Manuscript accepted on :23-10-2025
Published online on: 06-11-2025
Plagiarism Check: Yes
Reviewed by: Dr. Jagadish K
Second Review by: Dr. Hany Akeel
Final Approval by: Dr. Anton R Keslav
Karthik Kadhiri Karunakar,1* Binoy Varghese Cheriyan,2*
Binoy Varghese Cheriyan,2* Bhavya E1
Bhavya E1 , Akshaya Murugathirumal1
, Akshaya Murugathirumal1 , Abinaya Senthilkumar1
, Abinaya Senthilkumar1 , Ragavendran Anandakumar1
, Ragavendran Anandakumar1 , Gnanisha Murugan1
, Gnanisha Murugan1 , Krithikeshvaran Rajasekaran1
, Krithikeshvaran Rajasekaran1 , Rajeshkumar Shanumugam3
, Rajeshkumar Shanumugam3 , Sowmyalakshmi Venkataraman4
, Sowmyalakshmi Venkataraman4 , Nandhini Jayaprakash1
, Nandhini Jayaprakash1 and Lokeshvar Ravikumar1
and Lokeshvar Ravikumar1
1Department of Pharmacy Practice, Saveetha College of Pharmacy, Saveetha Institute of Medical and Technical Sciences, Chennai, Tamilnadu, India
*2 Department of Pharmaceutical Chemistry, Saveetha College of Pharmacy, Saveetha Institute of Medical and Technical Sciences, Chennai, Tamilnadu, India
3 Nano biomedicine Lab, Department of Pharmacology, Saveetha Dental College and Hospitals, Saveetha Institute of Medical and Technical Sciences, Chennai, Tamilnadu, India
4Department of Pharmaceutical Chemistry, Sri Ramachandra Faculty of Pharmacy, SRIHER, Chennai, Tamilnadu, India.
Corresponding Author lallybinoy@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3309
Abstract
Oral cancer is an aggressive malignancy that typically originates in surrounding tissues and spreads to the cervical lymph nodes, with no effective chemotherapy currently available to prevent its progression. Recent studies have explored selenium-containing compounds as potential chemotherapeutic agents. This study used a biological approach to synthesis bioinspired selenium nanoparticles (SeNPs), which were then analyzed using Fourier Transform Infrared Spectroscopy (FTIR), Ultraviolet-Visible (UV-Vis) spectroscopy, Energy Dispersive X-ray Spectroscopy (EDX), and Scanning Electron Microscopy (SEM).Their anticancer and anti-inflammatory activities were evaluated against human oral squamous cell carcinoma (HOSCC) KB-1 cell lines using the egg albumin assay, MTT assay, and cytotoxicity assay at concentrations of 15 and 31 µg/ml. The SeNPs significantly reduced cell viability, induced apoptosis, and exhibited potent anti-inflammatory effects. Morphological alterations, such as cytoplasmic membrane blebbing and cell shrinkage, were observed under both inverted phase contrast and fluorescence microscopy, indicating apoptotic cell death. These findings demonstrate that bioinspired selenium nanoparticles effectively induce cytotoxicity and apoptosis in HOSCC KB-1 cells, highlighting their potential as promising agents for oral cancer therapy.
Keywords
Anticancer Agent; Biological Synthesis; Nanotechnology; Oral Cancer; Selenium Nanoparticles (SeNPs)
Download this article as:| Copy the following to cite this article: Karunakar K. K, Cheriyan B. V, Bhavya E, Murugathirumal A, Senthilkumar A, Anandakumar R, Murugan G, Rajasekaran K, Shanumugam R, Venkataraman S, Jayaprakash N, Ravikumar L. Bioinspired Selenium Nanoparticles as a Potential Anticancer Agent in Oral Squamous Cell Carcinoma: Apoptosis-Induced Cytotoxicity. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Karunakar K. K, Cheriyan B. V, Bhavya E, Murugathirumal A, Senthilkumar A, Anandakumar R, Murugan G, Rajasekaran K, Shanumugam R, Venkataraman S, Jayaprakash N, Ravikumar L. Bioinspired Selenium Nanoparticles as a Potential Anticancer Agent in Oral Squamous Cell Carcinoma: Apoptosis-Induced Cytotoxicity. Biomed Pharmacol J 2025;18(4). Available from: https://bit.ly/3XesYiB |
Introduction
Oral cancer is one of the most aggressive malignancies, often originating in the mucosal tissues and metastasizing to the cervical lymph nodes. Despite advancements in cancer treatment, OSCC, or oral squamous cell carcinoma, is still a serious health concern due to its high mortality rate and limited therapeutic options. Conventional chemotherapy and radiation therapy are often associated with severe side effects and poor patient outcomes, necessitating the search for novel, effective, and biocompatible treatment strategies.1 Selenium (Se), an essential trace element, has gained considerable because of its strong anticancer effects, it has received attention in cancer research. Selenium-containing compounds, including selenium nanoparticles (SeNPs), have demonstrated significant promise as chemotherapeutic agents because of their capacity to trigger cell death, modulate oxidative stress, and inhibit tumor growth.2 Compared to conventional selenium compounds, SeNPs offer superior bioavailability, reduced toxicity, and enhanced therapeutic efficacy as mentioned in the study. The biogenic synthesis of SeNPs using plant extracts has emerged as a sustainable and eco-friendly approach, providing additional bioactive compounds that may further enhance their therapeutic potential.3 This work utilised extracts of Mangifera indica and Beta vulgaris as reducing agents for the biological synthesis of selenium nanoparticles (SeNPs) and assessed their anticancer efficacy against human oral squamous cell carcinoma (HOSCC) KB-1 cell lines. The synthesised SeNPs were characterised by FTIR, UV-Vis spectroscopy, Energy Dispersive X-ray Spectroscopy (EDX), and Scanning Electron Microscopy (SEM) to verify their structural and chemical properties. The anticancer activity of SeNPs was evaluated using in vitro cell viability (MTT assay), cytotoxicity (cytotoxicity assay), and anti-inflammatory (egg albumin assay) studies. Additionally, we explored the mechanism of SeNP-induced cytotoxicity in KB-1 cells by analysing apoptotic markers, such as cell shrinkage and cytoplasmic membrane blebbing, utilising an inverted phase contrast and fluorescent microscopy. Our research indicates that bioinspired SeNPs elicit apoptosis-dependent cytotoxicity, positioning them as a potential therapy option for oral cancer. This research underscores the therapeutic promise of bioinspired SeNPs as a novel anticancer agent and elucidates their molecular effect in oral cancer cells.
Materials and Methods
Plant material confirmation
Mangifera indica leaves and Beta Vulgaris root were obtained from a garden in Tamil Nadu, India. The leaves and roots were meticulously studied by a botanist from the Siddha Central Research Institute in Arumbakkam, Chennai, India, in order to precisely identify them. A botanist correctly recognized a herbarium specimen after it was meticulously prepared and assigned a unique registration number (PCOG002-ACF).
Plant extract preparation
The plants Mangifera indica and Beta vulgaris were obtained from the garden and were carefully cleansed twice using running water and Mil-Q water. Following this procedure, the leaves of Mangifera indica and Beta vulgaris were shade-dried at room temperature for 7–10 days, then pulverized into coarse powder using a mechanical grinder. Approximately 50 g of M. indica leaf powder and 50 g of B. vulgaris root powder were combined in a conical flask containing 1 L of distilled water. The mixture was heated for 20 minutes at 80 °C using a heating mantle to release phytochemicals. After that, the flask was heated for 20 minutes to 80°C in a heating mantle configuration. This makes it possible for the powder’s phytochemicals to become active. Whatman filter paper was used to filter the extract after it had been boiled and centrifuged at 5000 rpm for 10 minutes to remove particulate matter. The clarified supernatant was collected and stored at 4 °C until further use in nanoparticle synthesis..4
Preparation of sodium selenite
In this process, A stock solution of 2 mM sodium selenite (Na₂SeO₃; Sigma-Aldrich, ≥99% purity) is dissolved in 70 ml conical flask containing distilled water, and the mixture is maintained in an orbital shaker for 1 hour. The precursor will indicate the colour change.5
Synthesis of selenium nanoparticles
A 30 ml amount of plant extract was incorporated into a pre-prepared 70 mL solution of 2 mM sodium selenite (Na₂SeO₃; Sigma-Aldrich, ≥99% purity). The mixture was stirred magnetically for one hour at a speed of 380–420 revolutions per minute (rpm) and simultaneously placed in a shaker container to enhance particle mixing for the green synthesis process. The conversion of sodium selenite to selenium nanoparticles (SeNPs) was indicated by a visible colour change and further confirmed using UV-Vis spectroscopy (200–800 nm) at 24-hour intervals throughout a three-day period. Subsequently, the SeNPs solution underwent centrifugation at 8000 rpm for 10 minutes utilising a Lark cooled centrifuge. The resultant particles were gathered, rinsed twice with distilled water, and meticulously purified. The resulting pure pellet was further dried at temperatures between 100°C and 150°C for a 24-hour period. The dried nanoparticle powder was then collected and kept in a securely sealed Eppendorf jar at 4 °C.6
Characterization of selenium nanoparticle
To improve the synthesis parameters of selenium nanoparticles (SeNPs) using extracts from Mangifera indica and Beta vulgaris, the environmentally friendly synthesised SeNPs were examined by several characteriszation procedures. Optimal synthesis conditions were established by UV-Vis spectroscopy. The size and shape of the SeNPs were examined using scanning electron microscopy (SEM). Various levels of magnification were used to analyze the samples. Moreover, the elemental makeup of the SeNPs was investigated using energy dispersive X-ray spectroscopy (EDX).7
Anti-inflammatory activity
BSA Assay
Denaturation of BSA and egg albumin was employed to evaluate the anti-inflammatory effects of green selenium nanoparticles To denature BSA, 0.45 mL of 1% BSA solution (Himedia, India) was combined with 0.05 mL of SeNP suspensions synthesized from Mangifera indica and Beta vulgaris at concentrations ranging between 10 and 50 µg/mL. The pH of the mixture was adjusted to 6.3. The samples were first kept at room temperature for 10 min and then incubated in a water bath at 55 °C for 30 min. Dimethyl sulfoxide (DMSO) served as the control, while diclofenac sodium (100 µg/mL) was used as the reference standard. After cooling, absorbance was recorded at 660 nm using a Shimadzu UV-1800 spectrophotometer (Kyoto, Japan).8
The percentage of protein denaturation was determined utilizing the following equation:

Egg Albumin denaturation assay
The denaturation of egg albumin was employed to evaluate the anti-inflammatory effects of selenium nanoparticles (SeNPs).Fresh hen’s eggs were obtained from a local poultry supplier in Chennai, India, and albumin was separated immediately before use. The reaction mixture contained 0.2 mL of freshly prepared egg albumin and 2.8 mL of phosphate buffer (pH 6.3), to which SeNPs synthesized from Mangifera indica and Beta vulgaris were added at concentrations ranging from 10 to 50 µg/mL. The samples were first kept at room temperature for 10 min and subsequently incubated in a water bath at 55 °C for 30 min. Diclofenac sodium (100 µg/mL) served as the standard drug, while dimethyl sulfoxide (DMSO) was used as the control. After cooling, the absorbance was measured at 660 nm using a Shimadzu UV-1800 spectrophotometer (Kyoto, Japan).9
Percentage of protein denaturation was determined utilizing the following equation:

Maintenance of the cell line
Oral squamous cell carcinoma (KB-1) cell lines were obtained from the National Centre for Cell Science (NCCS, Pune, India). The cells were cultured in Dulbecco’s Modified Eagle Medium (DMEM; HiMedia, India) supplemented with 10% fetal bovine serum (FBS; Gibco, USA) and 1% antibiotic solution (penicillin 100 U/mL and streptomycin 100 µg/mL). Cultures were maintained in T25 flasks at 37 °C in a humidified atmosphere containing 5% CO₂. Sub-culturing was carried out by trypsinization with 0.25% trypsin-EDTA when the cells reached 80–90% confluence. The cells were then passaged into fresh culture flasks for further experiments.10
Cell viability assay
The MTT assay assessed oral cancer cell viability after treatment with M. Indica leaves and B. vulgaris SeNPs at 7.5 and 180 µg/mL doses. Insoluble purple formazan crystals are formed by soluble yellow tetrazolium salt is being converted by metabolically active cells. For plating, 96-well plates with 5 × 10³ KB cells per well were used. Following plating, cells were incubated for three hours at 37 °C in serum-free medium to synchronize growth and exhaust nutrients, as reported in previous literature. Twenty-four hours later, two 100 µL washes with serum-free medium were carried out. Cells were subjected to doses of M. Indica Leaves + B. vulgaris SeNPs (7.5–180 µg/mL) for 24 h following starvation. After treatment, 100 µL of DMEM containing 0.5 mg/mL MTT was added to each well, including treated and untreated control cells. Plates were incubated for four hours at 37 °C in a CO₂ incubator. Following incubation, the MTT-containing medium was carefully removed, and the cells were washed once with PBS. The formazan crystals formed were dissolved in 100 µL of DMSO, and the mixture was incubated in the dark for 1 h. Absorbance was then measured at 570 nm using a Micro ELISA plate reader. The ratio of absorbance of treated samples to control cells was used to determine cell viability. Untreated control cells showed 100% viability. The following formula assesses the vitality of cells.11
% cell viability = A570 nm of treated cellsA570 nm of control cells×100.
Morphology Analysis
The MTT experiment determined the optimal dosage (IC₅₀: 31 µg/mL) for further analysis. Cell morphological changes were examined using phase-contrast microscopy. M. indica leaves and B. vulgaris SeNPs were applied to 6-well plates seeded with 2 × 10⁵ cells at doses of 15 and 31 µg/mL for 24 h. After incubation, cells were washed with PBS (pH 7.4) to remove the medium. Plates were then examined under phase-contrast microscopy.12
Determination of mode of cell death by ethidium bromide (EtBr) dual staining
The study examined the effect of M. Indica leaves and B. Vulgaris SeNPs (15 and 31 µg/ml) on the mortality of oral cancer cells using AO/EtBr dual staining. Cold PBS was used to wash the cells after they had been treated for 24 hours with M. Indica leaves and B. vulgaris SeNPs (15 and 31 µg/ml). Pellets were reconstituted in 5 µl of ethidium bromide (1 mg/mL) and acridine orange (1 mg/mL). The apoptotic alterations of the labeled cells were subsequently assessed using a fluorescence microscope.13
Statistical analysis
The complete dataset was analysed via PRISM software, initially employing ANOVA in one way, then the student’s t-test. The findings are displayed as mean ± standard deviation (SD) in triplicate. Statistical significance was defined as a p-value of less than 0.05.14
Results
Result: Visual approval of the green synthesis of SeNPs
Green synthesis was used to make selenium nanoparticles (SeNPs) using [32] Mangifera indica and Beta vulgaris extracts as reducing and stabilizing agents. Plants aid synthesis in this method. The hue of Mangifera indica and Beta vulgaris extracts has declined from bright yellow to dark greyish-brown due to phytochemicals. Their hue affects the production and nucleation of selenium nanoparticles (SeNPs), which is crucial to their manufacture. A noticeable color change from light yellow to brownish-yellow indicates rapid SeNP development and clustering, according to the study. The brown color comes from surface plasmon resonance and the transition of Ag+ ions into Ag0 in liquid extracts. According to reference, a color shift in SeNPs-containing reaction solutions indicates metal ion reduction. The hue change during selenium nanoparticle generation can be utilized to track the chemical process. To validate its accuracy, an ultraviolet-visible spectral analysis was done after visual inspection.
UV-Visible Spectroscopy
The selenium ions in the aqueous solution of the selenium complex underwent reduction when exposed to extracts from Mangifera indica and Beta vulgaris. This reduction was clear from the color shift, which went from colorless to pale yellowish and then to reddish-brown. The observed alteration in colour signifies the formation of selenium nanoparticles as a result of the excitation of surface plasmon oscillations. The UV-visible absorption spectrum of the SeNPs exhibited a distinct peak at around 420 nm, providing confirmation of the presence of selenium nanoparticles. The magnitude of the absorption peak grew with time, suggesting a gradual rise in the concentration of nanoparticles. The persistent location of the peak over a period of time indicates the enduring stability of the synthesized nanoparticles. This finding aligns with prior studies on the production of SeNPs using plant extracts, which have consistently shown a peak of surface plasmon resonance at roughly 420 nm. The absorbance of selenium nanoparticles is shown in Figure 1 (SeNPs) at three distinct time intervals: 1 hour, 12 hours, and 24 hours.
 |
Figure 1: illustrates the use of a UV-visible spectrophotometer with a 1 nm resolution in the 400–500 nm range. The wavelength is displayed on the X-axis in nanometres (nm), while the absorbance is shown on the Y-axis. |
Scanning Electron Microscopy Analysis
Scanning electron microscopy (SEM) was used to examine the size distribution and surface appearance of the biologically synthesized selenium nanoparticles (SeNPs). The presence of both single selenium nanoparticles and clusters of nanoparticles was evident from the SEM images. Most of the particles were spherical in shape, with an average size of about 18 nm and diameters ranging from 10 to 25 nm. The occurrence of many secondary metabolites in the extracts of Mangifera indica and Beta vulgaris is accountable for the formation of aggregates. These metabolites function as agents that reduce and stabilize. The SEM investigation provided clear images of the selenium nanoparticles (SeNPs), highlighting their spherical morphology and uniform size. Moreover, there was a noticeable degree of particle agglomeration, The data suggest that the selection of plant extract significantly influences the nanoparticles’ dimensions and form. The histogram analysis of the particle size distribution confirmed that most particles were within the required size range, supporting the findings obtained using scanning electron microscopy (SEM) mentioned in Figure 2, illustrating the surface properties and dispersion of the SeNPs.
 |
Figure. 2: Illustrates SEM, used to examine the surface appearance and size distribution of biosynthesized selenium nanoparticles (SeNPs). The photos displayed spherical selenium nanoparticles (SeNPs) has an average size of 18 nm and a size range of 10–25 nm. |
EDX
The elemental composition of the selenium nanoparticles (SeNPs) synthesised during biosynthesis was analyzed using spectroscopy of energy dispersive X-rays (EDX). Metallic selenium’s existence as the main element was confirmed by the EDX spectrum, which showed a distinct signal at 3 keV, corresponding to the selenium (Se) L line. Furthermore, the EDX examination revealed the presence of supplementary elements, namely oxygen (O), carbon (C), potassium (K), chlorine (Cl), calcium (Ca), and aluminum (Al) as mentioned in figure 3. The existence of these constituents can be ascribed to the extracts derived from Mangifera indica and Beta vulgaris, indicating that the plant biomolecules attached to the surface of the nanoparticles and contributed to their stability. The EDX spectrum shows a distinct selenium peak, confirming the successful synthesis of SeNPs. The supplementary peaks detected in the spectrum correspond to constituents present in the plant extracts. These elements function as capping agents, enhancing the stability of the nanoparticles. Organic materials including proteins, polyphenols, and flavonoids are indicated by the presence of carbon and oxygen peaks. These chemicals have crucial roles in reducing and stabilizing selenium nanoparticles. The EDX elemental composition analysis reveals a significant amount of Selenium, indicating a successful reduction of Se⁺ ions to Se⁰ nanoparticles. The presence of carbon, oxygen, and other trace elements indicates that plant biomolecules have a role in stabilizing nanoparticles and preventing them from clumping together. This discovery is consistent with previous studies that used plant extracts to create and improve the durability of selenium nanoparticles 15. The EDX results confirm the findings from the UV spectroscopy and SEM studies, offering additional proof that the biosynthesized SeNPs mostly consist of selenium and are accompanied by a range of organic compounds obtained from the plant extracts. The elemental composition investigation reveals that extracts from Mangifera indica and Beta vulgaris provide a substantial contribution to the environmentally sustainable manufacture of stable and biocompatible selenium nanoparticles.
 |
Figure 3: A highly porous microstructure is seen in a SEM image, and the presence of metallic selenium (Se) in the EDX spectroscopy was used to confirm the presence of nanoparticles, which produced a significant signal at 3 keV. |
FTIR
The Fourier Transform Infrared Spectroscopy (FTIR) analysis was carried out to determine which functional groups were present on the surface of the selenium nanoparticles that were biosynthesized (SeNPs) and to gain insight into the possible biomolecules responsible for their synthesis and stabilization16. The FTIR spectrum, as shown in the figure 5, displays prominent absorption bands at several wavenumbers, indicating the presence of various functional groups.
A broad peak observed at 3390.59 cm⁻¹ corresponds to O-H stretching vibrations of the hydroxyl groups, indicating that the reduction and capping of the nanoparticles were aided by polyphenols or alcohol groups, which are frequently present in plant extracts. Aliphatic hydrocarbons’ C–H stretching vibrations are responsible for the peak at 2922.78 cm⁻¹. C=O stretching is indicated by a clear peak at 1690.01 cm⁻¹, which suggests the presence of carbonyl groups, which may also contribute to nanoparticle stabilization. Further, peaks at 1334.93 cm⁻¹ and 1034.65 cm⁻¹correspond to the C–O stretching of alcohols or esters and the C–N stretching of amine groups, respectively. These peaks verify the existence of several biomolecules, including proteins, flavonoids, and polysaccharides, which most likely served as stabilizing and reducing agents during the environmentally friendly synthesis of SeNPs17. Overall, the FTIR analysis supports the successful biosynthesis of selenium nanoparticles and reveals the presence of key functional groups that contribute to their stability and potential biological activity, including anticancer effects18.
 |
Figure 4: The FTIR spectrum of selenium nanoparticles shows transmittance (%) as a function of wavenumber (cm⁻¹), identifying key functional groups that the sample contains. |
Anti-inflammatory activity
The denaturation assays for egg albumin (EA) and bovine serum albumin (BSA) were used to assess the anti-inflammatory properties of M. indica and beta vulgaris nanoparticles. Protein denaturation is a well-known marker of inflammation, and agents that inhibit this process can exhibit potential anti-inflammatory properties. In the BSA assay, selenium nanoparticles (SeNPs) derived from M. Indica & Beta Vulgaris showed a concentration-dependent inhibition of protein denaturation, increasing from 45% at 10 µg/ml to 85% at 50 µg/ml, surpassing the standard control at each concentration. Similarly, in the EA assay, selenium nanoparticles (SeNPs) exhibited strong inhibitory effects, ranging from 70% at 10 µg/ml to 85% at 50 µg/ml, indicating a consistent protective effect against protein denaturation. These findings suggest that M. Indica & Beta Vulgaris-derived nanoparticles possess significant anti-inflammatory properties, likely due to their interaction with protein structures and stabilization against heat-induced denaturation. The results highlight their potential as therapeutic agents for inflammation-related disorders, warranting further mechanistic and in vivo evaluations. Figure 6 depicts graphical presentation of assays.
Various concentrations were used to measure the inhibitory activity (10–50 µg/ml) for M. Indica & Beta Vulgaris nanoparticles (SeNPs for BSA and EA), compared to the standard reference. Results indicate a dose-dependent increase in inhibition, with M. Indica & Beta Vulgaris selenium nanoparticles demonstrating comparable or superior inhibitory activity to the standard across all concentrations.
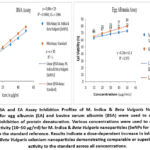 |
Figure 5: BSA and EA Assay Inhibition Profiles of M. Indica & Beta Vulgaris Nanoparticles. The assays for egg albumin (EA) and bovine serum albumin (BSA) were used to measure the percentage inhibition of protein denaturation. |
Cytotoxic Effects of Selenium Nanoparticles on KB-1 Oral Cancer Cells
Nanoparticles have emerged as promising therapeutic agents in cancer treatment due to their selective cytotoxicity and biocompatibility. In this work, we examined the cytotoxic capability of selenium nanoparticles (SeNPs) made from extracts of B. vulgaris and M. indica leaves against KB-1 oral cancer cells. Cell viability after a 24-hour exposure to varying doses of SeNPs was assessed using the MTT test. The findings showed a dose-dependent, significant decrease in cell viability, with doses > 30 µg/ml demonstrating strong cytotoxic effects. The IC50 value was determined to be 31.08 ± 4.952 µg/ml, indicating an effective concentration at which 50% of the cancer cells were inhibited. At higher doses (≥ 120 µg/ml), a drastic decrease in cell viability was observed, suggesting apoptosis-induced cell death as depicted in figure 7 and 8.
These results demonstrate the potential of bioinspired SeNPs as a strong therapeutic agent for the treatment of oral cancer. The cytotoxic mechanism may involve oxidative stress induction, mitochondrial dysfunction, and apoptotic pathway activation, warranting further molecular investigations.
Fig.6. Cytotoxic effect of Mangifera indica (M. indica) leaves and Beta vulgaris (B. vulgaris) selenium nanoparticles (SeNPs) KB-1 oral cancer cells, as determined by the MTT test. Cell viability was assessed using the MTT test after KB-1 cells were exposed to different concentrations of M. indica + B. vulgaris SeNPs (7.5–180 µg/ml) for a full day. The percentage of viable cells in comparison to the untreated control is shown in the bar graph. There was a dose-dependent reduction in cell viability, with higher concentrations of SeNPs (≥ 30 µg/ml) significantly reducing cell survival. The half-maximal inhibitory concentration (IC50) was determined to be 31.08 ± 4.952 µg/ml, suggesting a potent cytotoxic effect. Data are presented as mean ± standard deviation (SD) from three independent experiments (n = 3). Asterisks (*) indicate statistically significant differences (p < 0.05) compared to the control group.
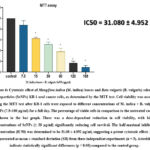 |
Figure 6: Cytotoxic effect of Mangifera indica (M. indica) leaves and Beta vulgaris (B. vulgaris) selenium nanoparticles (SeNPs) KB-1 oral cancer cells, as determined by the MTT test. |
(A–C) Phase-contrast microscopy images of KB-1 cells after 24-hour treatment with M. indica + B. vulgaris SeNPs at different concentrations. Normal morphology is displayed by untreated control cells. (B) 15 µg/ml-treated cells (C) Cells treated with 31 µg/ml SeNPs exhibiting a significant reduction in cell number, along exhibited noticeable cytoplasmic membrane blebbing and cell shrinkage, which are signs of apoptosis (D–F) Ethidium bromide (EtBr) staining of apoptotic cells observed under an inverted fluorescence microscope. (D) Control cells showing minimal fluorescence, indicating low levels of apoptosis. (E) Cells treated with 15 µg/ml SeNPs showing increased fluorescence, suggestive of early apoptotic changes. (F) Cells treated with 31 µg/ml SeNPs displaying intense fluorescence, signifying a high level of apoptosis.
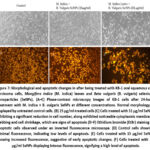 |
Figure 7: Morphological and apoptotic changes in after being treated with KB-1 oral squamous cell carcinoma cells, Mangifera indica (M. indica) leaves and Beta vulgaris (B. vulgaris) selenium nanoparticles (SeNPs). |
Discussion
Our study demonstrates that bioinspired selenium nanoparticles (SeNPs) exhibit significant anticancer properties against human oral squamous cell carcinoma (HOSCC) KB-1 cell lines, primarily through the induction of apoptosis. These findings align with existing literature on the anticancer potential of selenium-based compounds and provide new insights into their application in oral cancer therapy. The observed morphological changes in KB-1 cells, such as cell shrinkage and cytoplasmic membrane blebbing, are hallmark features of apoptosis. Previous studies have similarly reported that SeNPs can induce apoptosis in cancer cell lines through reactive oxygen species (ROS) formation, which results in mitochondrial dysfunction and caspase activation19.This suggests that SeNPs may exert their anticancer effects by disrupting the cellular redox balance and activating intrinsic apoptotic signaling20. In addition to their pro-apoptotic effects, SeNPs demonstrated significant anti-inflammatory properties in our study, as evidenced by the egg albumin assay. Chronic inflammation is a well-known contributor to oral squamous cell carcinoma (OSCC) progression, and the observed anti-inflammatory activity of SeNPs may therefore support their anticancer potential. Similar findings have been reported in earlier studies, where biogenic SeNPs decreased the synthesis of pro-inflammatory cytokines such as TNF-α and IL-6, thereby mitigating inflammation-driven tumorigenesis.While the egg albumin assay provides a general indication of anti-inflammatory potential, it is not specific to molecular markers involved in HOSCC. Nevertheless, previous studies indicate that SeNPs can modulate key pro-inflammatory mediators, including TNF-α, IL-6, and IL-1β, in cancer cells, suggesting that the observed effects in KB-1 cells may reflect similar anti-inflammatory pathways21.
The green synthesis of SeNPs using Mangifera indica and Beta vulgaris extracts offers additional advantages over conventional chemical methods. Plant-derived phytochemicals act as reducing and capping agents, improving nanoparticle stability, size uniformity, and biological compatibility. Studies have shown that such eco-friendly synthesis routes not only ensure biocompatibility but also enhance the therapeutic potential of SeNPs compared to chemically synthesized counterparts.22,23 The selective cytotoxicity of SeNPs towards KB-1 cells, coupled with their ability to induce apoptosis and reduce inflammation, underscores their potential as a therapeutic agent for OSCC.24 Traditional chemotherapeutic agents often lack selectivity and are associated with adverse side effects. In contrast, SeNPs offer a targeted approach with reduced toxicity, as they cause cancer cells to undergo apoptosis preferentially while leaving healthy cells unaffected. This selectivity is crucial for improving patient outcomes and minimizing treatment-related complications.25 It should be noted that the current study did not include a group treated with Mangifera indica or beta vulgaris extracts alone. while the primary aim was to evaluate the anticancer and anti-inflammatory effects of SeNPs,future studies should examine plant extract-only groups to determine whether the observed effects are exclusively due to SeNPs or are partially contributed by the bioactive compounds in the extract. Treatment with SeNPs resulted in a significant increase in intracellular ROS levels, leading to oxidative stress and mitochondrial dysfunction. This is consistent with findings from other studies where SeNPs induced ROS-mediated cytotoxicity. For instance, a study on PC-3 prostate cancer cells reported that biogenic SeNPs induced ROS overproduction, leading to necroptosis through tumor necrosis factor (TNF) activation.26 Similarly, in MCF-7 breast cancer cells, selenium nanoparticles synthesized using green methods induced ROS generation, resulting in mitochondrial dysfunction and apoptosis. These parallels suggest that ROS generation is a critical mediator of SeNP-induced cytotoxicity across various cancer cell types.27 The observed apoptosis in KB-1 cells was accompanied by the activation of caspase-3 and caspase-9, indicating the involvement of the intrinsic apoptotic pathway, While previous studies have reported that SeNPs can induce apoptosis through ROS generation and activation of caspase-3 and caspase-9, our current results primarily demonstrate morphological changes consistent with apoptosis. Further studies are required to confirm the involvement of ROS-mediated pathways and caspase activation in KB-1 cells.28 For example, selenium nanoparticles functionalized with galangin induced HepG2 cell death by ROS-mediated caspase-3 activation and modification of AKT and p38 signaling pathways. Additionally, targeted delivery of paclitaxel using functionalized SeNPs in MCF-7 cells resulted in enhanced apoptosis through ROS generation and subsequent caspase-3 activation.29 These findings reinforce the role of caspase-mediated pathways in SeNP-induced apoptosis. While limited studies have specifically investigated the effects of SeNPs on OSCC, our findings are in line with general observations of SeNP-induced apoptosis in various cancer cell lines. The mechanisms involving ROS generation and caspase activation appear to be conserved across different cancer types.30 Further research focusing on OSCC is necessary to elucidate the specific molecular interactions and potential therapeutic applications of SeNPs in oral cancer treatment. While our findings are promising, further research is necessary to fully elucidate the molecular mechanisms underlying SeNP-induced apoptosis in KB-1 cells. Investigating the involvement of specific signaling pathways, such as the mitochondrial (intrinsic) and death receptor (extrinsic) pathways, would provide a deeper understanding of SeNPs’ mode of action. Additionally, In vivo research is necessary to evaluate the safety profile and therapeutic efficacy of bioinspired SeNPs in animal models of OSCC. Evaluating parameters such as biodistribution, pharmacokinetics, and potential off-target effects will be critical steps towards clinical translation . Our research adds to the increasing amount of data demonstrating bioinspired SeNPs’ anticancer potential. SeNPs offer a possible path for the creation of innovative therapeutics since they induce apoptosis and have anti-inflammatory qualities strategies against OSCC. The green synthesis approach further enhances their appeal by offering a sustainable and biocompatible alternative to conventional therapies. Future studies should focus on detailed mechanistic investigations and in vivo validations to pave the way for clinical applications of SeNPs in oral cancer treatment31.
Conclusion
This research illustrates the robust anticancer and anti-inflammatory attributes of bioinspired selenium nanoparticles (SeNPs) produced utilising extracts from Mangifera indica and Beta vulgaris. The biologically synthesised SeNPs demonstrated considerable cytotoxicity against human oral squamous cell carcinoma (HOSCC) KB-1 cell lines, predominantly via the induction of apoptosis. Morphological alterations, such as cellular shrinkage and cytoplasmic membrane blebbing, were seen in SeNP-treated cells, substantiating their capacity to induce programmed cell death. These findings are consistent with previous studies suggesting SeNPs can induce apoptosis in cancer cells. Moreover, SeNPs exhibited potent anti-inflammatory activities, potentially advantageous in alleviating the chronic inflammation linked to OSCC progression. This study utilised a green synthesis technique that offered a cost-effective and environmentally sustainable way for the creation of SeNPs, while simultaneously improving their biocompatibility through the integration of plant-derived bioactive chemicals. In contrast to traditional chemotherapy, SeNPs provide a more targeted and less toxic option, positioning them as a possible choice for oral cancer treatment. Further studies are needed to clarify the molecular mechanisms underlying SeNP-induced apoptosis and inflammation modulation. Moreover, in vivo investigations are essential for evaluating the pharmacokinetics, biodistribution, and long-term safety of SeNPs. Subsequent investigations should concentrate on refining nanoparticle compositions to improve selectivity and therapeutic effectiveness. In conclusion, our results underscore the promise of bioinspired SeNPs as an innovative treatment agent for OSCC. Overall, these findings highlight the potential of bioinspired SeNPs as a therapeutic option for OSCC, combining pro-apoptotic and anti-inflammatory effects
Acknowledgement
The authors would like to thank Saveetha college of pharmacy for conducting the research.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability
This statement does not apply to this article
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Authors’ Contribution
- Karthik Kadhiri Karunakar: Planning and conceptualization of the research , outlined study objectives, and coordinated the overall project direction.
- Binoy Varghese Cheriyan: Supervised the study hypothesis and helped design the experimental approach.
- Rajesh shanumugam, Akshaya Murugathirumal, Abinaya Senthilkumar: Nanoparticle Synthesis, Characterization , Methodology.
- Ragavendran Anandakumar, Gananish Murugan: Cell line and Biological Testing and visualization.
- Krithikeshvaran Rajasekaran ,Sowmyalakshmi Venkataraman: Data Analysis and Validation Phase.
- Nandhini Jayaprakash, Lokesh Ravikumar, Maha Lakshmi: Project administration, Resources, Language polishing and Editing.
Reference
- Adeola HA, Papagerakis S, Papagerakis P. Systems biology approaches and precision oral health: A circadian clock perspective. Front Physiol. 2019;10(MAR). doi:10.3389/fphys.2019.00399.
CrossRef - Karthik KK, Cheriyan BV, Rajeshkumar S, Gopalakrishnan M. A review on selenium nanoparticles and their biomedical applications. Biomedical Technology. 2024;6. doi:10.1016/j.bmt.2023.12.001
CrossRef - Gharbavi M, Mousavi M, Pour-Karim M, Tavakolizadeh M, Sharafi A. Biogenic and facile synthesis of selenium nanoparticles using Vaccinium arctostaphylos L. fruit extract and anticancer activity against in vitro model of breast cancer. Cell Biol Int. 2022;46(10). doi:10.1002/cbin.11852
CrossRef - Witmer RK, Harris R, Niemiera AX. Frequent Irrigation Increases Growth of Pot-in-Pot Sugar and Red Maple. HortScience. 2019;33(3):474. doi:10.21273/hortsci.33.3.474
CrossRef - Hussain A, Lakhan MN, Hanan A, et al. Recent progress on green synthesis of selenium nanoparticles – a review. Materials Today Sustainability. 2023;23. doi:10.1016/j.mtsust.2023.100420
CrossRef - Perumal S, Gopal Samy MV, Subramanian D. Selenium nanoparticle synthesis from endangered medicinal herb (Enicostema axillare). Bioprocess Biosyst Eng. 2021;44(9):1853-1863. doi:10.1007/s00449-021-02565-z
CrossRef - Ramachandran T, Manoharan D, Natesan S, et al. Synthesis and Structural Characterization of Selenium Nanoparticles–Bacillus sp. MKUST-01 Exopolysaccharide (SeNPs–EPS) Conjugate for Biomedical Applications. Biomedicines. 2023;11(9). doi:10.3390/biomedicines11092520
CrossRef - Chung S, Zhou R, Webster TJ. Green synthesized BSA-coated selenium nanoparticles inhibit bacterial growth while promoting mammalian cell growth. Int J Nanomedicine. 2020;15:115-124. doi:10.2147/IJN.S193886
CrossRef - HDT M. In vitro Anti-Inflammatory Egg Albumin Denaturation Assay: An Enhanced Approach. Journal of Natural & Ayurvedic Medicine. 2023;7(3):1-6. doi:10.23880/jonam-16000411
CrossRef - Varadarajan PS, Roy A, Jagadeswaran D. Anticancer Property of Digera muricata Leaf Extract: An In Vitro Study. Cureus. Published online 2023. doi:10.7759/cureus.49276
CrossRef - Kumar P, Nagarajan A, Uchil PD. Analysis of cell viability by the MTT assay. Cold Spring Harb Protoc. 2018;2018(6):469-471. doi:10.1101/pdb.prot095505
CrossRef - Ghasemi M, Turnbull T, Sebastian S, Kempson I. The mtt assay: Utility, limitations, pitfalls, and interpretation in bulk and single-cell analysis. Int J Mol Sci. 2021;22(23). doi:10.3390/ijms222312827
CrossRef - Liu K, Liu P cheng, Liu R, Wu X. Dual AO/EB staining to detect apoptosis in osteosarcoma cells compared with flow cytometry. Med Sci Monit Basic Res. 2015;21:15-20. doi:10.12659/MSMBR.893327
CrossRef - Kulshrestha A, Katara GK, Ibrahim SA, et al. In vivo anti-V-ATPase antibody treatment delays ovarian tumor growth by increasing antitumor immune responses. Mol Oncol. 2020;14(10):2436-2454. doi:10.1002/1878-0261.12782
CrossRef - Jha N, Esakkiraj P, Annamalai A, Lakra AK, Naik S, Arul V. Synthesis, optimization, and physicochemical characterization of selenium nanoparticles from polysaccharide of mangrove Rhizophora mucronata with potential bioactivities. Journal of Trace Elements and Minerals. 2022;2:100019. doi:10.1016/j.jtemin.2022.100019
CrossRef - Abdelhamid AE, Ahmed EH, Awad HM, Ayoub MMH. Synthesis and cytotoxic activities of selenium nanoparticles incorporated nano-chitosan. Polymer Bulletin. 2024;81(2). doi:10.1007/s00289-023-04768-8
CrossRef - Kamnev AA, Dyatlova YA, Kenzhegulov OA, Vladimirova AA, Mamchenkova P V., Tugarova A V. Fourier transform infrared (FTIR) spectroscopic analyses of microbiological samples and biogenic selenium nanoparticles of microbial origin: Sample preparation effects. Molecules. 2021;26(4). doi:10.3390/molecules26041146
CrossRef - Tugarova A V., Mamchenkova P V., Dyatlova YA, Kamnev AA. FTIR and Raman spectroscopic studies of selenium nanoparticles synthesised by the bacterium Azospirillum thiophilum. Spectrochim Acta A Mol Biomol Spectrosc. 2018;192. doi:10.1016/j.saa.2017.11.050
CrossRef - Zhuang Y, Li L, Feng L, et al. Mitochondrion-targeted selenium nanoparticles enhance reactive oxygen species-mediated cell death. Nanoscale. 2020;12(3). doi:10.1039/c9nr09039h
CrossRef - Saraste A, Pulkki K. Morphologic and biochemical hallmarks of apoptosis. Cardiovasc Res. 2000;45(3):528-537. doi:10.1016/S0008-6363(99)00384-3
CrossRef - HDT M. In vitro Anti-Inflammatory Egg Albumin Denaturation Assay: An Enhanced Approach. Journal of Natural & Ayurvedic Medicine. 2023;7(3). doi:10.23880/jonam-16000411
CrossRef - Zambonino MC, Quizhpe EM, Jaramillo FE, et al. Green synthesis of selenium and tellurium nanoparticles: Current trends, biological properties and biomedical applications. Int J Mol Sci. 2021;22(3). doi:10.3390/ijms22030989
CrossRef - Vijayaram S, Razafindralambo H, Sun YZ, et al. Applications of Green Synthesized Metal Nanoparticles — a Review. Biol Trace Elem Res. 2024;202(1). doi:10.1007/s12011-023-03645-9
CrossRef - Huang Y, He L, Liu W, et al. Selective cellular uptake and induction of apoptosis of cancer-targeted selenium nanoparticles. Biomaterials. 2013;34(29). doi:10.1016/j.biomaterials.2013.04.067
CrossRef - Li H, Zhang Y, Xu M, Yang D. Current trends of targeted therapy for oral squamous cell carcinoma. J Cancer Res Clin Oncol. 2022;148(9). doi:10.1007/s00432-022-04028-8
CrossRef - Sonkusre P, Cameotra SS. Biogenic selenium nanoparticles induce ROS-mediated necroptosis in PC-3 cancer cells through TNF activation. J Nanobiotechnology. 2017;15(1). doi:10.1186/s12951-017-0276-3
CrossRef - Al-Otaibi AM, Al-Gebaly AS, Almeer R, Albasher G, Al-Qahtani WS, Abdel Moneim AE. Potential of green-synthesized selenium nanoparticles using apigenin in human breast cancer MCF-7 cells. Environmental Science and Pollution Research. 2022;29(31). doi:10.1007/s11356-022-19166-2
CrossRef - Ranjitha VR, Rai VR. Selenium nanostructure: Progress towards green synthesis and functionalization for biomedicine. J Pharm Investig. 2021;51(2). doi:10.1007/s40005-020-00510-y
CrossRef - Li Y, Guo M, Lin Z, et al. Multifunctional selenium nanoparticles with Galangin-induced HepG2 cell apoptosis through p38 and AKT signalling pathway. R Soc Open Sci. 2018;5(11). doi:10.1098/rsos.180509
CrossRef - Varlamova EG, Goltyaev M V., Mal’tseva VN, et al. Mechanisms of the cytotoxic effect of selenium nanoparticles in different human cancer cell lines. Int J Mol Sci. 2021;22(15). doi:10.3390/ijms22157798
CrossRef - Behera A, Dharmalingam Jothinathan MK, Saravanan S, Tamil Selvan S, Rajan Renuka R, Srinivasan GP. Green Synthesis of Selenium Nanoparticles From Clove and Their Toxicity Effect and Anti-angiogenic, Antibacterial and Antioxidant Potential. Cureus. Published online 2024. doi:10.7759/cureus.55605
CrossRef







