Manuscript accepted on :18-09-2025
Published online on: 26-09-2025
Plagiarism Check: Yes
Reviewed by: Dr. Moumita Hazra and Dr. Daya Shankar Gautam
Second Review by: Dr. Satya Namani
Final Approval by: Dr. Prabhishek Singh
Asaad Babker1* , Hanan Khalid Fadul Ahmed2, Salaheldein Elzaki 2
, Hanan Khalid Fadul Ahmed2, Salaheldein Elzaki 2 , Shawgi Elsiddig3
, Shawgi Elsiddig3 , Sarah Elsiddig Dafallah4, Tagwa Yousif Elsayed Yousif5
, Sarah Elsiddig Dafallah4, Tagwa Yousif Elsayed Yousif5 , Elryah Ali 6
, Elryah Ali 6 , Maha Mahagoub Osman Mahagoub7 and Vyacheslav Lyashenko8
, Maha Mahagoub Osman Mahagoub7 and Vyacheslav Lyashenko8
1Department of Medical Laboratory Sciences, College of Health Sciences, Gulf Medical University, Ajman, United Arab Emirates
2Department of Molecular Epidemiology, Tropical Medicine Research Institute, National Center for Research, Khartoum, Sudan
3Clinical Laboratory Sciences Department, College of Applied Medical Sciences, Jouf University, Sakaka, Saudi Arabia.
4Departrment of Family Medicine, Faculty of Medicine, Gadarif University, Gadarif, Sudan
5Department of Medical Laboratory Technology, College of Nursing and Health Sciences, Jazan University. Gizan, Saudi Arabia.
6Department of Medical Laboratory Technology, College of Applied Medical Sciences, Northern Border University, Arar, Saudi Arabia
7Department of Microbiology, Faculty of Medicine, Ibnsina university, Sudan
8Department of Media Systems and Technology, Kharkiv National University of Radio Electronics, Kharkiv, Ukraine
Corresponding Author E-mail: azad.88@hotmail.com
DOI : https://dx.doi.org/10.13005/bpj/3228
Abstract
This study aimed to investigate the prevalence of ACE I/D and PAI-1 4G/5G polymorphisms in Sudanese women experiencing recurrent pregnancy loss (RPL), and to explore their possible relationship with this condition. A retrospective study was carried out at Omdurman Maternity Hospital in Sudan. The study involved 125 participants: 64 RPL cases (defined as women with a history of at least three unexplained spontaneous abortions) and 61 healthy controls with normal success pregnancies and. A 5 mL EDTA blood sample was taken from each participant, and demographic, personal, and family medical information was collected using a questionnaire. Genomic DNA was extracted from blood leukocytes utilizing the GF-1 Blood DNA Extraction Kit. The polymorphisms of the PAI-1 4G/5G and ACE I/D genes were examined through polymerase chain reaction (PCR). Data analysis was conducted employing SPSS version 24. The ACE I/D polymorphism was found to have a notable association with recurrent pregnancy loss (RPL), with the I/D and D/D genotypes linked to an increased risk (odds ratio 1.29). Although the 4G allele and the 4G/4G genotype showed statistical significance, they were more prevalent in the control group (2.8% and 45.9%, respectively) compared to the cases (6.2%, p < 0.001). Homozygosity for the D allele of ACE, when combined with the PAI-1 4G/4G genotype, significantly raised the likelihood of RPL (p < 0.001). Despite a higher occurrence of the 4G/4G genotype in controls (17% versus 44.7%), these findings suggest complex interactions with other genetic factors. Overall, the homozygous D allele of ACE and the PAI-1 4G/4G genotype are more frequently observed among Sudanese women suffering from recurrent miscarriage, highlighting the commonality of these genetic variations within this population.
Keywords
ACE I/D Polymorphism; Abortion; Genetic Risk Factors; PAI-1 4G/5G; Recurrent Pregnancy Loss; Sudanese Women; Thrombophilia
Download this article as:| Copy the following to cite this article: Babker A, Ahmed H. K. F, Elzaki S, Elsiddig S, Dafallah S. E, Yousif T. Y. E, Ali E, Mahagoub M. M. O, Lyashenko V. Frequency of ACE I/D and PAI-1 4G/5G Polymorphisms in Women with Recurrent Pregnancy Loss in Sudan. Biomed Pharmacol J 2025;18(3). |
| Copy the following to cite this URL: Babker A, Ahmed H. K. F, Elzaki S, Elsiddig S, Dafallah S. E, Yousif T. Y. E, Ali E, Mahagoub M. M. O, Lyashenko V. Frequency of ACE I/D and PAI-1 4G/5G Polymorphisms in Women with Recurrent Pregnancy Loss in Sudan. Biomed Pharmacol J 2025;18(3). Available from: https://bit.ly/46pA1L0 |
Introduction
Recurrent pregnancy loss (RPL), previously characterized as three or more consecutive pregnancy losses, is now defined by two or more pregnancy losses.1 Clinically recognized pregnancy loss, indicating spontaneous termination before the 20th week of gestation, affects approximately 1-5% of women during their childbearing years .RPL is a multifaceted condition with a poorly understood underlying cause.2 Numerous factors, including chromosomal abnormalities, anatomical issues, endocrine imbalances, immunological factors, and infections, are believed to contribute to RPL.3, 4, 5 The renin-angiotensin system (ReAnS) is a crucial hormonal system essential for regulating blood pressure and fluid balance within the body.5 6, 7, 8 Angiotensin-converting enzymes (ACE) subsequently transforms angiotensin I into angiotensin II, a potent vasoconstrictor that narrows blood vessels and elevates blood pressure. Angiotensin II also stimulates aldosterone release, promoting sodium and water retention by the kidneys and consequently increasing blood volume and pressure .9 Endocrine secretions originating from the decidua, placenta, and ovary influence ReAnS throughout gestation. Specifically, the rise in AGT levels, triggered by estrogen during the first trimester, correlates directly with increased angiotensin II production.10 The uteroplacental unit expresses ReAnS components, emphasizing its local importance .11, 12 The uteroplacental ReAnS plays a vital role in endometrial regeneration following shedding, decidualization, implantation, and placentation.13 Furthermore, local ReAnS participates in prostaglandin production, estradiol release, and regulation of blood flow to the placenta and uterus.14 The D allele is linked to higher plasma levels of Angiotensin-Converting Enzyme (ACE) and, consequently, increased enzyme activity. Unlike individuals homozygous for the I allele (I/I), who have the lowest ACE levels, those with the D/D genotype generally possess twice the amount of the enzyme in circulation. Heterozygous individuals (I/D) typically exhibit intermediate levels of ACE in both plasma and tissues.17,16, 18
This research seeks to explore the involvement of ACE I/D and PAI-1 4G/5G gene polymorphisms in Sudanese women experiencing RPL, with the intention of evaluating their potential link to the condition.
Materials and Methods
A retrospective study was conducted at Omdurman Medical Hospital in Sudan. The study involved 232 women, divided into two groups: 119 women with recurrent spontaneous abortions forming the case group, and 113 healthy women with at least two successful pregnancies and no history of pregnancy loss serving as the control group. Participants in the case group were Sudanese women between 25 and 45 years of age who had experienced at least three consecutive pregnancy losses. The control group was age-matched and free from prior pregnancy loss. Women were excluded if they had a history of vascular thrombotic disorders, congenital fetal abnormalities, chromosomal abnormalities, uterine abnormalities, or known causes of abortion. Ethical approval and informed consent procedures were carefully followed in this study. The research received approval from the ethical committees of Omdurman Maternity Hospital in Sudan. Additionally, participants provided documented written consent by signing the agreement included within the questionnaire and Each participant provided informed consent after receiving detailed information about the study’s objectives, the confidentiality of their data, and the intended use of the information exclusively for the research purposes. All women provided written informed consent before participating. A structured questionnaire was used to collect data on demographics, medical history, family history, and obstetric history. A 5-milliliter sample was collected following informed consent, properly labeled, and stored for future laboratory analysis.
Molecular technique
DNA extraction
The extraction of genomic DNA was performed on up to 200 μL of whole blood using a GF-1 Blood DNA Extraction Kit. The process began with the addition of blood to a microcentrifuge tube, followed by a lysis buffer to disrupt cell membranes. The mixture was homogenized via pulsed vortexing and incubated at an elevated temperature (65°C) to facilitate lysis. Proteinase K was added to digest proteins, and RNase A was included to remove RNA contamination. After incubation, 100% ethanol was added to precipitate the DNA. The solution was then applied to a purification column and centrifuged to bind the DNA to the matrix. The column was washed with a series of wash buffers to remove impurities while retaining the DNA. Finally, the
purified DNA was eluted from the column using a heated elution buffer and stored at -20°C for subsequent use in PCR experiments. All centrifugation steps were conducted at 5,000 x g for the specified durations.
Molecular analysis
Detection of plasinogene Activator -1 4G/5G polymorphisms
Polymerase Chain Reaction (PCR) was used to identify polymorphisms in the PAI-1 4G/5G gene at position -675, employing the Amplification Refractory Mutation System (ARMS-PCR) method. The reaction mixture consisted of 0.4 pmol of each forward and reverse primer, 4 μL of 5X Green Mastermix, and 5 μL of genomic DNA, brought to a final volume of 25 μL.The primers included a common downstream primer and allele-specific primers for the 4G or 5G variants. These primers amplified the respective alleles at an annealing temperature of 55°C. The PCR cycling conditions were as follows: initial denaturation at 95°C for 3 minutes; 30 cycles of denaturation at 95°C for 20 seconds, annealing at 55°C for 10 seconds, and extension at 72°C for 20 seconds; with a final extension step at 72°C for 3 minutes.
PCR products were separated by 2% agarose gel electrophoresis, producing fragments of 138 bp for the 4G allele and 139 bp for the 5G allele.
Determination of AnCoEn genotyping
PCR-based methods were used to genotype the AnCoEn I/D polymorphism, targeting a region previously described in reference 20. Each 25 μL PCR reaction contained 5 μL of genomic DNA, 4 μL of 5X Green Master Mix, and 0.4 pmol of both forward and reverse primers specific to the AnCoEn I/D region. Amplification was performed with a Bio-Rad Peltier thermal cycler using the following thermal profile: an initial denaturation step at 94°C for 5 minutes, followed by 35 cycles of denaturation (94°C for 1 minute), annealing (58.5°C for 90 seconds), and extension (65°C for 4 minutes), with a final extension at 72°C for 7 minutes. After amplification, PCR products were separated by electrophoresis on a 1.5% agarose gel containing 1xTBE buffer, run at 100 volts for 45 minutes. Following electrophoresis, the gel was stained with ethidium bromide (0.5 μg/mL) and visualized under UV light. Genotypes were determined based on band size: the presence of a 490 bp fragment corresponded to the II genotype, a 190 bp fragment to the DD genotype, and both fragments (490 bp and 190 bp) to the ID genotype.
Genotyping of ANCoEn I/Genome D
The ANCoEn I/Genome D gene was genotyped using PCR amplification with primers flanking a specific region previously described in the literature. The PCR reaction volume was 25 μL, which included 5 μL of genomic DNA, 4 μL of 5X Green Mastermix, and 0.4 pmol of each forward and reverse primer per sample.
The PCR process involved initial denaturation at 94°C for 5 minutes, followed by 35 cycles of: denaturation at 94°C for 1 minute, annealing at 58.5°C for 90 seconds, and extension at 65°C for 4 minutes. A final extension step was performed at 72°C for 7 minutes, using a Bio-Rad Peltier thermal cycler.
PCR products were separated by electrophoresis at 100 volts for 45 minutes in 1.5% agarose gel prepared in 1X TBE buffer. The gel was stained with 0.5 μg of Ethidium Bromide and visualized under UV light.
Result interpretation
490 bp fragment indicates the II genotype
190 bp fragment indicates the DD genotype
Both 490 bp and 190 bp fragments indicate the ID genotype
Statistical analysis
Data entry and analysis were conducted using SPSS software (version 24.0). The prevalence of PAI-1 4G/5G and ANCoEn I/D polymorphisms among patients and controls was compared using the Chi-square test, with a significance level set at P < 0.05. Demographic data for the study population is presented as means in the text. The strength of the association between the polymorphisms and the outcomes was assessed using categorical frequencies expressed as percentages (%)
Results
The study groups were balanced regarding age, with mean ages of 31.3 ± 5.9 years for patients and 30.3 ± 5.4 years for controls (p = 0.285, Table I). Among patients, 25.2% experienced four or more abortions.
Genotyping of the AnCoEn polymorphism was performed using gel electrophoresis, as illustrated in Figure I. The lanes labeled 2, 3, 4, 8, and 12 display the homozygous I/I genotype with a 190 bp band. Lanes 5, 6, and 7 contain the heterozygous ID genotype characterized by both 190 bp and 490 bp bands. Lanes 9 and 11 show the D/D genotype with a 490 bp band.
PCR analysis for PAI-1 4G polymorphisms is shown in Figure 2. The gel reveals bands at approximately 154 bp, indicating the presence of the 4G allele in lanes 2, 3, 4, 6-11, confirming the detection of the mutant genotype.
The gel images for PlAcIn-1 polymorphism are presented in Figure 3. Lanes 1-10 display positive results for the 4G allele, evidenced by bands at the expected size, confirming the genotype as 4G carriers.
Statistical analysis revealed significant associations between certain genotypes and recurrent miscarriage. The D/I AnCoEn polymorphism showed significant differences (p = 0.006), with an odds ratio of 0.40 (95% CI: 0.21–0.77) (Table 2). Regarding the PAI-1 polymorphism, allele 4G and genotypes 4G/5G and 4G/4G differed significantly between patients and controls (Table 3, p < 0.001).
Analysis of the effect of AnCoEn and PAI-1 genotypes relative to abortion history among miscarriage patients showed no statistically significant associations (Table 4). However, when examining the interaction between genotypes across both groups, women with the D/D AnCoEn genotype exhibited a strong association with the 4G/4G, 4G/5G, and 5G/5G PAI-1 genotypes (Table 5, p < 0.001).
Table I: Baseline characteristic among patients with recurrent miscarriage and control groups.
| Characteristic | Patient(N=119) | Control (N=113) | P-value | |
| Age means +SD | 31.3+5.9 | 30.3+5.4 | 0.285 | |
| Abortion time N (%) | <4 | 89 (74.8) | – | – |
| ≥4 | 30 (25.2) | – | – | |
Table 2: AnCoEn polymorphism in miscarriage patient and control groups
| AnCoEn | Patient N=119(%) | Control N=113(%) | TotalN | P-value | Z- test | OR (95%CI) |
| D/D | 94(79.0) | 69(61.1) | 163 | – | – | 1 (reference) |
| D/I | 18(15.1) | 33(29.2) | 51 | 0.006* | -2.75 | 0.40(0.21 to 0.77) |
| I/I | 7(5.9) | 11(9.7) | 18 | 0.135 | -1.50 | 0.47(0.17 to 1.27) |
| Allele D | 206(86.6) | 171(75.7) | 377 | – | – | 1 (reference) |
| Allele I | 32(13.4) | 55(24.3) | 87 | 0.003* | 19.93 | 0.48(0.29 to 0.78) |
OR= odds ratio, CI=Confident Interval
*P-value<0.05 statistically significant association
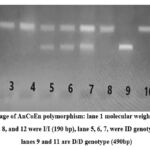 |
Figure I: Gel image of AnCoEn polymorphism: lane 1 molecular weight maher1Kb plus, lane, 2, 3, 4 and 8, and 12 were I/I (190 bp), lane 5, 6, 7, were ID genotype (190/490 bp), lanes 9 and 11 are D/D genotype (490bp). |
 |
Figure 2: Figure PCR amplification PAI-1 4G of polymorphisms PCR product loaded on 2% agarose gel disolved in 1X TBE buffer, stanied with ethidium bromide, Lane 1 molecular weight marker 100 bp, lane 2, 3, 4, 6, 7, 8, 9, 10, 11 were mutant type (154bp). |
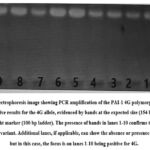 |
Figure 3: Gel electrophoresis image showing PCR amplification of the PAI-1 4G polymorphism. Lanes 1-10 display positive results for the 4G allele, evidenced by bands at the expected size (154 bp). |
Table 3: genotype and allele frequencies for miscarriage and controls
| PlAcIn | Patient N=119(%) | Control N=113(%) | Total | P-value | Z-test | OR (95%CI) |
| 5G/5G | 7(5.9) | 38(33.6) | 45 | – | – | 1 (reference) |
| 4G/5G | 52(43.7) | 47(41.6) | 99 | 0.000* | 3.92 | 6.01(2.45 to 14.74) |
| 4G/4G | 60(50.4) | 28(24.8) | 88 | 0.000* | 5.21 | 11.63(4.62 to 29.26) |
| Allele 5G | 66(27.7) | 123(54.4) | 189 | – | – | 1 (reference) |
| Allele 4G | 172(72.3) | 103(45.6) | 275 | 0.000* | 34.22 | 3.11(2.12 to 4.58) |
*P-value<0.001 highly statistically significant association
Table 4: Association of AnCoEn and PlAcIn Genotypes with Abortion Number in Women with Recurrent Pregnancy Loss
| genotypes | Abortion time | P-value | chix2 | ||
| <4N=98(%) | ≥4N=21(%) | ||||
| AnCoEn | D/D | 77(78.6) | 17(81.0) | 0.961 | 0.079 |
| D/I | 15(15.3) | 3(14.3) | |||
| I/I | 6(6.1) | 1(4.8) | |||
| PlAcIn | 4G/4G | 4(4.1) | 3(14.3) | 0.141 | 3.92 |
| 4G/5G | 42(42.9) | 10(47.6) | |||
| 5G/5G | 52(53.1) | 8(38.1) | |||
Table 5: Association between AnCoEn and PlAcIn genotypes on recurrent miscarriage patients and controls
| AnCoEn | PlAcIn | P-value | Chix2 | |||
| 4G/4G | 4G/5G | 5G/5G | ||||
| D/D | Patient N=94(%) | 6(6.4) | 44(46.8) | 44(46.8) | 0.000** | 19.93 |
| Control N=69(%) | 21(30.4) | 32(46.4) | 16(23.2) | |||
| D/I | Patient N=18(%) | 0 | 6(33.3) | 12(66.7) | 0.006* | 10.12 |
| Control N=33(%) | 12(36.4) | 11(33.3) | 10(30.3) | |||
| I/I | Patient N (%) | 1(14.3) | 2(28.6) | 4(57.1) | 0.343 | 3.03a |
| Control N (%) | 5(45.5) | 4(36.4) | 2(18.2) | |||
*P-value<0.05 statistically significant association, **P-value<0.001 highly statistically significant association
a: Fisher exact test
Discussion
Sudanese women face numerous health challenges, including pregnancy complications.19 and cancers such as breast cancer.20 which are significantly influenced by genetic and vascular factors. Recent research indicates that variations in genes related to blood clotting, such as ACE I/D and PAI-1 4G/5G polymorphisms, can increase the risk of recurrent pregnancy loss.
The findings of this study provide important insights into the genetic factors underlying recurrent pregnancy loss (RPL) in Sudanese women, with particular focus on ACE I/D and PAI-1 4G/5G polymorphisms. The significant association of the ACE D/D and I/D genotypes with increased susceptibility to RPL aligns with several previous studies that have reported a similar relationship, such as those conducted in Iran.21,22 These studies suggest that the ACE D allele, particularly in homozygous form, may contribute to the pathophysiology of RPL, possibly through its role in blood pressure regulation and vascular function, which are critical during pregnancy.23
Interestingly, this study found that the 4G allele and 4G/4G genotype of PAI-1 were more common in controls than in cases, and their association with RPL was statistically significant. This contrasts with the meta-analysis by Li et al.24 which demonstrated that the PAI-1 4G/5G polymorphism is generally linked to an increased risk of RPL across diverse populations, including Asians and Caucasians. Moreover, Wolf et al.25 and Adler et al.26 reported similar associations where the 4G/4G genotype was associated with a higher propensity for pregnancy loss. Our findings suggest that in the Sudanese population, the PAI-1 4G/4G genotype might have a different modulatory effect or gene-environment interaction impacting the risk differently than in other ethnic groups.
When recurrent abortion was compared in the current study to D/D genotype and D allele of the AnCoEn gene, there was a highly significant correlation 59 (92.2%). Su et al.27 they conducted a systematic review and meta-analysis to examine the relationship between the AnCoEn polymorphism and recurrent abortions. Under the assumption of dominant inheritance, data from 11 eligible studies, including 1275 patients and 2049 controls, were analysed. A significant association between the two was found. Additionally, they demonstrated a strong correlation between the AnCoEn I/D polymorphism and recurrent pregnancy loss, as well as a higher risk (1.29 OR) for women with the I/D and D/D genotypes compared to those with the I/I genotype . This study also demonstrated that, even if the prevalence is higher in controls28(45.9) than in patients4(6.2) (p-value 0.000*), the frequency of the 4G allele and 4G/4G genotype in the patient group is statistically significant. This result is consistent with two earlier investigations conducted in Iran by Soltanghoraee et al., and Aarabi et al., .28,29 In contrast, further results from Bulgaria and two research by Coulam and Jeyendran. 30 and Coulam et al.31 in the US do not demonstrate a significant relationship between RPL and 4G/4G genotypes.32,33 Therefore, even in research carried out in the same nation, there are discrepancies in the findings of PlAcIn-1 4G/5G polymorphism with recurrent abortions .
The prevalence of the 4G/4G polymorphism is larger in controls (17.7%) than in patients (4.8%), but our findings demonstrate that homozygosity for the D allele of the AnCoEn gene in combination with the PlAcIn-1 4G4G genotype was a significant risk factor for recurrent pregnancy loss (p-value0.000*). This result agrees with a prior report in the literature.34 The size of the population sample can have an impact on variations in the frequency of PlAcIn-1 4G4G polymorphisms. Smaller sample sizes did not reveal significant differences in PlAcIn-1 4G4G frequencies, but larger samples of controls (n = 1956) (Goodman & co-workers.35 compared to recurrent aborters indicated a significant increase in PlAcIn-1 4G4G.
Conclusion
In conclusion, the frequency of the ACE D allele homozygosity and the PAI-1 4G/4G genotype is significantly higher among women with recurrent miscarriage, indicating that these genetic variations are prevalent in this population. The combined occurrence of these polymorphisms occurs at a noteworthy rate and suggests a potential synergistic effect in increasing the risk of recurrent pregnancy loss. Monitoring the frequency of these genotypes can be valuable for understanding genetic predispositions and guiding risk assessment in affected women.
Acknowledgement
I would like to express my gratitude to everyone who participated in the study. I especially extend my heartfelt appreciation to the staff at Omdurman Maternity Hospital in Sudan for their support and assistance.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This study involved human participants; therefore, ethical approval was obtained from the ethical committees of Omdurman Maternity Hospital in Sudan (ETH/OMD/2019/0456). The research was conducted in accordance with the ethical standards and guidelines currently applied in the country, and all privacy rights of the human subjects were strictly observed.
Informed Consent Statement
This study involved human participants, specifically women experiencing recurrent pregnancy loss and healthy controls. Informed consent was obtained from all participants prior to sample collection and data inclusion. Participants were informed about the study’s objectives, procedures, potential risks, and benefits, and their participation was voluntary. The confidentiality and privacy rights of all human subjects were strictly observed throughout the research, complying with ethical standards and guidelines applicable in Sudan.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not applicable
Author Contributions
- Hanan Khalid Fadul Ahmed , Salaheldein G Elzaki and Asaad Babker: Visualization, Supervision, Review & Editing, Project Administration
- Asaad Babker and Vyacheslav Lyashenko: Conceptualization, Methodology, Writing – Original Draft, Editing, Data Collection, Analysis
- Hanan Khalid Fadul Ahmed and Asaad Babker: Methodology, Data Collection, Analysis.
- Shawgi A Elsiddig and Sarah Elsiddig Dafallah: Methodology, Writing – Original Draft.
References
- Babker A M, Gameel F E. Molecular characterization of prothrombin G20210A gene mutations in pregnant Sudanese women with spontaneous recurrent abortions. Rawal Med J. 2015;40(2):207-209
- Pei C Z, Kim Y J, Baek K H. Pathogenetic factors involved in recurrent pregnancy loss from multiple aspects. Obstet Gynecol Sci. 2019;62(4):212-223.
CrossRef - Hyde K J, Schust D J. Genetic considerations in recurrent pregnancy loss. Cold Spring Harb Perspect Med. 2015;5(3):a023119.
CrossRef - Babker A M, Gameel F E. Methylenetetrahydrofolate reductase C677T polymorphism in Sudanese women with recurrent spontaneous abortions. Kuwait Med J. 2016;48(2):100-104.
- Babker A M. The role of inherited blood coagulation disorders in recurrent miscarriage syndrome. J Crit Rev. 2019;7(1):20-20.
- Pahlavani M, Kalupahana N S, Ramalingam L, Moustaid-Moussa N. Regulation and functions of the renin-angiotensin system in white and brown adipose tissue. Compr Physiol. 2017;7(4):1137-1150.
CrossRef - Fountain J H, Lappin S L. Physiology, renin-angiotensin system. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2022. Available from: https://www.ncbi.nlm.nih.gov/books/NBK470410
- Nielsen A, Schauser K, Poulsen K. The uteroplacental renin-angiotensin system. 2000;21(5-6):468-477.
CrossRef - Zhang F, Liu H, Liu D, et al. Effects of RAAS inhibitors in patients with kidney disease. Curr Hypertens Rep. 2017;19(9):72.
CrossRef - Tan W S D, Liao W, Zhou S, Mei D, Wong W F. Targeting the renin–angiotensin system as novel therapeutic strategy for pulmonary diseases. Curr Opin Pharmacol. 2018;40:9-17.
CrossRef - Bernardi S, Michelli A, Zuolo G, Candido R, Fabris B. Update on RAAS modulation for the treatment of diabetic cardiovascular disease. J Diabetes Res. 2016;2016:8917578
CrossRef - Lumbers E R, Pringle K G. Roles of the circulating renin-angiotensin-aldosterone system in human pregnancy. Am J Physiol Regul Integr Comp Physiol. 2014;306(2):R91-R10
CrossRef - Irani R A, Xia Y. Renin-angiotensin signaling in normal pregnancy and preeclampsia. Semin Nephrol. 2011;31(1):47-58.
CrossRef - Gintoni I, Adamopoulou M, Yapijakis C. The angiotensin-converting enzyme insertion/deletion polymorphism as a common risk factor for major pregnancy complications. In Vivo. 2021;35(1):95-103
CrossRef - Yart L, Bahmanyar E, Cohen M, de Tejada B. Role of uteroplacental renin-angiotensin system in placental development and preeclampsia. 2021;9(10):1332
CrossRef - Nielsen A, Schauser K, Poulsen K. The uteroplacental renin-angiotensin system. 2000;21(5-6):468-477.
CrossRef - Gintoni I, Adamopoulou M, Yapijakis C. The angiotensin-converting enzyme insertion/deletion polymorphism as a common risk factor for major pregnancy complications. In Vivo. 2021;35(1):95-103.
CrossRef - Brenner B. Hypercoagulability and recurrent miscarriages. Clin Adv Hematol Oncol. 2010;8:467.
- Brown N J, Vaughan D E. The renin-angiotensin and fibrinolytic systems: co-conspirators in the pathogenesis of ischemic cardiovascular disease. Trends Cardiovasc Med. 1996;6(4):239-246.
CrossRef - Babker A, et al. Factor V Leiden G1691A, prothrombin G20210A, and MTHFR C677T mutations among Sudanese women with recurrent pregnancy loss. Int J Biomed. 2024;14(1):59-65
CrossRef - Shalabi M G, Eltahir Z, Ba M A. Evaluation of angiogenesis using CD105 and CD34 in Sudanese breast cancer patients. Pak J Biol Sci. 2021;24(11):1144-1151.
CrossRef - Kamali M, Hantoushzadeh S, Borna S, et al. Association between thrombophilic gene polymorphisms and recurrent pregnancy loss susceptibility in Iranian women: a systematic review and meta-analysis. Iran Biomed J. 2018;22(2):78-89
- Li X, Liu Y, Zhang R, Tan J, Chen L, Liu Y. Meta-analysis of the association between plasminogen activator inhibitor-1 4G/5G polymorphism and recurrent pregnancy loss. Med Sci Monit. 2015 Apr 11;21:1051-6. doi: 10.12659/MSM.892898. PMID: 25862335; PMCID: PMC4403499.
CrossRef - Wolf C E, Haubelt H, Pauer H U, et al. Recurrent pregnancy loss and its relation to FV Leiden, FII G20210A and polymorphisms of plasminogen activator and plasminogen activator inhibitor. Pathophysiol Haemost Thromb. 2003;33(2):134-137.
CrossRef - Adler G, Mahmutbegovic N, Valjevac A, et al. Association between -675 ID, 4G/5G PAI-1 gene polymorphism and pregnancy loss: a systematic review. Acta Inform Med. 2018;26(3):156-159.
CrossRef - Su M-T, Lin S-H, Chen Y-C, Kuo P-L. Genetic association studies of ACE and PAI-1 genes in women with recurrent pregnancy loss. Thromb Haemost. 2013;109(1):8
CrossRef - Aarabi M, Memariani T, Arefi S, et al. Polymorphisms of plasminogen activator inhibitor-1, angiotensin converting enzyme and coagulation factor XIII genes in patients with recurrent spontaneous abortion. J Matern Fetal Neonatal Med. 2011;24(6):545-549
CrossRef - Soltanghoraee H, Memariani T, Aarabi M, et al. Association of ACE, PAI-1, and coagulation factor XIII gene polymorphisms with recurrent spontaneous abortion in Iranian patients. J Reprod Infertil. 2007;7(4):324-330.
- Ivanov P, Komsa-Penkova R, Konova E, et al. Combined thrombophilic factors among women with late recurrent spontaneous abortions. Akush Ginekol (Sofiia). 2011;50(3):8-12.
- Coulam CB, Jeyendran R, Fishel LA, Roussev R. Multiple thrombophilic gene mutations are risk factors for implantation failure. Reprod Biomed Online. 2006;12(3):322-327.
CrossRef - Coulam CB, Jeyendran RS. Vascular endothelial growth factor gene polymorphisms and recurrent pregnancy loss. Am J Reprod Immunol. 2008;59(4):301-304.
CrossRef - Wolf CE, Haubelt H, Pauer HU, et al. Recurrent pregnancy loss and its relation to FV Leiden, FII G20210A, and polymorphisms of plasminogen activator and plasminogen activator inhibitor. Pathophysiol Haemost Thromb. 2003;33(2):134-137.
CrossRef - Buchholz T, Lohse P, Rogenhofer N, et al. Polymorphisms in the ACE and PAI-1 genes are associated with recurrent spontaneous miscarriages. Hum Reprod. 2003;18(10):2473-2477.
CrossRef - Goodman C S, Coulam C B, Jeyendran R S, Acosta V A, Roussev R. Which thrombophilic gene mutations are risk factors for recurrent pregnancy loss? Am J Reprod Immunol. 2006;56(3):230-235.
CrossRef







