Manuscript accepted on :09-09-2025
Published online on: 25-09-2025
Plagiarism Check: Yes
Reviewed by: Dr. Tapaskumar Mukeshkumar Shah and Dr. Shail Modi
Second Review by: Dr. M. K. Shukla
Final Approval by: Dr. Prabhishek Singh
Qurbonova Shakhnoza Bakhtiyorovna1 , Zhumaev Inoyat Zulfiqorovich1*
, Zhumaev Inoyat Zulfiqorovich1* , Boboev Sadriddin Nurillo Ugli1
, Boboev Sadriddin Nurillo Ugli1 , Usmanov Pulat Bekmuratovich1
, Usmanov Pulat Bekmuratovich1 , Rustamov Shavkat Yusubovich1
, Rustamov Shavkat Yusubovich1 , Zaripov Abdisalim Abdikarimovich1
, Zaripov Abdisalim Abdikarimovich1 and Zhurakulov Sherzod Niyatkobulovich2
and Zhurakulov Sherzod Niyatkobulovich2
1Department of Cell Biophysics, Institute of Biophysics and Biochemistry, National University of Uzbekistan, Tashkent, Uzbekistan
2Department of Alkaloids Chemistry, Institute of Chemistry of Plant Substances, Academy Sciences of Uzbekistan, Tashkent, Uzbekistan
Corresponding Author E-mail:inoyat8585@mail.ru
DOI : https://dx.doi.org/10.13005/bpj/3270
Abstract
During the study, the inotropic and antiarrhythmic properties of the alkaloid 1-(3ˊ-Bromophenyl)-6,7-methylenedioxy-1,2,3,4-tetrahydroisoquinoline hydrochloride (F-25) were investigated. The alkaloid F-25 was shown to exert a negative inotropic effect on the contractile force of rat cardiac papillary muscle, decreasing myocardial contractility by 87.6±4.2% at a concentration of 100 μM compared to the control. To assess the involvement of voltage-gated Na+ channels in providing the negative inotropic effect of the alkaloid F-25, experiments were conducted using lidocaine, a specific blocker of this channel. When the effect of F-25 alkaloid (100 μM) was tested in the existence of lidocaine (IC50=15.4 μM), the amplitude of papillary muscle contraction force was 32.8±3.9%, respectively. The influence of the alkaloid F-25 on Na⁺/Ca²⁺ exchange was examined using NiCl₂, a non-specific blocker of this exchanger. The negative inotropic effect of the alkaloid F-25 (100 μM) on papillary muscle contraction activity in the presence of a 10 mM concentration of NiCl2 in the incubation medium was 42.3±4.2%. The negative inotropic effect of the alkaloid F-25 is exerted mainly by modulating the function of Na+ channels and, in part, the Na+/Ca2+ exchange. The impact of the alkaloid F-25 was also evaluated using an aconitine-induced arrhythmia model, revealing that the compound exhibits strong antiarrhythmic properties
Keywords
Antiarrhythmic action; Isoquinoline alkaloid; Na+/Ca2+-exchanger; Na+-channel; Papillary muscle
Download this article as:| Copy the following to cite this article: Bakhtiyorovna Q. S, Zulfiqorovich Z. I, Ugli B. S. N, Bekmuratovich U. P, Yusubovich R. S, Abdikarimovich Z. A, Niyatkobulovich Z. S. Antiarrhythmic Effect of 1-(3ˊ-Bromophenyl)-6,7-methylenedioxy-1,2,3,4-tetrahydroisoquinoline Hydrochloride. Biomed Pharmacol J 2025;18(3). |
| Copy the following to cite this URL: Bakhtiyorovna Q. S, Zulfiqorovich Z. I, Ugli B. S. N, Bekmuratovich U. P, Yusubovich R. S, Abdikarimovich Z. A, Niyatkobulovich Z. S. Antiarrhythmic Effect of 1-(3ˊ-Bromophenyl)-6,7-methylenedioxy-1,2,3,4-tetrahydroisoquinoline Hydrochloride. Biomed Pharmacol J 2025;18(3). Available from: https://bit.ly/4gGE9K0 |
Introduction
Cardiovascular diseases remain one of the primary causes of illness and death across numerous countries worldwide. Cardiac arrhythmia, the most common cardiovascular disease, is one of the most important risk factors for sudden death.1 The pathogenesis of cardiac arrhythmia is based on functional damage to the heart, leading to a violation of the most important parameters of the myocardium: contraction frequency and contractile activity. The effect of most antiarrhythmic drugs is based on the correction of these parameters, but many of them do not fully meet the requirements of clinical practice and have a number of additional side effects. From this point of view, the creation and production of a new generation of safe and highly effective antiarrhythmic drugs is one of the urgent problems of modern pharmacology and cardiology.
The pathogenesis of cardiac arrhythmias is based on a violation of the electrophysiological properties of cardiomyocytes, which are responsible for excitability, automatism, and the generation of action potentials (AP).2,3 In this regard, the modern concept of preventing and treating cardiac arrhythmias places a leading role in the development of new approaches to correcting these disorders, including the creation of new generation antiarrhythmic drugs.
The normal rhythmic activity of the heart depends on the synchrony in the function of ion channels, and serious changes in this system are directly manifested in the form of arrhythmia.4,5 The mutation results in a phenotype of dysfunction of cardiomyocyte Na+ and K+ channels, leading to a number of arrhythmias.6-8 LQT syndrome, SQT syndrome, Brugada syndrome, catecholaminergic polymorphic tachycardia syndromes are associated with mutations in the genome of cardiomyocyte ion channels.9,10 Also, a number of arrhythmias, such as Andersen and Timothy syndromes, are not caused by a single mutation, but by multisystem disorders. In the cardiomyocyte, the Na+ channel is activated in AP 0-phase, its function disorders lead to tachyarrhythmia.11
At present, antiarrhythmic drugs are categorized based on their specific characteristics. The E. Vaughan-Williams classification system categorizes these drugs into four primary classes: Class I – sodium channel blockers; Class II – beta-adrenoceptor blockers; Class III – potassium channel blockers; and Class IV – calcium channel blockers.12
Class I includes Na+ channel blockers, which are characterized by slowing of the rate of AP depolarization, excitability, and signal transmission, and changes in the QT interval on the ECG. LQTS syndrome, associated with mutations in the genome of Na+ and K+ channels, occurs in both acquired and inherited forms, and in the acquired form, antihistamines and antiarrhythmic pharmacological drugs are used.13,14
Antiarrhythmic drugs used in the treatment of cardiac arrhythmias are based on inhibiting the function of Na+, Ca2+, and K+ channels and adrenoreceptors.15 Alkaloids exhibit a broad spectrum of pharmacological effects, and many have found applications in both traditional and modern medicine, as well as serving as key starting materials for drug development. Among them, the isoquinoline, indole, and purine alkaloids are currently the most extensively researched in pharmacological studies.16-18 The pharmacological properties of berberine include its antiarrhythmic activity and its potential application in the management of hypertension.19 Overall, medications primarily composed of isoquinoline alkaloids are currently being effectively utilized in the treatment of conditions such as pain, atherosclerosis, hypertension, myocardial infarction, cardiomyopathy, heart failure, and various types of arrhythmias.20-23 In this context, isoquinoline alkaloids hold significant potential as a valuable source for creating new antiarrhythmic drugs, thanks to their structural diversity and wide range of pharmacological actions. Investigating the biological activities of these alkaloids will lay the groundwork for discovering promising candidates to develop next-generation antiarrhythmic drugs aimed at treating and preventing cardiac arrhythmias.
Materials and Methods
The experiments were conducted at the Laboratory of Cellular Biophysics, Institute of Biophysics and Biochemistry, National University of Uzbekistan. The studies used white, non-purebred rats weighing between 200 and 250 grams. The international Declaration of Helsinki and the guidelines developed by the Council for International Organizations of Medical Sciences (CIOMS; The Council for International Organizations of Medical Sciences) (1985) were followed when working with experimental animals.
The mechanography method was used to study the mechanism of action of biologically active substances on the functional role of the myocardium of experimental animals In vitro. Experimental animals are a convenient object of research for elucidating the mechanisms of antiarrhythmic action of pharmacological agents In vitro, under normal physiological and pathological conditions.
The experimental animals were anesthetized using diethyl ether and euthanized by cervical dislocation. The thoracic cavity was then surgically opened to remove the heart. A papillary muscle sample (diameter 0.4-1.3 mm; length 2.5-3.8 mm) was isolated and placed in a Petri dish containing Krebs-Henseleit physiological solution. The preparation was subsequently transferred to a specialized horizontal experimental bath (type 813, volume 5 ml) (Hugo Sachs Electronic, Germany) designed for studying the muscle’s isometric contraction activity. The papillary muscle preparation was incubated in a Krebs-Henseleit physiological solution, the temperature of the solution was maintained constant (t=+36±0.5°C) using a thermostat (LOIP LT-108a, Russia), and aerated with carbogen (O2-95%, CO2-5%). Circulation of physiological solution (3-5 ml/minute) was ensured by LKB Bromma (Sweden) peristaltic pumps. The papillary muscle preparation was connected to an F30 mechanotron (Model D-79232; HSE, Germany), and the cardiac muscle preparation was stimulated with an electrical impulse ~20% above the threshold level at a frequency of 1 Hz (5-10 ms; 3 V) using an electrostimulator С type 224 (HSE, Germany) with two platinum (Pt) wire electrodes. The isometric mechanical activity of the papillary muscle was recorded on a computer in a special digital format using an analog-to-digital converter LabPro Logger Lite 1.2 (Vernier Software & Technology, Beaverton, USA) through an amplifier (TAM-A, HSE GmbH, Germany). The research used Krebs-Henseleit physiological solution with the following composition (mM): NaCl-118; KCl-4.7; CaCl2-2.5; MgSO4-1.2; KH2PO4-1.1; glucose-5.5; NaHCO3-25 (pH=7.4) [24].
Aconitine Arrhythmia Model
The experiments used a model of arrhythmia induced by aconitine. It is known that aconitine increases the activity of Na+ channels in cardiomyocytes, leading to an increase in the concentration of Na+ in the cytosol, and as a result of the entry of Ca2+ ions through the Na+/Ca2+– exchange system, the concentration of Ca2+ increases and arrhythmia occurs. Arrhythmia occurs 7-10 minutes after adding aconitine (1 μM) to the incubation medium.25,26 The antiarrhythmic effect of the isoquinoline alkaloid F-25 was evaluated in this experimental arrhythmia model.
Statistics
In this study, all results are expressed as mean ± SD. Analysis of variance was used to compare control values between groups. The statistical reliability of the values between the experimental results and the control group was calculated based on the Student’s t-test and was considered statistically reliable at values of р<0.05, р<0.01. The statistical analysis was conducted with Origin Pro version 9.1. (OriginLab Co., U.S.A).
Method for the synthesis of 1-(3′-bromophenyl)-6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline hydrochloride (F-25), C₁₇H₁₈O₂NBr·HCl
The compound 1-(3′-bromophenyl)-6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline hydrochloride (F-25) was synthesized using a previously described method. 27
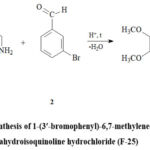 |
Scheme 1: Synthesis of 1-(3′-bromophenyl)-6,7-methylenedioxy-1,2,3,4-tetrahydroisoquinoline hydrochloride (F-25). |
The compound 1-(3′-bromophenyl)-6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline hydrochloride (F-25) was obtained by a new method, in contrast to a mixture of 5.0 g (0.027 mol) of 3,4-dimethoxyphenylethylamine (1) and 5.11 g (0.027 mol) of 3-bromobenzaldehyde (2) in 10 mL of trifluoroacetic acid was refluxed for 4 hours. The progress of the reaction was monitored by TLC. After completion, the reaction mixture was cooled and basified with 5% aqueous sodium hydroxide solution to pH 9–10. The product was then exhaustively extracted with chloroform. After evaporation of the chloroform, the crude product was dissolved in acetone and acidified with concentrated HCl to pH 5–6. The resulting precipitate of the hydrochloride was filtered and washed three times with acetone. Yield: 9.22 g (96%), m.p. 248–251 °C (from acetone), Rf 0.82 (chloroform:methanol = 12:1).
IR spectrum (KBr, νmax, cm-1): 3244, 3235, 2994, 2960, 2922, 2892, 2826, 2722, 1610, 1590, 1508, 1456, 1331, 1253, 1214, 1177, 1114, 1043, 1018, 969, 852, 818;
NMR 1Н (600 MHz, СDСl3, m.d., J/Hz): 1.85 (1H, с, NH), 2.72 (1Н, дт, J=4.7, 15.9, Ha-4), 2.90 (1Н, ддд, J= 5.3, 8.2, 15.9, Hb-4), 3.01 (1Н, ддд, J= 4.7, 8.1, 12.2, Ha-3), 3.16 (1H, дт, J= 5.3, 12.2, Hb-3), 3.64 (3Н, с, 7-ОСН3), 3.86 (3Н, с, 6-ОСН3), 4.99 (1Н, с, Н-1), 6.20 (1Н, с, Н-8), 6.62 (1Н, с, Н-5), 7.11-7.17 (2H, м, Ar-Н), 7.37-7.40 (2Н, м, Ar-Н);
NMR 13C (150 MHz, СDСl3, m.d.): 29.29 (С-4), 41.77 (С-3), 55.94 (6-ОСН3), 56.02 (7-ОСН3), 61.02 (С-1), 110.90 (С-8), 111.63 (С-5), 122.64 (С-3′), 127.69 (С-6′), 127.82 (С-4а), 129.04 (С-8a), 130.03 (С-5′), 130.59 (C-4′), 132.00 (С-2′), 147.24 (С-1′), 147.43 (С-7), 147.90 (С-6).
Results
Isoquinoline alkaloids, which are heterocyclic chemical compounds, exhibit a broad range of physiological effects.28-30 Specifically, isoquinoline alkaloids have been shown to possess antiarrhythmic and inotropic properties in cardiovascular diseases.31,32 Therefore, the mechanisms of antiarrhythmic action of the isoquinoline alkaloid F-25 have been clarified. The experiments investigated the dose-dependent impact of the alkaloid F-25 on the contractile function of rat cardiac papillary muscles. The alkaloid demonstrated a negative inotropic effect on the contractile activity of rat heart papillary muscle across all tested concentrations. Accordingly, when examining the effects of the alkaloid F-25 at concentrations ranging from 10 μM to 100 μM, the greatest effect was observed at 100 μM, where it reduced the muscle contraction force by 87.6±4.2% compared to the control. (Figures 1(A) and 1(B)). The half-maximal effective concentration (IC50) of the alkaloid F-25 was 34.4 μM, respectively.
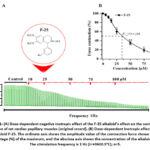 |
Figure 1: (A) Dose-dependent negative inotropic effect of the F-25 alkaloid’s effect on the contraction function of rat cardiac papillary muscles (original record). (B) Dose-dependent inotropic effect of the alkaloid F-25. |
During membrane depolarization of cardiac muscle cells, the entry of Na+ ions into the cardiomyocyte activates the sarcolemmal L-type Са2+ channel, and Са2+ ions entering the cytosol stimulate the release of Са2+ ions from the SR.33,34 As the amount of [Ca2+]i in the cytosol increases, Ca2+ ions bind to the troponin C protein in the myofilament, causing contraction.35,36 Blocking Na+ channels reduces the intracellular concentration of Na+ ions in cardiomyocytes, which leads to a decrease in [Ca2+]i and a subsequent reduction in the strength of cardiac muscle contraction.37-39
The negative inotropic effect of the alkaloid F-25 on the contractile function of rat cardiac papillary muscle may result from the inhibition of Na+ channels in cardiomyocytes.Therefore, to assess the role of voltage-gated Na+ channels in providing the negative inotropic effect of the alkaloid F-25, experiments were conducted using lidocaine, a specific blocker of these channels.
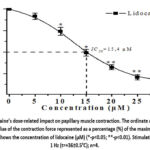 |
Figure 2: Lidocaine’s dose-related impact on papillary muscle contraction. The ordinate axis shows the amplitude value of the contraction force represented as a percentage (%) of the maximum, |
In initial control experiments, the dose-dependent effect of lidocaine (ranging from 5 to 30 μM) on papillary muscle contraction activity was examined at a stimulation frequency of 1 Hz. It was observed that lidocaine reduced the amplitude of papillary muscle contraction force by 89.6±4.2% at a concentration of 30 µM compared to the control. It was found that the concentration of lidocaine that inhibits muscle contraction by 50% (IC50) was 15.4 μM (Figure 2).
In cardiac muscle cells, lidocaine blocks the Na+ channel, resulting in a decrease in [Na+]i in the cytosol. Lidocaine causes a decrease in the force of papillary muscle contraction under lidocaine influence, which increases the release of Ca2+ through the Na+/Ca2+ exchange system and is accompanied by a decrease in the amount of [Ca2+]i in the cytosol.
In subsequent experiments, when the effect of alkaloid F-25 (100 μM) was tested in the presence of lidocaine (IC50=15.4 μM) in incubation conditions, the amplitude of papillary muscle contraction force was 32.8±3.9%, respectively (Figures 3(A) and 3(B)). The negative inotropic effect of alkaloid F-25 was found to decrease in conditions involving lidocaine. Analysis of the experimental results indicates that this alkaloid blocks Na+ channels located in the membrane of cardiomyocytes, resulting in a decrease in the amount of [Na+]i in the cytosol and, as a result, a decrease in the entry of Ca2+ ions into the cell through the Na+/Ca2+ exchange system. In addition, the negative inotropic effect of the alkaloid F-25 indicates that other ion transport systems are also involved in the alkaloid’s negative inotropic effect.
 |
Figure 3: (A) Influence of alkaloid F-25 on the contractile force of rat cardiac papillary muscle in the presence of lidocaine (original record). (B) The inotropic action of alkaloid F-25 on the contraction strength of papillary muscle in the presence of lidocaine. |
In cardiomyocytes, the entry of Na+ ions into the cytosol is ensured not only by Na+ channels but also by Na+/Ca2+ exchange.40 Na+/Ca2+ exchange is recognized as a key factor in regulating Ca2+ ion dynamics within cardiomyocytes and pacemaker function, and it can also play a role in the development of arrhythmias in certain situations.41,42 As a result of the increase in the concentration of [Ca2+]i in the cardiomyocyte cytosol, the outflow of Ca2+ ions from the SR increases, and the influx of Na+ ions through the Na+/Ca2+ exchanger increases, resulting in early afterdepolarization in the plateau phase of the AP and arrhythmia.43
Based on the above, the F-25 alkaloid may exert a negative inotropic effect by modulating the Na+/Ca2+ exchange function. To verify this hypothesis, further experiments were conducted to assess the impact of the F-25 alkaloid on Na+/Ca2+ exchange in the presence of its non-specific blocker, NiCl2. In the presence of 10 mM NiCl2 in the incubation medium, the negative inotropic effect of the F-25 alkaloid (100 μM) on the papillary muscle contraction activity was 42.3±4.2% (Figures 4(A) and 4(B)). These data indicate that Na+/Ca2+ exchange is partially involved in the negative inotropic effect of the alkaloid F-25.
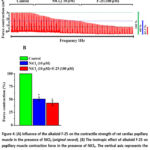 |
Figure 4: (A) Influence of the alkaloid F-25 on the contractile strength of rat cardiac papillary muscle in the presence of NiCl₂ (original record). (B) The inotropic effect of alkaloid F-25 on papillary muscle contraction force in the presence of NiCl₂. |
Class I antiarrhythmic drugs consist of pharmacological agents that are crucial for restoring the normal physiological function of Na⁺ channels in cardiomyocytes and correcting their dysfunction.44 In the next experiments, to evaluate the antiarrhythmic effect of the alkaloid F-25, its antiarrhythmic effect was tested on an experimental arrhythmia model induced by aconitine. In this case, exposure to 1 μM aconitine led to an increase in the force of papillary muscle contraction and, after 7-10 minutes, to the occurrence of spontaneous contraction, that is, arrhythmia.
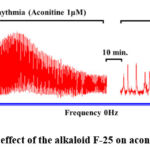 |
Figure 5: Antiarrhythmic effect of the alkaloid F-25 on aconitine-induced arrhythmia. |
The arrhythmia caused by aconitine results in an elevated concentration of Na⁺ ions, which subsequently enhances the Na⁺/Ca²⁺ exchange process, increasing the swapping of Na⁺ ions for Ca²⁺ ions. This type of activation of the Na+/Ca2+ exchange in cardiomyocytes enhances the entry of Ca2+ into the cytosol and leads to spontaneous release of Ca2+ from the SR and cardiac rhythm disturbances. Upon investigating the impact of the alkaloid F-25 on aconitine-induced arrhythmia, we observed a reduction in heart rate from 246±12 beats per minute to 65±6 beats per minute (Figure 5).
Discussion
Isoquinoline alkaloids have a very broad pharmacological activity, and one of their most important properties is their antiarrhythmic effect.45 In particular, pharmacological studies conducted on berberine show that it exhibits vasodilating and antiarrhythmic effects.46
Class I antiarrhythmic drugs are pharmacological agents that play a key role in restoring the normal physiological function of Na⁺ channels in cardiomyocytes by correcting their abnormal activity.47 To evaluate the antiarrhythmic effect of these alkaloids in the experiments, the antiarrhythmic effect on the experimental arrhythmia model induced by aconitine was examined. Aconitine-induced arrhythmia results in elevated intracellular Na+ levels, which subsequently enhances the exchange of Na+ for Ca2+ via the Na+/Ca2+ exchanger.48 This mode of Na⁺/Ca²⁺ exchanger activation in cardiomyocytes increases Ca²⁺ influx into the cytosol, triggering spontaneous Ca²⁺ release from the sarcoplasmic reticulum and resulting in cardiac rhythm disturbances. The alkaloid F-25 effectively eliminates disturbances in the contractile activity of the heart muscle that occur as a result of arrhythmias caused by Aconitine.
Conclusion
In cardiomyocytes, the Na+ channel is a substrate for the action of class I antiarrhythmic drugs.49,50 Open and inactive channels are more prone to blocking than quiescent channels. Binding to antiarrhythmic drugs occurs mainly during the action potential. And this block extends to the time interval between action potentials. Class I antiarrhythmic drugs can be classified according to their kinetic binding, with drugs exhibiting different effects and exhibiting fast, intermediate, and slow kinetic binding.51
Considering the evaluation of the findings from our research, it can be concluded that the antiarrhythmic effect of the alkaloid F-25 on the aconitine (1 μM)-induced arrhythmia model in rat papillary muscle is due to its interaction with voltage-gated Na+ channels in cardiomyocytes and modification of the transport of Na+ and Ca2+ ions (Figure 6).
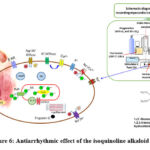 |
Figure 6: Antiarrhythmic effect of the isoquinoline alkaloid F-25 |
Acknowledgment
This work was supported by the Science and Technology Development Coordination Committee.
Funding source
This work was supported by the Ministry of Higher Education, Science and Innovation of Uzbekistan (grant AL-9224104346).
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
The experimental protocols complied with the standards and requirements for the humane treatment of animals and the provisions of the Ethical Commission of the IBB at the National University of Uzbekistan. (Protocol No. 7 BEC/IBB-NUU of 04/07/2022) on the use of laboratory animals. Preparations of isolated aortic segments were obtained using a known method.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Authors’ Contribution
- Qurbonova Shakhnoza Bakhtiyorovna: Conceptualization, Methodology, Writing – review & editing, Writing – Original Draft, Supervision.
- Zhumaev Inoyat Zulfiqorovich: Methodology, Writing – review & editing, Writing – original draft, Validation, Formal analysis, Project Administration.
- Boboev Sadriddin Nurillo Ugli: Methodology, Investigation, Formal analysis, Data curation.
- Usmanov Pulat Bekmuratovich: Conceptualization, Methodology, Writing – review & editing, Supervision, Resources, Funding acquisition.
- Rustamov Shavkat Yusubovich: Visualization, Resources.
- Zaripov Abdisalim Abdikarimovich: Writing – review & editing, Writing – Original Draft.
- Zhurakulov Sherzod Niyatkobulovich: Isolation of 1-(3ˊ-Bromophenyl)-6,7-methylenedioxy-1,2,3,4-tetrahydroisoquinoline hydrochloride.
References
- SrinivasanT., Schilling R.J. Sudden Cardiac Death and Arrhythmias. Arrhythm Electrophysiol Rev. 2018;7(2):111–117.
CrossRef - Antzelevitch Ch, Burashnikov Overview of Basic Mechanisms of Cardiac Arrhythmia. Card Electrophysiol Clin. 2011;3(1):23–45.
CrossRef - Issa Z.F, Miller J.M., Zipes D.P. 3 – Electrophysiological Mechanisms of Cardiac Arrhythmias. Clinical Arrhythmology and Electrophysiology (Third Edition). 2019;51-80.
CrossRef - Oshchepkova Yu.I., Uzbekov V.V., Jumayev I.Z., Rustamov Sh.Yu., Usmanov P.B., Salikhov Sh.I. Comparative study of antiarrhythmic and inotropic activity of amiodarone hydrochloride and its complexes with glycyrrhizic acid and monoammonium salt of glycyrrhizic acid. Eksperimental’naya i Klinicheskaya Farmakologiya, 2023;86(4):15–22.
- Uzbekov V.V., Abdullaev B.F., Jumayev I.Z., Oshchepkova I.Yu, Usmanov P.B. and Salikhov Sh.I. Comparative study of the antiarrhythmic activity of liposomal forms of lappaconitine hydrobromide and its complex with glycyrrhizic acid monoammonium salt in the aconitine arrhythmia model. Pharmaceutical Chemistry Journal, 2023;56(10):1327–1332.
CrossRef - Zhu Z.I., Clancy C.E. Genetic mutations and arrhythmia: simulation from DNA to electrocardiogram. Journal of Electrocardiology. 2007;40(6):47-50.
CrossRef - Tsumoto K, Ashihara, Naito N,. et all. Specific decreasing of Na+channel expression on the lateral membrane of cardiomyocytes causes fatal arrhythmias in Brugada syndrome. Scientifc Reports. 2020;10:19964.
CrossRef - GiudicessiR, Ackerman M.J. Potassium-channel mutations and cardiac arrhythmias—diagnosis and therapy. Nat Rev Cardiol. 2012;9(6):319–332.
CrossRef - KaufmanS. Mechanisms and clinical management of inherited channelopathies: long QT syndrome, Brugada syndrome, catecholaminergic polymorphic ventricular tachycardia, and short QT syndrome. Heart Rhythm. 2009;6(8):51-55.
CrossRef - GollobH, Redpath C.J and Roberts J.D. The Short QT Syndrome: Proposed Diagnostic Criteria. JACC. 2011;57 (7):802–812
CrossRef - Juárez Ch.K., Amin A.S., Offerhaus J.A., Bezzina C.R, Boukens B.J. Cardiac Repolarization in Health and Disease. JACC: Clinical Electrophysiology. 2023;9(1):124-138.
CrossRef - Barton A.K, McGowan M, Smyth A, Wright G.A, Gardner R.S. Classification and choice of antiarrhythmic therapies. Prescriber; 2020;31(3):11-17
CrossRef - MossJ, Kass R.S. Long QT syndrome: from channels to cardiac arrhythmias. J Clin Invest. 2005;115(8):2018–2024.
CrossRef - Santamaría D.B, Caballero K.L, Corso-Ramirez J.M et al. Genetic and Molecular Aspects of Drug-Induced QT Interval Prolongation. International Journal of Molecular Sciences (IJMS), 2021;2(15):1-27.
- Lei M, Wu L, Terrar D.A, Huang C.L Modernized Classification of Cardiac Antiarrhythmic Drugs. Circulation. 2018;138:1879-1896.
CrossRef - Heinrich M, Mah J, Amirkia Alkaloids Used as Medicines: Structural Phytochemistry Meets Biodiversity-An Update and Forward Look. Molecules. 2021;26(7):1-18.
CrossRef - Zhumaev I.Z., Boboev, S.N., Usmanov P.B. et al. Role of RyR2 and SERCA2a in the Cardioprotective Effects of Vincanine and Pyrazoline Alkaloids. Trends in Sciences, 2025;22(1):1-11.
CrossRef - Zhumaev I.Z., Boboev S.N., Usmanov P.B. et al. Mechanism of Positive Inotropic Effect of Vincanine on Cardiac Muscle Contraction Activity. Biomedical and Pharmacology Journal, 2022;15(4):2309–2316.
CrossRef - Zhang M, Feng L, Li J, Chen Therapeutic Potential and Mechanisms of Berberine in Cardiovascular Disease. Cardiovascular Pharmacology. 2016;2:281–292.
CrossRef - Rakhmanova K.A., Zhurakulov S.N., Tursunkhodjayeva F.M., Azamatov A.A., Saidkhodjayeva D.M., Jumayev I.Z. Analgesic and Anti-Inflammatory Effects of 1-(4′-Dimethylaminophenyl)-6, 7-Dimethoxy-1,2,3,4-Tetrahydroisoquinoline Hydrochloride. Biomedical and Pharmacology Journal, 2022;15(2):891–895
CrossRef - Zhang Y, Li M, Li X, Zhang T, Qin M, Ren Isoquinoline Alkaloids and Indole Alkaloids Attenuate Aortic Atherosclerosis in Apolipoprotein E Deficient Mice: A Systematic Review and Meta-Analysis. Front. Pharmacol., 2018;9:602:1-9.
CrossRef - Usmanov P.B., Jumayev I.Z., Rustamov S.Yu. et. al. The combined inotropic and vasorelaxant effect of DHQ-11, a conjugate of flavonoid dihydroquercetin with isoquinoline alkaloid 1-aryl-6,7-dimethoxy-1,2.3,4-tetrahydroisoquinoline. Biomedical and Pharmacology Journal, 2021;14(2):651–661.
CrossRef - Khushmatov S.S., Zhumaev I.Z., Zhurakulov S.N., Saidov A.S., Vinogradova V.I. Synthesis and Comparative Inotropic Effects of Several Isoquinoline Alkaloids. Pharmaceutical Chemistry Journal.2020;54(1):7–11
CrossRef - Jumayev I., Usmanov P., Rustamov S., Zhurakulov, S. Comparative inotropic effects of the some isoquinoline alkaloids. Biomedical and Pharmacology Journal. 2020;13(1):325–333
CrossRef - ZhouH, Piao Z.X , Liu X et al. Arrhythmogenesis Toxicity of Aconitine Is Related to Intracellular Ca2+ Signals. Int J Med Sci. 2013;10(9):1242–1249.
CrossRef - Ziyavitdinov Z.F., Ishimov U.Z., Berdiev N.S. et al. Supramolecular Complex of Lappaconitine Hydrobromide and the Monoammonium Salt of Glycyrrhizic Acid: Synthesis, Physicochemical Characteristics, and Antiarrhythmic Activity. Pharmaceutical Chemistry Journal.2022;56(2):167–173.
CrossRef - Azamatov A.A., Zhurakulov Sh.N., Vinogradova V.I. et. al. Evaluation of the Local Anesthetic Activity, Acute Toxicity, and Structure–Toxicity Relationship in Series of Synthesized 1-Aryltetrahydroisoquinoline Alkaloid Derivatives In Vivo and In Silico. Molecules, 2023; 28, 477
CrossRef - ShangF., Yang Ch.J, Morris-Natschke S et al. Biologically Active Isoquinoline Alkaloids covering. Med Res Rev. 2020;40(6):2212–2289.
CrossRef - Prasanta Dey, Amit Kundu, Anoop Kumar and et al. Analysis of alkaloids (indole alkaloids, isoquinoline alkaloids, tropane alkaloids) Recent Advances in Natural Products Analysis. 2020; 20:505–567.
CrossRef - Zaripov A.A., Jumayev I.Z., Usmanov, P.B. et al. Protective Effect of DHQ-11 against Hypoxia-induced Vasorelaxation. Trends in Sciences, 2024;21(11), 8192:1-12.
CrossRef - LawsonW. Antiarrhythmic activity of some isoquinoline derivatives determined by a rapid screening procedure in the mouse. J Pharmacol Exp Ther. 1968;160(1):22-31.
CrossRef - Yang J, Ye K., Zhang R. et al. The characteristics and molecular targets of antiarrhythmic natural products. Biomedicine & Pharmacotherapy. 2023;168:1-15.
CrossRef - Bers D., Despa.S. Na+ Transport in Cardiac Myocytes; Implications for Excitation-Contraction Coupling. IUBMB Life. 2009;61(3):215–221.
CrossRef - Eisner D.A., Caldwell J.L, Kistamás K, and Trafford A.W. Calcium and Excitation-Contraction Coupling in the Heart. Circulation Research. 2017;121(2):181-195
CrossRef - Bers D. Calcium Fluxes Involved in Control of Cardiac Myocyte Contraction. Circulation Research. 2000; 87(4):275-81.
CrossRef - LiX., Saude E.J., Wang X, Pearlstone J.R, Smillie L.B, Sykes B.D. Kinetic studies of calcium and cardiac troponin I peptide binding to human cardiac troponin C using NMR spectroscopy. European biophysics journal. 2002;31(4):245-56.
CrossRef - Belardinellia L., Antzelevitchb Ch., Fraserc H. Inhibition of late (sustained/persistent) sodium current: a potential drug target to reduce intracellular sodium-dependent calcium overload and its detrimental effects on cardiomyocyte function. European Heart Journal Supplements. 2004;6:13-17.
CrossRef - Luo M and AndersonE. Mechanisms of Altered Ca2+ Handling in Heart Failure. Circulation Research. 2013;113(6):690-708.
CrossRef - Faber G.M, Rudy Y. Action Potential and Contractility Changes in [Na+]i Overloaded Cardiac Myocytes: A Simulation Study. Biophysical journal. 2000;78(5):2392-2404.
CrossRef - BersM., Barry W.H., Despa S. Intracellular Na+ regulation in cardiac myocytes. Cardiovascular Research, 2003;57(4):897–912.
CrossRef - Yuea X, Hazanb A, Lotteaub S et al. Na/Ca exchange in the atrium: Role in sinoatrial node pacemaking and excitation-contraction coupling. Cell Calcium. 2020;87:1-7.
CrossRef - HayterA, Wehrens S M, Van Dongen H.PA. et al. Distinct circadian mechanisms govern cardiac rhythms and susceptibility to arrhythmia. Nature Communications. 2021;12:1-13.
CrossRef - Kistamás K, Veress R, Horváth B, Bányász T, NánásiP., Eisner D.A. Calcium Handling Defects and Cardiac Arrhythmia Syndromes. Front Pharmacol. 2020;11(72):1-25.
CrossRef - RodenM. Antiarrhythmic drugs: From mechanisms to clinical practice. Heart (British Cardiac Society), 2000; 84(3):339-346.
CrossRef - Iranshahy M., Quinn R.J., Milad I. ChemInform Abstract: Biologically Active Isoquinoline Alkaloids with Drug-Like Properties from the Genus Corydalis. RSC Advances 2014; 4(31):15900-15913.
CrossRef - Chi‐Wai L., Xiaoqiang Y., ZhenY.Ch. Cardiovascular Actions of Berberine. Cardiovascular Drug Reviews. 2001; 19(3):234-44.
CrossRef - Roden D.M. Antiarrhythmic drugs: From mechanisms to clinical practice. Heart (British Cardiac Society). 2000; 84(3):339-46.
CrossRef - Norbert N., Noémi T., and Péter P.N. Antiarrhythmic and Inotropic Effects of Selective Na+/Ca2+ Exchanger Inhibition: What Can We Learn from the Pharmacological Studies? J. Mol. Sci. 2022; 23(23):14651.
CrossRef - RodenM. Pharmacology and Toxicology of Nav1.5-Class 1 anti-arrhythmic drugs. Card Electrophysiol Clin. 2014;6(4):695-704.
CrossRef - Grant A.O. Cardiac Ion Channels. Circulation: Arrhythmia and Electrophysiology. 2009;2(1):185-194.
CrossRef - KirchhofF., Fabritz C.L. and Franz M.R. Postrepolarization Refractoriness Versus Conduction Slowing Caused by Class I Antiarrhythmic Drugs : Antiarrhythmic and Proarrhythmic Effects. Circulation, 1998;97(25):2567-2574.
CrossRef







