Manuscript accepted on :12-12-2025
Published online on: 22-12-2025
Plagiarism Check: Yes
Reviewed by: Dr. Karveer Aghade
Second Review by: Dr. Priya Gayathri
Final Approval by: Dr. Eman Refaat Youness
Ashwaq Taleb Kareem1* and Samer Khalid Ali2
and Samer Khalid Ali2
1Department of Pharmacognosy, College of Pharmacy, Thi-Qar University, Thi-Qar, Iraq.
2Pharmaceutic Department, College of Pharmacy, Al-Ayen Iraqi University, AUIQ, An Nasiriyah, Iraq
Corresponding Author E-mail:ashwaq.t.kareem@utq.edu.iq
DOI : https://dx.doi.org/10.13005/bpj/3280
Abstract
Parsley leaves (Petroselinum crispum) biennial herb native to the Mediterranean has diverse bioactive phytochemicals, like flavonoids and phenolic acids that known for their potent antioxidant and cytotoxic properties. This study aimed to isolate two phenolic compounds, Apigenin and Syringic acid from ethyl acetate fraction of parsley leaves extract and evaluate their cytotoxic activity against A375 cell line compared to normal cells. Ultrasonication was used to extract the active compounds using 90% ethanol, and PHPLC was used to isolate the two phenolic compounds. FT-IR was used to categorize the active functional groups comprising the two isolated phenolic compounds. The results showed the isolation of syringic acid and apigenin, apigenin exhibited cytotoxic activity with IC50% 40.8 μg /ml and Syringic acid exhibited cytotoxic activity with IC50 values 54.5 μg /ml, Apigenin showing a stronger cytotoxic activity than Syringic acid. Both isolated phenolic compounds exhibited strong cytotoxic activity depend on increasing concentration of each compound, and Apigenin showing particularly high cytotoxic activity against A375 cell line.
Keywords
A375cell line; Apigenin; Cancer; Parsley; Syringic acid
Download this article as:| Copy the following to cite this article: Kareem A. T, Ali S. K. Comparative Study on the Anticancer Potential of Apigenin and Syringic Acid Isolated from Parsley Leaves Against A375 Cell Line. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Kareem A. T, Ali S. K. Comparative Study on the Anticancer Potential of Apigenin and Syringic Acid Isolated from Parsley Leaves Against A375 Cell Line. Biomed Pharmacol J 2025;18(4). Available from: https://bit.ly/45lh7Uk |
Introduction
Parsley (Petroselinum crispum L.) is a widely cultivated biennial herb native to the Mediterranean, thriving in rich, well-drained soils under full or partial sunlight, and often grown from seeds planted in early spring.1 This vibrant green herb is celebrated for its diverse phytochemical profile, including abundant flavonoids like apigenin and luteolin glycosides, volatile oils such as apiol and myristicin, and phenolic acids like syringic and ferulic acids, which together confer potent antioxidant and anti-inflammatory properties.2
Traditionally, parsley has been used across cultures as a digestive aid, mild diuretic, breath freshener, and remedy for kidney or bladder issues, while in folk medicine it has also been employed to ease menstrual discomfort and reduce swelling, making it a plant of both culinary and medicinal significance .3
Cancer is a complex disease assigned by the unrestrained growth of abnormal cells, which can infiltrate neighboring tissues and spread to distant sites through metastasis.4 Its development is driven by a combination of genetic mutations, epigenetic modifications, and disturbances in key signaling pathways that regulate cellular proliferation, programmed cell death, and differentiation .5. Although conventional treatments like surgery, chemotherapy, and radiation therapy have advanced, they still face significant drawbacks, such as serious side effects, the emergence of treatment resistance, and a lack of selectivity for cancer cells over healthy ones.6,7 As a result, there is increasing interest in identifying new therapeutic agents, particularly natural compounds with potential anticancer properties, due to their promise of improved safety and effectiveness.8
Apigenin is a naturally occurring flavonoid found abundantly in many fruits, vegetables, and herbs. It is especially concentrated in parsley, chamomile, celery, and oranges, making these common dietary sources of this bioactive compound .9,10 Traditionally, plants rich in apigenin have been used in folk medicine across various cultures to treat conditions such as inflammation, anxiety, insomnia, digestive issues, and skin disorders.8 Chamomile tea, for example, is widely consumed as a calming remedy and owes much of its soothing effect to its apigenin content.11,12
Beyond its traditional applications, modern pharmacological research has revealed that apigenin possesses a varied spectrum of biological activities, as antioxidant, anti-inflammatory, antimicrobial, neuroprotective, and notably, anticancer activities.13–15 Studies have shown that apigenin can inhibit the growth of cancer cells, induce apoptosis, and suppress tumor angiogenesis and metastasis by modulating various molecular pathways involved in cell cycle regulation and survival .14,15
Because of these promising properties, apigenin is increasingly being explored as a potential therapeutic agent in cancer prevention and treatment strategies.
Syringic acid is a naturally occurring phenolic acid that belongs to the class of hydroxybenzoic acids. It is widely distributed in the plant kingdom and can be found in various fruits, vegetables, and medicinal herbs, including grapes, olives, dates, honey, and spices like turmeric and parsley. 16,17 This compound is also abundant in the bark and heartwood of certain trees, such as maple and walnut, as well as in red wine and other plant-derived foods and beverages.16-18
In traditional medicine, syringic acid-rich plants have long been valued for their health-promoting properties. Remedies containing syringic acid sources have been used for centuries to manage inflammation, pain, infections, and digestive problems in different cultures around the world. 19,20 These traditional uses laid the foundation for scientific investigations into its potential health benefits.Modern pharmacological research has highlighted syringic acid’s diverse biological activities, including antioxidant, anti-inflammatory, antimicrobial, antidiabetic, neuroprotective, and anticancer effects.21–23
Studies show that syringic acid can neutralize free radicals, reduce inflammatory responses, modulate key metabolic enzymes, and even induce cancer cell apoptosis by influencing signaling pathways related to cell proliferation and survival.22,23 These findings support syringic acid’s promise as a natural therapeutic agent for various chronic diseases, including cancer and metabolic disorders. In this study, we hope to isolate two phenolic compounds Apigenin and Syringic acid from ethyl acetate fraction of parsley leaves extract and compared their cytotoxic effects on A375 cell line.
Materials and Methods
Plant Collection and extraction
Parsley leaves were harvested from a home garden during the summer season and experienced authentication directed by expert Prof. Sukaena Abass. The collected leaves were thoroughly washed, shade-dried, and then mechanically ground into a fine powder. For extraction, 20 g of the powdered parsley leaves were mixed with 150 mL of 90% ethanol and subjected to ultrasonic-assisted extraction using an ultrasonic bath sonicator operating at ( 40 kHz and 45 °C for 60 minutes) which relies on the principle of acoustic resonance, where small bubbles form and then burst near the plant cell wall, leading to its destruction and the release of active substances into the solution. These resulting cavities are a consequence of the conversion of electrical energy into high-frequency sound waves transmitted through the liquid inside the tank. The resulting mixture was filtered through Whatman filter paper. The filtrate was evaporated to dryness, and the dried extract was subsequently suspended in a biphasic system comprising 150 mL of distilled water and 150 mL of ethyl acetate. The ethyl acetate fraction (EtOAc) was then subjected to Preparative High-performance Liquid Chromatography (PHPLC) for the isolation of phenolic compounds.24
Preparative High-performance Liquid Chromatography (PHPLC)Conditions
Quantitative analysis of phenolic compounds in the EtOAc was carried out using PHPLC. The phenolic constituents expected in the fractions were identified by comparing their retention times with those of standard compounds, namely apigenin and syringic acid. The chromatographic separation employed a mobile phase consisting of 1% aqueous acetic acid (solvent A) and acetonitrile (solvent B), with the flow rate set at 3 mL/min and an injection volume of 300 μL. A gradient elution was applied, starting with 10% B and increasing linearly to 40% B over 28 min.then from 40% to 60% B by 39 min. and finally from 60% to 90% B by 50 min.After reaching 90% B, the gradient was returned to the initial composition of 10% B (B:A = 10:90) at 55 min.and the system was allowed to equilibrate for an additional 10 min. before concluding the run.25,26
Analysis by Fourier transform infrared (FT-IR)
FT-IR spectra of isolated compounds were subjected in the FTIR spectrometer (Shimadzu)in the range of 500 to 4000 cm–1wave number
Materials
Apigenin and syringic acid St. Were from Sigma-Aldrich, A375 cell lines from Center for Biotechnology Research at Al-Nahrain University.
Cell Line Maintenance
The procedure of work was conducted for cell line maintenance according toFreshney RI (27).
MTT Assay
After treating the cells with apigenin and syringic acid for 72 hours, the culture medium in each well was gently removed. The wells were rinsed twice with phosphate-buffered saline to clear any residual compounds. Next, 100 μL of fresh cell culture medium and 15 μL of MTT staining solution were added to each well. Following a 4-hour incubation period, 100 μL of stop solution was carefully introduced to halt the MTT reaction.28 The plates were then kept in the incubator overnight to allow complete solubilization of the formazan crystals. Finally, cell viability was assessed by measuring at 570 nm optical density using a spectrophotometer ,Experiment was repeated three times .
Statistical analysis
Data analysis was performed with GraphPad Prism version 6, and results were appeared as the mean ± standard deviation depend on three independent replicates. Statistical comparisons between groups were made using an unpaired t-test, with differences considered statistically significant at P < 0.05.
Results
Extraction by Ultrasonic bath sonicator
Ultrasound-assisted extraction was employed as the method of choice for processing the parsley leaves, yielding approximately 11 g of crude extract from 20 g of dried leaf material, representing a notably high extraction efficiency.
Isolation of Apigenin and syringic acid by PHPLC
Apigenin and Syringic acid were isolated by PHPLC using the chromatographic settings that cited in the earlier paragraphs, as in Figures. from 1 to 5
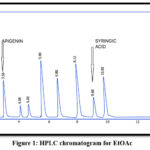 |
Figure 1: HPLC chromatogram for EtOAc |
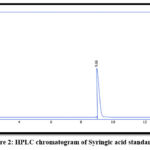 |
Figure 2: HPLC chromatogram of Syringic acid standard.
|
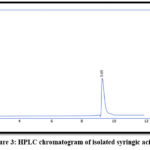 |
Figure 3: HPLC chromatogram of isolated syringic acid |
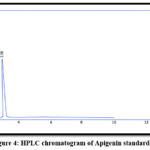 |
Figure 4: HPLC chromatogram of Apigenin standard. |
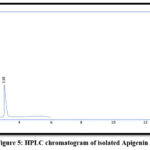 |
Figure 5: HPLC chromatogram of isolated Apigenin
|
Table 1: Retention time in min. for phenolic compounds in EtOAc fraction
| Phenolic compounds | Retention time forphenols in EtOAc fraction | Retention time of phenolic compounds standards |
| Syringic acid | 9.68 | 9.69 |
| Apigenin | 3.56 | 3.58 |
Analysis by FT-IR
FT-IR spectroscopy is generally used in phytochemical reports as a fingerprinting technique to compare normal compounds with synthetic standards. The IR spectra of the quarantined compound Apigenin is existing in figure 6. characteristic bands can be seen in the range of 3381-3000cm -1 (O-H of alcohols stretching vibration),2904-2620 cm-1 (C-H stretching vibration for aliphatic ),1650-1512 cm-1(C=O)stretching vibration.29while the bands present in FT-IR spectrum of isolated syringic acid in figure 7 exhibited distinct peaks at 3361 cm–1 (corresponding to –OH vibrations), 1710 cm − 1 (representing C = O stretching), and 1618 cm − 1 (indicating aromatic ring group C = C stretching). Others peaks were seen at 1367 cm–1, 1244 cm–1, and 1203 cm − 1, which corresponded to CH3-, C-O-C, and C-OH groups, respectively as in previous studies.30
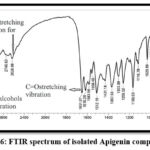 |
Figure 6: FTIR spectrum of isolated Apigenin compound |
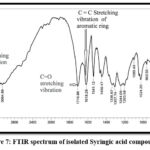 |
Figure 7: FTIR spectrum of isolated Syringic acid compound |
MTT Assay
The Cytotoxicity of Apigenin and Syringic acid isolated from plant leaves against A375 cell line compared with (Normal human dermal fibroblast) HdFn cell lines. both cell lines were exposed to serial concentrations from (6.25-400 μg/ml) of the Apigenin and Syringic acid to assess their effects on the viability of cell line as shown in figures (8 and 9) respectively. The decrease in A375 cell line viability (%) is represented in tables (2and3) respectively which exhibited a decrease in cell viability (%) with IC50 values of 40.8 μg /ml for apigenin and IC50 values of 54.5 μg /ml for syringic acid. These two compounds induced significant cell death that began at 25 μg /mL (P < 0.05, n = 6).
Table 2: Cytotoxicity of Apigenin on A375 cell line and HdFn Cell after 72 hours of Incubation at 37ºC
| Conc. of Apigenin in μg /ml | HdFn viable cell countMean ±SD | A375cell line countMean ±SD |
| 400 | 62.5386± 2.7419 | 22.3753 ±2.7365 |
| 200 | 74.4986 ±2.0764 | 29.9693± 2.5524 |
| 100 | 88.0846± 1.0499 | 39.3180± 1.5714 |
| 50 | 90.8653 ±1.1040 | 51.6603± 2.8454 |
| 25 | 93.6373± 0.4819 | 63.0000± 3.5990 |
| 12.5 | 94.0970 ±1.2520 | 76.2653 ±4.0336 |
| 6.25 | 95.0233 ±0.2315 | 85.5893± 1.1000 |
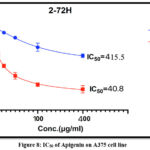 |
Figure 8: IC50 of Apigenin on A375 cell line |
Table 3: Cytotoxicity of Syringic acid on A375 cell line and HdFn Cell after 72 hours of Incubation at 37ºC
| Conc. of Syringic acid in μg /ml | HdFn viable cell countMean ±SD | A375cell line countMean ±SD |
| 400 | 65.5376± 3.7419 | 33.3653 ±2.7265 |
| 200 | 72.4976 ±3.07642 | 40.9593± 2.5424 |
| 100 | 90.0846± 1.04991 | 53.3180± 1.5724 |
| 50 | 92.8643 ±1.10405 | 62.6503± 1.8444 |
| 25 | 94.5373± 0.4819 | 73.0100± 3.5980 |
| 12.5 | 94.0970 ±1.2520 | 84.2643 ±4.0336 |
| 6.25 | 95.0433 ±0.2315 | 91.3893± 1.1000 |
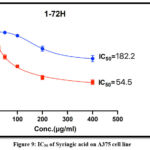 |
Figure 9: IC50 of Syringic acid on A375 cell line |
Discussion
The results were consistent with previous studies as ultrasonic analysis showed a significant increase in the conc.31 of extracted material. Twenty grams of plant leaves yielded 11 grams of crude extract material this is high percentage compared to using the soaking method or the Soxhlet extraction method due to Ultrasound waves made fast formation and collapse of microscopic bubbles in the solvent these bubbles explode the plant cell walls, causing cell disruption and freedom of intracellular phytochemical compounds. Furthermore, using 90% ethanol is an ideal solvent for extracting and isolating the two phenolic compounds, apigenin and syringic acid. The PHPLC results showed high efficiency in isolating the two phenolic compounds, as they had the same retention times compared to the standards compounds. Apigenin had the same retention times was 3.56 as standard apigenin retention time 3.58, and syringic acid showed the same retention time was 9.68 as the standard apigenin retention time 9.69.FT-IR bands gave indications of the presence of functional groups that characterize the apigenin compound, as the peak (3381–3000 cm⁻¹) for O-H group, (2904–2620 cm⁻¹)for C-H group, (1650–1512 cm⁻¹) for C=O group formed the chemical structure of the isolated apigenin compound. While syringic acid FT-IR spectroscopy showed three major band which 3361cm-1 for O-H group ,1710 cm-1for C=O group and 1618 cm-1 for C=C group the structural components of this compound. As for the MTT test, it showed a direct correlation between increasing concentration and the percentage decrease in live cancer cells. Apigenin had a clear effect, with a IC50 % 40.8 μg /ml which higher effect than that of syringic acid with IC50 54.5μg /ml %. This is due to the nature of the compound, as it has a more potent effect in inducing apoptosis than syringic acid, which has a simpler phenolic structure than apigenin with flavonoid structure with four hydroxyl group that have effect on PI3K/Akt and MAPK path way.32 The results also showed that both compounds had low activity against normal cells, exhibiting selective properties against cancer cells and being safer against normal cells. There are some limitations to this study. Only one cancer cell line was used, which limits the generalizability of the results to other cancer types. While the two compounds were isolated and characterized, further diagnostic testing is needed to determine their complete structural composition. Therefore, we anticipate conducting more comprehensive future studies to investigate the efficacy of these anticancer compounds.
Conclusion
The results indicate that two phenolic compounds isolated from parsley leaves Apigenin and Syringic acid exhibited an anti-cancer effect against A375 cell line that depended on increasing concentration of each compound. Apigenin was the more potent cytotoxic compound than Syringic acid. Both compounds demonstrated a safe and harmless effect on normal cells HdFn. Therefore, further preclinical studies to understand their mechanism of action in killing these cancer cells, with the aim of developing natural anti-cancer treatments.
Acknowledgement
We acknowledge the Department of Pharmacognosy and Plant science in College of Pharmacy, Thi- Qar University to facilitate our work
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article
Conflict of Interest
The author(s) declares no conflict of interest
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval Informed Consent Statement. This study did not involve human participants, and therefore, informed consent was not required.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Author contributions
- Ashwaq T. Kareem: contributed to data gathering, analysis, practical (follow the procedure), and written parts of the study.
- Samer khalid Ali gave final approval and agreement for all aspects of the study, supervision, revision, and rearrangement.
References
- Kadoglidou, K. I., Boutsika, A., Mellidou, I., Xanthopoulou, A., & Ganopoulos, I. Parsley: Breeding Approaches for Plant Improvement.Biodiversity and Genetic Improvement of Herbs and Spices, 2025; 261-294. https://doi.org/10.1007/978-3-031-81838-7_10
CrossRef - Karaca G, Koyuncu O, Gungor H, et al. A review of ethnobotanical, phytochemical and pharmacological properties of parsley (Petroselinum crispum).Int J Food Prop. 2019;22(1):1456–1471. https://doi.org/10.1079/cabicompendium.38808
CrossRef - Alobaidi, S. renal health benefits and therapeutic effects of parsley; areview. Frontiers in medicine,2024;11,1494740 https://doi.org/10.3389/fmed.2024.1494740
CrossRef - Martin, T. A., Ye, L., Sanders, A. J., Lane, J., & Jiang, W. G. Cancer invasion and metastasis: molecular and cellular perspective. InMadame Curie Bioscience Database. Landes Bioscience. https://doi.org/10.2741/4144
CrossRef - Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144(5):646-74. https://doi.org/10.1016/j.cell.2011.02.013
CrossRef - Vogelstein B, Papadopoulos N, Velculescu VE, et al. Cancer genome landscapes. Science. 2013;339(6127):1546-58. https://doi.org/10.1126/science.1235122
CrossRef - Torre LA, Siegel RL, Ward EM, et al. Global cancer incidence and mortality rates and trends—an update. Cancer Epidemiol Biomarkers Prev. 2016;25(1):16-27. https://doi.org/10.1158/1055-9965.epi-15-0578
CrossRef - Siegel RL, Miller KD, Fuchs HE, et al. Cancer statistics, 2024. CA Cancer J Clin. 2024;74(1):7-33. https://doi.org/10.3322/caac.21654
CrossRef - Newman DJ, Cragg GM. Natural products as sources of new drugs over the nearly four decades from 1981 to 2019. J Nat Prod. 2020;83(3):770-803. https://doi.org/10.1021/acs.jnatprod.9b01285
CrossRef - Salehi B, Venditti A, Sharifi-Rad M, et al. The therapeutic potential of apigenin. Int J Mol Sci. 2019;20(6):1305. https://doi.org/10.3390/ijms20061305
CrossRef - Shukla S, Gupta S. Apigenin: a promising molecule for cancer prevention. Pharm Res. 2010;27(6):962-78. https://doi.org/10.1007/s11095-010-0089-7
CrossRef - Srivastava JK, Shankar E, Gupta S. Chamomile: a herbal medicine of the past with a bright future. Mol Med Rep. 2010;3(6):895-901. https://doi.org/10.3892/mmr.2010.377
CrossRef - McKay DL, Blumberg JB. A review of the bioactivity and potential health benefits of chamomile tea (Matricaria recutita L.). Phytother Res. 2006;20(7):519-30. https://doi.org/10.3892/mmr.2010.377
CrossRef - Nabavi SF, Khan H, D’Onofrio G, et al. Apigenin as neuroprotective agent: of mice and men. Pharmacol Res. 2018;128:359-65. https://doi.org/10.1016/j.phrs.2017.10.008
CrossRef - Yan X, Qi M, Li P, et al. Apigenin in cancer therapy: anti-cancer effects and mechanisms of action. Cell Biosci. 2017;7:50. https://doi.org/10.1186/s13578-017-0179-x
CrossRef - Imran M, Salehi B, Sharifi-Rad J, et al. Apigenin: dietary sources, therapeutic perspectives and pharmacological activities. Biomed Pharmacother. 2019;110:958-74. https://doi.org/10.1002/ptr.6647
CrossRef - Peanparkdee M, Iwamoto S, Yamauchi R. Characterization of phenolic compounds in grape pomace extract and their antioxidant and antimicrobial activities. Food Chem. 2018;268:416-22. https://doi.org/10.1016/j.foodchem.2010.05.080
CrossRef - Rashed K, Butnariu M. Syringic acid: a review of its occurrence, biosynthesis, pharmacological and industrial importance. Chem Cent J. 2021;15(1):10. https://doi.org/10.1016/j.biopha.2018.09.069
CrossRef - Pinelo M, Arnous A, Meyer AS. Upgrading of grape skins: significance of plant cell-wall structural components and extraction techniques for phenol release. Trends Food Sci Technol. 2006;17(11):579-90. https://doi.org/10.1016/j.tifs.2006.05.003
CrossRef - Kumar N, Goel N. Phenolic acids: natural versatile molecules with promising therapeutic applications. Biotechnol Rep. https://doi.org/10.1016/j.btre.2019.e00370
CrossRef - Salehi B, Quispe C, Sharifi-Rad J, et al. Syringic acid: a review of its occurrence, biosynthesis, pharmacological and industrial importance. Biomed Pharmacother. 2021;137:111390. https://doi.org/10.1016/j.biopha.2018.09.069
CrossRef - Ringgit G, Cheong BE, Shah MD, et al. Syringic acid inCanarium odontophyllum for diabetes and obesity–a review. Cell Biochem Biophys. 2025;1-27.https://doi.org/10.1007/s12013-025-01773-8
CrossRef - Ferah Okkay I, Okkay U, Cicek B, et al . Syringic acid guards against indomethacin-induced gastric ulcer by alleviating inflammation, oxidative stress and apoptosis.Biotechnic Histochem. 2024;99(3):147-56.. https://doi.org/10.1080/10520295.2024.2344477
CrossRef - Mirza, A. C., Panchal, S. S., Allam, A. A., et al. Syringic acid ameliorates cardiac, hepatic, renal and neuronal damage induced by chronic hyperglycaemia in Wistar rats: a behavioural, biochemicalandhistologicalanalysis.Molecules, 2022;27(19):https://doi.org/10.3390/molecules27196722
CrossRef - Kareem, A. T., & Kadhim, E. J. Isolation and identification of hyperin and naringenin from guava cultivated in Iraq and evaluation of cytotoxic activity of hyperin.Plant Science Today. 2024 ; 11 (2). https://doi. org/10.14719/pst,
CrossRef - Kareem, A. T., & Hamad, M. N. Separation and identification of phenolic acid from Borago officinalis (F: Boraginaceae) cultivated in Iraq.Iraqi Journal of Pharmaceutical Sciences, 2020; 29(2):139-151. https://doi.org/10.31351/vol29iss2pp139-151
CrossRef - Freshney RI. Culture of animal cells: a manual of basic technique and specialized applications. John Wiley & Sons; 2015; 23. https://doi.org/10.1002/9780470649367
CrossRef - Al-Ziaydi AG, Al-Shammari AM, Hamzah MI, Kadhim HS, Jabir MS. Newcastle disease virus suppress glycolysis pathway and induce breast cancer cells death. Virusdisease. 2020;31(3):341-8. https://doi.org/10.1007/s13337-020-00612-z
CrossRef - Aldawsari MF, Ahmed MM, Fatima F, et al. Development and characterization of calcium-alginate beads of apigenin: in vitro antitumor, antibacterial, and antioxidant activities. Mar Drugs. 2021;19(8):467. https://doi.org/10.3390/md19080467
CrossRef - Rasheed A, Kamran SH, Hameed M, et al. Syringic acid loaded chitosan nanoparticles mitigate glycation-associated oxidative stress and inflammation in hyperglycaemic rat model. Sci Rep. 2025;15(1):22778. https://doi.org/10.1038/s41598-025-05469-0
CrossRef - Zuhair Abdul-lalil T. Ultrasound-Assisted Extraction of Fennel Leaves: Process optimization, Thin Layer Chromatography and Cytotoxic Activity of Ethanolic Extract. Iraqi J Pharm Sci. 2024;33(1):94-103. https://doi.org/10.31351/vol33iss1pp94-103
CrossRef - Tong, X., & C. Pelling, J. (2013). Targeting the PI3K/Akt/mTOR axis by apigenin for cancer prevention.Anti-Cancer Agents in Medicinal Chemistry-Anti-Cancer Agents), 13(7), 971-978. https://doi.org/10.2174/18715206113139990119
CrossRef







