Manuscript accepted on :12-09-2025
Published online on: 30-09-2025
Plagiarism Check: Yes
Reviewed by: Dr. Akmal El-Mazny and Dr. Ranjan Kumar Singh
Second Review by: Dr. Rekha Mehani
Final Approval by: Dr. Anton R Kiselev
Anwar Yonis Ibrahim1 and Nadheerah Falih Neamah2*
and Nadheerah Falih Neamah2*
1Department of Clinical Laboratory science department, College of Pharmacy, University of Basrah, Basrah, Iraq.
2Department of Pharmacology and Toxicology, College of Pharmacy, University of Basrah, Basrah, Iraq.
Corresponding Author E-mail:nadheerah.neamah@uobasrah.edu.iq
DOI : https://dx.doi.org/10.13005/bpj/3265
Abstract
Meteorin-like (Metrnl), or Subfatin, is a novel adipokine primarily secreted by adipose tissue and skeletal muscle. It plays a crucial role in energy metabolism, insulin sensitivity, and inflammatory regulation. However, its relationship with obesity and metabolic disturbances remains controversial.This study aimed to evaluate the association between serum Metrnl levels and markers of glucose and lipid metabolism in normal-weight, overweight, and obese individuals. A total of 102 participants (38 normal-weight, 33 overweight, and 31 obese; aged 30–70 years) attending Al-Mawanee General Hospital in Basrah, Iraq, were enrolled in this cross-sectional study (June 2022–December 2023). Anthropometric indices, fasting blood glucose (FBG), insulin, HbA1c, lipid profile, liver enzymes, and Metrnl concentrations (ELISA) were measured. Insulin resistance was assessed by the homeostasis model assessment of insulin resistance (HOMA-IR). Statistical analysis included ANOVA, correlation, and regression models.Serum Metrnl levels were significantly lower in overweight (188.6 ± 18.5 pg/ml) and obese (137.1 ± 15.5 pg/ml) individuals compared with normal-weight participants (229.6 ± 16.5 pg/ml, p<0.001). Circulating Metrnl showed strong negative correlations with BMI, TC, TG, FBG, insulin, and HOMA-IR (all p<0.001), and a positive correlation with HDL-C (r=0.947, p<0.001). Multiple regression analysis confirmed that lipid and glucose homeostasis parameters were independently associated with serum Metrnl concentrations. Based on the present study results, reduced serum Metrnl levels are closely associated with obesity, dyslipidemia, and insulin resistance. These findings suggest that Metrnl may serve as a potential biomarker and therapeutic target for obesity-related metabolic disturbances, particularly atherogenic dyslipidemia. Further longitudinal studies are warranted to clarify the causal role of Metrnl in metabolic disorders.
Keywords
Drug absorption; In vitro study; Intestinal motility; Iworx system; Sodium Copper Chlorophyllin
Download this article as:| Copy the following to cite this article: Ibrahim A. Y, Neamah N. F. Serum Meteorin-Like (Metrnl) as a Potential Biomarker of Obesity-Related Dyslipidemia and Insulin Resistance. Biomed Pharmacol J 2025;18(3). |
| Copy the following to cite this URL: Ibrahim A. Y, Neamah N. F. Serum Meteorin-Like (Metrnl) as a Potential Biomarker of Obesity-Related Dyslipidemia and Insulin Resistance. Biomed Pharmacol J 2025;18(3). |
Introduction
Obesity is a disease characterized by the excessive accumulation of body fat that impairs health. The primary cause of weight gain and obesity is an energy imbalance between calories consumed and calories expended.1-4 Body Mass Index (BMI) is a common screening tool used to estimate body fat based on an individual’s weight relative to their height. Following a kilogram weight measurement, the result is divided by the square of the person’s height in meters (kg/m2). A high resultant may suggest high body fat.5
The state of Weight determined by BMI, where various categories are indicated by particular ranges. A BMI of less than 18.5 is considered underweight, whereas a normal (healthy) BMI falls between 18.5 and 24.9. If the average body mass index (BMI) is between 25 and 29.9, you are considered overweight. Obesity has additional subcategories: A BMI between 30 and 34.9 is considered class I obesity, 35 to 39.9 is class II obesity, and extremely severe obesity is class III obesity.5
Both hereditary and environmental factors can contribute to obesity, which is a complex, multivariate, non-communicable disease. Among the known reasons include endocrine problems, mental illnesses, drugs, hereditary predisposition, diet, and physical activity.6
Obesity is strongly linked to a number of important causes of morbidity and mortality, including diabetes mellitus, Type 2 diabetes (T2DM), insulin resistance, fatty liver disease, a metabolic disorder, dyslipidemia, cardiovascular disease (CVD), cancer, and high blood pressure atherosclerosis.7–10 Considering all that, obesity has been identified as a serious worldwide health concern.7
Subfatin, also referred to as Meteorin-like (Metrnl), is a novel adipokine released by adipose tissue and skeletal muscle. Metrnl has been detected in adipose tissue from both humans and animals, according to a study by Li et al.11–13 It is extensively found in barrier tissues, such as the respiratory system, the intestinal tract, and skin epithelium.14–15 Clinical research focuses on the connection between metabolic disorders and inflammatory diseases like coronary artery disease, type 2 diabetes, etc.16–21
In white adipose tissue, MERTRL expression can be induced by exercise and severe cold exposure. In addition to raising circulating MERT levels, mice’s glucose tolerance is improved and energy expenditure is encouraged.22 Additionally, Metrnl can control adipocyte differentiation, lipid-mediated inflammation, and insulin resistance by promoting the expression of genes linked to thermogenesis in beige/brown adipose tissue.23–24
In addition, there still exists disagreements about circulating Metrnl concentrations in T2DM and obesity.25–28 Thus, this study aimed to evaluate the association between serum Metrnl levels and glucose and lipid metabolism in obese individuals.
Materials and Methods
Ethical Considerations
Written informed consent was obtained from all participants prior to enrollment. (Verbal consent is less common for research; written is standard). Both participating hospital and college of Pharmacy local ethical committees gave their approval to the study.
Research Participants
This cross-sectional research involved participants aged 30 to 70 years who under went regular health examinations conducted in Basrah City, Iraq from June 2022 to December 2023. who were recruited from a leading hospital in Basrah City (Almawanee General Hospital). Type 1 DM Myocardial infarction , stroke history, cardiac problems, renal or hepatic disorders, pregnancy or lactation, thyroid disorders, cancer, chronic inflammation, autoimmune diseases, acute infection, and usage of drugs that impacted lipid and blood glucose levels —for instance an antidiabetic, statin, corticosteroid, or estrogen hormone medication—were among the exclusion criteria.
Measurements of anthropometry and biochemistry
BMI was calculated as weight in kilograms divided by height in meters squared (kg/m²).
Participants were categorized into three groups: normal-weight (BMI < 25 kg/m²), overweight (BMI 25–29.9 kg/m²), and obese (BMI ≥ 30 kg/m²).29
Methods
Venous blood samples were taken in the morning following an overnight fast. A Randox kit (GLMC PAP) and the glucose oxidase technique were used to quantify the glucose after the samples were put in a gel tube and centrifuged for 10 minutes at 6000 rpm to separate the serum. Fasting insulin was measured using a two-site immune enzymatic assay kit, USA, utilizing the TOSOH device.
A kit from Bio-Rad, USA, was utilized to assay hemoglobin A1c using ion exchange high-performance liquid chromatography (HPLC). (Kit Reference. No. 220-02021). Lipid profile: total cholesterol assessed using the cholesterol CHOD PAP kit, Triglyceride determined by the triglyceride GOP method kit (BioLABo SA) France Ref. No. 86516, HDL using HDL-Cholesterol kit (BIOLABO SA, France). Utilizing Cell Biolabs Inc. Alanine Aminotransferase (ALT) Enzyme Assay Kit (Colorimetric) MET-5123, ALT was calculated, whereas aspartate aminotransferase (AST) was evaluated with the aspartate aminotransferase AST Kit (cell biology 2805), and meteorin like (Metrnl) was quantified using the human meteorin-like protein kit (Metrnl Elisa kit).
FBG (mmol/L) × FINS (mIU/L)/22.5 is the equation30 was used to calculate the homeostasis model assessment for insulin resistance (HOMAIR).
Biochemical parameters
low risk of fasting blood glucose (mg/dl) was between (100-125.9 mg/dl), the high risk was at the concentration ≥ 126 mg/dl. Good glycemic control of HbA1c (%) was at concentration < 7.5%, poor glycemic control was at concentration > 7.5% Sensitive Insulin (μu/ml) was normal range at concentration < 10 μu/ml, high risk at concentration ≥ 10 μu/ml (31).
The risk associated with HOMA-IR at this value was > 2.5, whereas the usual value was ≤ 2.5 (32). The lipid profiles associated with atherogenesis: Hypercholesterolemia (hyper-TC), for instance, was classified as TC equaling or above 200 mg/dl, and high triglycerides (hyper-TG) as TG exceeding or equal to 150 mg/dl. HDL-C levels below 40 mmol/L are indicative of hypo-HDL cholesterolemia, or hypo-HDL.33
Statistical evaluation
IBM SPSS version 26.0 was used for all analyses. For continuous variables, information was presented as the average ± standard deviation. The categorical variables were displayed as a percentage. An analysis that was one-way was used to compare groups.
ANOVA. The association between serum Metrnl concentrations and metabolic indicators was evaluated using Spearman and partial correlation analysis. binary, multiple, Using category logistic regression, the relationship between serum MTR levels and atherogenic dyslipidemia was further examined.
The variables deemed clinically significant or demonstrating a noteworthy correlation with the Metrnl values were taken into account. It was deemed statistically significant when the two-tailed P value was less than 0.05.
Results
Table 1 displays the attributes of the individuals, with mean ages of 46±9 for the normal group, 49±9.5 for the overweight group, and 51±10 for the obese group. Based on their BMI, the individuals were divided into three groups. Age and sex differences between the groups under study were not statistically significant. Conversely, the overweight and obese groups showed significantly higher BMI, FBG, insulin, TG, and TC levels, as well as HOMA-IR values (p<0.001 for all), alongside significantly lower HDL and Metrnl levels.
Table 1: displays the clinical and biochemical characteristics of the study participants.
| Variable | N=(38)
normal |
N=(33)
Over Wight |
N=(31)
Obese |
p-value | |||
| Mean± | S.D | Mean | S.D | Mean | S.D | ||
| metrnal | 229.5789 | 16.4508 | 188.5938 | 18.50869 | 137.0667 | 15.48511 | 0.000 |
| TC | 136.7632 | 19.95594 | 219.8125 | 14.42318 | 275.5667 | 15.66389 | 0.000 |
| TG | 99.94737 | 26.87403 | 153.3594 | 33.12105 | 217.5 | 16.37018 | 0.000 |
| HDL | 55.81579 | 21.7664 | 41.97813 | 28.62535 | 26.57 | 1.845666 | 0.000 |
| FBS | 86.375 | 3.275498 | 92.90781 | 1.481722 | 99.71 | 2.65237 | 0.000 |
| INSU | 6.671053 | 1.38015 | 11.10125 | 1.293729 | 15.60333 | 1.599996 | 0.000 |
| HOM | 1.434763 | 0.33932 | 2.550219 | 0.333652 | 3.847333 | 0.496324 | 0.000 |
| Age | 46.05263 | 9.013346 | 49.34375 | 9.512672 | 51.43333 | 9.583331 | 0.367 |
| Sex | 1.5 | 0.506712 | 1.5 | 0.508001 | 1.466667 | 0.507416 | 0.521 |
| BMI | 31.38026 | 32.02232 | 26.29688 | 6.379344 | 26.51167 | 5.994183 | 0.000 |
Data were presented as the mean ± S.D., and p values for the binary (sex) variable were computed by Employing binary logistic regression, p-values for the categorical (BMI) variable were determined by utilizing ordinal logistic regression, along with p_values for continuous variables (TG, TC, HDL, FBS, Insulin, HOMA IR Values were determined through standard multiple regression, with p_values < 0.05. Body mass index (BMI); triglycerides (TG); TC, meaning total cholesterol High density lipoprotein cholesterol, abbreviated HDL-C; fasting plasma glucose, or simply FBG; fasting insulin levels, or as FINS; HOMA-IR, homeostasis model evaluation, insulin sensitivity, and insulin resistance assessment; Meteorin-like, Metrnl figure (1): demonstrated lower circulating levels of metrnl in overweight and obese individuals compared to normal ones, with means of 188.6, 137, and 229.6 (pg/ml) respectively.
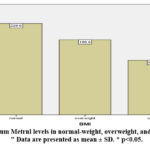 |
Figure 1: Serum Metrnl levels in normal-weight, overweight, and obese groups.” Data are presented as mean ± SD. * p<0.05.
|
Association of serum Metrnl concentrations and clinical variables
Correlation analysis was used to evaluate the relationship between each participant’s blood Metrnl levels and metabolic parameters as in Table 2, also, Figures 2, 3and 4. Circulating Metrnl levels were positively correlated with HDL-C (r = 0.947, P < 0.001) and negatively correlated with BMI (r =-0.914), TG (r =-0.948), TC (r =-0.990,), FBS (r =-0.982), FINS (r =-0.992), and HOMA-IR (r =-0.992), P < 0.001.
Table 2: Examination of the relationship between clinical factors and serum Mternl levels
| S.Metrnl | S.Metrnl* | |||
| r | p | r | P* | |
| Age | -.063 | .532 | ||
| BMI | -.914 | .000 | ||
| S.TC | -.990 | .000 | -.822 | .000 |
| S.TG | -.948 | .000 | -.627 | .000 |
| S.HDL | .947 | .000 | .180 | .076 |
| FBS | -.982 | .000 | -.806 | .000 |
| S.insulin | -.992 | .000 | -.931 | .000 |
| HOMA-IR | -.992 | .000 | -.915 | .000 |
Spearman’s correlation analysis and *partial correlation analysis, which accounts for age, sex, BMI, and eGFR, were used to calculate P values. P value < 0.05 is indicated in bold.
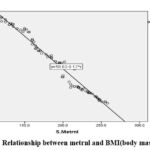 |
Figure 2: Relationship between metrnl and BMI(body mass index).
|
 |
Figure 3: Relationship between serum metrnl with lipid profile .
|
 |
Figure 4: Relationship between serum metrnl with glucose homeostasis .
|
The logistic regression analysis for serum metrnl levels regarding to different variables
Multiple logistic regression analysis was performed to identify independent associations between circulating Metrnl levels and selected variables (Table3).
Parameters of lipid metabolism (TC, TG, HDL) and glucose homeostasis (FBS, Insulin, HOMA-IR) were identified as independently and significantly associated with Metrnl levels. No significant association was found with age. Binary logistic regression investigation was approved out in table 4 , Regarding to sex no significant association with circulating metrnl . Regarding to body mass index(BMI) by category logistic regression in table 5 ,observed significantly negative association with metrnl levels
Table 3: Multiple logistic regression between serum levels of metrnl with different variable
| Variable | Coefficient (β) | p-value | 95.0% Confidence Interval for B | |
| Lower Bound | Upper Bound | |||
| Age | -.091 | .367 | -.067 | .025 |
| S.TC | -.972 | .000 | -1.482 | -1.345 |
| S.TG | -.925 | .000 | -1.326 | -1.125 |
| S.HDL | .521 | .000 | .202 | .401 |
| FBS | -.967 | .000 | -.149 | -.134 |
| S.insulin | -.989 | .000 | -.097 | -.091 |
| HOMA-IR | -.987 | .000 | -.026 | -.024 |
Table 4: Binary logistic regression between serum levels of metrnl with Sex,
| Variable | p-value | Exp(B) | 95% C.I.for EXP(B) | |
| Lower | Upper | |||
| Sex b | 0.521 | 0.997 | 0.987 | 1.006 |
Table 5: Category logistic regression between serum levels of metrnl with BMI .
| BMIa | p-value | Exp(B) | 95% Confidence Interval for Exp(B) | ||
| Lower Bound | Upper Bound | ||||
| overweight | Intercept | .000 | |||
| S.Metrnl | .000 | -0.862 | .797 | .931 | |
| obese | Intercept | .027 | |||
| S.Metrnl | .033 | -0.326 | .117 | .912 | |
The reference category is: normal.
In figure 4 showed relationship between different veriables FBG, insulin, HOMA-IR,TC, TG and Metrnl values regarding to BMI and elucidated lower in overweight and obese
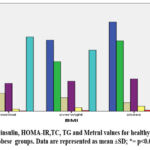 |
Figure 5: FBG, insulin, HOMA-IR,TC, TG and Metrnl values for healthy and overweight ,obese groups. Data are represented as mean ±SD; *= p<0.05
|
Discussion
Meteorin-like (Metrnl) is a secreted protein expressed in peripheral tissues and plays a vital role in several physiological and pathological processes. Increasing evidence suggests that Metrnl contributes to the regulation of metabolic homeostasis, particularly under pathological conditions such as obesity.
Several studies have highlighted the association between obesity and circulating Metrnl levels.34-46 For instance, Wang et al.34 demonstrated that both serum and adipose tissue from obese mice displayed elevated concentrations of Metrnl. Similarly, obese individuals were reported to have higher Metrnl levels,36 and Loffler et al.35 observed consistently increased Metrnl expression in the adipose tissue of obese children compared with their lean counterparts. In contrast, other investigations reported decreased circulating hormone concentrations in obesity.37-41 Moreover, some studies found no significant correlation between serum Metrnl levels and body mass index (BMI) in physically examined participants.43 Interestingly, several reports demonstrated a negative correlation between circulating Metrnl and both BMI and visceral adiposity.37-39,44,45
In line with these findings, the present study revealed a negative relationship between serum Metrnl levels and BMI. These results are consistent with other recent reports suggesting that overweight and obese individuals exhibit reduced circulating Metrnl concentrations. Metrnl is primarily secreted by muscle and adipose tissue, given that obesity is often accompanied by sarcopenia and adipose tissue dysfunction, the decreased Metrnl levels observed may result from impaired adipose tissue and muscle mass loss.37-39,44,45
Furthermore, growing evidence suggests that Metrnl plays a significant role in lipid metabolism. Through activation of fatty acid oxidation (FAO) in skeletal muscle, mediated by AMPK or PPARγ signaling, Metrnl upregulates genes involved in lipid metabolism and enhances lipase activity in adipose tissue.49 In addition, tissue-specific Metrnl expression has been implicated in regulating blood lipid components in mice.50 Therefore, reduced circulating Metrnl levels may impair FAO and promote triglyceride (TG) synthesis in the liver and adipose tissue by inhibiting lipoprotein lipase, thereby leading to hypertriglyceridemia and impaired metabolism of cholesteryl esters (HDL-C, LDL-C).51,52
Another important finding of the present study is the negative correlation between serum Metrnl levels, glucose concentration, and insulin resistance indices (serum insulin, HOMA-IR). This suggests that reduced Metrnl may act as a trigger for insulin resistance and, consequently, the progression of diabetes mellitus. Mechanistically, Metrnl enhances insulin sensitivity and glucose tolerance through activation of peroxisome proliferator-activated receptor gamma (PPARγ), 53 improves glucose metabolism by promoting browning of white adipose tissue,54 and stimulates adipose tissue macrophages to reinforce thermogenic and anti-inflammatory gene expression programs.55
Despite these promising observations, certain limitations should be acknowledged. The cross-sectional design and relatively small sample size of the present study limit the ability to establish causality between circulating Metrnl and metabolic disorders. Although our findings demonstrate an inverse association between circulating Metrnl and indices of obesity, insulin resistance, and dyslipidemia, several alternative interpretations must be considered. First, the cross-sectional design precludes causal inference; reduced Metrnl may represent a consequence rather than a cause of metabolic dysfunction, reflecting sarcopenia or adipose tissue impairment commonly observed in obesity.39,46,47 Second, unmeasured confounding factors—including body composition, physical activity, systemic inflammation, and medication use—may have influenced the observed associations. Prior reports suggest that circulating Metrnl is modulated by exercise, inflammatory cytokines, and anti-diabetic therapies.37-39,53 Third, the heterogeneity of findings in the literature, with some studies showing elevated or unchanged Metrnl in obesity,34,36,54 underscores the possibility of non-linear or context-dependent effects. For example, compensatory upregulation at early stages of adipose expansion may be followed by downregulation in advanced obesity.
To assess robustness, sensitivity analyses adjusting for age, sex, and common medications were performed, and the associations between Metrnl and BMI, HOMA-IR, and lipid parameters remained materially unchanged. However, residual confounding cannot be excluded, particularly by visceral adiposity and muscle mass, which were not directly measured. Future longitudinal and interventional studies are required to clarify whether low Metrnl precedes or results from metabolic impairment, and whether changes in Metrnl with lifestyle or pharmacological interventions mediate improvements in cardiometabolic health.
The investigation has a number of important benefits. This study contributes to an emerging field by focusing on Metrnl, a relatively novel adipokine whose role in obesity and metabolic diseases is not yet fully understood. Rigorous exclusion criteria and meticulous participant selection strengthened the study by reducing the impact of confounding comorbidities and medications. The thorough biochemical profiling that was carried out, which comprised fasting blood glucose, HbA1c, insulin, HOMA-IR, lipid profile, and liver enzymes, is another strength. This allowed for an integrated assessment of Metrnl in connection to both glucose and lipid metabolism. While sensitivity analyses validated the constancy of the connections, the use of various statistical techniques—ANOVA, correlation, and regression analyses—further enhanced the findings’ robustness. Additionally, the data support the reliability and possible therapeutic significance of these findings because they concur with other research showing decreased circulating Metrnl in obesity and its correlation with dyslipidemia and insulin resistance.
However, several limits must be recognized. Causal inference is prevented by the cross-sectional design, and the data’ portability may be limited by the very small sample size. Furthermore, circulating Metrnl concentrations might have been impacted by residual confounding variables that were not directly evaluated, such as visceral adiposity, body composition, physical activity, and dietary practices. Lastly, the study’s external validity may be limited because it was only carried out at one center in Basrah, Iraq. Therefore, in order to determine the causative involvement of Metrnl in the development of metabolic diseases associated with obesity and to assess its potential as a biomarker and therapeutic target, bigger longitudinal and interventional studies are necessary.
Conclusion
Reduced serum Metrnl levels are strongly associated with obesity, dyslipidemia, and insulin resistance. These findings highlight the potential of Metrnl as a biomarker and therapeutic target for obesity-related metabolic disturbances, particularly atherogenic dyslipidemia. Future longitudinal studies are warranted to confirm the causal role of Metrnl in the development of metabolic diseases
Acknowledgement
The authors would like to express their sincere gratitude to the staff of Almawanee General Hospital for their invaluable support, cooperation, and assistance throughout the course of this work. Their dedication and commitment greatly facilitated the completion of this study.
We also extend our deep appreciation to the College of Pharmacy for providing guidance, academic support, and essential resources that contributed significantly to the success of this research.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
The human subjects study was approved by the ethical panel of the Basrah Health Directorate of the Ministry of Health in Iraq (181 at 1-6-2024). The studies were conducted in accordance with local laws and institutional norms. The subjects gave their written informed consent to participate in this investigation. All research participants provided written informed permission. The study respected patient privacy and complied with the Declaration of Helsinki.
Informed Consent Statement
Written informed consent was obtained from all participants prior to enrollment. (Verbal consent is less common for research; written is standard). Both participating hospital and college of Pharmacy local ethical committees gave their approval to the study.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Author Contributions
- Anwar Yonis Ibrahim: Conceptualization, Methodology, Investigation, Writing – Review & Editing.
- Nadheerah F. Neamah: Investigation, Data Curation, Writing – Original Draft, Writing – Review & Editing.
References
- Powell-Wiley TM, Poirier P, Burke LE, et al. Obesity and cardiovascular disease: a scientific statement from the American Heart Association. Circulation. 2021;143(21):e984-e1010. doi:10.1161/CIR.0000000000000973
CrossRef - Upadhyay J, Farr O, Perakakis N, Ghaly W, Mantzoros C. Obesity as a disease. Med Clin North Am. 2018;102(1):13-33. doi:10.1016/j.mcna.2017.08.004
CrossRef - Adams J. Addressing socioeconomic inequalities in obesity: democratizing access to resources for achieving and maintaining a healthy weight. PLoS Med. 2020;17(7):e1003243. doi:10.1371/journal.pmed.1003243
CrossRef - Anekwe CV, Jarrell AR, Townsend MJ, Gaudier GI, Hiserodt JM, Stanford FC. Socioeconomics of obesity. Curr Obes Rep. 2020;9(3):272-279. doi:10.1007/s13679-020-00398-7
CrossRef - Kanazawa M, Yoshiike N, Osaka T, Numba Y, Zimmet P, Inoue S. Criteria and classification of obesity in Japan and Asia-Oceania. World Rev Nutr Diet . 2005;94:1-12. doi:10.1159/000088200
CrossRef - Ang YN, Wee BS, Poh BK, Ismail MN. Multifactorial influences of childhood obesity. Curr Obes Rep. 2013;2(1):10-22. doi:10.1007/s13679-012-0042-7
CrossRef - Bagnall A, Radley D, Jones R, et al. Whole systems approaches to obesity and other complex public health challenges: a systematic review. BMC Public Health. 2019;19(1):8. doi:10.1186/s12889-018-6274-z
CrossRef - Blüher M. Obesity: global epidemiology and pathogenesis. Nat Rev Endocrinol. 2019;15(5):288-298. doi:10.1038/s41574-019-0176-8
CrossRef - Piché ME, Tchernof A, Després JP. Obesity phenotypes, diabetes, and cardiovascular diseases. Circ Res. 2020;126(11):1477-1500. doi:10.1161/CIRCRESAHA.120.316101
CrossRef - Speakman JR, Hall KD. Carbohydrates, insulin, and obesity. Science. 2021;372(6542):577-578. doi:10.1126/science.aav0448
CrossRef - Li ZY, Zheng SL, Wang P, et al. Subfatin is a novel adipokine and unlike meteorin in adipose and brain expression. CNS Neurosci Ther. 2014;20(4):344-354. doi:10.1111/cns.12219
CrossRef - Rao RR, Long JZ, White JP, et al. Meteorin-like is a hormone that regulates immune-adipose interactions to increase beige fat thermogenesis. Cell. 2014;157(6):1279-1291. doi:10.1016/j.cell.2014.03.065
CrossRef - Zheng SL, Li ZY, Song J, Liu JM, Miao CY. Metrnl: a secreted protein with new emerging functions. Acta Pharmacol Sin. 2016;37(5):571-579. doi:10.1038/aps.2016.9
CrossRef - Ushach I, Burkhardt AM, Martinez C, et al. METEORIN-LIKE is a cytokine associated with barrier tissues and alternatively activated macrophages. Clin Immunol. 2015;156:119-127. doi:10.1016/j.clim.2014.12.001
CrossRef - Li ZY, Fan MB, Zhang SL, et al. Intestinal metrnl released into the gut lumen acts as a local regulator for gut antimicrobial peptides. Acta Pharmacol Sin. 2016;37(11):1458-1466. doi:10.1038/aps.2016.96
CrossRef - Chung HS, Hwang SY, Choi JH, et al. Implications of circulating Meteorin-like (Metrnl) level in human subjects with type 2 diabetes. Diabetes Res Clin Pract. 2018;136:100-107. doi:10.1016/j.diabres.2017.11.031
CrossRef - Dadmanesh M, Aghajani H, Fadaei R, Ghorban K. Lower serum levels of Meteorin-like/Subfatin in patients with coronary artery disease and type 2 diabetes mellitus are negatively associated with insulin resistance and inflammatory cytokines. PLoS One. 2018;13(10):e0204180. doi:10.1371/journal.pone.0204180
CrossRef - Liu ZX, Ji HH, Yao MP, et al. Serum Metrnl is associated with the presence and severity of coronary artery disease. J Cell Mol Med. 2019;23(1):271-280. doi:10.1111/jcmm.13915
CrossRef - Zuo L, Ge S, Ge Y, et al. The adipokine Metrnl ameliorates chronic colitis in Il-10-/- mice by attenuating mesenteric adipose tissue lesions during spontaneous colitis. J Crohns Colitis. 2019;13(7):931-941. doi:10.1093/ecco-jcc/jjz006
CrossRef - Bridgewood C, Russell T, Weedon H, et al. The novel cytokine Metrnl/IL-41 is elevated in psoriatic arthritis synovium and inducible from both entheseal and synovial fibroblasts. Clin Immunol. 2019;208:108253. doi:10.1016/j.clim.2019.108253
CrossRef - Jung TW, Lee SH, Kim HC, et al. METRNL attenuates lipid-induced inflammation and insulin resistance via AMPK or PPARδ-dependent pathways in skeletal muscle of mice. Exp Mol Med. 2018;50(9):1-11. doi:10.1038/s12276-018-0147-5
CrossRef - Li ZY, Song J, Zheng SL, et al. Adipocyte metrnl antagonizes insulin resistance through PPARγ signaling. Diabetes. 2015;64(12):4011-4022. doi:10.2337/db15-0274
CrossRef - AlKhairi I, Cherian P, Abu-Farha M, et al. Increased expression of meteorin-like hormone in type 2 diabetes and obesity and its association with irisin. Cells. 2019;8(10):1283. doi:10.3390/cells8101283
CrossRef - Schmid A, Karrasch T, Schäffler A. Meteorin-like protein (Metrnl) in obesity, during weight loss and in adipocyte differentiation. J Clin Med. 2021;10(19):4338. doi:10.3390/jcm10194338
CrossRef - Moradi N, Fadaei R, Roozbehkia M, et al. Meteorin-like protein and asprosin levels in children and adolescents with obesity and their relationship with insulin resistance and metabolic syndrome. Lab Med. 2023;54(5):457-463. doi:10.1093/labmed/lmac152
CrossRef - Willett K, Jiang R, Lenart E, Spiegelman D, Willett W. Comparison of bioelectrical impedance and BMI in predicting obesity-related medical conditions. Obesity (Silver Spring). 2006;14(3):480-490. doi:10.1038/oby.2006.63
CrossRef - Schmid A, Karrasch T, Schäffler A. Meteorin-like protein (Metrnl) in obesity, during weight loss and in adipocyte differentiation. J Clin Med (2021) 10(19):4338. doi: 10.3390/jcm10194338.
CrossRef - Moradi N,Fadaei R, Roozbehkia M, Nourbakhsh M, Nourbakhsh M, Razzaghy-Azar M, Larijani B . Meteorin-like Protein and Asprosin Levels in Children and Adolescents with Obesity and Their Relationship with Insulin Resistance and Metabolic Syndrome .Laboratory Medicine. 2023;54( 5):457–463 https://doi.org/10.1093/labmed/lmac152
CrossRef - Willett, K. et al., Comparison of bioelectrical impedance and BMI in predicting obesity-related medical conditions. Obes. (Silver Spring), 14(3):480–490.
CrossRef - Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner Homeostasis model assessment: Insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985;28(7):412–9. doi: 10.1007/bf00280883 .
CrossRef - I. Sherwani, H. A. Khan, A. Ekhzaimy, A. Masood, and M. K. Sakharkar, “Significance of HbA1c test in diagnosis and prognosis of diabetic patients,” Biomarker Insights, 2016;11(July 03):95–104. doi: 10.4137/Bmi.s38440.
CrossRef - A, W. K, D. R, W. LE, and H. SM, “The natural course of beta-cell function in nondiabetic and diabetic individuals: the Insulin Resistance Atherosclerosis Study,” Diabetes, 2006;55(4):1114–1120 doi: 10.2337/DIABETES.55.04.06.DB05-1100.
CrossRef - Chinese Medicine Association. Guideline for primary care of dyslipidemias: Practice version. Chin J Gen Pract 2019;018(005):417–21. doi: 10.3760/ cma.j.issn.1671-7368.2019.05.004 .
- Wang K , Li F , wang e , Deny Y , Cao Z , Cu. Y , et al . Serum level of Meteorin-like ( Metrnal ) are increased in patients with newly diagnosed type 2 diabetes mellitus and are associated with resistance. Medsci morit 2019;25:233y-43. do:10.12639//msm.915331.
CrossRef - Loffler D , Landgrat K , Rockstron D , Schwartze J , Dunzentorfor H , kiess W , et al . Metrnl decreases during adipogenesis and inhibits adipocyte differentiation leading to adipocyte hypertrophy in human . Int J obes (Lord) . 2017;41:112_9
CrossRef - Alkhairi I , cherian P , Abu_Farha M , Madhour AA , Nizam R , Methem M , et al . Increased expression of Meteorin _ like hormon in type 2 diabetes and obesity and its associated with irisin. cells. 2019:8:1283.
CrossRef - Pellibero S , Piquer_Garcia I , Ferrar_Carrin G , Puig R , Martinezz E , Mareno P , et al . Opposite changes in meteorin _ like and oncostatin levels are associated with metabolic improvements after bariatric surgery. Int J Oba ( Lond ) 2018;42(4): 919-22. doi : 10.1038 lijo. 2017.268.
CrossRef - Dn Y , Ye X , Lu A , Zhao D , Lin J , Cheng J , et al . Inverse relationships between serum metrnl levels and visceral fat obesity ( VFO ) in patients with type 2 diabetes. Diabetes Res Elin pract ( Zeoza ) 2020;161:108068. doi:10.1016 / j.diabres2020.108068 .
CrossRef - Ding X , Chang X , Wang J , Bian N , An Y , et al . Serum metrnl levels are decreased in subjects with overweight or obesity and are independently associated with adverse lipid profile. Front Endocrine. 2022;13:938541. doi:10.3389/fendo.938341.
CrossRef - schmid A, Karrasch T , Schaffer A , meteorin like protein ( metrnl) in obesity , during weight loss and in adiposity differentiation , J clin med 2021;10(19):4338.doi: 10.3390/jcm 10184338 .
CrossRef - Jung J , Chio S , obesity and its metabolic components the role of adipose and relationship between obesity, inflammation resistance , dystopian and non- alcoholic fatty liner discase . IntJ Mol wc 2014;15(4):6184-223.
CrossRef - Li Zy , Song J , Zheng Sl , For MB , Gwan YF , QuY , etal. Adipocyte , Metrnl antgonizes insulin resistance signatling, Diabetes, 2015;64:4011-22.
CrossRef - El-Ashmawy HM , Selim Fo , Hosny TAM , Atmassy HN , Association of low seram metcorin like (Metrnl )concatenation with waresing of glucose tolerance improved endothelial function and atherosclerosis. Diabetes Res. Clin pract 2019;150:57-63. DoI : 10.1016/j. Diabetes.02.026.
CrossRef - Dadmanesh M , Aghajani H , Faclaci R , Ghorban K . Lawer scream levels of meteorin like /subfation in particle with coranary with insulin resistance, and inflammatory cytokines. Plos-one .2018,13:co2014180.
CrossRef - Fononi FZ , Fadaei R , Moradi N , zandieh Z , Ausaripoer S , Yekaniruejad Ms , et al . Circulating level of meteorin _ like protein in poly cystic ovary syndrome. A case control study. Plos one . 2020;15: eo231943.
CrossRef - Rao RR, Long JZ, White JP, Svensson KJ, Lou J, Lokurkar I, et al. Meteorinlike is a hormone that regulates immune-adipose interactions to increase beige fat thermogenesis. Cell 2014;157(6):1279–91. doi: 10.1016/j.cell.2014.03.065
CrossRef - Li ZY, Zheng SL, Wang P, Xu TY, Guan YF, Zhang YJ, et al. Subfatin is a novel adipokine and unlike meteorin in adipose and brain expression. CNS Neurosci Ther 2014;20(4):344–54. doi: 10.1111/cns.12219 .
CrossRef - Bamba V, Rader DJ. Obesity and atherogenic dyslipidemia. Gastroenterology 2007;132(6):2181–90. doi: 10.1053/gastro.2007.03.056 .
CrossRef - Jung TW, Lee SH, Kim HC, Bang JS, Abd El-Aty AM, Hacımüftüoğlu A, et al. METRNL attenuates lipid-induced inflammation and insulin resistance via AMPK or PPARd-dependent pathways in skeletal muscle of mice. Exp Mol Med 2018;50(9):1–11. doi: 10.1038/s12276-018-0147-5.
CrossRef - Qi Q, Hu WJ, Zheng SL, Zhang SL, Le YY, Li ZY, et al. Metrnl deficiency decreases blood HDL cholesterol and increases blood triglyceride. Acta Pharmacol Sin 2020;41(12):1568–75. doi: 10.1038/s41401-020-0368-8 .
CrossRef - Jung T , Lee S, Kim H , Bang J , Hacımüftüoğlu A , Shin Y and Jeong J . METRNL attenuates lipid-induced inflammation and insulin resistance via AMPK or PPARδ-dependent pathways in skeletal muscle of mice. Experimental & Molecular Medicine 2018;50:122 DOI 10.1038/s12276-018-0147-5.
CrossRef - Zhou Y; Liu L, Jin B, Wu U , Xu L , Chang X, Wang G , Huang Y et al . Metrnl Alleviates Lipid Accumulation by Modulating Mitochondrial Homeostasis in Diabetic Nephropathy . Diabetes. 2023 May; 72(5): 611–626.
CrossRef - AlKhairi et al., “Increased Expression of Meteorin-Like Hormone in Type 2 Diabetes and Obesity and Its Association with Irisin,” Cells, 2019;8(10). doi: 10.3390/cells8101283.
CrossRef - L. Zheng, Z. Y. Li, J. Song, J. M. Liu, and C. Y. Miao, “Metrnl: A secreted protein with new emerging functions,” Nature Publishing Group Acta Pharmacologica Sinica, 2016;37(5);571–579. doi: 10.1038/aps.2016.9.
CrossRef - Y. Li et al., “Adipocyte metrnl antagonizes insulin resistance through pparg signaling,” Diabetes, 2015;64(12): 4011–4022. doi: 10.2337/db15-0274.
CrossRef







