Manuscript accepted on :05-09-2025
Published online on: 30-09-2025
Plagiarism Check: Yes
Reviewed by: Dr. Amit Gupta and Dr. Shoheb Shaikh
Second Review by: Dr. Mihaela Ionescu
Final Approval by: Dr. Mariia Shanaida
Nazeerullah Rahamathullah1,2 , Sovan Bagchi1
, Sovan Bagchi1 , Manisha Nanda3,4
, Manisha Nanda3,4 and Abhaya Dutta5*
and Abhaya Dutta5*
1Department of Biomedical Sciences, College of Medicine, Gulf Medical University, Ajman, UAE.
2Thumbay Research Institute for Precision Medicine, Gulf Medical University, Ajman, UAE.
3Department of Biotechnology, Dolphin Institute of Biomedical and Natural Sciences, Dehradun, India.
4Department of Microbiology, Graphic Era University, Dehradun, India.
5Department of Physiology, Vidyasagar College for Women, Kolkata, India.
Corresponding Author E-mail: duttaabhaya8@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3251
Abstract
Catharanthus roseus, which is commonly called Nayantara, is a member of the Apocynaceae family. Traditional medicine has long used it to treat a variety of conditions, including microbial infections. This study aimed to evaluate the antibacterial qualities of C. roseus leaf extracts against a variety of common microorganisms. Using a conventional procedure, C. roseus leaf extracts were made with ethanol, methanol, and water. A qualitative phytochemical analysis was then conducted. Extracts from the leaves of C. roseus were examined for their antibacterial properties against Gram-positive bacteria, Staphylococcus aureus, and Gram-negative bacteria: Pseudomonas aeruginosa, Salmonella typhi, and Escherichia coli. Alkaloids and phenolic compounds, among other medicinal substances, were detected in the C. roseus leaf extracts according to a phytochemical test. All three extracts exhibited the maximum antibacterial activity against E. coli. The lowest antibacterial activity was noted in the ethanolic, methanolic, and aqueous extracts of C. roseus against P. aeruginosa, S. typhi, and S. aureus, respectively. The leaf of C. roseus has great promise as a source of bioactive chemicals with considerable health advantages, even though it is a byproduct. These findings open doors to customized extraction methods and the exploration of other parts of C. roseus for broader health applications, offering a sustainable pathway for drug discovery that harnesses natural resources to promote both global health and environmental well-being.
Keywords
Antibacterial activity; Bioactive compounds; Catharanthus roseus; Microbial infections; Phytochemicals; Traditional medicine
Download this article as:| Copy the following to cite this article: Rahamathullah N, Bagchi S, Nanda M, Dutta A. In Vitro Antibacterial Efficacy of Crude Leaf Extracts from Catharanthus roseus: A Natural Therapeutic Prospect. Biomed Pharmacol J 2025;18(3). |
| Copy the following to cite this URL: Rahamathullah N, Bagchi S, Nanda M, Dutta A. In Vitro Antibacterial Efficacy of Crude Leaf Extracts from Catharanthus roseus: A Natural Therapeutic Prospect. Biomed Pharmacol J 2025;18(3). |
Introduction
Antimicrobial resistance (AMR) has emerged as a major public health challenge. The rise of multidrug-resistant (MDR) bacteria is rendering many antimicrobial therapies ineffective, leading to prolonged hospitalizations and higher mortality rates.¹˒² For centuries, herbs and medicinal plants have been utilized for their therapeutic properties. Compared to synthetic drugs, these natural remedies are often associated with fewer side effects and greater efficacy. Their diverse molecular structures and biological activities play a crucial role in the development of novel therapeutic agents.3
Apocynaceae family member Nayantara, scientifically known as C. roseus, is a perennial shrub of medicinal importance. It is cultivated primarily for its valuable alkaloid compounds, which exhibit significant anticancer and antidiabetic activities.4-6 Several studies have reported the antibacterial potential of crude extracts from different parts of C. roseus, including leaves, stems, roots, and flowers, against clinically significant single or multidrug-resistant bacteria.7,8 The leaf extracts of C. roseus have been shown to be effective against several bacterial pathogens.9,10 Additionally, leaf extracts are noted for their antihypertensive and antihyperlipidemic activity.11,12 Furthermore, anti-Alzheimer’s activity has also been shown by inhibiting the acetylcholine esterase enzyme and increasing cerebral blood flow by its constituent alkaloids, hence showing immense neuroprotective potential.13,14 Even though these extracts appear to have potential health advantages, more study is required to completely comprehend their effects and determine the best ways to apply them in clinical settings.
The entire plant is commonly utilized for alkaloid extraction, but it also comprises numerous other chemical constituents, such as tannins, phenolic compounds, steroids, and flavonoids, all of which possess therapeutic applications. Due to their polar nature, these compounds necessitate specific procedures and solvents for extraction, typically water or water-alcohol mixtures.
The efficacy of plant extracts is primarily determined by the concentration and synergistic interactions of their bioactive compounds, both of which are significantly affected by the selection of extraction solvent, as it influences the chemical composition and overall biological activity of the resultant extract.15,16 The current study aimed to investigate the phytochemical composition and antibacterial activity of C. roseus leaf extracts using three different solvents against S. aureus, E. coli, S. typhi, and P. aeruginosa. This study also highlights the importance of using the appropriate solvent to ensure that the C. roseus extract works effectively.
Materials and Methods
Processing and Extraction of Plant Leaves
Fresh leaves of C. roseus were harvested, manually sorted, and left to wilt at room temperature for 15 days. After being cleaned with tap water, the leaves were rinsed with distilled water and allowed to dry. The dried leaves were then mechanically ground to obtain a fine powder. A total of 15 grams of powdered leaves was loaded into a Soxhlet apparatus with 300 mL of solvent. Extractions were performed separately using ethanol, methanol, and distilled water. The Soxhlet extraction was carried out at 65 °C for 24 hours.17 It was noted that all three extracts had a dark green hue, sticky in consistency, and semi-solid in appearance.
Preliminary Phytochemical Screening
Qualitative Detection of Alkaloids
A few milliliters of diluted HCl were added to 50 milligrams of the solvent-free extract, and the mixture was filtered. The resulting filtrate was then analyzed using a variety of alkaloidal reagents, including Mayer’s, Wagner’s, and Hager’s tests for alkaloid detection.18,19
Test for Carbohydrates
Benedict’s qualitative test, Fehling’s test, and Molisch’s test were performed to detect carbohydrates.20
Test for Proteins and Amino Acids
A 100 milligrams of the extract was dissolved in 10 mL of distilled water and filtered using Whatman filter paper. The resulting filtrate was then subjected to protein and amino acid analysis, including the Ninhydrin test for amino acid detection.21
Test for Sterols and Triterpenoids
Sterols and triterpenoids were detected using the sulfur powder test and Salkowski’s assay.22
Test for Tannins and Phenol Compounds
The ferric chloride test was used to identify the presence of phenolic compounds.23
Antimicrobial Activity Test
Four bacterial cultures obtained from the Microbial Type Culture Collection (MTCC), each with distinct characteristics and identification numbers, were used in the study. E. coli was designated as MTCC#43, S. aureus as MTCC#87, P. aeruginosa as MTCC#2488, and S. typhi as MTCC#96. To evaluate antibacterial activity, a loopful of each culture was inoculated into sterile nutrient broth and incubated overnight at 37 °C.
Preparation of Culture Media
For antimicrobial testing, 38 grams of Muller-Hinton agar (MHA) in 1000 mL was sterilized for 15–30 minutes at 121°C.
Concentration of Extract
Stock extract concentrations were prepared in dilutions of 100 mg/mL, 200 mg/mL, and 300 mg/mL in ethanol, methanol, and water.
Use of Antibiotics
Penicillin (10 U), Tetracycline (10 µg), and Ampicillin (25 µg) were used to identify standard zones of inhibition. Antibiotic discs (3 mm) were placed on MHA agar and incubated at 37°C for 24 hours, and zones of inhibition were noted.
Antimicrobial Activity via Well Diffusion Method
The agar well diffusion method was employed for the assay. Bacterial strains were cultured in nutrient broth for 24 hours, after which 100 μL of the cell suspension was spread onto Mueller–Hinton Agar (MHA) plates. Wells with a diameter of 9 mm were filled with 200 μL of the respective extracts: 100 mg/mL ethanol extract, 200 mg/mL methanol extract, and 300 mg/mL aqueous extract, which also served as controls. Following incubation at 37 °C for 24 hours, the plates were examined for zones of inhibition.22
Minimum Inhibitory Concentration and Minimum Bactericidal Concentration
The antibacterial activity of C. roseus leaf extracts prepared in ethanol, methanol, and water against E. coli, S. typhi, P. aeruginosa, and S. aureus was evaluated using the broth dilution method. Except for the first well, each well of the microtiter plate was filled with 95 μL of Mueller–Hinton Broth (MHB) and 5.0 μL of bacterial inoculum, with each column assigned to a different bacterium. A 100μL aliquot of the test extract was added to the first and second wells, followed by serial dilution from the second to the eleventh well, using the first and twelfth wells as controls. After incubation at 37 °C for 16–18 hours, 20 μL of resazurin dye was added to each well and incubated for an additional 2–3 hours to assess bacterial growth. For MBC determination, 50 μL from each MIC well was inoculated onto Mueller–Hinton Agar (MHA) plates using the lawn culture method and incubated at 37 °C for 24 hours. The lowest concentration showing no visible growth was recorded as the MBC.24-26
Results
Phytochemical Screening of C. roseus Leaf Extracts
The phytochemical profiles of the ethanol, methanol, and aqueous extracts of C. roseus leaves are presented in Table 1. Qualitative tests were performed to detect carbohydrates, proteins, amino acids, alkaloids, glycosides, saponins, tannins, phenolic compounds, and steroids. The presence (+) or absence (–) of each compound was recorded for each extract type.
Table 1: Phytochemical analysis of ethanol, methanol, and aqueous extracts of C. roseus leaves.
| Name of test | Ethanol | Methanol | Aqueous |
| 1. Alkaloidsa) Mayer’s test
b) Wagner’s test c) Hager’s test |
a) –ve
b) –ve c) +ve |
a) –ve
b) –ve c) +ve |
a) –ve
b) –ve c) +ve |
| 2. Carbohydrates and glycosidesa) Fehling’s test
b) Molisch’s test c) Benedict’s test |
a) -ve
b) +ve c) -ve |
a) -ve
b) +ve c) +ve |
a) -ve
b) +ve c) -ve |
| 3. Proteins and amino acidsNinhydrin test | +ve | +ve | +ve |
| 4. SaponinsFoam test | –ve | –ve | –ve |
| 5. Tannins & Phenol compoundsFerric chloride test | +ve
|
–ve
|
–ve
|
| 6. Steroidsa) Salkowasky’s test
b) Sulphur powder test |
a) +veb) +ve | a) +veb) +ve | a) +veb) +ve |
Screening of C. roseus leaf extracts for antibacterial activity
Each extract’s zone of inhibition (diameter in millimeters) was assessed and contrasted with that of common antibiotics such as ampicillin, tetracycline, and penicillin. The maximum inhibitory zone was observed with a concentration of 300 mg/mL of ethanolic extract against E. coli (23±1.02 mm) and S. typhi (21±1 mm). Similarly, the maximum inhibitory zone was observed against P. aeruginosa (22.3±1.52 mm) with the concentration of 300 mg/mL of aqueous extract. In the case of S. aureus, the maximum inhibitory zone was recorded (20.3±1.5 mm) with the concentration of 300 mg/mL of methanolic extract. The details of antibacterial activities of ethanol, methanol, and aqueous extracts against E. coli, P. aeruginosa, S. aureus, and S. typhi are given in Table 2 and Figure 1.
Screening of C. roseus Leaf Extracts for Antibacterial Activity
The antibacterial activity of each extract was evaluated by measuring the zone of inhibition (diameter in millimeters) and comparing it with standard antibiotics such as ampicillin, tetracycline, and penicillin. The largest inhibitory zones were observed with 300 mg/mL concentrations of the extracts: ethanolic extract showed maximum inhibition against E. coli (23 ± 1.02 mm) and S. typhi (21 ± 1 mm), the aqueous extract was most effective against P. aeruginosa (22.3 ± 1.52 mm), and the methanolic extract exhibited the highest inhibition against S. aureus (20.3 ± 1.5 mm). Detailed antibacterial activity data for ethanol, methanol, and aqueous extracts against E. coli, P. aeruginosa, S. aureus, and S. typhi are presented in Table 2 and Figure 1.
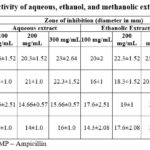 |
Table 2: Antibacterial activity of aqueous, ethanol, and methanolic extracts of C. roseus leaf.Click here to view Table |
aP – Penicillin, bTE – Tetracycline, cAMP – Ampicillin
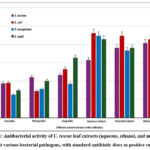 |
Figure 1: Antibacterial activity of C. roseus leaf extracts (aqueous, ethanol, and methanol) against various bacterial pathogens, with standard antibiotic discs as positive controls.Click here to view Figure |
In this study, following the initial assessment of antibacterial activity using the well diffusion method, the MIC and MBC of C. roseus leaf extracts were determined for the four bacterial strains via the broth dilution method (Table 3). For S. aureus and E. coli, the ethanolic extract exhibited MBCs of 50 and 55 mg/mL, respectively, while the MIC for both was 25 mg/mL. The same extract showed MICs of 45 and 55 mg/mL and MBCs of 65 and 75 mg/mL against P. aeruginosa and S. typhi, respectively. The methanolic extract demonstrated MICs and MBCs of 40 and 60 mg/mL for P. aeruginosa, 20 and 45 mg/mL for S. typhi, and 50 and 75 mg/mL for E. coli and S. aureus. The aqueous extract showed an MIC of 20 mg/mL and an MBC of 45 mg/mL against E. coli, P. aeruginosa, and S. typhi, while for S. aureus, the MIC and MBC were 40 mg/mL and 65 mg/mL, respectively (Figure 2). In all cases, the MBC values were higher than the MIC values, indicating that a higher concentration of extract is required to completely kill the bacteria compared to the amount needed to inhibit their growth.
Table 3: MIC and MBC of C. roseus leaf extracts prepared in ethanol, methanol, and aqueous solvents against various bacterial pathogens.
| Plant extract | Bacterial pathogens | |||||||
| S. aureus | E. coli | P. aeruginosa | S. typhi | |||||
| MIC(mg/mL) | MBC(mg/mL) | MIC(mg/mL) | MBC(mg/mL) | MIC(mg/mL) | MBC(mg/mL) | MIC(mg/mL) | MBC(mg/mL) | |
| Aqueous extract | 40 | 65 | 20 | 45 | 20 | 45 | 20 | 45 |
| Ethanolic extract | 50 | 75 | 50 | 75 | 40 | 60 | 20 | 45 |
| Methanolic extract | 25 | 55 | 25 | 50 | 45 | 65 | 55 | 75 |
 |
Figure 2: Radar plot showing the MIC and MBC of C. roseus leaf extracts against different bacterial pathogens.Click here to view Figure |
Discussion
The current study makes a substantial addition to the fields of phytotherapeutics and natural product science by advancing our knowledge of the antibacterial properties of C. roseus leaf extracts. Examining different extracts (ethanol, methanol, and aqueous) and their efficacy against the nosocomial pathogens like E. coli, S. typhi, P. aeruginosa, and S. aureus. The thorough chemical analysis of the C. roseus leaf extracts is a crucial component of this study. The diverse composition of these extracts is evidenced by the presence of a wide range of bioactive constituents, including alkaloids, phenols, glycosides, flavonoids, terpenoids, steroids, carbohydrates, proteins, and amino acids. These results highlight the possible therapeutic use of C. roseus extracts and are in line with other studies of a similar nature.14,27,28
The study demonstrates that the ethanolic extract of C. roseus leaves exhibits strong antibacterial activity, as evidenced by the largest zones of inhibition, which reflect its antimicrobial potency. This significant finding is in line with previous research findings and supports the potential of C. roseus leaf extracts as strong substitutes for traditional antibiotics.17,29,30 The bacteria P. aeruginosa, S. typhi, and E. coli are Gram-negative, while S. aureus has a Gram-positive cell wall structure and is naturally sensitive to the active ingredients of the extract. There are variances in how different bacteria react to C. roseus leaf extracts. The main method these extracts fight bacteria is by breaking down the structure of their cell walls, including peptidoglycan and other cell wall components, which prevents the bacterial cells’ vital enzymatic functions.9,10,31 Polyphenols, a class of chemicals renowned for their capacity to bind with and destroy important bacterial components such as enzymes, proteins, and adhesins involved in cell transport, are primarily responsible for this impact. Additionally, by interfering with bacterial DNA, these polyphenols can prevent bacterial growth and multiplication.32 Furthermore, it has been discovered that the extraction solvents such as methanol and ethanol, have an impact on the efficacy.33,34 These solvents are more effective than water at dissolving the active ingredients, which emphasizes how crucial the extraction technique is in establishing the extracts’ efficacy. This highlights how important it is to select the right solvent when removing different advantageous compounds from plant materials and figuring out how they work against bacteria. Ethanol extracts may be more effective than water at solubilizing these bioactive chemicals, which could explain their enhanced potency in this study. Given that it affects the kinds and amounts of phytochemicals that are extracted from any plant material, it is clear that the extraction solvent can also have a significant impact on the mechanism of action.35,36 This study is notable for its comprehensive analysis of C. roseus leaf extracts’ antibacterial properties.
These findings underscore the considerable therapeutic potential of the extracts, suggesting their use as natural agents against nosocomial infections, particularly in chronic wounds and bedsores in long-term hospitalized patients. The results provide a foundation for the development of plant-based antimicrobial agents and support further research into the underlying mechanisms and clinical efficacy of these extracts.
Stdy Limitations
In this study, C. roseus leaves were used; other parts of the plant, such as the shoot, root, and stem, were not utilized. To analyze the phytochemical components and their structure, and identity of the molecules, the following techniques, such as High-Performance Liquid Chromatography (HPLC), High-Performance Thin-Layer Chromatography (HPTLC), and Nuclear Magnetic Resonance (NMR) spectroscopy, were not used in this study. The molecular level of structural interactions of the leaf extracts with various cellular components of the bacterial pathogens has not been studied. The other drug-resistant nosocomial pathogens, Enterobacter aerogenes, Streptococcus pyogenes, Klebsiella pneumoniae, and Acinetobacter baumannii, are not used in this study to determine the antimicrobial activity of different solvent extractions of the plant products. These important limitations should be addressed in future studies.
Conclusion
This study highlights the significant potential of C. roseus leaf extracts as a sustainable source of bioactive compounds with potent antibacterial properties against common nosocomial pathogens. The presence of diverse phytochemicals such as alkaloids, flavonoids, steroids, tannins, cardiac glycosides, and terpenoids highlights the plant’s rich chemical profile and the pivotal role of solvent selection in extracting targeted compounds. By transforming an often-overlooked plant into a valuable natural resource, this research aligns with the Sustainable Development Goals (SDGs), particularly Goal 3 (Good Health and Well-being) and Goal 12 (Responsible Consumption and Production), by promoting both public health and resource efficiency. These findings open doors to customized extraction methods and the exploration of other parts of C. roseus for broader health applications, offering a sustainable pathway for drug discovery that harnesses natural resources to promote both global health and environmental well-being.
Acknowledgement
We thank the staff and faculty of Dolphin Institute of Biomedical and Natural Sciences, Dehradun, India, for their continuous support and encouragement.
Funding source
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest
The author(s) do not have any conflict of interest
Data Availability
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not applicable
Authors’ Contribution
- Nazeerullah Rahamathullah: Conceptualization, Methodology, Analyses, Writing – Original Draft.
- Sovan Bagchi: Conceptualization, Supervision, Project administration, Writing – Review & Editing.
- Manisha Nanda: Visualization, Reference search, and interpretation of the data.
- Abhaya Dutta: Conceptualization, Methodology, Analysis, Methodology, Project administration, and Writing – Review & Editing.
Refereneces
- Talaat M, Zayed B, Tolba S, et al. Increasing antimicrobial resistance in World Health Organization Eastern Mediterranean Region, 2017–2019. Emerg Infect Dis. 2022;28(4):717–724.
CrossRef - Centers for Disease Control and Prevention (CDC). Antibiotic Resistance Threats Report. Atlanta, GA: US Department of Health and Human Services, CDC; 2023. https://www.cdc.gov/antimicrobial-resistance/data-research/threats/index.html.
- Salm S, Rutz J, van-den-Akker M, Blaheta RA, Bachmeier BE. Current state of research on the clinical benefits of herbal medicines for non-life-threatening ailments. Front Pharmacol. 2023;8:1–21.
CrossRef - Sharma P, Singla N, Kaur R, Bhardwaj U. A review on phytochemical constituents and pharmacological properties of Catharanthus roseus (L.) G. Don. J Med Plants Stud. 2024;12(3):131–156.
CrossRef - Lahane S, Dhikale Y, Jagadale R. Catharanthus roseus treatment of anti-cancer. Int J Novel Res Dev. 2024;9(6):1–4.
- Lahare RP, Yadav HS, Dashahre AK, Bisen YK. An updated review on phytochemical and pharmacological properties of Catharanthus rosea. Saudi J Med Pharm Sci. 2020;6(12):759–766.
CrossRef - Alibi S, Crespo D, Navas J. Plant-derivatives small molecules with antibacterial activity. 2021;10(3):231.
CrossRef - Yadav AK, Ambasta SK, Prasad SK, Trivedi MP. In-vitro evaluation of antibacterial property of Catharanthus roseus (Linn.) G. Don. var. “Rosea” and “Alba”. Int J Pharm Pharm Sci. 2018;10(5):55.
CrossRef - Roy SS, Rahman MA, Rahman MM. Phytochemical analysis and antibacterial activity of organic extract of Catharanthus roseus flower against gram-positive and gram-negative bacteria. J Agric Food Environ. 2020;1(4):87–93.
CrossRef - Padmaa PM. Catharanthus roseus Linn—A review. Acta Sci Pharm Sci. 2019;3(10):19–24
CrossRef - Pan Q. Monoterpenoid indole alkaloids biosynthesis and its regulation in Catharanthus roseus: A literature review from genes to metabolites. Phytochem Rev. 2016;15(2):221–250.
CrossRef - Azam K, Rasheed MA, Omer MO, Altaf I, Akhlaq A. Anti-hyperlipidemic and anti-diabetic evaluation of ethanolic leaf extract of Catharanthus roseus alone and in combination therapy. Braz J Pharm Sci. 2022;58:e18672:1–8.
CrossRef - Tolambiya P, Mathur S. A study on potential phytopharmaceuticals assets in Catharanthus roseus Int J Life Sci Biotechnol Pharm Res. 2016;5(1):1–6.
- Roy A, Khan A, Ahmad I, et al. Flavonoids is a bioactive compound from medicinal plants and its therapeutic applications. Biomed Res Int. 2022; 2022:5445291.
CrossRef - Goswami S. Preliminary phytochemical screening and standardization of leaves of Catharanthus roseus (L.). Indian J Res Pharm Biotechnol. 2013;1(1):24–27.
- Rao BM, Vedavijaya T, Ramani YR, Sayana SB. Protective role of methanol leaf extract of Catharanthus roseus in lipid profile modulation in diabetic Wistar rats. 2025;17(1):e77420.
- Rajashekara S, Reena D, Mainavi MV, et al. Biological isolation and characterization of Catharanthus roseus (L.) G. Don methanolic leaves extracts and their assessment for antimicrobial, cytotoxic, and apoptotic activities. BMC Complement Med Ther. 2022;22:328.
CrossRef - Kancherla N, Dhakshinamoorthi A, Chitra K, Komaram RB. Preliminary Analysis of Phytoconstituents and Evaluation of Anthelminthic Property of Cayratia auriculata (In Vitro). Maedica A Journal of Clinical Medicine. 2019;14(4):350–356.
CrossRef - Sabdoningrum EK, Hidanah S, Chusniati S, Soeharsono. Characterization and Phytochemical Screening of Meniran (Phyllanthus niruri Linn) Extract’s Nanoparticles Used Ball Mill Method. Pharmacogn J. 2021; 13(6)Suppl:1568-1572.
CrossRef - Indrakumar I, Selvi V, Gomathi R, Karpagam S. Phytochemical Analysis of Leaf Extracts of Calophyllum inophyllum L. and Cananga odorata (Lam.)f. & Thomson. IOSR J Pharm Biol Sci. 2012;3(2):35-37.
CrossRef - Jeong S, Jeon Y, Mun J, Jeong SM, Liang H, Chung K, et al. Ninhydrin Loaded Microcapsules for Detection of Natural Free Amino Acid. Chemosensors. 2023;11(1):49.
CrossRef - Ali S, Khan MR, Irfanullah, Sajid M, Zahra Z. Phytochemical investigation and antimicrobial appraisal of Parrotiopsis jacquemontiana (Decne) BMC Complementary and Alternative Medicine. 2018; 18:43.
CrossRef - Farag RS, Abdel-Latif MS, Abd El Baky HH, Tawfeek LS. Phytochemical screening and antioxidant activity of some medicinal plants’ crude juices. Biotechnol Rep (Amst). 2020; 7;28:e00536.
CrossRef - Balinadoa LO, Chan MA. Determination of Antimicrobial Properties of Crude Aqueous Leaves Extracts of Selected Medicinal Plants Using Resazurin-Based Microtiter Broth Dilution Method. Arabian Journal of Medicinal & Aromatic Plants. 2018;4(2):1-14.
- The European Committee on Antimicrobial Susceptibility Testing. Clinical Breakpoints—Bacteria (v 10.0). 2020. Available online:https://www.eucast.org/fileadmin/src/media/PDFs/EUCAST _files/ Breakpoint_ tables/ v_10.0_Breakpoint_Tables.pdf (accessed on 15 March 2024).
- Clinical and Laboratory Standards Institute (CLSI). Performance Standards for Antimicrobial Susceptibility Testing, 28th ed.; CLSI Supplement M100; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2018; ISBN1 978-1-68440-066-9. [Print]; ISBN2 978-1-68440-067-6. [Electronic].
- Khan A, Maparu K, Aran KR. Catharanthus roseus: A comprehensive review of its phytochemicals, therapeutic potential, and mechanisms of action. Nat Cell Sci. 2024;000(000):000–000.
CrossRef - Nisar A, Mamat A, Dzahir MIHM, Aslam MS, Ahmad MS. Antioxidant and total phenolic content of Catharanthus roseus using deep eutectic solvent. Rec Adv Biomed. 2017;3:7–10.
CrossRef - Pham HNT, Vuong QV, Bowyer MC, Scarlett CJ. Phytochemicals derived from Catharanthus roseus and their health benefits. 2020;8(4):80.
CrossRef - Akhtar N, Ihsan-ul-Haq, Mirza B. Phytochemical analysis and comprehensive evaluation of antimicrobial and antioxidant properties of 61 medicinal plant species. Arab J Chem. 2018;11(8):1223–1235.
CrossRef - Kumar RR, Kabesh K, Ragunathan R. Phytochemical analysis of Catharanthus roseus plant extract and its antimicrobial activity. Indian J Pure Appl Biosci. 2015;3(2):162–172.
- Raza ML, Nasir M, Abbas T, Naqvi SB. Antibacterial activity of different extracts from the Catharanthus roseus. Clin Exp Med J. 2009;3(1):81–85.
CrossRef - Kale S, Pathare TB, Lavale SA. Identification of phytochemicals and alkaloids in Catharanthus roseus and study of their antimicrobial activity. Int J Res Pharm Pharm Sci. 2018;6(3):431–433.
- Koel M, Kuhtinskaja M, Vaher M. Extraction of bioactive compounds from Catharanthus roseus and Vinca minor. Sep Purif Technol. 2020;252:117438.
CrossRef - Goyal P, Khanna A, Chauhan A, Chauhan G, Kaushik P. In vitro evaluation of crude extracts of Catharanthus roseus for potential antibacterial activity. Int J Green Pharm. 2008:176–181.
CrossRef - Wagay SA, Dwivedi SD, Sharma M, Tripathi J, Ahmad M. Antimicrobial activity of Catharanthus roseus. Chem Mater Res. 2013;3(9):61–64.
Abbreviations List
AMR – Antimicrobial Resistance,
MDR – Multidrug-Resistant,
MIC – Minimum Inhibitory Concentration,
MBC – Minimum Bactericidal Concentration,
MHA – Mueller-Hinton Agar,
MHB – Mueller-Hinton Broth.







