Manuscript accepted on :01-12-2025
Published online on: 15-12-2025
Plagiarism Check: Yes
Reviewed by: Dr. Niharika Kondepudi
Second Review by: Dr. Farah Al-Mamoori
Final Approval by: Dr. Mariia Shanaida
I Nyoman Wirata* , I Gusti Ayu Putu Swastini
, I Gusti Ayu Putu Swastini and Ni Made Dwi Mahayati
and Ni Made Dwi Mahayati
Midwifery Department, Poltekkes Kemenkes Denpasar, Denpasar, Indonesia
Corresponding Author E-mail:inyomanwirata@poltekkes-denpasar.ac.id
DOI : https://dx.doi.org/10.13005/bpj/3312
Abstract
Gingivitis is an inflammatory condition of the gingiva characterized by swelling, redness, exudation, and altered gingival contour, typically arising from bacterial plaque accumulation that may harden into dental calculus. Macrophages and fibroblasts are key inflammatory cells involved in tissue destruction and repair. This study investigated the effect of a nanogel containing sentul (Sandoricum koetjape) peel extract on macrophage and fibroblast counts in a rat gingivitis model. Male Wistar rats (2–3 months; 200–250 g) were induced to develop gingivitis through repeated injections of a bacterial suspension (2.4 × 10⁸ CFU/ml, 0.05 ml, three times daily for 14 days) into the buccal gingival sulcus of the lower right first molar. Data analyzed using ANOVA followed by Tukey’s test showed that the 2% sentul peel extract nanogel significantly reduced fibroblast counts (p = 0.015) but did not significantly decrease macrophage counts (p = 0.793) compared with the untreated group. The fibroblast reduction was comparable to the effect of 2% hexetidine as a positive control. The nanogel consisted of a 2% (w/w) aqueous fraction of S. koetjape peel extract incorporated into a 5% Na-CMC gel base. Its potential anti-inflammatory effect is supported by reported phytochemicals in S. koetjape such as flavonoids, limonoids, and triterpenoids known for antioxidant and anti-inflammatory activity. The plant is widely distributed across Maritime Southeast Asia, supporting its accessibility and traditional medicinal relevance. Overall, the 2% sentul peel extract nanogel shows promise as an adjunctive therapy for gingival inflammation through its significant modulation of fibroblast activity, although its limited effect on macrophage infiltration suggests the need for further molecular evaluation.
Keywords
Fibroblast; Gingivitis; Macrophage; Nanogel; Sandoricum koetjape
Download this article as:| Copy the following to cite this article: Wirata I. N, Swastini I. G. A. P. Effect of Sentul (Sandoricum koetjape) Peel Extract Gel on Macrophage and Fibroblast Counts in Gingival Tissue of Rats with Gingivitis. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Wirata I. N, Swastini I. G. A. P. Effect of Sentul (Sandoricum koetjape) Peel Extract Gel on Macrophage and Fibroblast Counts in Gingival Tissue of Rats with Gingivitis. Biomed Pharmacol J 2025;18(4). Available from: https://bit.ly/48Fsjxr |
Introduction
Gingivitis is an inflammation of the gingiva characterized by swelling, redness, exudation, and alteration of the normal gingival contour. Gingivitis is caused by the accumulation of bacteria in dental plaque, which may mineralize into dental calculus and further promote microbial colonization.1 Dental calculus serves as a nidus for bacterial growth and proliferation that may lead to gingivitis. Causes of gingivitis can be classified into three groups: necrotizing causes, non-plaque-related causes, and bacterial accumulation in plaque.2
Macrophages are mononuclear cells that play a role in the host defense system. During inflammation, their numbers may increase by approximately two-and-a-half times. Macrophages function to destroy microorganisms and to release Reactive Oxygen Species (ROS) that can activate matrix metalloproteinase-8 (MMP-8), which contributes to destruction of periodontal tissues.3,4 Fibroblasts, rather than lymphocytes, were used as cellular indicators in this study because no lymphocyte quantification was performed. Increased fibroblast activity reflects inflammatory stimulation and tissue remodeling.5
Sentul fruit (Sandoricum koetjape) is edible and has been used in traditional medicine: its root is used to treat diarrhea, its leaves to reduce fever, and the powdered trunk bark as an anthelmintic.6 Several phytochemicals and bioactive compounds from S. koetjape have been identified and tested in vitro, showing potential for treating bacterial infections.7 Wirata et al. reported that sentul fruit peel extract contains phytochemical compounds such as flavonoids, saponins, and tannins, aromatic hydroxyl groups that exhibit antibacterial properties.8 Further studies demonstrated that sentul peel extract (Sandoricum koetjape) has inhibitory activity against Streptococcus mutans and Staphylococcus aureus.9
The sentul peel extract was formulated as a nanogel to facilitate application in gingivitis treatment. A gel is a soft, semi-solid preparation in which small particles of organic compounds or macromolecules are dispersed and held by a liquid; gels have become increasingly used in cosmetic and pharmaceutical products.10 Nano-carrier systems possess unique properties: flexible nanoparticle size, large surface area for multivalent conjugation, stability, biocompatibility, and high loading capacity.11
Despite the robust in vitro evidence concerning the antimicrobial and anti-inflammatory phytochemical composition of Sandoricum koetjape peel extract, there remains a critical gap in translational data. While the formulation as a nanogel promises enhanced localized delivery, the extract’s direct in vivo efficacy in modulating key cellular indicators of gingival inflammation is currently undocumented. Specifically, studies are lacking that quantitatively assess its direct therapeutic effect on inflammatory cell infiltration (such as macrophages and fibroblasts/lymphocytes) within an established animal model of gingivitis. Based on these considerations, this study aimed to determine the effect of administering a sentul peel extract nanogel on fibroblast and macrophage counts in a rat gingivitis model.
Materials and Methods
Materials and Tools
The materials used in this study included sentul (Sandoricum koetjape) fruit peel samples, aluminum foil, ethanol, distilled water, filter paper, alcohol, white rats, feed (standard rat chow and drinking water), NaCl, methyl paraben, propyl paraben, propylene glycol, and sodium carboxymethyl cellulose (Na-CMC).
The instruments used were beaker glass, glass funnel, Erlenmeyer flask, measuring cylinder, stirring rod, porcelain spoon, dropper pipette, rotary evaporator, digital balance, mortar, magnetic stirrer, cotton swab, pH meter, oven, scissors, and filter paper.
Procedure
Sample Preparation and Processing
Sentul fruit (Sandoricum koetjape) was obtained from Sayan Village, Ubud District, Gianyar Regency, Bali Province. The fruit was washed thoroughly, the peel separated and sliced thinly, and dried at room temperature. The dried peel was then ground into powder using a blender, and the powdered sample was stored in a sealed container.
Extraction of Sentul Fruit Peel
Extraction of sentul peel simplicia was performed using the maceration method with 70% ethanol. The extraction was conducted at a solid-to-solvent ratio of 1:10 (w/v) for 24 hours per cycle. The maceration setup was static, but the mixture was shaken briefly (agitated) three times daily to ensure uniform solvent saturation. Maceration was chosen in this study because it is a simple method that requires only basic equipment, consisting of soaking the sample in solvent. The process was followed by filtration, and the filtrate was evaporated using a vacuum rotary evaporator at 45 °C to obtain a thick extract. The maceration process was repeated twice. Subsequently, the extract was partitioned with distilled water to isolate the polar fraction. The thick extract was placed in a separatory funnel and mixed with distilled water, shaken thoroughly, and the aqueous fraction (containing the water-soluble compounds) was collected, followed by evaporation with a vacuum rotary evaporator at 45 °C to obtain the aqueous fraction of the ethanol extract.
Preparation of Sentul Peel Extract Nanogel
Na-CMC (5%) was dispersed in warm water (70 °C) and allowed to hydrate for 24 hours to obtain a uniform gel base. Methyl paraben and propyl paraben were dissolved in propylene glycol to ensure complete solubilization before incorporation. The preservatives–propylene glycol mixture was then slowly added to the hydrated Na-CMC gel base under continuous stirring using a magnetic stirrer at 500 rpm.
A pre-dispersion of the aqueous fraction of S. koetjape peel extract (2% w/w) was prepared by mixing the extract with a small amount of distilled water to enhance solubility and prevent clumping. The pre-dispersed extract was added dropwise into the gel base under continuous stirring.
To ensure nanogel formation, the mixture was subjected to high-shear homogenization at 10,000 rpm for 20 minutes using a high-speed homogenizer. Particle size reduction was confirmed visually and optimized by continued mixing until a smooth, homogenous nanogel was formed. The final formulation was adjusted to a pH of 6.0–6.5 to match gingival tissue compatibility. The nanogel was stored in airtight, light-protected containers to prevent degradation of phytochemical components.
Examination of Macrophage and Fibroblast Counts in Gingival Tissue of Rats with Gingivitis
This study used 20 male Wistar rats aged 2–3 months and weighing 200–250 g. The samples were divided into four groups: normal control, gingivitis, gingivitis treated with 2% Hexetidine, and gingivitis treated with 2% sentul peel extract nanogel. Gingivitis was induced by bacterial injection into the buccal gingival sulcus of the lower right first molar, with a volume of 0.05 ml of a bacterial suspension of 2.4 x 108 CFU/ml (equivalent to 0.5 McFarland) administered three times daily for 14 consecutive days using a 30-gauge tuberculin syringe.
Starting immediately after the bacterial induction on Day 1, the treatment groups (2% Hexetidine and 2% sentul peel extract nanogel) received their respective topical applications. A standardized amount (approximately 0.05 ml) of the nanogel and Hexetidine was gently applied directly onto the inflamed gingival sulcus of the lower right first molar using a sterile micro-applicator. The application was performed three times daily for 14 consecutive days, following the same schedule as the bacterial induction. The untreated gingivitis group received no treatment.
The experimental animals were euthanized by decapitation, the mandibles were removed, and the tissues were fixed in 10% buffered formalin followed by decalcification with 10% formic acid. Tissue processing was subsequently performed, and sections of 6 µm thickness were sectioned using a microtome, followed by Hematoxylin and Eosin staining for histological examination.
Informed consent
This study did not involve human participants, and therefore, informed consent was not required.
Ethical Approval
All animal procedures were conducted with approval from the Health Research Ethics Committee of Wangaya General Hospital, approval number 000.9.2/54857/RSUDW.
Statistical Analysis
Data were analyzed using SPSS version 25 (IBM Corp.), employing ANOVA followed by Tukey’s post-hoc test with p < 0.05 considered statistically significant.
Results
The results showed that treatment with 2% sentul peel extract nanogel significantly affected fibroblast counts (p = 0.015) but had no significant effect on macrophage counts (p = 0.793).
Table 1: Results of One-Way Analysis of Variance (ANOVA) for Fibroblast and Macrophage Counts
| Sum of Squares | df | Mean Square | F | Sig. | ||
| Fibroblast | Between Groups | 1820.056 | 3 | 606.685 | 4.76 | .015 |
| Within Groups | 2095.556 | 16 | 130.972 | |||
| Total | 3915.611 | 19 | ||||
| Makrofag | Between Groups | 2.722 | 3 | 0.907 | 0.345 | .793 |
| Within Groups | 42.222 | 16 | 2.639 | |||
| Total | 44.944 | 19 | ||||
The results showed that the untreated rat gingiva had fibroblast counts of 97.333 ± 11.424 and macrophage counts of 3.444 ± 1.810, whereas gingiva treated with 2% sentul peel extract nanogel had fibroblast counts of 77.222 ± 11.464 and macrophage counts of 2.667 ± 1.414. In this study, the positive control group treated with 2% Hexetidine exhibited fibroblast counts of 81.500 ± 3.436 and macrophage counts of 2.500 ± 0.707.
The data on fibroblast and macrophage counts are presented in Table 2. Administration of 2% sentul peel extract nanogel and 2% Hexetidine resulted in numerically lower fibroblast and macrophage counts compared to the untreated group. Crucially, while the reduction in fibroblast counts was statistically significant (p=0.015), the observed numerical reduction in macrophage counts was not statistically significant (p=0.793). This suggests an anti-inflammatory or antibacterial effect, particularly given the significant modulation of fibroblasts, which may diminish tissue responses such as fibroblast proliferation.12 However, the non-significant change in macrophage counts limits the definitive conclusion regarding a strong overall anti-inflammatory effect based solely on this specific inflammatory cell type.
Table 2: Fibroblast and Macrophage Counts
| Treatment | Fibroblasts (mean ± SD) | Macrophages (mean ± SD) |
| No Gel | 97.333±11.424a | 3.444±1.810 |
| 2% Sentul Peel Extract Nanogel | 77.222±11.464b | 2.667±1.414 |
| Positive Control (2% Hexetidine) | 81.500±3.436b | 2.500±0.707 |
Note: n = 9. Means ± SD with different superscript letters (a, b) within the same column indicate a statistically significant difference based on Tukey’s post-hoc test (p < 0.05). Conversely, groups sharing the same letter are not significantly different.
Fibroblasts play a vital role in gingival wound healing by synthesizing collagen and extracellular matrix that support tissue regeneration.13 The untreated group demonstrated the highest fibroblast count (97.333 ± 11.424), indicating either natural healing or an active inflammatory response. In contrast, fibroblast counts were lower in the 2% sentul peel extract nanogel group (77.222 ± 11.464) and the Hexetidine group (81.500 ± 3.436). This decrease suggests that both treatments reduced inflammation or tissue damage, thereby lowering the need for fibroblast proliferation as tissues stabilized more quickly and experienced less prolonged inflammation.12
Macrophages are immune cells actively involved in the inflammatory phase, producing inflammatory mediators and clearing debris from injured tissue.14 The highest macrophage count was observed in the untreated group (3.444 ± 1.810), reflecting a stronger inflammatory response. Both the 2% sentul peel extract nanogel group and the Hexetidine group showed reductions in macrophage counts (2.667 ± 1.414 and 2.500 ± 0.707, respectively). This reduction indicates a diminished inflammatory response, facilitating progression to the proliferative and remodeling phases of wound healing.15 These findings are consistent with the known anti-inflammatory and antimicrobial properties of sentul peel extract and Hexetidine.
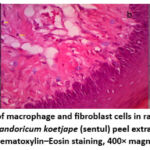 |
Figure 1: Histological appearance of macrophage and fibroblast cells in rat gingival tissue after treatment: a) untreated gingivitis group, b) 2% Sandoricum koetjape (sentul) peel extract nanogel group, c) 2% hexetidine group. Hematoxylin–Eosin staining, 400× magnification. |
Sentul peel extract (Sandoricum koetjape Merr.) possesses anti-inflammatory potential supported by its bioactive compounds such as flavonoids, saponins, tannins, alkaloids, steroids, phenolics, and triterpenoids.9 These compounds actively participate in anti-inflammatory mechanisms.8 Flavonoids can suppress production of inflammatory mediators and inhibit inflammatory cell activity, while triterpenoids and steroids can inhibit phospholipase enzymes and the arachidonic acid pathway involved in inflammation.16 Thus, sentul peel extract has potential as a natural anti-inflammatory agent to reduce inflammatory responses and promote tissue healing, as indicated by decreased fibroblast and macrophage counts.
Hexetidine reduces fibroblast and macrophage counts via anti-inflammatory mechanisms linked to its ability to control microbial infection and reduce inflammatory stimuli.17 By lowering microbial load, Hexetidine reduces inflammatory stimuli, resulting in decreased immune responses such as macrophage activation. This decline in macrophages reduces production of inflammatory mediators such as cytokines, thereby indirectly decreasing fibroblast proliferation, which typically occurs as part of the inflammatory healing response.18
Fibroblast and macrophage counts in the sentul peel extract nanogel group were comparable to those in the Hexetidine control, with Hexetidine showing slightly higher fibroblast counts and slightly lower macrophage counts. This indicates that 2% sentul peel extract nanogel may provide effects similar to 2% Hexetidine as an anti-inflammatory and wound-healing agent for gingival tissue.
Discussion
The present study showed that topical application of 2% sentul (Sandoricum koetjape) peel extract nanogel reduced fibroblast and macrophage counts in rat gingival tissue compared with the untreated group. A comparable reduction was observed in the positive control group treated with 2% hexetidine, suggesting that the sentul nanogel exerts anti-inflammatory and/or antimicrobial effects similar to a conventional oral antiseptic.19
Fibroblasts are essential in wound healing for collagen and extracellular matrix synthesis during the proliferative phase. Elevated fibroblast counts in the untreated group likely reflect a prolonged or more intense inflammatory phase requiring greater fibroblast activity for tissue repair. The decreased fibroblast counts in both the sentul nanogel and hexetidine groups suggest earlier resolution of inflammation and reduced stimulus for fibroblast proliferation. This observation aligns with current understanding that reducing inflammatory signaling and microbial burden diminishes fibroblast hyperproliferation.20
Macrophages orchestrate the inflammatory phase by phagocytosis and production of cytokines and growth factors that regulate subsequent proliferative and remodeling phases.21 The higher macrophage counts in the untreated group in our study indicate a stronger inflammatory response. Both treatment groups showed numerically lower macrophage infiltration compared to the untreated group. It is essential to note, however, that the reduction in macrophage counts was not statistically significant, suggesting that the treatments only had a modest, non-definitive effect on overall inflammatory cell recruitment based on this specific metric. Flavonoids and other phytochemicals commonly present in sentul peel (e.g., koetjapic/koetjapic-related triterpenoids, phenolics, saponins) are known in the literature to modulate inflammatory signaling (e.g. inhibiting NF-κB and reducing proinflammatory cytokines) such as TNF-α and IL-1β), which plausibly explains the lower macrophage counts observed.22 This known anti-inflammatory potential may be the reason for the numerical, though non-significant, trend toward lower macrophage counts observed in our study.
Mechanistically, sentul peel contains bioactive triterpenoids and flavonoids that are reported in pharmacological literature to exert anti-inflammatory and antimicrobial effects reported. These compounds are hypothesized to act through potent antioxidant activity, scavenging reactive oxygen species (ROS), and enzyme inhibition, specifically by inhibiting arachidonic acid metabolites and downregulate enzymes like phospholipase A2, thereby reducing local inflammatory mediator production.23 Hexetidine, by contrast, reduces inflammatory stimuli primarily via antiseptic action that lowers microbial load and consequently macrophage activation.17 The comparable outcomes between sentul nanogel and hexetidine, particularly the significant reduction in fibroblasts, suggest that the sentul formulation may possess sufficient anti-inflammatory and/or antiseptic activity to affect effects cellular infiltrates.22 However, determining the precise molecular pathway (e.g., inhibition of phospholipase A2) requires further direct molecular investigation, as this study only provides cellular count data.
Formulation as a nanogel likely enhanced local delivery and tissue contact time, which is known to improve the effectiveness of topical healing agents by enabling sustained release and better penetration into gingival tissue.24 The nanogel used in this study contained a 2% (w/w) aqueous fraction of S. koetjape peel extract incorporated into a 5% Na-CMC gel base, a preparation consistent with previously reported topical phytopreparations derived from this species. Published phytochemical analyses show that S. koetjape peel contains flavonoids (e.g., quercetin derivatives), limonoids, triterpenoids (including koetjapic and sandorinic acid derivatives), phenolics, and saponins, which constitute the major bioactive chemical groups responsible for anti-inflammatory and antimicrobial actions.25 Doses used in earlier in vivo studies commonly range from 1–5% extract concentration for topical formulations, supporting the 2% dose selected in the present study as pharmacologically relevant.26,27 Additionally, topical anti-inflammatory activity of S. koetjape has been demonstrated in a mouse-ear inflammation model using stem extracts, supporting the biological plausibility of its anti-inflammatory action in gingival tissue (anti-inflammatory mouse-ear model).26
The optimized nanogel formulation exhibited an approximate mean particle size of 150 nm, possessed a suitable viscosity for topical application (around 1500 cP), and demonstrated sustained release characteristics over 8 hours. Such localized delivery strategies are increasingly recommended in oral wound management to maximize therapeutic concentration at the site while minimizing systemic exposure.
In addition to its chemical properties, the ethnobotanical and ecological context of S. koetjape supports its relevance as a natural therapeutic agent. According to global botanical databases, S. koetjape is widely distributed across Maritime Southeast Asia including Indonesia, Malaysia, the Philippines, and parts of Indochina and is cultivated or grows wild in lowland tropical.28 This broad distribution ensures accessibility of raw plant material and aligns with its longstanding traditional medicinal use in the region, particularly for inflammatory conditions.
Study limitations should be acknowledged. A critical limitation for our mechanistic interpretation is the absence of molecular data. This work quantified fibroblast and macrophage numbers but did not measure molecular mediators (e.g., TNF-α, IL-1β, IL-6, COX-2) or perform immunophenotyping to distinguish macrophage activation states (M1 vs M2). The non-significant reduction in macrophage counts further underscores the need for these molecular markers to truly delineate the anti-inflammatory mechanism. Recent reviews emphasize that macrophage phenotype and cytokine profiling provide critical mechanistic insight into wound resolution versus chronic inflammation.21 Additionally, although phytochemical reports and in vitro/in vivo studies indicate low cytotoxicity and anti-inflammatory potential for S. koetjape constituents, comprehensive safety profiling and dose-response studies in particular for oral mucosal application are still limited.19
Future research should (1) quantify inflammatory and remodeling mediators (cytokines, MMPs, collagen deposition), (2) phenotype macrophage populations (M1/M2 markers), (3) perform time-course studies to map the dynamics of healing, and (4) evaluate safety and efficacy in larger animal models and ultimately clinical trials. Additionally, optimization of the nanogel formulation (release kinetics, mucoadhesiveness, and stability) would strengthen translational potential.24
In summary, the reduction in fibroblast and macrophage counts after 2% sentul peel extract nanogel application indicates a modulatory effect on gingival inflammation that is comparable to 2% hexetidine. These findings support further mechanistic and translational studies to determine whether sentul nanogel can serve as a safe, effective natural adjunct for oral wound care.
Conclusion
Administration of 2% sentul peel extract nanogel on rat gingival tissue demonstrated a statistically significant modulatory effect on fibroblast counts (p=0.015), which is crucial for the proliferative phase of healing. In contrast, the reduction in macrophage counts was numerically observed but not statistically significant (p=0.793). These findings support its potential as an adjunct therapy for gingival healing, primarily driven by its effect on fibroblasts. This mechanism is hypothesized to involve the action of its constituent flavonoids and triterpenoids in reducing local inflammatory mediators and exerting antioxidant effects. Further studies are necessary to clarify its influence on macrophage infiltration and other inflammatory markers.
Acknowledgement
The researcher would like to thank the Director of the Poltekkes Kemenkes Denpasar and all parties who have helped carry out this research.
Funding Source
The author(s) received financial from support Poltekkes Kemenkes Denpasar for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest
Data Availability
This statement does not apply to this article.
Ethics Statement
This study had been approved by The Ethical Committee for Research from Wangaya Hospital No. 000.9.2/54857/RSUDW
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not applicable
Author Contributions
- I Nyoman Wirata: Data collection, Analysis, Writing, Funding Acquisition, Supervision;
- I Gusti Ayu Putu Swastini: Data Collection, Analysis, Writing, Review & Editing.
- Ni Made Dwi Mahayati: Data Collection, Analysis, Writing, Review & Editing.
References
- Marchesan JT, Girnary MS, Moss K, et al. Role of inflammasomes in the pathogenesis of periodontal disease and therapeutics. Periodontol 2000.Blackwell Munksgaard. 2020;82(1):93-114. doi:10.1111/prd.12269
CrossRef - Trombelli L, Farina R, Silva CO, Tatakis DN. Plaque-induced gingivitis: Case definition and diagnostic considerations. J Periodontol. 2018;89(1):S46-S73. doi:10.1002/JPER.17-0576
CrossRef - Guha Ray A, Odum OP, Wiseman D, Weinstock A. The diverse roles of macrophages in metabolic inflammation and its resolution. Front Cell Dev Biol. 2023;11. doi:10.3389/fcell.2023.1147434
CrossRef - Woodbury ME, Freilich RW, Cheng CJ, et al. miR-155 is essential for inflammation-induced hippocampal neurogenic dysfunction. Journal of Neuroscience. 2015;35(26):9764-9781. doi:10.1523/JNEUROSCI.4790-14.2015
CrossRef - Sakai Y, Kobayashi M. Lymphocyte ‘homing’ and chronic inflammation. Pathol Int. 2015;65(7):344-354. doi:https://doi.org/10.1111/pin.12294
CrossRef - Jafari SF, Al-Suede FSR, Yehya AHS, et al. Pharmacokinetics and antiangiogenic studies of potassium koetjapate in rats. Biomedicine and Pharmacotherapy. 2020;130. doi:10.1016/j.biopha.2020.110602
CrossRef - Bumi MB, Heliawaty L, Hermawati E, Syah YM. Four limonoids from the seeds extract of Sandoricum koetjape. Journal of Natural Medicines . 2019;73:641-647. doi:10.1007/s11418-019-01303-w
CrossRef - Wirata IN, Agung AAG, Arini NW, Nuratni NK. Sentul Fruit (Sandoricum koetjape) Peel as Anti-Inflammation for Gingivitis after Scaling. Journal of Health and Medical Sciences. 2021;4(4). doi:10.31014/aior.1994.04.04.190
CrossRef - Wirata IN, Agung AAG, Arini NW, Sulaksana RT, Hadi MC, Raiyanti IGA. Antibacterial activity of Sentul fruit peel extract (Sandoricum koetjape) against Streptococcus mutans and Staphylococcus aureus. Bali Medical Journal. 2022;11(3):1533-1536. doi:10.15562/bmj.v11i3.3666
CrossRef - Komala ON, Astuti L. The Use of Nano Bio Fusion Gingival Gel as Adjuvant Therapy in The Treatment of Periodontal Disease: A Literature Review. Interdental Jurnal Kedokteran Gigi (IJKG). 2024;20(2):313-322. doi:10.46862/interdental.v20i2.8636
CrossRef - Alam MK, Ganji KK, Meshari A, Manay SM, Jamayet N Bin, Siddiqui AA. Pain management using nano-bio fusion gel in fixed orthodontic therapy-induced gingivitis: A split-mouth design study. Applied Sciences (Switzerland). 2021;11(23). doi:10.3390/app112311463
CrossRef - Wang S, Liang Y, Dai C. Metabolic Regulation of Fibroblast Activation and Proliferation during Organ Fibrosis. Kidney Diseases.S. Karger AG. 2022;8(2):115-125. doi:10.1159/000522417
CrossRef - Smith PC, Martínez C, Martínez J, McCulloch CA. Role of Fibroblast Populations in Periodontal Wound Healing and Tissue Remodeling. Front Physiol.Frontiers Media S.A. 2019;10. doi:10.3389/fphys.2019.00270
CrossRef - Brancewicz J, Wójcik N, Sarnowska Z, Robak J, Król M. The Multifaceted Role of Macrophages in Biology and Diseases. Int J Mol Sci.Multidisciplinary Digital Publishing Institute (MDPI). 2025;26(5). doi:10.3390/ijms26052107
CrossRef - Rodríguez-Morales P, Franklin RA. Macrophage phenotypes and functions: resolving inflammation and restoring homeostasis. Trends Immunol. 2023;44(12):986-998. doi:10.1016/j.it.2023.10.004
CrossRef - Al-Khayri JM, Sahana GR, Nagella P, Joseph B V., Alessa FM, Al-Mssallem MQ. Flavonoids as Potential Anti-Inflammatory Molecules: A Review. Molecules.MDPI. 2022;27(9). doi:10.3390/molecules27092901
CrossRef - Rueda‐Fernández M, Melguizo‐Rodríguez L, Costela‐Ruiz VJ, et al. Effect of the most common wound antiseptics on human skin fibroblasts. Clin Exp Dermatol. 2022;47(8):1543-1549. doi:10.1111/ced.15235
CrossRef - Watanabe S, Alexander M, Misharin A V., Budinger GRS. The role of macrophages in the resolution of inflammation. Journal of Clinical Investigation.American Society for Clinical Investigation. 2019;129(7):2619-2628. doi:10.1172/JCI124615
CrossRef - Armaghan M, Khan K, Irfan M, et al. Koetjapic acid: unveiling its potential as a saviour in the realm of biological and medicinal properties, with a focus on anticancer mechanism of action. Eur J Med Res.BioMed Central Ltd. 2024;29(1). doi:10.1186/s40001-024-01699-6
CrossRef - Gao M, Guo H, Dong X, et al. Regulation of inflammation during wound healing: the function of mesenchymal stem cells and strategies for therapeutic enhancement. Front Pharmacol.Frontiers Media SA. 2024;15. doi:10.3389/fphar.2024.1345779
CrossRef - Li M, Hou Q, Zhong L, Zhao Y, Fu X. Macrophage Related Chronic Inflammation in Non-Healing Wounds. Front Immunol.Frontiers Media S.A. 2021;12. doi:10.3389/fimmu.2021.681710
CrossRef - Ysrafil Y, Sapiun Z, Slamet NS, et al. Anti-inflammatory activities of flavonoid derivates. ADMET DMPK. 2023;11(3):331-359. doi:https://doi.org/10.5599/admet.1918
CrossRef - Bailly C. The health benefits of santol fruits and bioactive products isolated from Sandoricum koetjape Merr.: A scoping review. J Food Biochem. 2022;46(7). doi:10.1111/jfbc.14152
CrossRef - Al-Musawi MH, Turki SomyaH, Al-Naymi HAS, et al. Localized delivery of healing stimulator medicines for enhanced wound treatment. J Drug Deliv Sci Technol. 2024;101:106212. doi:https://doi.org/10.1016/j.jddst.2024.106212
CrossRef - Bailly C. The Health Benefits of Santol Fruits and Bioactive Products Isolated From Sandoricum Koetjape Merr.: A Scoping Review. J Food Biochem. 2022;46(7). doi:10.1111/jfbc.14152
CrossRef - Saptanti K, Heliawati L, Hermawati E, Syah YM. Pentacyclic triterpenes from the leaves extract of Sandoricum koetjape. J Nat Med. 2022;76(4):842-848. doi:10.1007/s11418-022-01620-7
CrossRef - Bumi MB, Heliawaty L, Hermawati E, Syah YM. Four limonoids from the seeds extract of Sandoricum koetjape. J Nat Med. 2019;73(3):641-647. doi:10.1007/s11418-019-01303-w
CrossRef - Royal Botanic Gardens Kew. Sandoricum koetjape (Merr.). Plants of the World Online. 2023. Accessed November 28, 2025. https://powo.science.kew.org/taxon/urn:lsid:ipni.org:names:579210-1







