Manuscript accepted on :08-09-2025
Published online on: 22-09-2025
Plagiarism Check: Yes
Reviewed by: Dr Dunya Abdal-Malik and Dr. Ramesh Gupta
Second Review by: Dr. Mu, Tianhong
Final Approval by: Dr. Prabhishek Singh
Department of Oral and Maxillofacial Surgery and Diagnostic Sciences, College of Dentistry, Jazan University – Saudi Arabia.
Corresponding Author E-mail:dr.alqahtani44@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3218
Abstract
Oral mucosal diseases such as lichen planus, aphthous ulcers, and mucositis are chronic conditions that markedly impair quality of life and often require long-term treatment. Conventional systemic therapies frequently lead to poor targeting and adverse effects, while standard topical formulations face rapid clearance, poor retention, and low patient adherence. To overcome these limitations, research has focused on key strategies such as site-specific mucoadhesion, stimuli-responsive (‘intelligent’) release, and advanced carrier engineering. Mucoadhesive patches, films, and nanoparticles improve drug residence time and precision targeting. Thermo-responsive hydrogels and stimuli-sensitive systems enable sustained release in response to temperature, pH, or enzymatic changes, aligning with real-time disease activity. Advanced carriers like liposomes and lipid-based nanoparticles protect labile drugs and improve mucosal penetration. Innovations such as 3D printing and biosensor-integrated smart patches further personalize oral drug delivery, enhancing both efficacy and patient compliance. This review highlights the current advances in topical drug-delivery systems for oral mucosal diseases and illustrates how biological barriers, materials science, and digital-health technologies intersect. The aim of this comprehensive review is also to discuss emerging platforms and to identify translational gaps that will guide future precision-based mucosal therapeutics.
Keywords
Bioadhesives; Drug Carriers; Gels; Hydrogels; (MDDS) Mucoadhesive Drug Delivery Systems; Nanoparticles; Patches; Thermosensitive Hydrogels
Download this article as:| Copy the following to cite this article: Alqahtani A. S. Topical Drug Delivery in Oral Mucosal Diseases: Challenges, Carriers, and Innovations: A Comprehensive Review. Biomed Pharmacol J 2025;18(3). |
| Copy the following to cite this URL: Alqahtani A. S. Topical Drug Delivery in Oral Mucosal Diseases: Challenges, Carriers, and Innovations: A Comprehensive Review. Biomed Pharmacol J 2025;18(3). Available from: https://bit.ly/3Iwx5mu |
Introduction
Oral mucosal diseases such as recurrent aphthous stomatitis (RAS), oral lichen planus (OLP), candidiasis, oral mucositis, pemphigus vulgaris (PV), leucoplakia, and others often cause pain, ulcers, redness, inflammation, and various functional restrictions (e.g., difficulty speaking or chewing), severely diminishing quality of life. Their etiologies range from immune dysregulation and infections to idiopathic and treatment-related factors. Many of these follow a chronic, relapsing course that requires ongoing management.1, 2
Moderate to severe cases often require systemic therapy with corticosteroids, antifungals, immunosuppressants, or immunomodulators.1, 3, 4 However, oral administration subjects drugs to first-pass hepatic metabolism and gut interactions, yielding low lesion concentrations.3, 5-7 Long-term use can have systemic side effects such as hepatotoxicity, immunosuppression, and endocrine disturbances.4, 8, 9 Injectable or IV formulations act rapidly but are invasive and impractical.6
Topical therapies (gels, pastes, sprays, patches, films) deliver drugs directly to lesions, enhancing local bioavailability and minimizing systemic exposure.2, 10, 11 The non-keratinized oral mucosa, with its rich vascularity and high permeability, favors rapid drug uptake.7, 10-12 Yet rapid salivary clearance, epithelial turnover, enzymatic degradation, and constant mucosal movement limit retention, while poor adhesion, unpleasant taste or odor, and solubility issues undermine patient compliance.10, 13-15
Researchers are developing advanced topical carriers tailored to the oral mucosa to address these barriers. Mucoadhesive polymers, lipid-based nanoparticles, in situ gels, layered films, and bioinspired materials aim to improve drug retention, absorption, and sustained release in oral mucosa.5, 13, 16, 17 Mucoadhesive patches and films prolong the retention time of the drugs at the site of application and reduce the frequency of drug administration.18 Nanocarriers like liposomes and lipid-based particles enhance drug absorption and protect sensitive labile therapeutic agents from enzymatic degradation.8, 19-21 Natural adhesive-based materials, including mussel protein, demonstrate strong mucoadhesion and serve to be promising agents for stable drug retention in the dynamic oral milieu.4
Recent technologies are transforming oral drug delivery through innovations like 3D-printed dosage forms, sensor-integrated smart patches, and personalized delivery systems. These tools enable precise dosing, real-time therapeutic monitoring, and seamless integration with digital health platforms.22, 23 These approaches align with precision-medicine principles and are customized to the oral cavity’s unique anatomy and physiology. While systemic drugs retain value, their non-specific distribution and systemic side effects underscore the need for such targeted topical systems. These are customized to the unique anatomical and physiological environment of the oral cavity. This review aims to evaluate advanced topical drug delivery systems, such as mucoadhesive patches, nanocarriers, and smart technologies, for their ability to overcome oral mucosal barriers, improve patient compliance, and address translational challenges to enhance clinical outcomes in chronic oral mucosal diseases. Continued research into these advanced carriers and smart technologies is essential to bridge translational gaps and improve clinical outcomes in oral mucosal disease management.24-28
Challenges in Oral Topical Drug Delivery
Topical oral drug delivery encounters barriers such as salivary washout, enzymatic degradation, and patient variability, prompting the use of mucoadhesive systems, enzyme inhibitors, and novel biomaterials for improved efficacy.
Salivary washout and enzymatic degradation
Continuous salivary secretion (0.5–1.5 L/day) rapidly clears topical drugs, reducing residence time and therapeutic efficacy in the oral mucosa.1,2,6 Saliva and mucosal tissues contain proteases, esterases, and amylases that degrade peptide-based and labile drugs, particularly large biopharmaceuticals.12,15 Protein-based agents may lose up to 90% of their activity within 30 minutes of exposure, underscoring their instability.9 Mucoadhesive polymers such as chitosan and carbopol increase contact time but show inconsistent adhesion influenced by patient-specific factors.17, 29 Nanocarriers including liposomes and solid lipid nanoparticles protect drugs from enzymatic degradation and improve mucosal penetration, achieving up to 3.2-fold higher delivery with more than 80% protection.20, 30 Stimuli-responsive hydrogels further enhance targeting by releasing drugs in response to physiological triggers. For example, pH-sensitive nanospheres released 80% of metronidazole at pH 6.5 compared with less than 50% at pH 7.4.31 Despite these advances, clinical translation remains limited by formulation instability, scalability issues, and insufficient long-term safety data.32
Mucosal permeability limitations
The oral mucosa shows region-specific permeability, with non-keratinized sites such as the buccal, sublingual, and ventral tongue mucosa being optimal for drug delivery because of their thin epithelium (100–200 µm), rich vascularity, and higher permeability. These features enable rapid absorption and support both local and systemic delivery.1, 7, 13 The stratified squamous epithelium (15–30 layers), combined with a dense mucin coat (50–500 µm) and tight intercellular junctions, forms a strong barrier for hydrophilic molecules and macromolecules like peptides, proteins, and nucleic acids, which often show absorption rates below 5–10%.11, 33 Keratinized regions such as the gingiva and hard palate provide mechanical resilience but demonstrate 2- to 5-fold lower permeability due to thicker epithelium (up to 500 µm) and compact lipid architecture.13 This heterogeneity necessitates careful site selection and rational design of delivery systems. Advanced strategies including penetration enhancers, chitosan-modified nanoparticles, and muco-penetrating carriers transiently loosen tight junctions or bypass mucus, markedly improving drug flux and therapeutic efficacy.12, 19
Anatomical and functional constraints
The oral cavity’s dynamic activity significantly limits topical drug retention. Swallowing occurs 0.5–2 times per minute, while mastication exerts forces of 20–30 N, quickly displacing non-adherent formulations.2, 12 Chewing and speaking reduce the residence time of conventional gels and liquids to less than 5–10 minutes, restricting absorption.6, 34 Rapid epithelial turnover every 5–7 days further decreases drug contact duration and promotes uneven distribution.1, 11 Tongue mobility and continuous salivary flow compound detachment, especially in non-keratinized, high-mobility sites. Non-mucoadhesive systems retain less than 15% of drug content after one hour, emphasizing the inadequacy of conventional formulations.8, 13 Mucoadhesive patches and films provide marked improvement, with feasibility studies reporting retention beyond 100 minutes in vivo. These systems demonstrate clear superiority by maintaining prolonged contact despite anatomical movements and functional disruptions, offering a more reliable approach for sustained topical drug delivery.34, 35
Patient-centric barriers
Patient-related factors strongly influence the success of topical oral drug delivery. Unpleasant taste, mucosal irritation, and frequent dosing schedules markedly reduce adherence, with up to 60% of patients discontinuing therapy within the first week due to discomfort or inconvenience.1, 11 Elderly patients and those with orofacial pain often struggle with precise application because of reduced manual dexterity, which compromises dosing accuracy.18 Xerostomia and Sjögren’s syndrome, affecting 10–15% of older adults, further impair drug dissolution and diminish the effectiveness of mucoadhesive systems that depend on adequate salivary moisture for activation.12, 24 These limitations highlight the urgent need for taste-masked, once-daily, and moisture-independent delivery platforms to enhance compliance and optimize outcomes in chronic oral mucosal diseases.
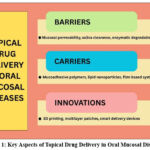 |
Figure 1: Key Aspects of Topical Drug Delivery in Oral Mucosal DiseasesClick here to view Figure |
Table 1: Translational Challenges and Strategies for Enhancing Topical Drug Delivery in Oral Mucosa
| Barrier/Challenge | Underlying Mechanism | Impact on Drug Delivery | Mitigation Strategies | Limitations | Recommendations | References |
| Saliva flow and clearance | Continuous production and flow of saliva dilutes and removes drug formulations | Reduced residence time and bioavailability of the drug | Mucoadhesive polymers (e.g., chitosan, carbopol), buccal patches | Inconsistent adhesion, discomfort, and limited retention time | Smart mucoadhesive nanoparticles, multilayer films, mussel-inspired adhesives | 2, 5, 36, 37 |
| Permeability of oral mucosa | Stratified squamous epithelium, tight junctions limit drug penetration | Poor systemic and local absorption | Use of penetration enhancers, prodrugs, and nanocarriers | Potential for irritation, toxicity, and variable absorption | Muco-penetrating and mucus-inert nanoparticles, enzyme-sensitive carriers | 2, 11, 13, 19, 20 |
| Enzymatic degradation | Presence of salivary enzymes (e.g., proteases, amylases) | Premature degradation of sensitive drugs, especially peptides/proteins | Enzyme inhibitors, encapsulation in protective vehicles | Partial protection, added complexity | Enzyme-resistant formulations, biodegradable nanogels, and liposomes | 6, 14, 20, 22 |
| Mucosal irritation/sensitivity | High vascularity and sensory innervation of oral tissues | Discomfort, inflammation, and poor patient compliance | Use of biocompatible carriers and natural polymers | Still risk of irritation with some chemical enhancers | Biopolymers (e.g., hyaluronic acid), propolis-loaded nanofibers | 11, 18, 21 |
| Inconsistent patient compliance | Difficulty in retaining formulations in situ | Ineffective dosing, reduced treatment adherence | Fast-dissolving films, bioadhesive tablets | May fall off or dissolve too quickly | 3D-printed patches, wearable intraoral devices, personalized films | 21, 25, 27, 37, 38 |
| Disease-related changes | Lesions, ulcers, and inflammation alter mucosal barrier function | Variable drug absorption and local effects | Localized application of corticosteroids, mouth rinses | Lack of site-specific delivery and targeting | Targeted nanoparticle therapy, stimuli-responsive delivery systems | 4, 8, 20, 39 |
| Formulation stability | Moist and enzymatically active environment leads to instability | Decreased drug efficacy and shelf life | Use of stabilizers and encapsulation technologies | Limited long-term stability, regulatory hurdles | In situ gelling systems, lyophilized films, spray-dried nanocarriers | 9, 15, 40, 41 |
| Limited dosage form variety | Fewer options compared to systemic routes | Limits drug types/formulations for oral diseases | Patches, gels, films, sprays | Limited patient-specific tailoring | Smart wearable patches, digital integration for dose control | 26, 27, 31, 37, 38 |
Note: LNs: Solid lipid nanoparticles; PEG: Polyethylene glycol; PEO–PPO–PEO: Polyethylene oxide–polypropylene oxide–polyethylene oxide; PNIPAAm: Poly(N-isopropylacrylamide); 3D: Three-dimensional; DNA: Deoxyribonucleic acid; RNA: Ribonucleic acid; HA: Hyaluronic acid; EPO: Eudragit polymer; PLA: Polylactic acid; PLGA: Poly (lactic-co-glycolic acid); ROS: Reactive oxygen species.
Conventional Drug Delivery Systems in Oral Mucosal Therapy
Clinicians continue to depend on traditional topical treatments such as gels, rinses, and sprays because they apply them easily, access them readily, and use them cost-effectively to manage localized or short-term oral mucosal diseases.
Gels and semi-solid formulations
Gels serve as common formulations in oral mucosal therapy, where viscosity supports mucoadhesion and enables localized delivery of drugs such as 0.1% triamcinolone acetonide (Orabase®), miconazole, clotrimazole, and benzocaine for oral lichen planus, candidiasis, and ulcers.1, 11 Conventional gels provide short residence times of only 30–60 minutes because salivary washout (0.5–1.5 L/day) and oral movements during speech or swallowing displace them rapidly.2, 12 Epithelial turnover every 5–7 days further limits contact duration.33 These factors require reapplication three to four times daily, lowering patient adherence.6 Acceptance also remains poor, with up to 60% of patients especially children and elderly individuals reporting issues such as unpleasant taste, stickiness, or burning sensation.1, 38 Mucoadhesive polymers like chitosan and carbopol can extend retention, yet variable salivary pH and mucosal dynamics restrict consistent performance.14 Advanced systems with sustained release and improved tolerability are needed to overcome these drawbacks.
Mouth rinses and liquid solutions
Oral rinses ensure uniform drug distribution across widespread lesions and are useful in conditions such as radiation-induced mucositis and erosive oral lichen planus. Agents like chlorhexidine (0.12–0.2%), budesonide, and tacrolimus provide antimicrobial, anti-inflammatory, and immunomodulatory effects.1, 4 Their clinical impact remains limited because salivary clearance reduces mucosal contact to less than 5 minutes, severely lowering bioavailability.2, 12 Alcohol-based formulations often cause burning and aggravate irritation in ulcerated tissues.11 Accidental swallowing of steroid rinses such as budesonide can result in systemic exposure, raising risks of adrenal suppression or oral candidiasis.6 Rinses offer convenience but their drawbacks highlight the need for mucoadhesive or sustained-release formulations that prolong retention and lower safety risks during long-term therapy.
Topical sprays
Oral sprays provide localized and precise drug delivery with minimal tissue contact, making them useful for painful conditions such as erosive lichen planus and oral mucositis, particularly after surgery.4, 6 Agents like clobetasol and benzydamine hydrochloride act rapidly, and some sprays include mucoadhesive polymers such as carbopol to prolong retention.11 Delivery efficiency remains limited, as only 40–60% of the sprayed dose reaches posterior regions like the oropharynx or retromolar trigone, while user variability causes dosing inconsistencies of up to 30%.2, 12 Challenges include salivary washout (0.5–1.5 L/day), enzymatic degradation, and low permeability of hydrophilic or large-molecule drugs.1, 7 Xerostomia in nearly 30% of elderly adults, reduced manual dexterity, and taste-related non-adherence with discontinuation rates up to 60% limit treatment success.1, 18 These drawbacks restrict conventional sprays to transient relief and emphasize the need for advanced mucoadhesive, nano-enabled, and sustained-release systems for chronic oral diseases.8, 17
Mucoadhesive Drug Delivery Systems (Mdds) in Oral Therapy
MDDS are an innovative approach to oral therapeutic agents. They apply natural adhesion between polymers and mucosal surfaces, thus permitting drugs to act at the target site for a prolonged period.
Mechanism of mucoadhesion
Mucoadhesive drug carriers attach to the oral mucosa through a two-step mechanism. In the contact phase, the dosage form hydrates and swells in saliva, allowing close interaction with the mucosal surface. In the consolidation phase, polymer chains such as chitosan or carbopol interpenetrate with mucin glycoproteins and form intermolecular bonds through hydrogen bonding, van der Waals forces, and electrostatic interactions, particularly between cationic polymers and negatively charged sialic acid residues.16, 29 Strong adhesion is difficult to maintain because salivary flow (0.5–1.5 L/day) can wash away the system before consolidation, while proteases and esterases degrade polymer–mucin complexes.1, 2 Rapid epithelial turnover every 5–7 days and tissue changes in inflamed or ulcerated regions further weaken adhesion, limiting effective retention to 2–4 hours in vivo.11, 17 Advanced polymers and bioinspired designs such as catechol-functionalized systems are being developed to strengthen wet adhesion and extend durability in the dynamic oral environment To overcome these challenges.37
Polymers used in mucoadhesive systems
Various types of natural and synthetic polymers reinforce the design of MDDS due to their favorable physicochemical properties and biocompatibility. Chitosan, a cationic polysaccharide, binds strongly to mucins through electrostatic interactions, increasing retention time on the oral mucosa.17, 29 It also provides antimicrobial and hemostatic benefits, making it suitable for treating infected oral ulcers.13 Carbopol, a high-molecular-weight polyacrylic acid, adheres to mucin via hydrogen bonding and swells extensively, supporting sustained drug release and retention of up to 8–12 hours in optimized systems.11, 16 Hydroxypropyl methylcellulose (HPMC), a non-ionic cellulose ether, is valued for its film-forming ability, safety, and adjustable release profile. Modulating viscosity grades allows precise control of drug release for tailored therapeutic effects.21, 38 These polymers form the backbone of modern MDDS that provides stability, prolonged residence, and flexibility for effective oral mucosal drug delivery.
Innovations in mucoadhesive formulations
Recent innovations in MDDS are advancing precision in oral therapeutics. pH-responsive carriers release drugs selectively in acidic microenvironments of inflamed or ulcerated tissues, improving local efficacy and reducing systemic exposure.17, 40 Multilayered films introduce structural sophistication, combining an adhesive base for retention, a protective backing to prevent drug loss, and a reservoir enabling controlled release, as shown in bilayer patches for oral mucositis.21, 36 Bioinspired catechol-functionalized polymers, such as polydopamine-based materials, replicate mussel adhesion, producing strong yet reversible bonding under wet conditions without mucosal injury.37 Smart MDDS integrate microneedles, biosensors, and wireless modules to track biomarkers and deliver feedback-based dosing.25, 27 They extend mucosal residence, improve adherence, and enable personalized drug delivery in oral diseases.
Nanocarrier-Based Systems for Oral Mucosal Drug Delivery
Nanocarrier-based systems (liposomes and solid lipid nanoparticles), improve drug stability, mucosal adhesion, and controlled release, thus enhancing therapeutic efficacy in various oral mucosal lesions.
Nanocarrier-based drug delivery systems
Nanocarrier-based drug delivery systems, including solid lipid nanoparticles (SLNs) and liposomes, encapsulate both hydrophilic and lipophilic drugs. They shield therapeutic agents from enzymatic degradation in the oral cavity and improve transmucosal delivery.23, 30, 42 Their nanometric size and mucoadhesive surface modifications, such as chitosan coating, increase mucosal retention and enhance permeability through paracellular pathways.19, 41, 42 These carriers release drugs in a sustained and controlled manner through diffusion or stimuli-responsive mechanisms, which lowers dosing frequency and raises therapeutic efficiency.20, 43 Nanocarriers increase bioavailability and limit systemic exposure by protecting labile drugs and directing them to target sites. In oral mucosal diseases including oral lichen planus, candidiasis, and mucositis, these systems show strong potential to improve patient compliance and treatment outcomes with localized, long-acting, and biocompatible formulations.20, 30, 42, 43
Liposomes: structure and applications in oral mucosal therapy
Liposomes consist of phospholipid bilayers that encapsulate hydrophilic drugs in the aqueous core and lipophilic agents within the lipid bilayer, allowing dual drug loading and targeted delivery to ulcerated oral mucosa.20, 41 Their biocompatibility, membrane fluidity, and ability to fuse with epithelial cells enhance drug transport through membrane fusion or endocytosis, supporting intracellular delivery of corticosteroids, antifungals, and anticancer agents.14, 20, 23. Mucoadhesive surface modifications, such as chitosan coating, increase mucosal adhesion and extend residence time. Chitosan-coated liposomes, for example, achieved a 3.2-fold rise in buccal permeability and sustained drug release for 12 hours in ex vivo models.20 In clinical studies, liposomal triamcinolone acetonide reduced pain and lesion size in erosive oral lichen planus (OLP) by 50% compared with conventional formulations, while lowering systemic absorption by 70%.35 Liposomal nystatin maintained prolonged antifungal activity and reduced fungal load by 80% in oral candidiasis models through improved mucosal penetration and retention.20 These outcomes position liposomes as effective nanocarriers for localized, sustained, and biocompatible therapy in oral mucosal diseases.
Solid lipid nanoparticles (SLNs)
Solid lipid nanoparticles (SLNs) are nanocarriers made from biocompatible lipids that remain solid at both room and body temperature, such as stearic acid and glyceryl monostearate. Their solid lipid core provides excellent physical stability and allows efficient encapsulation of both hydrophilic and lipophilic drugs, making them well suited for oral mucosal delivery.42, 44 With particle sizes typically ranging from 50 to 300 nm, SLNs can be surface-modified with mucoadhesive polymers like chitosan to improve adhesion and extend residence time in the oral cavity.14, 44 The lipid matrix enables controlled release for 8–24 hours, improves local targeting, and minimizes systemic absorption.13, 22 By shielding drugs from salivary proteases and esterases, SLNs enhance stability and bioavailability.9 Preclinical models show corticosteroid-loaded SLNs maintain prolonged anti-inflammatory activity, while antifungal-loaded SLNs improve mucosal retention and therapeutic outcomes in oral candidiasis.14, 30
Emerging hybrid nanocarrier systems
Hybrid nanocarriers, integrating liposomes and SLNs, enhance drug release profiles and improve the stability of encapsulated bioactive agents.13, 23, 42 Nanostructured lipid carriers (NLCs), incorporating both solid and liquid lipids, exhibit superior drug-loading capacity and reduced expulsion compared to SLNs.21, 44 NLCs delivering anti-inflammatory cytokines and antioxidants significantly decreased ulceration severity and accelerated epithelial recovery in oral mucositis models.21, 44 These carriers are greatly combined with mucoadhesive gels, films, and patches to improve mucosal adhesion, easy application, and better patient adherence.20, 36, 38 These systems protect labile drugs, enable site-specific targeting, and control release, improving outcomes in OLP, candidiasis, and mucositis. Emerging technologies like 3D printing and smart biomaterials support personalized, responsive, and precision-based oral drug delivery.13, 20, 23, 25, 42 Table 2 shows preclinical studies on advanced mucoadhesive and smart drug delivery systems for oral and periodontal diseases.
 |
Table 2: Preclinical Studies on Advanced Mucoadhesive and Smart Drug Delivery Systems for Oral and Periodontal DiseasesClick here to view Table |
Thermo-Responsive Gelation: Principles and Polymer Systems
Thermo-responsive hydrogels utilize polymers that shift from liquid to gel with temperature changes. Poloxamer 407 (Pluronic F127), a PEO–PPO–PEO triblock copolymer, remains liquid at room temperature and gels near body temperature (~30–37°C), aiding oral drug delivery.32, 40, 44 This occurs through micellization, where PPO blocks clump together and PEO blocks keep the gel stable in water.40 The gel forms a 3D network that encapsulates the drug, thus facilitating sustained release. Thermosensitive, mucoadhesive materials like methylcellulose, chitosan, and PNIPAAm enhance oral mucosal retention.3, 5, 44
Novel systems respond to specific activators like pH, enzymes, or oxidative stress to release drugs when required.19, 32, 40 Local alterations in pH and enzymatic activity during oral diseases activate thermo-responsive gels, thereby facilitating their release of drugs, particularly at lesional sites while reducing systemic exposure.19, 23 Mucoadhesive polymers like Carbopol, Eudragit, and chitosan prolong the mucosal contact by interacting with mucin thus improving drug retention and therapeutic efficacy at inflamed oral mucosa.16, 17 Figure 2 illustrates thermo-responsive hydrophilic polymers undergoing LCST-induced sol–gel transition, forming biocompatible scaffolds for targeted oral mucosal drug delivery.
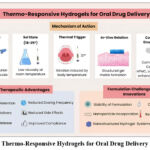 |
Figure 2: Thermo-Responsive Hydrogels for Oral Drug Delivery SystemsClick here to view Figure |
Therapeutic Advantages and Clinical Potential
Prolonged mucosal retention and sustained release
Thermo-responsive gels undergo sol-to-gel transition at body temperature (~30–37°C), forming a semi-solid depot that adheres to the oral mucosa and releases drugs for 24–72 hours.29, 30 This localized system raises drug concentration at the lesion site while lowering systemic exposure. A poloxamer-based hydrogel loaded with dexamethasone increased bioavailability 3.5-fold and extended release to 24 hours, with a half-life of 20.7 hours in rat models.37 In beagle dogs, a thermo-reversible hydrogel containing doxycycline and lipoxin A₄ maintained stable drug levels in periodontal pockets for 4 days, releasing 70% within 24 hours.43 Such extended retention reduces dosing frequency from three to four times daily to once daily, which improves compliance in chronic conditions like oral lichen planus and mucositis.4, 18, 22
Dual or synergistic drug delivery
Thermo-responsive gels co-deliver hydrophilic and hydrophobic drugs, facilitating synergistic treatment of complex oral diseases. A chitosan-modified poloxamer hydrogel encapsulated clotrimazole (antifungal, 85% efficiency) and triamcinolone acetonide (corticosteroid, 78% efficiency), providing dual anti-inflammatory and antifungal activity for 48 hours.22 This design benefits candidiasis-associated OLP, where infection and inflammation overlap. In preclinical studies, these systems reduced fungal load by 80% and lowered inflammatory cytokines (TNF-α, IL-6) by 60–70% compared to monotherapy.20 Delivering multiple agents through one system enhances efficacy, limits resistance, and simplifies treatment, positioning thermo-responsive gels as a strong platform for mixed oral pathologies.4, 22, 29
Enhanced delivery to posterior and complex anatomical sites
Thermo-responsive gels improve drug delivery to anatomically challenging regions such as the oropharynx, retromolar trigone, and tonsillar pillars, where conventional sprays and gels show poor retention due to gravitational drainage.29, 30 Their low viscosity at room temperature allows application by syringe, followed by gelation at 37°C, ensuring coverage of irregular mucosal surfaces.43 In a study, a thermosensitive hydrogel applied to sublingual mucosa achieved 90% drug coverage in ulcerated areas, compared with 40% using standard gels.34 This property proves useful in radiation-induced mucositis, where lesions are extensive and deep. By adhering and releasing drugs uniformly for 5–7 days, these gels support healing and pain reduction in high-burden oral lesions.31
Formulation Challenges and Innovations in Thermo-Responsive Gels
Thermo-responsive gels show strong clinical potential but face key formulation challenges, including storage instability, premature drug degradation, and batch variability. Poloxamer-based systems such as Pluronic F127 often undergo syneresis (water expulsion) and phase separation during storage at temperatures above 4°C, which disrupts gelation behavior and shortens shelf life.12 Encapsulated drugs, particularly biologics like peptides and proteins, remain vulnerable to hydrolytic and oxidative degradation in aqueous environments. Reports indicate up to 30% loss of drug activity within two weeks under ambient conditions.32 Several strategies address these limitations. Lyophilized precursors of thermosensitive gels provide higher stability and can be stored as powders for later reconstitution into functional gels. A lyophilized poloxamer-chitosan system retained 95% of drug content after six months at 25°C compared with 70% in liquid formulations.12 Hybrid designs further improve performance by embedding nanocarriers such as SLNs or liposomes within the hydrogel matrix. These systems achieve multi-phase release with an initial burst followed by sustained delivery for 24–72 hours. A curcumin-SLN-loaded thermo-gel reached 88% encapsulation efficiency, maintained 70% drug release over 48 hours, and improved bioavailability 3.1-fold in murine models.22 Such modular approaches allow adjustment of drug release to disease severity and improve reproducibility, stability, and pharmacokinetic control, supporting translation into scalable, clinically viable formulations.
Emerging Applications of Thermo-Responsive Gels in Advanced Therapeutics
Integration of thermo-responsive gels into next-generation platforms expands their role in precision oral therapy. Applications include 3D-printed scaffolds, personalized mucoadhesive patches, and biosensor-based smart systems that adjust drug delivery to local physiological changes. 3D-printed poloxamer scaffolds fabricated with CAD-CAM imaging of patient lesions demonstrated sol–gel transition near 32°C and provided unidirectional release, achieving 90% drug localization at ulcerated sites in radiation-induced mucositis.25 Biosensor-integrated hydrogels detect pH, temperature, or inflammatory biomarkers such as IL-6 and TNF-α to trigger controlled drug release. A digital health-integrated patch that combined a thermosensitive gel with wireless biosensors enabled remote monitoring and app-controlled corticosteroid delivery in erosive oral lichen planus, reducing flare-ups by 60% over eight weeks compared with standard treatment.28 Thermo-responsive gels also serve as carriers for gene and immunotherapy. A PLGA nanoparticle-loaded gel that delivered siRNA against TNF-α in a murine oral inflammation model achieved 75% gene silencing and marked ulcer reduction.23 Another system carrying immune checkpoint inhibitors for oral squamous cell carcinoma produced a 50% reduction in tumor volume over 14 days.41 By combining mucoadhesion, sol–gel transition, and sustained release, thermo-responsive gels provide a foundation for programmable and patient-specific oral therapeutics.
Mucoadhesive Films and Patches: Form and Function
Fabrication and Structural Design
Mucoadhesive films and patches offer design versatility and enhanced patient satisfaction. Techniques like solvent casting, electrospinning, and 3D printing facilitate accurate modulation of film thickness, porosity, and drug distribution to achieve customized release kinetics.21, 27, 36 Most of these systems comprise a bioadhesive layer for secure mucosal attachment, a drug-loaded reservoir, and an impermeable backing layer to direct unidirectional release and prevent detachment. Intraoral imaging facilitates 3D-printed patch fabrication customized for anatomically complex sites hence improving retention and treatment efficiency.25, 36
Therapeutic and mechanical benefits
These systems ensure prolonged mucosal adhesion and controlled drug release, and enhance local bioavailability while reducing systemic absorption. Their optimized tensile strength and elasticity support retention during mastication, speech, and salivation.2, 11, 13, 17, 36, 37
Patient comfort and future directions
Patients, particularly in pediatric and geriatric populations, benefit from the ease of clean, non-invasive application and discreet use of mucoadhesive systems, which significantly enhances treatment adherence and overall therapeutic outcomes.17, 21, 38 Future strategies incorporating biosensitive polymers, 3D customization, and mobile-based digital monitoring position MDDS as a transformative approach in personalized oral therapeutics.17, 25, 37
Future Innovations and Research Directions
Bio-responsive drug delivery systems
Bio-responsive drug delivery systems enhance therapeutic precision by responding to internal cues like pH variations, enzymatic activity, or inflammatory cytokines. pH-sensitive hydrogels selectively release corticosteroids or antimicrobials in acidic mucosal sites, minimizing systemic exposure and drug loss.40 Enzyme-sensitive nanocarriers respond to increased levels of proteases and esterases in diseased tissues to activate site-specific and temporally controlled drug release, thus enhancing therapeutic efficacy. Cytokine-responsive platforms utilize receptors or ligands attuned to inflammatory biomarkers like TNF-α and IL-1β, enabling flare-specific drug release in conditions such as OLP or autoimmune ulcerations, thereby minimizing systemic exposure and enhancing therapeutic precision.17, 19, 28, 44
3D-printed oral delivery devices
3D printing has revolutionized personalized oral drug delivery by producing anatomically customized dosage forms. Innovations like multilayer mucoadhesive films, customized tablets, and site-specific patches with biocompatible polymers has facilitated accurate control over drug load, release kinetics, and structure.26, 36 CAD-CAM mapping improves lesion targeting, while 3D-printed dexamethasone or lidocaine patches enhance adhesion and sustained drug release. These systems manage complex processes effectively by incorporating multiple drugs and bio-responsive features with spatial and temporal accuracy.9, 15, 25
Smart patches with app integration and remote monitoring
Smart patches enhance oral drug delivery by integrating controlled release mechanisms with real-time biosensing, enabling site-specific therapy. They adhere to moist mucosa and respond dynamically to local pH shifts or cytokine surges for precision treatment.27, 28, 37 Clinical assessments validate their efficacy in delivering biologics during mucosal healing post-radiotherapy or chemotherapy. Integration with mHealth tools improves adherence monitoring, automated reminders, and individualized management of chronic oral mucosal disorders.27
Personalized medicine and digital health integration
Personalized advanced oral therapies are driven by genetic, immunological, and environmental variability. Pharmacogenomics, salivary proteomics, and microbiome profiling provide accurate sustenance to patient stratification for personalized therapy. Digital tools capture real-time data, while AI integrates wearables, reports, and electronic health records to predict disease exacerbations and enable app-controlled, cytokine-triggered corticosteroid release in OLP cases.22, 27, 28
Conclusion
Thermo-responsive gels, mucoadhesive patches, and bioresponsive delivery systems provide targeted, sustained, and patient-centered therapy for oral mucosal disorders. “Advances in polymer science, nanotechnology, and digital health directly overcome barriers such as salivary washout, enzymatic degradation, and limited mucosal retention. Preclinical studies show that 3D-printed scaffolds, stimuli-responsive hydrogels, and biosensor-driven devices enable controlled, site-specific, and on-demand drug release, improving efficacy in conditions like oral lichen planus, mucositis, and recurrent aphthous stomatitis. Clinical translation, however, remains limited. Long-term safety, reproducibility, and large-scale validation in diverse populations are not yet established. Regulatory pathways for combined drug–device systems are unclear, and challenges in manufacturing scalability, cost, and accessibility persist. Moving forward, coordinated efforts among researchers, clinicians, regulators, and industry are required to generate robust clinical evidence and streamline approval. With focused innovation and collaboration, these platforms hold strong potential to advance oral therapeutics by improving adherence, minimizing systemic toxicity, and enhancing quality of life.
Acknowledgment
The author would like to thank Jazan University for granting the opportunity for research work.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) does not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not applicable.
Author Contributions:
The sole author was responsible for the conceptualization, methodology, data collection, analysis, writing, and final approval of the manuscript.
References
- Sankar V, Hearnden V, Hull K, et al. Local drug delivery for oral mucosal diseases: challenges and opportunities. Oral Dis. 2011;17 Suppl 1:73–84. https://doi.org/10.1111/j.1601-0825.2011.01793.x.
CrossRef - Hearnden V, Sankar V, Hull K, et al. New developments and opportunities in oral mucosal drug delivery for local and systemic disease. Drug Deliv. Rev. 2012;64(1):16–28. https://doi.org/10.1016/j.addr.2011.02.008.
CrossRef
- Sosnik A, Augustine R. Challenges in oral drug delivery of antiretrovirals and the innovative strategies to overcome them. Drug Deliv. Rev. 2016;103:105–120. https://doi.org/10.1016/j.addr.2015.12.022.
CrossRef
- Caro V, Sutera FM, Giannola LI. In situ delivery of corticosteroids for treatment of oral diseases. Deliv. 2017;8(10):899–914. https://doi.org/10.4155/tde-2017-0055.
CrossRef
- Remiro PFR, Nagahara MHT, Azoubel RA, et al. Polymeric biomaterials for topical drug delivery in the oral cavity: advances on devices and manufacturing technologies. 2022;15(1):1–15. https://doi.org/10.3390/pharmaceutics15010012.
CrossRef
- Lou J, Duan H, Qin Q, et al. Advances in oral drug delivery systems: challenges and opportunities. 2023;15(2):1–15. https://doi.org/10.3390/pharmaceutics15020484.
CrossRef
- Patel VF, Liu F, Brown MB. Advances in oral transmucosal drug delivery. Control. Release. 2011;153(2):106–116. https://doi.org/10.1016/j.jconrel.2011.01.027.
CrossRef
- Brako F, Boateng J. Transmucosal drug delivery: prospects, challenges, advances, and future directions. Expert Opin. Drug Deliv. 2025;22(4):525–553. https://doi.org/10.1080/17425247.2025.2470224.
CrossRef
- Truong-Le V, Lovalenti PM, Abdul-Fattah AM. Stabilization challenges and formulation strategies associated with oral biologic drug delivery systems. Drug Deliv. Rev. 2015;93:95–108. https://doi.org/10.1016/j.addr.2015.08.001.
CrossRef - Singh Malik D, Mital N, Kaur G. Topical drug delivery systems: a patent review. Expert Opin. Ther. Pat. 2016;26(2):213–228. https://doi.org/10.1517/13543776.2016.1131267.
CrossRef
- Paderni C, Compilato D, Giannola LI, et al. Oral local drug delivery and new perspectives in oral drug formulation. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2012;114(3):e25–34. https://doi.org/10.1016/j.oooo.2012.02.016.
CrossRef
- Nguyen S, Hiorth M. Advanced drug delivery systems for local treatment of the oral cavity. Deliv. 2015;6(5):595–608. https://doi.org/10.4155/tde.15.5.
CrossRef
- Dubashynskaya NV, Petrova VA, Skorik YA. Biopolymer drug delivery systems for oromucosal application: recent trends in pharmaceutical R&D. J. Mol. Sci. 2024;25(10):1–20. https://doi.org/10.3390/ijms25105359.
CrossRef
- Guilherme VA, Ribeiro LNM, Tofoli GR, et al. Current challenges and future of lipid nanoparticles formulations for topical drug application to oral mucosa, skin, and eye. Pharm. Des. 2017;23(47):1–12. https://doi.org/10.2174/1381612823666171122103849.
CrossRef
- Homayun B, Lin X, Choi HJ. Challenges and recent progress in oral drug delivery systems for biopharmaceuticals. 2019;11(3):1–20. https://doi.org/10.3390/pharmaceutics11030129.
CrossRef
- Golshani S, Vatanara A, Amin M. Recent advances in oral mucoadhesive drug delivery. Pharm. Pharm. Sci. 2022; 25:201–217. https://doi.org/10.18433/jpps32705.
CrossRef
- Kumar R, Islam T, Nurunnabi M. Mucoadhesive carriers for oral drug delivery. Control. Release. 2022; 351:504–559. https://doi.org/10.1016/j.jconrel.2022.09.024.
CrossRef
- Hosseinpour-Moghadam R, Mehryab F, Torshabi M, et al. Applications of novel and nanostructured drug delivery systems for the treatment of oral cavity diseases. Ther. 2021;43(12):e377–e402. https://doi.org/10.1016/j.clinthera.2021.10.016.
CrossRef
- Zheng B, Liu D, Qin X, et al. Mucoadhesive-to-mucopenetrating nanoparticles for mucosal drug delivery: a mini review. J. Nanomedicine. 2025; 20:2241–2252. https://doi.org/10.2147/IJN.S505427.
CrossRef
- Rahma MN, Suhandi C, Mohammed AFA, et al. The role and advancement of liposomes for oral diseases therapy. J. Nanomedicine. 2025; 20:1865–1880. https://doi.org/10.2147/IJN.S492353.
CrossRef - Maheen S, Awan MS, Abbas G, et al. Design, statistical optimization, in vitro and biological characterization of Eudragit L100-gelatin based buccoadhesive bilayer patch for managing oral mucositis. J. Biol. Macromol. 2025;308(Pt 2):142070. https://doi.org/10.1016/j.ijbiomac.2025.142070.
CrossRef
- Zhang Y, Jiang R, Lei L, et al. Drug delivery systems for oral disease applications. Appl. Oral Sci. 2022;30: e20210349. https://doi.org/10.1590/1678-7757-2021-0349.
CrossRef
- Huang P, Li W, Guan J, et al. Synthetic vesicle-based drug delivery systems for oral disease therapy: current applications and future directions. Funct. Biomater. 2025;16(1):1–20. https://doi.org/10.3390/jfb16010025.
CrossRef
- Almuqbil RM, Aldhubiab B. Bioadhesive nanoparticles in topical drug delivery: advances, applications, and potential for skin disorder treatments. 2025;17(2):1–20. https://doi.org/10.3390/pharmaceutics17020229.
CrossRef
- Pandey M, Choudhury H, Fern JLC, et al. 3D printing for oral drug delivery: a new tool to customize drug delivery. Drug Deliv. Transl. Res. 2020;10(4):986–1001. https://doi.org/10.1007/s13346-020-00737-0.
CrossRef
- Mancilla-De-la-Cruz J, R-S M. Applications of 3D printing technologies in oral drug delivery. Today Proc. 2022; 70:477–482. https://doi.org/10.1016/j.matpr.2022.09.464.
CrossRef
- Khadka B, Lee B, Kim KT. Drug delivery systems for personal healthcare by smart wearable patch system. 2023;13(6):1–15. https://doi.org/10.3390/biom13060929.
CrossRef
- Raijada D, Wac K, Greisen E, et al. Integration of personalized drug delivery systems into digital health. Drug Deliv. Rev. 2021; 176:113857. https://doi.org/10.1016/j.addr.2021.113857.
CrossRef
- Asane GS, Nirmal SA, Rasal KB, et al. Polymers for mucoadhesive drug delivery system: a current status. Drug Dev. Ind. Pharm. 2008;34(11):1246–1266. https://doi.org/10.1080/03639040802026012.
CrossRef
- Vergara D, Sanhueza C, Mendez S, et al. Evaluation of preclinical efficacy of curcumin-loaded bicosome systems in amelioration of oral mucositis. 2025;17(2):1–15. https://doi.org/10.3390/pharmaceutics17020181.
CrossRef
- Lin JH, Feng F, Yu MC, et al. Modulation of periodontitis progression using pH-responsive nanosphere encapsulating metronidazole or N-phenacylthialzolium bromide. Periodontal Res. 2018;53(1):22–28. https://doi.org/10.1111/jre.12481.
CrossRef
- Ezike TC, Okpala US, Onoja UL, et al. Advances in drug delivery systems, challenges and future directions. 2023;9(6):e17488. https://doi.org/10.1016/j.heliyon.2023.e17488.
CrossRef
- Liang J, Peng X, Zhou X, et al. Emerging applications of drug delivery systems in oral infectious diseases prevention and treatment. 2020;25(3):1–16. https://doi.org/10.3390/molecules25030516.
CrossRef
- Chang PC, Chao YC, Hsiao MH, et al. Inhibition of periodontitis induction using a stimuli-responsive hydrogel carrying naringin. Periodontol. 2017;88(2):190–196. https://doi.org/10.1902/jop.2016.160189.
CrossRef
- Colley HE, Said Z, Santocildes-Romero ME, et al. Pre-clinical evaluation of novel mucoadhesive bilayer patches for local delivery of clobetasol-17-propionate to the oral mucosa. 2018; 178:134–146. https://doi.org/10.1016/j.biomaterials.2018.06.009.
CrossRef
- Shakeri H, Tolabi H, Nazarpak MH, et al. Design and manufacture of propolis loaded multilayer nanofibrous mucoadhesive patch for the treatment of oral mucosal lesions. Drug Deliv. Sci. Technol. 2025;106630. https://doi.org/10.1016/j.jddst.2025.106630.
CrossRef
- Hu S, Pei X, Duan L, et al. Author correction: A mussel-inspired film for adhesion to wet buccal tissue and efficient buccal drug delivery. Commun. 2024;15(1):3713. https://doi.org/10.1038/s41467-024-48275-4.
CrossRef
- Jacob S, Nair AB, Boddu SHS, et al. An updated overview of the emerging role of patch and film-based buccal delivery systems. 2021;13(8):1–20. https://doi.org/10.3390/pharmaceutics13081206.
CrossRef
- Suharyani I, Mohammed FAA, Muchtaridi M, Wathoni N, Abdassah M. Evolution of drug delivery systems for recurrent aphthous stomatitis. Drug Des. Devel. Ther. 2021; 15:4071–4089. https://doi.org/10.2147/DDDT.S328371.
CrossRef
- Vigani B, Rossi S, Sandri G, Bonferoni MC, Caramella CM, Ferrari F. Recent advances in the development of in situ gelling drug delivery systems for non-parenteral administration routes. 2020;12(9):1–18. https://doi.org/10.3390/pharmaceutics12090859.
CrossRef
- Ketabat F, Pundir M, Mohabatpour F, et al. Controlled drug delivery systems for oral cancer treatment—current status and future perspectives. 2019;11(7). https://doi.org/10.3390/pharmaceutics11070302.
CrossRef
- Pridgen EM, Alexis F, Farokhzad OC. Polymeric nanoparticle drug delivery technologies for oral delivery applications. Expert Opin Drug Deliv. 2015;12(9):1459–1473. https://doi.org/10.1517/17425247.2015.1018175.
CrossRef
- Wang B, Booij-Vrieling HE, Bronkhorst EM, et al. Antimicrobial and anti-inflammatory thermo-reversible hydrogel for periodontal delivery. Acta Biomater. 2020; 116:259–267. https://doi.org/10.1016/j.actbio.2020.09.018.
CrossRef
- Sato H, Yamada K, Miyake M, et al. Recent advancements in the development of nanocarriers for mucosal drug delivery systems to control oral absorption. 2023;15(12). https://doi.org/10.3390/pharmaceutics15122708.
CrossRef
- Mou J, Liu Z, Liu J, et al. Hydrogel containing minocycline and zinc oxide-loaded serum albumin nanoparticle for periodontitis application: preparation, characterization and evaluation. Drug Deliv. 2019;26(1):179–187. https://doi.org/10.1080/10717544.2019.1571121.
CrossRef








