Manuscript accepted on :05-09-2025
Published online on: 22-09-2025
Plagiarism Check: Yes
Reviewed by: Dr. Karthikeyan and Dr. Samiya Faruq
Second Review by: Dr. Armerinayanti Pranata
Final Approval by: Dr. Anton R Keslav
Afraa Asif1 , Yusra Khan1
, Yusra Khan1 , Manahel Khuram1
, Manahel Khuram1 , Mohammed Khalil1
, Mohammed Khalil1 , Sunil Bylappa2
, Sunil Bylappa2 and Abid Shaheer1*
and Abid Shaheer1*
1Department of Biomedical Sciences, College of Medicine, Gulf Medical University, United Arab Emirates.
2 Department of Pathology, Thumbay Labs, Gulf Medical University, United Arab Emirates.
Corresponding Author E-mail:abidshaheer@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3267
Abstract
This prospective comparative study evaluated the diagnostic accuracy of Elastic Tissue Staining (ETS) versus Hematoxylin and Eosin (H&E) staining for detecting venous invasion in colorectal, breast and thyroid malignancies, with emphasis on clinical implications. Archived cancer specimens from 94 patients with histologically confirmed colorectal (n=30), breast (n=32) and thyroid cancers (n=32) were analyzed. ETS, performed using Verhoeff-Van Gieson (VVG) staining, was compared with consensus H&E staining, which is considered the gold standard for diagnosis. Diagnostic performance was assessed using sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), accuracy, the Chi-square test, the Phi coefficient and McNemar's test. ETS demonstrated perfect sensitivity (100%) and NPV (100%) across all cancer types, effectively eliminating false-negative results. Specificity is highest in colorectal cancer (80%), moderate in breast cancer (63.6%) and lowest in thyroid cancer (50%). ETS achieved significantly higher diagnostic accuracy (91.3%, p<0.001) when compared to H&E, particularly its superior detection of venous invasion in thyroid cancers (81.2% with ETS vs. 62.5% with H&E, p=0.031). ETS substantially improves the diagnostic accuracy of venous invasion compared with conventional H&E staining, ensuring no false-negative cases and enhancing clarity in histologically challenging tissues, especially thyroid cancer. Although specificity varied, the consistently exceptional sensitivity strongly supports the routine integration of ETS into pathological practice to improve staging accuracy, prognostic assessment and clinical management.
Keywords
Breast Neoplasms; Colorectal Neoplasms; Elastic Tissue; Thyroid Neoplasms; Venous Invasion
Download this article as:| Copy the following to cite this article: Asif A, Khan Y, Khuram M, Khalil M, Bylappa S, Shaheer A. Elastic Tissue Staining Significantly Enhances Detection of Venous Invasion Compared to Conventional H and E Staining in Colorectal, Breast and Thyroid Carcinomas. Biomed Pharmacol J 2025;18(3). |
| Copy the following to cite this URL: Asif A, Khan Y, Khuram M, Khalil M, Bylappa S, Shaheer A. Elastic Tissue Staining Significantly Enhances Detection of Venous Invasion Compared to Conventional H and E Staining in Colorectal, Breast and Thyroid Carcinomas. Biomed Pharmacol J 2025;18(3). Available from: https://bit.ly/3KisEMx |
Introduction
Vascular invasion is defined as the infiltration of malignant tumor cells into blood vessels, particularly veins, due to their thinner walls and is a critical histopathological indicator of tumor aggressiveness and metastatic potential.¹ Vascular invasion represents a complex biological process involving key molecular and cellular events, such as epithelial-mesenchymal transition, extracellular matrix degradation, tumor cell intravasation and eventual colonization of distant anatomical sites.¹˒² The presence of vascular invasion profoundly influences cancer staging, prognosis and treatment across various malignancies. Venous invasion is an independent prognostic factor in colorectal cancer and is closely associated with adverse outcomes such as hepatic metastasis, often necessitating aggressive adjuvant chemotherapy.²˒³ Venous invasion serves as a strong predictor of axillary lymph node involvement and distant dissemination in breast cancer, thereby guiding systemic therapeutic decisions, including chemotherapy and hormonal therapy.⁴˒⁵ The identification and extent of venous invasion in thyroid carcinoma significantly affect tumor classification, risk stratification and therapeutic strategies, guiding decisions on radioactive iodine therapy and surgical management.⁶ Moreover, microvascular invasion in hepatocellular carcinoma strongly correlates with early recurrence and reduced overall survival, underscoring the prognostic gravity of vascular invasion detection across multiple cancer types.¹˒⁷
Despite its clear prognostic importance, accurate detection of venous invasion using conventional Hematoxylin and Eosin (H&E) staining remains diagnostically challenging. Subtle foci of vascular invasion are frequently overlooked or misinterpreted as retraction artifacts or stromal clefts, resulting in diagnostic inconsistencies and variability among pathologists.⁸ This limitation risks underdiagnosing venous invasion, potentially compromising cancer staging accuracy and clinical management decisions. Recognizing these diagnostic challenges, specialized histochemical staining techniques such as Elastic Tissue Staining (ETS), particularly Verhoeff-Van Gieson (VVG), have been developed and refined to enhance diagnostic sensitivity and reproducibility. VVG provides superior contrast, clear visualization of elastic fibers and greater reliability in distinguishing true venous invasion from artifacts, making it the preferred technique over other elastic tissue staining methods in routine diagnostic practice. ETS distinctly highlights elastic fibers within vascular walls, effectively delineating vessel boundaries and thereby significantly improving identification of tumor emboli within veins.⁹
Previous studies have demonstrated the diagnostic superiority of ETS in colorectal cancer by revealing venous invasion in cases initially considered negative by standard H&E, thereby enabling more accurate cancer staging and improved prognostication.²˒³ Elastin staining also significantly improves venous invasion detection, raising detection rates from 44% with H&E alone to approximately 56%, thereby substantially influencing adjuvant treatment decisions in clinical practice.⁹ Additionally, ETS has shown promise in reducing interobserver variability by providing clear, reproducible histological markers of vessel boundaries, thus increasing diagnostic consistency among pathologists.² The accurate identification of venous invasion has profound implications: in node-negative colorectal cancer, confirmed venous invasion may result in disease upstaging and indicate the necessity for adjuvant chemotherapy.² In thyroid cancer, the precise quantification of venous invasion critically influences decisions regarding radioactive iodine therapy and aggressive surgical management.⁶ Likewise, reliable detection of vascular invasion in breast cancer directly impacts systemic chemotherapy or the extension of endocrine therapy, potentially altering patient outcomes significantly.⁴
Furthermore, reduced expression of fibulin-2, a protein integral to maintaining the structural integrity of elastic fibers, is associated with increased vascular invasion and decreased recurrence-free survival in breast cancer patients. This observation underscores the biological significance of elastic fibers in vascular integrity, with the loss of fibulin-2 potentially facilitating malignant cell invasion into blood vessels.¹⁰ ETS techniques such as Elastic Van Gieson or Orcein stains have proved particularly valuable in challenging thyroid cancer cases, where routine H&E staining frequently yields inconclusive results. These specialized stains substantially enhance visualization of the elastic lamina within venous walls, allowing definitive differentiation between true venous invasion and histological artifacts.¹¹ Specifically, in thyroid tumors exhibiting follicular patterns, elastic stains reliably identify venous invasion even when vessel walls are markedly distorted by tumor infiltration or surrounding inflammation, providing robust histological evidence of malignancy.¹²
Despite their clear diagnostic advantages, elastin-based staining techniques remain inconsistently utilized outside gastrointestinal malignancies, representing a critical gap in standardized pathological practice.¹³ Consequently, the primary objective of this study is to systematically evaluate and directly compare the diagnostic accuracy and clinical utility of ETS against standard H&E staining in detecting venous invasion across colorectal, breast and thyroid malignancies. By clarifying the added diagnostic value of ETS, this study seeks to advocate for its routine integration into pathological evaluations, ultimately promoting improved cancer staging accuracy, treatment strategies and patient outcomes.
Materials and Methods
Study Design and Ethical Approval
This study utilized a prospective, observational, comparative design, allowing real-time data collection and systematic comparison of staining techniques for detecting venous invasion. Ethical approval was secured from the Institutional Review Board (IRB) and the University Research and Ethics Committees at Gulf Medical University (Approval No: IRB-COM-STD-11-JULY-2024), ensuring adherence to ethical standards, patient confidentiality and robust research governance protocols.
Study Population
The study population comprised 94 archived formalin-fixed, paraffin-embedded (FFPE) tissue specimens retrospectively collected from patients histologically diagnosed with colorectal (n=30), breast (n=32), or thyroid carcinomas (n=32) at Thumbay Laboratory, Gulf Medical University, from September 2024 to April 2025. Inclusion criteria were explicitly defined as FFPE resection specimens histologically confirmed as malignant tumors, specifically of colon, breast, or thyroid origin. Specimens were systematically excluded if they represented benign lesions, malignancies from other tissues, contained insufficient viable tumor tissue, demonstrated extensive necrosis, exhibited poor slide quality, or lacked comprehensive clinical and pathological documentation, as these factors could negatively affect the reliability of vascular invasion assessments.
Histological Processing and Staining Procedures
Standardized histological processing was performed following validated laboratory protocols and in accordance with College of American Pathologists (CAP) guidelines to guarantee reproducibility and diagnostic accuracy. Tissue grossing was conducted using the GrossPath 1500 grossing station. Subsequently, specimens underwent automated processing with STP 120 Micron and ASP6025 Leica tissue processors, followed by embedding with the EC-350-1-2 embedding system. Serial tissue sections of 4 µm thickness were prepared using a HistoCore HM-325 microtome. Routine H&E staining was executed using an automated HMS760x robotic stainer for baseline assessment. To enhance the precision of vascular invasion detection, VVG special staining was performed on additional serial sections from each tissue block to distinctly visualize elastic fibers within vessel walls.
Microscopic Evaluation and Diagnostic Consensus
Microscopic evaluation was conducted using a BX-51 high-resolution light microscope, facilitating detailed visualization of tissue morphology. Diagnostic assessment was executed in two stages: initially, all H&E-stained slides were independently evaluated by an experienced senior pathologist to establish baseline consensus diagnoses for venous invasion. Subsequently, the same slides underwent re-assessment after ETS, first by the senior pathologist and subsequently by four trained student researchers. Each researcher independently examined both H&E and corresponding ETS slides, following identical assessment sequences to ensure methodological consistency. All staining procedures and slide preparations were rigorously supervised by qualified laboratory personnel.
To minimize interobserver variability, structured training sessions led by the senior pathologist were conducted. Training emphasized essential morphological criteria for reliably identifying venous invasion on both H&E and ETS slides, focusing explicitly on distinguishing true venous invasion from histological artifacts. Periodic cross-validation sessions were held to monitor interobserver reliability, ensuring excellent agreement (Cohen’s Kappa statistic ≥0.8).
Statistical Analysis
The primary aim was to ascertain whether ETS improved diagnostic sensitivity, specificity and clarity of venous invasion detection compared to conventional H&E staining in colorectal, breast and thyroid carcinomas. The diagnostic performance of ETS was systematically compared across staining techniques and among multiple observers to comprehensively evaluate its added diagnostic value. Enhanced diagnostic visibility, consistently reported by both pathologists and researchers, was interpreted as evidence supporting the routine integration of ETS in clinical practice.
Statistical analyses included calculations of sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV) and overall diagnostic accuracy. Comparative analysis utilized Chi-square and Phi coefficient tests to evaluate the strength and statistical significance of associations between ETS and consensus H&E diagnoses. The McNemar’s test was specifically employed to assess the statistical significance of diagnostic discrepancies between the two staining methods. All statistical analyses were performed using IBM SPSS software (Version 28.0), with a significance threshold defined at p-values <0.05.
Results
The study cohort comprised 94 participants diagnosed with breast, colorectal and thyroid malignancies. The mean age of the participants was 51.59 years (SD ±18.17), with a median age of 52.5 years and an age range spanning from 17 to 86 years, thereby representing a wide spectrum of adult age groups. The most frequently represented age bracket was 51-60 years, accounting for 22.3% of the sample. Females constituted a majority of the cohort (59.6%), reflecting the inclusion of breast cancer cases. The distribution of cancer types was approximately balanced, with breast cancer comprising 34%, colorectal cancer 31.9% and thyroid cancer 34% of cases.
The poorly differentiated Grade 3 tumors were the most prevalent, representing 45.7% of cases, indicating a predominance of more aggressive tumor biology. In contrast, well-differentiated Grade 1 tumors accounted for the smallest proportion (22.3%). Tumor size was largely substantial, with 66% of tumors exceeding 4.0 cm in greatest dimension, suggesting that many patients presented with a considerable tumor burden. Consistent with this, nearly 47% of cases were classified as Stage III, underscoring the predominance of advanced disease within this patient population. The diagnostic accuracy of ETS for identifying venous invasion was evaluated against the consensus H&E staining method, regarded as the gold standard. The key findings of this comparative analysis are summarized in Table 2.
Across all three cancer types, ETS demonstrated a remarkable sensitivity of 100%, indicating that no cases of venous invasion were missed. This perfect sensitivity underscores the robust ability of ETS to detect all true positive cases, effectively eliminating false negatives, a critical factor for guiding clinical management and therapeutic decision-making. The specificity of ETS reflects its ability to correctly identify patients without venous invasion, which varies among cancer types. Colorectal cancer exhibited the highest specificity at 80%, indicating limited false positives and high diagnostic clarity. In breast cancer, specificity was moderate at 63.6%, suggesting a moderate proportion of false-positive findings. Thyroid cancer showed the lowest specificity at 50%, implying that ETS may tend to overestimate venous invasion in this histologically complex tissue type. Despite this variation, ETS achieved an exceptional NPV of 100% across all cancer types, meaning that a negative ETS result reliably excludes venous invasion. The PPV ranged from 76.9% in thyroid cancer to 96.2% in colorectal cancer, indicating that positive ETS results are generally trustworthy, particularly in colorectal tumors.
Diagnostic accuracy, defined as the overall proportion of correctly classified cases (true positives and true negatives), further supports the clinical utility of ETS. Colorectal cancer cases showed the highest accuracy at 96.9%, reflecting a combination of excellent sensitivity and specificity. Breast cancer demonstrated an accuracy of 93.3%, indicating strong performance despite a slightly increased false positive rate. Thyroid cancer exhibited an accuracy of 86.7%, which, while comparatively lower, still represents a substantial diagnostic capability given the tissue’s complex architecture. ETS achieved an overall diagnostic accuracy of 91.3%, underscoring its consistent reliability across diverse histologies.
Table 1: Descriptive Statistics of the Study Cohort
| Variable | Category | Frequency (n) | Percentage (%) |
| Age Group | <20 years | 4 | 4.2 |
| 21-30 years | 12 | 12.8 | |
| 31-40 years | 10 | 10.6 | |
| 41-50 years | 17 | 18.1 | |
| 51-60 years | 21 | 22.3 | |
| 61-70 years | 14 | 14.9 | |
| 71-80 years | 10 | 10.6 | |
| >80 years | 6 | 6.4 | |
| Gender | F | 56 | 59.6 |
| M | 38 | 40.4 | |
| Cancer Type | Breast Cancer | 32 | 34 |
| Colorectal Cancer | 30 | 31.9 | |
| Thyroid Cancer | 32 | 34 | |
| Tumor Grade | 1 | 21 | 22.3 |
| 2 | 30 | 31.9 | |
| 3 | 43 | 45.7 | |
| Tumor Size (cm) | <2.0 cm | 20 | 21.3 |
| 2.0-4.0 cm | 12 | 12.8 | |
| >4.0 cm | 62 | 66 | |
| Tumor Stage | I | 20 | 21.3 |
| II | 30 | 31.9 | |
| III | 44 | 46.8 |
These results confirm that ETS is highly effective in detecting venous invasion, particularly in colorectal and breast cancers and remains a valuable adjunct in thyroid cancer despite some limitations in specificity. The combination of perfect sensitivity, high accuracy and a definitive negative predictive value renders ETS a dependable screening and diagnostic tool. Statistical validation using Chi-square tests and Phi coefficient analyses (Table 2) further reinforces these findings. All cancer types exhibited statistically significant associations between ETS and H&E diagnoses, with p-values less than 0.05. Phi coefficients ranged from 0.612 to 0.859, reflecting moderate to strong correlations and demonstrating the substantial concordance between the two staining modalities.
Table 2: Sensitivity and Specificity Analysis of Elastic Tissue Staining vs. Consensus H&E
| Cancer Type | Sensitivity (%) | Specificity (%) | PPV (%) | NPV (%) | Accuracy (%) | Chi-square (χ²) | Phi Coefficient | p-value |
| Colorectal (n=32) | 100 | 80.0 | 96.2 | 100 | 96.9 | 23.85 | 0.859 | < 0.001 |
| Breast (n=30) | 100 | 63.6 | 90.9 | 100 | 93.3 | 15.32 | 0.716 | < 0.001 |
| Thyroid (n=30) | 100 | 50.0 | 76.9 | 100 | 86.7 | 11.25 | 0.612 | 0.001 |
| Total (n=92) | 100 | 60.7 | 88.6 | 100 | 91.3 | 48.74 | 0.728 | < 0.001 |
ETS demonstrated a superior detection rate for venous invasion across the entire cohort, identifying 82% of cases compared to 70% with H&E staining. This difference was statistically significant (p < 0.001), highlighting the enhanced diagnostic sensitivity of ETS. When analyzed by cancer subtype, ETS maintained its advantage compared to H&E staining. In thyroid cancer, ETS detected venous invasion in 81.2% of cases, compared to 62.5% with H&E. This difference reached statistical significance (p = 0.031), underscoring the particular utility of ETS in a tissue type known for its complex vascular and stromal architecture. In colorectal cancer, ETS detected venous invasion in 86.7% of cases, slightly higher than the 83.3% detected by H&E. However, this difference was not statistically significant (p = 1.000), suggesting that conventional H&E staining already provides a high diagnostic yield in this subgroup. For breast cancer, ETS identified venous invasion in 78% of cases compared to 66% with H&E. While the trend favoured ETS, the difference did not reach statistical significance (p = 0.125), which may be attributed to a modest sample size or interpretive challenges associated with breast tumor histology. Importantly, in no instance was a case positive by H&E but negative by ETS. This observation confirms that ETS did not miss any case of venous invasion identified by H&E, reinforcing its role as a superior or complementary diagnostic tool. These findings collectively support the incorporation of ETS into routine histopathological evaluation, particularly in cases where venous invasion has critical prognostic and therapeutic implications.
Table 3: Diagnostic Agreement between Consensus H&E and Consensus Elastic Staining
| Cancer Type | Total Pairs | Discordant Pairs | H&E Detection % | Elastic Detection % | Sig.
(2-tailed) |
| All Cases | 94 | 11 | 70% | 82% | < 0.001 |
| Thyroid Cancer | 32 | 6 | 62.5% | 81.2% | 0.031 |
| Colorectal Cancer | 30 | 1 | 83.3% | 86.7% | 1.000 |
| Breast Cancer | 32 | 4 | 66% | 78% | 0.125 |
Representative H&E-stained sections for breast, colorectal and thyroid carcinomas are shown below, depicting cases with and without histologically confirmed venous invasion. These sections were critically reviewed during the diagnostic assessment and served as the reference standard for validating vascular involvement.
Figure 1 shows that blood vessels appear collapsed or distorted due to surrounding stromal fibrosis, but no tumor emboli are visible within the vascular lumen. The vessel walls are thin and difficult to delineate, which limits diagnostic clarity.
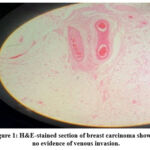 |
Figure 1: H&E-stained section of breast carcinoma showing no evidence of venous invasion. |
Figure 2 indicates that a cluster of tumor cells is seen within a vessel-like space. However, due to the absence of a clearly defined endothelial or elastic layer, it is difficult to confirm true venous invasion versus a retraction artifact. This illustrates one of the key limitations of H&E in identifying venous invasion with certainty.
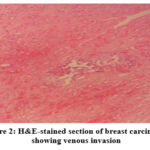 |
Figure 2: H&E-stained section of breast carcinoma showing venous invasion |
Figure 3 shows that several blood vessels are observed adjacent to the tumor margin, but their lumens are free of tumor cells. The endothelial lining appears intact and no disruption is evident, suggesting a true negative for vascular involvement.
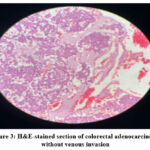 |
Figure 3: H&E-stained section of colorectal adenocarcinoma without venous invasion |
Figure 4 indicates that tumor cells appear to be present within a vascular structure. However, the lack of a well-defined elastic lamina makes it difficult to distinguish between true vessel invasion and interstitial spread. Further staining is often required to confirm invasion.
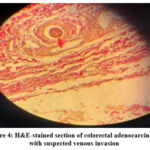 |
Figure 4: H&E-stained section of colorectal adenocarcinoma with suspected venous invasion |
Figure 5 shows that small blood vessels are compressed within the fibrotic stroma and no tumor cells are observed within their lumens. The thin and fragmented nature of vessels in thyroid tissue makes assessment particularly challenging with H&E alone.
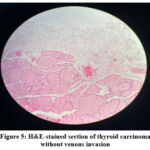 |
Figure 5: H&E-stained section of thyroid carcinoma without venous invasion |
Figure 6 indicates a suspicious cluster of tumor cells partially filling a space that may represent a vessel. The confirmation is difficult due to indistinct vessel wall features and surrounding fibrosis, which highlights that thyroid tissue architecture may obscure vascular landmarks in standard H&E staining.
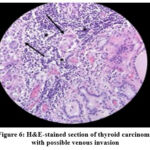 |
Figure 6: H&E-stained section of thyroid carcinoma with possible venous invasion |
H&E staining often showed limited clarity in detecting venous invasion across breast, colorectal and thyroid cancers. In many cases, vessel walls were poorly defined and distinguishing true invasion from artifacts was challenging. These findings highlight that while H&E is widely used, it may not reliably reveal venous invasion, emphasizing the need for complementary stains like ETS for more accurate diagnosis. ETS was applied to sections of breast, colorectal and thyroid cancers to assess venous invasion. In each case, this special stain enhanced the contrast between vessel walls and surrounding tissues by selectively highlighting elastic fibers in a distinct blue-black color. This allowed for precise delineation of vascular structures, particularly the internal elastic lamina, which is often difficult to appreciate with H&E alone. In the following images, the blue-black-stained vessel walls provide clear evidence of tumor cells within vascular lumina, confirming venous invasion. The enhanced visibility reduces ambiguity and helps distinguish true vascular invasion from mimics such as retraction artifacts or stromal clefts.
Figures 7 to 9 demonstrate how elastic staining significantly improves diagnostic confidence in identifying venous invasion by providing a strong contrast between vessel structures and surrounding tissue. The blue-black coloration of elastic fibers serves as a reliable marker to confirm the presence of tumor cells within true vascular spaces.
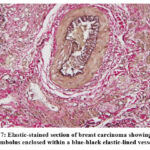 |
Figure 7: Elastic-stained section of breast carcinoma showing tumor embolus enclosed within a blue-black elastic-lined vessel |
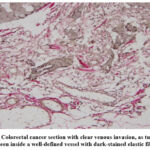 |
Figure 8: Colorectal cancer section with clear venous invasion, as tumor cells are seen inside a well-defined vessel with dark-stained elastic fibers. |
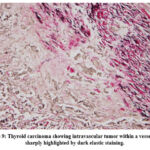 |
Figure 9: Thyroid carcinoma showing intravascular tumor within a vessel wall, sharply highlighted by dark elastic staining. |
Discussion
The descriptive analysis provided a comprehensive demographic and clinical profile of the 94 patients included, forming an essential foundation for interpreting subsequent analytical results. The broad age distribution (17-86 years; mean 51.59 years, median 52.50 years) ensured representation across the adult lifespan, consistent with epidemiological data indicating higher cancer incidence among middle-aged and older populations.14,15 The prominent concentration of patients (22.3%) aged 51-60 years is reflective of established age-related cancer risk profiles, with lower frequencies at extreme age groups aligning with typical clinical demographics observed in cancer registries.14 The females comprised the majority (59.6%) of the cohort, influenced by the inclusion of breast cancer cases. This gender distribution is clinically significant, as gender-based differences influence tumor biology, disease progression and the expression of diagnostic markers.16,17
The balanced representation of breast (34%), colorectal (31.9%) and thyroid cancers (34%) facilitated a robust comparative analysis across diverse malignancies characterized by distinct natural histories and patterns of vascular invasion.18,19 Nearly half (45.7%) of the cohort exhibited poorly differentiated Grade 3 tumors, indicative of aggressive tumor biology, aligning with recent data highlighting increased aggressiveness at advanced disease stages.20,21 Furthermore, the predominance of large tumors (>4.0 cm in 66% of cases) underscores significant tumor burden at diagnosis, associated with increased risk for vascular invasion and worse clinical outcomes.22,23 The stage distribution, predominantly advanced (Stage III, 46.8%), further emphasized the clinical complexity of this cohort and the critical importance of accurate and sensitive diagnostic approaches for detecting venous invasion.24 These demographic and clinical characteristics underscore the clinical need for effective vascular invasion detection methods to improve diagnostic accuracy, staging precision and informed clinical decision-making.25,26
This study underscored the considerable diagnostic utility of ETS in accurately detecting venous invasion across colorectal, breast and thyroid malignancies compared to conventional H&E staining. ETS demonstrated perfect sensitivity (100%) across all cancers, reliably identifying all true positive cases and ensuring no false negatives. ETS serves as an exceptionally reliable screening tool, providing absolute confidence in excluding venous invasion when results are negative, an essential attribute given the severe clinical consequences of missing venous invasion, such as inaccurate staging and suboptimal therapeutic interventions, as highlighted in recent studies.27,28
The sensitivity and NPV remain uniformly high and the specificity varies significantly based on the tumor. Colorectal cancer exhibited the highest specificity (80%), suggesting that ETS effectively minimized false-positive results and provided high diagnostic accuracy (96.9%) and PPV (96.2%). This finding confirms ETS as a robust adjunct tool for colorectal cancer pathology, consistent with previous research emphasizing its enhanced sensitivity and clinical utility in this setting, as explained in previous studies.16,29 Breast cancer demonstrated moderate specificity (63.6%) and slightly reduced PPV (90.9%), yet its consistently high NPV (100%) and significant statistical association (χ²=15.32, p<0.001; Φ=0.716) maintained clinical relevance, particularly in reliably excluding venous invasion when ETS was negative.
Thyroid cancer exhibited the lowest specificity (50%), indicating higher false-positive rates, likely influenced by histological complexity, dense stromal architecture and frequent presence of vessel-wall fragmentation or fibrosis, complicating definitive interpretation as stated in previous studies.30,31 ETS maintained perfect sensitivity and achieved statistical significance (p=0.001), underscoring its diagnostic value. Given these specificity variations, ETS findings in thyroid cancer must be carefully interpreted within morphological contexts and corroborated with additional diagnostic markers or clinical correlation to reduce interpretative ambiguity, as mentioned in previous studies.32,33
The significant statistical associations (p<0.001) and robust Phi coefficients (0.612-0.859) reinforced the diagnostic relevance of ETS and supported its broader clinical integration, particularly where venous invasion critically informs staging, prognosis and treatment planning. The aggregated diagnostic accuracy (91.3%) further established ETS as universally applicable, despite specificity challenges in certain cancers.
The superior detection rates by ETS (82%) compared to H&E (70%) clearly highlight the capability of ETS to reveal venous invasion otherwise obscured or ambiguous in standard H&E staining, a finding consistent with previous high-impact studies.²⁹˒³⁴˒³⁵ ETS significantly enhanced detection in thyroid cancers (81.2% vs. 62.5%, p=0.031), demonstrating particular utility in thyroid malignancies where subtle vascular features are frequently overlooked on routine stains. ETS showed only marginal and statistically non-significant improvements in colorectal cancer (86.7% vs. 83.3%, p=1.000) and breast cancer (78% vs. 66%, p=0.125). The consistent detection of additional cases supports its role as a complementary confirmatory method in diagnostically ambiguous scenarios, as discussed in recent studies.³⁶˒³⁷
The practical significance of additional positive findings by ETS remains a crucial topic for further investigation. Previous research underscores the prognostic significance of venous invasion detection methods, reinforcing the clinical imperative for reproducible and standardized techniques such as ETS, particularly in colorectal cancers, where elastic lamina invasion directly influences patient outcomes.³¹˒³⁸ The established prognostic importance of vascular invasion across cancers justifies routine incorporation of ETS in diagnostic workflows.³⁹˒⁴⁰ The integration of complementary immunohistochemical markers (e.g., CD31, D2-40) could further improve diagnostic accuracy, particularly in breast cancer, where subtle invasion is frequently overlooked.³⁷˒⁴¹ Such multimodal diagnostic strategies may also help reduce false positives in tissues with complex histomorphology or extensive fibrosis.
A critical limitation of the present study was the relatively small sample size in subgroup analysis, limiting statistical power, generalizability and interpretative robustness, particularly for thyroid and breast cancers. The single-centered design of the study could restrict the broader applicability of the findings due to variations in histological practice and patient demographics. Future research should therefore include larger multicenter cohorts with diverse histological subtypes, investigating the histopathological mechanisms underlying enhanced sensitivity of ETS while explicitly correlating diagnostic findings with clinical outcomes such as recurrence, metastasis and survival, to firmly establish the prognostic and clinical utility of ETS.
Conclusion
ETS consistently demonstrated superior sensitivity and high diagnostic accuracy for venous invasion detection across colorectal, breast and thyroid cancers, strongly supporting its routine integration into diagnostic practice. However, critical histological interpretation remains essential, as specificity varies by tumor type, highlighting the potential for false positives in histologically complex tissues. ETS emerges as a complementary diagnostic approach, providing critical diagnostic clarity and significantly enhancing pathological evaluation, while careful morphological interpretation remains essential to account for specificity limitations. ETS strengthens staging accuracy and optimizes patient management when integrated into routine diagnostic practice.
Acknowledgment
The authors extend their sincere gratitude to the laboratory staff of Thumbay Lab, Gulf Medical University, for their invaluable assistance and unwavering support throughout the research process.
Funding Source
This study was funded by Gulf Medical University, Ajman, United Arab Emirates (GMU/COM/PRO402/G8/2024-07-11).
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
The study was conducted in full compliance with the ethical guidelines of Gulf Medical University, Ajman, United Arab Emirates and approved by the Institutional Review Board (IRB-COM-STD-11-JULY-2024).
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Authors Contributions
- Abid Shaheer: Supervised the entire research process and ensured successful execution.
- Afraa Asif and Yusra Khan: Conducted the complete research work.
- Manahel Khuram and Mohammed Khalil: Conducted the complete research work.
- Sunil Bylappa: Ensured the accuracy and reliability of the results.
References
- Mattila K, Vainio P, Jaakkola P. Prognostic factors for localized clear cell renal cell carcinoma and their application in adjuvant therapy. Cancers (Basel). 2022;14(1):239.
CrossRef - Lee S, Go J, Ahn B, et al. Lymphovascular invasion is an independent prognostic factor in breast cancer irrespective of axillary node metastasis and molecular subtypes. Front Oncol. 2023;13:1269971.
CrossRef - Kim H, Choi G, Seo A, et al. Prognostic value of venous invasion detected by elastin stain may surpass lymph node status in colon cancer. Dis Colon Rectum. 2021;64(8):955-963.
CrossRef - Wong K, Barletta J. Challenges in Encapsulated Follicular-Patterned Tumors: How Much Is Enough? Evaluation of Nuclear Atypia, Architecture and Invasion. Surg Pathol Clin. 2023;16(1):27-44.
CrossRef - Bahmad H, Alloush F, Salami A, et al. Routine elastin staining improves venous invasion detection in colorectal carcinoma. Ann Diagn Pathol. 2023;66:152170.
CrossRef - Shalkamy M, Bae J, Lee C, et al. Oncological impact of vascular invasion in colon cancer might differ depending on tumor sidedness. J Minim Invasive Surg. 2022;25(2):53-62.
CrossRef - Sari A, Cyr D, Brar A, et al. Routine Elastin Staining in Surgically Resected Colorectal Cancer: Impact on Venous Invasion Detection and its Association With Oncologic Outcomes. Am J Surg Pathol. 2022;46(2):200-212.
CrossRef - Fujisawa M, Omori M, Doihara H, et al. Elastin and collagen IV double staining: A refined method to detect blood vessel invasion in breast cancer. Pathol Int. 2020;70(9):612-623.
CrossRef - Kato T, Tsukamoto S, Miyake M, et al. Prognostic impact of extramural venous invasion detected by contrast-enhanced CT colonography in colon cancer. BJS Open. 2024;8(1):zrad121.
CrossRef - Klingen T, Chen Y, Aas H, Wik E, Akslen L. Fibulin-2 expression associates with vascular invasion and patient survival in breast cancer. PLoS One. 2021;16(4):e0249767.
CrossRef - Misra S, Dhawan S, Badwal S, et al. Evaluation of the follicular patterned thyroid lesions based on the WHO 2022 criteria with an emphasis on the grey-zone lesions. Ann Diagn Pathol. 2024;71:152282.
CrossRef - WHO Classification of Tumours Editorial Board. Endocrine and Neuroendocrine Tumours. 5th ed. Lyon, France: International Agency for Research on Cancer; 2025.
- Yamazaki H, Sugino K, Katoh R, et al. New Insights on the Importance of the Extent of Vascular Invasion in Widely Invasive Follicular Thyroid Carcinoma. World J Surg. 2023;47(11):2767-2775.
CrossRef - Sung H, Ferlay J, Siegel R, et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71(3):209-249.
CrossRef - Pilleron S, Soto-Perez-de-Celis E, Vignat J, et al. Estimated global cancer incidence in the oldest adults in 2018 and projections to 2050. Int J Cancer. 2021;148(3):601-608.
CrossRef - Nevola R, Tortorella G, Rosato V, et al. Gender Differences in the Pathogenesis and Risk Factors of Hepatocellular Carcinoma. Biology (Basel). 2023;12(7):984.
CrossRef - Ckhert E, Lansinger O, Ritter V, et al. Breast Cancer Diagnosis, Treatment and Outcomes of Patients From Sex and Gender Minority Groups. JAMA Oncol. 2023;9(4):473-480.
CrossRef - Halada S, Casado-Medrano V, Baran J, et al. Hormonal Crosstalk Between Thyroid and Breast Cancer. Endocrinology. 2022;163(7):bqac075.
CrossRef - Shirouzu K, Hisaka T, Fujita F, Yoshida T, Koushi K. The clinical significance of elastic lamina invasion in patients with pStage II colorectal cancer: a notable prognostic indicator. World J Surg Oncol. 2024;22(1):274.
CrossRef - Park K, Hong Y, Choi Y, Kang J. Clinicopathologic characteristics of early-onset colorectal cancer. Ann Coloproctol. 2022;38(5):362-369.
CrossRef - Ye Z, Zhuang L, Liu X, Song M, Zhang J, Cao G. The clinical outcomes of patients with vascular invasion after deceased donor liver transplantation. J Gastrointest Oncol. 2024;15(4):1686-1697.
CrossRef - Zhang Y, Wang H, Zhao H, He X, Wang Y, Wang H. Prognostic significance and value of further classification of lymphovascular invasion in invasive breast cancer: a retrospective observational study. Breast Cancer Res Treat. 2024;206(2):397-410.
CrossRef - Xiao J, Shen K, Fan H, et al. Prognostic Value of Tumor Size in Gastric Cancer: A Retrospective Cohort Study Based on SEER Database. Int J Surg Pathol. 2023;31(7):1273-1282.
CrossRef - Lin Y, Liu T, Hu Y, et al. Assessment of vascular invasion of pancreatic ductal adenocarcinoma based on CE-boost black blood CT technique. Insights Imaging. 2024;15(1):293.
CrossRef - Vass D, Ainsworth R, Anderson J, Murray D, Foulis A. The value of an elastic tissue stain in detecting venous invasion in colorectal cancer. J Clin Pathol. 2004;57(7):769-772.
CrossRef - Gonzalez J, Bahmad H, Ocejo S, et al. The Usefulness of Elastin Staining to Detect Vascular Invasion in Cancer. Int J Mol Sci. 2023;24(20):15264.
CrossRef - Fei L, Patel S, Popa T, et al. Venous invasion detectable only by elastic stain shows weak prognostic value in colon cancer. Histopathology. 2024;84(6):1038-1046.
CrossRef - Chen K, Collins G, Wang H, Toh J. Pathological Features and Prognostication in Colorectal Cancer. Current Oncology. 2021; 28(6):5356-5383.
CrossRef - Kang J, Park H, Song I, et al. Detection of venous invasion in colorectal cancer: Reappraisal using elastin immunohistochemistry and its clinical significance. Histopathology. 2020;77(5):769-781.
- Zhou L, Wang J, Zhang X, et al. Prognostic Value of Preoperative NLR and Vascular Reconstructive Technology in Patients With Pancreatic Cancer of Portal System Invasion: A Real World Study. Front Oncol. 2021;11:682928.
CrossRef - Kirsch R, Messenger D, Riddell R, Pollett A. Venous invasion in colorectal cancer: impact of morphology and staining technique. Histopathology. 2013;62(5):751-762.
- Duan K, Chow B, Tsui W, et al. Impact of tissue sampling on detection of venous invasion in colorectal cancer: a prospective analysis. Histopathology. 2023;83(6):891-902.
CrossRef - Limberg J, Lee-Saxton Y, Egan C, et al. Perineural Invasion in Papillary Thyroid Cancer: A Rare Indicator of Aggressive Disease. Ann Surg Oncol. 2023;30(6):3570-3577.
CrossRef - Sung Y, Kim Y, Datuin A, Jang I, Sim J. Routine Elastic Staining Helps Detection of Vascular Invasion in Colorectal Cancer: A Comprehensive Analysis of T3 or Higher Tumors Without Lymph Node Metastasis. Anticancer Res. 2024;44(11):5059-5065.
CrossRef - Watanabe J, Ichimasa K, Kataoka Y, et al. Additional staining for lymphovascular invasion is associated with increased estimation of lymph node metastasis in patients with T1 colorectal cancer: Systematic review and meta-analysis. Dig Endosc. 2024;36(5):533-545.
CrossRef - Messenger D, Driman D, Kirsch R, et al. Routine elastic staining is required to detect vascular invasion in stage II colon cancer. Am J Surg Pathol. 2012;36(2):193-201.
- Vosough Z, Golbini S, Sharbatdaran M, Hosseini A. D2-40 A Helpful Marker in Assessment of Lymphatic Vessel Invasion in Carcinoma of Breast. Iran J Pathol. 2021;16(2):96-102.
CrossRef - Liu W, Sun L, Guan F, Wang F, Zhang G. Prognostic Value of Lymphovascular Invasion in Upper Urinary Tract Urothelial Carcinoma after Radical Nephroureterectomy: A Systematic Review and Meta-Analysis. Dis Markers. 2019;2019:7386140.
CrossRef - Hwang C, Lee S, Kim A, Kim Y, Ahn S, Park D. Venous Invasion in Colorectal Cancer: Impact of Morphologic Findings on Detection Rate. Cancer Res Treat. 2016;48(4):1222-1228
CrossRef - Fathi M, Taefehshokr S, Rashidi M, et al. Vascular mimicry: changing the therapeutic paradigms in cancer. Mol Biol Rep. 2020;47(6):4749-4765.
CrossRef - Wei Y, Jiao Z, Sun T, Lai Z, Wang X. Molecular Mechanisms Behind Vascular Mimicry as the Target for Improved Breast Cancer Management. Int J Womens Health. 2023;15:1027-1038.
CrossRef
Abbreviations List
ETS: Elastic Tissue Staining
H&E: Hematoxylin and Eosin
PPV: Positive Predictive Value
NPV: Negative Predictive Value
VVG: Verhoeff-Van Gieson
IRB: Institutional Review Board
FFPE: Formalin-Fixed, Paraffin-Embedded
CAP: College of American Pathologists
SPSS: Statistical Package for the Social Sciences







