Manuscript accepted on :03-12-2025
Published online on: 22-12-2025
Plagiarism Check: Yes
Reviewed by: Dr. Subhasis Chakraborty
Second Review by: Dr. Hina Imran
Final Approval by: Dr. Patorn Piromchai
Abduvali Abdubokiev1* , Shunkor Khushmatov1
, Shunkor Khushmatov1 , Esohon Komilov1, Sirojiddin Ikramov1 and Sherzod Zhurakulov1
, Esohon Komilov1, Sirojiddin Ikramov1 and Sherzod Zhurakulov1
1Department of Physiology and Anatomy, Namangan State University, Namangan, Uzbekistan.
2Department for the Development of Scientific and Innovative Activities, Ministry of Higher Education, Science and Innovation of the Republic of Uzbekistan, Tashkent, Uzbekistan.
3Department of Molecular Biophysics, Institute of Biophysics and Biochemistry under the National University of Uzbekistan, Tashkent, Uzbekistan.
4Department of Medicine, University of Business and Science, Namangan, Uzbekistan.
5Department of Alkaloid Chemistry, Institute of the Chemistry of Plant Substances named after Academician S.Yu. Yunusov, Academy of Sciences of the Republic of Uzbekistan, Tashkent, Uzbekistan.
Corresponding Author’s E-mail: abduvaliabduboqiyev38@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3332
Abstract
Carbon tetrachloride (CCl4)-induced hepatotoxicity is characterized by profound oxidative stress, disruption of hepatocyte membrane integrity, and sharp elevations in alanine aminotransferase (ALT) and aspartate aminotransferase (AST). In this study, the hepatoprotective potential of isoquinoline alkaloids (F-5, F-24) and their semi-synthetic derivatives (KV-6, KV-8) was evaluated using a CCl4-induced toxic hepatitis model in mice. CCl4 intoxication increased serum ALT and AST levels by up to 300% compared to the control, confirming severe hepatocellular injury. Administration of the tested compounds (25-100 mg/kg) produced a concentration-dependent reduction in ALT and AST activities. Among the alkaloids, F-5 (100 mg/kg) decreased ALT to 8.19±0.14 microkat/l in males and 7.14±0.25 microkat/l in females, while F-24 reduced these values to 9.43±0.18 and 8.45±0.26 μkat/l, respectively. The derivatives KV-6 and KV-8 demonstrated stronger hepatoprotective effects, with KV-6 (100 mg/kg) showing the greatest efficacy – reducing ALT to 5.10±0.17 and 4.04±0.23 microkat/l, and AST to 7.56±0.35 and 8.42±0.29 microkat/l in male and female mice, respectively. Calculation of the hepatoprotective action coefficient confirmed the effectiveness of the compounds in the order: F-24<F-5<KV-8<KV-6, with KV-6 exhibiting the highest protective activity (64-82% restoration relative to CCl4 pathology). These findings indicate that isoquinoline alkaloids and their derivatives mitigate CCl4-induced hepatocyte damage by normalizing aminotransferase activity, suggesting their promise as hepatoprotective agents. The results align with existing evidence on the protective effects of polyphenolic compounds in oxidative-stress–mediated liver injury.
Keywords
ALT; AST; CCl4-induced hepatotoxity; Hepatoprotective effect; Isoquinoline alkaloids
Download this article as:| Copy the following to cite this article: Abdubokiev A, Khushmatov S, Komilov E, Ikramov S, Zhurakulov S. Analysis of Hepatoprotective Activity of Isoquinoline Alkaloids (F-5, F-24), and their Derivatives (KV-6, KV-8) in CCl₄-Induced Hepatotoxity in Mice. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Abdubokiev A, Khushmatov S, Komilov E, Ikramov S, Zhurakulov S. Analysis of Hepatoprotective Activity of Isoquinoline Alkaloids (F-5, F-24), and their Derivatives (KV-6, KV-8) in CCl₄-Induced Hepatotoxity in Mice. Biomed Pharmacol J 2025;18(4). Available from: https://bit.ly/4qqujQ1 |
Introduction
According to World Health Organization statistics, approximately 3 million people will die from liver diseases by 2024, and this rapidly increasing rate represents a serious medical, socioeconomic, and global challenge. Therefore, developing new approaches to treating these disorders and creating effective next-generation pharmaceuticals is of critical importance.1-7 Numerous studies have described in detail the origins of liver diseases, their pathogenesis, and the mechanisms of action of hepatoprotective agents. In particular, it has been noted that the therapeutic effects of broad-spectrum hepatoprotectors (including polyphenolic compounds and flavonoids) involve stimulation of the antioxidant system in hepatocytes, antiradical activity (suppression of ROS formation), and optimization of enzyme function, among other mechanisms.8-10
Isoquinoline alkaloids and their derivatives represent one promising group of agents in this field. The biological activity of isoquinoline alkaloids – known for their wide range of pharmacological effects (anticancer, antibacterial, antiviral, antineurodegenerative, analgesic, antidepressant, cardioprotective, etc.) – has been extensively studied.11-14 It has been demonstrated that isoquinoline alkaloids optimize lipid metabolism in hepatocytes by modulating the AMPK/SIRT1/PPAR-alfa signaling cascade, enhance the activity of antioxidant enzymes, and exert antioxidant and anti-apoptotic effects through modulation of the MAPK/NF-kappaB and ERK/JNK pathways.15-18 It is also noteworthy that many hepatoprotective medications contain components with antioxidant, anti-inflammatory, detoxifying, and choleretic properties. A significant proportion of these components are polyphenolic compounds (such as flavonoids) that inhibit lipid peroxidation (LPO) and increase the stability of lipid biomembranes. In particular, the hepatoprotective activity of quercetin, closely related to its chemical structure, has been widely documented.19-22
The aim of this study was to compare the hepatoprotective activity of several isoquinoline alkaloids (F-5, F-24) and their derivatives (KV-6, KV-8) by evaluating their effects on plasma transaminase enzymes – alanine aminotransferase (ALT) and aspartate aminotransferase (AST) – in a mouse model of CCl4-induced toxic hepatitis.
Materials and Methods
The experiments were conducted in 2023-2024 in the Metabolomics laboratory of the Institute of Biophysics and Biochemistry of the National University of Uzbekistan named after Mirzo Ulugbek.
Experimental Animals
The objects of the study were white mice (male/female, m=20.5-26.2 g), which were fed standard food (water) under standard vivarium conditions (room temperature +20±5°C, relative air humidity 75±10%, light regime 12:12 hours).23,24
Ethical Approval
When working with experimental animals in scientific research, the requirements of the rules developed by the International Council for International Organizations of Medical Sciences (1985), the European Convention for the Protection of Vertebrate Animals used for Experimental and other Scientific Purposes (Strasbourg, 1986), the Declaration developed by the European Union (86/609/EEC), and the Bioethical Statement of the Institute of Biophysics and Biochemistry of the National University of Uzbekistan (No. BRC/IBB; N44/2024/75-1) were observed.25-28
Chemicals and Drugs
Isoquinoline alkaloids (F-5, F-24) and their derivatives with the flavonoid quercetin (KV-6, KV-8), provided by the staff of the S.Yu.Yunusov Institute of Plant Chemistry of the Academy of Sciences of the Republic of Uzbekistan (Table. 1) were used as objects of research.29-33
 |
Table 1: Chemical structure of isoquinoline alkaloids F-5, F-24, and their derivatives Click here to view Table |
Experimental Design
The hepatoprotective activity of isoquinoline alkaloids and their derivatives was assessed using a standard method on a model of experimental toxic hepatitis in mice caused by CCl4.34,35,36,37,38 To create an experimental model of toxic hepatitis, we used white outbred mice of both sexes (male/female, m=20.5-26.2 g) aged 2-3 months, kept in vivarium conditions on standard food and water. During the experiments, animals were administered intraperitoneal CCl4 solution (diluted in olive oil) at a dose of 1 ml/kg for 14 days (on the 1st, 5th and 11th days).39-56
Biochemical Analysis of Enzymes
In biological research, spectrophotometric analysis of ALT, AST and other substances allows for the acquisition of highly accurate data.57,58
The activity of ALT and AST in blood serum was analyzed using a standard method.59-65
Aminotransferases or transaminases that use phosphopyridoxal and phosphopyridoxamine as coenzymes catalyze the reverse transfer of the amino group from an amino acid to an α-keto acid. Formed in the transamination reaction of an amino acid determination of the concentration of α-ketoacid forms the basis of the assessment of the activity of transaminases. Aspartic acid is converted to oxaloacetate by AST, and alanine is converted to pyruvic acid by ALT. During the enzymatic reaction, oxaloacetate is converted to pyruvate acid. Adding 2,4-dinitrophenylhydrazine to the medium halts the process, forming a brown pyruvate dinitrophenylhydrazone. The color intensity was used to estimate the concentration, which in turn, indicates the aminotransferase activity (expressed in micromol/h×mL), formed during 1 hour of incubation in 1 mL of the test serum at 37°C). The blood sample was centrifuged at 3000 rpm for 10 minutes, the serum was separated with a pipette into a test tube, dinitrophenylhydrazine (0.25 ml) was added to the test sample (0.05 ml), incubated for 60 minutes at t=+37±0.5°C, 0.4 M NaOH (2.5 ml) was added, after 10 minutes of incubation, the color intensity was assessed by optical density in the wavelength range 490-520 nm and the activity of ALT and AST was determined using a calibration graph. Based on the results of spectrophotometric analysis, a curve of the dependence of optical density and enzyme activity was constructed, where the optical density is plotted along the ordinate axis and the enzyme activity is plotted along the abscissa axis. The activity of ALT and AST (microkat/l) in the blood serum of mice was analyzed using a biochemical analysis reagent kit from Cupress Diagnostica (Belgium) and an Agilent Cary 60 Uv-Vis spectrophotometer (Agilent Technologies, USA).66-80 A blood sample (1 ml) taken from a rat vein was centrifuged at 12,600 rpm for 10 minutes (t=+4±0.5°C), after which ALT and AST were analyzed spectrophotometrically.81,82,83 In the spectrophotometric analysis of ALT activity, a method developed by Reitman S., Frenkel S. is used, based on the transfer of an amino group from the amino acid alanine to 2-oxoglutarate in the presence of ALT, the formation of pyruvate and glutamate, and in the next stage – the formation of lactate and NAD+ in the reaction of pyruvate (pyruvic acid) and NADH+ (H+) in the presence of lactate dehydrogenase and the assessment of the amount of ALT by the level of absorption of NADH+ (H+) in the range of 340 nm (Alanine + 2-oxoglutarate Pyruvate + Glutamate Pyruvate + NADH+ (H+) Lactate + NAD+).AST activity is analyzed by assessing the amount of oxaloacetate hydrazone formed from 2,4-dinitrophenylhydrazine under the action of oxaloacetate in the reaction of L-aspartate and 2-hydroxyglutarate with the participation of AST.84-87
Statistical Analysis
Mathematical and statistical processing of the obtained experimental results was carried out by standard methods using special software packages “Microsoft Excel 2007” (Microsoft, USA), OriginPro v. 8.5 SR1 (EULA, USA). The results are presented as M±m, where M is the arithmetic mean and m is the standard error of the mean, calculated based on the results of experiments conducted with n=3-4 repetitions. Also, the level of statistical significance of the values between the experimental results and the control group was calculated based on Student’s t-test and was considered statistically significant at values of p<0.05 and p<0.01. The level of reliability of the difference between the values of the two experimental groups was calculated using the Student’s t-test.88,89,90,91,92 In the studies, the value of the hepatoprotective coefficient (HC) of isoquinoline alkaloids (F-5, F-24) and their derivatives (KV-6, KV-8) was calculated using the following formula (1):
HC=[1-(E-C)/HC-C)]×100% (1)
HC – is the hepatoprotective activity coefficient (%), E – is the experimental value (CCl4+substance), C – is the control (healthy animal), and PC – is the pathological control value (CCl4). The closer the HC – value is to 100%, the higher the level of hepatoprotective activity of the substance.
Results
In the pathogenesis of liver diseases under the influence of oxidative stress, a sharp change in enzyme activity is observed.88,89
Aminotransferases (AST/ALT) – catalyze the reaction of amination of the aminogroup of an aminoacid to alfa-ketoglutaric acid (in the human body under physiological conditions, ALT is 0.68-1.08 microkat/l, AST – 0.67-0.83 microkat/l), and in experimental animals it has been confirmed that under conditions of hepatotoxic exposure to CCl4 their level increases up to 300% compared to the control.90-94 Under the influence of CCl4, the permeability of biological membranes in hepatocytes is disrupted, the activity of transaminases in the cytoplasm increases, the extracellular matrix is destroyed, and the amount of gamma-globulins decreases.95 In turn, the next series of studies analyzed the effect of isoquinoline alkaloids and some of their derivatives on the ALT and AST enzymes in blood plasma in experimental toxic hepatitis caused by CCl4. In the control group, ALT activity averaged 1.05±0.02 and 0.94±0.01 microkat/l in males and females, respectively, and with CCl4 intoxication, these indicators increased to 12.23±0.19 and 10.53±0.16 microkat/l, respectively. Isoquinoline alkaloids (F-5, F-24) and their derivatives (KV-6, KV-8) were found to cause concentration-dependent (25-100 mg/kg) inhibition of ALT activity under CCl4 intoxication conditions. It was established that the isoquinoline alkaloid F-5 at a maximum concentration of 100 mg/kg reduced ALT activity in male and female mice during CCl4 intoxication to 8.19±0.14 and 7.14±0.25 microkat/l, respectively (Fig. 1A). It was also noted that the isoquinoline alkaloid F-24 at a maximum concentration of 100 mg/kg reduced ALT activity in experimental toxic hepatitis caused by CCl4 to 9.43±0.18 and 8.45±0.26 microkat/l in male and female mice, respectively (Fig. 1B).
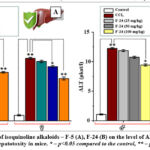 |
Figure 1: The effect of isoquinoline alkaloids – F-5 (A), F-24 (B) on the level of ALT in the blood serum in CCl4-induced hepatotoxity in mice. |
It was found that the KV-6 at a maximum concentration of 100 mg/kg reduced the activity of ALT in experimental toxic hepatitis CCl4 in male and female mice to 5.10±0.17 and 4.04±0.23 microkat/l, respectively, while the KV-8 (100 mg/kg) had this indicator of 7.13±0.28 and 6.05±0.15 microkat/l, respectively (Fig. 2A, B).
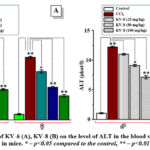 |
Figure 2: The effect of KV-6 (A), KV-8 (B) on the level of ALT in the blood serum in CCl4-induced hepatotoxity in mice. |
It was also established that isoquinoline alkaloids (F-5, F-24), and their derivatives (KV-6, KV-8) inhibit AST activity depending on the concentration (25-100 mg/kg) under conditions of CCl4-intoxication. It was noted that the isoquinoline alkaloid F-5 at a maximum concentration of 100 mg/kg reduced AST activity in male and female mice under CCl4-intoxication to 10.35±0.27 and 12.64±0.32 microkat/l, respectively. For the isoquinoline alkaloid F-24 (100 mg/kg), a decrease in the value of this indicator was noted to 13.43±0.16 and 15.82±0.29 microkat/l, respectively (Fig. 3A, B).
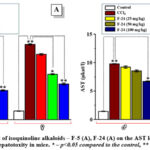 |
Figure 3: The effect of isoquinoline alkaloids – F-5 (A), F-24 (A) on the AST level in the blood serum in CCl4-induced hepatotoxity in mice. |
It was found that the KV-6 at a maximum concentration of 100 mg/kg reduced ALT activity in male and female mice poisoned with CCl4 to 7.56±0.35 and 8.42±0.29 microkat/l, respectively, while the KV-8 (100 mg/kg) had this indicator down to 8.37±0.21 and 9.68±0.16 microkat/l, respectively (Fig. 4A, B).
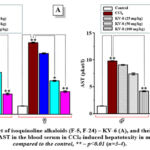 |
Figure 4: The effect of isoquinoline alkaloids (F-5, F-24) – KV-6 (A), and their derivatives KV-8 (B) on the level of AST in the blood serum in CCl4-induced hepatotoxity in mice.
|
The increase in ALT and AST levels during CCl4-intoxication is explained by impaired liver function.96-102
Under the influence of CCl4, the generation of free radicals in hepatocytes, structural and functional disturbances of the biological membrane of the LPO process, and changes in the activity of enzyme complexes such as alkaline phosphatase, lactate dehydrogenase, ALT and AST are observed.103 Also, the increase in ALT and AST during CCl4 intoxication is explained by the release of these enzymes into the blood as a result of damage to mitochondrial membranes during hepatopathogenesis.104-107 In this case, changes in serum activity of ALT, AST, total protein and other indicators against the background of CCl4-toxic hepatitis are used as objective indicators.108-111 The hepatoprotective activity of alkaloids and flavonoids, including a decrease in the level of ALT and AST in the blood serum, has been confirmed in studies. It has been established that flavonoids and other polyphenols optimize the levels of ALT, AST, and bilirubin in experimental animals under conditions of toxic hepatitis.112 Under the influence of various stress factors, pathological changes occur in the liver of rats, in which the amount of ALT, AST and bilirubin in the blood serum increases, and quercetin optimizes these indicators.113 Studies have confirmed that quercetin (100-500 μg/kg) reduces the activity of ALT and AST in blood serum, decreases the amount of malondialdehyde in liver homogenate, optimizes the activity of glutathione peroxidase and superoxide dismutase, and has a therapeutic effect on the morphoanatomical and histopathological changes in hepatocytes under conditions of CCl4-induced hepatotoxity in mice.114 Polyphenols have been shown to optimize the activity of ALT, AST, malondialdehyde, catalase and glutathione peroxidase, and also improve the cytological structure of hepatocytes in experimental animals with CCl4-induced hepatotoxity.115 Flavonoids have been shown to exert hepatoprotective effects on hepatocytes by activating the PI3K/Akt cascade, inhibiting ROS formation, activating the antioxidant cascade (activation of antioxidant enzymes) through modulation of MAPK/Nrf2 signaling, reducing serum ALT and AST levels, regulating metabolism, inhibiting apoptosis through modulation of the MicroRNA-205/NF-κB cascade, optimizing lipogenesis, and improving mitochondrial function. Thus, under conditions of CCl4-induced hepatotoxity, an increase in the formation of LPO and ROS in the liver of mice, a significant decrease in the activity of the antioxidant enzyme complex – superoxide dismutase, glutathione peroxidase, glutathione reductase, as well as an increase in the activity of iNOS, ALT and AST are observed. In liver diseases, structural and functional changes in hepatocytes, disturbances in carbohydrate, protein and lipid metabolism, and an increase in the number of enzymes (including aminotransferases) in the blood serum have been confirmed. At the next stage of the experiments, based on the data on the activity of ALT and AST, the values of the coefficient of hepatoprotective action of isoquinoline alkaloids (F-5, F-24) and their derivatives (KV-6, KV-8) were calculated (Table 2).
Table 2: The effect of isoquinoline alkaloids (F-5, F-24) and their derivatives (KV-6, KV-8) on the activity of ALT and AST in blood serum in in CCl4-induced hepatotoxity in mice (M±m)
| Index | Types of gender | Control | CCl4 | Concentration (100 mg/kg) | |||
| F-5 | F-24 | KV-6 | KV-8 | ||||
| In blood serum | |||||||
| ALT (μkat/L) | Male | 1,05±0,02 | 12,23±0,19 | 8,19±0,14** | 9,43±0,18* | 5,10±0,17** | 7,13±0,28** |
| Female | 0,94±0,01 | 10,53±0,16 | 7,14±0,25** | 8,45±0,26* | 4,04±0,23** | 6,05±0,15** | |
| AST (μkat/L) | Male | 2,87±0,08 | 19,60±0,10 | 10,35±0,27** | 13,43±0,16* | 7,56±0,35** | 8,37±0,21** |
| Female | 3,06±0,09 | 26,64±0,22 | 12,64±0,32** | 15,82±0,29* | 8,42±0,29** | 9,68±0,16** | |
Note: “kat” (katal) is the enzyme activity that converts 1 mol of substrate into product (P) in 1 second, and 1 microkat/L = [(10–6×(mol/sec)]/L (represents the activity that converts 10–6 mol of substrate into product (P) in 1 second in 1 L of medium [Lothar, 1999; pp.771; Baltierra-Trejo et al., 2015; pp.126-131]. * – p<0.05 compared to control, ** – p<0.01 (n=3-4).
Based on the values of the analyzed parameters (ALT and AST activity in blood serum), the values of the coefficient of hepatoprotective action of isoquinoline alkaloids (F-5, F-24) and their derivatives (KV-6, KV-8) were calculated (Table 3).
Table 3: alues of the hepatoprotective coefficient of isoquinoline alkaloids (F-5, F-24) and their derivatives (KV-6, KV-8) for ALT and AST activity
| Index | Types of gender | Control | CCl4 | Hepatoprotective coefficient (%) | |||
| F-5 | F-24 | KV-6 | KV-8 | ||||
| In blood serum | |||||||
| ALT (μkat/L) | Male | 1,05±0,02 | 12,23±0,19 | 36 | 25 | 64 | 46 |
| Female | 0,94±0,01 | 10,53±0,16 | 35 | 22 | 68 | 47 | |
| AST (μkat/L) | Male | 2,87±0,08 | 19,60±0,10 | 55 | 37 | 72 | 67 |
| Female | 3,06±0,09 | 26,64±0,22 | 59 | 47 | 77 | 72 | |
Note: Based on the change in ALT activity (microkat/l) in the blood of mice (male) under conditions of toxic hepatitis caused by CCl4, for the isoquinoline alkaloid F-5 (100 mg/kg), the coefficient of hepatoprotective action is: HCF-5=[(1-(8,19-1,05)/(12,23-1,05)]×100=36%.
Based on the analysis of the values of the analyzed parameters under control conditions, CCl4-induced hepatotoxity and the action of isoquinoline alkaloids (F-5, F-24) and their derivatives (KV-6, KV-8) at a concentration of 100 mg/kg, it was established that the coefficient of the hepatoprotective action of these substances has a characteristic increase in the series F-24<F-5<KV-8<KV-6. The maximum coefficient of hepatoprotective action was noted in the KV-6 (100 mg/kg), while it was established that the values of ALT (microkat/l), AST (microkat/l) in the blood serum of mice amounted to 64, 72, 82, 67, 73% of the values of CCl4-induced hepatotoxity in males and females, respectively.
Mice are convenient subjects for various medical and physiological experimental studies, with the mean serum ALT level being 0.85 microkat/L and 0.82 microkat/L in male and female mice, respectively, and the mean AST level being 1.85 microkat/L and 1.95 microkat/L in male and female mice, respectively. Under conditions of CCl4-induced hepatotoxity, ALT activity in the serum of mice increased from 0.51 microkat/l to 2.80 microkat/l in the control group, and AST activity increased from 0.84 microkat/l to 2.34 microkat/l in the control group. At the same time, gender differences in the values of the analyzed indicators in the blood of experimental animals were confirmed.
In the normal physiological norm in the human body, ALT in men is on average 41 units/l, in women 31 units/l, AST – 37 and 31 units/l, respectively, and an increase in its level (hepatopathologies are characterized by an increase in ALT, AST by 10-20 times compared to the control) indicates liver pathogenesis. In the CCl4-induced hepatotoxity, changes (increases) in ALT and AST activities are used as sensitive objective indicators for assessing the functional state of the liver. CCl4 exposure has been shown to induce fibrosis in hepatocytes, including TNF-α and transforming rowth factor (TGF), suggesting that plant extracts with anti-inflammatory activity may have beneficial effects.
Using an experimental model of acute hepatitis caused by CCl4, it was confirmed that plant substances containing polyphenolic compounds and flavonoids (hepatoprotectors) significantly reduce the induction of lipid peroxidation, an increase in the concentration of toxic metabolites, necrosis and fibrosis in hepatocytes.
It is assumed that the hepatoprotective effect of Taraxacum officinale extract, consisting of polyphenolic compounds, in CCl4-induced hepatotoxity is associated with the blockade of the KEAP1/JAK2 (Kelch-like ECH-related protein 1/Janus kinase 2) signaling pathway, which is involved in the activation of Nrf2 in the oxidative stress cascade in hepatocytes.
Discussion
Carbon tetrachloride (CCl4)–induced hepatotoxicity is a well-established experimental model for evaluating liver injury and hepatoprotective agents, as it closely mimics oxidative stress–mediated liver damage observed in human hepatopathologies. In the present study, acute CCl4 intoxication resulted in a pronounced elevation of serum aminotransferase activities (ALT and AST), reflecting severe hepatocellular injury and increased membrane permeability of hepatocytes. These findings are consistent with earlier reports demonstrating that CCl4 exposure leads to excessive generation of reactive oxygen species (ROS), initiation of lipid peroxidation (LPO), and subsequent disruption of mitochondrial and cytoplasmic membranes, culminating in enzyme leakage into the bloodstream.
Under physiological conditions, ALT and AST are predominantly localized within hepatocytes and participate in amino acid metabolism via transamination reactions. Their sharp increase during CCl4 intoxication – up to 10–20-fold compared with control values – confirms extensive hepatocyte damage and impaired liver function. The present data clearly demonstrate that CCl4 intoxication caused a statistically significant rise in ALT and AST activities in both male and female mice, with a tendency toward higher AST elevation, indicating mitochondrial involvement, as AST is partially localized within mitochondria. Gender-related differences observed in enzyme activities may reflect sex-dependent variations in xenobiotic metabolism, antioxidant capacity, and hormonal regulation of hepatic enzymes.
Administration of isoquinoline alkaloids (F-5, F-24) and their derivatives (KV-6, KV-8) markedly attenuated the CCl4-induced increase in serum ALT and AST activities in a dose-dependent manner. Among the tested compounds, KV-6 demonstrated the most pronounced hepatoprotective effect, followed by KV-8, F-5, and F-24. This ranking was consistently reflected in both absolute enzyme activity values and calculated hepatoprotective coefficients, indicating a clear structure–activity relationship within the isoquinoline alkaloid series.
The observed decrease in aminotransferase activity suggests that these compounds effectively stabilize hepatocyte membranes, limit enzyme leakage, and preserve cellular integrity under oxidative stress conditions. Such effects are likely mediated by the antioxidant properties of isoquinoline alkaloids, including their ability to scavenge free radicals, inhibit lipid peroxidation, and modulate redox-sensitive signaling pathways. Previous studies have shown that alkaloids and polyphenolic compounds can restore antioxidant enzyme activity (superoxide dismutase, glutathione peroxidase, glutathione reductase), suppress inducible nitric oxide synthase (iNOS), and reduce malondialdehyde accumulation in liver tissue during toxic hepatitis.
The superior efficacy of KV-6 may be attributed to enhanced bioavailability, stronger interaction with membrane lipids, or improved modulation of intracellular signaling pathways involved in oxidative stress responses. It is noteworthy that flavonoids and structurally related polyphenols exert hepatoprotective effects through activation of the PI3K/Akt pathway, induction of the Nrf2-dependent antioxidant response, inhibition of MAPK-mediated ROS generation, and suppression of apoptosis via regulation of NF-kappaB and microRNA-dependent mechanisms. Similar molecular mechanisms may underlie the protective effects observed for the tested isoquinoline alkaloids.
Furthermore, CCl4-induced hepatotoxicity is associated not only with oxidative stress but also with inflammatory and fibrotic processes mediated by cytokines such as TNF-alfa and transforming growth factor-beta (TGF-beta). The reduction of ALT and AST activities observed in this study suggests that the tested compounds may also interfere with inflammatory signaling cascades and fibrogenic pathways, thereby limiting progression from acute hepatocellular damage to chronic fibrosis. This assumption is supported by literature data indicating that plant-derived hepatoprotectors reduce necrosis, fibrosis, and accumulation of toxic metabolites in hepatocytes.
The calculated hepatoprotective coefficients provide an integrated quantitative assessment of liver protection, allowing direct comparison between compounds. The increasing order of hepatoprotective efficacy (F-24 < F-5 < KV-8 < KV-6) highlights the importance of chemical modification of isoquinoline alkaloids for enhancing biological activity. Notably, KV-6 restored ALT and AST activities to levels approaching physiological norms, indicating substantial recovery of liver function even under severe toxic stress.
Conclusion
Thus, during the experiments it was established that the ALT activity in the control group of male and female mice was 1.05±0.02 and 0.94±0.01 microkat/l, respectively, and with CCl4-intoxication it increased to 12.23±0.19 and 10.53±0.16 microkat/l. It was found that isoquinoline alkaloids F-5 and F-24 (25-100 mg/kg) significantly reduce the activity of ALT and AST during CCl4-intoxication, with F-5 (100 mg/kg) reducing ALT activity to 8.19±0.14 microkat/l in males and 7.14±0.25 microkat/l in females, while for alkaloid F-24 these values are 9.43±0.18 and 8.45±0.26 microkat/l, respectively. It has been confirmed that derivatives of isoquinoline alkaloids F-5 and F-24 (KV-6, KV-8) have a more pronounced hepatoprotective effect than the alkaloids, with KV-6 reducing ALT activity in male mice by 5.10±0.17 and in females by 4.04±0.23 microkat/l, while for KV-8 these values are 7.13±0.28 and 6.05±0.15 microkat/l, respectively. Based on the analysis of the coefficient of hepatoprotective action, it was established that hepatoprotective activity increases in the series F-24<F-5<KV-8<KV-6. The obtained results can be used as a scientific basis for the development of hepatoprotective agents depending on the chemical structure based on isoquinoline alkaloids and their derivatives.
Acknowledgement
The author would like to thank Abdullaev Izzatullo and Gayibov Ulugbek, Ergashev Nurali for recommendations
Funding Source
This study was supported by the Project carriedout at the Institute of Biophysics and Biochemistry at the National Universityof Uzbekistan in 2021-2024 “Development of simulation models of pathological conditions and diseases of humans and animalsin vitro and in vivo” (F-OT-2021-465).
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article
Ethics statement
When working with experimental animals in scientific research, the requirements of the rules developed by the International Council for International Organizations of Medical Sciences (1985), the European Convention for the Protection of Vertebrate Animals used for Experimental and other Scientific Purposes (Strasbourg, 1986), the Declaration developed by the European Union (86/609/EEC), and the Bioethical Statement of the Institute of Biophysics and Biochemistry of the National University of Uzbekistan (No. BRC/IBB; N44/2024/75-1) were observed.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not applicable
Author Contributions
- Abduvali Abdubokiev: Investigation, Methodology, Writing-review and editing.
- Khushmatov Shunkor: Conceptualization, Formal Analysis, Funding acquisition, Methodology, Project administration, Supervision, Writing-review and editing.
- Esohon Komilov: Supervision, Writing-review and editing.
- Sirojiddin Ikromov: Data curation, Formal Analysis, Methodology, Writing-original draft.
- Sherzod Zhurakulov: Funding acquisition, Methodology, Project administration.
References
- Alhassan AJ, Sule MS, Aliyu SA, Aliyu MD. Ideal hepatotoxicity model in rats using carbon tetrachloride (CCl₄). Bayero J Pure Appl Sci. 2009;2(2):185–187.
CrossRef - Ali AA. Biochemical and histological study of rat liver injury induced by carbon tetrachloride (CCl₄). Wasit J Pure Sci. 2025;4(1):156–167.
CrossRef - Alkinani KB, Ali EMM, Al-Shaikh TM, et al. Hepatoprotective effects of (−)-epicatechin in CCl₄-induced toxicity model are mediated via modulation of oxidative stress markers in rats. Evid Based Complement Alternat Med. 2021;2021:4655150.
CrossRef - Al-Zharani M, Mubarak M, Rudayni HA, et al. Quercetin as a dietary supplementary flavonoid alleviates oxidative stress induced by lead toxicity in male Wistar rats. Nutrients. 2023;15:1888.
CrossRef - Azamatov AA, Zhurakulov SN, Vinogradova VI, et al. Evaluation of the local anesthetic activity, acute toxicity, and structure–toxicity relationship in a series of synthesized 1-aryltetrahydroisoquinoline alkaloid derivatives in vivo and in silico. Molecules. 2023;28:477.
CrossRef - Begum R, Papia SA, Begum MM, et al. Evaluation of hepatoprotective potential of polyherbal preparations in CCl₄-induced hepatotoxicity in mice. Adv Pharmacol Pharm Sci. 2022;2022:3169500.
CrossRef - Bishnolia M, Yadav P, Singh SK, et al. Methyl donor ameliorates CCl₄-induced liver fibrosis by inhibiting inflammation and fibrosis through downregulation of EGFR and DNMT-1 expression. Food Chem Toxicol. 2025;196:115230.
CrossRef - Byun JH, Kim J, Choung SY. Hepatoprotective effect of standardized Ecklonia stolonifera formulation on CCl₄-induced liver injury in Sprague–Dawley rats. Biomol Ther (Seoul). 2018;26(2):218–223.
CrossRef - Chen Y, Li R, Hu N, et al. Baihe Wuyao decoction ameliorates CCl₄-induced chronic liver injury and liver fibrosis in mice through blocking TGF-β1/Smad2/3 signaling, anti-inflammation, and anti-oxidation effects. J Ethnopharmacol. 2020;263:113227.
CrossRef - Ciumarnean L, Milaciu MV, Runcan O, et al. The effects of flavonoids in cardiovascular diseases. Molecules. 2020;25(18):4320.
CrossRef - Cui Y, Han Y, Yang X, Sun Y, Zhao Y. Protective effects of quercetin and quercetin-5′,8-disulfonate against carbon tetrachloride-caused oxidative liver injury in mice. Molecules. 2014;19:291–305.
CrossRef - Czaja MJ, Xu J, Alt E. Prevention of carbon tetrachloride-induced rat liver injury by soluble tumor necrosis factor receptor. Gastroenterology. 1995;108:1849–1854.
CrossRef - Ernst L, Zieglowski L, Schulz M, et al. Severity assessment in mice subjected to carbon tetrachloride. Sci Rep. 2020;10:15790.
CrossRef - Fang L, Wang HF, Chen YM, Bai RX, Du SY. Baicalin confers hepatoprotective effect against alcohol-associated liver disease by upregulating microRNA-205. Int Immunopharmacol. 2022;107:108553.
CrossRef - Fedekar F, Madkour D, Khalil WF, Dessouki AA. Protective effect of ethanol extract of Sargassum dentifolium in carbon tetrachloride-induced hepatitis in rats. Int J Pharm Pharm Sci. 2012;4(3):637–641.
- Fu X, Jiang B, Zheng B, et al. Heterogenic transplantation of bone marrow-derived rhesus macaque mesenchymal stem cells ameliorates liver fibrosis induced by carbon tetrachloride in mice. PeerJ. 2018;6:e4336.
CrossRef - Handoussa H, Ayoub N, Mahran L. Liver Protection by Natural Means: Hepatoprotective Agents from Medicinal Plants. Saarbrücken: LAP LAMBERT Academic Publishing; 2015:132.
- Hu Q, Zhang S, Liu J, et al. A review: hepatoprotective compounds and their mechanisms of medicine-food homology resources. Agric Prod Process Storage. 2025;1(5):1–20.
CrossRef - Huang D, Zhou S, Cai J, et al. Effect of matrine on alcoholic steatohepatitis in rats based on MAPK/NF-κB pathway. Chin Tradit Pat Med. 2023;45(11):3769–3774.
- Huang XJ, Choi YK, Im HS, Yarimaga O, Yoon E, Kim HS. Aspartate aminotransferase (AST/GOT) and alanine aminotransferase (ALT/GPT) detection techniques. Sensors. 2006;6:756–782.
CrossRef - Jain SK, Sahu RK, Soni P, Soni V. Plant-Derived Hepatoprotective Drugs. Sharjah: Bentham Science Publishers; 2023:197.
- Kainan L, Wenjun X, Jun C. Effects of silibinin capsules combined with routine therapy on serum oxidative injury and liver function of patients with alcoholic liver disease complicated with early liver fibrosis. Chin Pharm J. 2018;29(5):686–689.
- Ke BJ, Lee CL. Cordyceps cicadae NTTU 868 mycelium prevents CCl₄-induced hepatic fibrosis in BALB/c mice via inhibiting the expression of pro-inflammatory and pro-fibrotic cytokines. J Funct Foods. 2018;43:214–223.
CrossRef - Khalaf AAA, Mekawy MEM, Moawad MS, Ahmed AM. Comparative study on the protective effect of some antioxidants against CCl₄ hepatotoxicity in rats. Egypt J Nat Toxicol. 2009;6(1):59–82.
- Lai Y, Zhou C, Huang P, et al. Polydatin alleviates alcoholic liver injury in zebrafish larvae through ameliorating lipid metabolism and oxidative stress. J Pharmacol Sci. 2018;138(1):46–53.
CrossRef - Lee YS, Cho IJ, Kim JW, et al. Hepatoprotective effects of blue honeysuckle on CCl₄-induced acute liver damage in mice. Food Sci Nutr. 2018;6(3):1–17.
CrossRef - Limdi JK, Hyde GM. Evaluation of abnormal liver function tests. Postgrad Med J. 2003;79(932):307–312.
CrossRef - Liu B, Fang Y, Yi R, Zhao X. Preventive effect of blueberry extract on liver injury induced by carbon tetrachloride in mice. Foods. 2019;8(48):1–13.
CrossRef - Liu YS, Yuan MH, Zhang CY, et al. Puerariae lobatae radix flavonoids and puerarin alleviate alcoholic liver injury in zebrafish by regulating alcohol and lipid metabolism. Biomed Pharmacother. 2021;134:111121.
CrossRef - Luckey SW, Petersen DR. Activation of Kupffer cells during the course of carbon tetrachloride-induced liver injury and fibrosis in rats. Exp Mol Pathol. 2001;71:226–240.Ma Y, Bao Y, Wu L, Ke Y, Tan L, Ren H, Song J, Zhang Q, Jin Y. IL-8 exacerbates CCl₄-induced liver fibrosis in human IL-8-expressing mice via the PI3K/Akt/HIF-1α pathway. Mol Immunol. 2022;152:111–122.
CrossRef - Mak LY, Liu K, Chirapongsathorn S, et al. Liver diseases and hepatocellular carcinoma in the Asia-Pacific region: burden, trends, challenges and future directions. Nat Rev Gastroenterol Hepatol. 2024;21:834–851.
CrossRef - Masuda A, Nakamura T, Abe M, et al. Promotion of liver regeneration and anti-fibrotic effects of the TGF-β receptor kinase inhibitor galunisertib in CCl₄-treated mice. Int J Mol Med. 2020;46:427–438.
CrossRef - McClure DE. Clinical pathology and sample collection in the laboratory rodent. Vet Clin North Am Exot Anim Pract. 2017;19(2):565–590.
CrossRef - McClure DE. Clinical pathology and sample collection in the laboratory rodent. In: Commercial Rodent Diagnostic Laboratories (“Charles River Laboratories, Inc”). Vet Clin North Am Exot Anim Pract. 1999;2(3):565–590.
CrossRef - Musolino V, Cardamone A, Mare R, et al. Antioxidant and in vitro hepatoprotective activities of a polyphenol-rich fraction from the peel of Citrus lumia Risso (Rutaceae). Plants. 2025;14(8):1209.
CrossRef - Nagata K, Suzuki H, Sakaguchi SJ. Common pathogenic mechanism in development and progression of liver injury caused by non-alcoholic or alcoholic steatohepatitis. Toxicol Sci. 2007;32:453–468.
CrossRef - Nhung TH, Nam NH, Nguyen NTK, et al. Establishment of a standardized mouse model of hepatic fibrosis for biomedical research. Biomed Res Ther. 2014;1(2):43–49.
CrossRef - Otto GP, Rathkolb B, Oestereicher MA, et al. Clinical chemistry reference intervals for C57BL/6J, C57BL/6N, and C3HeB/FeJ mice (Mus musculus). J Am Assoc Lab Anim Sci. 2016;55(4):375–386.
- Ouassou H, Bouhrim M, Daoudi NE, et al. Evaluation of hepatoprotective activity of Caralluma europaea stem extract against CCl₄-induced hepatic damage in Wistar rats. Adv Pharmacol Pharm Sci. 2021;2021:8883040.
CrossRef - Patel J, Roy H, Chintamaneni PK, Patel R, Bohara R. Advanced strategies in enhancing the hepatoprotective efficacy of natural products: integrating nanotechnology, genomics, and mechanistic insights. ACS Biomater Sci Eng. 2025;11(5):2528–2549.
CrossRef - Pullaiah T, Ramaiah M. Handbook of Research on Herbal Liver Protection: Hepatoprotective Plants. 1st ed. Boca Raton: Apple Academic Press; 2021:812.
CrossRef - Rafiq H, Ayaz M, Khan HA, et al. Therapeutic potential of stem cell and melatonin on the reduction of CCl₄-induced liver fibrosis in experimental mice model. Braz J Biol. 2024;84:e253061.
CrossRef - Reitman S, Frankel S. A colorimetric method for the determination of serum glutamic oxalacetic and glutamic pyruvic transaminases. Am J Clin Pathol. 1957;28(1):56–63.
CrossRef - Reitman S, Frankel S. A method of assaying liver enzymes in human serum. Am J Clin Pathol. 1957;28:56–58.
CrossRef - Sahoo D, Rukmini M, Ray R. Quantitative analysis of serum alanine and aspartate aminotransferases, γ-glutamyl transferase and alkaline phosphatase as predictors of liver diseases. Am Int J Res Formal Appl Nat Sci. 2014;9(1):51–55.
- Salim NS, Abo El-Maati MF, Abdelnour SA, Abdel-Alim ME. Hepatoprotective activity of Taraxacum officinale extract against CCl₄-induced liver injury in rats. Food Biosci. 2025;68:106708.
CrossRef - Scholten D, Trebicka J, Liedtke C, Weiskirchen R. The carbon tetrachloride model in mice. Lab Anim. 2015;49(S1):4–11.
CrossRef - Shah G, Patel BG, Shah GB. Development of carbon tetrachloride-induced chronic hepatotoxicity model in rats and its application in evaluation of hepatoprotective activity of silymarin. Asian J Pharm Clin Res. 2017;10(8):274–278.
CrossRef - Shim JY, Kim MH, Kim HD, et al. Protective action of the immunomodulator ginsan against carbon tetrachloride-induced liver injury via control of oxidative stress and inflammatory response. Toxicol Appl Pharmacol. 2010;242(3):318–325.
CrossRef - Silva J, Yu X, Moradian R, et al. Dihydromyricetin protects the liver via changes in lipid metabolism and enhanced ethanol metabolism. Alcohol Clin Exp Res. 2020;44(5):1046–1060.
CrossRef - Singh P, Arif Y, Bajguz A, Hayat S. The role of quercetin in plants. Plant Physiol Biochem. 2021;166:10–19.
CrossRef - Tabet E, Genet V, Tiaho F, et al. Chlordecone potentiates hepatic fibrosis in chronic liver injury induced by carbon tetrachloride in mice. Toxicol Lett. 2016;255:1–10.
CrossRef - Tang Y, Gao C, Xing M, et al. Quercetin prevents ethanol-induced dyslipidemia and mitochondrial oxidative damage. Food Chem Toxicol. 2012;50(5):1194–1200.
CrossRef - Tuzimski T, Petruczynik A. New trends in the practical use of isoquinoline alkaloids as potential drugs applied in infectious and non-infectious diseases. Biomed Pharmacother. 2023;168:115704.
CrossRef - Wang DY, Zhao Y, Jiao YD, et al. Antioxidative and hepatoprotective effects of the polysaccharides from Zizyphus jujube cv. Shaanbeitanzao. Carbohydr Polym. 2012;88:1453–1459.
CrossRef - Wang JY, Chen J, Yang J, et al. Effects of surface charges of gold nanoclusters on long-term in vivo biodistribution, toxicity, and cancer radiation therapy. Int J Nanomedicine. 2016;11:3475–3485.
CrossRef - Wang PY, Kaneko T, Tsukada H, et al. Time courses of hepatic injuries induced by chloroform and by carbon tetrachloride: comparison of biochemical and histopathological changes. Arch Toxicol. 1997;71(10):638–645.
CrossRef - Wang W, Liu M, Fu X, et al. Hydroxysafflor yellow A ameliorates alcohol-induced liver injury through PI3K/Akt and STAT3/NF-κB signaling pathways. Phytomedicine. 2024;132:155814.
CrossRef - Wang YX, Wang Q, Tian Y, et al. Mechanism of different chemicals in animal models of liver injury. Chin J Comp Med. 2022;32:109–114.
- Wei H, Zhao T, Liu X, et al. Mechanism of action of dihydroquercetin in the prevention and therapy of experimental liver injury. Molecules. 2024;29:3537.Xue Y, Deng Q, Zhang Q, Ma Z, Chen B, Yu X, Peng H, Yao S, Liu J, Ye Y, Pan G. Gigantol ameliorates CCl₄-induced liver injury via preventing activation of JNK/cPLA2/12-LOX inflammatory pathway. Sci Rep. 2020;10:22265.
CrossRef - Yang X, Bu T, Ma Y, et al. An updated review of isoquinoline alkaloids: biological activity and mode of action, structural modifications, and agricultural applications. Ind Crops Prod. 2025;234:121591.
CrossRef - Yao P, Nussler A, Liu L, et al. Quercetin protects human hepatocytes from ethanol-derived oxidative stress by inducing heme oxygenase-1 via the MAPK/Nrf2 pathways. J Hepatol. 2007;47(2):253–261.
CrossRef - Yu C, Wang F, Jin C, et al. Increased carbon tetrachloride-induced liver injury and fibrosis in FGFR4-deficient mice. Am J Pathol. 2002;161(6):1–8.
CrossRef - Zhang L, Liu C, Yin L, Huang C, Fan S. Mangiferin relieves CCl₄-induced liver fibrosis in mice. Sci Rep. 2023;13:4172.
CrossRef - Zhang P, Ma D, Wang Y, et al. Berberine protects liver from ethanol-induced oxidative stress and steatosis in mice. Food Chem Toxicol. 2014;74:225–232.
CrossRef - Zhu Y, Jia Y, Zhang E. Oxidative stress modulation in alcohol-related liver disease: from Chinese botanical drugs to exercise-based interventions. Front Pharmacol. 2025;16:1516603.
CrossRef - Zhurakulov ShN, Babkin VA, Chernyak EI, et al. Aminomethylation of 1-aryl-6,7-dimethoxy-1,2,3,4-tetrahydroisoquinolines by dihydroquercetin. Chem Nat Compd. 2015;51(1):57–61.
CrossRef - Zhurakulov ShN, Babkin VA, Chernyak EI, et al. Aminomethylation of 1-aryl-6,7-dimethoxy-1,2,3,4-tetrahydroisoquinolines by dihydroquercetin. Chem Nat Compd. 2015;51(1):57–61.
CrossRef - Zhurakulov ShN, Vinogradova VI, Levkovich MG. Synthesis of 1-aryltetrahydroisoquinoline alkaloids and their analogs. Chem Nat Compd. 2013;49(1):70–74.
CrossRef - Zimmerman HJ, Kodera Y, West M. Effects of carbon tetrachloride poisoning on the plasma levels of cytoplasmic and mitochondrial enzymes in animals with nutritional fatty metamorphosis. J Lab Clin Med. 1965;66:324–333.
- Abdulkhakova GV, Komilov ED, Ergashev NA, Asrarov MI, Makhmudov RR, Kenzhaeva MF. Effects of tannins hexagalloyl-glucose and heptagalloyl-glucose on ATP-dependent potassium channels in vitro. Universum: Chem Biol. 2025;5(131):59–64.
- Abrashova TV, Gushin YaA, Kovaleva MA, et al. Physiological, Biochemical, and Biometric Reference Indicators of Laboratory Animals (Handbook). Petersburg: LeMA; 2013:116.
- Archakov AI, Karuzina NN. Molecular mechanisms of carbon tetrachloride interaction with endoplasmic reticulum membranes of the liver. Adv Hepatol. 1973;4:39–59.
CrossRef - Basu S. Lipid peroxidation induced by carbon tetrachloride: eicosanoid formation and its regulation by antioxidant nutrients. Toxicology. 2003;189:113–127.
CrossRef - Besednova NN, Zaporozhets TS, Kuznetsova TA, et al. Hepatoprotective effects of extracts and polysaccharides of marine algae. Antibiot Chemother. 2014;59(3-4):30–37.
- Blyuger AF, Kartashova OYa. Modeling pathological processes in the liver. In: Experimental Pathology of the Liver (Series 16). 1983:7–16.
- Brattin VYu, Glende YeA, Recknagel RO. Pathological mechanisms of carbon tetrachloride hepatotoxicity. J Free Radic Biol Med. 1985;1:27–38.
CrossRef - Glantz S. Medical and Biological Statistics. Moscow: Praktika; 1999:250–459.
- Davydova VV. Hepatoprotective Properties of Extracts from Coriander Herb (Coriandrum sativum L.) in Toxic Liver Injury. PhD thesis (Pharmaceutical Sciences). Pyatigorsk; 2018:153.
- Dergacheva DI, Klyain OI, Marinichev AA, et al. Hepatoprotective effect of polyphenols in experimental toxic liver pathology induced by thioacetamide. Vestn RGMU. 2019;6:76–82.
- Dorkina EG. Hepatoprotective Properties of Flavonoids (Pharmacodynamics and Prospects for Clinical Study). Doctoral abstract (Biology). Volgograd; 2010:48.
- Dospekhov BA. Field Experiment Methodology (With Basics of Statistical Processing of Research Results). 5th ed. Moscow: Agropromizdat; 1985:347–415.
- Jo‘raqulov ShN. Synthesis, Modification, and Biological Activity of Tetrahydroisoquinoline and Quinazolinone Alkaloids and Their Derivatives. DSc abstract. Tashkent; 2023:5–74. (in Uzbek).
- Zapadnyuk IP, Zapadnyuk VI, Zakhariya YeA, Zapadnyuk BV. Laboratory Animals: Breeding, Maintenance, and Use in Experiments. 3rd ed. Kyiv: Vyshcha Shkola; 1983:380.
- Kaldybaeva AO, Abdusamatov AA. Effects of licorice root, immortelle flowers, and mummy on lipid peroxidation in chronic toxic hepatitis. Med J Uzbekistan. 2010;3:99–101.
- Kamyshnikov VS. Handbook of Clinical-Biochemical Studies and Laboratory Diagnostics. Moscow: Medpress-Inform; 2004:920.
- Kamyshnikov VS. Handbook of Clinical-Biochemical Laboratory Diagnostics. Minsk; 2000:363.
- Karimova GA, Ziyaeva ShT, Kaldybaeva AU. Effect of Darmonal on liver functional status in toxic hepatitis. Austria-Science. 2017;7:16–19.
- Ketova ES, Bibik EYu, Batisheva GA, Krivokolysko SG. Comprehensive evaluation of hepatoprotective activity of new α-cyanothioacetamide derivatives. Far East Med J. 2024;1:22–27.
CrossRef - Kishkun AA. Guide to Laboratory Diagnostic Methods. Moscow: GEOTAR-Media; 2009:800.
- Kishkun AA, Beganskaya LA. Clinical Laboratory Diagnostics. 2nd ed. In 3 vols. Moscow: GEOTAR-Media; 2023:624.
- Kovalenya TA. Effects of Flavonoids and Their Cyclodextrin Complexes on Functional Activity, Structure, and Ca²⁺-Induced Processes in Rat Heart and Liver Mitochondria. PhD abstract (Biology). Grodno; 2024:1-24.
- Kolb VG, Kamyshnikov VS. Handbook of Clinical Chemistry. Minsk; 1982:336.
- Kravchenko LV, Trusov NV, Uskova MA, et al. Characteristics of acute toxic effects of carbon tetrachloride as a model of oxidative stress. Toxicological Bull. 2009;1:12-17.
- Lakin GF. Biometrics. Moscow: Vysshaya Shkola; 1990:284.
- Mallaeva MM, Mustafakulov MA. Effect of polyphenols on activities of antioxidant enzymes in rat liver during hepatitis. Informatics Eng Technol (Conf Proc). 2023;1(2):391-394. (in Uzbek).
- Makhmudov LU, Aripov TF, Vypova NL, et al. Antioxidant and hepatoprotective properties of monoammonium glycyrrhizinate complexes with amino acids and phenolic compounds. Exp Clin Pharmacol. 2023;86(3):23-28.
- Mekhtiev SN, Mekhtieva OA, Berko OM. Rational choice of hepatoprotectors in therapy of chronic liver diseases. Eff Pharmacother. 2023;19(35):50-58.
CrossRef - Miroshnikov MV, Makarova MN. Variability of blood biochemical parameters and establishing reference intervals in preclinical studies (Report 4: mice). Lab Anim Sci Res. 2021;3:63-69.
CrossRef - Mozhokina GN, Elistratova NA, Mikhaylova LP, Makarova OV. Choosing a model of drug-induced liver injury to study new hepatoprotectors in experiments. Antibiot Chemother. 2016;61(5-6):3-7.
- Pechenkina IG, Kozin SV, Bulanov DV. Histomorphological evaluation of hepatoprotective action of phytoadaptogens in mouse liver injury induced by carbon tetrachloride under intensive physical load. Bull Volgograd State Med Univ. 2014;2(50):78-81.
- Pinaeva OG. Structural-Functional State of Liver in White Rats Subjected to Antenatal Hypoxia and Correction by Leu-enkephalin Analogs. Doctoral thesis (Medicine). Khabarovsk; 2017:151.
- Plotnikova OM, Evdokimov AN, Grigorovich MA. Activity of liver enzymes after administration of methylphosphonic acid to laboratory mice. Bull ChGPU. 2011;7:321-327.
- Plokhinskiy NA. Biometrics. Moscow: Moscow State University Press; 1970:20-367.
- Rebrova OYu. Statistical Analysis of Medical Data (Using the STATISTICA Package). Moscow: Media Sfera; 2002:5-312.
- Ronin VS, Starobinets GB. Manual for Practical Classes on Clinical Laboratory Methods. Moscow: Meditsina; 1989:320.
- Safaeva ShT. Indicators of bilirubin metabolism in rats with acute toxic hepatitis receiving Asfervon in a preventive regimen. Infect Immun Pharmacol. 2021;2:253-262.
- Terenteva E, Sasmakov S, Azimov Sh, et al. Antimicrobial activity and toxicity of alkyl-tetrahydroisoquinolines. Bull Taras Shevchenko Natl Univ Kyiv (Biol). 2017;2(74):51-55.
CrossRef - Titova AA, Bilyalov AI, Kiyasov AP, Titova MA. Laboratory Animals for Scientific Research. Kazan: Kazan University; 2021:71.
- Titova NM, Zamay TN, Borovkova GI. Biochemistry and Molecular Biology (Laboratory Manual). Krasnoyarsk: Siberian Federal University; 2007:106.
- Udut VV, Vengerovskiy AI, Korshunov DA, Karkishenko NN. Effect of phospholipid-based hepatoprotectors on bioenergetics and lipid peroxidation of the liver in paracetamol-induced pathology. Biomedicine. 2012;1:120-27.
- Shorina ED, Koshkina DA, Grebnev DYu. Modeling liver fibrosis in mice. In: Proceedings of the VI International (76th All-Russian) Scientific-Practical Conference “Current Issues of Modern Medical Science and Healthcare.” 2021:1515-1520.
- Shutov DV. Hepatoprotective effect of Bergenia extract and silymarin in experimental inhibition of fatty acid β-oxidation induced by 4-pentenic acid. Bull Siber Med. 2007;4:64-70.
CrossRef - Khushmatov SS. Makhmudov RR. Antiarrhythmic activity of the flavonoid fraction of Plantago major extract. Pharmaceutical Chemistry Journal. 2018;52(12):30-33.
CrossRef - Khushmatov SS., Makhmudov RR, Mavlyanov SM. Comparison of inotropic and antiarrhythmic activity of flavonoids – quercetin, rutin and (+)-catechin. Russian Journal of Cardiology. 2015;(11):35-41







