Manuscript accepted on :25-11-2025
Published online on: 10-12-2025
Plagiarism Check: Yes
Reviewed by: Dr. Ilya Nikolaevich Medvedev
Second Review by: Dr. Mina Girgiss
Final Approval by: Dr. Prabhishek Singh
Ashok Kumar Sah1 , Darla Srinivasarao2*
, Darla Srinivasarao2* Nuwo David3
Nuwo David3 , Shagun Agarwal4
, Shagun Agarwal4 , Vijaya Simha3
, Vijaya Simha3 , Pallav Mishra5
, Pallav Mishra5 , Rabab H Elshaikh1
, Rabab H Elshaikh1 , Asaad MA Babker6
, Asaad MA Babker6 , Rashmi A Saibannavar7
, Rashmi A Saibannavar7 and Ranjay Kumar Choudhary1
and Ranjay Kumar Choudhary1
1Department of Medical Laboratory Sciences, College of Applied and Health Sciences, A’ Sharqiyah University, Ibra, Oman.
2Department of Medical Laboratory Sciences, Regional Institute of Paramedical and Nursing Sciences, Aizawl, Mezoram, India.
3Department of Medical Laboratory Science, University Institute of Allied Health Sciences, Chandigarh University, Punjab, India.
4Department of Physiotherapy, School of Allied Health Sciences, Galgotias University, Greater Noida, Uttar Pradesh, India.
5Department of physiotherapy, University Institute of Allied Health Sciences, Chandigarh University, Punjab, India.
6Department of Medical Laboratory Sciences, College of Health Sciences, Gulf Medical University, Ajman, United Arab Emirate.
7Department of Physical Therapy and Health Rehabilitation, College of Applied Medical Sciences, Majmaah University, Majmaah 11981, Saudi Arabia.
Corresponding author: Email: darlasrinivas10@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3283
Abstract
The significant mortality and morbidity linked to Diabetes mellitus Type 2 are triggered by many of vascular complications, consisting of insulin resistance and persistent hyperglycemia. T2DM is a serious issue for people all around the world. Macrovascular outcomes include peripheral artery disease, cardiovascular disease, cerebrovascular accidents, and abdominal aortic aneurysms, all of which are brought on by increasing atherosclerosis due to endothelial cell dysfunction, oxidative damage, and vascular calcification. Microvascular complications, including retinopathy, nephropathy, peripheral neuropathy, as well as delayed healing of wounds, contribute to additional impairment. The challenges that patients with type 2 diabetes frequently experience from an endovascular standpoint are rather distinct and involve diffuse and calcified tumors, decreased collateral damage, smaller blood vessel caliber, as well as a greater likelihood of restenosis following angioplasty or stenting. In this study, we look at the present level of knowledge on the detrimental effects of type 2 diabetes, discuss how it affects endovascular implications, and highlight the difficulties and potential solutions to lessen the disease's vascular impact.
Keywords
Atherosclerosis; Endothelial Dysfunction; Hyperglycemia; Insulin Resistance; Vascular Complications
Download this article as:| Copy the following to cite this article: Sah A. K, Srinivasarao D, David N, Agarwal S, Simha V, Mishra P, Elshaikh R. H, Babker A. M. A, Saibannavar R. A, Choudhary R. K. A Review on Type 2 Diabetes Mellitus: Pathophysiology, Risk Factors, and Vascular Complications. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Sah A. K, Srinivasarao D, David N, Agarwal S, Simha V, Mishra P, Elshaikh R. H, Babker A. M. A, Saibannavar R. A, Choudhary R. K. A Review on Type 2 Diabetes Mellitus: Pathophysiology, Risk Factors, and Vascular Complications. Biomed Pharmacol J 2025;18(4). Available from: https://bit.ly/4iUgPcW |
Introduction
Among the majority of prevalent metabolic disorders worldwide, type 2 diabetes mellitus, also known as T2DM, is primarily brought up through a combination of two primary variables: the pancreatic cells’ lack of ability to produce insulin and the tissues’ inability of responding to insulin.1 The molecular mechanisms behind the production and activity of the hormone, along with the insulin reaction in tissues, need to be strictly controlled since the release of insulin and activity must precisely correspond to the metabolic demand. Thus, the pathophysiology of diabetes of type 2 could stem from an imbalance in metabolism triggered by defects in any one of the mechanisms involved.2 The primary features of diabetes type 2 are discussed in this review, including the molecular processes and mechanisms involved in insulin synthesis as well as the relationship between the condition and cardiac pathology.3 This review discusses the rise of type 2 diabetes worldwide as well as the importance of key risk factors, such as genetic characteristics, gut dysbiosis, epigenetics, obesity, lifestyle variables, and mitochondrial dysfunction.4 It focus on the different molecular and physiological processes that result in type 2 diabetes and its effects. According to the World Health Organization, diabetes mellitus is a chronic metabolic disease marked by high blood sugar levels that eventually damage the kidneys, heart, eyes, blood vessels, and nerves. Tissue resistance to insulin, a poor compensatory insulin secretory response, and insufficient insulin synthesis by pancreatic islet cells are characteristics of type 2 diabetes (T2DM), which accounts for more than 90% of all cases of diabetes mellitus. A high blood sugar level is caused by insulin secretion’s inability to preserve glucose homeostasis as the disorder worsens. Despite a variety of autoimmune processes, including the increased levels of free fatty acids, or FFA, and improper regulation of adipokines, the breakdown of adipose tissue in such a scenario promotes IR.6 The global increase in overweight and obese people, a lack of exercise, calorie-dense meals, and older people are the main drivers of the type 2 diabetes increase, which has quadrupled both the prevalence and incidence of the disease.7 The liver, muscle in the skeleton, renal system, the brain, small intestine, adipose tissue, and pancreas make up the organs associated in the onset of T2DM.8,9 Insulin dysregulation, inflammation, and changes in the gut-microbiota have all been recognized as important disease variables according to increasing research. People within the ages of forty and fifty-nine constitute the most significant category of patients who have diabetes. Geographical location influences the occurrence and severity of type 2 diabetes; more than eighty percent of those affected reside in middle-income or lower nations, thereby rendering efficient treatment harder to achieve. Having cardiovascular disease (CVD), the prevalent cause of death and morbidity rate associated to type 2 diabetes, elevates the likelihood of death from all causes among individuals with the condition by fifteen percent compared to people without the disease.10,11 Both heredity and environmental factors have an influence on the incidence of T2DM. After getting exposed to an atmosphere that is characterized by sedentary behavior as well as elevated calorie intake, hereditary Factors exhaust the disorder.12 While genome-wide association research have identified prevalent glycemic gene variations for type 2 diabetes, these merely account ten percent of the variance associated with the trait, highlighting the significance of rare variants. People from various ethnic groups might show distinct phenotypes that enhance vulnerability to cluster risk factors for cardiovascular disease, which include dyslipidemia, insulin resistance, and hypertension.5
Normal Blood Glucose
Considering the fact that T2DM is linked with an elevated level in blood sugar levels, it is crucial to first figure out the concentration of glucose in the bloodstream, before analyzing the endogenous as well as exogenous variables which influence the concentration of sugar in the blood circulation. In accordance with a person’s size, the levels of sugar in the blood circulation is strictly controlled which can vary between five to ten grams at any given time.13 Multiplying a hundred milligrams per deciliter by one gram or a thousand milligrams by ten deciliters per liter and five liters of blood will yield this outcome.14 There may be a forty percent of glucose due to the red blood cell reserve because red blood cells constitute approximately 40% of the whole volume of blood and the level of glucose is in equilibrium.
Key Exogenous Factors Rising Blood Glucose
Carbohydrates from diet are the main external factor that raises blood sugar levels. Given that the average American in 2021 consumes 200–300 grams of dietary carbohydrates daily, almost all of which are broken down and absorbed as glucose, which the body then sends to the cells through the circulatory system to try to maintain blood sugar levels within normal ranges.15 For instance, a person who eats between 200 and 300 grams of carbohydrates per day will experience between 200,000 and 300,000 milligrams (200 grams = 200,000 milligrams) of glucose flowing into their bloodstream, which is made up of up to ten grams of glucose and has a mean concentration of about 100 milligrams/deciliter.16
Endogenous factors that cause blood glucose levels to rise
The blood glucose is raised by a variety of endogenous factors. Glucagon, Hormone adrenaline, and cortisol constitute the three hormones which increase blood glucose levels.17 By stimulating glycogenolysis and gluconeogenesis, these hormones can raise glucose in the blood levels.18 Through the mechanisms of glucagon production, gluconeogenesis, glycogen storage, and glycogenolysis, the average human body may produce sufficient glucose without any dietary carbohydrates.19
Principal Exogenous Elements that Minimize Blood Glucose
The level of blood glucose can be decreased by consuming less carbohydrates in the diet. Blood levels of glucose often decline with higher physical activity or exercise. There are several drugs that utilize a variety of techniques to reduce blood sugar.19 Drugs can decrease gluconeogenesis (biguanides), decrease insulin resistance (biguanides, thiazolidinediones), boost insulin secretion (sulfonylureas, meglitinides, and GLP-1 agonists and DPP-4 blockers), slow stomach emptying (GLP-1 agonists, DPP-4 inhibitors), increase glucose elimination into the urine (SGLT-2 blockers), and hinder the breakdown of starch and sucrose by the body (alpha-glucosidase inhibitors). Using medication may have negative consequences.20
Principal endogenous factors that reduce blood glucose
Obtaining glucose inside the cells is the primary endogenous technique for decreasing the level of blood glucose (all cells can utilize glucose). It will be possible for glucose to get excreted through the urine if the blood sugar level increases above 180 milligrams per deciliter. Cellular uptake uses glut transporters to decrease levels of glucose in the blood.21 Others have transporters that are not dependent on insulin to activate, whereas certain cells possess transporters that respond to insulin’s presence to activate (glut4).22 Insulin-responsive transporters of glucose in adipose and muscle cells cause blood sugar levels to drop, especially following meals high in carbohydrates. The insulin-sensitive glut4 transporters enable glucose enter adipose tissue and muscle swiftly during physical activity, when the metabolic needs of skeletal muscle may rise by over one hundred times, and throughout the absorptive period (after a meal), preventing major swings in blood glucose levels .23,24
An insulin-dependent glucose transporter’s potential dual function (GLUT4)
The glucose/insulin transporter mechanism is often compared to a lock and key mechanism. Insulin-responsive glut4 transporters are considered to help accelerate the absorption of glucose when insulin levels in the blood are high. However, a lock can be utilized to permit someone inside or prevent them out.25,26 Thus, a particular role of the hormone-responsive glut-4 transporter is to keep glucose from entering muscle and fat cells when insulin levels are low. Notwithstanding, with the demand for insulin, glucose may reach cells that depend on it (known as “glucose-dependent” cells) through non-insulin-responsive transporters. Based on a teleological perspective, cells that need glucose would “make absolutely no sense” if they had insulin-sensitive glut4 transporters. Insulin does not affect the glut1, glut2, glut3, and glut5 transporters found in glucose-dependent cells. Reiterating the key/lock example, it makes no sense to lock a door that you wouldn’t want people to use. In baseline (low insulin) conditions, the cerebral cortex uses the great majority of glucose, and non-insulin responsive glut1 and glut3 transport it as well. Therefore, it is thought that one role of adrenaline-responsive glucose absorption in muscle and adipose tissue is to release glucose for glucose-dependent tissue (such as the renal medulla and blood cells) by preventing glucose from accessing these cells under baseline (low insulin) conditions.27,28
 |
Figure 1: Illustrates the characteristics linked to the risks factors for T2DM, a chronic illness and metabolic disorder in which a patient is resistant to insulin as well as unable to controlglucose levels. Obesity, which is linked to the most prevalent and dangerous kind of body fat centrally situated fat is one of the major contributing factors. |
IR and T2DM: What causes it?
Is it conceivable that insulin resistance (IR) is an adapted (protective) response to an overabundance of glucose accessibility, taking into consideration the possible role of the surroundings? considering that carbohydrates do not provide essential nutrients and that changes in diets in the past few decades have contributed to a spike in the ingestion of processed sugar and bread, hyperinsulinemia could be the primary cause of IR and type 2 diabetes, as cells struggle against elevated insulin and glucose levels.27
Pathophysiology and Mechanisms Contributing to T2DM
Molecular Mechanisms and Pathophysiological Insights
Physiological and dysfunctional Mechanism for the secretion of Insulin in Type 2 Diabetes
Protecting proper cell function involves both maintaining cell integrity as well as careful monitoring of the various channels and mechanisms that control the physiological activity of cells. The synthesis of insulin that is produced from the precursor Preproinsulin to proinsulin is tightly controlled by cells. The development process of pre-proinsulin to proinsulin requires a change in conformation that is supported by the many endoplasmic reticulum (ER) proteins components.29 Insulin precursor subsequently travels to the Golgi apparatus (GA) from the ER, and there it enters immature secretion capillaries and degrades into insulin and C-peptide.30 When insulin becomes mature, it is kept inside the particles until it is ready to release it. High levels of glucose are the primary variable triggering the release of insulin. It is essential to keep in mind that other compounds that could trigger the release of insulin includes chemicals, fatty acids, and amino acids.31 When the messenger molecules generated through binding of ligand or metabolism of nutrients activate the channel, RYR increases Ca2+ signals and leads to the enhancement of the release of insulin.32,33 Yet, additional cellular impulses may additionally assist cells build up and release extra insulin. cAMP could be a particularly significant messenger among them, boosting the release of insulin, accumulated proof suggests that by lowering intracellular calcium reservoirs and increasing extracellular Ca2+ concentrations, cAMP promotes the release of insulin-rich secretory vesicles.
The Mechanisms of Cell Dysfunction
Cell dysfunction has historically been linked to cell destruction. However, recent studies indicate that the cells undergo degeneration in type 2 diabetes could be caused by an intricate network of interactions between the outside world and several biochemical reactions related to the biology of cells.34,35 In a state of excessive food intake, such as that associated with obesity, elevated blood sugar levels and elevated cholesterol levels are common, leading to IR and ongoing inflammation. Due to anomalies associated with their genetic tendency, cells are susceptible to toxic stress in such circumstances, including inflammatory trauma, endoplasmic reticulum (ER) stress, oxidative and metabolic stress, and amyloid stress. The islet’s function could ultimately vanish as a result of these pressures.36 High blood sugar levels and excessive FFAs induce ER stress, which stimulates the apoptotic unfolded protein reaction (UPR) pathways resulting in cell failure. By blocking the sarco/endoplasmic reticulum Ca2+ ATPase (SERCA), a protein in control of ER Ca2+ absorption, triggering IP3 receptors or directly affecting ER homeostasis, stress caused via elevated saturated FFA levels may activate the UPR cascade. Moreover, persistently high levels of glucose promote the production of the proinsulin molecules as well as islets of amyloid polypeptides (IAAP) in cells, and this leads to the development of incorrectly folded insulin as well as IAAP, and this in turn boosts the generation of ROS, or reactive oxygen species, as a consequence of naturally occurring protein folding. These variables additionally influence physiologic ER Ca2+ mobilization in the setting of growing proapoptotic signals, promoting insulin mRNA breakdown, and the production of interleukin (IL)-1, which draws macrophages and increases local islet inflammation. As mentioned before, precise regulation of insulin release is necessary to effectively satisfy this metabolic requirement.37,38 Thus, it is necessary to uphold sufficient islet viability in order that cells may respond to demands from metabolism. In cases of disease, the previously discussed mechanism could ultimately end in a breakdown of the islet organization as well as its integrity, interfere with optimum communication between cells inside the islets of the pancreas, trigger insufficient control of glucagon and insulin release, and eventually worsen hyperglycemia.39
Pathological Factors Leading to Type 2 Diabetes
Aspects of Nutrition
In combination with circulating VLDLs, the chylomicrons, and their remnants, which are formed up of triglycerides (TG), the elevated amounts of carbohydrates and lipids found in the large-calorie Western diet additionally increase blood sugar levels.40,41 As a result, levels of reactive oxygen species, also known as Reactive Oxygen Species (ROS), leading to an excessive manufacturing process for inflammatory chemicals. Since inflammation has been shown to cause the process of oxidative stress, the two conditions cooperate to increase negative postprandial effects after eating a big meal. The continuous and considerable increase in consistent ROS levels has a major impact on the development of T2DM and IR. Thus, the pro-oxidant environment results in the generation of superoxide (O2), stress within the ER, mitochondrial dysfunction, and the activation of a metabolic enzyme called NADPH oxidase (NOX).31 Increased intracellular reactive oxygen species via these channels results in impaired angiogenesis in the aftermath of ischemia, triggering many pro-inflammatory routes and leading to persistent epigenetic alterations that promote inflammatory transcription regardless of when blood sugar levels return to normal. In addition, elevated blood concentrations of fatty acids induce mitochondrial dysfunction via two separate mechanisms: disrupt the transmission of electrons across the respiratory chain of the mitochondria and integrate FFAs into the cell membranes of the mitochondria, which probably favours electron leakage. Figure 1.1 shows the different factors that lead to diabetes.
Exercise
Obesity and type 2 diabetes have been associated with elevated indicators of continuous low-grade chronic inflammation, in addition to a decrease in vigorous exercise and physical activity plus an increase in inactive lifestyles. In this situation, chemicals such as interleukin-6 (IL-6), C-Reactive Protein (CRP), the tumor necrosis factor (TNF-), or IL-1 penetrate the bloodstream as well as cause metabolic inflammation, which is an inflammation that takes place inside specific organs. In fact, the immune system’s attack to pancreatic cells is influenced by IL-1. 42Cell activity decreases and mortality is encouraged when the nuclear factor stimulation combined cell activity reduce the gene transcription regulator kappa-light chain promoter of activated B cells (NF-B). Insulin sensitivity has been improved through the elimination of the macromolecular complex NLRP3 the inflammasome, which generates IL-1 and IL-18, based on preclinical studies. It indicates that reducing inflammation could assist overweight and prediabetic people avoid developing diabetes of the type 2 variety.43 The most common strategy for increasing the response to insulin and, in certain cases, postponing the onset of type 2 diabetes in those suffering from obesity and diabetes is to deliberately reduce their weight. As a consequence of frequent physical activity, anti-inflammatory cytokines such as bioavailable TNF receptors (s-TNF-R) and IL-1 receptors antagonist (IL-1Ra), which are antagonists of TNF- and IL-1, are created more readily as calorie burn levels rise.44 Over time, vigorous activity may reduce oxidative damage caused by type 2 diabetes by increasing the production of radicals such as glutathione (GSH), an important non-enzymatic antioxidant, along with other cell-protective enzymes. Exercise-induced myokine irisin, which increases glucose tolerance, is at least produced by fat and skeletal muscles.45 Irisin levels within the circulatory system are decreased in people with type 2 diabetes than in individuals who do not have the condition, and diabetic individuals with CVD enjoyed substantially lower plasma irisin concentrations than individuals without the condition. In people with type 2 diabetes, lower blood iris in levels have been linked with a 1.6-fold increased risk of CVD incidence.46,47
T2DM and Endovascular
Macrovascular complication
Studies reveal that soon after vascular surgeries such as angioplasties or stenting are performed, those suffering from diabetes are more susceptible to experience restenosis following these procedures.48 Diabetes has been associated to an approximately 43 percent greater relative risk of developing primary restenosis following endovascular therapy, such as balloon angioplasty, or stenting, based on a new meta-analysis encompassing 20 randomized controlled studies with more than 31,000 individuals.49,50 In relation to a separate research study on individuals undergoing coronary stenting or angioplasty, the restenosis rates had been 36.7 percent in those with diabetes as opposed to approximately twenty-five percent in those who were not diabetic; stents with drug-eluting properties were found to decrease but not entirely eradicate the increased risk. Smaller blood vessel size, longer injury length, insulin dependency, and diminished vascular healing effectiveness are all markers for restenosis in patients with diabetes. T2DM also comes along with a greater risk of cerebrovascular disease.51 The increased likelihood of stroke that is ischemic can be triggered by the same mechanisms that include inflammatory processes, dysfunction of endothelial cells, lipid imbalance, and high blood pressure. Even though there have been fewer research investigations that concentrate on the endovascular therapy for cerebrovascular disorders in people with diabetes, the treatment can be difficult partly because of the vascular surroundings, consisting of constricted arteries, diminished endothelial degradation recovery, and an increased likelihood for thrombosis to occur.52 Additionally, one of the most considered clinically significant macrovascular complications is arterial disease of the peripheral arteries, particularly associated with the lower part of the body. More distal (in tibial vessels), widespread, and solidified (medial calcification of arterial walls or Mönckeberg’s disease sclerosis) peripheral arterial disease (PAD) is more prevalent in patients with diabetes.53
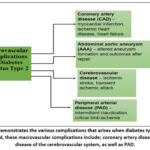 |
Figure 2: demonstrates the various complications that arises when diabetes type 2 is not managed, these macrovascular complications include; coronary artery disease, AAA, disease of the cerebrovascular system, as well as PAD. |
Microvascular complications
The initial step of the earliest possible and most widespread complications of microvascular disease is retinopathy caused by diabetes.54 The hardening of the blood vessel wall, calcium, small vessel rupture, and necrosis in proliferation disorders are all manifestations of chronic high blood sugar, and this additionally triggers oxidative damage, accelerated aging (AGEs), and inappropriate growth hormone (like the vascular endothelial growth factor) expression. If left untreated, these modifications may result in visual impairment by reducing retinal perfusion.
Injury to the glomerular microvasculature, characterized by glomerular excess filtration, thickening within the glomerular basement membrane, the mesangial matrix proliferation, and embryonic podocyte injury, leading to diabetic nephropathy.55,56 Decreased filtration, the appearance of elevated protein levels, and subsequently end-stage renal disease, or ESRD, are the consequences of damage to the endothelial parts of the kidney’s capillary walls and the blood vessel. Both metabolic nerve damage and tiny vessel damage of the nerves are seen in diabetic neuropathy.57 The thickening of the subcutaneous membrane, inadequate blood circulation, and capillary shortages impact the microvessels which nourish nerves; this leads to in hypoxia of the tissues, or an underlying inadequate supply of oxygen, and that can also deplete peripheral nerves of nourishment, resulting in motor, sensory, and autonomic disorders. A person with diabetes who suffers from both neuropathy (deformed sense of touch), peripheral arterial disease and specific biological domain changes (decreased immune capabilities) acquires ulcers in their feet due to diabetes, which all too frequently heal slowly and are prone to infection. Poor wound recovery is another common microvascular complication with severe clinical consequences, in particular to the lower limbs. Unhealed ulcers in diabetic feet may result in blisters and sometimes amputation.58
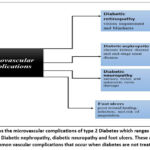 |
Figure 3: shows the microvascular complications of type 2 Diabetes which ranges from Diabetic retinopathy, Diabetic nephropathy, diabetic neuropathy and foot ulcers. |
Core mechanism of endothelium dysfunction
Microvascular embolism within the nervous system and metabolic damage to nerves are both involved in neuropathy caused by diabetes. Membrane basement thickening, inadequate blood circulation, and blood vessel permeability influence the tiny blood vessels that carry blood to nerves; all of these conditions work together to lead to hypoxia in the tissues, or an approximate absence of oxygen, and this may additionally deprive the peripheral nervous system of nourishment, resulting in motor, sensory, and autonomic disorders.59 When an individual experiences both neuropathy (loss of sensation), peripheral arterial disease and other external domain variations (decreased immune capacities), they experience ulcers in their feet from diabetes, and this typically heal gradually and are vulnerable to infection. Poor wound repair is a separate tiny blood vessel condition with significant clinical impact, especially in lower extremities. Persistent ulcers caused by diabetes may end up in inflammation and on occasion amputation. Many researchers emphasize that dysfunction of the endothelial system happens frequently at the beginning of the progression of an illness and, in certain circumstances, even during pre-diabetes, and is detectable in many people. Early vascular damage can be triggered by a variety of variables, such as diminished vascular dilation capacity, a higher expression of attachment molecules (including VCAM-1 and ICAM-1), higher levels in endothelial released microparticles, glycocalyx breakdown, and increased permeability. Good glycemic control, hypertension management, dyslipidemia therapy, and agents with multiple endothelial advantages represent a few of the treatment options to combat endothelial dysfunction.58,59
Conclusion
The relevance of investigating diabetes and homeostasis of glucose has not diminished. In fact, research on this subject has been steadily growing because of the rapid rate of globalization, the accepted norm of a lack of physical activity, and an increase in diabetes, obesity, and other illnesses resulting from these diseases. To mitigate, manage, or reverse the pathophysiology of type 2 diabetes underlying its complications, it is necessary to understand the processes that govern every phase of the progression of the condition and complications. With increasing knowledge of the pathophysiology and fundamental mechanisms of diabetes type 2, personalized medicine could possibly be utilized to target therapies via molecular genetic tools. These tools may be employed to identify particular variations that are linked to the occurrence of the condition and to identify biomarkers that measure its development and response to various therapies. To find out if the microbiota of the gut contributes directly to the pathophysiology of diabetes type 2 and how well therapies work, additional research is required. Type 2 diabetes possesses a variety of serious complications, both microvascular and macrovascular. A vital variable that influences their pathophysiology is dysfunction of endothelial cells. In accordance with endovascular hypothesis, diabetic patients are more inclined to experience slow healing of wounds, less favorable sac shrinkage following abdominal aortic aneurysm (AAA) repair, restenosis following angioplasty or stenting, and procedural difficulties (caused by calcification forming or distal disease). Better results could be achieved, though, with careful planning of the procedure, the use of the appropriate device technology, attentive monitoring, and optimal management of blood pressure, lipids, and glycemic control. In order to maximize endovascular outcomes in type 2 diabetes, research is required to elucidate the relationship between the device, technique, and patient metabolic control.
Acknowledgment
The authors express their sincere gratitude to the Department of Medical Laboratory Sciences, A’ Sharqiyah University, Oman, and the Regional Institute of Paramedical and Nursing Sciences, Aizawl, Mizoram, India, for providing valuable academic and research support during the preparation of this manuscript.
The authors also thank Chandigarh University, Galgotias University, Gulf Medical University, and Jouf University for their cooperation and encouragement in facilitating scholarly collaboration.
Special appreciation is extended to all colleagues and mentors who provided insightful comments, guidance, and assistance throughout the development of this review articl
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not applicable
Author Contributions
- Ashok Kumar Sah: Conceptualization, Literature Review, and Manuscript Drafting.
- Darla Srinivasarao: Corresponding Author; Conceptualization, Critical Revision, and Final Approval of the Manuscript.
- Nuwo David: Data Collection, Literature Curation, and Draft Editing.
- Shagun Agarwal: Review of Physiological and Clinical Sections; Figure and Table Preparation.
- Vijaya Simha: Data Interpretation and Manuscript Proofreading.
- Rajesh Prasad Jayaswal: Support in Literature Analysis and Editing.
- Rabab Elshaikh: Review of Biochemical and Pathophysiological Mechanisms.
- Asaad Babker: Technical Review and Validation of Scientific Content.
- Ranjay Kumar Choudhary: Conceptual Assistance and Supervision of the Overall Work.
References
- Galicia-Garcia U, Benito-Vicente A, Jebari S, Larrea-Sebal A, Siddiqi H, Uribe KB, et al. Pathophysiology of type 2 diabetes mellitus. Int J Mol Sci. 2020;21(17):1–34.
CrossRef - Galicia-Garcia U, Benito-Vicente A, Jebari S, Larrea-Sebal A, Siddiqi H, Uribe KB, Ostolaza H, Martín C. Pathophysiology of Type 2 Diabetes Mellitus. Int J Mol Sci. 2020 Aug 30;21(17):6275. doi: 10.3390/ijms21176275.
CrossRef - Shah MS, Brownlee M, Scherer PE, Hill JA. Molecular and cellular mechanisms of cardiovascular disorders in diabetes. Circ Res. 2016;118(11):1808–1829.
CrossRef - Qaid MM, Abdelrahman MM. Role of insulin and other related hormones in energy metabolism—a review. Cogent Food Agric. 2016;2(1):1267691.
CrossRef - Hameed I, Masoodi SR, Mir SA, Nabi M, Ghazanfar K, Ganai BA. Type 2 diabetes mellitus: From a metabolic disorder to an inflammatory condition. World J Diabetes. 2015;6(4):598–612.
CrossRef - Kasuga M. Insulin resistance and pancreatic β-cell failure. J Clin Invest. 2006;116(7):1756–1760.
CrossRef - Kreier F, Kap YS, Mettenleiter TC, Van Heijningen C, Van Der Vliet J, Kalsbeek A, et al. Tracing from fat tissue, liver, and pancreas: A neuroanatomical framework for the role of the brain in type 2 diabetes. 2006;147(3):1140–1147.
CrossRef - Carvalho BM, Abdalla Saad MJ. Influence of gut microbiota on subclinical inflammation and insulin resistance. Mediators Inflamm. 2013;2013(1):986734.
CrossRef - Glass CK, Olefsky JM. Inflammation and lipid signaling in the etiology of insulin resistance. Cell Metab. 2012;15(5):635–645.
CrossRef - Chen L, Magliano DJ, Zimmet PZ. The worldwide epidemiology of type 2 diabetes mellitus—present and future perspectives. Nat Rev Endocrinol. 2012;8(4):228–236.
CrossRef - Rob A, Hoque A, Asaduzzaman MM, Khatoon MAA, Khalil R, Thomas D, et al. The global challenges of type 2 diabetes. Bangladesh J Med. 2025;36(2):92–98.
CrossRef - Srinivasarao D. Risk stratification based on obesity, hypertension, hyperglycemia, and metabolic syndrome in patients with confirmed bone disorders. Afr J Biomed Res. 2024;27:225–235.
CrossRef - Güemes M, Rahman SA, Hussain K. What is a normal blood glucose? Arch Dis Child. 2016;101(6):569–574.
CrossRef - Hajjawi OS. Glucose transport in human red blood cells. Am J Biomed Life Sci. 2013;1(3):44–52.
CrossRef - World Health Organization. Carbohydrate intake for adults and children: WHO guideline. Geneva: WHO; 2023.
- Wolever TMS. Carbohydrate and the regulation of blood glucose and metabolism. Nutr Rev. 2003;61(Suppl 5):S40–S48.
CrossRef - Nirmalan N, Nirmalan M. Hormonal control of metabolism: Regulation of plasma glucose. Anaesth Intensive Care Med. 2020;21(11):578–583.
CrossRef - Hantzidiamantis PJ, Awosika AO, Lappin SL. Physiology, glucose. 2024.
- Blood glucose levels. Clin Biochem Rev. 2020;41:Pages.
- Houglum J, Harrelson G, Seefeldt T. Drugs for treating diabetes mellitus. Principles of Pharmacology for Athletic Trainers. 3rd ed. 2024;269–290.
CrossRef - Fine J. Glucose content of normal urine. Br Med J. 1965;1(5444):1209.
CrossRef - Zheng Y, Scow JS, Duenes JA, Sarr MG. Mechanisms of glucose uptake in intestinal cell lines: Role of GLUT2. 2012;151(1):13–25.
CrossRef - Russell WR, Baka A, Björck I, Delzenne N, Gao D, Griffiths HR, et al. Impact of diet composition on blood glucose regulation. Crit Rev Food Sci Nutr. 2016;56(4):541–590.
CrossRef - Treem WR. Beta-oxidation defects: Biochemistry and clinical. Clin Liver Dis. 1999;3(1):49–67.
CrossRef - Wang L, Tao T, Su W, Yu H, Yu Y, Qin J. A disease model of diabetic nephropathy in a glomerulus-on-a-chip microdevice. Lab Chip. 2017;17(10):1749–1760.
CrossRef - Furtado LM, Somwar R, Sweeney G, Niu W, Klip A. Activation of the glucose transporter GLUT4 by insulin. Can J Physiol Pharmacol. 2011;80(5):569–578.
CrossRef - Hedeskov CJ. Mechanism of glucose-induced insulin secretion. Physiol Rev. 1980;60(2):442–509.
CrossRef - Vagn Korsgaard T, Colding-Jørgensen M. Time-dependent mechanisms in beta-cell glucose sensing. J Biol Phys. 2006;32(3–4):289–306.
CrossRef - Nonogaki K. Dysglycemia and cardiovascular risk. J Am Coll Cardiol. 2012;60(12):1121–1123.
CrossRef - Grundy SM. Pre-diabetes, metabolic syndrome, and cardiovascular risk. J Am Coll Cardiol. 2012;59(7):635–643.
CrossRef - Galicia-Garcia U, Benito-Vicente A, Jebari S, Larrea-Sebal A, Siddiqi H, Uribe KB, et al. Pathophysiology of type 2 diabetes mellitus. Int J Mol Sci. 2020;21(17):1–34.
CrossRef - Alam S, Hasan MK, Neaz S, Hussain N, Hossain MF, Rahman T. Diabetes mellitus: Insights from epidemiology, biochemistry, risk factors, diagnosis, complications and comprehensive management. 2021;2(2):36–50.
CrossRef - Petersen JL, McGuire DK. Impaired glucose tolerance and impaired fasting glucose—a review of diagnosis, clinical implications and management. Diab Vasc Dis Res. 2005;2(1):9–15.
CrossRef - Wysham C, Shubrook J. Beta-cell failure in type 2 diabetes: Mechanisms, markers, and clinical implications. Postgrad Med. 2020;132(8):676–686.
CrossRef - Ahmad E, Lim S, Lamptey R, Webb DR, Davies MJ. Type 2 diabetes. 2022;400(10365):1803–1820.
CrossRef - Duckworth W, Abraira C, Moritz T, Reda D, Emanuele N, Reaven PD, et al. Glucose control and vascular complications in veterans with type 2 diabetes. N Engl J Med. 2009;360(2):129–139.
CrossRef - Cao H, Oghenemaro EF, Latypova A, Abosaoda MK, Zaman GS, Devi A. Advancing clinical biochemistry: addressing gaps and driving future innovations. Front Med (Lausanne). 2025;12:1521126.
CrossRef - Kamangar F, Dores GM, Anderson WF. Patterns of cancer incidence, mortality, and prevalence across five continents. J Clin Oncol. 2006;24(14):2137–2150.
CrossRef - Lind PM, Lind L. Endocrine-disrupting chemicals and risk of diabetes: An evidence-based review. 2018;61(7):1495–1502.
CrossRef - Serbis A, Giapros V, Tsamis K, Balomenou F, Galli-Tsinopoulou A, Siomou E. Beta cell dysfunction in youth- and adult-onset type 2 diabetes: An extensive narrative review. 2023;15(9):2217.
CrossRef - Garduño-Diaz SD, Khokhar S. Prevalence, risk factors and complications associated with type 2 diabetes in migrant South Asians. Diabetes Metab Res Rev. 2012;28(1):6–24.
CrossRef - Sniderman AD, Furberg CD. Age as a modifiable risk factor for cardiovascular disease. 2008;371(9623):1547–1549.
CrossRef - Liu S, van der Schouw YT, Soedamah-Muthu SS, Spijkerman AMW, Sluijs I. Intake of dietary saturated fatty acids and risk of type 2 diabetes. Eur J Nutr. 2019;58(3):1125–1136.
CrossRef - Maclean HM. Patterns of diet-related self-care in diabetes. Soc Sci Med. 1991;32(6):689–696.
CrossRef - Standl E, Khunti K, Hansen TB, Schnell O. The global epidemics of diabetes in the 21st century: Current situation and perspectives. Eur J Prev Cardiol. 2019;26(Suppl 2):7–14.
CrossRef - Gujral UP, Pradeepa R, Weber MB, Narayan KMV, Mohan V. Type 2 diabetes in South Asians: Similarities and differences with other populations. Ann N Y Acad Sci. 2013;1281(1):51–63.
CrossRef - Matheus ASDM, Tannus LRM, Cobas RA, Palma CCS, Negrato CA, Gomes MDB. Impact of diabetes on cardiovascular disease: An update. Int J Hypertens. 2013;2013:653789.
CrossRef - Yahagi K, Kolodgie FD, Lutter C, Mori H, Romero ME, Finn AV, et al. Pathology of human coronary and carotid artery atherosclerosis and vascular calcification in diabetes mellitus. Arterioscler Thromb Vasc Biol. 2017;37(2):191–204.
CrossRef - Del Turco S, Basta G. An update on advanced glycation end-products and atherosclerosis. 2012;38(4):266–274.
CrossRef - Pricci F, Leto G, Amadio L, Iacobini C, Cordone S, Catalano S, et al. Oxidative stress in diabetes-induced endothelial dysfunction: Involvement of nitric oxide and protein kinase C. Free Radic Biol Med. 2003;35(6):683–694.
CrossRef - Maida CD, Daidone M, Pacinella G, Norrito RL, Pinto A, Tuttolomondo A. Diabetes and ischemic stroke: An overview of the close interaction between these diseases. Int J Mol Sci. 2022;23(4):2397.
CrossRef - Campia U, Gerhard-Herman M, Piazza G, Goldhaber SZ. Peripheral artery disease: Past, present, and future. Am J Med. 2019;132(10):1133–1141.
CrossRef - Faglia E. Characteristics of peripheral arterial disease and its relevance to the diabetic population. Int J Low Extrem Wounds. 2011;10(3):152–166.
CrossRef - Carlo Tenore G, Maisto M, Ya J, Bayraktutan U. Vascular ageing: Mechanisms, risk factors, and treatment strategies. Int J Mol Sci. 2023;24(14):11538.
CrossRef - Cugati S, Varma DD, Chen CS, Lee AW. Treatment options for central retinal artery occlusion. Curr Treat Options Neurol. 2013;15(1):63–77.
CrossRef - Kriz W, Löwen J, Gröne HJ. The complex pathology of diabetic nephropathy in humans. Nephrol Dial Transplant. 2023;38(10):2109–2119.
CrossRef - Mauricio D, Gratacòs M, Franch-Nadal J. Diabetic microvascular disease in non-classical beds: The hidden impact beyond the retina, kidney, and peripheral nerves. Cardiovasc Diabetol. 2023;22(1):1–21.
CrossRef - Li Y, Liu Y, Liu S, Gao M, Wang W, Chen K, et al. Diabetic vascular diseases: Molecular mechanisms and therapeutic strategies. Signal Transduct Target Ther. 2023;8(1):1–29.
CrossRef - Beckman JA, Duncan MS, Damrauer SM, Wells QS, Barnett JV, Wasserman DH, et al. Microvascular disease, peripheral artery disease, and amputation. 2019;140(6):449–458.
CrossRef







