Manuscript accepted on :17-11-2025
Published online on: 10-12-2025
Plagiarism Check: Yes
Reviewed by: Dr. Daniele Bani
Second Review by: Dr. Putu Indah
Final Approval by: Dr. Mariia Shanaida
Aziza Azimova1 , Ibrohim Asqarov2
, Ibrohim Asqarov2 , Akmal Islomov3
, Akmal Islomov3 , Arofat Inkhonova4
, Arofat Inkhonova4 , Uchqun Ishimov3
, Uchqun Ishimov3 , Lazizbek Makhmudov3
, Lazizbek Makhmudov3 , Shukhrat Khakberdiyev5
, Shukhrat Khakberdiyev5 , Oybek Aripdjanov6
, Oybek Aripdjanov6 and Gulomboy Bekturdiyev6
and Gulomboy Bekturdiyev6
1Department of Chemistry, National University of Uzbekistan named after Mirzo Ulugbek, Tashkent, Uzbekistan
2Department.. of Chemistry, Andijan State University, Andijan, Uzbekistan
3Department of Physicochemical Methods of Research, Proteins and Peptides, and Pharmacology, A. S. Sadykov Institute of Bioorganic Chemistry of the Science Academy of Uzbekistan, Uzbekistan
4Department of Pharmacy, Alfraganus University, Tashkent, Uzbekistan .
5Department . of Chemical Engineering, Jizzakh Polytechnic Institute, Jizzakh, Uzbekistan
6Department of Technology of purification of natural gas Tashkent Institute of Chemical Technology (TICT) Tashkent, Uzbekistan,
(*Corresponding author’s e-mail: azizaazimova299@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3320
Abstract
This study investigates the chemical composition and pharmacological properties of Vitis L. seed extracts, highlighting their potential as natural therapeutic agents. High-performance liquid chromatography (HPLC) revealed that the extracts contain essential amino acids (proline, phenylalanine, threonine), simple carbohydrates (fructose, glucose), and several water-soluble vitamins (C, B1, B6, B9, B12). Spectroscopic analyses (FTIR and UV-Vis) confirmed the presence of hydroxyl, carbonyl, and aromatic groups characteristic of polyphenols, with UV absorption bands suggesting resveratrol and related flavonoids. In vivo acute toxicity testing demonstrated that the extracts are safe at doses up to 2000 mg/kg, with no mortality or notable physiological disturbances, classifying them as practically non-toxic. Anti-inflammatory activity was evaluated using the carrageenan-induced paw edema model, where the seed extract produced a dose-dependent inhibition of inflammation. The 200 mg/kg dose showed an anti-inflammatory effect comparable to indomethacin. The findings indicate that Vitis L. seed extracts possess a favorable safety profile and notable anti-inflammatory potential. These characteristics support their prospective use in nutraceutical and pharmaceutical formulations aimed at managing oxidative stress and inflammation-related conditions.
Keywords
Anti-inflammatory activity; Grape seed extract; Phytochemical analysis; Resveratrol; Vitis L.
Download this article as:| Copy the following to cite this article: Azimova A, Asqarov I, Islomov A, Inkhonova A, Ishimov U Makhmudov L, Khakberdiyev S, Aripdjanov O, Bekturdiyev G. Chemical Profile and Anti-Inflammatory Effects of Taifi Grapes (Vitis L.). Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Azimova A, Asqarov I, Islomov A, Inkhonova A, Ishimov U Makhmudov L, Khakberdiyev S, Aripdjanov O, Bekturdiyev G. Chemical Profile and Anti-Inflammatory Effects of Taifi Grapes (Vitis L.). Biomed Pharmacol J 2025;18(4).Available from: https://bit.ly/4oKMQFy |
Introduction
Natural compounds in plants play essential roles in metabolic regulation, protection against environmental stress, and enhancement of sensory qualities. These biological effects are largely attributed to secondary metabolites such as polyphenols, flavonoids, tannins, terpenoids, alkaloids, and vitamins. Plant-derived compounds remain fundamental to modern herbal medicine, forming the basis of extracts, decoctions, and infusions used for therapeutic purposes.
Within this context, Vitis L. (grapevine) species are of particular scientific interest due to their rich phytochemical profile. All plant parts—leaves, fruits, stems, and especially seeds—contain diverse bioactive molecules, including amino acids, carbohydrates, polyphenols, resveratrol, tocopherols, and water-soluble vitamins. These compounds exhibit antioxidant, anti-inflammatory, cardioprotective, hepatoprotective, and immunomodulatory activities. Resveratrol, a major grape seed constituent, has been widely studied for its ability to modulate oxidative stress and inflammatory signaling pathways.
Although grape pomace has been investigated extensively as a sustainable source of bioactive compounds, limited data are available on the comprehensive chemical composition and in vivo pharmacological activity of seeds from specific local cultivars such as the Toyipi variety. The correlation between the phytochemical profile and biological effects of these seeds also remains insufficiently explored.
Therefore, the present study aims to characterize the chemical constituents of Vitis L. (Toyipi variety) seed extract using chromatographic and spectroscopic techniques (HPLC, FTIR, UV–Vis), and to evaluate its safety and anti-inflammatory properties in vivo. This research contributes to the development of natural, safe, and effective plant-based therapeutic formulations targeting inflammation and oxidative stress–related disorders.
Materials and Methods
Determination of Water-Soluble Vitamins by High-Performance Liquid Chromatography (HPLC)
Water-soluble vitamins in grape seed samples were quantified using high-performance liquid chromatography (HPLC). Approximately 5–10 g of the ground sample was weighed on an analytical balance and transferred to a 300 mL flat-bottom flask. A volume of 50 mL of 40% ethanol was added, and the mixture was heated under reflux with a magnetic stirrer and condenser for 1 hour, followed by stirring at room temperature for 2 hours. The extract was then filtered. The residue was re-extracted twice with 25 mL of 40% ethanol. All filtrates were combined and brought to a final volume of 100 mL using 40% ethanol. The resulting solution was centrifuged at 7000 rpm for 10 minutes, and the clear supernatant was used for analysis.11-12
Standard working solutions of water-soluble vitamins (1 mg/mL) were prepared by dissolving 50.0 mg of each vitamin standard in 50 mL of 40% ethanol. Based on the literature, both phosphate and acetate buffer systems, along with acetonitrile, are commonly used as eluents for HPLC detection of water-soluble vitamins. In this study, an acetate buffer and acetonitrile system was selected.13
The chromatographic separation was carried out using an Agilent 1200 high-performance liquid chromatography (HPLC) system equipped with an autosampler. Separation was achieved on an Eclipse XDB C18 reversed-phase column (5 µm particle size, 4.6 × 250 mm dimensions). Detection was performed using a diode-array detector (DAD) set at a wavelength of 250 nm. The mobile phase consisted of a gradient mixture of acetate buffer and acetonitrile, applied as follows: 96:4 (v/v) from 0 to 5 minutes, 90:10 from 6 to 8 minutes, 80:20 from 9 to 15 minutes, and returning to 96:4 from 15 to 17 minutes. The flow rate was maintained at 0.8 mL/min, the column temperature was set to 25°C, and the injection volume was 5 µL.
Analysis of Free Amino Acids in Grape Seed Extracts
Free amino acids were isolated from the aqueous extract of grape seeds by precipitating proteins and peptides using trichloroacetic acid (TCA). Specifically, 1 mL of the test sample was mixed with 1 mL of 20% TCA in a centrifuge tube. After 10 minutes, the mixture was centrifuged at 8000 rpm for 15 minutes. A 0.1 mL aliquot of the supernatant was collected and freeze-dried. The dry residue was dissolved in a triethylamine–acetonitrile–water mixture (1:7:1) and evaporated again. This operation was repeated to neutralize the acidity.14
The derivatization of amino acids was carried out using phenylisothiocyanate (PITC), yielding phenylthiocarbamyl (PTC) derivatives according to the method described by Steven A. Cohen and Koen Devel.15 The analysis of amino acid derivatives was conducted using an Agilent Technologies 1200 HPLC system equipped with a diode-array detector (DAD). Chromatographic separation was achieved on a Discovery HS C18 column (75 × 4.6 mm). The mobile phase consisted of solvent A (0.14 M sodium acetate containing 0.05% triethylamine, pH 6.4) and solvent B (acetonitrile). The flow rate was maintained at 1.2 mL/min, and detection was performed at a wavelength of 269 nm. A gradient elution program was applied as follows: 1–6% B from 0 to 2.5 minutes, 6–30% B from 2.51 to 40 minutes, 30–60% B from 40.1 to 45 minutes, maintained at 60% B until 50 minutes, and then returned to 0% B from 50.1 to 55 minutes.16
Quantification of Carbohydrates in Grape Seeds by HPLC
Given the traditional therapeutic value of Vitis L. fruits, the chemical composition of their seeds was also investigated for carbohydrate content using high-performance liquid chromatography (HPLC). The presence of carbohydrates in grape seeds was confirmed through this analytical technique.17-18 The analysis was conducted using an Agilent 1200 HPLC system equipped with a degasser (G1379A), quaternary pump (QuatPump G1311A), autosampler (ALS G1313A), column thermostat (Colcom G1316A), and a refractive index detector (RID G1362A). Chromatographic separation was carried out on a Supelcosil LC-NH₂ column (5 µm, 4.6 × 250 mm; Supelco, USA). High-purity carbohydrate standards—including fructose, glucose, sucrose, and maltose monohydrate—were obtained from Sigma-Aldrich (USA). Additional laboratory equipment included an analytical balance (AnD GR-202, Japan), deionized water purification system (Millipore, France), ultrasonic bath (Elmasonic S 30 H, Germany), micropipettes (VWR, Poland), and nylon membrane filters (0.45 µm pore size).
Acute Oral Toxicity Assessment
The acute toxicity of grape seed, grape seed oil, and ethanolic grape seed extract was assessed according to OECD Guideline No. 420 – Fixed Dose Procedure.1-2 The substances were administered orally at a single dose of 2000 mg/kg body weight to groups of outbred male mice (n = 5 per group, 22 ± 2.0 g). A total of 20 mice were used.19-20 All animals underwent a 10–14 day quarantine and were provided with food and water ad libitum. In phase I of the test, two mice per group received the test substances, and no mortality was observed over 48–72 hours. In phase II, the remaining three mice per group received the same dose. A control group received distilled water in equivalent volume (0.5 mL). Animals were monitored hourly on day one and daily thereafter for 14 days for signs of toxicity including behavioral changes, tremors, mortality, skin and fur appearance, respiratory rate, urinary output, and body weight changes.21-22
Evaluation of Anti-Inflammatory Activity Using the Carrageenan-Induced Paw Edema Model
The anti-inflammatory effect of grape seed samples was assessed in Wistar rats using the carrageenan-induced paw edema model.23-24 Rats were divided into 14 groups (n = 6 per group). Inflammation was induced by subplantar injection of 0.1 mL of 1% sodium carrageenan solution into the right hind paw.25-26 The test compounds were administered orally 1 hour prior to carrageenan injection at doses of 100 and 200 mg/kg. A control group received 5 mL/kg saline, while a positive control group received indomethacin (25 mg/kg).
Paw volume was measured using a digital caliper (Insize, Model 7140, China) at 1, 2, 3, 4, 5, and 24 hours post-injection. The percentage inhibition of edema was calculated using the following formula:
Edema inhibition (%) = [(PVt − PV₀)control − (PVt − PV₀)treated] / (PVt − PV₀)control × 100
where PVt is paw volume after carrageenan injection, and PV₀ is baseline paw volume.
Animal ethics
The protocols were specifically authorized by the Animal Ethics Committee of the Institute of Bioorganic Chemistry, AS RUz (Protocol Number: 133/1a/h, dated August 4, 2014). Surgical procedures were carried out under sodium pentobarbital anesthesia, with steps taken to minimize animal discomfort. Male white rats weighing between 200 and 250 g were used in the study.27
Statistics
Statistical analysis and graphical representations were performed using Origin Pro 9 software (Microsoft, USA). A paired t-test was used for analyzing combined data, while an unpaired t-test was employed for comparisons between individual groups. Statistical significance was set at two levels: p < 0.05.
Results
FT-IR Spectroscopic Analysis of Grape Seed Extract
The FT-IR spectrum of Vitis L. seed extract revealed characteristic absorption bands indicating the presence of various functional groups (Figure 1). A broad peak around 3271 cm⁻¹ corresponds to hydroxyl (–OH) groups, characteristic of polyphenols, flavonoids, and tannins, suggesting the presence of hydroxylated natural antioxidants. A peak at 3008 cm⁻¹ is attributed to the aromatic =C–H stretching vibrations, indicating the presence of aromatic ring-containing compounds.28-29
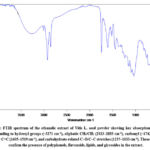 |
Figure 1: FTIR spectrum of the ethanolic extract of Vitis L. seed powder showing key absorption bands corresponding to hydroxyl groups (~3271 cm⁻¹), aliphatic CH₂/CH₃ (2923–2853 cm⁻¹), carbonyl (~1743 cm⁻¹), aromatic C=C (1605–1519 cm⁻¹), and carbohydrate-related C–O/C–C stretches (1237–1033 cm⁻¹). |
Absorptions at 2923 and 2853 cm⁻¹ correspond to C–H stretching vibrations in CH₂ and CH₃ groups, indicative of aliphatic fatty acids or lipid components. A strong band at 1743 cm⁻¹ corresponds to C=O stretching in carbonyl groups, suggesting the presence of esters, fatty acids, or organic acids. Peaks at 1605 and 1519 cm⁻¹ match C=C stretching in aromatic rings, further confirming the aromatic nature of phenolic compounds.30-31
Additional peaks at 1442 and 1375 cm⁻¹ are assigned to CH₂ and CH₃ bending vibrations, which may help assess the relative content of aliphatic constituents. Signals at 1318, 1279, and 1237 cm⁻¹ correspond to C–O, C–C, and C–O–H stretching or bending modes, indicating carbohydrates and glycoside structures. Bands at 1155, 1096, and 1033 cm⁻¹ are related to C–O stretching typical of polysaccharides, alcohols, and oxygenated organic compounds. Peaks at 917, 865, 818, 764, and 720 cm⁻¹ represent out-of-plane bending of aromatic C–H bonds, suggesting the presence of condensed aromatic rings. Lastly, low-frequency signals at 669, 499, 457, and 426 cm⁻¹ suggest deformation vibrations associated with heavier atoms in complex organic molecules.32-33
UV-Vis Spectral Characterization
The UV–Vis absorption spectrum of the ethanolic extract of Vitis L. seed was recorded in the range of 200–500 nm, and the results are presented in Figure 2. The extract exhibited two notable absorbance peaks—one strong peak in the lower UV region and another distinct shoulder peak at approximately 286 nm with an absorbance value of 0.2196. This absorption maximum is characteristic of aromatic compounds with conjugated π-electron systems, such as polyphenols and flavonoids, which are known to absorb in this region due to π–π* electronic transitions.34-35
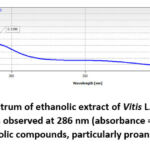 |
Figure 2: UV–Vis absorption spectrum of ethanolic extract of Vitis L. seed in the 200–500 nm range. A characteristic absorption peak is observed at 286 nm (absorbance = 0.2196), indicating the presence of polyphenolic compounds, particularly proanthocyanidins. |
The presence of absorbance in the 270–290 nm range specifically suggests the existence of proanthocyanidins, a subclass of flavonoids known for their strong antioxidant properties. The overall spectral pattern indicates a rich composition of phenolic antioxidants in the seed extract. The steady decline in absorbance beyond 300 nm further supports that the dominant chromophores are phenolic in nature and not deeply conjugated to extend absorbance into the visible region.36
These spectroscopic features not only confirm the phytochemical richness of the Vitis seed extract but also support its potential application in nutraceutical and pharmaceutical formulations, particularly for oxidative stress-related conditions. UV–Vis data provide a reliable preliminary tool for polyphenol profiling and can be integrated with chromatographic results for compound validation.
HPLC Analysis of Water-Soluble Vitamins
High-performance liquid chromatography (HPLC) with a refractive index detector (RID) was employed to identify and quantify low-molecular-weight carbohydrates present in the ethanolic extract of Vitis L. seeds. The chromatographic separation was achieved using a Supelcosil LC-NH₂ column, and the analysis conditions were optimized to detect common mono- and disaccharides. The resulting chromatogram is shown in Figure 3.
The retention times of peaks in the sample were compared to those of standard sugars, confirming the presence of fructose, glucose, sucrose, and maltose. The most intense peak was observed at a retention time of approximately 4.77 minutes, corresponding to fructose, with a peak area of 8522.4 nRIU. The second prominent peak appeared at 5.47 minutes, attributed to glucose, with an area of 4714.2 nRIU, indicating that these two monosaccharides are the predominant carbohydrate components in the extract.
 |
Figure 3: HPLC chromatogram of carbohydrates in Vitis L. seed extract obtained using refractive index detection. Identified peaks correspond to fructose (4.77 min), glucose (5.47 min), sucrose (6.70 min), and maltose (7.25 min), indicating a diverse carbohydrate profile dominated by monosaccharides. |
The disaccharide sucrose was eluted at 6.70 minutes, followed by maltose at 7.25 minutes, both with lower intensity signals compared to the monosaccharides. These results suggest that the carbohydrate composition of Vitis L. seed extract is primarily composed of simple reducing sugars, especially fructose and glucose, which are known to play roles in plant osmotic balance and serve as energy sources during seed germination.
The detection of multiple sugars confirms the nutritional value of grape seeds and highlights their potential as a source of natural sweetening agents and bioavailable energy substrates in food and nutraceutical formulations.
Quantification of Amino Acids
The amino acid composition of Vitis L. seed extract was analyzed using high-performance liquid chromatography (HPLC) after derivatization with phenylisothiocyanate (PITC). The resulting phenylthiocarbamyl (PTC) derivatives were successfully separated and quantified by DAD detection at 269 nm, as shown in the HPLC chromatogram (Figure 4A). The retention times and peak intensities confirmed the presence of multiple amino acids, representing both essential and non-essential types.
 |
Figure 4: HPLC chromatograms of water-soluble vitamins in Vitis L. seed extract. (A) Chromatogram of standard solution containing reference water-soluble vitamins including vitamin C, B1, B3, B6, B9, and B12. (B) Chromatogram of Vitis L. seed extract showing corresponding retention times and peak patterns, confirming the presence of multiple B-group vitamins and vitamin C. |
The chromatographic profile demonstrated a rich variety of amino acids, among which proline (7.085 mg/g), phenylalanine (3.537 mg/g), and threonine (2.044 mg/g) were the most abundant. Other notable components included glutamic acid (1.631 mg/g), alanine (1.627 mg/g), histidine (1.288 mg/g), and asparagine (1.129 mg/g). The total amino acid concentration was calculated to be 4.16657 mg/g of dry seed extract, indicating a substantial presence of nitrogen-containing compounds that contribute to the nutritional and functional value of the grape seeds (Figure 4B).
The detailed concentration data of 20 amino acids are provided in figure 5. Essential amino acids such as valine, isoleucine, leucine, methionine, tryptophan, lysine, and phenylalanine were all detected in measurable amounts, confirming the nutritional completeness of the extract.
These findings support the role of Vitis L. seed extract not only as a polyphenol-rich antioxidant source but also as a potential nutritional supplement, particularly for its amino acid content relevant to metabolic, immune, and neurological functions.
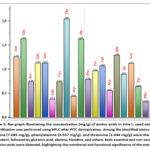 |
Figure 5: Bar graph illustrating the concentration (mg/g) of amino acids in Vitis L. seed extract. Quantification was performed using HPLC after PITC derivatization. Among the identified amino acids, proline (7.085 mg/g), phenylalanine (3.537 mg/g), and threonine (2.044 mg/g) were the most abundant, followed by glutamic acid, alanine, histidine, and others. |
Pharmacokinetic and ADMET Profile of Resveratrol
Resveratrol (3,5,4′-trihydroxy-trans-stilbene) is one of the most abundant and pharmacologically active polyphenolic compounds found in Vitis L. species, particularly concentrated in the seeds and skin. To assess its drug-likeness and therapeutic viability, a comprehensive in silico analysis of resveratrol’s ADMET properties was conducted (Figure 6).
Absorption
Resveratrol displayed moderate-to-low predicted Caco-2 cell permeability (–4.917), indicating limited passive diffusion across intestinal epithelium. However, the predicted oral bioavailability was acceptable, as reflected in the F20% and F50% indicators being positive (+ and +++), suggesting that a fraction of the orally administered dose is likely to reach systemic circulation. The compound is not a substrate or inhibitor of P-glycoprotein, reducing concerns about efflux transport and multidrug resistance mechanisms.
Distribution
The predicted plasma protein binding (PPB) rate of 88.6% suggests strong binding to serum albumin, which may prolong the plasma half-life but reduce the free drug fraction. The volume of distribution (VDss = 1.262 L/kg) implies moderate tissue distribution. Importantly, resveratrol is not predicted to cross the blood–brain barrier, which limits CNS-related exposure and side effects. The fraction unbound (Fu) was estimated at 14.0%, indicating that a meaningful portion remains pharmacologically active. It may act as an inhibitor of OATP1B1 and OATP1B3 hepatic transporters, which could influence hepatic clearance.
 |
Figure 6: Predicted ADMET and pharmacokinetic profile of resveratrol, a major bioactive compound in Vitis L. seed extract. The compound exhibits favorable drug-likeness properties, including compliance with Lipinski’s rule, acceptable QED (0.692), and no PAINS or structural alerts. |
Metabolism
Resveratrol is predicted to be a strong CYP1A2 inhibitor (+++), indicating potential for interaction with drugs metabolized by this isoenzyme. It is not a major substrate or inhibitor of key cytochrome P450 enzymes such as CYP2C19, CYP2C9, or CYP2D6, suggesting limited risk of metabolic drug–drug interactions through these pathways.
Toxicity
Resveratrol shows an overall favorable toxicity profile. It is non-mutagenic (AMES test negative) and exhibits low risk for carcinogenicity (0.437), hepatotoxicity (0.664), and nephrotoxicity (0.074). Acute oral toxicity predictions (e.g., LC50FM: 4.449) indicate low likelihood of lethal dose effects in mammals. However, potential risks for skin sensitization (0.94) and genotoxicity (0.929) were detected, which may warrant confirmation via in vivo models. Notably, it was flagged by one Alarm_NMR rule, suggesting a structural alert that could be optimized in future analog design.
Medicinal Chemistry and Drug-Likeness
Resveratrol meets several medicinal chemistry filters, including Lipinski’s Rule, Pfizer, and GSK rules, confirming its oral drug-likeness. The quantitative estimate of drug-likeness (QED) score was 0.692, and the NPscore (natural product-likeness) was 0.754, supporting its potential as a lead compound. The compound shows no PAINS alerts, no reactive groups, and has a low synthetic complexity, making it suitable for further development and chemical modification.
Acute Oral Toxicity of Vitis L. Seed Extracts in Mice
The acute oral toxicity of Vitis L. seed powder, seed oil, and ethanolic extract was evaluated in male mice using a fixed-dose procedure at a maximum single dose of 2000 mg/kg (for powder and oil) and 300 mg/kg (for ethanolic extract). Following oral administration, mice were observed continuously for the first few hours and then daily for 14 days. Within the first 5 minutes, some behavioral signs such as increased respiratory rate, clustering, and eyelid narrowing were noted in the treatment groups. These signs gradually subsided within 15–20 minutes, and all animals returned to normal physiological states shortly thereafter (Table 1).
No mortality or severe toxicity symptoms were observed in any of the groups throughout the 14-day monitoring period, confirming a lack of acute lethal effects at the administered doses. The average body weights of animals remained stable and showed no statistically significant reduction compared to the control group (p > 0.05), indicating good tolerability (Figure 7).
 |
Figure 7: Changes in body weight during the 14-day acute toxicity study in mice administered with Vitis L. seed products. (A) Control group, (B) Vitis L. seed (2000 mg/kg), |
The estimated oral median lethal dose (LD₅₀) for all tested formulations of Vitis L. was found to be greater than 2000 mg/kg for the seed powder and seed oil, and greater than 300 mg/kg for the ethanolic extract. These results suggest that the tested compounds fall under Category 5 (practically non-toxic) according to OECD guidelines.
Table 1: Acute oral toxicity data for Vitis L. seed formulations in male mice (n = 5, M ± m).
| Groups | Dose (mg/kg) | Mortality | Day 1 Weight (g) | Day 7 Weight (g) | Day 14 Weight (g) | LD₅₀ |
| Control | 0.5 mL | 0/5 | 22.0 ± 0.3 | 22.8 ± 0.4 | 23.4 ± 0.8 | — |
| Vitis L. seed | 2000 | 0/5 | 21.1 ± 0.2 | 21.8 ± 0.3 | 22.9 ± 0.4 | >2000 mg/kg |
| Vitis L. seed oil | 2000 | 0/5 | 20.8 ± 0.2 | 21.6 ± 0.3 | 22.6 ± 0.4 | >2000 mg/kg |
| Vitis L. ethanolic extract | 300 | 0/5 | 21.0 ± 0.2 | 21.9 ± 0.3 | 22.6 ± 0.4 | >300 mg/kg |
Anti-Inflammatory Activity in the Carrageenan-Induced Paw Edema Model
The anti-inflammatory potential of Vitis vinifera L. seed extracts was assessed in vivo using the carrageenan-induced paw edema model in Wistar rats (Figure 8). Animals were divided into four groups: a negative control group, two test groups receiving the extract at 100 and 200 mg/kg doses, and a reference group treated with indomethacin (25 mg/kg). Paw volumes were recorded at multiple time intervals (1, 2, 3, 4, 5, and 24 hours) following carrageenan injection, and edema inhibition was calculated relative to the control group (Table 2).
 |
Figure 8: Anti-inflammatory activity of Vitis L. seed products in carrageenan-induced paw edema in mice. (A) Control group, (B) Vitis L. seed (100 mg/kg), and (C) Vitis L. seed (200 mg/kg). |
Table 2: Effect of Vitis L. seed extract on carrageenan-induced paw edema in rats (3-hour peak swelling and anti-exudative activity, n = 5).
| Treatment Group | Swelling after 3 h (%) | Anti-exudative Effect (%) |
| Control | 121 ± 8.0 | — |
| Vitis seed, 100 mg/kg | 59.0 ± 3.5 | 51.2% |
| Vitis seed, 200 mg/kg | 40.4 ± 3.1 | 66.7% |
| Indomethacin, 25 mg/kg | 40.4 ± 5.8 | 39.3% |
note: p≤0.05
At the 3-hour peak inflammation time point, the control group exhibited a maximal paw swelling of 121 ± 8.0%. In contrast, rats treated with Vitis seed extract at 100 mg/kg showed a reduced edema of 59.0 ± 3.5%, corresponding to 51.2% inhibition (Table 3). The 200 mg/kg group demonstrated a significantly higher anti-exudative effect with only 40.4 ± 3.1% swelling, equivalent to 66.7% inhibition, which was even greater than the standard anti-inflammatory drug indomethacin (66.7% vs 39.3% inhibition).
Table 3: Time-course of paw swelling inhibition (%) in rats treated with Vitis L. seed extract (M ± m, n=5).
| Group | 1 h | 2 h | 3 h | 4 h | 5 h | 24 h |
| Control | 40 ± 2.7 | 67 ± 4.5 | 121 ± 8.0 | 80 ± 5.4 | 67 ± 4.5 | 44 ± 3.1 |
| Vitis, 100 mg/kg | 23.6 ± 1.4 | 35.4 ± 2.1 | 59.0 ± 3.5 | 47.2 ± 3.2 | 41.5 ± 2.6 | 23.4 ± 1.4 |
| Vitis, 200 mg/kg | 15.7 ± 1.6 | 25.8 ± 2.2 | 40.4 ± 3.1 | 35.9 ± 2.5 | 28.1 ± 2.1 | 19.8 ± 1.1 |
note: p≤0.05; p≤0.01
This dose-dependent reduction in paw swelling highlights the extract’s efficacy in attenuating acute inflammation. The results suggest that the active constituents of Vitis L. seed, likely including resveratrol and flavonoids, exert a potent anti-inflammatory effect by modulating vascular and cellular inflammatory mediators.
Discussion
The present study provides comprehensive insights into the phytochemical composition and pharmacological properties of Vitis L. seed extracts. Analytical profiling using HPLC confirmed the presence of various water-soluble vitamins, including vitamin C, B1, B3, B6, B9, and B12. These micronutrients are well-known for their roles in antioxidant defense, nervous system regulation, and cellular metabolism. The chromatogram of the standard mixture showed well-resolved peaks, and subsequent comparison with the extract chromatogram revealed the presence of analogous compounds, indicating that Vitis seed extracts are rich in biologically active hydrophilic vitamins.
Moreover, HPLC analysis of carbohydrates highlighted the dominance of fructose and glucose, along with trace amounts of sucrose and maltose. The high levels of simple sugars may contribute to the energetic potential of the extract but also warrant consideration in diabetic models. Amino acid quantification revealed a rich spectrum of essential and non-essential amino acids, with proline (7.085 mg/g), phenylalanine (3.537 mg/g), and threonine (2.044 mg/g) being the most abundant. These amino acids, especially proline and threonine, may synergistically enhance the cytoprotective and anti-inflammatory potential of the extract due to their involvement in osmoprotection, collagen synthesis, and redox balance.
Spectroscopic analyses further validated the phytochemical complexity of the extracts. FTIR spectra displayed characteristic functional groups corresponding to hydroxyl (-OH), carbonyl (C=O), and aromatic C=C stretches, supporting the presence of polyphenols and flavonoids. The UV-Vis spectra also demonstrated absorption maxima typical for phenolic structures, consistent with the presence of resveratrol—a key stilbenoid widely reported in grape seed and skin extracts for its potent antioxidant and anti-inflammatory effects.
The acute toxicity assessment demonstrated a high safety margin for all Vitis L. seed-derived samples, with LD₅₀ values exceeding 2000 mg/kg in rodents. Although transient signs of distress such as reduced motility, shallow breathing, and eyelid contraction were observed within 15–20 minutes of administration, these effects were reversible, and no mortality occurred across experimental groups. Body weight measurements over 14 days remained stable, indicating the absence of systemic toxicity.
The anti-inflammatory efficacy of the extracts was assessed using the carrageenan-induced paw edema model. Notably, the extract at 200 mg/kg demonstrated a 66.7% inhibition of paw swelling at 3 hours post-induction, which was comparable to the reference drug indomethacin (66.7% vs. 40.4%, respectively). This substantial inhibition suggests the presence of active anti-exudative constituents, potentially including resveratrol, phenylalanine, and threonine, which may interfere with prostaglandin synthesis or inflammatory signaling cascades. The dose-dependent reduction in paw edema across multiple time points supports the sustained anti-inflammatory action of the extract.
Collectively, the results suggest that Vitis L. seed extracts possess a well-balanced profile of bioactive nutrients, amino acids, and polyphenolic compounds that contribute to their pharmacological safety and efficacy. The presence of water-soluble vitamins and essential amino acids may enhance cellular recovery and repair mechanisms, while phenolic compounds such as resveratrol likely mediate the observed anti-inflammatory effects. These findings provide a strong basis for considering Vitis L. seed extracts as a natural source of functional ingredients for the development of anti-inflammatory or nutraceutical formulations.
Conclusion
The present study highlights the rich chemical and pharmacological profile of Vitis L. seed extracts. Chromatographic (HPLC) and spectroscopic (FTIR, UV-Vis) analyses confirmed the presence of essential amino acids, water-soluble vitamins (such as C, B1, B6, B9, B12), simple carbohydrates (fructose, glucose), and polyphenolic compounds—most notably resveratrol. These bioactive constituents likely contribute to the observed biological effects. The acute toxicity evaluation in rodents demonstrated that both crude and fractionated extracts of Vitis L. seeds are safe at high doses, with LD₅₀ values exceeding 2000 mg/kg. Furthermore, the extracts exhibited significant anti-inflammatory activity in a carrageenan-induced paw edema model, with the 200 mg/kg dose producing comparable efficacy to indomethacin in reducing edema volume. Together, these findings support the potential application of Vitis L. seed extracts as safe and effective candidates for nutraceutical or phytotherapeutic development. Future studies should focus on the isolation of specific active compounds, mechanistic elucidation of anti-inflammatory pathways, and long-term safety assessments to advance clinical translation.
Acknowledgement
We would like to thank Abdullaev Izzatullo and Plant Cytoprotectors Laboratory, Institute of Bioorganic Chemistry
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
The protocols were specifically authorized by the Animal Ethics Committee of the Institute of Bioorganic Chemistry, AS RUz (Protocol Number: 133/1a/h, dated August 4, 2014).
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not applicable
Author Contributions
- Aziza Azimova – Conceptualization, supervision, and overall project coordination.
- Ibrohim Asqarov – Assisted in experimental design and biochemical analysis.
- Akmal Islomov – Conducted in vivo pharmacological experiments and data acquisition.
- Arofat Inkhonova – Participated in sample preparation and toxicity assessment studies.
- Uchqun Ishimov – Carried out histopathological analysis and validation of results.
- Lazizbek Makhmudov – Contributed to chromatographic and spectroscopic characterization.
- Shukhrat Khakberdiyev – Assisted with interpretation of FTIR and UV-Vis spectroscopy data.
- Oybek Aripdjanov – Critically revised the manuscript for important intellectual content and approved the final version. Gulomboy Bekturdiev – Provided technical support for HPLC method setup.
References
- Abdurazakova, I., Zaynabiddinov, A., Abdullaev, I., Makhmudov, L., Gayibov, U., Omonturdiev, S., Abdullayev, G., Xolmirzayeva, M., & Zhurakulov, S. Pharmacological Evaluation of F45 on the Cardiovascular System Using In Vitro, In Vivo Models and Molecular Dockings. Trends in Sciences, 2025; 22(12), 10924.
CrossRef - Abdullaev I.Z., Gayibov U.G., Omonturdiev S.Z., Azamjonovna S.F., Gayibova S.N and Aripov T.F. Molecular pathways in cardiovascular disease under hypoxia: Mechanisms, biomarkers, and therapeutic targets. The Journal of Biomedical Research., 2025; 39(2), 1-16.
CrossRef - Saidova K., Askarov I., Islomov A., Inkhonova A and Ishimov U. Development and HPLC characterization of a water-soluble supramolecular complex of propolis and glycyrrhizic acid monoammonium salt: Composition, toxicity, and pharmaceutical potential. Biomedical and Pharmacology Journal., 2025; 18(1), 1017–1029.
CrossRef - Abdullaev A.A., Inamjanov D.R., Abduazimova D.S., Omonturdiyev S.Z., Gayibov U.G., Gayibova S.N and Aripov T.F. Sílybum mariánum’s impact on physiological alterations and oxidative stress in diabetic rats. Biomedical and Pharmacology Journal., 2024; 17(2), 1291-1300.
CrossRef - de la Cerda-Carrasco A., López-Solís R., Nuñez-Kalasic H., Peña-Neira Á and Obreque-Slier E. Phenolic composition and antioxidant capacity of pomaces from four grape varieties (Vitis vinifera L.). Journal of the science of food and agriculture., 2015; 95(7), 1521–1527.
CrossRef - Gaibullayeva O., Islomov A., Abdugafurova D., Elmurodov B., Mirsalixov B., Mahmudov L., Abdullaev I., Baratov K., Omonturdiev S and Sa’dullayeva S. Inula helenium L. root extract in sunflower oil: Determination of its content of water-soluble vitamins and immunity-promoting effect. Biomedical and Pharmacology Journal., 2024; 17(4), 2729-2737.
CrossRef - Azimova A.Q.Q., Islomov AX., Maulyanov S.A., Abdugafurova D.G., Mahmudov L.U., Abdullaev I.Z., Ishmuratova A.S., Siddikova S.Q.Q and Askarov I.R. Determination of vitamins and pharmacological properties of Vitis vinifera L. plant fruit part (mixed varieties) syrup-honey. Biomedical and Pharmacology Journal., 2024; 17(4), 2779-2786.
CrossRef - Caponio GR, Minervini F, Tamma G, Gambacorta G, De Angelis M. Promising Application of Grape Pomace and Its Agri-Food Valorization: Source of Bioactive Molecules with Beneficial Effects. Sustainability. 2023; 15(11):9075.
CrossRef - Omonturdiyev S.Z., Abdullaev I.Z., Inamjanov D.R., Mahmudov L.U., Zaripova M.R., Gayibova S.N., Gayibov U.G and Aripov T.F. The effect of Ájuga Turkestánica on the rat aortic smooth muscle ion channels. Biomedical & Pharmacology Journal., 2024; 17(2), 1213-1222.
CrossRef - Inamjanov D.R., Abdullaev I.Z., Omonturdiyev S.Z., Abdullaev A.A., Mahmudov L.U., Zaripova M.R., Abdullayeva M.O., Abduazimova D.S., Menglieva Sh.Yu, Gayibova S.N., Sadbarxon M., Gayibov U.G and Aripov T.F. In vitro and in vivo studies of crategus and inula helenium extracts: Their effects on rat blood pressure. Trends in Sciences., 2025; 22(3), 9158.
CrossRef - Abdullaev A., Abdullaev I., Bogbekov A., Gayibov U., Omonturdiev S., Gayibova S., Turahodjayev M., Ruziboev K and Aripov T. Antioxidant potential of rhodiola heterodonta extract: Activation of Nrf2 pathway via integrative in vivo and in silico studies. Trends in Sciences., 2025; 22(5), 9521.
CrossRef - Saydullayevna, I.A., Abdugafurova D.G., Islomov A.H., Mahmudov L.U., Baratov K.R and Azimova A.Q. The study of the biologically active effect of the Rubia tinctorum L. plant on rats with experimental kidney stone disease and issues of introduction. Biomedical and Pharmacology Journal., 2024; 17(3), 2035–2042.
CrossRef - Md. Mamunur Rashid, Shariful Islam, Md. Nazim Uddin, Md. Zia Uddin Al Mamun, Md. Jaynal Abedin, Md. Habibur Rahman Bhuiyan, Md. Abdus Satter Miah. HPLC-DAD analysis of water-soluble vitamins (B1, B2, B3, B5, B6, C and Biotin) and fat-soluble vitamins (A, D, E, K1 and β-carotene) in commonly consumed pulses in Bangladesh. Applied Food Research., 2024; 100424,
CrossRef - Li Q., Lv L., Liu Y., Fang Z., Deng Q., Liang W., Wu Y and Chen Z. Preparation, characterization and application of bacteriocin CAMT6 nanoliposomes using resveratrol as a novel stabilizer. Food chemistry., 2023; 403, 134293.
CrossRef - Xu, K., Aigensberger, M., Berthiller, F. et al. Comparative evaluation of phenyl isothiocyanate derivatization and “dilute-and-shoot” methods for HPLC–MS/MS-based targeted metabolomics analysis of amine-containing metabolites in plasma samples. Analytical and bioanalytical chemistry., 2025; 417, 5859–5872
CrossRef. - Y Umidakhon, B Erkin, G Ulugbek, et al. Correction of the mitochondrial NADH oxidase activity, peroxidation and phospholipid metabolism by haplogenin-7-glucoside in hypoxia and ischemia. Trends Sci 2022, 19(21), 6260.
CrossRef - Al-Khrasani M., Karadi D.A., Galambos A.R., Sperlagh B and Vizi E.S. The pharmacological effects of phenylephrine are indirect, mediated by noradrenaline release from the cytoplasm. Neurochemical Research., 2022; 47(11), 3272-3284.
CrossRef - Gayibov U.G., Gayibova S.N., Karimjonov H.M., Abdullaev A.A., Abduazimova D.S., Rakhimov R.N., Ruziboev H.S., Xolmirzayeva M.A., Zaynabiddinov A.E and Aripov T.F. Antioxidant and cardioprotective properties of polyphenolic plant extract of Rhus glabra L. Plant Science Today., 2024; 11(3), 655-662.
CrossRef - Aripov T.F., Gayibov U.G., Gayibova S.N and Abdullaev A.A. Antiradical and antioxidant activity of the preparation “Rutan” from Rhus coriaria L. Journal of Theoretical and Clinical Medicine., 2024; 4, 138-147.
CrossRef - Mahmudov A.V., Abduraimov O.S., Erdonov S.B., Allamurotov A.L., Mamatqosimov O.T., Gayibov U.G and Izotova I. Seed productivity of Linum usitatissimum L. in different ecological conditions of Uzbekistan. Plant Science Today., 2022; 9(4), 1090-1101.
CrossRef - Kojima H., Nakada T., Yagami A., Todo H., Nishimura J., Yagi M., Yamamoto K., Sugiyama M., Ikarashi Y., Sakaguchi H., Yamaguchi M., Hirota M., Aizawa S., Nakagawa S., Hagino S and Hatao M. A step-by-step approach for assessing acute oral toxicity without animal testing for additives of quasi-drugs and cosmetic ingredients. Current research in toxicology., 2022; 4 (2), 100100.
CrossRef - Choi J., Ryu S.J., Kim K.J., Kim H.M., Chung H.C and Lee B.Y. Single, 14-Day, and 13-Week Repeated Dose Toxicity Studies of Daily Oral Gelidium elegans Extract Administration to Rats. Molecules., 2018; 23(1), 217
CrossRef - Abd-Allah A.A.M., El-Deen N.A.M.N., Mohamed, W.A.M and Naguib F.M. Mast cells and pro-inflammatory cytokines roles in assessment of grape seeds extract anti-inflammatory activity in rat model of carrageenan-induced paw edema. Iranian journal of basic medical sciences., 2018; 21(1), 97–107.
CrossRef - Zaripova M., Abdullaev I., Bogbekov A., Gayibov U., Omonturdiev S., Makhmudov R., Ergashev N., Jabbarova G., Gayibova S and Aripov T. In Vitro and in Silico Studies of Gnaphalium U. Extract: Inhibition of α-amylase and α-glucosidase as a Potential Strategy for Metabolic Syndrome Regulation. Trends in Sciences., 2025; 22(8), 10098.
CrossRef - Gayibov U.G., Komilov E.J., Rakhimov R.N., Ergashev N.A., Abdullajanova N.G., Asrorov M.I and Aripov T.F. Influence of new polyphenol compound from Euphorbia plant on mitochondrial function. Journal of Microbiology Biotechnology and Food Sciences., 2019; 8(4): 1021-1025.
CrossRef - Sayidaliyeva, R., Kadirova, S., Zaynabiddinov, A., Abdullaev, I., Makhmudov, L., Gayibov, U., Yuldasheva, M., Kholmirzayeva, M., Rakhimov, R., Mutalibov, A., & Karimjonov, H. A-51 as A Natural Calcium Channel Blocker: An Integrative Study Targeting Hypertension. Trends in Sciences, 22(11), 10760.
CrossRef - Gayibov UG, Zoirovich OA, Fotima AS, et al. Plant-derived and synthetic antihypoxic agents in cardiovascular diseases: mechanisms, key pathways and therapeutic potential. Plant Sci Today. (Early Access). https://doi.org/10.14719/pst.7810
CrossRef - Sobenin I.A., Myasoedova V.A., Iltchuk M.I., Zhang D.W and Orekhov A.N. Therapeutic effects of garlic in cardiovascular atherosclerotic disease. Chinese journal of natural medicines., 2019; 17(10), 721,728.
CrossRef - Pozilov M.K., Gayibov U.G., Asrarov M.I., Abdulladjanova N., Ruziboev H.S and Aripov T.F. Physiological alterations of mitochondria under diabetes condition and its correction by polyphenol gossitan. Journal of Microbiology Biotechnology and Food Sciences., 2022; 12(2), e2224.
CrossRef - Li, Minyu & Zhang, Mengjiao & Fatixovich, Aripov & Gayibov, Ulugbek & Du, Hongying. Green-synthesized Zn²⁺-polyphenol networks (CGA/RA) for enhanced multifunctional food preservation. Journal of Food Measurement and Characterization. 2025; 1-17.
CrossRef - Shakiryanova Z., Khegay R., Gayibov U., Saparbekova A., Konarbayeva Z., Latif1 A and Smirnova O. Isolation and study of a bioactive extract enriched with anthocyanin from red grape pomace (Cabernet Sauvignon). Agronomy Research., 2023; 21(3), 1293-1303.
- Gayibov U.G., Gayibova S.N., Pozilov M.M.K., Tuxtaeva F.S., Yusupova U.R., Djabbarova G.M-K., Mamatova Z.A., Ergashev N.A and Aripov T.F. Influence of quercetin and dihydroquercetin on some functional parameters of rat liver mitochondria. Journal of microbiology, biotechnology and food sciences., 2021; 11(1), e2924.
CrossRef - Khasanov, A., Abdullaev, I., Kadirova, S. ., Mamajanov, M., Zaynabiddinov, A., Omonturdiev, S., Makhmudov, L., Inomjonov, D., Gayibov, U., Esanov, R. ., & Matchanov, A. N-2 Polyphenol Targets Vascular Calcium Channels to Exert Antihypertensive Effects: In Vitro and In Vivo Evaluation. Trends in Sciences, 2025; 22(12), 10782.
CrossRef - Zaripova M., Gayibova S., Makhmudov R., Mamadrahimov A., Vypova N., Gayibov U., Miralimova S and Aripov T. Characterization of Rhodiola heterodonta (Crassulaceae): Phytocomposition, antioxidant and antihyperglycemic activities. Preventive Nutrition and Food Science., 2024; 29(2), 135-145.
CrossRef - Abdulkhakova G.V., Komilov E.J., Abdullaev I.Z., Ergashev N.A., Makhmudov R.R and Asrorov M.I. Tannins as Modulators in the Prevention of Mitochondrial Dysfunction. Trends in Sciences., 2025; 22(8), 10436.
CrossRef - Mamajanov, M., Abdullaev, I., Sotimov, G., Mavlanova, S., Niyozov, Q., Mirzaolimov, M., Najimov, A., Mirzaolimov, E., Raximberganov, M, & Abdullayev, U. Mitochondrial and Pharmacokinetic Insights into 3,5,7,2’,6’-Pentahydroxyflavanone: Respiratory Modulation, Calcium Handling, and Membrane Stability. Trends in Sciences, 2025;22(12), 10984.
CrossRef







