;302:134826.
CrossRef - Gunasekhar P, Vijayalakshmi S. Optimal biomarker selection using adaptive Social Ski-Driver optimization for liver cancer detection. Biocybernetics and Biomedical Engineering. 2020 Oct 1;40(4):1611-25.
CrossRef - Chauhan D, Chandra R, Kumar S. Hemocompatible functionalized hydrogen substituted graphdiyne based highly durable biosensor for liver cancer detection. ACS Applied Bio Materials. 2023 May 17;6(6):2257-65.
CrossRef - Lv S, Zhang K, Zeng Y, Tang D. Double photosystems-based ‘Z-Scheme’photoelectrochemical sensing mode for ultrasensitive detection of disease biomarker accompanying three-dimensional DNA walker. Analytical Chemistry. 2018 May 18;90(11):7086-93.
CrossRef - Qiu Z, Shu J, Tang D. Near-infrared-to-ultraviolet light-mediated photoelectrochemical aptasensing platform for cancer biomarker based on core–shell NaYF4: Yb, Tm@ TiO2 upconversion microrods. Analytical chemistry. 2018 Jan 2;90(1):1021-8.
CrossRef - Ren Y, He S, Huttad L, Chua MS, So SK, Guo Q, Cheng Z. An NIR-II/MR dual modal nanoprobe for liver cancer imaging. Nanoscale. 2020;12(21):11510-7.
CrossRef - Kim YJ, Jang H, Lee K, Park S, Min SG, Hong C, Park JH, Lee K, Kim J, Hong W, Jung H. PAIP 2019: Liver cancer segmentation challenge. Medical image analysis. 2021 Jan 1;67:101854.
CrossRef - Tzartzeva K, Singal AG. Testing for AFP in combination with ultrasound improves early liver cancer detection. Expert review of gastroenterology & hepatology. 2018 Oct 3;12(10):947-9.
CrossRef - Tayob N, Kanwal F, Alsarraj A, Hernaez R, El-Serag HB. The performance of AFP, AFP-3, DCP as biomarkers for detection of hepatocellular carcinoma (HCC): a phase 3 biomarker study in the United States. Clinical Gastroenterology and Hepatology. 2023 Feb 1;21(2):415-23.
CrossRef - Li D, Mallory T, Satomura S. AFP-L3: a new generation of tumor marker for hepatocellular carcinoma. Clinica chimica acta. 2001 Nov 1;313(1-2):15-9.
CrossRef - Jiang D, Zhang Y, Wang Y, Xu F, Liang J, Wang W. Diagnostic accuracy and prognostic significance of Glypican-3 in hepatocellular carcinoma: a systematic review and meta-analysis. Frontiers in Oncology. 2022 Sep 23;12:1012418.
CrossRef - Deng H, Shang W, Wang K, Guo K, Liu Y, Tian J, Fang C. Targeted-detection and sequential-treatment of small hepatocellular carcinoma in the complex liver environment by GPC-3-targeted nanoparticles. Journal of nanobiotechnology. 2022 Mar 24;20(1):156.
CrossRef - Yao M, Yao DF, Bian YZ, Wu W, Yan XD, Yu DD, Qiu LW, Yang JL, Zhang HJ, Sai WL, Chen J. Values of circulating GPC-3 mRNA and alpha-fetoprotein in detecting patients with hepatocellular carcinoma. Hepatobiliary & Pancreatic Diseases International. 2013 Apr 15;12(2):171-9.
CrossRef - Zeng R, Qiu M, Wan Q, Huang Z, Liu X, Tang D, Knopp D. Smartphone-based electrochemical immunoassay for point-of-care detection of SARS-CoV-2 nucleocapsid protein. Analytical chemistry. 2022 Oct 17;94(43):15155-61.
CrossRef - Lv S, Zhang K, Zhu L, Tang D, Niessner R, Knopp D. H2-based electrochemical biosensor with Pd nanowires@ ZIF-67 molecular sieve bilayered sensing interface for immunoassay. Analytical chemistry. 2019 Aug 22;91(18):12055-62.
CrossRef - Zou Y, Gu H, Yang J, Zeng T, Yang J, Zhang Y. A high sensitivity strategy of nitrite detection based on CoFe@ NC nanocubes modified glassy carbon electrode. Carbon Letters. 2023 Dec;33(7):2075-86.
CrossRef - Yi K, Xu S, Cheng H, Chen S, Jiang S, Tu J. A label-free sensor based on a carbon nanotube-graphene platform for the detection of non-Hodgkin lymphoma genes. Alexandria Engineering Journal. 2023 Dec 1;84:93-9.
CrossRef - Zhu Y, Liu TH, Zhou W, Shi M, Wu M, Shi P, Zhao N, Li X, Zhang Z, Zhang D, Lv Y. An “On‐Site Transformation” strategy for electrochemical formation of TiO2 nanoparticles/Ti3C2Tx Mxene/reduced graphene oxide heterojunction electrode controllably toward ultrasensitive detection of uric acid. Small Structures. 2024 Aug;5(8):2400034.
CrossRef - Su J, Su X. Determination of tartrazine in sports drinks by a disposable electrochemical sensor modified with Co2O3. Journal of Food Measurement and Characterization. 2023 Dec;17(6):5856-63.
CrossRef - Wang X, Wang H, Wan X, Wei Q, Zeng Y, Tang D. Smartphone-based point-of-care photoelectrochemical immunoassay coupling with ascorbic acid-triggered photocurrent-polarity conversion switching. Biosensors and Bioelectronics. 2025 Jan 1;267:116749.
CrossRef - Zeng R, Gong H, Li Y, Li Y, Lin W, Tang D, Knopp D. CRISPR-Cas12a-derived photoelectrochemical biosensor for point-of-care diagnosis of nucleic acid. Analytical chemistry. 2022 May 12;94(20):7442-8.
CrossRef - Tang M, Guo J, Shen Z. Rapid detection of carbendazim residue in tea by machine learning assisted electrochemical sensor. Journal of Food Measurement and Characterization. 2023 Dec;17(6):6363-9.
CrossRef - Xu F, Ai QY, Wang AJ, Mei LP, Song P, Liu W, Feng JJ, Cheang TY. Pronounced signal enhancement with gourd-shaped hollow PtCoNi bunched nanochains for electrochemical immunosensing of alpha-fetoprotein. Sensors and Actuators B: Chemical. 2025 Jan 1;422:136608.
CrossRef - Wang AJ, Zhu XY, Chen Y, Yuan PX, Luo X, Feng JJ. A label-free electrochemical immunosensor based on rhombic dodecahedral Cu3Pt nanoframes with advanced oxygen reduction performance for highly sensitive alpha-fetoprotein detection. Sensors and Actuators B: Chemical. 2019 Jun 1;288:721-7.
CrossRef - Li Z, Liu H. Study on electrochemical properties of lead calcium tin anode for hydrometallurgy. Alexandria Engineering Journal. 2023 Nov 1;82:389-95.
CrossRef - Luo Z, Zhang L, Zeng R, Su L, Tang D. Near-infrared light-excited core–core–shell UCNP@ Au@ CdS upconversion nanospheres for ultrasensitive photoelectrochemical enzyme immunoassay. Analytical chemistry. 2018 Jun 25;90(15):9568-75.
CrossRef - Luo Z, Qi Q, Zhang L, Zeng R, Su L, Tang D. Branched polyethylenimine-modified upconversion nanohybrid-mediated photoelectrochemical immunoassay with synergistic effect of dual-purpose copper ions. Analytical chemistry. 2019 Feb 22;91(6):4149-56.
CrossRef - Liang Y, Xu Y, Tong Y, Chen Y, Chen X, Wu S. Graphene-based electrochemical sensor for detection of hepatocellular carcinoma markers. Frontiers in Chemistry. 2022 Apr 8;10:883627.
CrossRef - Yu Z, Xu Z, Zeng R, Xu M, Zou M, Huang D, Weng Z, Tang D. Tailored metal–organic framework‐based nanozymes for enhanced enzyme‐like catalysis. Angewandte Chemie. 2025 Feb 10;137(7):e202420200.
CrossRef - Zhou Q, Lin Y, Zhang K, Li M, Tang D. Reduced graphene oxide/BiFeO3 nanohybrids-based signal-on photoelectrochemical sensing system for prostate-specific antigen detection coupling with magnetic microfluidic device. Biosensors and Bioelectronics. 2018 Mar 15;101:146-52.
CrossRef - Lin Y, Zhou Q, Li J, Shu J, Qiu Z, Lin Y, Tang D. Magnetic graphene nanosheet-based microfluidic device for homogeneous real-time electronic monitoring of pyrophosphatase activity using enzymatic hydrolysate-induced release of copper ion. Analytical Chemistry. 2016 Jan 5;88(1):1030-8.
CrossRef - Gao Y, Tang J, Zhou Q, Yu Z, Wu D, Tang D. Excited-state intramolecular proton transfer-driven photon-gating for photoelectrochemical sensing of CO-releasing molecule-3. Analytical Chemistry. 2024 Mar 14;96(12):5014-21.
CrossRef - Lu L, Zeng R, Lin Q, Huang X, Tang D. Cation exchange reaction-mediated photothermal and polarity-switchable photoelectrochemical dual-readout biosensor. Analytical chemistry. 2023 Oct 25;95(44):16335-42.
CrossRef - Shu J, Tang D. Recent advances in photoelectrochemical sensing: from engineered photoactive materials to sensing devices and detection modes. Analytical chemistry. 2019 Nov 18;92(1):363-77.
CrossRef - Wang Y, Zeng R, Tian S, Chen S, Bi Z, Tang D, Knopp D. Bimetallic single-atom nanozyme-based electrochemical-photothermal dual-function portable immunoassay with smartphone imaging. Analytical Chemistry. 2024 Aug 10;96(33):13663-71.
CrossRef - Yaman D, Jimenez M, Ferreira-Gonzalez S, Corrigan D. Current trends in electrochemical approaches for liver biomarker detection: a mini-review. 2024.
CrossRef - Zhang J, Chen G, Zhang P, Zhang J, Li X, Gan DN, Cao X, Han M, Du H, Ye YA. The threshold of alpha-fetoprotein (AFP) for the diagnosis of hepatocellular carcinoma: A systematic review and meta-analysis. PLoS One. 2020 Feb 13;15(2):e0228857.
CrossRef - Zhou JM, Wang T, Zhang KH. AFP-L3 for the diagnosis of early hepatocellular carcinoma: A meta-analysis. Medicine. 2021 Oct 29;100(43):e27673.
CrossRef - Zhang YS, Chu JH, Cui SX, Song ZY, Qu XJ. Des-γ-carboxy prothrombin (DCP) as a potential autologous growth factor for the development of hepatocellular carcinoma. Cellular Physiology and Biochemistry. 2014 Aug 21;34(3):903-15.
CrossRef - Sauzay C, Petit A, Bourgeois AM, Barbare JC, Chauffert B, Galmiche A, Houessinon A. Alpha-foetoprotein (AFP): A multi-purpose marker in hepatocellular carcinoma. Clinica chimica acta. 2016 Dec 1;463:39-44.
CrossRef - Ofuji K, Saito K, Yoshikawa T, Nakatsura T. Critical analysis of the potential of targeting GPC3 in hepatocellular carcinoma. Journal of Hepatocellular Carcinoma. 2014 May 21:35-42.
CrossRef - Shih TC, Wang L, Wang HC, Wan YJ. Glypican-3: a molecular marker for the detection and treatment of hepatocellular carcinoma. Liver research. 2020 Dec 1;4(4):168-72.
CrossRef - Zhang J, Li D, Zhang R, Gao P, Peng R, Li J. The miR-21 potential of serving as a biomarker for liver diseases in clinical practice. Biochemical Society Transactions. 2020 Oct 30;48(5):2295-305.
CrossRef - Bandiera S, Pfeffer S, Baumert TF, Zeisel MB. miR-122–a key factor and therapeutic target in liver disease. Journal of hepatology. 2015 Feb 1;62(2):448-57.
CrossRef - Ye D, Zhang T, Lou G, Liu Y. Role of miR-223 in the pathophysiology of liver diseases. Experimental & molecular medicine. 2018 Sep;50(9):1-2.
CrossRef - Braconi C, Henry JC, Kogure T, Schmittgen T, Patel T. The role of microRNAs in human liver cancers. In Seminars in oncology 2011 Dec 1 (Vol. 38, No. 6, pp. 752-763). WB Saunders.
CrossRef - Kopystecka A, Patryn R, Leśniewska M, Budzyńska J, Kozioł I. The Use of ctDNA in the diagnosis and monitoring of hepatocellular carcinomaliterature review. International Journal of Molecular Sciences. 2023 May 26;24(11):9342.
CrossRef - Zhang Z, Chen P, Xie H, Cao P. Using circulating tumor DNA as a novel biomarker to screen and diagnose hepatocellular carcinoma: A systematic review and meta‐analysis. Cancer medicine. 2020 Feb;9(4):1349-64.
CrossRef - Xing C, Sun SG, Yue ZQ, Bai F. Role of lncRNA LUCAT1 in cancer. Biomedicine & Pharmacotherapy. 2021 Feb 1;134:111158.
CrossRef - Kim YA, Park KK, Lee SJ. LncRNAs act as a link between chronic liver disease and hepatocellular carcinoma. International journal of molecular sciences. 2020 Apr 20;21(8):2883.
CrossRef - Li Z, Zhu M. Detection of pollutants in water bodies: electrochemical detection or photo-electrochemical detection?. Chemical Communications. 2020;56(93):14541-52.
CrossRef - Trotter M, Borst N, Thewes R, von Stetten F. Electrochemical DNA sensing–Principles, commercial systems, and applications. Biosensors and Bioelectronics. 2020 Apr 15;154:112069.
CrossRef - Sun D, Lu J, Chen Z, Yu Y, Mo M. A repeatable assembling and disassembling electrochemical aptamer cytosensor for ultrasensitive and highly selective detection of human liver cancer cells. Analytica Chimica Acta. 2015 Jul 23;885:166-73.
CrossRef - Chikhaliwala P, Rai R, Chandra S. Simultaneous voltammetric immunodetection of alpha-fetoprotein and glypican-3 using a glassy carbon electrode modified with magnetite-conjugated dendrimers. Microchimica Acta. 2019 Apr;186:1-2.
CrossRef - Kamel RM, Abdel-aal FA, Mohamed FA, Abdeltawab A, Abdel-Malek MO, Othman AA, Mohamed AM. Copper@ eggshell nanocomposite/chitosan gelified carbon paste electrode as an electrochemical biosensor for l-tyrosine analysis as a biomarker in the serum of normal and liver disease patients. Microchemical Journal. 2024 Jun 1;201:110703.
CrossRef - Leung WH, Pang CC, Pang SN, Weng SX, Lin YL, Chiou YE, Pang ST, Weng WH. High-sensitivity dual-probe detection of urinary miR-141 in cancer patients via a modified screen-printed carbon electrode-based electrochemical biosensor. Sensors. 2021 May 3;21(9):3183.
CrossRef - Wu H, Zhang G, Yang X. Electrochemical immunosensor based on Fe3O4/MWCNTs-COOH/AuNPs nanocomposites for trace liver cancer marker alpha-fetoprotein detection. Talanta. 2023 Jul 1;259:124492.
CrossRef - Sheikhpour M, Golbabaie A, Kasaeian A. Carbon nanotubes: A review of novel strategies for cancer diagnosis and treatment. Materials Science and Engineering: C. 2017 Jul 1;76:1289-304.
CrossRef - Ruiyi L, Fangchao C, Haiyan Z, Xiulan S, Zaijun L. Electrochemical sensor for detection of cancer cell based on folic acid and octadecylamine-functionalized graphene aerogel microspheres. Biosensors and Bioelectronics. 2018 Nov 15;119:156-62.
CrossRef - Fu X, Li X, Han D, Yang W, Liu C, Fan L, Ding S, Ma Y. Ultrasensitive electrochemical biosensor for des-gamma-carboxy prothrombin analysis based on core-shell Pd@ PtCu-alloy loaded on WS2 nanosheet. Journal of Electroanalytical Chemistry. 2021 May 1;888:115213.
CrossRef - Masud MK, Na J, Younus M, Hossain MS, Bando Y, Shiddiky MJ, Yamauchi Y. Superparamagnetic nanoarchitectures for disease-specific biomarker detection. Chemical Society Reviews. 2019;48(24):5717-51.
CrossRef - Giannetto M, Mori L, Mori G, Careri M, Mangia A. New amperometric immunosensor with response enhanced by PAMAM-dendrimers linked via self assembled monolayers for determination of alpha-fetoprotein in human serum. Sensors and Actuators B: Chemical. 2011 Nov 28;159(1):185-92.
CrossRef - Purohit B, Vernekar PR, Shetti NP, Chandra P. Biosensor nanoengineering: Design, operation, and implementation for biomolecular analysis. Sensors International. 2020 Jan 1;1:100040.
CrossRef - Taheri N, Khoshsafar H, Ghanei M, Ghazvini A, Bagheri H. Dual-template rectangular nanotube molecularly imprinted polypyrrole for label-free impedimetric sensing of AFP and CEA as lung cancer biomarkers. Talanta. 2022 Mar 1;239:123146.
CrossRef - Liu S, Ma Y, Cui M, Luo X. Enhanced electrochemical biosensing of alpha-fetoprotein based on three-dimensional macroporous conducting polymer polyaniline. Sensors and Actuators B: Chemical. 2018 Feb 1;255:2568-74.
CrossRef - Al-Shami A, Oweis RJ, Al-Fandi MG. Developing an electrochemical immunosensor for early diagnosis of hepatocellular carcinoma. Sensor Review. 2021 May 17;41(2):125-34.
CrossRef - Gangopadhyay B, Roy A, Paul D, Panda S, Das B, Karmakar S, Dutta K, Chattopadhyay S, Chattopadhyay D. 3-polythiophene acetic acid nanosphere anchored few-layer graphene nanocomposites for label-free electrochemical immunosensing of liver cancer biomarker. ACS Applied Bio Materials. 2024 Jan 2;7(1):485-97.
CrossRef - Yaiwong P, Anuthum S, Sangthong P, Jakmunee J, Bamrungsap S, Ounnunkad K. A new portable toluidine blue/aptamer complex-on-polyethyleneimine-coated gold nanoparticles-based sensor for label-free electrochemical detection of alpha-fetoprotein. Frontiers in Bioengineering and Biotechnology. 2023 May 22;11:1182880.
CrossRef - Pusta A, Tertis M, Graur F, Cristea C, Al Hajjar N. Aptamers and new bioreceptors for the electrochemical detection of biomarkers expressed in hepatocellular carcinoma. Current Medicinal Chemistry. 2022 Aug 1;29(25):4363-90.
CrossRef - Ahmad OS, Bedwell TS, Esen C, Garcia-Cruz A, Piletsky SA. Molecularly imprinted polymers in electrochemical and optical sensors. Trends in biotechnology. 2019 Mar 1;37(3):294-309.
CrossRef - Moussa FB. Molecularly imprinted polymers meet electrochemical cancer chemosensors: A critical review from a clinical and economic perspective. Microchemical Journal. 2023 Aug 1;191:108838.
CrossRef - Liu C, Liu T. A graphene-assisted electrochemical sensor for detection of alpha-fetoprotein in serum. International Journal of Electrochemical Science. 2023 Apr 1;18(4):100081.
CrossRef - Forouzanfar S, Alam F, Pala N, Wang C. A review of electrochemical aptasensors for label-free cancer diagnosis. Journal of The Electrochemical Society. 2020 Mar 27;167(6):067511.
CrossRef - Sanko V, Kuralay F. Label-free electrochemical biosensor platforms for cancer diagnosis: Recent achievements and challenges. Biosensors. 2023 Mar 1;13(3):333.
CrossRef - Li G, Wang B, Zhao L, Shi X, Wu G, Chen W, Sun L, Liang J, Zhou Z. Label-free detection of glypican-3 using reduced graphene oxide/polyetherimide/gold nanoparticles enhanced aptamer specific sensing interface on light-addressable potentiometric sensor. Electrochimica Acta. 2022 Sep 10;426:140808.
CrossRef - Kim DH, Oh HG, Park WH, Jeon DC, Lim KM, Kim HJ, Jang BK, Song KS. Detection of alpha-fetoprotein in hepatocellular carcinoma patient plasma with graphene field-effect transistor. Sensors. 2018 Nov 19;18(11):4032.
CrossRef - Li G, Wang B, Li S, Li X, Yan R, Tan X, Liang J, Zhou Z. Competitive electrochemical aptasensor for high sensitivity detection of liver cancer marker GP73 based on rGO-Fc-PANi nanocomposites. Bioelectrochemistry. 2024 Dec 1;160:108767.
CrossRef - Olorundare FO, Sipuka DS, Sebokolodi TI, Kodama T, Arotiba OA, Nkosi D. An electrochemical immunosensor for an alpha-fetoprotein cancer biomarker on a carbon black/palladium hybrid nanoparticles platform. Analytical Methods. 2023;15(29):3577-85.
CrossRef - Zhang JH, Liu M, Zhou F, Yan HL, Zhou YG. Homogeneous electrochemical immunoassay using an aggregation–collision strategy for alpha-fetoprotein detection. Analytical Chemistry. 2023 Jan 24;95(5):3045-53.
CrossRef - Li J, Xing H, Jin P, Li M, Liu H. Electrochemical immunosensing based on signal amplification strategy for alpha-fetoprotein detection. International Journal of Electrochemical Science. 2022 Oct 1;17(10):22107.
CrossRef - Zhang T, Yang L, Yan F, Wang K. Vertically-ordered mesoporous silica film based electrochemical aptasensor for highly sensitive detection of alpha-fetoprotein in human serum. Biosensors. 2023 Jun 6;13(6):628.
CrossRef - Shao H, Liu Z. Epitope imprinted electrochemical sensor for highly sensitive detection of alpha-fetoprotein. Electrochimica Acta. 2024 Apr 20;484:144094.
CrossRef - Upan J, Youngvises N, Tuantranont A, Karuwan C, Banet P, Aubert PH, Jakmunee J. A simple label-free electrochemical sensor for sensitive detection of alpha-fetoprotein based on specific aptamer immobilized platinum nanoparticles/carboxylated-graphene oxide. Scientific Reports. 2021 Jul 7;11(1):13969.
CrossRef - Zhao S, Liu N, Wang W, Xu Z, Wu Y, Luo X. An electrochemical biosensor for alpha-fetoprotein detection in human serum based on peptides containing isomer D-Amino acids with enhanced stability and antifouling property. Biosensors and Bioelectronics. 2021 Oct 15;190:113466.
CrossRef - Liao X, Wang X, Li P, Chen S, Zhang M, Mei L, Qi Y, Hong C. Electrochemical immunosensor using artificial enzyme-induced metallization for the ultra-sensitive detection of alpha fetoprotein. Sensors and Actuators B: Chemical. 2021 Oct 1;344:130258.
CrossRef - Rahmati Z, Roushani M, Hosseini H. Hierarchical nickel hydroxide nanosheets grown on hollow nitrogen doped carbon nanoboxes as a high-performance surface substrate for alpha-fetoprotein cancer biomarkers electrochemical aptasensing. Talanta. 2022 Jan 15;237:122924.
CrossRef - Wu H, Yang X. Biofunctional photoelectrochemical/electrochemical immunosensor based on BiVO4/BiOI-MWCNTs and Au@ PdPt for alpha-fetoprotein detection. Bioelectrochemistry. 2024 Dec 1;160:108773.
CrossRef - Shu Y, Su T, Lu Q, Shang Z, Feng J, Jin D, Zhu A, Xu Q, Hu X. based electrochemical immunosensor device via Ni-Co MOF nanosheet as a peroxidase mimic for the label-free detection of alpha-fetoprotein. Sensors and Actuators B: Chemical. 2022 Dec 15;373:132736.
CrossRef - Gong L, Wu S, Liu J, Zhang M, Zhuang J, Xu D. Construction of an immunosensor based on Cys/Au@ TiO2 modification for the detection of liver cancer marker PIVKA-II. 2024 Aug 1;275:126082.
CrossRef - Li G, Feng H, Shi X, Chen M, Liang J, Zhou Z. Highly sensitive electrochemical aptasensor for Glypican-3 based on reduced graphene oxide-hemin nanocomposites modified on screen-printed electrode surface. Bioelectrochemistry. 2021 Apr 1;138:107696.
CrossRef - Lu W, Xie X, Lan X, Wu P, Peng H, He J, Zhong L, Liu X, Deng Z, Tan Z, Wu A. An electrochemical immunosensor for the detection of Glypican-3 based on enzymatic ferrocene-tyramine deposition reaction. Biosensors and Bioelectronics. 2023 Apr 1;225:115081.
CrossRef - Zhou Z, Zhao L, Li W, Chen M, Feng H, Shi X, Liang J, Li G. Glypican-3 electrochemical aptamer nanobiosensor based on hemin/graphene nanohybrids peroxidase-like catalytic silver deposition. Microchimica Acta. 2020 May;187:1-1.
CrossRef - Shi X, Chen M, Feng H, Zhou Z, Wu R, Li W, Liang J, Chen J, Li G. Glypican-3 electrochemical aptasensor based on reduced graphene oxide‐chitosan‐ferrocene deposition of platinum–palladium bimetallic nanoparticles. Journal of Applied Electrochemistry. 2021 May;51:781-94.
CrossRef - Li G, Guo F, Liang J, Wan B, Liang J, Zhou Z. Sandwich-type supersensitive electrochemical aptasensor of glypican-3 based on PrGO-Hemin-PdNP and AuNP@ PoPD. Microchimica Acta. 2024 Jun;191(6):340.
CrossRef - Li G, Feng H, Li X, Li S, Liang J, Zhou Z. A dual-signal output electrochemical aptasensor for glypican-3 ultrasensitive detection based on reduced graphene oxide-cuprous oxide nanozyme catalytic amplification strategy. Bioelectrochemistry. 2024 Aug 1;158:108709.
CrossRef - Li G, Wang B, Li L, Li X, Yan R, Liang J, Zhou X, Li L, Zhou Z. H-rGO-Pd NPs nanozyme enhanced silver deposition strategy for electrochemical detection of glypican-3. 2023 Feb 28;28(5):2271.
CrossRef - Ozer T, Henry CS. Recent advances in sensor arrays for the simultaneous electrochemical detection of multiple analytes. Journal of The Electrochemical Society. 2021 May 7;168(5):057507.
CrossRef - Kaya SI, Cetinkaya A, Bakirhan NK, Ozkan SA. Trends in sensitive electrochemical sensors for endocrine disruptive compounds. Trends in Environmental Analytical Chemistry. 2020 Dec 1;28:e00106.
CrossRef - Gunasekaran D, Gerchman Y, Vernick S. Electrochemical detection of waterborne bacteria using bi-functional magnetic nanoparticle conjugates. 2022 Jan 12;12(1):36.
CrossRef - Mollarasouli F, Zor E, Ozcelikay G, Ozkan SA. Magnetic nanoparticles in developing electrochemical sensors for pharmaceutical and biomedical applications. Talanta. 2021 May 1;226:122108.
CrossRef - Hassanpour S, Hasanzadeh M. Label-free electrochemical-immunoassay of cancer biomarkers: Recent progress and challenges in the efficient diagnosis of cancer employing electroanalysis and based on point of care (POC). Microchemical Journal. 2021 Sep 1;168:106424.
CrossRef - Mohammadniaei M, Nguyen HV, Tieu MV, Lee MH. 2D materials in development of electrochemical point-of-care cancer screening devices. Micromachines. 2019 Sep 30;10(10):662.
CrossRef - Du J, Tao H. Recent advances in electrochemical detection methods for liver cancer biomarkers. Int J Electrochem Sci. 2025;20(1):100921. doi:10.1016/j.ijoes.2024.100921.
CrossRef
Abbreviations:
HCC – Hepatocellular Carcinoma , AFP – Alpha-fetoprotein, AFP-L3 – AFP isoform L3 (Lectin-reactive AFP), DCP – Des-gamma-carboxy prothrombin, PIVKA-II – Protein Induced by Vitamin K Absence or Antagonist-II, GPC-3 – Glypican-3, miRNA – MicroRNA, lncRNA – Long non-coding RNA, ctDNA – Circulating Tumor DNA, CNT – Carbon Nanotube, AuNPs – Gold Nanoparticles, MWCNTs – Multi-Walled Carbon Nanotubes, PANI – Polyaniline, MIP – Molecularly Imprinted Polymer, SAM – Self-Assembled Monolayers, rGO – Reduced Graphene Oxide, FET – Field Effect Transistor, G-FET – Graphene Field Effect Transistor, LAPS – Light Addressable Potentiometric Sensor, BSA – Bovine Serum Albumin, GCE – Glassy Carbon Electrode, SPE – Screen Printed Electrode, SPCE – Screen Printed Carbon Electrode, PtNPs – Platinum Nanoparticles, PdNPs – Palladium Nanoparticles, Ni(OH)₂@N-C – Nickel Hydroxide/Carbon Nano boxes, TiO₂ – Titanium Dioxide, MOF – Metal-Organic Framework, AI – Artificial Intelligence, POC – Point-of-Care, PEG – Polyethylene Glycol, ELISA – Enzyme-Linked Immunosorbent Assay, DNA – Deoxyribonucleic Acid, RNA – Ribonucleic Acid, HULC – Highly Upregulated in Liver Cancer (lncRNA), MALAT1 – Metastasis Associated Lung Adenocarcinoma Transcript 1, HOTAIR – HOX Transcript Antisense RNA, GP73 – Golgi Protein 73, Fc – Ferrocene, CS – Chitosan, ITO – Indium Tin Oxide, BNPs – Bimetallic Nanoparticles, UCNP – Up conversion Nanoparticles







