Manuscript accepted on :23-07-2025
Published online on: 25-08-2025
Plagiarism Check: Yes
Reviewed by: Dr. Putu Indah
Second Review by: Dr . Mais Emad
Final Approval by: Dr. Mariia Shanaida
Muhammad Aiman Haiqal bin Ismail1,3,4 , Koh Soo Peng2
, Koh Soo Peng2 , Sanimah Simoh3
, Sanimah Simoh3 , Armania Nurdin1,4
, Armania Nurdin1,4 , Machap Chandradevan3
, Machap Chandradevan3 and Muhammad Nazrul Hakim1,4*
and Muhammad Nazrul Hakim1,4*
1Department of Biomedical Sciences, Faculty of Medicine and Health Sciences, University Putra Malaysia, UPM Serdang, Selangor, Malaysia.
2Food Science and Technology Research Centre, Malaysian Agricultural Research Development Centre, MARDI, Persiaran MARDI-UPM, Serdang, Selangor, Malaysia.
3Biotechnology and Nanotechnology Research Centre, Malaysian Agricultural Research Development Centre, MARDI, Persiaran MARDI-UPM, , Selangor, Malaysia.
4Laboratory of UPM-MAKNA Cancer Research (CANRES), Institute of Bioscience, and Halal Product Institute, Universiti Putra Malaysia, UPM Serdang, Selangor, Malaysia.
Correspondence author Email: nazrulh@upm.edu.my
DOI : https://dx.doi.org/10.13005/bpj/3266
Abstract
Herbal remedies are increasingly studied for their potential as safer alternatives to conventional anti-inflammatory drugs. However, the anti-inflammatory efficacy and interaction of many herbal combinations remain poorly understood. This study addresses this gap by investigating the anti-inflammatory activity, herb-herb interactions, and LC-ESI-MS/MS metabolite profiles of Cleome gynandra and Melicope ptelefolia leaf extracts. Fresh leaves of both plants were extracted and tested for cytotoxicity and nitric oxide (NO) inhibition using the murine RAW 264.7 macrophage cell line. Cell viability was assessed via MTT assay, while NO inhibition was measured for individual and combined extracts at varying ratios. LC-ESI-MS/MS analysis was performed to identify the major bioactive metabolites. Cleome gynandra extract showed higher cell viability compared to M. ptelefolia, indicating lower cytotoxicity. Among all tested combinations, the 70:30 (w/w) ratio of C. gynandra to M. ptelefolia exhibited the highest cell viability. Although C. gynandra demonstrated limited NO inhibition at 0.5 mg/mL, it still more potent than M. ptelefolia and their formulations. The LC-MS/MS analysis revealed that M. ptelefolia contains flavonoids like kaempferol neohesperidoside and derivatives with sugars such as rhamnose and arabinose, known to inhibit nitric oxide synthase and reduce NO production. Isorhamnetin derivatives also support its anti-inflammatory potential by modulating signaling pathways. In C. gynandra, the presence of quercetin rutinoside (rutin), kaempferol rutinoside, and phenolic acids like caffeic acid contribute to its anti-inflammatory effects. Quercetin rutinoside inhibits enzymes like COX-2 and iNOS and suppresses cytokines such as TNF-α and IL-6. Caffeic acid, a potent antioxidant, inhibits NF-κB activation, collectively supporting its anti-inflammatory capacity. In conclusion, C. gynandra demonstrates promising potential as a safer anti-inflammatory agent compared to M. ptelefolia, although its efficacy remains lower than that of conventional drugs such as diclofenac sodium.
Keywords
Anti-inflammatory; Cleome gynandra; Liquid Chromatography - Mass Spectrometry (LCMS); Melicope ptelefolia; Nitric Oxide (NO) inhibition; Cytotoxicity; Herb-herb Interactions
Download this article as:| Copy the following to cite this article: Ismail M. A. H. B, Peng K. S, Simoh S, Nurdin A, Chandradevan M, Hakim M. N. Phytochemical Profiling and Anti-Inflammatory Activity of Leaf Extract from Cleome gynandra Linn. and Melicope ptelefolia Champ. ex Benth. Biomed Pharmacol J 2025;18(3). |
| Copy the following to cite this URL: Ismail M. A. H. B, Peng K. S, Simoh S, Nurdin A, Chandradevan M, Hakim M. N. Phytochemical Profiling and Anti-Inflammatory Activity of Leaf Extract from Cleome gynandra Linn. and Melicope ptelefolia Champ. ex Benth. Biomed Pharmacol J 2025;18(3). Available from: https://bit.ly/45IXJl7 |
Introduction
Cleome gynandra (Cleomaceae), commonly known as ‘Maman,’ is a widely consumed leafy vegetable and a well-known herb in traditional Indian medicine, used for treating various inflammatory conditions.1 Traditionally, its leaves and seeds have been used to manage ailments such as ear inflammation, epileptic seizures, stomachaches, constipation, and other inflammation-related disorders.2 It is particularly popular among low-income communities due to its affordability and high nutritional value, being rich in vitamins A and C, as well as essential minerals like iron and calcium.3-5 Extensive research has demonstrated the biological activities of C. gynandra, including antidiabetic,6 antioxidant,7 anti-inflammatory,4 and anticancer effects.8 Phytochemical analysis has also confirmed the presence of bioactive compounds such as flavonoids, tannins, alkaloids, terpenoids, steroids, and carotenoids, which contribute to its pharmacological potential.9
Melicope ptelefolia (Rutaceae), locally known as ‘Tenggek burung’ in Malaysia,10 ‘Uam sam ngam’ in Thailand, and ‘Sampang uam’ in Indonesia, is another herb traditionally used in Southeast Asia.11 In Malaysia, the young leaves and shoots are commonly consumed fresh as ‘ulam’ (traditional salad)12 and are believed to possess medicinal properties, particularly for reducing inflammation and treating skin irritations.13 Scientific studies have reported various pharmacological properties of M. ptelefolia leaf extract, including antipyretic, antioxidant, analgesic, and anti-inflammatory effects. These benefits are largely attributed to its rich content of benzopyrans, alkaloids, and glycosides.14-18
Inflammation, although a natural part of the healing process, is often associated with pathological consequences when prolonged or uncontrolled.19 Macrophages play a central role in initiating and regulating inflammation by releasing nitric oxide (NO), pro-inflammatory cytokines, and other mediators in response to stimuli such as bacterial lipopolysaccharides (LPS).20 While non-steroidal anti-inflammatory drugs (NSAIDs) like diclofenac, aspirin, and indomethacin are commonly used to manage inflammation,21-22 their long-term use is associated with serious side effects, including gastrointestinal bleeding, renal toxicity, hypertension, and cardiovascular risks.23-25
Given the drawbacks of NSAIDs, there is increasing interest in exploring herbal alternatives that are believed to be safer and less toxic.26 Both C. gynandra and M. ptelefolia have demonstrated anti-inflammatory potential individually. However, a significant gap in the literature exists regarding the combined use (herb-herb interaction) of these two plants as a formulation for anti-inflammatory therapy. Most studies to date have focused on single-herb activity,27 with little understanding of possible synergistic or antagonistic effects when used in combination.28
To address this research gap, this study aims to evaluate the cytotoxicity and anti-inflammatory properties of C. gynandra, M. ptelefolia, and their various formulations. In addition, LC-ESI-MS/MS-based metabolite profiling will be conducted to identify the active compounds responsible for their pharmacological activities.29 By optimizing the extraction protocol and analyzing both individual and combined effects, this study seeks to contribute to the development of effective and safer herbal-based anti-inflammatory treatments.
Materials and Methods
This study did not involve human participants, animals and therefore, ethical approval or informed consent was not required.
Chemicals and Reagents
Analytical-grade chemicals and solvents were all utilized. Saline phosphate buffer, 3-[4,5-dimethylthiazol-2-yl]Sigma-Aldrich Co. (St. Louis, MO, USA) supplied the 2,5-diphenyltetrazolium bromide (MTT), diclofenac sodium salt, Griess reagent, and lipopolysaccharides (LPS); SciencellTM (San Diego, CA) supplied trypan blue; HiMedia® Laboratories (Mumbai, India) supplied Dulbecco’s Modified Eagle Medium (DMEM); Amresco Inc. (Ohio, USA) supplied sodium dodecyl sulfate and dimethyl sulfoxide (DMSO); Fisher Scientific (Loughborough, Leicestershire, UK) supplied fetal bovine serum (FBS); and Gibco (Life Technologies, USA) supplied the antibiotic mixture of penicillin-streptomycin.
Collection of plant Material
Fresh C. gynandra and M. ptelefolia leaves were supplied by the Biotechnology and Nanotechnology Research Centre, Malaysian Agricultural Research and Development Institute (MARDI), Malaysia. The leaves were collected before noon to maintain their freshness. A voucher specimen of C. gynandra (voucher specimen number MDI 12840) and M. ptelefolia (voucher specimen number MDI 12450) were deposited in MARDI’s herbarium. The plant was verified by a botanist, Dr. Mohd Norfaizal Ghazalli. Lab tissue paper was used to gently dry the gathered leaves after they had been completely cleaned with tap water. A mortar and pestle were then used to grind the clean leaves into a powder in liquid nitrogen. Prior to examination, the powder samples were held at -20 °C after being instantly freeze-dried using a Labconco 6 Plus freeze drier in Kansas City, USA.
Extraction of Samples
The extraction of the powder leaves was performed as described by Chandradevan et al.30 with slight modification. Generally, 0.5 g of grounded and dried powder of leaves of C. gynandra and M. ptelefolia were macerated individually in 70% methanol at a ratio of 1:80 (w/v). The grounded and dried powder leaves of C. gynandra and M. ptelefolia were extracted individually in 40 ml of 70% methanol, respectively. The mixture was then homogenised for 1 min at 262 g using a homogeniser (Ultra Turrax, IKA, Germany), followed by shaking at room temperature for 30 min at 72 g using an orbit shaker (Multi Reax, Heidolph, Germany). A 125 mm diameter filter paper (Advantec, Japan) was used to filter the supernatant. A rotary evaporator (RII, Buchi, Switzerland) was used to concentrate the collected supernatant to create a crystallized extract.
Herbal Combinations
Herbal combinations were prepared by mixing the crystallised C. gynandra and M. ptelefolia extract in a ratio of 30:70 (w/w), 50:50 (w/w) and 70:30 (w/w) based on previously reported herbal formulation study.31
Cell Culture
The American Type Culture Collection (ATCC®) (ATCC accession no. TIB-71) (Manassas, VA, USA) provided the murine RAW 264.7 macrophage cell line. The cells were cultivated in DMEM with 5% FBS added, and they were then incubated at 37 °C in a humidified room with 5% CO2. Each experiment employed cells with less than 18 passage numbers.
MTT Assay
The MTT assay of the C. gynandra and M. ptelefolia and its herbal formulation extracts in RAW 264.7 cells were performed as described by Chung et al.32 with slight modifications. Concisely, the cells (2 × 104 cells per well) were seeded in a 96-well plate and incubated for 24 h Different concentrations of the individual herbs and herbal formulation (0.25–10 mg/mL) were applied to the adhering cells. After 24 hours of treatment, 30 µL of the MTT solution (2 mg/mL) was applied to each well, and the wells were then incubated for 4 hours at 37°C in a dark environment. To dissolve the formazan crystals, 100 µL of DMSO was applied to each well after the supernatant was aspirated. A microplate reader (Biotek EL800, Cambridge Scientific Products, Watertown, USA) set to 570 nm was used to measure the absorbance. The following formula was used to calculate the percentage of cell viability. LPS-induced RAW 264.7 was then used to further select the non-cytotoxic concentration with at least 90% cell viability for the anti-inflammatory test. The MTT assay for each of the extract was conducted in triplicate of three independent experiments. Detailed calculation of cell viability is published previously.32
Nitric Oxide (NO) Inhibition Activity
To determine the anti-inflammatory activity of C. gynandra, M. ptelefolia extract and its herbal formulation, the NO inhibition assay was performed according to procedure described by Chandradevan et al.26 with slight modification. In brief, RAW 264.7 cells (5 × 104 cells per well) were seeded in 96-well plates and left in an incubator for overnight. Then, cells were induced with 10 µg/mL LPS (50 μL, diluted in culture media), followed by treatment with different concentration of plant extract (0.25–10 mg/mL, w/v). Diclofenac sodium salt (0.6 µM) was used as the positive control. The treated cells were incubated in an environment containing 5% CO2 for 24 hours at 37°C. Griess reagent was used to determine the studied extracts’ inhibitory impact on NO generation. Following a 24-hour incubation period, 50 µL of the plate’s medium was cautiously moved into a fresh 96-well plate. After adding the same amount of Griess reagent to each well, the plate was placed in the dark for ten minutes. Sodium nitrite (NaNO2) at varying concentrations (0.156 to 100 µM) served as a standard control in this experiment. A microplate reader (E16 OneTech Medical Equipment Co., Ltd., Guangzhou, China) was then used to measure the absorbance at 540 nm. The NO inhibition assay for each tested extract was performed in triplicate of three independent experiments. Detailed calculation is published previously.26
Separation and tentative identification of selected peaks via LC-ESI-MS/MS
Separation and identification of metabolites were achieved according to the method previously described in Chandradevan and Indu Bala.33 A HPLC system (1200 Series, Agilent Technologies, Germany) was paired with a mass spectrometry (3200 QTrap, ABSciex, USA) was used in this study. A reversed phase C18 column (Thermo Hypersil, 5 µm, 150 x 4.6 mm, Thermo Scientific, USA) was used to ease the separation. Ten microliter of extracts from both herbs at 20 mg/mL was injected into HPLC with the flow rate set at 1 mL/min. A gradient setting was set using 0.1% formic acid as solvent A and 0.1% formic acid in acetonitrile as solvent B. The gradient setting was set as follow: Solvent B was increased from 5% to 40% in 40 min before further increased to 95% for 15 min. A steep decrement of solvent B to 5% within a minute and maintained at the same percentage for another 5 min for equilibrating the column. Ionisation of metabolites was done using electron spray ionization (ESI) at negative mode. The ion source was heated up at 500°C and the entrance potential, de-clustering potential and collision energy were set at -15V, -20V and -25V respectively. Enhance mode was activated in order to obtain the MS/MS fragmentations. The entire analysis was performed using Analyst software (version 1.4.2). Identification of selected peaks from generated chromatograms was done by comparing their mass fragments with previously reported data and online databases. 34, 35
Statistical Analysis
The mean ± standard error of mean (SEM) was used to represent the data. The Statistical Program for Social Sciences (SPSS for Windows, Version 22) (International Business Machines Corp. IBM, New York, USA) was used to do the statistical analysis, and a p-value of less than 0.05 was deemed significant. One-way analysis of variance (ANOVA) was used to evaluate the results, and the Tukey or Dunnet tests were used to determine whether the plant extracts differed significantly.
Results
Cytotoxicity of Raw 264.7 Cells Treated With C. Gynandra, M. Ptelefolia, and Herbal Formulations
Following a 24-hour treatment, the cell viability percentages of RAW 264.7 macrophages significantly decreased (p < 0.05) in a concentration-dependent manner upon exposure to various concentrations (0.25–10 mg/mL, w/v) of the single herbs C. gynandra and M. ptelefolia. Specifically, C. gynandra (Figure 1A) demonstrated a higher tolerated concentration, with cells maintaining at least 90% viability at doses below 4 mg/mL, whereas M. ptelefolia achieved similar viability only at concentrations below 1 mg/mL. Notably, C. gynandra exhibited the highest percentage of cell viability compared to M. ptelefolia, indicating that C. gynandra is less cytotoxic.
 |
Figure 1: Percentages of cell viability of RAW 264.7 cells after treated with the C. Gynandra, M. Ptelefolia and its herbal formulation as determined by MTT assay: (A) between single herb, C. gynandra and M. ptelefolia and (B) herb formulation, C. gynandra: M. ptelefolia at different ratios of 30:70, 50:50 and 70:30. |
When evaluating the herbal formulations combinations of C. gynandra and M. ptelefolia at ratios of 50:50, 70:30, and 30:70 (w/w), similar trends were observed (Figure 1B). The cell viability decreased with increasing concentration, yet the formulation with the highest proportion of C. gynandra (70:30) showed the greatest viability across tested concentrations. Specifically, at concentrations ranging from 0.25–2 mg/mL, cell viability remained above 90%, which is considered non-cytotoxic based on prior standards. The formulations at 50:50 and 30:70 ratios also maintained viability above 90% at these concentrations but showed slightly decreased viability at higher doses. Literature supports these findings; Anbazhagi et al.7 reported that higher concentrations of C. gynandra contain more flavonoids, which can induce cytotoxicity, while Kabir et al.36 observed that M. ptelefolia, with high alkaloid content, can decrease cell viability and cause cell hypotonicity, shrinkage, and death. This information was instrumental in selecting the concentration range of 0.25–2 mg/mL for subsequent anti-inflammatory assays, as these concentrations did not cause cytotoxicity in RAW 264.7 cells.
Anti-Inflammatory Activity of C. gynandra, M. ptelefolia, and Herbal Formulations
The anti-inflammatory potential was assessed via nitric oxide (NO) inhibition in lipopolysaccharide (LPS)-activated RAW 264.7 cells (Figure 2A and 2B). LPS activation induces macrophages to produce pro-inflammatory mediators, including NO, tumor necrosis factor-alpha (TNF-α), interleukins, and enzymes like cyclooxygenase-2 (COX-2). Diclofenac sodium, a nonsteroidal anti-inflammatory drug (NSAID), served as the positive control.
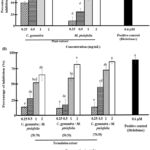 |
Figure 2: Comparison of C. gynandra, M. ptelefolia and its herbal formulation (0.25–2 mg/mL) on the percentage of NO inhibition in LPS-induced RAW 264.7 cells: (A) between single herb of C. gynandra and M. ptelefolia and (B) herb formulation, C. gynandra: M. ptelefolia at different ratios of 30:70, 50:50 and 70:30. |
Initial screening revealed that all extracts, including single herbs and herbal formulations, exhibited insignificant NO inhibition compared to diclofenac sodium. C. gynandra showed a dose-dependent increase in NO inhibition, reaching around 50% at 0.5 mg/mL, but the inhibition was not statistically significant beyond that point. Similarly, M. ptelefolia’s NO inhibition was insignificant starting from 1 mg/mL. The herbal formulations (30:70, 50:50, 70:30 ratios) exhibited increased NO inhibition with rising concentrations; however, at 0.5 mg/mL, their inhibitory effects were minimal and not significantly different from untreated controls. At higher doses (1 and 2 mg/mL), the formulations’ NO inhibition approached that of diclofenac sodium, with the 70:30 formulation achieving approximately 65% inhibition at 2 mg/mL.
Comparison showed that single herb C. gynandra had superior NO inhibitory activity at 0.5 mg/mL, while the herbal formulations only matched the positive control at the highest tested concentrations (1 and 2 mg/mL). Overall, the data suggest that the herbal formulations do not significantly enhance NO suppression compared to C. gynandra alone, indicating a lack of synergistic effect under the tested conditions.
Identification of Metabolites and Evaluation of Synergism
Identification of metabolites from selected peaks was done to ascertain the possible metabolites that might contribute to the anti-inflammatory activities. A total of 13 and 10 metabolites (Numbers in parenthesis are peaks detected) were identified from M. ptelofolia and C. gynandra respectively (Table 1) . Figures 3 and 4 show the chromatograms of M. ptelofolia and C. gynandra corresponding to Table 1. Analysis from the identified metabolites indicates that both herbs are rich in flavonoids and its derivatives. In M. ptelofolia extract, metabolites (4, 5, 6, 8 and 10) were tentatively identified as kaempferol neohesperidoside and its derivatives, conjugated with either a rhamnose or arabinose sugars. Other flavonoids such as isorhamnetin glucoside conjugated with arabinose sugar (9), isorhamnetin-3-O rutinoside (11), isorhamnetin glucoside (12) and isorhamnetin conjugated with 2 arabinose or xylose sugars (13) were identified too. Coumaroylquinic hydrate (1), isomer of coumaroylquinic hydrate (2) and P-coumaroyl glucoside (3) were eluted out approximately at 11.01, 13.02 and 16.58 min respectively.
Quercetin rutinoside (22), an important flavonoid in C. gynandra extract was identified at 13.8 min. The presence of quercetin rutinoside or rutin as one of the main marker compound in C. gynandra was validated in a documented patent by Msika et al.48 Another flavonoid, kaempferol rutinoside (23) was detected after the elution of rutin. Phenolics from the cluster of hydroxycinnamic acids were found abundantly in C. gynandra extract too. Caffeic acid (19) and its derivative (17), dimethoxycinnamic glucoside (18), dimethoxycinnamoyl coumaric acid (20) and dimethoxycinnamoyl glucoronide (21) were some of the hydroxycinnamic acids identified. Other phenolics such as citric acid (14), caffeoylglucaric acid (15) and esculetin glucoside (16) were tentatively identified as well.
The naturally occurring phenolic acids in plants have always been associated with their health benefiting properties. Presence of kaempferol and its derivative in M. ptelefolia extract could explain the high inhibition activity of nitric oxide production. Kaempferol was shown to reduced significantly inducible nitric oxide synthase (iNOS) protein level in a concentration-dependent manner and as well the mRNA of iNOS.57 The same pattern was observed in our study where the RAW 264.7 cells were induced using LPS, as to mimic the induction of NOS. When the concentration of M. ptefolia extract was increased from 0.25 to 2.0 mg/mL, the production of NO ceased significantly. On the other hand, high concentration of quercetin rutinoside could lead to possible anti-inflammatory activity in C. gynandra extract. Lee and colleague58 demonstrated on how quercetin rutinoside attenuated various pro-inflammatory mediators in LPS induced RAW 264.7 cells. Besides, hydroxycinnamic acids such as dimethoxycinnamic derivatives and caffeic acid in C. gynandra have potent anti-inflammation property too, as indicated by Gunasekaran and colleague.59
Even though both herbs possessed significant anti-inflammatory activity, mixing their extracts in certain ratio does not necessarily enhance their activity as shown in this study. The dogma of ‘two better than one’ in herbal extract formulations follows the logic of interaction between the phytochemicals in formulated extracts.60 When the formulated herbal extracts have better activity compared to the individual ones, the net effect is said to be synergistic. The contrary to the synergism is known as antagonistic when the formulated herbal extracts have poor activity than the individual extracts. However, when the activity of herbal extract formulation is not significant compared to the individual ones, additive term will be used instead.61 An additive effect was observed in all formulations at 2 mg/mL compared to their respective individual extracts in inhibiting NO (Figure 2). However, the interaction pattern changes to antagonistic when the concentrations were reduced from 1 mg/mL to 0.25 mg/mL. None of the formulations had better efficacy in inhibiting NO at 0.25 mg/mL compared to single extract of C. gynandra which registered at 50% inhibition.
Table 1: Tentative identification of selected peaks from M. ptefolia and C. gynandra extracts
| Peak number | TR (min) | [M-H]– m/z | MS/MS m/z | Tentatively identified metabolites | Reference |
|
M. ptelefolia extract |
|||||
| 1 | 11.01 | 355.21 | 337, 209, 191, 163, 147, 129, 119 | Coumaroylquinic hydrate | Harbaum et al., 2007 [49] |
| 2 | 13.02 | 355.22 | 337, 209, 191, 172, 163, 144, 128, 119 | Isomer of coumaroylquinic hydrate | Harbaum et al., 2007 [49] |
| 3 | 16.58 | 325.22 | 163,145,133,117,69,59 | P-coumaroyl glucoside | Nuengchamnong & Ingkaninan, 2009[50] |
| 4 | 23.95 | 739.34 | 593, 575, 473, 429, 393, 327, 284, 255, 227, 199, 179, 151 | Kaempferol neohesperidoside with rhamnose moiety | Sanchez-Rabaneda et al., 2003 [51] |
| 5 | 24.37 | 739.38 | 593, 575, 547, 473, 393, 327, 284, 255, 227, 199, 179, 151 | Isomer of kaempferol neohesperidoside with rhamnose moiety | Sanchez-Rabaneda et al., 2003 [51] |
| 6 | 25.55 | 725.31 | 593, 575, 327, 284, 255, 277, 151 | Kaempferol neohesperidoside with arabinose moiety | Sanchez-Rabaneda et al., 2003 [51] |
| 7 | 25.83 | 755.32 | 723, 623, 605, 489, 314, 299, 285, 271, 243 | Isorhamnetin-3-O rutinoside with arabinose moiety | Sanchez-Rabaneda et al., 2003 [51] |
| 8 | 26.40 | 593.30 | 547, 447, 429, 363, 327, 284, 255, 227, 211, 151, 107 | Kaempferol neohesperidoside | Sanchez-Rabaneda et al., 2003 [51] |
| 9 | 26.90 | 609.40 | 315, 357, 300, 285, 271, 25, 243, 227, 151 | Isorhamnetin glucoside with arabinose moeity | Sanchez-Rabaneda et al., 2003 [51] |
| 10 | 28.17 | 593.32 | 547, 447, 429, 363, 327, 284, 255, 227, 211, 151, 107 | Isomer of kaempferol neohesperidoside | Sanchez-Rabaneda et al., 2003 [51] |
| 11 | 28.80 | 623.33 | 445, 357, 315, 299, 285, 271, 255, 243, 227, 150 | Isorhamnetin-3-O rutinoside | Sanchez-Rabaneda et al., 2003 [51] |
| 12 | 29.90 | 477.31 | 314, 229, 285, 271, 257, 243, 227, 214, 199 | Isorhamnetin glucoside | Harbaum et al., 2007 [49] |
| 13 | 30.90 | 579.27 | 447, 314, 299, 285, 271, 257, 243, 227, 187, 151 | Isorhamnetin glucoside with 2 arabinose/xylose moieties | Sanchez-Rabaneda et al., 2003 [51] |
|
C. gynandra extract |
|||||
| 14 | 2.30 | 191.02 | 173, 111, 87, 85, 67, 57 | Citric acid | Lacine et al., 2013 [52] |
| 15 | 5.55 | 371.06 | 209, 191, 179, 173, 85 | Caffeoylglucaric acid | Chen et al., 2014 [53] |
| 16 | 7.40 | 339.07 | 225, 203, 177, 133 | Esculetin glucoside | Simigiortis et al., 2015 [54] |
| 17 | 8.13 | 297.06 | 179, 161, 135, 117, 89 | Derivative of caffeic acid | Fang et al., 2002 [55] |
| 18 | 9.63 | 369.05 | 207, 189, 127, 99, 83 | Dimetoxycinnamic glucoside | Li et at., 2016 [56] |
| 19 | 9.92 | 179.03 | 135, 117, 107, 89 | Caffeic acid | Chen et al., 2014 [53] |
| 20 | 11.83 | 353.04 | 207, 189, 127, 119, 99, 83 | Dimethoxycinnamoyl coumaric acid | Li et al., 2016 [56] |
| 21 | 12.84 | 383.06 | 207, 189, 127, 99, 83 | Dimethoxycinnamoyl glucoronide | Li et al., 2016 [56] |
| 22 | 13.80 | 609.15 | 343, 301, 179, 151 | Quercetin rutinoside | Lacine et al., 2013 [52] |
| 23 | 14.74 | 593.15 | 327, 285, 151 | Kaempferol rutinoside | Lacine et al., 2013 [52] |
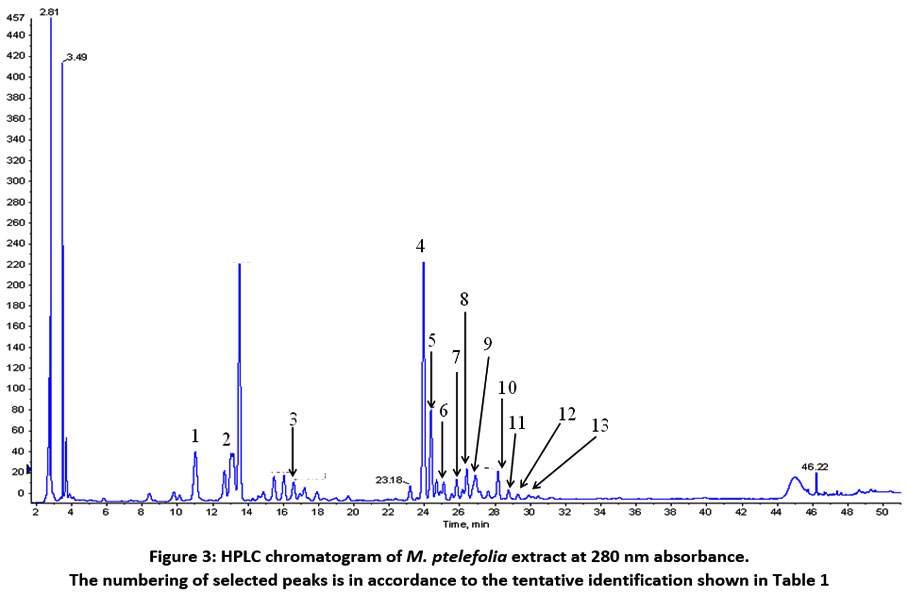 |
Figure 3: HPLC chromatogram of M. ptelefolia extract at 280 nm absorbance. The numbering of selected peaks is in accordance to the tentative identification shown in Table 1. |
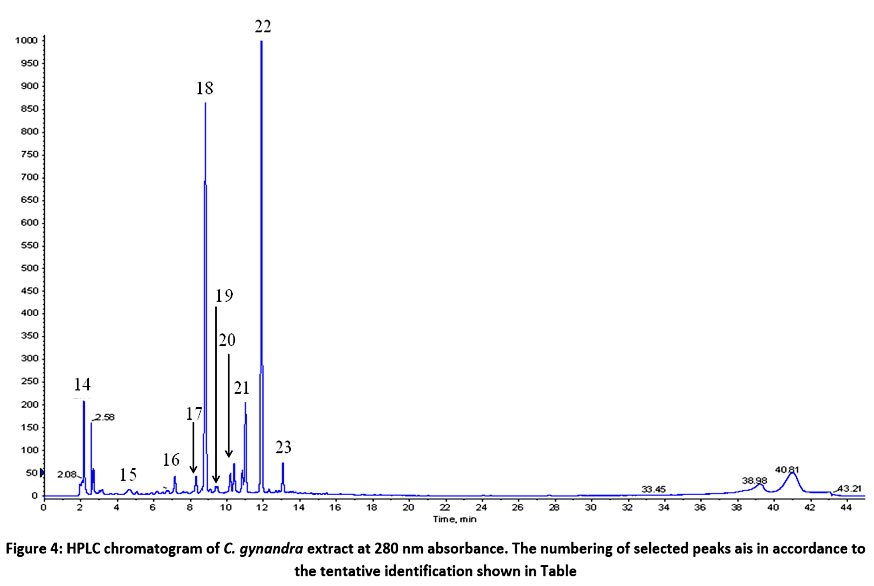 |
Figure 4: HPLC chromatogram of C. gynandra extract at 280 nm absorbance. The numbering of selected peaks ais in accordance to the tentative identification shown in Table 1. |
Discssion
The present study provides a comprehensive evaluation of the cytotoxic and anti-inflammatory properties of C. gynandra, M. ptelefolia, and their herbal formulations, with results supported by phytochemical profiling. The cytotoxicity assays confirmed that at concentrations ranging from 0.25 to 2 mg/mL, the extracts and formulations are safe for RAW 264.7 macrophages, aligning with prior research indicating the safety of herbal extracts at moderate doses. Notably, C. gynandra demonstrated higher cell viability across tested concentrations compared to M. ptelefolia, which suggests that C. gynandra contains fewer cytotoxic constituents or that its bioactive compounds are less damaging to macrophage cells. These findings are consistent with earlier reports where flavonoids such as quercetin and phenolic acids like caffeic acid, prevalent in C. gynandra, are known to exert anti-inflammatory effects without significant cytotoxicity at moderate doses.
The observed decrease in cell viability at higher concentrations (>4 mg/mL for C. gynandra and >1 mg/mL for M. ptelefolia) can be attributed to the high content of bioactive phytochemicals, particularly flavonoids and alkaloids, which at elevated doses can induce oxidative stress, cell membrane disruption, or apoptosis. Anbazhagi et al.7 indicated that flavonoids, while generally beneficial, can exert cytotoxic effects at high concentrations, possibly due to pro-oxidant activity. Similarly, Kabir et al.36 reported that alkaloids in M. ptelefolia could cause cell hypotonicity and shrinkage, leading to cell death, especially at higher doses. These findings reinforce the importance of dose optimization in herbal medicine research to maximize therapeutic efficacy while minimizing toxicity37.
The anti-inflammatory activity assessed via NO inhibition revealed that C. gynandra possesses a more potent effect compared to M. ptelefolia, especially at lower concentrations (0.5 mg/mL). Although none of the extracts or formulations showed statistically significant inhibition compared to diclofenac sodium, the trend suggests that C. gynandra‘s phytochemicals are more effective in modulating macrophage inflammatory responses38. This aligns with phytochemical profiling that identified key bioactive compounds such as quercetin rutinoside (rutin) and hydroxycinnamic acids, both well-documented for their anti-inflammatory mechanisms—namely, downregulation of iNOS, suppression of pro-inflammatory cytokines, and inhibition of nuclear factor-kappa B (NF-κB) activation.39
The LC-MS/MS analysis provided critical insights into the phytochemical composition underlying these activities. In M. ptelefolia, flavonoids like kaempferol neohesperidoside and its derivatives, conjugated with sugars such as rhamnose and arabinose, are known to inhibit nitric oxide synthase activity. Kaempferol, in particular, has been shown to significantly reduce iNOS expression, thereby decreasing NO production, a key mediator in inflammatory processes. The detection of isorhamnetin derivatives further supports anti-inflammatory potential, as these compounds are also recognized for their ability to modulate inflammatory signaling pathways.40 These phytochmicals maybe nanoencapsulated to improve their efficacy. 41-43
In C. gynandra, the presence of quercetin rutinoside (rutin) and kaempferol rutinoside, along with phenolic acids like caffeic acid and its derivatives, underscores its anti-inflammatory capacity. Quercetin rutinoside has demonstrated efficacy in attenuating pro-inflammatory mediators by inhibiting enzymes such as COX-2 and inducible nitric oxide synthase, and suppressing cytokines like TNF-α and IL-643,44. Hydroxycinnamic acids like caffeic acid are potent antioxidants and have been shown to inhibit NF-κB activation, thus reducing inflammation. These phytochemicals may act synergistically to produce anti-inflammatory effects, although the exact interactions require further elucidation.
Despite the promising individual activities, the combination of C. gynandra and M. ptelefolia in herbal formulations did not produce synergistic effects in NO inhibition. Instead, the interaction pattern was predominantly additive at the higher concentration tested (2 mg/mL), implying that the combined effects simply sum up without amplification. At lower concentrations (0.25–1 mg/mL), the interactions shifted toward antagonism, where the formulations exhibited less NO inhibition than the individual extracts. This phenomenon can be explained by phytochemical interactions, where certain compounds may compete for the same targets or interfere with each other’s absorption, distribution, or cellular activity. For example, flavonoids and alkaloids could potentially inhibit each other’s binding to inflammatory mediators or modulate enzyme activity in opposing ways.
The lack of observed synergism underscores the complexity of herbal interactions and the importance of ratio optimization. While traditional medicine often relies on synergistic effects, modern scientific validation indicates that not all combinations produce enhanced efficacy. The phytochemical profile suggests that specific compounds like quercetin rutinoside and kaempferol derivatives are primarily responsible for anti-inflammatory effects; however, their interactions within complex mixtures can vary depending on concentration, extraction method, and the presence of other constituents.
Furthermore, this study highlights the necessity for detailed mechanistic studies to understand how phytochemicals interact at molecular levels. For instance, future investigations could examine the effects of these extracts on key inflammatory signaling pathways such as NF-κB, MAPK, or STAT, and explore their effects on cytokine profiles beyond NO, including TNF-α, IL-1β, and IL-645. Additionally, exploring different ratios, extraction techniques, or formulations (e.g., nanoemulsions, encapsulation) might reveal conditions under which synergistic effects become apparent.46-47
Conclusion
This study assessed the cytotoxicity and anti-inflammatory potential of Cleome gynandra and Melicope ptelefolia leaf extracts, both individually and in combination using RAW 264.7 macrophage cells. Among the two, C. gynandra exhibited higher cell viability and lower cytotoxicity, indicating its greater safety profile. In nitric oxide (NO) inhibition assays, C. gynandra showed moderate activity, while M. ptelefolia and their combinations demonstrated limited inhibitory effects, particularly at lower concentrations.
LC-ESI-MS/MS metabolite profiling revealed several bioactive compounds in both extracts. C. gynandra was rich in quercetin, kaempferol, and gallic acid, all known for their potent anti-inflammatory and antioxidant activities through modulation of nitric oxide synthase and suppression of pro-inflammatory cytokines. M. ptelefolia primarily contained scopoletin, auraptene, and rutin, which are associated with inhibition of COX-2 and inflammatory mediators. The presence of these compounds supports the traditional use of both plants in treating inflammatory conditions.
However, the combination of C. gynandra and M. ptelefolia at the tested ratios (especially 70:30 w/w) did not show a synergistic improvement in anti-inflammatory activity, highlighting the complexity of herb-herb interactions. These findings underscore the need to better understand phytochemical compatibility and optimize formulation ratios in herbal product development.
In conclusion, C. gynandra demonstrates greater potential as a standalone anti-inflammatory agent compared to M. ptelefolia, primarily due to its richer content of flavonoids and phenolic acids. Future research should focus on refining extract ratios, exploring detailed molecular mechanisms of action, and validating these effects through in vivo studies to support the development of safe and effective plant-based anti-inflammatory therapeutics.
Acknowledgement
The authors would like to thank the Malaysian Agricultural Research and Development Institute (MARDI) and University Putra Malaysia for facilitating this research.
Funding Source
This study was supported by the Malaysian Agricultural Research and Development Institute (MARDI) beneath Government Development RMK-11 Fund (Grant No: P-RB403-1001).
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Author Contribution’s
- Muhammad Aiman Haiqal bin Ismail – original draft, Formal analysis, Data curation.
- Koh Soo Peng– Supervision, Formal analysis, Funding acquisition, Conceptualization.
- Sanimah Simoh – LC-ESI-MS/MS Profiling.
- Armania Nurdin – Supervision, Formal analysis.
- Machap Chandradevan – LC-ESI-MS/MS Profiling.
- Muhammad Nazrul Hakim – Supervision, Formal analysis, Project administration.
References
- Narendhirakannan RT, Subramanian S, Kandaswamy M. Anti-inflammatory and lysosomal stability actions of Cleome gynandra L. studied in adjuvant induced arthritic rats. Food Chem Toxicol. 2007; 45(6): 1001-1012. https://doi.org/10.1016/j.fct.2006.12.009
CrossRef - Khuntia A, Martorell M, Ilango K, et al. Theoretical evaluation of Cleome species’ bioactive compounds and therapeutic potential: A literature review. Biomed Pharmacother. 2022; 151: 113161. https://doi.org/10.1016/j.biopha.2022.113161 .
CrossRef - Mashamaite CV, Manyevere A, Chakauya E. Cleome gynandra: A wonder climate-smart plant for nutritional security for millions in semi-arid areas. Front Plant Sci. 2022; 13:1003080. https://doi.org/10.3389/fpls.2022.1003080
CrossRef - Mohammad Rosdi MN, Awang MA, Abu Bakar MH. A perspective on anti-inflammatory properties of Cleome gynandra. Food Res. 2025; 9: 153-158. https://doi.org/10.26656/fr.2017.9(1).212
CrossRef - Chatterjee A, Pakrash SC. The Treatise on Indian Medicinal Plants. 1991; Vol. 3. National Inst. Of Science Communication and Information Resources.
- Nirosa T, Sravanthi K, Brahma RR. Evaluation of antidiabetic activity of Cleome gynandra leaves. Indo Am J Pharmaceu Sci. 2016; 3(5): 482–486.[Google Scholar]
- Anbazhagi T, Kadavul K, Suguna G, Petrus AJA. Studies on the pharmacognostical and in vitro antioxidant potential of Cleome gynandra Linn. leaves. Nat Prod Radiance. 2009. 8: 151–157. [Google Scholar]
- Bala A, Kar B, Haldar PK, Mazumder UK, Bera S. Evaluation of anticancer activity of Cleome gynandra on Ehrlich’s Ascites Carcinoma treated mice. J Ethnopharmacol. 2010; 129: 131–134. https://doi.org/10.1016/j.jep.2010.03.010
CrossRef - Sowunmi LI, Afolayan AJ. Phytochemical constituents and antioxidant properties of acetone extract of Cleome gynandra (L.) growing in the Eastern Cape, South Africa. Com Alt Med. 2015;12(3): 1-8. https://doi.org/10.21010/ajtcam.v12i3.1
CrossRef - Agarwal A. Current issues in quality control of natural products. Pharma Times. 2005; 39(6): 9-11.
- Rukayah A. Tumbuhan liar berkhasiat ubatan. 1st ed. Kuala Lumpur: Dewan Bahasa dan Pustaka. 2006; 1:1-118.
- Abas F, Shaari K, Israf DA, Syafri S, Zainal Z, Lajis NH. LC–DAD–ESI-MS analysis of nitric oxide inhibitory fractions of Tenggek burung (Melicope ptelefolia Champ. ex Benth.). J Food Compos Analysis. 2010; 23: 107–112. https://doi.org/10.1016/j.jfca.2009.03.009
CrossRef - Loi DT. Glossary of Vietnamese medicinal plants. Science and Technics Publication. 1977; 1:145.
CrossRef - Chong YJ, Musa NF, Ng CH, et al. Barrier protective effects of 2, 4, 6-trihydroxy-3-geranyl acetophenone on lipopolysaccharides-stimulated inflammatory responses in human umbilical vein endothelial cells. J Ethnopharmacol. 2016192: 248-255. https://doi.org/10.1016/j.jep.2016.07.032
CrossRef - Sulaiman MR, Mohd Padzil A, Shaari K, et al. Antinociceptive activity of Melicope ptelefolia ethanolic extract in experimental animals. J Biomed Biotech. 2010; 2010: 1-6. https://doi.org/10.1155/2010/937642
CrossRef - Johnson AJ, Kumar AR, Sherin A, et al. Antipyretic, analgesic, anti-inflammatory and antioxidant activities of two major chromenes from Melicope lunu-ankenda. J Ethnopharmacol. 2010; 130: 267-71. https://doi.org/10.1016/j.jep.2010.05.003
CrossRef - Mahadi M, Rahman NA, Viswanathan D, et al. The potential effects of Melicope ptelefolia root extract as an anti-nociceptive and anti-inflammatory on animal models. Bull Fac Pharmacy, Cairo University. 2016; 54(2): 237–41. https://doi.org/10.1016/j.bfopcu.2016.06.005
CrossRef - Nguyen NH, Ha TK, Choi S, et al. Chemical constituents from Melicope ptelefolia leaves. Phytochem. 2016; 130: 291-300. https://doi.org/10.1016/j.phytochem.2016.06.011
CrossRef - Nathan C. Points of control in inflammation. Nature. 2002; 420(6917): 846-852. https://doi.org/10.1038/nature01320
CrossRef - Liu X, Yin S, Chen Y, et al. LPS‑induced pro-inflammatory cytokine expression in human airway epithelial cells and macrophages via NF‑κB, STAT3 or AP‑1 activation. Mol Med Rep. 2018; 17(4): 5484-5491. https://doi.org/10.3892/mmr.2018.8542
CrossRef - Punchard NA, Whelan CJ, Adcock I. The journal of inflammation. Biomed Central. 2004; 1: 1-4. https://doi.org/10.1186/1476-9255-1-1
CrossRef - Wongrakpanich S, Wongrakpanich A, Melhado K, Rangaswami J. A comprehensive review of non-steroidal anti-inflammatory drug use in the elderly. Aging Dis. 2018; 9(1): 143. https://doi.org/10.14336/AD.2017.0306
CrossRef - Somchit N, Sanat F, Gan EH, Shahrin IW, Zuraini A. Liver injury induced by the non-steroidal anti-inflammatory drug mefenamic acid. Singapore Med J. 2004; 45(11): 530-2. PMID: 15510325.
- Hussein Al Ali SH, Al-Qubaisi M, Hussein MZ, et al. Controlled release and angiotensin-converting enzyme inhibition properties of an antihypertensive drug based on a perindopril erbumine-layered double hydroxide nanocomposite. Int J Nanomed. 2012; 7: 2129–2141. https://doi.org/10.2147/IJN.S30461
CrossRef - Somchit MN, Sanat F, Gan EH, Shahrin IW, Zuraini A. Mefenamic Acid Induced Nephrotoxicity: An Animal Model. Ad Pharmaceu Bull. 2014; 4(4): 401–404. DOI: 10.5681/apb.2014.059
- Chandradevan M, Simoh S, Mediani A, Ismail IS, Abas F. 1 H NMR-Based Metabolomics Approach in Investigating the Chemical Profile, Antioxidant and Anti-Inflammatory Activities of Gynura procumbens and Cleome gynandra. Plant Foods Hu Nut. 2020; 2020: 1-9. https://doi.org/10.1007/s11130-020-00805-3
CrossRef - Parasuraman S, Thing GS, Dhanaraj SA. Polyherbal formulation: Concept of ayurveda. Pharmacognosy Rev, 2014; 8(16): 73. https://doi.org/10.4103/0973-7847.134229
CrossRef - Locatelli M, Governatori L, Carlucci G, et al. Recent application of analytical methods to phase I and phase II drugs development: a review. Biomed Chromatography. 2012; 26(3): 283– 300. https://doi.org/10.1002/bmc.1674
CrossRef - Do QD, Angkawijaya AE, Tran-Nguyen PL, et al. Effect of extraction solvent on total phenol content, total flavonoid content, and antioxidant activity of Limnophila aromatic. J Food Drug Anal. 2014; 22(3): 296–302. https://doi.org/10.1016/j.jfda.2013.11.001
CrossRef - Chandradevan M, Simoh S, Mediani A, et al. UHPLC-ESI-Orbitrap-MS Analysis of Biologically Active Extracts from Gynura procumbens (Lour.) Merr. and Cleome gynandra L. Leaves. Evi Com Alt Med. 2020; 2020: 1-14. https://doi.org/10.1155/2020/3238561
CrossRef - Kusmayadi A, Adriani L, Abun A, Muchtaridi M, Tanuwiria UH. The microencapsulation of mangosteen peel extract with maltodextrin from arenga starch: formulation and characterisation. J App Pharmaceu Sc. 2019; 9(03): 033-040. https://doi.org/10.7324/JAPS.2019.90306
CrossRef - Chung HJ, Lee J, Shin JS, et al. In vitro and in vivo anti-allergic and anti-inflammatory effects of eBV, a newly developed derivative of bee venom, through modulation of IRF3 signaling pathway in a carrageenan-induced edema model. PloS One. 2016; 11(12): e0168120. https://doi.org/10.1371/journal.pone.0168120
CrossRef - Chandradevan M, Indu Bala J. High throughput analysis on selected polyphenol production and principal component analysis (PCA) in Phyllanthus watsonii grown under different environmental condition. J Trop Agri Food Sci. 2014; 42(2): 157-167.
- Wishart DS, Feunang YD, Marcu A, et al. HMDB 4.0: the human metabolome database for 2018. Nuc Acids Res. 2008; 46: 608-617. https://doi.org/10.1093/nar/gkx1089
CrossRef - National Center for Biotechnology Information, U.S. National Library of Medicine. Rockville Pike, MD 2018. Available at https://www.ncbi.nlm.nih.gov. Accessed on 1st September 2024.
- Kabir MF, Ali JM, Abolmaesoomi M, Hashim OH. Melicope ptelefolia leaf extracts exhibit antioxidant activity and exert anti-proliferative effect with apoptosis induction on four different cancer cell lines. BMC Com Alt Med. 2017; 17(1): 1-18. https://doi.org/10.1186/s12906-017-1761-9
CrossRef - Tungmunnithum D, Thongboonyou A, Pholboon A, Yangsabai A. Flavonoids and other phenolic compounds from medicinal plants for pharmaceutical and medical aspects: An overview. Med. 2018; 5(3): 93. https://doi.org/10.3390/medicines5030093
CrossRef - Nemudzivhadi V, Masoko P. In vitro assessment of cytotoxicity, antioxidant, and anti-inflammatory activities of Ricinus communis (Euphorbiaceae) leaf extracts. Evi-Based Com Alt Med. 2014; 2014: 1-8. https://doi.org/10.1155/2014/625961
CrossRef - Oh YC, Cho WK, Oh JH, et al. Fermentation by Lactobacillus enhances anti-inflammatory effect of Oyaksungisan on LPS-stimulated RAW 264.7 mouse macrophage cells. BMC Com Alt Med. 2012; 12(1):17. https://doi.org/10.1186/1472-6882-12-17
CrossRef - Liu A, Ding S. Anti-inflammatory Effects of Dopamine in Lipopolysaccharide (LPS)-stimulated RAW264.7 Cells via Inhibiting NLRP3 Inflammasome Activation. Ann Clin Lab Sci. 2019; 49(3):353-360.
- Salman MF, Al-Mudallal NHAL, Ahmed ME. Cytotoxic Effect of Biogenic Selenium Nanoparticles Using Bacteriocin of Acinetobacter baumannii Isolated from Burns and Wound Infections. (2025). Iraqi J Sci, 66(4), 1535-1549. https://doi.org/10.24996/ijs.2025.66.4.13
CrossRef - Ahmed ME, Alzahrani KK, Fahmy NM, et al. Colistin-Conjugated Selenium Nanoparticles: A Dual-Action Strategy Against Drug-Resistant Infections and Cancer. Pharmaceutics. 2025; 17(5):556. https://doi.org/10.3390/pharmaceutics17050556
CrossRef - Goh JZ, Tang SN, Chiong HS, et al. Evaluation of antinociceptive activity of nanoliposome-encapsulated and free-form diclofenac in rats and mice. Int J Nanomedicine. 2014; 10: 297-303. doi: 10.2147/IJN.S75545.
CrossRef - Chiong HS, Yong YK, Hijaz MSM, et al. Improved Anti-nociceptive, Anti-pyretic and Anti-inflammatory Effects of Orally Administered Liposome-encapsulated Piroxicam. Biomed Pharmacol J. 2024;17(2): 795-811. DOI : https://dx.doi.org/10.13005/bpj/2905
CrossRef - Pang Z, Wang G, Ran N, et al. Inhibitory effect of methotrexate on rheumatoid arthritis inflammation and comprehensive metabolomics analysis using ultra-performance liquid chromatography-quadrupole time of flight-mass spectrometry (UPLC-Q/TOF-MS). Intl J Mol Sc. 2018; 19(10): 2894. https://doi.org/10.3390/ijms19102894
CrossRef - Anyasor GN, Okanlawon AA, Ogunbiyi B. Evaluation of anti-inflammatory activity of Justicia secunda Vahl leaf extract using in vitro and in vivo inflammation models. Clin Phytosci. 2019; 5(1): 49. https://doi.org/10.1186/s40816-019-0137-8
CrossRef - Djuichou Nguemnang SF, Tsafack EG, Mbiantcha M, et al. In Vitro Anti-Inflammatory and In Vivo Antiarthritic Activities of Aqueous and Ethanolic Extracts of Dissotis thollonii Cogn. (Melastomataceae) in Rats. Evi-Based Com Alt Med. 2019; 2019: 1-17. https://doi.org/10.1155/2019/3612481
CrossRef - Msika P, Saunois A, Baudouin C, Leclere-Bienfait S, Debrock S. Extract of the above-ground portions of Gynandropsis gynandra or Cleome gynandra, and cosmetic, dermatological or pharmaceutical compositions. Washington, DC: U.S. Patent and Trademark Office. 2016; (9) 370-541.
- Harbaum B, Hubbermann EM, Wolff C, et al. Identification of flavanoids and hydroxycinnamic acids in pak choi varieties (Brassica campestris ssp. chinensis var. communis) by HPLC-ESI-MSn and NMR and their quantification by HPLC-DAD. J Agri Food Chem. 2007; 55(20): 8251-8260. https://doi.org/10.1021/jf071314+
CrossRef - Nuengchamnong N. Ingkaninan K. On-line characterization of phenolic antioxidants in fruit wines from family myrtaceae by liquid chromatography combined with electrospray ionization tandem mass spectrometry and radical scavenging detection. LWT – Food Sci Tech. 2009; 42(1): 297-302. https://doi.org/10.1016/j.lwt.2008.04.012
CrossRef - Sanchez-Rabaneda F, Jauregui O, Casals I, et al. Liquid chromatography/electrospray ionization tandem mass spectrometric study of the phenolic composition of cocoa (Theobroma cacao). J Mass Spectro. 2003; 38(1): 35-42. https://doi.org/10.1002/jms.395
CrossRef - Lacine A, Erdi K, Yasin A, Zeyneb A, Mustafa K. Free radical scavenging activity, total phenolic content, total antioxidant status, and total oxidant status of endemic Thermopsis turcica. Saudi J Biol Sci. 2013; 3:235-239. https://doi.org/10.1016/j.sjbs.2013.02.003
CrossRef - Chen F, Long X, Liu Z, Shao H, Liu L. Analysis of phenolic acids of Jurusalem artichoke (Helianthus tuberosus) responding to salt-stress by liquid chromatography/tandem mass spectrometry. Scientific World J. 2014; 1-8. https://doi.org/10.1155/2014/568043
CrossRef - Simirgiotis MJ, Benites J, Areche C, Sepulveda B. Antioxidant capacities and analysis of phenolic compounds in three endemic Nolana species by HPLC-PDA-ESI-MS. Molecules. 2015; 20: 11490-11507. https://doi.org/10.3390/molecules200611490
CrossRef - Fang N, Yu S, Prior RL. LC/MS/MS characterization of phenolic constituents in dried plums. J Agri Food Chem. 2002; 50: 3579-3585. https://doi.org/10.1021/jf0201327
CrossRef - Li S, Lin Z, Jiang H, et al. Rapid identification and assignation of the active ingredients in Fufang Banbianlian injection using HPLC-DAD-ESI-IT-TOF-MS. J Chromato Sci. 2016; 54(7): 1225-1237. https://doi.org/10.1093/chromsci/bmw055
CrossRef - Garcia-Mediavilla V, Creapo I, Collado PS, et al. The anti-inflammatory flavones quercetin and kaempferol cause inhibition of inducible nitric oxide synthase, cyclooxygenase-2 and reactive C-protein, and down-refulation of the nuclear factor kappaB pathway in Chang Liver cells. Eur J Pharmacol. 2007; 557:221-229. https://doi.org/10.1016/j.ejphar.2006.11.014
CrossRef - Lee SJ, Lee SY, Ha HJ, et al. Rutin attenuates lipopolysaccharide-induced nitric oxide production in macrophage cells. J Food Nutri Res. 2015; 3(3): 202-205. https://doi.org/10.12691/jfnr-3-3-12
CrossRef - Gunasekaran S, Venkatachalam K, Namasivayam N. Anti-inflammatory and anticancer effect of p-methoxycinnamic acid, an active phenylpropanoic against 1,2-dimethylhydrazine-induced rat colon carcinogenesis. Mol Cell Biochem. 2019; 451: 117-129. https://doi.org/10.1007/s11010-018-3398-5
CrossRef - Zhou X, Seto SW, Chang D, et al. Synergistic effects of Chinese herbal medicine: a comprehensive review of methodology and current research. Front Pharmacol. 2016; 7: 1-16. https://doi.org/10.3389/fphar.2016.00201
CrossRef - Roell KR, Reif DM, Motsinger-Reif AA. An introduction of terminology and methodology of chemical synergy-perspectives from across disciplines. Front Pharmacol. 2017; 8: 1-11. https://doi.org/10.3389/fphar.2017.00158
CrossRef







