Manuscript accepted on :09-10-2025
Published online on: 04-11-2025
Plagiarism Check: Yes
Reviewed by: Dr. Hassan Shora
Second Review by: Dr. Elina Margarida Ribeiro Marinho
Final Approval by: Dr. Patorn Piromchai
Kajal Pansare* , Yogesh Ahire
, Yogesh Ahire and Vinod Bairagi
and Vinod Bairagi
Department of Pharmacology, Savitribai Phule Pune University Affiliated, KBHSS Trusts Institute of Pharmacy, Malegaon, Nashik, India.
Corresponding Author E-mail: kajalpansare11@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3323
Abstract
Gentamicin, historically widely used and still employed in specific clinical settings, often causes nephrotoxicity primarily through oxidative stress and inflammation, among other mechanisms. This study evaluates the protective effect of Naringenin, a flavonoid antioxidant, and Bromelain, an anti-inflammatory proteolytic enzyme, against gentamicin-induced renal injury in male Wistar rats. Animals were divided into eight groups, including normal and gentamicin controls, standard treatment (N-acetyl cysteine), varying doses of Naringenin and Bromelain, and their combination. Nephrotoxicity was induced with gentamicin (80 mg/kg/day, i.p., for 8 days). Parameters such as body/kidney weight, urine output, biochemical markers, oxidative stress indicators, cytokines, and histopathology were assessed. Gentamicin caused significant renal damage, evidenced by increased serum urea (51.27 ± 6.10 mg/dL) and creatinine (6.13 ± 1.37 mg/dL) compared to control (p < 0.001). Treatment with Naringenin (20 mg/kg) and Bromelain (30 mg/kg) significantly reduced serum creatinine to 3.80 ± 0.71 mg/dL and 4.06 ± 0.58 mg/dL, respectively (p < 0.01 vs. gentamicin). The combination therapy produced the most pronounced effect, lowering serum creatinine to 2.85 ± 0.59 mg/dL and restoring total protein levels by nearly 90% compared to the control group (p < 0.001). Histopathological analysis further confirmed marked recovery of renal architecture, with the combination group showing minimal inflammatory infiltration and near-normal morphology. Overall, the combination therapy showed statistically significant (p < 0.001) superior nephroprotection compared to individual treatments, highlighting its potential as a natural, synergistic alternative for mitigating drug-induced nephrotoxicity.
Keywords
Bromelain; Combination therapy; Gentamicin; N-acetyl cysteine; Naringenin; Nephropathy; Nephroprotective agents; Oxidative stress
Download this article as:| Copy the following to cite this article: Pansare K, Ahire Y, Bairagi V. A Novel Combination Approach using Naringenin and Bromelain for the Amelioration of Gentamicin-Induced Nephrotoxicity in Rats. Biomed Pharmacol J 2025;18(4). |
| Copy the following to cite this URL: Pansare K, Ahire Y, Bairagi V. A Novel Combination Approach using Naringenin and Bromelain for the Amelioration of Gentamicin-Induced Nephrotoxicity in Rats. Biomed Pharmacol J 2025;18(4). Available from: https://bit.ly/3Lk7fmQ |
Introduction
Gentamicin is an aminoglycoside antibiotic still used in specific clinical settings for severe gram-negative infections, owing to its potent bactericidal activity.1-3 However, its clinical efficacy is significantly limited by nephrotoxicity, which affects patients at variable rates, ranging from 10–25%, depending on dosage, treatment duration, and individual patient risk factors.4-5 Gentamicin-induced nephrotoxicity is characterized by acute tubular necrosis and is primarily mediated by oxidative stress, inflammation, and apoptosis in renal tissues.6-10 The accumulation of gentamicin in the renal cortex leads to the generation of reactive oxygen species (ROS), lipid peroxidation (LPO), nitric oxide (NO) overproduction, and the release of pro-inflammatory cytokines such as TNF-α and IL-6, contributing to progressive renal damage.11-14
Given the limitations of conventional therapeutic approaches, increasing attention has been directed towards natural compounds with antioxidant and anti-inflammatory properties for the prevention and management of drug-induced nephrotoxicity.15 Naringenin, a flavonoid mainly found in citrus fruits, shows strong antioxidant, anti-inflammatory, and free radical scavenging activities. It has been reported to modulate oxidative stress and inflammatory pathways in various experimental models of organ injury.16-17 Bromelain, a proteolytic enzyme extracted from the stem and fruit of Ananas comosus (pineapple), has demonstrated significant anti-inflammatory, antioxidant, and immunomodulatory effects, with emerging evidence suggesting its role in protecting renal function.18
Naringenin and Bromelain have been studied alone for their pharmacological benefits,19-20 but no data exist regarding their combined effect against nephrotoxicity. Considering their complementary mechanisms of action, this study evaluates the individual and synergistic nephroprotective effects of Naringenin and Bromelain in a gentamicin-induced rat model of renal toxicity. The study focuses on evaluating renal function markers, oxidative stress parameters, and pro-inflammatory cytokine levels to understand the protective potential of these natural agents.
Materials and Methods
Materials
Gentamicin and N-Acetyl Cysteine was purchased from Sigma-Aldrich, USA. Naringenin (≥98% purity, HPLC grade) and Bromelain (≥97% purity, enzymatic activity grade) were purchased from Yucca Enterprises and Biolaxi Enzymes Pvt. Ltd., Mumbai, respectively. Biochemical, spectrophotometric and ELISA assay kits for serum and antioxidant parameters were obtained from Krishgen Biosystems, Mumbai. All other chemicals as well as reagents used were of analytical grade.
Experimental animals
Healthy adult male albino Wistar rats (200–250 g) were procured from a certified animal house facility. A total of 56 rats were initially acquired to account for potential losses during acclimatization or experimentation, of which 48 rats were finally included and randomized into the study groups. The animals were housed in standard polypropylene cages under controlled environmental conditions: 12-hour light/dark cycle, temperature of 22 ± 2 °C, and relative humidity of 55–65%. They had free access to a standard pellet diet and water ad libitum.21-22 All experimental procedures were carried out in accordance with the guidelines of the Committee for Control and Supervision of Experiments on Animals (CCSEA) for the care and use of laboratory animals. The study protocol was approved by the Institutional Animal Ethics Committee (IAEC) of KBHSS Trust’s Institute of Pharmacy, Malegaon (Approval No.: KBH/IAEC/2024/07-02).
Preparation of drug solutions
Gentamicin (80 mg/kg) was dissolved in normal saline and administered intraperitoneally (i.p.) as a single dose to induce nephropathy.23 N-Acetyl Cysteine (150 mg/kg, p.o.) was dissolved in saline and administered orally.24 Naringenin and Bromelain were suspended separately in 0.5% carboxymethylcellulose for oral administration. All solutions were administered in a standardized volume of 10 mL/kg body weight to ensure dose consistency across groups. All drug preparations were made freshly each day before administration.
Dose selection for Naringenin and Bromelain
Dose of Naringenin and Bromelain was selected from the previously reported literature. The same dose was also standardized in our laboratory in the gentamicin model. Based on this the dose of Naringenin (10 and 20 mg/kg, p.o.) and Bromelain (15 and 30 mg/kg, p.o.) was selected in the present study.25-26 The Bromelain used in this study was derived from pineapple stem with ≥97% enzymatic activity grade, ensuring reproducibility of pharmacological effects.
Induction of nephropathy
Nephropathy was induced in male albino Wistar rats by intraperitoneal administration of gentamicin at a dose of 80 mg/kg body weight, once daily for eight consecutive days. Gentamicin was administered using an insulin syringe under light anesthesia with ketamine (50 mg/kg, i.p.) and xylazine (5 mg/kg, i.p.) to minimize animal discomfort. Following the induction, animals were monitored daily for signs of toxicity, including changes in behavior, food and water intake, and general activity level.27-29
Experimental protocol
A total of 48 male albino Wistar rats were randomly divided into eight groups (n = 6 per group) as follows:25-26
Group I (Normal Control): Received no treatment.
Group II (Disease Control): Received gentamicin (80 mg/kg, i.p.) once daily for eight days.
Group III (Standard): Received N-acetyl cysteine (150 mg/kg, p.o.).
Group IV: Received naringenin low dose (10 mg/kg, p.o.).
Group V: Received naringenin high dose (20 mg/kg, p.o.).
Group VI: Received bromelain low dose (15 mg/kg, p.o.).
Group VII: Received bromelain high dose (30 mg/kg, p.o.).
Group VIII (Combination): Received naringenin (20 mg/kg, p.o.) and bromelain (30 mg/kg, p.o.).
All treatments were administered orally once daily, given 1 hour after gentamicin injection to allow for drug absorption and to assess their protective effect following nephrotoxic insult. This sequence of administration may have influenced the pharmacodynamic interactions observed.
The number of animals per group (n = 6) was selected based on prior published nephrotoxicity studies and standard practice in preclinical pharmacology.23 Although no formal statistical sample size calculation was performed, this group size was considered sufficient to detect biologically and statistically meaningful differences, while adhering to the principle of reduction in animal experimentation.
Initial and final body weights and urine volumes were recorded weekly to monitor nephrotoxic effects and treatment responses.30 On Day 9, animals were euthanized to collect blood samples for biochemical estimations. The kidneys were carefully excised; one kidney from each animal was used to prepare homogenates for the estimation of antioxidant and inflammatory markers, while the other was fixed in 10% formalin for histopathological examination.31-32
Estimation of antioxidant parameters
Kidney tissues were excised, minced, and homogenized in phosphate buffer (pH 7.0) containing a protease inhibitor to prevent protein degradation. A 10% (w/v) tissue homogenate was prepared in accordance with the protocols provided by the respective antioxidant assay kit manufacturers. The homogenate was centrifuged at 10,000 rpm for 20 min at 4 °C, and the resulting supernatant was collected for the estimation of oxidative stress and antioxidant markers. LPO was assessed by measuring thiobarbituric acid reactive substances, and the results were expressed as malondialdehyde (MDA) equivalents. NO levels were estimated based on the accumulation of nitrite using the Griess reagent.33 The antioxidant enzymes assessed included superoxide dismutase (SOD), evaluated based on its ability to inhibit the reduction of nitroblue tetrazolium; catalase (CAT), determined by monitoring the spectrophotometric decomposition of hydrogen peroxide at 240 nm; and reduced glutathione (GSH), estimated as protein-free sulfhydryl groups using 5,5′-dithiobis-2-nitrobenzoic acid.34-36
Estimation of anti-inflammatory parameters
Kidney tissues collected for ELISA were stored at −80 °C until analysis. Prior to processing, the tissues were thawed at room temperature for 15 min and homogenized in the ice-cold buffer provided with the ELISA kits. The homogenates were centrifuged at 12,000 rpm for 30 min at 4 °C, and the resulting supernatants were collected and aliquoted for cytokine estimation. Levels of pro-inflammatory cytokines, including TNF-α and IL-6, were quantified using commercially available ELISA kits, following the instructions of manufacturer. Absorbance was recorded using a microplate reader, and cytokine concentrations were calculated from the standard curves generated for each assay.37 The ELISA kits used had a sensitivity of <2 pg/mL and specificity >95% for rat cytokines, ensuring reliable quantification.
Histopathology
For histopathological evaluation, kidney tissues were fixed in 10% neutral-buffered formalin, embedded in paraffin, and sectioned at a thickness of 5 µm. The sections were stained with hematoxylin and eosin (H&E) to visualize cellular and tissue architecture. Stained slides were examined under a light microscope for histopathological alterations, including tubular necrosis, glomerular damage, and inflammatory cell inflammation.38-39 A blinded pathologist performed a semi-quantitative assessment of renal histological damage using a standardized 0–4 scale, where 0 indicated no abnormality detected (NAD), 1 represented minimal changes, 2 denoted mild changes, 3 reflected moderate changes, and 4 indicated severe pathological alterations. Scoring was carried out independently for glomerular atrophy, tubular degeneration, and inflammatory infiltration.
Statistical analysis
All data are presented as mean ± standard error of the mean (SEM). Statistical analysis was performed using one-way analysis of variance (ANOVA) followed by Tukey’s multiple comparison post hoc test. A p-value < 0.05 was considered statistically significant.40 GraphPad Prism software (version 5.0) was used for data analysis and graphical representation.
Results
General parameters
The effects of various treatments on body weight, kidney weight, and hypertrophy index are summarized in Table 1. Gentamicin administration (Group II) led to a statistically significant (p < 0.001) reduction in body weight on Day 8 compared to Day 0, along with marked increases in absolute kidney weight, relative kidney weight, hypertrophy index, and urine volume (p < 0.001 vs. Group I), indicating nephrotoxicity and renal hypertrophy typically associated with oxidative damage and tubular dysfunction. In contrast, rats treated with the standard antioxidant N-acetyl cysteine (Group III) showed a significant reversal of these changes (p < 0.05 vs. Group II), with improved body weight gain, reduced kidney hypertrophy, and normalized urine volume, demonstrating its partial nephroprotective effect.41 Naringenin-treated groups also exhibited a dose-dependent improvement. The low-dose group (Group IV) significantly ameliorated kidney weights and hypertrophy index (p < 0.05 vs. Group II), while the high-dose group (Group V) showed more pronounced nephroprotection, restoring the values close to the control group. This suggests potent antioxidative and anti-inflammatory properties of Naringenin in attenuating gentamicin-induced renal damage.42 Similarly, Bromelain treatment in both low (Group VI) and high doses (Group VII) resulted in significant decreases in kidney weight and urine output (p < 0.05 vs. Group II), with the high dose being more effective. The combination of Naringenin and Bromelain (Group VIII) offered the most significant protection, with almost normalized body and kidney parameters and urine volume, suggesting a synergistic effect in mitigating gentamicin-induced nephrotoxicity.43 In Table 1, the symbol “#” denotes statistical significance compared to the normal control group (Group I), while “*” denotes significance compared to the disease control group (Group II). The hypertrophy index was calculated as the ratio of kidney weight to body weight (g/100 g), which reflects renal enlargement relative to systemic body mass.
Table 1: Effect of various treatments on body weight, kidney weight, and hypertrophy index in gentamicin-induced nephropathy in rats.
| Group | Body weight | Absolute kidney weight | Relative kidney weight | Hypertrophy index | Final urine volume (ml/24hrs) | |
| Day 0 | Day 8 | |||||
| Group I | 232.5±2.68 | 250.4±3.44 | 1.73±0.07 | 0.65±0.03 | 0.006±0.001 | 6.53±0.39 |
| Group II | 226.1±3.56 | 221.0±3.58### | 2.42±0.07### | 1.08±0.05### | 0.012±0.001### | 13.03±0.77### |
| Group III | 230.6±2.37 | 244.2±3.89* | 1.78±0.08* | 0.70±0.11* | 0.007±0.001* | 6.87±0.76* |
| Group IV | 226.3±3.10 | 232.6±4.07 | 2.21±0.11* | 0.92±0.07* | 0.009±0.001* | 11.22±0.53* |
| Group V | 229.3±3.31 | 241.7±3.05* | 1.90±0.13* | 0.78±0.07* | 0.008±0.001* | 7.92±0.44* |
| Group VI | 226.1±2.48 | 230.0±6.98 | 2.23±0.05* | 0.94±0.05* | 0.009±0.001* | 11.27±0.62* |
| Group VII | 228.5±3.58 | 237.8±2.72* | 1.92±0.11* | 0.78±0.10* | 0.008±0.001* | 7.98±0.42* |
| Group VIII | 230.2±3.38 | 242.8±4.32* | 1.92±0.10* | 0.74±0.09* | 0.007±0.001* | 7.15±0.51* |
Values are expressed as mean ± SEM; n = 6. One-way ANOVA followed by Tukey’s post hoc test.
Significance value: ###p < 0.001 vs. Control (Group I); *p < 0.05, **p < 0.01, ***p < 0.001 vs. Disease Control (Group II).
Biochemical parameters in blood
The biochemical evaluation of serum parameters across various treatment groups (as shown in Fig. 1 (a–e) and summarized in Tables 2) highlights the nephroprotective efficacy of Naringenin and Bromelain in a gentamicin-induced nephropathy model. Gentamicin administration (Group II) induced significant nephrotoxicity in rats, as evidenced by elevated serum urea, uric acid, and creatinine levels, alongside a marked reduction in serum total protein and albumin compared with the normal control group (Group I). These changes confirm acute renal damage and impaired kidney function due to gentamicin-induced oxidative stress and tubular necrosis.44 Treatment with the standard nephroprotective agent, N-acetyl cysteine (Group III), significantly restored serum urea, uric acid, creatinine, total protein, and albumin levels, reflecting effective renal protection.45 Naringenin and Bromelain both showed dose-dependent nephroprotective effects. High-dose Naringenin (Group V) significantly improved serum urea, uric acid, creatinine, total protein, and albumin, outperforming the low-dose group (Group IV). Similarly, high-dose Bromelain (Group VII) led to significant improvements in serum urea, uric acid, creatinine, total protein, and albumin over its low-dose counterpart (Group VI). The combination treatment group (Group VIII) exhibited the most pronounced protective effects among all test groups, showing marked reductions in serum urea, uric acid, and creatinine, along with near-complete restoration of total protein and albumin.46 These results align with previous studies reporting that Naringenin mitigates renal oxidative damage by enhancing antioxidant status and reducing serum creatinine and urea,47 while Bromelain has been shown to improve biochemical markers of nephrotoxicity through anti-inflammatory and proteolytic mechanisms.48
Table 2: Effect of different experimental groups on serum urea, uric acid, creatinine, total protein and albumin levels.
| Group | Serum urea(mg/dL) | Uric acid(mg/dL) | Serum creatinine(mg/dL) | Total protein(g/L) | Serum albumin(g/L) |
| Group I | 15.50 ± 2.15 | 22.03 ± 3.27 | 0.46 ± 0.04 | 7.77 ± 0.78 | 4.73 ± 0.58 |
| Group II | 51.27 ± 6.10### | 52.22 ± 4.34### | 6.13 ± 1.37### | 2.55 ± 0.62### | 1.70 ± 0.55### |
| Group III | 19.67 ± 3.61*** | 25.04 ± 3.62*** | 2.56 ± 0.63** | 6.82 ± 0.49*** | 4.10 ± 0.72*** |
| Group IV | 38.82 ± 4.84* | 42.04 ± 6.82* | 4.73 ± 0.89* | 4.34 ± 0.54* | 2.74 ± 0.79* |
| Group V | 29.15 ± 2.99** | 35.37 ± 3.90** | 3.80 ± 0.71** | 5.41 ± 0.68** | 3.69 ± 0.18** |
| Group VI | 42.70 ± 3.61* | 44.32 ± 3.42* | 4.96 ± 0.57* | 3.94 ± 0.75* | 2.65 ± 0.78* |
| Group VII | 31.03 ± 4.52** | 37.84 ± 3.49** | 4.06 ± 0.58** | 5.19 ± 0.58** | 3.49 ± 0.77** |
| Group VIII | 21.38 ± 3.63*** | 30.04 ± 3.23*** | 2.85 ± 0.59*** | 6.19 ± 0.38*** | 4.13 ± 0.54*** |
Values are expressed as mean ± SEM; n = 6. One-way ANOVA followed by Tukey’s multiple comparison tests.
Significance value: ###p < 0.001 vs. Control (Group I); *p < 0.05, **p < 0.01, ***p < 0.001 vs. Disease Control (Group II).
 |
Figure 1: Effect of various treatments on (a) serum urea, (b) uric acid, (c) creatinine, (d) total protein, and (e) albumin levels in gentamicin-induced nephropathy in rats. |
Estimation of antioxidant parameters
Gentamicin administration (Group II) produced a marked depletion of antioxidant defense mechanisms, as evidenced by decreased levels of GSH, SOD, and CAT, along with a significant increase in lipid peroxidation (MDA levels) (p < 0.001 vs. Group I), as shown in Fig. 2 (a–e) and summarized in Table 3. Treatment with N-acetyl cysteine (Group III) restored antioxidant enzyme levels and reduced LPO, confirming its nephroprotective potential.49
Naringenin treatment exhibited a dose-dependent restoration of antioxidant parameters.50 High-dose Naringenin (Group V) produced a more pronounced significativamente higher increase in GSH, SOD, and CAT compared to the low-dose group (Group IV) (*p < 0.05 to ***p < 0.001 vs. Group II). Bromelain also improved antioxidant markers in a dose-dependent manner, with Group VII (30 mg/kg) showing superior effects over Group VI (15 mg/kg). Importantly, the combination therapy (Group VIII) provided the most profound antioxidant protection,51 restoring GSH, SOD, and CAT levels close to those of the control group and reducing MDA levels by nearly 70% compared to gentamicin-treated animals (p < 0.001 vs. Group II).
Mechanistically, Naringenin enhances antioxidant capacity by activating the nuclear factor erythroid 2–related factor 2 (Nrf2)/ARE pathway, leading to the upregulation of detoxifying enzymes and direct scavenging of free radicals.52 Bromelain reduces oxidative stress indirectly by inhibiting nuclear factor kappa B (NF-κB)–mediated inflammation and promoting the activity of endogenous antioxidant enzymes.53 Their individual actions augment intracellular GSH replenishment and stabilize SOD and CAT activity, resulting in significant antioxidant effects. These findings align with recent studies reporting that Naringenin restores renal antioxidant status in drug-induced nephrotoxicity,54 while Bromelain exhibits complementary antioxidant and anti-inflammatory properties.55
Table 3: Effect of different experimental groups on LPO, NO, SOD, CAT and GSH levels.
| Group | LPO level(nmol MDA/mg protein) | NO level(µmol/g tissue) | GSH level(µmol/g tissue) | SOD level(U/mg protein) | CAT level(U/mg protein) |
| Group I | 6.30 ± 0.82 | 5.23 ± 0.96 | 6.54 ± 0.78 | 16.64 ± 2.34 | 15.44 ± 1.83 |
| Group II | 30.83 ± 4.34### | 21.90 ± 1.87### | 1.88 ± 0.24### | 5.86 ± 1.44### | 4.68 ± 1.27### |
| Group III | 10.42 ± 1.56*** | 7.84 ± 1.98*** | 5.97 ± 1.27*** | 14.34 ± 1.94*** | 13.86 ± 2.15*** |
| Group IV | 23.70 ± 3.75* | 16.80 ± 2.32* | 3.41 ± 0.47* | 9.08 ± 2.19* | 9.29 ± 2.00* |
| Group V | 19.67 ± 1.43** | 12.63 ± 3.25** | 4.79 ± 0.71** | 11.84 ± 1.44** | 11.69 ± 1.28** |
| Group VI | 24.75 ± 3.21* | 17.85 ± 1.67* | 3.18 ± 0.46* | 8.99 ± 1.77* | 8.78 ± 1.41* |
| Group VII | 20.08 ± 2.75** | 13.60 ± 2.25** | 4.09 ± 0.94** | 11.24 ± 2.46** | 10.65 ± 1.61** |
| Group VIII | 12.03 ± 2.22*** | 9.20 ± 1.58*** | 5.99 ± 0.64*** | 14.90 ± 2.59*** | 13.81 ± 1.59*** |
Values are expressed as mean ± SEM; n = 6. One-way ANOVA followed by Tukey’s multiple comparison tests.
Significance values: ###p < 0.001 vs. Control (Group I); *p < 0.05, **p < 0.01, ***p < 0.001 vs. Disease Control (Group II).
 |
Figure 2: Effect of various treatments on (a) LPO, (b) NO, (c) GSH, (d) SOD and (e) CAT levels in gentamicin-induced nephropathy in rats. |
Estimation of anti-inflammatory parameters
Gentamicin administration (Group II) produced a significant elevation of pro-inflammatory cytokines TNF-α and IL-6 (p < 0.001 vs. Group I), confirming the role of inflammatory pathways in drug-induced nephrotoxicity.56 Treatment with N-acetyl cysteine (Group III) significantly reduced cytokine levels, validating its nephroprotective role.57 Both Naringenin and Bromelain produced dose-dependent decreases in TNF-α and IL-6 compared to the disease group, with high-dose groups (V and VII) showing greater efficacy. Notably, the combination group (VIII) exhibited the most profound anti-inflammatory response, with cytokine levels restored nearly to control values (p < 0.001 vs. Group II).58
Mechanistically, Naringenin is known to suppress NF-κB activation and downregulate pro-inflammatory cytokines, while Bromelain exerts anti-inflammatory effects through modulation of NF-κB and MAPK pathways, leading to reduced TNF-α and IL-6 release. Both agents individually produce inhibitory effects on renal inflammatory signaling, thereby enhancing protection against gentamicin-induced damage. These findings are in agreement with recent reports showing that Naringenin attenuates nephroinflammation via NF-κB inhibition and Nrf2 activation in nephrotoxic models,59 while Bromelain has been demonstrated to lower renal cytokine expression and oxidative-inflammatory markers in experimental nephropathy.60
Table 4: Effect of different experimental groups on TNF-α and IL-6 levels.
| Group | TNF-α level(pg/mL protein) | IL-6 Level(pg/mL protein) |
| Group I | 95.79 ± 8.58 | 39.77 ± 7.18 |
| Group II | 242.81 ± 16.04### | 132.48 ± 7.70### |
| Group III | 122.24 ± 16.67*** | 54.94 ± 9.07*** |
| Group IV | 195.81 ± 18.87* | 115.52 ± 7.75** |
| Group V | 152.90 ± 21.39** | 94.93 ± 6.38* |
| Group VI | 198.32 ± 31.59* | 117.37 ± 6.63** |
| Group VII | 162.21 ± 14.93** | 98.36 ± 6.47* |
| Group VIII | 128.40 ± 11.05*** | 60.18 ± 6.93*** |
Values are expressed as mean ± SEM; n = 6. One-way ANOVA followed by Tukey’s multiple comparison tests.
Significance values: ###p < 0.001 vs. Control (Group I); *p < 0.05, **p < 0.01, ***p < 0.001 vs. Disease Control (Group II).
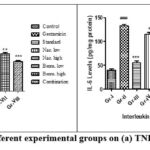 |
Figure 3: Effect of different experimental groups on (a) TNF-α and (b) IL-6 levels. |
Histopathology
The histopathological evaluation of kidney tissues across the experimental groups as illustrated in Fig. 4 (a-h) reveals significant insights into the nephroprotective potential of Naringenin and Bromelain against gentamicin-induced nephropathy. The control group (Group I) exhibited normal renal architecture with intact glomeruli and tubules, confirming the baseline of healthy kidney structure.61 In contrast, the gentamicin-treated group (Group II) demonstrated extensive renal damage characterized by glomerular atrophy, tubular degeneration, and marked inflammatory infiltration, validating the induction of nephrotoxicity.62 Treatment with the standard antioxidant, N-acetyl cysteine (Group III), showed noticeable structural recovery with reduced inflammation and preserved glomerular integrity, though not complete restoration.63 Administration of Naringenin and Bromelain individually at low doses (Groups IV and VI) resulted in modest protective effects with partial recovery of renal histology.64 However, their high-dose counterparts (Groups V and VII) displayed moderate improvements in tissue architecture, with better preservation of glomeruli and tubules and reduced inflammatory signs.65 Remarkably, the combination therapy group (Group VIII), receiving high doses of both Naringenin and Bromelain, exhibited significant improvement in kidney morphology, with minimal evidence of damage or inflammation, suggesting a synergistic protective effect.66-68 These findings collectively indicate that both Naringenin and Bromelain possess nephroprotective properties, and their combination provides enhanced protection against gentamicin-induced renal injury. The semi-quantitative histological scores are summarized in Table 5, highlighting significant reductions in glomerular atrophy, tubular degeneration, and inflammatory infiltration, particularly in the combination group.
Table 5: Semi-quantitative histopathological scores of renal tissues in different treatment groups.
| Group No. | Group Description | Glomerular Atrophy | Tubular Degeneration | Inflammatory Infiltration |
| I | Control | 0 | 0 | 0 |
| II | Gentamicin-treated | 3 | 4 | 3 |
| III | N-acetyl cysteine | 1 | 1 | 2 |
| IV | Naringenin (10 mg/kg) | 3 | 2 | 2 |
| V | Naringenin (20 mg/kg) | 2 | 1 | 2 |
| VI | Bromelain (15 mg/kg) | 2 | 3 | 2 |
| VII | Bromelain (30 mg/kg) | 1 | 2 | 2 |
| VIII | Naringenin (20 mg/kg) + Bromelain (30 mg/kg) | 0 | 1 | 1 |
Scoring Criteria: 0 = No abnormality detected (NAD); 1 = Minimal changes; 2 = Mild changes; 3 = Moderate changes; 4 = Severe changes.
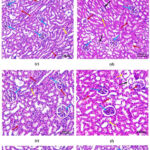 |
Figure 4: Representative photomicrographs of kidney sections stained with Hematoxylin and Eosin (H&E) illustrating renal histology. |
(A) Group I – Control: Normal renal architecture with intact glomerulus indicated by blue arrows. (B) Group II – Cisplatin-treated: Severe renal damage observed—orange arrows show glomerular atrophy, black arrows indicate tubular degeneration, and yellow arrows highlight areas of inflammatory cell infiltration. (C) Group III – NAC: Noticeable structural recovery—blue arrows indicate preserved glomeruli, yellow arrows indicate mild residual inflammation, and red arrows show areas of histological improvement. (D) Group IV – Naringenin (Low Dose): Partial recovery noted—blue arrows denote glomeruli, black arrows show tubular degeneration, yellow arrows indicate inflammation, and red arrows show areas of moderate recovery. (E) Group V – Naringenin (High Dose): Marked improvement—blue arrows denote glomeruli, yellow arrows indicate mild inflammation, and red arrows highlight evident recovery. (F) Group VI – Bromelain (Low Dose): Moderate protective effect—blue arrows show glomeruli, yellow arrows indicate inflammation, black arrows represent tubular regeneration, and red arrows point to recovering tissue. (G) Group VII – Bromelain (High Dose): Significant recovery—blue arrows show intact glomeruli, yellow arrows indicate mild inflammation, black arrows shows tubular degeneration, and red arrows show histological improvement. (H) Group VIII – Naringenin + Bromelain (High Dose): Near-complete restoration of renal structure—blue arrows indicate preserved glomeruli, and red arrows show minimal residual damage with substantial tissue recovery.
Discussion
This study systematically evaluated the nephroprotective potential of Naringenin and Bromelain, both alone and in combination, against gentamicin-induced nephrotoxicity in a rat model. Gentamicin, although clinically valuable, is notorious for causing acute kidney injury via oxidative stress, inflammation, and tubular epithelial cell damage.69 Our findings reinforce the role of oxidative and inflammatory pathways in gentamicin-induced renal dysfunction and demonstrate the effectiveness of Naringenin and Bromelain in mitigating these effects.
The administration of gentamicin induced characteristic renal dysfunction, as evidenced by significant weight loss, increased kidney mass, and elevated hypertrophy index, along with marked derangements in serum renal biomarkers. These outcomes align with established models of gentamicin-induced nephropathy, highlighting the clinical relevance of the model.70 N-acetyl cysteine, a known antioxidant, offered moderate nephroprotection, confirming the important role of oxidative stress in the pathology.
Both Naringenin and Bromelain, known for their free radical-scavenging and anti-inflammatory properties, showed clear dose-dependent nephroprotective effects. Their administration improved renal function markers, decreased oxidative burden, and downregulated pro-inflammatory cytokines TNF-α and IL-6.71 Importantly, the low- and high-dose groups demonstrated statistically significant differences, reinforcing the dose–response relationship observed in this study. High-dose treatments yielded greater benefits, indicating a dose-response relationship. Notably, the combination therapy was most effective across all evaluated parameters, indicating synergistic protective effects.
Mechanistically, Naringenin preserves mitochondrial integrity, inhibits apoptosis, activates the Nrf2 signaling pathway to upregulate antioxidant enzymes (SOD, CAT, GSH), and suppresses NF-κB–mediated inflammation.72 Bromelain similarly inhibits NF-κB and downregulates pro-inflammatory mediators while potentially modulating TGF-β1, a key fibrogenic cytokine, thereby reducing the risk of renal fibrosis and progression to chronic kidney disease.73
Pharmacokinetically, Naringenin shows limited oral bioavailability due to first-pass metabolism,74 whereas Bromelain is absorbed in its active form and retains systemic proteolytic activity after oral administration. These complementary profiles may explain the enhanced nephroprotection observed with their combination.75
The restoration of antioxidant enzymes (GSH, SOD, CAT), reduction in LPO and NO levels, and suppression of inflammatory cytokines collectively demonstrate the ability of these compounds to modulate redox balance and immune responses in renal tissues.76 Histopathological findings further confirmed the protective effects, with the combination group showing the greatest preservation of renal architecture.
However, certain limitations must be acknowledged. The absence of molecular-level validation, such as Western blotting or PCR to confirm Nrf2, NF-κB, or TGF-β1 involvement, restricts mechanistic insights. Additionally, long-term toxicity and safety studies were not conducted, which are essential for clinical translation. Future research should incorporate molecular assays, pharmacokinetic studies, and chronic toxicity models to strengthen the therapeutic relevance of this combination strategy.
Conclusion
The present study demonstrates that both Naringenin and Bromelain alone exhibit significant nephroprotective effects against gentamicin-induced renal toxicity through their antioxidant and anti-inflammatory properties. Gentamicin administration led to marked renal dysfunction, oxidative stress, and inflammatory responses, as evidenced by alterations in body and kidney weight, biochemical markers, cytokine levels, antioxidant defenses, and histopathological features. Treatment with N-acetyl cysteine, a known nephroprotective agent, validated the study model and offered partial protection.
Notably, the combination of high-dose Naringenin and Bromelain provided the most comprehensive improvement in renal parameters, reducing oxidative and inflammatory markers and preserving kidney architecture. However, given the preclinical nature of this work, these findings should be interpreted as a potential therapeutic approach rather than a definitive strategy. A key limitation is that the results are derived from an animal model, and further mechanistic studies as well as clinical trials will be required before translation to human use. Additionally, although no overt adverse effects were observed during the short-term study period, the long-term safety profile of Naringenin and Bromelain in combination remains to be established and must be carefully assessed in future investigations. In summary, while the findings highlight the potential of Naringenin and Bromelain as complementary agents against gentamicin-induced nephrotoxicity, their clinical applicability will depend on further validation and safety evaluation.
Acknowledgement
The authors thank the management of KBHSS Trusts Institute of Pharmacy, Malegaon for providing necessary facilities and support. We also acknowledge the Institutional Animal Ethics Committee for study approval and the laboratory staff for their technical assistance.
Funding Source
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
The study protocol was approved by the Institutional Animal Ethics Committee of KBHSS Trust’s Institute of Pharmacy, Malegaon, under reference number KBH/IAEC/2024/07-02.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable.
Author Contributions
- Kajal Pansare conducted the experimental work, data collection, statistical analysis, and manuscript drafting as part of her doctoral research.
- Vinod Bairagi, as research guide, conceptualized and supervised the study, provided critical insights into the experimental design, and revised the manuscript for intellectual content.
- Yogesh Ahire assisted in refining the methodology, provided guidance during data interpretation, and contributed to manuscript review and editing.
References
- Mishra P, Mandlik D, Arulmozhi S, Mahadik K. Nephroprotective role of diosgenin in gentamicin-induced renal toxicity: biochemical, antioxidant, immunological and histopathological approach. Future Journal of Pharmaceutical Sciences. 2021 Aug 26;7(1):169. https://doi.org/10.1186/s43094-021-00318-z
CrossRef - Ahmed HE, Abdlrahman SH, Ahmed Mohamed S, Bilal TA, Elghazali FE, Elbadwi SM. Nephroprotective effect of acacia senegal (Gum Arabic) against gentamicin induced nephrotoxicity in rats. J. Res.-Granthaalayah. 2022;10:120-128.
CrossRef - Thy M, Timsit JF, de Montmollin E. Aminoglycosides for the treatment of severe infection due to resistant Gram-negative pathogens. Antibiotics (Basel). 2023;12(5):860. https://doi.org/10.3390/antibiotics12050860
CrossRef - Bai R, Fan J, Wang Y, Wang Y, Li X, Hu F. Protective effect of Cistanche deserticola on gentamicin-induced nephrotoxicity in rats. Chinese Herbal Medicines. 2023 Jan 1;15(1):102-109.
CrossRef - Amir S, Abid M, Nadeem H, Tipu MK, Irshad N. The nephroprotective potential of selected synthetic compound against gentamicin induced nephrotoxicity. BMC Pharmacology and Toxicology. 2024 Sep 27;25(1):68. https://doi.org/10.1186/s40360-024-00765-3
CrossRef - Bencheikh N, Bouhrim M, Kharchoufa L, et al. The nephroprotective effect of Zizyphus lotus L.(desf.) fruits in a gentamicin-induced acute kidney injury model in rats: a biochemical and histopathological investigation. Molecules. 2021 Aug 8;26(16):4806. https://doi.org/10.3390/molecules26164806
CrossRef - Mamri S, Daoudi NE, Marghich M, et al. Protective effect of Crocus sativus stamens extract on gentamicin-induced nephrotoxicity and oxidative damage in rat kidney. Journal of Experimental Biology and Agricultural Sciences. 2022;10(1):73-82.
CrossRef - Zeima NM, EL-Gawish AM. The Prophylactic Effect of some herbs extract on Gentamicin Induced Nephrotoxicity in Albino Rats. Egyptian Journal of Nutrition and Health. 2021 Jul 1;16(2):35-52.
CrossRef - Zhang M, Zhou Y, Wang X, Li Y, Wu X. Gentamicin aggravates renal injury by affecting mitochondrial dynamics, altering renal transporters expression, and exacerbating apoptosis. Toxicol Lett. 2025;412:55-67. https://doi.org/10.1016/j.toxlet.2025.07.1418
CrossRef - Abouzed TK, Sherif EA, Barakat ME, et al. Assessment of gentamicin and cisplatin-induced kidney damage mediated via necrotic and apoptosis genes in albino rats. BMC Veterinary Research. 2021 Dec;17:1-9.
CrossRef - Tomşa AM, Răchişan AL, Pandrea SL, et al. Accelerated lipid peroxidation in a rat model of gentamicin nephrotoxicity. Exp Ther Med. 2021;22(5):1218. https://doi.org/10.3892/etm.2021.10652
CrossRef - Meka Kedir W, Dukassa Dubiwak A, Tofik Ahmed E. Nephroprotective effect of asparagus africanus lam. root extract against gentamicin‐induced nephrotoxicity in swiss albino mice. Journal of Toxicology. 2022;2022(1):8440019. https://doi.org/10.1155/2022/8440019
CrossRef - Khalili N, Ahmadi A, Ghodrati Azadi H, Moosavi Z, AbedSaeedi MS, Baghshani H. Protective effect of betaine against gentamicin-induced renal toxicity in mice: a biochemical and histopathological study. Comparative Clinical Pathology. 2021 Dec;30:905-12. https://doi.org/10.1007/s00580-021-03285-2
CrossRef - Nassan MA, Soliman MM, Aldhahrani A, Althobaiti F, Alkhedaide AQ. Ameliorative impacts of Glycyrrhiza glabra root extract against nephrotoxicity induced by gentamicin in mice. Food science & nutrition. 2021 Jul;9(7):3405-3413.
CrossRef - Pansare K, Upasani C, Upaganlwar A, Sonawane G, Patil C. Streptozotocin and alloxan induced diabetic nephropathy: protective role of natural products. Journal of the Maharaja Sayajirao University of Baroda. 2021;55(1):86-102.
- Pansare K, Sonawane G, Mahajan T, Khairnar S, Patil C, Andhale A. Protective role of natural products in alloxan and streptozotocin induced diabetic nephropathy: a concise review. NVEO-Natural Volatiles & Essential Oils Journal. 2021;8(6):3977-3994.
- Hakyemez IN, Cevizci MN, Aksoz E, Yilmaz K, Uysal S, Altun E. Protective effects of p-coumaric acid against gentamicin-induced nephrotoxicity in rats. Drug and Chemical Toxicology. 2022 Nov 2;45(6):2825-2832.
CrossRef - Pansare K, Ahire Y, Bairagi V. Naringenin: A Potential Flavonoid Phytochemical For Diabetes Management. Biotech Res Asia 2025;22(3). Available from: https://bit.ly/48kQSQe
CrossRef - Mohamed SA, Abo-Elghiet F, Ahmed SF, Abd Al Haleem EN, El-Tantawy WH, Yasin NA. Melodorum fruticosum Lour. Leaves Methanolic Extract Ameliorates Gentamicin-Induced Renal Toxicity in Rats via Antioxidant, Anti-Inflammatory, and Anti-Apoptotic Pathways. Egyptian Journal of Chemistry. 2024 Aug 1;67(8):547-567.
CrossRef - Ezrari S, Khadda ZB, Boutagayout A, et al. Health risks and toxicity mechanisms of medicinal and aromatic plants (MAPs): A comprehensive review of adverse effects on organ systems, genotoxicity and reproductive toxicity. Fitoterapia. 2025 May 19:106630. https://doi.org/10.1016/j.fitote.2025.106630
CrossRef - Solanki S, Modi CM, Patel HB, Patel UD, Singh V. Protective Effect of Polyherbal Extract on Gentamicin-Induced Renal Injury in Wistar Rats. Indian Journal of Pharmaceutical Sciences. 2022 Mar 1;84(2):369-379.
CrossRef - Ijaz MU, Akbar A, Ashraf A, Alkahtani S, AlKahtane AA, Riaz MN. Antioxidant, anti-inflammatory and anti-apoptotic effects of amentoflavone on gentamicin-induced kidney damage in rats. Journal of King Saud University-Science. 2023 Oct 1;35(7):102791. https://doi.org/10.1016/j.jksus.2023.102791
CrossRef - Erseçkin V, Mert H, İrak K, Yildirim S, Mert N. Nephroprotective effect of ferulic acid on gentamicin-induced nephrotoxicity in female rats. Drug and chemical toxicology. 2022 Mar 4;45(2):663-669.
CrossRef - Elsayed A, Elkomy A, Elkammar R, et al. Synergistic protective effects of lycopene and N-acetylcysteine against cisplatin-induced hepatorenal toxicity in rats. Scientific reports. 2021 Jul 7;11(1):13979. https://doi.org/10.1038/s41598-021-93196-7
CrossRef - A Das, V Bhardwaj, GL Viswanatha, H Shylaja, H Gajendra and CHKVLSN Anjana Male. Unlocking Naringin’s potential: a systematic review and meta-analysis of its nephroprotective effects in pre-clinical models. Journal of Biochemical and Molecular Toxicology 2025; 39(2), e70149. https://doi.org/10.1002/jbt.70149
CrossRef - Acar MD, Özdemir D, Erdal A, Koc Sahin S. Histopathological evaluation of the effect of bromelain on myringosclerosis in rats. J Int Adv Otol. 2025;21(1):1-6. https://doi.org/10.5152/iao.2025.241591
CrossRef - Hilmi SR, Dewan ZF, Kabir AN, et al. Effect of Silymarin on Gentamicin Induced Nephrotoxicity in Rats. Bangladesh Journal of Infectious Diseases. 2023 Dec 31;10(2):71-76.
CrossRef - Fatimah SN, Usman SM. Protective role of salicylic acid in gentamicin induced nephrotoxicity. Pakistan Journal of Pharmaceutical Sciences. 2021 May 1;34(3):869–874.
- Bhat MM, Bhat VR, Parida A, Sushma RK, Poojar B, Manju V. Evaluation of nephroprotective effect of vortioxetine in gentamicin-induced renotoxicity in wistar rats. Research Journal of Pharmacy and Technology. 2023;16(5):2223-2228.
CrossRef - Ahmed MS, Eami EJ. Histological changes of the kidney in gentamycin induced nephrotoxicity and protected role of fish oil. Central Medical College Journal. 2024 Dec 15;8(1):12-16.
CrossRef - Pansare K, Upasani C, Upaganlwar A, Sonawane G, Patil C. Lycopene alone and in combination with olive oil ameliorates streptozotocin-induced diabetic nephropathy in rats. Vidyabharati International Interdisciplinary Research Journal (Special Issue). 2021;1:320–332.
- Şahin Y, Alçığır ME, Şenol A, et al. Protective Effect of Krill Oil Against Gentamicin Induced Oxidative Stress Mediated Nephrotoxicity in Rats. Kocatepe Veterinary Journal. 2022 Feb 16;15(1):38-46.
CrossRef - Elsawah HK, Mokhimar HM, Kandiel MM, Farid AS, El-Mahmoudy AM. Potential protective effect of alpha lipoic acid against testicular oxidative stress and altered gene expression induced by gentamicin treatment. Научные результаты биомедицинских исследований. 2024;10(2):234-243.
CrossRef - Genid AA, Ibrahim A, Elagwany AM, Yehia MA, Biram DM. The Effect of Ciprofloxacin on Renal Cortex of Adult Male Albino Rat and the Possible Protective Role of Olive Oil: Anatomical and Histological Study. Egyptian Journal of Histology. 2023 Sep 1;46(3):1494-1511.
- Rajesham VV, Kandoti HS, Raghavendra M, Ali PR, Rao TR. Nephroprotective Activity of Ethanolic Extract of Carissa carandas Leaves against Gentamicin-Induced Acute Kidney Injury in Wistar Albino Rats. Journal of Pharmaceutical Research International. 2021 Dec 15;33(58A):279-286.
CrossRef - Akram E, Waheed A, Munawar R, Mumal S, Azeem A. Comparative effects of parsley leaves aqueous extract and ramipril on gentamicin induced nephrotoxicity in rats. Journal of University Medical & Dental College. 2022 Nov 3;13(4):478-482.
CrossRef - Ijaz N, Iftikhar J, Tayyab M, Javed S, Aslam H, Salim S. Effect of N Acetyl Cysteine and Aqueous Extract of Berberis Lycium Royale Root Bark on Uric Acid of Albino Rats. Annals of Punjab Medical College. 2021 Jun 30;15(2):101-104.
- Tawfeek SE, Abd El Salam LA. Histological and Biochemical Study on the Possible Protective Effect of Nebivolol on Gentamicin Induced Nephrotoxicity in Adult Male Albino Rats. Egyptian Journal of Histology. 2021 Mar 1;44(1):286-294.
- Adikwu E, Nnanna TB, Safia K. Antituberculosis drug-induced hepatotoxicity: Preclinical benefit of glutamine. Eurasian Journal of Toxicology. 2024 Dec 1;6(3):43-49.
CrossRef - Oliveira SL, Costa CC, Conde G, et al. Safety of oral doxycycline treatment in Nile tilapia, Oreochromis niloticus. Ars Veterinaria. 2022 Sep 28;38(3):127-138.
CrossRef - Gamaan MA, Zaky HS, Ahmed HI. Gentamicin-induced nephrotoxicity: A mechanistic approach. Azhar International Journal of Pharmaceutical and Medical Sciences. 2023 Jun 1;3(2):11-19.
- Thangarajoo T, Hsin YK, Pandey M, et al. A stimuli-responsive in situ spray hydrogel co-loaded with naringenin and gentamicin for chronic wounds. Open Chemistry. 2023 Jul 20;21(1):20220357. https://doi.org/10.1515/chem-2022-0357
CrossRef - Wajih N, Erali RA, Forsythe SD, et al. Enhancing the efficacy of HIPEC through Bromelain: a preclinical investigation in appendiceal cancer. Annals of surgical oncology. 2024 Aug;31(8):5377-5389.
CrossRef - Laaroussi H, Bakour M, Ousaaid D, et al. Protective Effect of Honey and Propolis against Gentamicin‐Induced Oxidative Stress and Hepatorenal Damages. Oxidative medicine and cellular longevity. 2021;2021(1):9719906. https://doi.org/10.1155/2021/9719906
CrossRef - Elhalaby B, Said A, Youssef G, Aboubakr M. Antioxidant and anti-apoptotic roles of sesame oil and N acetylcysteine against gentamicin-induced nephrotoxicity in rats. Benha Veterinary Medical Journal. 2022 Sep 1;42(2):82-87.
CrossRef - Gorain B, Pandey M, Leng NH, et al. Advanced drug delivery systems containing herbal components for wound healing. International Journal of Pharmaceutics. 2022 Apr 5;617:121617. https://doi.org/10.1016/j.ijpharm.2022.121617
CrossRef - Valle-Velázquez E, Zambrano-Vásquez OR, Cortés-Camacho F, Sánchez-Lozada LG, Guevara-Balcázar G, Osorio-Alonso H. Naringenin – a potential nephroprotective agent for diabetic kidney disease: A comprehensive review of scientific evidence. Biomolecules & Biomedicine. 2024;24(6):1441-51. https://doi.org/10.17305/bb.2024.10511
CrossRef - El-Demerdash FM, Hussien DM, Ghanem NF, Al-Farga AM. Bromelain modulates liver injury, hematological, molecular, and biochemical perturbations induced by aluminum via oxidative stress inhibition. Biomed Res Int. 2022;2022:5342559. https://doi.org/10.1155/2022/5342559
CrossRef - Antar S, Al-Karmalawy AA, Mourad A, et al. Protective effects of mirazid on gentamicin-induced nephrotoxicity in rats through antioxidant, anti-inflammatory, JNK1/iNOS, and apoptotic pathways; novel mechanistic insights. Pharmaceutical Sciences. 2022 Jan 29;28(4):525-540.
CrossRef - Satvati M, Salehi-Vanani N, Nouri A, Heidarian E. Protective effects of N-acetyl cysteine against oxidative stress in ibuprofen-induced hepatotoxicity in rats. Comparative Clinical Pathology. 2022 Apr;31(2):293-301.
CrossRef - Veiko AG, Olchowik-Grabarek E, Sekowski S, et al. Antimicrobial activity of quercetin, naringenin and catechin: Flavonoids inhibit Staphylococcus aureus-induced hemolysis and modify membranes of bacteria and erythrocytes. Molecules. 2023 Jan 27;28(3):1252. https://doi.org/10.3390/molecules28031252
CrossRef - Mehranfard N, Ghasemi M, Rajabian A, Ansari L. Protective potential of naringenin and its nanoformulations in redox mechanisms of injury and disease. Heliyon. 2023;9(12):e22820. https://doi.org/10.1016/j.heliyon.2023.e22820
CrossRef - Bakare AO, Owoyele BV. Bromelain reduced pro-inflammatory mediators as a common pathway that mediate antinociceptive and anti-anxiety effects in sciatic nerve ligated Wistar rats. Sci Rep. 2021;11(1):289. https://doi.org/10.1038/s41598-020-79421-9
CrossRef - Saati AA. Naringenin’s neuroprotective effect on diazino-induced cerebellar damage in male albino rats, with modulation of acetylcholinesterase. Brain Sci. 2025;15(3):242. https://doi.org/10.3390/brainsci15030242
CrossRef - Kansakar U, Trimarco V, Manzi MV, Cervi E, Mone P, Santulli G. Exploring the therapeutic potential of bromelain: Applications, benefits, and mechanisms. Nutrients. 2024;16(13):2060. https://doi.org/10.3390/nu16132060
CrossRef - Akbaribazm M, Goodarzi N, Rahimi M, Naseri L, Khazaei M. Anti-inflammatory, anti-oxidative and anti-apoptotic effects of Heracleum persicum L. extract on rats with gentamicin-induced nephrotoxicity. Asian Pacific Journal of Tropical Biomedicine. 2021 Feb 1;11(2):47-58.
CrossRef - Abdelrahaman D, Shanab O, Abdeen A, et al. Bee venom ameliorates gentamicin-induced kidney injury by restoring renal aquaporins and enhancing antioxidant and anti-inflammatory activities in rats. Frontiers in Pharmacology. 2025 Apr 17;16:1525529. https://doi.org/10.3389/fphar.2025.1525529
CrossRef - Al-Amer HA, Al-Sowayan NS, Alfheeaid HA, et al. Oral administration of naringenin and a mixture of coconut water and Arabic gum attenuate oxidative stress and lipid peroxidation in gentamicin-induced nephrotoxicity in rats. European Review for Medical & Pharmacological Sciences. 2023 Nov 1;27(21). https://doi.org/10.26355/eurrev_202311_34317
- Zhang B, Wan S, Liu H, et al. Naringenin alleviates renal ischemia reperfusion injury by suppressing ER stress-induced pyroptosis and apoptosis through activating Nrf2/HO-1 signaling pathway. Oxid Med Cell Longev. 2022;2022:5992436. https://doi.org/10.1155/2022/5992436
CrossRef - Pereira IC, Sátiro Vieira EE, de Oliveira Torres LR, Carneiro da Silva FC, de Castro E Sousa JM, Torres-Leal FL. Bromelain supplementation and inflammatory markers: A systematic review of clinical trials. Clin Nutr ESPEN. 2023;55:116-27. https://doi.org/10.1016/j.clnesp.2023.02.028
CrossRef - Hassan DS, Rakha GM, Mousa SA, Korany R. Evaluation of gentamicin induced nephrotoxicity in canine: clinical, hematological, biochemical, ultrasonographic and histopathological findings. Exploratory Animal & Medical Research. 2022 Jun 1;12(1):33-45.
CrossRef - Sabra MS, Allam EAH, Darwish MHA, et al. A novel nanoparticle glutathione and Lepidium sativum treatment for gentamicin-induced acute renal failure in rats. Sci Rep. 2025;15:20158. https://doi.org/10.1038/s41598-025-05385-3
CrossRef - Ye M, Lin W, Zheng J, Lin S. N-acetylcysteine for chronic kidney disease: a systematic review and meta-analysis. Am J Transl Res. 2021;13(4):2472-85.
- Ammar NM, Hassan HA, Abdallah HMI, Afifi SM, Elgamal AM, Farrag ARH, El-Gendy AEG, Farag MA, Elshamy AI. Protective effects of naringenin from Citrus sinensis (var. Valencia) peels against CCl4-induced hepatic and renal injuries in rats assessed by metabolomics, histological and biochemical analyses. Nutrients. 2022;14(4):841. https://doi.org/10.3390/nu14040841
CrossRef - Weinzierl A, Harder Y, Schmauss D, Menger MD, Laschke MW. Bromelain protects critically perfused musculocutaneous flap tissue from necrosis. Biomedicines. 2022;10(6):1449. https://doi.org/10.3390/biomedicines10061449
CrossRef - Sokar SS, Abu-Risha SE, Alkabbani MA, et al. Protective effects of melatonin and naringenin against acitretin-induced hepatotoxicity via modulation of oxidative stress and inflammatory signaling. Sci Rep. 2025;15:31629. https://doi.org/10.1038/s41598-025-16740-9
CrossRef - Recinella L, Pinti M, Libero ML, et al. Beneficial effects induced by a proprietary blend of a new bromelain-based polyenzymatic complex plus N-acetylcysteine in urinary tract infections: results from in vitro and ex vivo Antibiotics (Basel). 2024;13:985. https://doi.org/10.3390/antibiotics13100985
CrossRef - Pansare K, Ahire Y, Bairagi V. Nephroprotective effect of naringenin and bromelain against cisplatin-induced renal toxicity in rats. Trends in Sci. 2025;22(11):10681. https://doi.org/10.48048/tis.2025.10681
CrossRef - Matouk AI, Awad EM, Mousa AAK, et al. Dihydromyricetin protects against gentamicin-induced nephrotoxicity via upregulation of renal SIRT3 and PAX2. Life Sci. 2024;336:122318. https://doi.org/10.1016/j.lfs.2023.122318
CrossRef - Gumbar S, Bhardwaj S, Mehan S, et al. Renal mitochondrial restoration by gymnemic acid in gentamicin-mediated experimental nephrotoxicity: evidence from serum, kidney and histopathological alterations. Front Pharmacol. 2023;14:1218506. https://doi.org/10.3389/fphar.2023.1218506
CrossRef - Stabrauskiene J, Kopustinskiene DM, Lazauskas R, Bernatoniene J. Naringin and naringenin: Their mechanisms of action and the potential anticancer activities. Biomedicines. 2022;10:1686. https://doi.org/10.3390/biomedicines10071686
CrossRef - Bwambale W, Oka VO, Onaadepo O, et al. Advancing nephroprotective strategies: the role of naringenin in preventing gentamicin-induced nephrotoxicity. Discov Med. 2025;2:115. https://doi.org/10.1007/s44337-025-00315-8
CrossRef - Insuan O, Janchai P, Thongchuai B,et al. Anti-inflammatory effect of pineapple rhizome bromelain through downregulation of the NF-κB- and MAPKs-signaling pathways in lipopolysaccharide (LPS)-stimulated RAW264.7 cells. Curr Issues Mol Biol. 2021;43(1):93-106. https://doi.org/10.3390/cimb43010008
CrossRef - Flores-Peña R, Monroy-Ramirez HC, Caloca-Camarena F, et al. Naringin and naringenin in liver health: A review of molecular and epigenetic mechanisms and emerging therapeutic strategies. Antioxidants. 2025;14(8):979. https://doi.org/10.3390/antiox14080979
CrossRef - Varilla C, Marcone M, Paiva L, Baptista J. Bromelain, a group of pineapple proteolytic complex enzymes (Ananas comosus) and their possible therapeutic and clinical effects: a summary. Foods (Basel). 2021;10(10):2249. https://doi.org/10.3390/foods10102249
CrossRef - Gusti AMT, Qusti SY, Alshammari EM, Toraih EA, Fawzy MS. Antioxidants-related superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPX), glutathione-S-transferase (GST), and nitric oxide synthase (NOS) gene variants analysis in an obese population: a preliminary case-control study. Antioxidants (Basel). 2021;10(4):595. https://doi.org/10.3390/antiox10040595
CrossRef
Abbreviations List
LPO: Lipid peroxidation
NO: Nitric oxide
GSH: Reduced glutathione
SOD: Superoxide glutathione
CAT: Catalase
NAC: N-acetyl cysteine
TNF-α: Tumor necrosis factor-alpha
IL-6: Interleukin-6
Nrf2: Nuclear factor erythroid 2–related factor 2
NF-κB: Nuclear factor kappa B
TGF-β1: Transforming growth factor-beta 1
i.p.: Intra-peritoneal
p.o.: Per oral
NAD: No abnormality detected
H&E: Hematoxylin and eosin
SEM: Standard error of the mean
ANOVA: Analysis of variance







