Manuscript accepted on :16-06-2025
Published online on: 08-09-2025
Plagiarism Check: Yes
Reviewed by: Dr. Dhyanendra Jain
Second Review by: Dr. Kasthuri NMK
Final Approval by: Dr. Patorn Piromchai
Yusminah Hala , A Irma Suryani*
, A Irma Suryani* , Muhammad Junda
, Muhammad Junda , Hartono
, Hartono , Rachmawaty Muhtar
, Rachmawaty Muhtar , Andi Annisa
, Andi Annisa and Andi Fahirah
and Andi Fahirah
Department of Biology, Faculty of Mathematics and Natural Sciences, Makassar State University, Makassar, Indonesia
Corresponding Author Email: a.irma.suryani@unm.ac.id
DOI : https://dx.doi.org/10.13005/bpj/3231
Abstract
Biosynthesis offers a safe and eco-friendly approach for producing nanoparticles using microorganisms or plant-derived reducing agents. This study explores the novelty of using endophytic bacteria isolated from the roots and stems of Lannea coromandelica, a medicinal plant with untapped potential in green nanotechnology, for the biosynthesis of silver nanoparticles (AgNPs). Four bacterial isolates were used Enterococcus (AM 2), Azotobacter (AM 5), Micrococcus (BM 2), and Enterococcus (BM 4), in reaction with 2–3 mM silver nitrate (AgNO₃). Characterization was performed using Particle Size Analyzer (PSA), UV-Visible Spectrophotometry (UV-Vis), Fourier Transform Infrared Spectroscopy (FTIR), and Scanning Electron Microscopy (SEM). The synthesized AgNPs had particle sizes ranging from 60 to 105.9 nm. Notably, AgNPs synthesized using Azotobacter (AM 5) and Micrococcus (BM 2) showed optimal size and stability, with PI (Polydispersity Index) values of 0.029673 and 0.01835, respectively, indicating uniform particle distribution. UV-Vis spectra confirmed stability for up to five days. FTIR revealed the presence of functional groups O-H, C-N, and Ag-O, validating biosynthesis. SEM analysis demonstrated spherical to semi-spherical morphologies, with AM 5 exhibiting the most homogeneous structure. These findings highlight the promising role of L. coromandelica-derived endophytes in the eco-friendly synthesis of AgNPs for potential nanomedicine and antimicrobial applications.
Keywords
Endophytic Bacteria; Fourier Transform Infrared; Lannea coromandelica; Scanning Electron Microscopy; Silver Nanoparticle
Download this article as:| Copy the following to cite this article: Hala Y, Suryani A. I, Junda M, Hartono H, Muhtar R, Annisa A, Fahirah A. Endophytic Bacteria from Lannea coromandelica for Enhancing Silver Nanoparticle Biosynthesis. Biomed Pharmacol J 2025;18(3). |
| Copy the following to cite this URL: Hala Y, Suryani A. I, Junda M, Hartono H, Muhtar R, Annisa A, Fahirah A. Endophytic Bacteria from Lannea coromandelica for Enhancing Silver Nanoparticle Biosynthesis. Biomed Pharmacol J 2025;18(3). Available from: https://bit.ly/3VDNOHq |
Introduction
Nanotechnology is a prominent research field focused on the creation of unique materials known as nanostructures. These include various forms such as nanoparticles, nanocrystals, and nanotubes, which are characterized by their size and shape at the nanoscale.1 Nanoparticles are particles with dimensions less than 100 nm, encompassing metals, polymers, fullerenes, and ceramics. Biological methods for their synthesis are preferred due to their low toxicity, cost-effectiveness, and environmental friendliness.2 One of the flagship products of nanotechnology is silver nanoparticles (AgNPs), which have attracted significant attention due to their optical, electrical, and magnetic properties that can be modified to suit various applications, such as antibacterial, anticancer, anti-inflammatory, and antioxidant activities.3 AgNPs have been shown to exhibit excellent bactericidal properties against a wide range of microorganisms, with mechanisms that disrupt cell membrane functions, inhibit metabolism, and interfere with bacterial DNA replication.4
However, conventional chemical synthesis methods for producing AgNPs often involve hazardous chemicals that have negative impacts on the environment and human health. Therefore, the biosynthesis of AgNPs presents an appealing alternative, being more environmentally friendly, cost-effective, and capable of producing AgNPs with stable and effective characteristics.5 Biosynthesis using microorganisms, such as endophytic bacteria, has emerged as a promising method. Endophytic bacteria are microorganisms that live within plant tissues without causing disease symptoms and are capable of producing secondary metabolites that play a role in the reduction of silver ions to AgNPs.6
Green particle synthesis using plants offers significant advantages over other biological methods, such as lower production costs, simpler and safer processes, the ability to scale up production, and faster synthesis times.7 In the context of this study, the biosynthesis of silver nanoparticles by endophytic bacteria from Lannea coromandelica supports the development of environmentally friendly nanotechnology, particularly for nanomedicine applications, by utilizing an efficient and sustainable biological approach.
In this context, the use of endophytic bacteria from Lannea coromandelica (commonly known as Java wood) provides a novel and underexplored source for AgNP production. L. coromandelica is known to host diverse endophytes that produce bioactive compounds such as saponins, flavonoids, polyphenols, and tannins—substances that can serve as natural reducing agents.8
To date, there have been few reports on the biosynthesis of AgNPs using endophytic bacteria specifically from the roots and stems of L. coromandelica. This research fills that gap by exploring the biosynthetic potential of these endophytes and characterizing the resulting nanoparticles. The biosynthesis process involves several steps, including bacterial isolation and nanoparticle characterization using Particle Size Analyzer (PSA), Fourier Transform Infrared Spectroscopy (FTIR), UV-Visible Spectrophotometry (UV-Vis), and Scanning Electron Microscopy (SEM). The findings are expected to contribute significantly to the advancement of green nanotechnology and its biomedical applications, particularly in nanomedicine for targeted drug delivery.9
The biosynthesis process of AgNPs involves several stages, from the isolation of endophytic bacteria to the characterization of the AgNPs. Characterization is carried out using various instruments such as PSA, FTIR, UV-Vis, and SEM to determine the size, morphology, functional groups, and stability of the nanoparticles. This study aims to explore the potential of endophytic bacteria from the roots and stems of L. coromandelica in the biosynthesis of AgNPs and to examine the characteristics of the resulting AgNPs. The findings of this research are expected to contribute to the development of environmentally friendly nanotechnology, particularly in health applications such as nanomedicine, which utilizes nanoparticles for effective drug delivery.
Materials and Methods
Research Location and Time
This study is a descriptive type of research that involves several stages, including sample collection, isolation of endophytic bacteria, biosynthesis of silver nanoparticles, and characterization of silver nanoparticles. The research was conducted from March 2024 to December 2024. Laboratories used in this study included Biology Laboratory, FMIPA, Universitas Negeri Makassar; Chemistry Laboratory, Faculty of Science and Technology, UIN Alauddin Makassar; Integrated Laboratory, Universitas Negeri Yogyakarta (PSA); Microstructure Engineering Laboratory, Universitas Muslim Indonesia (SEM).
Materials
Laboratory equipment used in this study included standard glassware, an analytical balance, an ultrasonic bath, Shimadzu FTIR (Fourier Transform InfraRed), Microtrac Particle Size Analyzer (PSA), Shimadzu UV-Vis Spectrophotometer, and JEOL JCM 6000 Scanning Electron Microscopy (SEM). Additionally, a Laminar Air Flow (LAF) cabinet, shaker incubator, Bunsen burner, vials and glass bottles, syringes, stirring rods, hot plate, autoclave, refrigerator, centrifuge, and inoculating needles were used to support various stages of the research. The main materials used in this study included the roots and stems of L. coromandelica as the source of endophytic bacteria, Enterococcus and Azotobacter endophytic bacteria, and distilled water. The media used consisted of Nutrient Agar (NA) and Luria Broth (LB) media. Silver nitrate (AgNO3) solutions with concentrations of 0.5 mM, 1 mM, 1.5 mM, 2 mM, and 3 mM were also used in the biosynthesis process of silver nanoparticles. Additional materials included nystatin, 70% ethanol, aluminum foil, tissue, spirit, cotton swabs, cotton, roll wrap, and labels to support the research process.
Research Procedure
Sample Collection and Isolation of Endophytic Bacteria
The surfaces of the roots and stems of L. coromandelica were sterilized using 70% ethanol, 5.25% sodium hypochlorite, and 70% ethanol again. After washing and drying, the roots and stems were ground, serially diluted up to 10⁻⁴, and inoculated onto NA media containing nystatin. After incubation for 2×24 hours, the growing endophytic bacterial colonies were purified using the streaking method on NA and LB agar media. The pure cultures were then inoculated into LB Broth media for cell growth before the biosynthesis of silver nanoparticles. The AgNO3 solution was prepared by dissolving AgNO3 powder (Mr = 170 g/mol) in distilled water. The concentrations of the solutions prepared were 0.5 mM, 1 mM, 1.5 mM, 2 mM, and 3 mM.
Biosynthesis of Silver Nanoparticles
The endophytic bacteria inoculated in liquid LB media were centrifuged at 10,000 rpm for 10 minutes to obtain the pure supernatant. The supernatant (10 mL) was mixed with 90 mL of AgNO3 solution at each concentration in a sterile flask. The mixture was sonicated for 60 minutes at 40 kHz and 30°C, then incubated on a shaker at 200 rpm and 37°C for 48 hours. A color change of the solution to dark brown indicated the formation of silver nanoparticles.
Characterization of Silver Nanoparticles
PSA was used to measure the size and distribution of nanoparticles based on the principle of Photon Correlation Spectroscopy. Small particles in suspension move randomly and are analyzed based on their movement patterns.
SEM was used to analyze the surface morphology of silver nanoparticles. Magnification was performed at 5,000x and 10,000x to obtain detailed structural information.
FTIR was used to identify the functional groups in the nanoparticles. The analysis was conducted in the wavelength range of 400-4000 cm⁻¹ to reveal the structural features of the nanoparticles.
The UV-Vis spectrophotometer was used to measure the stability of the nanoparticles based on UV-Vis absorption spectra. The analysis was conducted daily for five consecutive days at wavelengths between 400-450 nm.
Results
Biosynthesis of Silver Nanoparticles
The biosynthesis of silver nanoparticles was carried out using endophytic bacteria from the stems and roots of L. coromandelica. The bacteria used included Micrococcus (BM 2), Enterococcus (BM 4 and AM 2), and Azotobacter (AM 5). The biosynthesis process lasted for 48 hours with incubation on a shaker (200 rpm, 37°C) in the dark, followed by sonication for 1 hour at 40 kHz. A color change from cloudy to dark brown at certain concentrations indicated the formation of silver nanoparticles (AgNPs), as shown in Table 1, marked by the color change to dark brown (+). This result is consistent with other studies, which reported that a color change to dark brown or deep brown was observed after 24-48 hours of incubation. The color change is an indication of the formation of AgNPs.10
 |
Table 1: Color Change in the Biosynthesis Reaction of Silver Nanoparticles by Endophytic Bacteria |
Characterization of Silver Nanoparticles
Particle Size Analyzer (PSA)
The PSA test showed at Table 2 that the nanoparticle size falls within the nano range (1–100 nm).2
Table 2: Particle Size and Polydispersity Index (PI) of Silver Nanoparticles Synthesized by Endophytic Bacteria
| AgNPs Nanoparticle | Particle Size (nm) | Standard Deviation (nm) | Polydispersity Index (PI) |
| AgNPs AM 2 (Enterococcus) | 86.5 | 3.2 | 0.1600 |
| AgNPs AM 5 (Azotobacter) | 60 | 2.1 | 0.029673 |
| AgNPs BM 2 (Micrococcus) | 64.1 | 1.8 | 0.01835 |
| AgNPs BM 4 (Enterococcus) | 105.9 | 4.0 | 0.2573 |
The polydispersity index (PI) value indicates that the formed nanoparticles are relatively homogeneous, especially in BM 2 and AM 5.
Fourier Transform Infrared (FTIR)
FTIR analysis showed at Table 3, the presence of functional groups O-H, C-N, and Ag-O in all samples. The O-H group acts as a reducing and stabilizing agent, while the C-N and Ag-O groups indicate interactions between bacterial biomolecules and silver nanoparticles.
Table 3: Functional Groups and Wavelengths of Silver Nanoparticles Synthesized by Endophytic Bacteria
| Silver Nanoparticles | Functional Groups | Wavelength Range (cm⁻¹) | Wavelength (cm⁻¹) |
| AgNPs AM 2(Enterococcus) | O-H | 3200-3600 | 3450.25 |
| C-N | 1600-1650 | 1636.74 | |
| Ag-O | 400-600 | 575.38 | |
| AgNPs AM 5(Azotobacter) | O-H | 3200-3600 | 3450.32 |
| C-N | 1600-1650 | 1636.84 | |
| Ag-O | 400-600 | 573.70 | |
| AgNPs BM 2 (Micrococcus) | Ag-O | 400-600 | 576.19 |
| C-N | 1600-1650 | 1636.74 | |
| O-H | 3200-3600 | 3449.89 | |
| Ag-O | 400-600 | 574.93 | |
| AgNPs BM 4 (Enterococcus) | C-N | 1600-1650 | 1636.80 |
| O-H | 3200-3600 | 3449.59 |
UV-Vis Spectrophotometer
The stability of the nanoparticles was tested over 5 days with a maximum wavelength (λ max) around 425 nm. The absorbance values showed small fluctuations, indicating the stability of the nanoparticles.
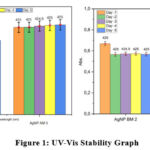 |
Figure 1: UV-Vis Stability GraphClick here to view Figure |
Based on Figure 1, the AgNP samples AM2 and AM5 exhibited good stability from day 1 to day 5. The absorbance values of the nanoparticles at the maximum wavelength (λ max) showed small fluctuations, with AgNP AM2 ranging from 0.55 to 0.57 and AgNP AM5 ranging from 0.67 to 0.68. This indicates that the amount of nanoparticles absorbing light did not change significantly during this period. However, these small fluctuations could be caused by factors such as nanoparticle aggregation and temperature changes.11
AgNPs BM2 (Micrococcus) and AgNPs BM4 (Enterococcus) showed on day 1 a wavelength of 425 nm with absorbance values of 0.66892 and 0.82033, respectively; on day 2, a wavelength of 425 nm with absorbance values of 0.56584 and 0.7329, respectively; on day 3, a wavelength of 424.9 nm with absorbance values of 0.5711 and 0.74567, respectively; on day 4, a wavelength of 425 nm with absorbance values of 0.57243 and 0.74317, respectively; and on day 5, a wavelength of 425 nm with absorbance values of 0.5683 and 0.7442, respectively.
Scanning Electron Microscopy (SEM)
The SEM results in Figure 2 show the morphology of the nanoparticles, which are needle-like crystals (BM 2 and BM 4) and irregular semi-spherical shapes (AM 2). The AM 5 nanoparticles have a homogeneous spherical shape. Minimal aggregation in AM 5 indicates better stability compared to the other samples
 |
Figure 2: Morphology SEM AgNPs Endophytic bacteria from roots (AM) and stem (BM) L. coromandelica |
Discussion
Biosynthesis of silver nanoparticles using endophytic bacteria results in a color change of the solution from cloudy to dark brown. This change indicates the reduction of silver ions (Ag⁺) to silver nanoparticles (Ag⁰) through the activity of biomolecules such as enzymes and secondary metabolites produced by the bacteria. This process is in accordance with studies that state that the enzyme nitrate reductase plays a key role in catalyzing the reduction reaction.12 The biosynthesis process is carried out through an extracellular method, where bioactive compounds such as enzymes and proteins act as biocatalysts.13 Sonication for 1 hour at 40 kHz helps break down protein aggregates and nanoparticles into smaller and more homogeneous units. This allows for uniform particle distribution, supporting the stability of the nanoparticle solution. Differences in AgNO₃ concentration affect the efficiency of nanoparticle formation. At low concentrations (0.5–1 mM), the reduction of silver ions is suboptimal, as seen from the solution color not changing significantly. In contrast, at concentrations of 2–3 mM, stable nanoparticles are formed, as indicated by the color change to dark brown. This optimal concentration shows that the available silver ions are sufficient to interact with the reducing biomolecules without causing excessive aggregation.
PSA analysis results show that the nanoparticle size ranges from 60–105.9 nm, with a polydispersity index (PI) of < 0.5 for most samples. Smaller particle sizes, such as those of AM 5 (60 nm), indicate that these bacteria produce enzymes and metabolites that are more effective in controlling nanoparticle growth. Nanoparticle homogeneity is important for biomedical applications, as uniform particles tend to have consistent physicochemical properties. The differences in nanoparticle size between BM 2 (64.1 nm) and BM 4 (105.9 nm) may be due to variations in the types of biomolecules produced by each bacterium. Bacteria with more specific enzymes or higher concentrations of secondary metabolites tend to produce smaller nanoparticles with higher stability.14,15
FTIR analysis shows the presence of functional groups O-H, C-N, and Ag-O, which play an important role in the biosynthesis process. O-H group: Derived from hydroxyl compounds in secondary metabolites such as polyphenols, this group acts as a reducing agent that converts Ag⁺ to Ag⁰. Additionally, the O-H group helps stabilize nanoparticles through hydrogen bonding with the nanoparticle surface.16 C-N group: This group indicates the involvement of proteins in the reduction and stabilization process. Proteins with specific amino acid residues, such as serine and threonine, can bind to silver ions, forming a stable complex before reduction occurs.17 Ag-O group: This bond indicates direct interaction between silver nanoparticles and bacterial biomolecules, which strengthens the stability of the nanoparticles, supported by Kasim’s research.15 This study reported that the FTIR spectrum of silver nanoparticles showed absorption at a wave number of 364 cm⁻¹, identified as Ag-O bond vibrations. This indicates the interaction between silver nanoparticles and biomolecules from water hyacinth leaf extract used as a bioreductor. The presence of these groups indicates that the biosynthesis of silver nanoparticles involves not only chemical reduction but also complex biological interactions between silver ions and bacterial biomolecules.18
UV-Vis spectrophotometer measurements show that the nanoparticles have good stability for 5 days. The maximum wavelength (λ max) remains constant at around 425 nm with small absorbance fluctuations. This indicates that the nanoparticles did not experience significant aggregation during the testing period. Small fluctuations in absorbance values may be due to temperature changes or minor aggregation during storage. However, the low standard deviation values indicate that the nanoparticles remained stable under test conditions19. This stability is important for biomedical applications, where nanoparticles must maintain their properties during storage or use.
SEM results show that the nanoparticle morphology varies depending on the type of bacteria used. BM 2 and BM 4: Needle-like crystals with a large surface area, suitable for catalytic applications. AM 5: Homogeneous spherical shape with a smooth surface, ideal for biomedical applications due to better stability. Aggregation observed in some samples, such as AM 2, may be caused by incomplete nanoparticle separation during centrifugation. This aggregation may affect the physical and chemical properties of the nanoparticles, including their size and homogeneity.20 The silver nanoparticles produced have broad application potential, particularly in the biomedical field. High antibacterial activity, supported by the ability of AgNPs to generate reactive oxygen species (ROS) that damage bacterial cell membranes. Catalysis, the needle-like shape of BM 2 and BM 4 with a large surface area enhances efficiency in catalytic reactions. Nanotechnology, the stability of nanoparticles for 5 days shows potential for use in nanotechnology-based product formulations.
Conclusion
Based This study demonstrated that endophytic bacteria isolated from the roots and stems of Lannea coromandelica are capable of biosynthesizing silver nanoparticles (AgNPs), with physicochemical characteristics influenced by both bacterial strain and silver nitrate (AgNO₃) concentration. At 3 mM AgNO₃, AM 2 (Enterococcus) and AM 5 (Azotobacter) successfully synthesized AgNPs, with AM 5 producing smaller, more uniform nanoparticles (60 nm, spherical, smooth-edged) compared to AM 2 (86.5 nm, semi-spherical, aggregated). Similarly, BM 2 (Micrococcus) and BM 4 (Enterococcus) from stem tissues showed positive AgNP formation at 2 mM, indicated by a dark brown color change and confirmed through PSA, UV-Vis, FTIR, and SEM analyses. AgNPs from BM 2 had a size of 64.1 nm with a low polydispersity index (PI = 0.01835), while BM 4 produced larger particles (105.9 nm, PI = 0.2573). Across all samples, FTIR detected O-H, C-N, and Ag-O functional groups, suggesting the role of bacterial metabolites in nanoparticle formation and stabilization.
These findings highlight the potential of L. coromandelica-derived endophytes as eco-friendly biological agents for silver nanoparticle biosynthesis. While the AgNPs produced exhibit characteristics suitable for further development, their application in nanomedicine or antimicrobial systems should be explored through additional biological and functional validation studies.
Acknowledgment
The authors would like to thank Universitas Negeri Makassar for supporting this research.
Funding Source
This work was supported by PNBP funding from the Professor Council and the Postgraduate Program, Universitas Negeri Makassar, Number SP DIPA – 023.17.2.677523/2025
Conflicts of Interest
The authors declare that they have no conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Authors Contribution
- Yusminah Hala carried out the laboratory work, conceived the idea, supervised the project, and facilitated funding acquisition.
- A Irma Suryani supervised the project, reviewed, and edited the draft.
- Hartono, Muhammad Junda and Rachmawaty Muhtar provided input and suggestions for the research.
- Andi Annisa and Andi Fahira performed bacterial isolation and laboratory testing.
References
- Rani NJ, Sharma P, Gangwar A. Role and application of nanostructures in biotechnology. Nanoscience Nanotechnology Asia. 2023;13.
CrossRef - Salari P, Ghaffari Moghaddam M, Bahreini M, Sharifmoghadam MR. Green synthesis of ZnO nanoparticles from Foeniculum vulgare Mill. seed extract and its antibacterial effects on foodborne bacteria. J Med Plants. 2023;22(86):14–26.
CrossRef - Du J, Hu Z, Dong WJ, Wang Y, Wu S, Bai Y. Biosynthesis of large-sized silver nanoparticles using Angelica keiskei extract and its antibacterial activity and mechanisms investigation. Microchem J. 2019;147:333–8.
CrossRef - Yin IX, Zhang J, Zhao IS, Mei ML, Li Q, Chu CH. The antibacterial mechanism of silver nanoparticles and its application in dentistry. Int J Nanomedicine. 2020;15:2555–62.
CrossRef - Alkhulaifi MM, Alshehri JH, Alwehaibi MA, Awad MA, Al-Enazi NM, Aldosari NS, et al. Green synthesis of silver nanoparticles using Citrus limon peels and evaluation of their antibacterial and cytotoxic properties. Saudi J Biol Sci. 2020;27(12):3434–41.
CrossRef - Singh M, Kumar A, Singh R, Pandey KD. Endophytic bacteria: a new source of bioactive compounds. Biotech. 2017;7(3):1–14.
CrossRef - Ghafarzadegan R, Yaghoobi M, Momtaz S, Ashoory N, Ghiaci Yekta M, Hajiaghaee R. Process optimization for green synthesis of iron nanoparticles by extract of fenugreek (Trigonella foenum-graecum L.) seeds. J Med Plants. 2022;21(81):22–32.
CrossRef - Hala Y, Ali A, Suryani AI. Indole acetic acid (IAA) production by endophytic bacteria from stem and roots of Indian ash tree (Lannea coromandelica). Bionature. 2024;25(2):52–62.
CrossRef - Barkah DC, Ahmad F, Putri DIS, Ningsih SNR, Yuniarsih N. Nanoparticles as a targeted drug delivery system through injection. J Bidang Ilmu Kesehatan. 2023;13(3):288–95.
CrossRef - El-Deeb NM, El-Sherbiny IM, El-Aassar MR, El Gohary MI. Novel alginate-based nanoparticle adheres to stomach mucosa and has anti-Helicobacter pylori activity. Carbohydr Polym. 2014;112:280–8.
- Elraies SSE, Adaila KE, Ahmad NA, Omar AA, Mhanna WE, Shaban MA, et al. Exploiting pear leaves in biosynthesis of silver nanoparticles. J Appl Sci Technol. 2023;11(1):1–10.
- Nefri FM, Djamaan RA. Biological synthesis of silver nanoparticles by bacteria and its characterizations: a review. IOSR J Agric Vet Sci. 2020;13:40–7.
- Nalawati AN, Suyatma NE, Wardhana DI. Silver nanoparticles (NPAg) synthesis with bioreductors of jatropha seed extracts and studies of antibacterial activities. J Teknol Ind Pangan. 2021;32(2):98–106.
CrossRef - Mukhopadhyay R. Green synthesis of silver nanoparticles using Azadirachta indica (neem) leaf extract and evaluation of their antimicrobial activity. Nanoscience Nanotechnology. 2014;4(2):30–5.
- Kasim S, Paulina T, Ruslan, Romianto. Silver nanopartikel synthesis uses water hyacinth leaf extract (Eichornia crassipes) as a bioredux. Kovalen: J Riset Kimia. 2020;6(2):126–33.
CrossRef - Kalishwaralal K, Deepak V, Pandian SBRK, Kottaisamy M, BarathManiKanth S, Kartikeyan B, et al. Biosynthesis of silver and gold nanoparticles using Brevibacterium casei. Colloids Surf B Biointerfaces. 2010;77(2):257–62.
CrossRef - Singh M, Kumar A, Singh R, Pandey KD. Endophytic bacteria: a new source of bioactive compounds. Biotech. 2017;7(3):1–14.
CrossRef - Rusnaenah R, Suhartono E, Arfianti D. Silver nanoparticle synthesis uses guava leaf extract (Psidium guajava L.) and antibacterial activity test. J Kimia Riset. 2016;1(1):37–44.
- Apriandanu DOB, Wahyuni S, Hadisaputro S. Silver nanoparticle synthesis uses the polyol method with a polivinylalkohol stabilizer agent (PVA). Indones J Math Nat Sci. 2013;36(2):157–68.
- Zhang XF, Liu ZG, Shen W, Gurunathan S. Silver nanoparticles: synthesis, characterization, properties, applications, and therapeutic approaches. Int J Mol Sci. 2016;17(9):1534.
CrossRef
Abbreviations list
AgNPs: Silver Nanoparticles
PSA: Particle Size Analyzer
FTIR: Fourier Transform Infrared Spectroscopy







