Manuscript accepted on :02-09-2025
Published online on: 22-09-2025
Plagiarism Check: Yes
Reviewed by: Dr. Tolmas Hamroyev
Second Review by: Dr. Feng Li
Final Approval by: Dr. Prabhishek Singh
Nuha Khalid Neyazi1* , Hend Maarof Tag2,3
, Hend Maarof Tag2,3 , Naglaa Elshafey4
, Naglaa Elshafey4 and Nashwa Hagagy1,5
and Nashwa Hagagy1,5
1Department of Biology, College of Science and Arts at Khulis, University of Jeddah, Jeddah, Saudi Arabia
2Department of Nursing, College of Applied Medical Sciences, University of Jeddah, Jeddah, Saudi Arabia
3Department of Zoology, Faculty of Sciences, Suez Canal University, Egypt
4Botany and Microbiology Department, Faculty of Science, Arish University, Al-Arish, Egypt
5Botany and Microbiology Department, Faculty of Science, Suez Canal University, Ismailia, Egypt
Corresponding Author E-mail:nons-iris@outlook.com
DOI : https://dx.doi.org/10.13005/bpj/3258
Abstract
Biodegradable polyhydroxyalkanoates (PHAs), consisting of monomers and heteropolymers, this is an alternative for polymers generated by fossil fuels. Halophilic archaea produced polyhydroxyalkanoates (PHAs) as intracellular granules reserves energy in response to nutrients scarcity. Nevertheless, there is a scarcity of comprehensive understanding of these processes and the capacity of halophilic archaea to accumulate PHAs. This study focused on isolation of haloarchaea from Saudi Arabia's Red Sea solar saltern and investigate their growth and synthesis of polyhydroxybutyrate (PHB) under varying pH levels, temperatures, and NaCl concentrations. In addition, the study aimed to assess the accumulation of polyhydroxybutyrate (PHB) under nutrient-limited conditions utilizing cost-effective sources of carbon, like carboxymethyl cellulose (CMC), glycerol, glucose, starch and sucrose. Identification of candidate strains was achieved via 16S rRNA genome sequencing. Subsequently, the sequences were added to the NCBI GenBank and given accession numbers OQ366239-OQ366242.Haloferax sp. strain HSM17 produced the greatest quantity of PHB (50.94%) when starch was the only source of carbon. The best growth was shown at 65°C, a pH 7, and 10% salinity. The chemical combination of the biopolymer was evaluated by applying GC-MS. Hemolysis was then measured to determine the cytotoxicity of the extracted PHB towards red blood cells. The results showed a considerable decrease in damage to human red blood cells parallel to the positive control (P<0.05). Haloferax sp. strain HSM17 has great potential as a producer of polyhydroxybutyrate (PHB) using cheap carbon sources.
Keywords
Biodegradable materials; Cytotoxic assays; Extremophiles; Halophilic archaea; Microbial biopolymers; Saltern ecosystems
Download this article as:| Copy the following to cite this article: Neyazi N. K, Tag H. M, Elshafey N, Hagagy N. Biopolymer (Polyhydroxybutrates) Production from Promising Haloarchaea: Characterization, Optimization with in Vitro Cytotoxic Activity. Biomed Pharmacol J 2025;18(3). |
| Copy the following to cite this URL: Neyazi N. K, Tag H. M, Elshafey N, Hagagy N. Biopolymer (Polyhydroxybutrates) Production from Promising Haloarchaea: Characterization, Optimization with in Vitro Cytotoxic Activity. Biomed Pharmacol J 2025;18(3). Available from: https://bit.ly/4nHIBue |
Introduction
Undegradable plastic pollution is a significant universal problem needs urgently process. The high demand for plastic products, lack of proper recycling after use, and widespread environmental pollution require a fundamental change in plastics technologies to tackle this huge challenge.1,2 Bioplastics are composed of many elements and typically emphasize the use of renewable resources such as starch, cellulose, or bioethanol.3 Bioplastics are biopolymers that microorganisms store as carbon reserves for energy in the shape of intracellular granules.4 Polyhydroxyalkanoates (PHAs) are naturally occurring polyesters that accumulate as storage granules inside microorganisms. They possess physicochemical properties that are comparable to those of petrochemical polymers.5 The most popular PHAs are polyhydroxybutyrate (PHB) and polyhydroxybutyrate-co-hydroxy valerate (PHBV) because of their outstanding optical properties and remarkable UV resistance.6 These polymers exhibit distinctive characteristics and have been widely utilized in several industries, such as medicine (e.g., medical devices and medication delivery systems), food, agriculture, biofuels, and packaging.6 Polyhydroxyalkanoates (PHAs) are extensively produced by different organisms, such as bacteria, cyanobacteria, and archaea, as well as eukaryotes such as algae and plants. Among these Haloarchaea, which thrive in hypersaline environments, are characterized by their ability to thrive without requiring sterile conditions and uses of lower in cost substrates, making them more economical.7 Two haloarchaeal species from the Natrialbaceae family have the ability to manufacture PHB and PHV.8
Halophilic microorganisms represent a unique group of extremophiles that have recently attracted considerable interest in biopolymer research due to their ability to grow in hypersaline environments with minimal contamination risks and without the need for costly sterile conditions.4 Compared to conventional microbial producers such as bacteria, cyanobacteria, or algae, haloarchaea offer significant advantages, including tolerance to extreme salinity, utilization of inexpensive substrates, and robustness in large-scale cultivation. The Red Sea, in particular, provides a distinctive ecological niche characterized by high salinity, elevated temperatures, and unique nutrient compositions, making its native haloarchaeal strains genetically and physiologically different from those reported in other saline environments.5 These natural adaptations render Red Sea isolates highly promising for the sustainable and economical production of polyhydroxybutyrates (PHB) and polyhydroxyalkanoates (PHA).
Accordingly, the aim of the current research is to examine halophilic microorganisms from the Red Sea capable of producing PHB/PHA, to optimize their growth conditions for maximal biopolymer production, and to determine the biosafety of the extracted polymers through in vitro cytotoxicity assays. Ultimately, this study seeks to contribute to reducing environmental contamination by producing eco-friendly, biodegradable plastics that are both economically viable and safe for public health, with the added advantage of being derived from renewable sources that do not harm the environment
Materials and Methods
Site description and sampling
Samples of brine and sediment have been collected from a solar saltern in the Kingdom of Saudi Arabia’s Red Sea. Located at Al-shaeiba (20°48’06.6″N 39°27’40.2″E), Alkurnayash Southern (21°19’50.5″N 39°06’37.3″E), (saltern) located (21°10’16.04″N 39°11’5.94″E), Obhur Alshamaliyah (21°44’52.7″N 39°07’56.3″E), Thuwal (22°16’17.7″N 39°05’09.7″E) and King Abdullah Economic city (22°23’06.4″N 39°05’48.6″E), Via plastic bags and bottles with a 5-meter distance between each sample. All samples were stored at a temperature of 4 OC upon arrival.
The selection of these specific sites was based on their ecological diversity, as they represent different hypersaline environments along the Red Sea coast that vary in salinity, temperature, and organic matter availability. Such environmental variations increase the probability of isolating novel haloarchaeal strains with distinct physiological traits and enhanced biopolymer production capabilities. Moreover, sampling from both brine and sediment niches ensures a broader representation of microbial communities, thereby providing a more comprehensive assessment of haloarchaeal diversity and potential for sustainable bioplastic production.
Isolation and culture conditions
The samples were enhanced through cultivation in a medium for PHA accumulation reported by 9 which involves BM (per liter; KH2PO4, 37.5 mg; FeSO4.7H2O, 50 mg; MnCl2.4H2O, 0.36 mg; MgSO4.7H2O, 0.1 g; KCl, 2.0 g; Trisodium Citrate,3.0 g; Na2CO3, 8.0 g; NaCl, 200 g; and yeast extract, 1 g; pH regulated to 9.7), the medium was complemented with glucose 10 g/L as the carbon source, and cultures were vibrated on 180rpm at 37 ° C for 14 days. In order to isolate halophilic archaeal strains that accumulate PHA/PHB, a serial dilution method was performed. 1 ml of each dilution was then placed onto BM medium. Pure isolates were obtained by serial cultivation on BM and kept on the same medium.
Screening for PHAs-producing haloarchaeal strains.
A total of four distinct haloarchaeal isolates labeled with (HSM17, HSM20, HSM35, HSM38), obtained after two weeks of incubation at 37oC, were screened their capability as PHB/PHA producers through Sudan Black B staining as described by.10 The cells were smeared then heat-fixed on a sterile glass slide, from early stationary phase of growth, and stained for 10 minutes by a 3% (w/v in ethanol 70%). Nile blue stain 1% was applied for 10 minutes to all purified isolates, then wash the slides with tap water. After that Immerge the slides in Glacial acetic acid 8% solution for 1min. washed with tap water afterward check in microscope. Sudan Black B (Sigma), then the slides submerged in xylene till totally decolorized.11 The slides were counterstained with a 5% (w/v) aqueous safranin solution (Sigma) for 10 seconds, followed by a gentle rinse and air‑drying. Specimens were then examined using phase‑contrast microscopy (Nikon, Japan), and cells appearing blue‑black were identified as PHB/PHA‑positive strains.12,13 Escherichia coli was used as a negative control strain.
Identification of the potential PHAs–producing haloarchaeal strains
Genomic DNA was isolated from each candidate strain using two different extraction protocols; a modified protocol from “Experimental Techniques in bacterial genetics” as described by,14 and using a HiPurA® genomic DNA purification kit (HiGenoMB, MB505, India) as manufacturer’s instructions. Invitrogen (USA) primers were used to amplify the 16S rRNA from the archaeal isolates. These primers targeted the Escherichia coli 16S rRNA gene sequence at locations 6-25 and 1540-1521.15 PCR amplifications were performed in 50μL reaction volumes over 30 cycles With the following specifications: a 5-minute first denaturation at 95°C, followed by a 1-minute cycle of denaturation at 94°C, a 1-minute annealing at 60°C, and a 1-minute 30-second extension at 72°C. A final extension was performed for ten minutes at 72°C., followed by a hold at 4°C. After purification, the PCR products were produced at 50ng/μl concentration for further analysis.
Phylogenetic tree construction and Accession numbers for nucleotide sequences.
A phylogenetic tree was constructed using 16S rRNA gene sequences that closely matched the isolates, obtained from the NCBI database. The partial sequences were aligned through Multiple alignment CLUSTAL-W and neighbor joining tree was subsequently with MEGA X software package.16,17 All nucleotide sequence databases from this study with 16S were uploaded to the NCBI and GenBank databases.
Optimizing growth condition for haloarchaeal strains
A 100 µL sample of each PHA-producing haloarchaeal strain in the exponential phase was used to inoculate 100 mL of BM medium. Growth in the PHA-production medium was monitored spectrophotometrically at 600 nm every 48 hours over a two-week period. To assess the effects of different conditions on isolate growth, experiments were conducted in PHA/PHB production medium under varying; pH levels (5-9); temperatures (4, 20, 37, 45, 55, and 65 °C); Sodium chloride (NaCl) concentrations (100, 150, 200, 250, 300, and 350 g/L); Carbon sources (glycerol, carboxymethyl cellulose (CMC), starch, glucose and sucrose).
Biopolymer (polyhydroxybutrates) extraction
PHAs were regained from desiccated cells applying sodium hypochlorite method as stated by.18 The gained pellet was sequentially rinsed with distilled water, acetone and ethanol. then dissolved in warm chloroform (60-65°C) until vaporization. The percentage of PHB in the dried extract was calculated using the formula.19,20
%PHB/PHA accumulation= (PHB extract dry weight (g/ml) × 100)/(biomass (CDW))
Cell dry weight (CDW) = Falcon tube weight included dried pellets – Clear falcon tube weight.
PHB extract dry weight = Filter paper with dried filtered PHB weight – Clear filter paper weight.
Characterization of the biopolymer (polyhydroxybutrates).
Gas chromatography–mass spectrometry (GC–MS).
GC-MS analysis of the extracted PHB/PHA polymers was conducted using the methodology described by Ojha and Das.19 To determine the sample’s PHB content a thermo scientific Trace GC1310-ISQ mass spectrometer (Austin, Texas, USA) was attached to a TG-5MS direct capillary column (30 m x 0.25 mm x 0.25 µm film thickness). The oven temperature of the column was first kept at 50°C, then increased by 5°C every minute to 230°C, where it remained for two minutes. To attain the final temperature of 290°C, it was elevated by 30°C each minute and held there for two minutes. The temperatures of the MS transfer line and injector were kept up at 260°C and 250°C, respectively. The helium carrier flowed at a steady 1 ml/min. A µL diluted split sample was automatically injected with a solvent delay of three minutes using the Autosampler AS1300 in split mode with GC. EI mass spectra were obtained in complete scan mode spanning the m/z 40–1000 range at ionization voltages of 70 eV. The ion source temperature was set at 200 °C. The identification of components was accomplished by comparing their mass spectral profiles and retention times with those in the WILEY 09 and NIST 11 mass spectral databases
Cytotoxicity testing (Hemolytic Activity)
Hemolytic assay, as described by,21 was used to assess the cytotoxicity of the HSM17 biopolymer. To create an erythrocyte suspension, A volume of 1 milliliter of freshly obtained blood was centrifuged at a rate of 10,000 rpm for a period of five minutes. 5ml of a normal saline solution containing 0.9% NaCl was mixed with 200μl of the erythrocytes precipitate. Subsequently, the concoction was centrifuged at 4000 rpm in 15 minutes. The liquid portion was discarded, and the procedure was repeated three times. Finally, the washed red blood cells were combined with a normal saline solution (0.9% NaCl) to create a cell suspension with a concentration of 5%. HSM17 biopolymers were prepared using a 10% solution of DMSO at six distinct doses (3.125, 6.25, 12.5, 25, 50, 100μg/ml). Three replicates were used to test each concentration of HSM17 biopolymer. 500μl aliquots of HSM17 biopolymer were aseptically transferred into a microcentrifuge tube. Subsequently, 500μl of RBC suspension and 3 ml of normal saline were added and gently mixed. For 60 minutes, the samples were incubated at 35°C. They were then centrifuged for 10minutes at 2500 rpm and 4°C. At a wavelength of 540 nm, the absorbance (Abs) of the supernatant was measured. As a positive control, 500μL of a 5% blood suspension combined with 3mL of distilled water demonstrated 100% hemolysis, whereas normal saline without any hemolysis served as the negative control. The formula was used to calculate the percentage of hemolysis which provided by 22.
% Hemolysis= [(Absorbance (test)-Absorbance (control) )/(Absorbance (control))] ×100
Statistical analyses
The information was analyzed with statistical software SPSS (IBM Corp., New York, USA). According to the process outlined by Faizi and Alvi. To assess group differences, the study used a one-way analysis of discrepancy (ANOVA). Duncan’s multiple range test, as described in,23 was used to investigate pairwise comparisons between groups in more detail.
Results
Morphological characterization for haloarchaea strains
Four isolates were selected based on their strong similarity to the recommended minimum standards for haloarchaea, including cell appearance, motility, pigmentation. The strains that grew on BM agar plates were small, circular, convex, and viscous. Additionally, some colonies exhibited pink-red coloring, while others were colorless.
Screening for PHB/PHA-producing haloarchaea strains.
The ability of all the Haloarchaea strains to accumulate PHB/PHA was examined. Nile Blue A and Sudan Black B staining revealed that only four strains (HSM 17, HSM20, HSM35, and HSM38) were promising strains (Figure. 1 and 2)
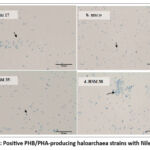 |
Figure 1: Positive PHB/PHA-producing haloarchaea strains with Nile Blue A.Click here to view Figure |
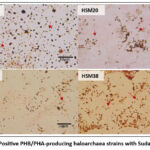 |
Figure 2: Positive PHB/PHA-producing haloarchaea strains with Sudan black B.Click here to view Figure |
Effects of carbon sources, Temperature, pH and Salinity on Growth of Potential Halophilic PHB /PHA Producers
The growth patterns of the promising strains were examined in PHA production medium at various temperatures, salinities, pH levels, and carbon sources. The effects of these parameters were examined in order to increase the PHA yield. The temperature range in which the four strains grew was 4–65°C.The optimum temperature for growth by four strains was 65˚C, 37oC,45oC, and 45oC for Strain HSM17, Strain HSM20, Strain HSM35 and Strain HSM38 respectively (Graph 1). The pH variety for growth of all HSM17, Strain HSM20, Strain HSM35 and Strain HSM38 strains was 5–9 with an optimum 7,7,9, and 8 respectively (Graph 2). With an optimal growth at 15% (Graph 3), the four strains were able to support NaCl at concentrations ranging from 20% to 35% (w/v). Additionally, strains HSM17, HSM20, HSM35, and HSM38 show the highest growth and PHA yield when using starch as the sole carbon source, with respective yields of 50.94%, 23.83%, 33.4%, and 27.29%. These strains are able to grow in a variety of carbon sources, including starch, carboxy-methyl cellulose (CMC), sucrose, glucose, and glycerol. This suggests that strain HSM17 has higher PHB/PHA yields and maximal specific growth ratios than other strains.
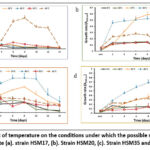 |
Graph 1: The impact of temperature on the conditions under which the possible strains that produce PHB/PHA proliferate (a). strain HSM17, (b). Strain HSM20, (c). Strain HSM35 and (d). Strain HSM38Click here to view Graph |
 |
Graph 2: The impact of pH on the conditions under which the possible strains that produce PHB/PHA proliferate (a). strain HSM17, (b). Strain HSM20, (c). Strain HSM35 and (d). Strain HSM38Click here to view Graph |
 |
Graph 3: The impact of NaCl concentration on the conditions under which the possible strains that produce PHB/PHA proliferate on the growth conditions for the potential PHB/PHA-producing strains (a). strain HSM17, (b). Strain HSM20, (c). Strain HSM35 and (d). Strain HSM38Click here to view Graph |
Molecular identification of the potential haloarchaeal strains
BLAST was used to identify the DNA sequences and compare the results with information from the NCBI GenBank database. According to phylogenetic analysis based on incomplete 16S rRNA gene sequences, the most promising strains belonged to the genera Haloarcula and Haloferax and exhibited sequence similarity of ≥95%. The NCBI and GenBank nucleotide sequence databases have received the 16S rRNA gene sequencing, and the accession numbers range from OQ366239 to OQ366242 (Figure.3).
 |
Figure 3: Using 16S rRNA gene sequences and maximum likelihood analysis, a phylogenetic tree was produced. Using information from the GenBank database, Click here to view Figure |
Characterization of the biopolymer by Gas chromatography–mass spectrometry (GC–MS)
Gamma-aminobutyric acid, phenol, 2,4-bis(1,1-dimethylethyl)-, dodecyl acrylate, 2-propenoic acid, 2-methyl-,1,5-pentanediyl ester, hexadecenoic acid, cis-vaccenic acid, oleic acid, 9-octadecenoic acid (z)-, butyl citrate, and isochiapin b were among the ten constituents identified by GC-MS analysis of the extracted polymer’s chemical structure to ascertain its monomeric composition. (Table1).
Table 1: Components information of the extracted PHB from HSM 17 occurred by GC-MS
| NO. | RT | Compound name | Area % | MF | MolecularFormula | MW |
| 1 | 13.36 | Gamma-Aminobutyric acid | 8.24 | 958 | C4H9NO2 | 103 |
| 2 | 17.19 | Phenol,2,4-bis(1,1-dimethylethyl)- | 4.60 | 945 | C14H22O | 206 |
| 3 | 21.39 | Dodecyl acrylate | 1.48 | 895 | C15H28O2 | 240 |
| 4 | 23.46 | 2-Propenoic acid, 2-methyl-,1,5-pentanediyl ester |
4.04 | 669 | C13H20O4 | 240 |
| 5 | 27.00 | Hexadecenoic acid | 7.16 | 910 | C16H32O2 | 256 |
| 6 | 30.29 | Cis-Vaccenic acid | 11.72 | 885 | C18H34O2 | 282 |
| 7 | 30.38 | Oleic acid | 2.16 | 865 | C18H34O2 | 282 |
| 8 | 30.73 | 9-octadecenoic acid (z)- | 2.38 | 808 | C18H34O2 | 282 |
| 9 | 31.00 | Butyl citrate | 56.35 | 925 | C18H32O7 | 360 |
| 10 | 38.94 | Isochiapin b | 1.87 | 825 | C19H22O6 | 346 |
RT: retention time, MW: molecular weight
In vitro cytotoxic effect of PHB/PHA extract (Hemolytic Activity percentage)
The hemolytic activity of the PHB/PHA extracted from Haloferax sp strain HSM17 is presented as percentage hemolysis and reported as mean ± standard deviation of three duplicates. All concentration showed significantly lower hemolytic impact toward human Erythrocytes liken to positive control (P<0.05) (Figure.4). Cytotoxicity was examined against erythrocytes, at concentrations differ from (62.5 to 2000µg/ml). The findings showed that hemolysis accelerated with increasing PHB dosage, reaching 0.633% at 62.5µg/ml and 3.517% at 2000µg/ml.
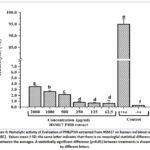 |
Figure 4: Hemolytic activity of Evaluation of PHB/PHA extracted from HSM17 on human red blood cells (RBC). Values mean ± SD; the same letter indicates that there is no meaningful statistical difference between the averages.Click here to view Figure |
Discussion
Archaea have earned considerable attention and research interest owing to their unique characteristics and remarkable capabilities. particularly their ability to produce bioplastics. In addition, the high salt concentration in the culturing medium means that not necessarily difficult sterile conditions. Furthermore, recovery of PHA/PHB granules is facilitated by the fact that haloarchaea cell walls and membranes can be disrupted simply by employing distilled water.5 Thus, it was thought to be a good model for creating biotechnological processes that would allow microorganisms to be employed as large-scale cell factories for the manufacturing of bioplastics.24 This study utilized low-cost carbon sources and Nile Blue A staining to screen haloarchaea isolated from the Red Sea in Saudi Arabia for their ability to produce PHB/PHA.11 demonstrated the effectiveness of this screening strategy for detecting PHA-producing species in the Archaea domain.
Haloarchaea have been the subject of numerous studies aimed at identifying the ideal circumstances for the highest possible yield of bioplastic. Optimizing the culture conditions for polymer formation in the potential strains HSM17, HSM20, HSM35, and HSM38 were the goal of this investigation. When starch was the only carbon source, strain HSM17 produced the highest yield (50.94%). followed by a yield of 40.51% when the only carbon source utilized was glycerol. HSM20, HSM35, and HSM38 produced the highest yields (23.83%, 33.4%, and 27.29%, respectively) when starch was the sole carbon source. In a similar with,25 demonstrated that the Haloferax sp strain NRS20 could create 23.83% of bioplastic when starch was used as the carbon source. Additionally, found that Haloarcula tradensis also exhibited this capability.26 This superior yield obtained from strain HSM17 may be explained by its enhanced metabolic flexibility and efficient carbon channeling mechanisms under hypersaline conditions. Haloarchaea are known to redirect excess carbon into intracellular PHB granules as a protective strategy against osmotic stress and nutrient fluctuations.8 In the case of HSM17, the organism demonstrated a stronger ability to utilize starch and glycerol compared to the other isolates, which likely increased the metabolic flux toward acetyl-CoA and subsequently to PHB biosynthesis. Previous studies have suggested that such high-yielding strains may possess elevated activity of key enzymes involved in the PHA pathway, such as β-ketothiolase, acetoacetyl-CoA reductase, and PHA synthase, which collectively contribute to superior polymer accumulation.20 Therefore, the high PHB yield observed in HSM17 is likely the result of both its environmental adaptation and its intrinsic enzymatic efficiency.
Regardless of the differences across strains, starch consistently proved to be the favored carbon source for promoting development. The finding aligns with prior studies that suggest glucose, when present at an ideal concentration of 25%, supports the highest level of bioplastic formation by haloarchaea.26-28 In contrast, strain NRS20 grew best at 15% and PH 7, with growth observed at NaCl concentrations ranging from 10% to 35% (w/v).25 Also, the study performed by Pramanik et al. in 2012, it was found that Haloarcula marismortui was able to create polyhydroxybutyrate (PHB) utilizing vinasse as a carbon source. Furthermore, it has been shown that additional species of Haloarcula are also capable of accumulating PHB.9 This study is the first to document the production of PHB and/or PHA by haloarchaea in a southern Saudi Arabian solar saltern. It is important to mention this fact. Moreover, the Haloarcula genus exhibits significant potential for biotechnology applications as a result of its well-studied genetic composition.26,29 Therefore, more research is required to investigate new species of Haloarcula that can manufacture PHB.
Additionally, Gas Chromatography Mass Spectrometry (GC-MS) is an influential analytical method which is frequently utilized to find and examine combinations in a range of samples. The method is based on using gas chromatography to separate the various components of a sample, then mass spectrometry to identify and detect individual molecules.30 By analyzing the mass spectra, the compounds present in the sample can be identified based on their characteristic fragmentation patterns and ion signals, this makes GC-MS a particularly useful instrument for identifying unknown compounds, investigating chemical reactions, and monitoring the purity tested material.31 Regarding GC MS analysis of biopolymer compounds extracted from were 10 compounds. The fact that biosafety is made from harmless components either human or environment made the biosafety of such evolved biodegradable polymers beside the nano-safety of their composite still negligible. However, most components run through tests to ensure they are safe to consume.
On the other hand, Researchers evaluate the toxicity of xenobiotics using newly gathered human erythrocytes through hemolysis or erythron-toxicity test.32 RBCs were selected for this investigation because they only have one membrane and no internal organelles, which makes them suitable for examining the fundamental relationships between chemical substances and cell membranes.33 this study found that different concentration of PHB product significantly influenced hemolysis with the percent hemolysis does not exceed 5% hemolysis until concentration (2000µg/ml).34 By determining the hemolytic activity of tested extract for human erythrocytes, it was observed that this extract has low toxicity. As highlighted previously, haloarchaea have been identified as promising and valuable biotechnological platforms to produce industrially important compounds such as bio plastic PHAs.1,35 The most important goal is to protect and restore natural landscapes while also reducing plastic pollution, which is one of the world’s biggest threats, as well as protect and rehabilitate natural landscapes.
Conclusion
Bioplastics provides a sustainable solution to the global challenge of plastic pollution. Extremophiles, particularly haloarchaea, represent promising microbial platforms due to their ability to thrive in hypersaline environments. In this study, four haloarchaeal isolates from the Red Sea (HSM17, HSM20, HSM35, and HSM38) were evaluated for their potential to produce polyhydroxybutyrate (PHB) and related polymers. All isolates demonstrated the ability to synthesize PHB granules under varying environmental conditions, with starch identified as the most effective carbon source. Notably, Haloferax sp. strain HSM17 exhibited the highest yield under optimized growth conditions and minimal cytotoxicity, confirming its promise as an eco-friendly and economical alternative to petrochemical plastics. These findings highlight Red Sea haloarchaea as valuable candidates for sustainable bioplastic production, while further studies are needed to explore additional strains and the use of industrial waste as alternative substrates to enhance yield and scalability.
Acknowledgment
We sincerely thank Jeddah University for the support and resources that made this research possible.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement-
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Authors’ Contribution
- Nuha Neyazi: Data Collection, Implement Experiment, Photography, Writing, Editing.
- Hend Tag: Conception, Supervision, Analysis, Writing, Review.
- Naglaa Elshafey: Review.
- Nashwa Hagagy: Conception, Supervision, Methodology, Writing.
Reference
- Mukherjee A, Koller M. Microbial PolyHydroxyAlkanoate (PHA) Biopolymers—Intrinsically Natural. Bioengineering. 2023;10(7):855.
CrossRef - Nikolaivits E, Pantelic B, Azeem M, et al. Progressing Plastics Circularity: A Review of Mechano-Biocatalytic Approaches for Waste Plastic (Re)valorization. Front Bioeng Biotechnol. 2021;9:696040. doi:10.3389/fbioe.2021.696040
CrossRef - Napathorn SC, Visetkoop S, Pinyakong O, Okano K, Honda K. Polyhydroxybutyrate (PHB) Production Using an Arabinose-Inducible Expression System in Comparison With Cold Shock Inducible Expression System in Escherichia coli. Front Bioeng Biotechnol. 2021;9:661096. doi:10.3389/fbioe.2021.661096
CrossRef - Khan AK, Anjum I, Hano C, Abbasi BH, Anjum S. An Overview on Feasible Production of Bioplastic Polyhydroxyalkanoate (PHA) in Transgenic Plants. In: Kuddus M, Roohi, eds. Bioplastics for Sustainable Development. Springer Singapore; 2021:555–579.
CrossRef - Moopantakath J, Imchen M, Anju VT, et al. Bioactive molecules from haloarchaea: Scope and prospects for industrial and therapeutic applications. Front Microbiol. 2023;14:1113540. doi:10.3389/fmicb.2023.1113540
CrossRef - Shaikh S, Yaqoob M, Aggarwal P. An overview of biodegradable packaging in food industry. Curr Res Food Sci. 2021;4:503–520. doi:10.1016/j.crfs.2021.07.005
CrossRef - Rekhi P, Goswami M, Ramakrishna S, Debnath M. Polyhydroxyalkanoates biopolymers toward decarbonizing economy and sustainable future. Crit Rev Biotechnol. Aug 2022;42(5):668–692. doi:10.1080/07388551.2021.1960265
CrossRef - Ben Abdallah M, Karray F, Sayadi S. Production of Polyhydroxyalkanoates by Two Halophilic Archaeal Isolates from Chott El Jerid Using Inexpensive Carbon Sources. Biomolecules. 2020;10(1):109.
CrossRef - Han J, Hou J, Liu H, et al. Wide distribution among halophilic archaea of a novel polyhydroxyalkanoate synthase subtype with homology to bacterial type III synthases. Appl Environ Microbiol. Dec 2010;76(23):7811–9. doi:10.1128/aem.01117-10
CrossRef - Narayanan M, Kandasamy S, Kumarasamy S, Gnanavel K, Ranganathan M, Kandasamy G. Screening of polyhydroxybutyrate producing indigenous bacteria from polluted lake soil. Heliyon. 10/30 2020;6doi:10.1016/j.heliyon.2020.e05381
CrossRef - Legat A, Gruber C, Zangger K, Wanner G, Stan-Lotter H. Identification of polyhydroxyalkanoates in Halococcus and other haloarchaeal species. Appl Microbiol Biotechnol. Jul 2010;87(3):1119–27. doi:10.1007/s00253-010-2611-6
CrossRef - Luhana K, Patel V. Quantitative Extraction and Analysis of Bioplastic (PHA) Accumulated in Bacterial Isolates of Paint Industry Effluent. International Journal of Chemtech Applicaions. 09/01 2013;2:52–62.
- Mostafa YS, Alrumman SA, Otaif KA, Alamri SA, Mostafa MS, Sahlabji T. Production and Characterization of Bioplastic by Polyhydroxybutyrate Accumulating Erythrobacter aquimaris Isolated from Mangrove Rhizosphere. Molecules. 2020;25(1):179.
CrossRef - Maloy SR. Experimental techniques in bacterial genetics / Stanley R. Maloy. The Jones and Bartlett series in biology. Jones and Bartlett; 1989.
- Sánchez-Porro C, Martín S, Mellado E, Ventosa A. Diversity of moderately halophilic bacteria producing extracellular hydrolytic enzymes. J Appl Microbiol. 2003;94(2):295–300. doi:10.1046/j.1365-2672.2003.01834.x
CrossRef - Elshafey N, Hamedo H, Mansour M, Awny A. Molecular identification of Halophilic Microorganisms producing Exo-Halozymes by a culture-dependent approach from the North Sinai Solar saltern. THE EGYPTIAN JOURNAL OF EXPERIMENTAL BIOLOGY (Botany). 08/10 2022;doi:10.5455/egyjebb.20220726105555
CrossRef - Tamura K, Stecher G, Peterson D, Filipski A, Kumar S. MEGA6: Molecular Evolutionary Genetics Analysis version 6.0. Mol Biol Evol. Dec 2013;30(12):2725–9. doi:10.1093/molbev/mst197
CrossRef - Bhattacharyya S, Crippa JA, Allen P, et al. Induction of psychosis by Δ9-tetrahydrocannabinol reflects modulation of prefrontal and striatal function during attentional salience processing. Arch Gen Psychiatry. Jan 2012;69(1):27–36. doi:10.1001/archgenpsychiatry.2011.161
CrossRef - Ojha N, Das N. Optimization and Characterization of Polyhydroxyalkanoates and its Copolymers Synthesized by Isolated Yeasts. Research Journal of Pharmacy and Technology. 03/01 2017;10:861. doi:10.5958/0974-360X.2017.00161.5
CrossRef - Sathiyanarayanan G, Bhatia SK, Song HS, et al. Production and characterization of medium-chain-length polyhydroxyalkanoate copolymer from Arctic psychrotrophic bacterium Pseudomonas sp. PAMC 28620. Int J Biol Macromol. Apr 2017;97:710–720. doi:10.1016/j.ijbiomac.2017.01.053
CrossRef - Powell WA, Catranis CM, Maynard CA. Design of self-processing antimicrobial peptides for plant protection. Lett Appl Microbiol. Aug 2000;31(2):163–8. doi:10.1046/j.1365-2672.2000.00782.x
CrossRef - Brovedani V, Sosa S, Poli M, et al. A revisited hemolytic assay for palytoxin detection: Limitations for its quantitation in mussels. Toxicon. Sep 1 2016;119:225–33. doi:10.1016/j.toxicon.2016.06.013
CrossRef - Faizi N, Alvi Y. Chapter 1 – Introduction to biostatistics. In: Faizi N, Alvi Y, eds. Biostatistics Manual for Health Research. Academic Press; 2023:1–16.
CrossRef - Simó-Cabrera L, García-Chumillas S, Hagagy N, et al. Haloarchaea as Cell Factories to Produce Bioplastics. Marine Drugs. 2021;19(3):159.
CrossRef - Hagagy N, Saddiq A, Tag H, Abdelgawad H, Selim S. Characterization of bioplastics produced by haloarchaeon Haloarcula sp strain NRS20 using cost-effective carbon sources. Materials Research Express. 10/01 2021;8:105404. doi:10.1088/2053-1591/ac3166
CrossRef - Karray F, Ben Abdallah M, Baccar N, Zaghden H, Sayadi S. Production of Poly(3-Hydroxybutyrate) by Haloarcula, Halorubrum, and Natrinema Haloarchaeal Genera Using Starch as a Carbon Source. Archaea. 2021;2021:8888712. doi:10.1155/2021/8888712
CrossRef - Hermann-Krauss C, Koller M, Muhr A, Fasl H, Stelzer F, Braunegg G. Archaeal production of polyhydroxyalkanoate (PHA) co- and terpolyesters from biodiesel industry-derived by-products. Archaea. 2013;2013:129268. doi:10.1155/2013/129268
CrossRef - Lynch EA, Langille MG, Darling A, et al. Sequencing of seven haloarchaeal genomes reveals patterns of genomic flux. PLoS One. 2012;7(7):e41389. doi:10.1371/journal.pone.0041389
CrossRef - Han J, Lu Q, Zhou L, Zhou J, Xiang H. Molecular characterization of the phaECHm genes, required for biosynthesis of poly(3-hydroxybutyrate) in the extremely halophilic archaeon Haloarcula marismortui. Appl Environ Microbiol. Oct 2007;73(19):6058–65. doi:10.1128/aem.00953-07
CrossRef - Santos FJ, Galceran MT. Modern developments in gas chromatography-mass spectrometry-based environmental analysis. J Chromatogr A. Jun 6 2003;1000(1-2):125–51. doi:10.1016/s0021-9673(03)00305-4
CrossRef - Garcia A, Barbas C. Gas chromatography-mass spectrometry (GC-MS)-based metabolomics. Methods Mol Biol. 2011;708:191–204. doi:10.1007/978-1-61737-985-7_11
CrossRef - Jeswani G, Alexander A, Saraf S, Saraf S, Qureshi A, Ajazuddin. Recent approaches for reducing hemolytic activity of chemotherapeutic agents. J Control Release. Aug 10 2015;211:10–21. doi:10.1016/j.jconrel.2015.06.001
CrossRef - Robles-Loaiza AA, Pinos-Tamayo EA, Mendes B, et al. Traditional and Computational Screening of Non-Toxic Peptides and Approaches to Improving Selectivity. Pharmaceuticals. 2022;15(3):323.
CrossRef - Pagano M, Faggio C. The use of erythrocyte fragility to assess xenobiotic cytotoxicity. Cell Biochem Funct. Aug 2015;33(6):351–5. doi:10.1002/cbf.3135
CrossRef - Cánovas V, Garcia-Chumillas S, Monzó F, et al. Analysis of Polyhydroxyalkanoates Granules in Haloferax mediterranei by Double-Fluorescence Staining with Nile Red and SYBR Green by Confocal Fluorescence Microscopy. Polymers. 2021;13(10):1582.
CrossRef
Abbreviation list
16S rRNA: 16S ribosomal
RNA; Abs: absorbance;
BLAST: Basic Local Alignment Search Tool;
CDW: Cell dry weight;
CMC: carboxymethyl cellulose;
DNA Deoxyribonucleic acid;
GC–MS: Gas chromatography–mass spectrometry;
MW molecular weight;
NaCl: Sodium chloride;
NCBI National Center for Biotechnology Information;
PHAs: Polyhydroxyalkanoates;
PHB: polyhydroxybutyrate;
PHBV: polyhydroxybutyrate-co-hydroxy valerate;
RT retention time.







