Manuscript accepted on :04-12-2025
Published online on: 15-12-2025
Plagiarism Check: Yes
Reviewed by: Dr. Fariha Jasin Mansur
Second Review by: Dr. Dunya Abd Al- Malik Mohammed Salih
Final Approval by: Dr. Anton R Keslav
Sri Masyeni1* , Ratna Kartika Dewi2
, Ratna Kartika Dewi2 , Kadek Suryawan3
, Kadek Suryawan3 , Saraswati Laksmi Dewi1
, Saraswati Laksmi Dewi1 , Arya Giri Prebawa1
, Arya Giri Prebawa1 , Pande Ayu Naya Kasih Permatananda2
, Pande Ayu Naya Kasih Permatananda2 , Rois M Fatawy4
, Rois M Fatawy4 and Erni Juwita Nelwan5
and Erni Juwita Nelwan5
1Department of Internal Medicine, Faculty of Medicine and Health Sciences, Universitas Warmadewa, Bali, Indonesia
2Faculty of Medicine and Health Sciences, Universitas Warmadewa, Bali, Indonesia;
3Department of Microbiology, Faculty of Medicine and Health Sciences, Universitas Warmadewa, Bali, Indonesia;
4Department of Infectious Disease and Immunology Research Center, Indonesia Medical Education and Research Institute, Faculty of Medicine, Universitas Indonesia, Jakarta, Indonesia;
5Department of Internal Medicine, Faculty of Medicine, Universitas Indonesia, Jakarta, Indonesia.
Corresponding Author E-mail:sri.masyeni@warmadewa.ac.id
DOI : https://dx.doi.org/10.13005/bpj/3290
Abstract
The rising incidence of Streptococcus suis (S. suis)-associated meningitis, often associated with sudden deafness, warrants further investigation into its clinical characteristics and outcomes. This cross-sectional study analyzed medical records of patients with suspected S. suis-associated meningitis from 2022 to 2024, diagnosing the condition based on symptoms such as fever, headache, neck stiffness, and altered mental status, with confirmation via blood cultures. A notable limitation was the absence of CSF testing. Among 67 patients, 34 (50.7%) experienced deafness, although 3 had negative cultures. Key symptoms included fever (100%), neck stiffness (100%), and severe headache (28.4%). S. suis was identified in 10.5% of blood cultures, with 30 probable (44.8%) and seven confirmed cases (10.4%). Deafness occurred in 85.7% of S. suis bacteremia cases, but no significant link was found between raw pork consumption and deafness (p = 0.151). The overall case fatality rate was 4.5%, with a 1.5% mortality rate among confirmed cases. Common complications included septic shock and sudden deafness. Increased awareness and preventive measures related to pig exposure are essential to address this public health concern.
Keywords
Bacteremia; Meningitis; Sudden deafness; S. Suis; Zoonotic
Download this article as:| Copy the following to cite this article: Masyeni S, Dewi R. K, Suryawan K, Dewi S. L, Prebawa A. G, Permatananda P. A. N. K, Fatawy R. M, Nelwan E. J. Detection of Streptococcus suis Bacteremia in a Subset of Meningitis Patients with Sudden Deafness. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Masyeni S, Dewi R. K, Suryawan K, Dewi S. L, Prebawa A. G, Permatananda P. A. N. K, Fatawy R. M, Nelwan E. J. Detection of Streptococcus suis Bacteremia in a Subset of Meningitis Patients with Sudden Deafness. Biomed Pharmacol J 2025;18(4). Available from: https://bit.ly/4oTElYC |
Introduction
Streptococcus suis-associated meningitis is a significant zoonotic disease caused by Streptococcus suis, particularly prevalent in regions with close human–pig interactions. This Gram-positive bacterium is a common pathogen in pigs and is transmitted to humans through direct contact with pigs or pork products. 1 The disease is primarily diagnosed in rural areas and among individuals with occupational exposure, making it a leading cause of bacterial meningitis in adults, especially in Southeast Asia.2,3 As the leading cause of bacterial meningitis in adults, particularly in areas where close contact between humans and pigs is common, the incidence of S. suis-associated meningitis is reported to be increasing in Southeast Asian countries, including Vietnam, Thailand, Hong Kong, China, and Japan.4 Cases of this type of meningitis have also been identified in Europe, North America, South America, Oceania, and Africa.5–8 The reported annual incidence of S. suis meningitis in Thailand is alarmingly high, reaching between 0 and 0.381 cases per 100,000 individuals 9. In Vietnam, the estimated annual incidence rate is approximately 0.25-0.32 cases per 100,000 population.10 Hong Kong reports a yearly incidence of 0.09 cases per 100,000 people, underscoring the persistent threat posed by this infection.11 Furthermore, between 2011 and 2014, China’s estimated annual incidence rate of S. suis infections in humans ranged from 0.249 to 0.324 cases per 100,000 population, highlighting a notable public health issue that warrants attention.10
Key virulence factors include the capsular polysaccharides, the toxin suilysin, biofilm formation, and genes related to immune evasion, metabolism, and stress responses.12–14 Streptococcus suis comprises 29 serotypes, classified by capsular polysaccharide (CPS) composition.15 S. suis infection induces inflammasome activation, leading to the secretion of pro-inflammatory cytokines and the disruption of blood–brain barrier and tight junction proteins, facilitating the pathogen’s invasion into the central nervous system.16,17 The infection can present with various clinical manifestations, including fever, chills, headache, decreased appetite, generalized pain, signs of meningitis, sepsis, irreversible hearing loss, toxic shock syndrome, arthritis, skin manifestations, and vestibular dysfunction.18–20
Key risk factors for contracting this infection include consuming raw or undercooked pork, such as undercooked pig blood or intestine, and handling sick or dead pigs.15,21 Since 2014, cases of S. suis-associated meningitis have been reported to increase in Bali, Indonesia, where pig farms are densely concentrated.22 During ceremonies, the Balinese often serve high-risk dishes, including raw pig blood, undercooked suckling pig, or pig intestine.23
Despite its prevalence in certain regions, S. suis infections are frequently underreported due to diagnostic challenges and limited awareness.2 This study presents the clinical characteristics of meningitis cases in which patients experienced sudden deafness related to S. suis, as observed in a tertiary hospital with limited resources.
Materials and Methods
Study design
This study is a retrospective analysis of the medical records of patients diagnosed with suspected S. suis-associated meningitis between 2022 and 2024 who were admitted to Sanjiwani Hospital, a tertiary referral hospital in Bali with 241 beds. The inclusion criteria for this study were primarily based on clinical diagnosis and comprised adult patients aged 18 years or older who had been diagnosed with suspected S. suis-associated meningitis. The clinical diagnosis of meningitis was based on the classic triad of fever, neck stiffness, and altered mental status or headache.25 Incomplete medical record data were excluded from the study. In this study, cases were defined as confirmed S. suis-associated meningitis if the patient had a positive blood culture for Streptococcus suis. Cases were classified as probable S. suis infection if the patient had a documented history of consuming raw pork, a known risk factor for the disease. The term meningitis cases refers to all instances in which S. suis was suspected as the cause of meningitis but not confirmed through blood culture.
Laboratory and imaging investigation
The study faced limitations, particularly regarding the reliance on selective blood cultures. This approach can introduce bias, as not all patients with suspected meningitis may have had blood cultures performed, potentially leading to an underestimation of the true incidence of S. suis-associated meningitis. Additionally, the absence of cerebrospinal fluid (CSF) analysis limits our ability to definitively confirm meningitis. CSF testing is crucial for distinguishing between different types of meningitis and understanding the complete clinical picture. The lack of CSF data may hinder the assessment of the severity and specific characteristics of the meningitis cases.
At the tertiary hospital, lumbar puncture to obtain cerebrospinal fluid (CSF) is often deferred due to concerns about the risk of cerebral herniation. Additionally, the high cost of CSF analysis prevented its routine use to confirm the cause of meningitis. As the number of probable S. suis-related meningitis cases increases, the hospital has implemented a non-invasive diagnostic tool, blood culture, for meningitis cases, including those with or without sudden deafness.
Blood samples from patients exhibiting clinical signs of meningitis were introduced onto 5% defibrinated sheep blood agar plates (DSBAPs) and incubated in 5% CO2 at 37°C for 18–24 hours.26 According to the Clinical and Laboratory Standards Institute guidelines, the resulting colonies were isolated for identification and drug susceptibility testing using a fully automatic VITEK 2 COMPACT system.27 Selected colonies were cultured in tryptic soy broth, incubated at 37°C for 18–24 hours, and preserved at –80°C in 50% glycerol. Next, the S. suis isolates were cultured on DSBAPs and incubated in 5% CO2 at 37°C for 18–24 h for further analysis.
The imaging approach used in this study was head computed tomography (CT), although CT is not sensitive to detecting meningitis.
Ethical considerations
The study protocol was reviewed and approved by the Research Ethics Committee of Sanjiwani Hospital, Bali (document number 420/40268/RSU). This study is a retrospective analysis of hospital medical records; therefore, informed consent was not required.
Statistical analysis
SPSS Version 25 (SPSS Inc., Chicago, IL) was used to analyze the collected statistical data. A probability value of p ≤ 0.05 was considered statistically significant. Numerical data were compared using the independent-samples t-test or the Mann–Whitney test, depending on their distribution. The Chi-Square test was used for categorical data.
Results
We identified 74 suspected S. suis-associated meningitis cases from hospital medical records of patients admitted between 2022 and 2024. Of these, 67 cases had complete data for analysis (Table 1). The first patients with meningitis to present with sudden deafness were admitted in April 2022. Among the 67 cases, 34 patients presented with deafness, representing 50.7%. There were 30 cases of probable S. suis-associated meningitis (44.8%) and 7 cases of confirmed S. suis-associated meningitis (10.4%) (Table 1). Therefore, the overall number of S. suis-associated meningitis cases was 31 out of 67 (46.3%), including one culture-positive patient with no history of consuming raw pork. Statistical analysis revealed no significant association between raw pork consumption and the presentation of deafness (p=0.151; odds ratio =1.538, 95% CI=0.584-4.055).
Table 1: Demographic and clinical characteristics of subjects.
| Variables | Total (N =67) | Meningitis with sudden deafness
(N=34) |
Meningitis without sudden deafness
(N=33) |
p |
| Sex, N, (%)Male
Female |
52 (77.7)
15 (22.4) |
27 (51.9)
7 (46.7) |
25 (48.1)
8 (53.3) |
0.474# |
| Age, (year), mean ±SD | 53.8 (14.9) | 53.7 (16.9) | 53.8 (12.9) | 0.965$ |
| Fever onset (day), mean ±SD | 2.8 (1.4) | 2.9 (1.6) | 2.6 (1.3) | 0.335$ |
| Length of stay (days), mean ±SD | 11.4 (3.8) | 11.4 (3.4) | 11.4 (4.3) | 0.044$ |
| Raw pork consumption, N (%) | 30 (44.8) | 17 (56.7) | 13 (43.3) | 0.151# |
| Sign and symptoms, N, (%)Headache
Convulsion Ataxia Vomiting Hemiparesis Neck rigidity Mental state disturbance Septic arthritis |
66 (98.5)
8 (11.9) 4 (58.1) 63 (94) 6 (9) 67 (100) 43 (64.2) 9 (13.4) |
33 (50)
4 (50) 2 (50) 31 (49.2) 2 (33.3) 34 (50.7) 19 (44.2) 4 (44.4) |
33 (50)
4 (50) 2 (50) 32 (58.8) 4 (66.7) 33 (49.3) 24 (55.8) 5 (55.6) |
0.507*
0.628* 0.682* 0.318* 0.322* NA 0.151# 0.480* |
| ICU treatment | 3 ( 4.5) | 0 | 3 (100) | 0.114* |
| OutcomeSurvival | 64 (95.5) | 33 (51.6) | 31 (48.4) | 0.489* |
Note: $ The independent t-test was used to compare the groups.
# The chi-square test was used to compare the groups.
*Fisher’s exact test; significance value at p < 0.05. NA: not assessed.
All samples exhibited fever and neck stiffness, with both systems present in 100% of cases. Among the subjects with clinical meningitis presenting with sudden deafness, only 56.7% reported consuming raw pork blood. The chief complaints of the subjects included decreased consciousness (52.2%), severe headache (28.4%), fever (13.4%), altered mental status (4%), convulsion (1.5%), and hearing loss (1%). Three subjects (4.5%) experienced both septic arthritis and deafness. Septic shock occurred in 11 subjects (16.4%), 3 of whom (4.5%) died. In total, septic shock complications occurred in 3 patients in the group with meningitis and sudden deafness (4.5%). Among these subjects with clinical meningitis and sudden deafness, mortality due to septic shock was one case (1.5%). Consequently, the mortality rate for S. suis-associated meningitis was also 1 (1.5%).
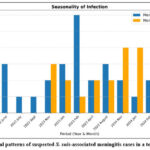 |
Figure 1: Seasonal patterns of suspected S. suis-associated meningitis cases in a tertiary hospital. |
The first suspected cases of S. suis-associated meningitis patients presenting with sudden deafness were reported in April 2022 (Figure 1). Most meningitis cases were reported in 2024. In 2022, the total number of cases with sudden deafness was 6 (9%), followed by 4 (6%) in 2023 and an enormous increase to 24 (35.8%) in 2024. The highest number of meningitis cases with sudden deafness was reported in January and February 2024.
This study did not evaluate the CSF analysis or CSF culture results. The only non-invasive tool for diagnosing S. suis-associated meningitis was blood culture, which was performed selectively due to cost considerations. The identification of S. suis in the blood of patients with clinical meningitis was confirmed with the diagnosis of S. suis-associated meningitis.28 The blood culture results are described in Figure 2. Blood cultures were performed in only 10 patients, revealing S. suis bacteremia in 7 of 67 (10.4%) cases. Additionally, 1 case (1.5%) was positive for Staphylococcus bacteremia, while 2 out of 67 (3%) exhibited no growth. In this study, S. suis bacteremia caused sudden deafness in 6 cases (85.7%), with 83.3% occurring in males.
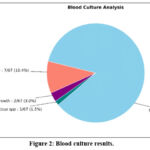 |
Figure 2: Blood culture results. |
The main findings of the computed tomography (CT) scans were typical, with 58.9% showing no meningeal enhancement. Others reveal meningeal enhancement or nonspecific findings such as hypodense lesions. The laboratory findings are summarized in Table 2. No significant differences were observed in laboratory results between those presenting with meningitis and sudden deafness and those without sudden deafness, except for the mean WBC count, which was higher in the group with sudden deafness (14.3 vs. 10.9 × 10^3/mL, p = 0.039).
Table 2: Laboratory results of the subjects at admission.
| Total(N=67) | Meningitis with sudden deafness(N=34) | Meningitis without sudden deafness(N=33) | p | |
| CBC at admissionHemoglobin (g/dL)
Hematocrit (%) Leucocyte (x103/mL) Thrombocyte (x103/mL) |
13.3 (1.7)a
39.2 (5)a 13.6 (6.2)a 305 (125.3)a |
13.3 ± 1.9a
39.2 ± 5.7a 14.3 (7-38.2)b 302.9 ±130.1a |
13.3 ± 1.5a
39.2 ± 4.3a 10.9 (5.3-26.3)b 307.2 ± 122a |
NA
NA 0.039# 0.892* |
| AST, IU/L | 47.6 (48)a | 37.5 (11-330)b | 33 (4-133)b | 0.261# |
| ALT, IU/L | 45.9 (37)a | 36 (19-224)b | 32 (13-85)b | 0.961 |
| BUN, mg/dL | 51 (9-211)a | 36.2 (22.9-188.2)b | 34.2 (16.1-65.2)b | 0.363# |
| SC, mg/dL | 41 (23.5)a | 0.7 (0.2-4.5)b | 0.7 (0.3-1.2)b | NA |
| Blood sugar, mg/dL | 138.2 (47.3)a | 125 (93-199)b | 123 (84-454)b | 0.337# |
Note: a data presented as mean (±SD), b data presented as median (minimum–maximum).
*Independent t-test. #Mann—Whitney test.
Values are significant at p < 0.05.
NA: not assessed.
All patients were treated with 2 grams of intravenous ceftriaxone every 12 hours for 10 to 14 days, in addition to dexamethasone at a dose of 10 mg intravenously every six hours for four days. Among the 67 patients with meningitis, 95.5% survived, resulting in an overall mortality rate of 4.5%.
Discussion
The diagnosis of S. suis-related meningitis does not rely on detecting S. suis in the cerebrospinal fluid (CSF); the presence of S. suis bacteremia can also confirm the diagnosis.10,28 The consumption of raw pork in Bali raises concerns due to its association with S. suis bacteremia. However, the current study found no significant link between raw pork consumption and meningitis with sudden deafness, with an odds ratio of 1.538 and a wide confidence interval indicating non-significance. Despite this, the elevated odds ratio suggests a potential risk and warrants further investigation, especially given the small sample size of 67 participants.
The odds ratio for acquiring an S. suis infection after consuming raw pork is 4.63 (95% CI 2.94–7.29). In comparison, the odds ratio for infection from exposure to a pig is 4.01 (95% CI 2.61–6.15), while for consuming pork, it is 3.03 (95% CI 1.61–5.68).2 Furthermore, a study conducted in Bali between 2014 and 2017 reported that the incidence of S. suis-associated meningitis reached 62%.22
Clinical manifestations of meningitis typically include headaches, neck stiffness, vomiting, and fever.29 In adults, common symptoms include neck stiffness, fever, and altered mental state.25 This study reported that all subjects presented with fever and signs of meningism, alongside other symptoms such as convulsions, ataxia, and severe headaches.
Laboratory findings are crucial for diagnosing bacterial meningitis; elevated white blood cell (WBC) counts indicate an inflammatory response to bacterial infection. The analysis revealed a significant difference in median WBC levels between cases with sudden deafness and those without. In one reported case of S. suis meningitis, the WBC count was exceptionally high at 27.28 × 10^9/L, with 94.20% neutrophils.30 This elevation in WBCs is linked to the host’s inflammatory response to S. suis, which stimulates macrophages to secrete pro-inflammatory cytokines such as IL-1β, TNF-α, IL-6, and IL-8, which are essential for recruiting and activating leukocyte populations.31 Indeed, the capsular material of S. suis significantly enhances prostaglandin E2 and matrix metalloproteinase nine production in macrophages, contributing to tissue destruction and blood–brain barrier disruption.32
The diagnosis of meningitis in this study was based on the classic triad of fever, neck rigidity, and altered mental state or headache. However, the high mortality rate of bacterial meningitis, ranging from 10% to 20% in developed countries, underscores the need for robust diagnostic systems to enable timely treatment. The sensitivity of clinical judgment in diagnosing bacterial meningitis varies, with the classic triad showing only 40% to 50% sensitivity.33
In this study, blood cultures were performed in only 10 cases, yielding 70% identification of S. suis, 10% for Staphylococcus spp., and no growth in 20%. The misidentification of S. suis as viridans streptococci has been documented elsewhere, and follow-up investigations have confirmed that a significant percentage of viridans cases were S. suis.34 Other studies have confirmed S. suis-associated meningitis through blood culture, cerebrospinal fluid (CSF) culture, real-time polymerase chain reaction (RT-PCR), or a combination of these methods.28
CSF analysis plays a crucial role in diagnosing bacterial meningitis by assessing cytochemical characteristics, including white blood cell (WBC) count, protein concentration, and glucose levels. An elevated CSF WBC count exceeding 10 cells/µL is a standard diagnostic threshold.35 A predominance of neutrophils suggests bacterial meningitis, whereas lymphocyte dominance indicates a viral infection. A CSF glucose level below 60 mg/dL or a CSF/serum glucose ratio below 0.44 generally indicates bacterial meningitis.36 Elevated protein levels are also typically observed in bacterial cases, while a CSF lactate level exceeding 30 mg/dL indicates bacterial meningitis.37,38 These findings underscore the utility of CSF analysis as a practical tool in resource-limited settings where advanced diagnostic facilities may not be available. Further analysis of CSF includes Gram staining, culture, and/or sensitivity testing. Gram staining may detect bacteria in 50-80% of cases, while culture yields a positive result in more than 80%; however, a study reported low sensitivity in CSF cultures.39 Accurately identifying the causative organism without culture results can be challenging, potentially leading to incorrect diagnoses or ineffective treatments.
Given the importance of CSF analysis, the absence of such assessments and RT-PCR testing in this study introduces several confounding factors. Without comprehensive CSF analysis, accurately confirming meningitis becomes challenging, potentially leading to the misclassification of other conditions. The symptoms of meningitis can overlap with those of various illnesses, complicating the clinical picture. Additionally, without conclusive CSF investigations, it is not easy to assess the impact of co-infections or other pathogens that may contribute to clinical outcomes.
The complications observed in this study included septic shock, arthritis, and sudden deafness. Previous research has documented additional complications, such as renal or liver involvement, acute respiratory distress syndrome, endophthalmitis, spinal abscess, and skin rashes.28 Hearing loss and vestibular dysfunction are common sequelae; one study reported that up to 75% of subjects with S. suis meningitis experienced hearing loss.10,40 The primary mechanism linking S. suis meningitis to hearing loss is suppurative labyrinthitis, which can damage the cochlea and auditory nerve. High bacterial loads can increase the permeability of the blood–labyrinth barrier, allowing bacteria and inflammatory cells to enter the cochlea and causing direct damage to the inner ear’s sensory cells.41
The case fatality rate (CFR) for S. suis-associated meningitis in this study was 1.5%. In contrast, previous studies in Bali reported a CFR of 11.4% among confirmed cases. In regions like Togo, within the African meningitis belt, the CFR was reported at 7%, highlighting the variability in outcomes depending on geographic and healthcare factors.5 An elevated CFR associated with S. suis infection can be attributed to rapid disease progression and nonspecific early symptoms, complicating timely diagnosis and treatment.42,43 The inflammatory response induced by S. suis, including the generation of IL-17A, can lead to significant tissue damage and disruption of the blood–brain barrier, facilitating nervous system infection.44
Although the results did not indicate a significant association between raw pork consumption and S. suis infection, they underscore the public health risks in regions with cultural practices involving undercooked pork. We advocate for improved diagnostic approaches and effective public health strategies to mitigate the rising incidence of S. suis infections, particularly in high-risk populations. By fostering a deeper understanding of the clinical manifestations and risks associated with this infection, this study aims to improve health outcomes and prevention efforts in affected communities.
Beyond acute deafness, emerging evidence indicates that S. suis meningitis survivors frequently experience persistent neurological deficits—including vestibular dysfunction, cognitive impairment, and gait disturbances—that are often underreported in acute care settings. A long-term follow-up study in Thailand reported vestibular impairment in 50 percent of meningitis survivors after a mean of 17 months,45,46 and a Vietnamese case–control study found elevated vestibular sequelae persisting through 9 months post-discharge (prevalence ratio ~2.4).10 In our Bali cohort, high rates of mental state changes (64 %) and ataxia (58%) suggest similar long-term risks, yet rehabilitation access remains scarce in this resource-limited setting. These findings underscore an urgent need for: (1) standardized neurocognitive and vestibular screening protocols at discharge, (2) vestibular and gait rehabilitation programs (especially for cochlear-implant candidates), and (3) community-based support networks for survivors coping with chronic disability—a critical and currently overlooked component of zoonotic meningitis management.
Since this study is retrospective, its limitations primarily stem from a lack of critical data, such as CSF analysis and culture results, and from the limited availability of other tools for detecting S. suis by RT-PCR. As it is retrospective, the study relies on the accuracy and completeness of the medical record data, which may introduce bias. Furthermore, a small sample size may limit the generalizability of the findings to the population. We included all subjects diagnosed with suspected S. suis-associated meningitis during the designated study period who had complete data. The generalizability of our study is limited due to demographic and geographic factors. The study population is predominantly adults, which may limit the applicability of the findings to other age groups with distinct immune responses and clinical presentations. Additionally, the geographic limitations of the current study, in which the risk factors for S. suis infection — such as a dense pig population and specific cultural attitudes — are prevalent, may also influence the results.
Conclusions
The study indicates a significant public health concern in Bali due to the high incidence of S. suis bacteremia, particularly influenced by the region’s pig density. To mitigate infection risks, preventive measures such as safe pig slaughter and proper cooking are essential. Future research should focus on diverse populations and prospective cohort studies to enhance data quality and enable advanced diagnostic techniques, such as next-generation sequencing and RT-PCR testing. Additionally, examining the link between dietary practices and infection rates will help develop effective public health strategies. A comprehensive understanding of S. suis epidemiology is crucial for addressing this emerging public health threat.
Acknowledgement
We gratefully acknowledge all participants in the study, the nurses, and the research team in the Faculty of Medicine and Health Sciences at Universitas Warmadewa for their support.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any materials that require ethical approval.
Informed Consent Statement
This study did not involve human participants; therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Authors’ contributions
- Sri Masyeni: the conceptualization, supervision, and writing of the original draft of the manuscript.
- Ratna Kartika Dewi and Arya Giri Prebawa: data curation,
- Pande Ayu Naya Kasih Permatananda : formal analysis.
- Saraswati Laksmi Dewi: methodology.
- Erni Juwita Nelwan, Sri Masyeni, Rois M Fatawy: supervision, review, and editing of the manuscript
- Kadek Suryawan and Rois M Fatawy: writing the original draft. All authors read and approved the final version of the manuscript.
References
- Hlebowicz M, Jakubowski P ST. Streptococcus suis Meningitis: Epidemiology, Clinical Presentation and Treatment. Vector Borne Zoonotic Dis. 2019;19(8):557-562.
CrossRef - Rayanakorn A, Goh BH, Lee LH, Khan TM, Saokaew S. Risk factors for Streptococcus suis infection: A systematic review and meta-analysis. Sci Rep. 2018;8(1):1-9.
CrossRef - Neila-Ibáñez C, Napp S, Pailler-García L, et al. Risk factors associated with Streptococcus suis cases on pig farms in Spain. Vet Rec. 2023;193(5):e3056.
CrossRef - van Samkar A, Brouwer MC, Schultsz C, van der Ende A, van de Beek D. Streptococcus suis Meningitis: A Systematic Review and Meta-analysis. PLoS Negl Trop Dis. 2015;9(10):1-10.
CrossRef - Tall H, Njanpop-Lafourcade BM, Mounkoro D, et al. Identification of Streptococcus suis Meningitis through Population-Based Surveillance, Togo, 2010–2014. Emerg Infect Dis. 2016;22(7):1262-1264.
CrossRef - Rieckmann K, Pendzialek SM, Vahlenkamp T, Baums CG. A critical review speculating on the protective efficacies of autogenous Streptococcus suis bacterins as used in Europe. Porc Heal Manag. 2020;6(1):12.
CrossRef - van de Beek D, Spanjaard L, de Gans J. Streptococcus suis meningitis in the Netherlands. J Infect. 2008;57(2):158-161.
CrossRef - Dutkiewicz J, Sroka J, Zając V, et al. Streptococcus suis: a re-emerging pathogen associated with occupational exposure to pigs or pork products. Part I – Epidemiology. Ann Agric Environ Med. 2017;24(4):683-695.
CrossRef - Praphasiri P, Owusu JT, Thammathitiwat S, et al. Streptococcus suis Infection in Hospitalized Patients, Nakhon Phanom Province, Thailand. Emerg Infect Dis. 2015;21(2):345-348.
CrossRef - Huong VTL, Ha N, Huy NT, et al. Epidemiology, Clinical Manifestations, and Outcomes of Streptococcus suis Infection in Humans. Emerg Infect Dis. 2014;20(7).
CrossRef - MA E, CHUNG PH, SO T, et al. Streptococcus suis infection in Hong Kong: an emerging infectious disease? Epidemiol Infect. 2008;136(12):1691-1697.
CrossRef - Arenas J, Bossers-de Vries R, Harders-Westerveen J, et al. In vivo transcriptomes of Streptococcus suis reveal genes required for niche-specific adaptation and pathogenesis. Virulence. 2019;10(1):334-351.
CrossRef - Li G, Wang G, Si X, et al. Inhibition of suilysin activity and inflammation by myricetin attenuates Streptococcus suis virulence. Life Sci. 2019;223:62-68.
CrossRef - Zhu Y, Dong W, Ma J, et al. Comparative genetic analyses provide clues about capsule switching in Streptococcus suis 2 strains with different virulence levels and genetic backgrounds. Microbiol Res. 2021;250:126814.
CrossRef - Kerdsin A, Segura M, Fittipaldi N, Gottschalk M. Sociocultural Factors Influencing Human Streptococcus suis Disease in Southeast Asia. Foods. 2022;11(9):1190.
CrossRef - Zheng C, Wei M, Jia M, Cao M. Involvement of Various Enzymes in the Physiology and Pathogenesis of Streptococcus suis. Vet Sci. 2020;7(4):143.
CrossRef - Auger JP, Payen S, Roy D, Dumesnil A, Segura M, Gottschalk M. Interactions of Streptococcus suis serotype 9 with host cells and role of the capsular polysaccharide: Comparison with serotypes 2 and 14. Singh AK, ed. PLoS One. 2019;14(10):e0223864.
CrossRef - Domado AM, Itable J. Streptococcus suis: Bacteremia presenting with fever, rashes, arthritis and neurologic deficits. Androl Gynecol Curr Res. 2017;05(03).
CrossRef - Li K, Li S, Hong J, et al. Meningitis and sepsis caused by Streptococcus suis in an elderly woman: A CARE-compliant case report. Medicine (Baltimore). 2023;102(43):e35780.
CrossRef - He N, Li H, Zhang X. Human Streptococcus suis Infection. In: Radiology of Infectious Diseases: Volume 2. Springer Netherlands; 2015:113-119.
CrossRef - Ho DTN, Le TPT, Wolbers M, et al. Risk Factors of Streptococcus suis Infection in Vietnam. A Case-Control Study. Smith T, ed. PLoS One. 2011;6(3):e17604.
CrossRef - Susilawathi NM, Tarini NMA, Fatmawati NND, et al. Streptococcus suis –Associated Meningitis, Bali, Indonesia, 2014–2017. Emerg Infect Dis. 2019;25(12):2235-2242.
CrossRef - Aryasa IA, Widiasari NPA, Susilawathi NM, et al. <em>Streptococcus suis</em> meningitis related to processing and consuming raw pork during Balinese tradition, <em>Mebat</em>. Med J Indones. 2020;29(1):88-92.
CrossRef - Tarini NMA, Setiabudy M, Susilawathi N, et al. Misidentification of S. suis as a Zoonotic Agent. Open Access Maced J Med Sci. 2019;7(14):2309-2312.
CrossRef - Scarborough M, Thwaites GE. The diagnosis and management of acute bacterial meningitis in resource-poor settings. Lancet Neurol. 2008;7(7):637-648.
CrossRef - Lehman DC, Mahon CR SK. Textbook of Diagnostic Microbiology.; 2016.
- Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Testing; Twenty-Fifth Informational Supplement (M100-S27). Wayne (PA): The Institute; 2017.
- Wertheim HFL, Nguyen HN, Taylor W, et al. Streptococcus suis, an Important Cause of Adult Bacterial Meningitis in Northern Vietnam. Ratner AJ, ed. PLoS One. 2009;4(6):e5973.
CrossRef - Sharma RR, Sharma A. Meningitis: Current Understanding and Management. In: The Microbiology of Central Nervous System Infections. Elsevier; 2018:3-27.
CrossRef - Liu Y, Kou Z, Wang X, Chen S, Li R, Wang Q. Case report: One human Streptococcus suis infection in Shandong Province, China. Medicine (Baltimore). 2023;102(14):e33491.
CrossRef - Tanabe S ichi, Gottschalk M, Grenier D. Hemoglobin and Streptococcus suis cell wall act in synergy to potentiate the inflammatory response of monocyte-derived macrophages. Innate Immun. 2008;14(6):357-363.
CrossRef - Jobin MC, Gottschalk M, Grenier D. Upregulation of prostaglandin E2 and matrix metalloproteinase 9 production by human macrophage-like cells: Synergistic effect of capsular material and cell wall from Streptococcus suis. Microb Pathog. 2006;40(1):29-34.
CrossRef - Viallon A, Bothelo-Nevers E, Zeni F. Clinical decision rules for acute bacterial meningitis: current insights. Open Access Emerg Med. Published online April 2016:7.
CrossRef - Fongcom A, Pruksakorn S, Netsirisawan P, Pongprasert R, Onsibud P. Streptococcus suis infection: a prospective study in northern Thailand. Southeast Asian J Trop Med Public Health. 2009;40(3):511-517.
- Lucke IM, Peric S, van Lieverloo GGA, et al. Elevated leukocyte count in cerebrospinal fluid of patients with chronic inflammatory demyelinating polyneuropathy. J Peripher Nerv Syst. 2018;23(1):49-54.
CrossRef - Zeinalizadeh M, Shadkam M, Afarinesh Khaki P, Abdollahi A, Douraghi M, Salehi M. Cerebrospinal Fluid Analysis in Patients with Post-neurosurgical Procedures: Meningitis vs. Non-meningitis. Iran J Pathol. 2024;19(3):342-347.
CrossRef - O’Leary C, Pittet LF, Beaumont R, et al. Predictive performance of cerebrospinal fluid parameters for diagnosis of meningitis in infants: a cohort study. Arch Dis Child. 2025;110(3):209-215.
CrossRef - Domingues RB, Fernandes GBP, Leite FBV de M, Senne C. Performance of lactate in discriminating bacterial meningitis from enteroviral meningitis. Rev Inst Med Trop Sao Paulo. 2019;61.
CrossRef - Manning L, Laman M, Mare T, Hwaiwhanje I, Siba P, Davis TME. Accuracy of cerebrospinal leucocyte count, protein and culture for the diagnosis of acute bacterial meningitis: a comparative study using <scp>B</scp> ayesian latent class analysis. Trop Med Int Heal. 2014;19(12):1520-1524.
CrossRef - Kerdsin A. Human Streptococcus suis Infections in Thailand: Epidemiology, Clinical Features, Genotypes, and Susceptibility. Trop Med Infect Dis. 2022;7(11):359.
CrossRef - Domínguez-Punaro MC, Koedel U, Hoegen T, Demel C, Klein M, Gottschalk M. Severe cochlear inflammation and vestibular syndrome in an experimental model of Streptococcus suis infection in mice. Eur J Clin Microbiol Infect Dis. 2012;31(9):2391-2400.
CrossRef - Xia X, Wang X, Wei X, Jiang J, Hu J. Methods for the detection and characterization of Streptococcus suis: from conventional bacterial culture methods to immunosensors. Antonie Van Leeuwenhoek. 2018;111(12):2233-2247.
CrossRef - Ágoston Z, Terhes G, Hannauer P, Gajdács M, Urbán E. Fatal case of bacteremia caused by Streptococcus suis in a splenectomized man and a review of the European literature. Acta Microbiol Immunol Hung. 2020;67(3):148-155.
CrossRef - Xu L, Lu X, Xiao P, et al. Interleukin-17A Contributed to the Damage of Blood-CNS Barriers During Streptococcus suis Meningitis. Mol Neurobiol. 2022;59(4):2116-2128.
CrossRef - Wangkaew S, Chaiwarith R, Tharavichitkul P et al. Streptococcus suis infection: a series of 41 cases from Chiang Mai University Hospital. 2006;52(6):455-460.
CrossRef - Navacharoen N, Chantharochavong V, Hanprasertpong C et al. Hearing and vestibular loss in Streptococcus suis infection from swine and traditional raw pork exposure in northern Thailand. J Laringol Otol. 2009;123(8):857-862.
CrossRef







