Manuscript accepted on :09-05-2025
Published online on: 17-07-2025
Plagiarism Check: Yes
Reviewed by: Dr. Raju Thenge
Second Review by: Dr. Divya Madhuri Kallam
Final Approval by: Dr. Mariia Shanaida
Ram Niwas Jangir* , Gyan Chand Jain
, Gyan Chand Jain , Aditya Jain
, Aditya Jain and Tanvi Jangid
and Tanvi Jangid
Department of Zoology, University of Rajasthan, Jaipur, Rajasthan, India.
Corresponding Author E-mail: ramanjangir@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3244
Abstract
Diabetes mellitus (DM) has been linked to reproductive impairments. Medicinal plants have shown potential in alleviating diabetes-induced reproductive dysfunction in male. The Primary aim of the study was to assess the influence of an extract derived from Cassia fistula pod on reproductive hormone levels and testicular dysfunction in diabetic rats. A streptozotocin (STZ) dose (60 mg/kg b.wt.) was intraperitoneally (i.p.) injected to Wistar male rats to induce diabetes. 36 male rats were randomly assigned to six different groups: a healthy control group, a diabetic control group, three diabetic groups administered varying amount of Cassia fistula extract (100, 250, 500 mg/kg body weight per day), and a diabetic group receiving glibenclamide (5 mg/kg body weight per day). The treatment was given every day for 60 consecutive days. Levels of reproductive hormones including follicle-stimulating hormone (FSH), testosterone and luteinizing hormone (LH), along with oxidative stress in testicular tissue, were assessed. Histomorphometric and histopathological alterations in the testes were also examined. Diabetic control group exhibited significant decline in testicular weight, the testicular germ cells population, seminiferous tubular diameter and reproductive hormones like testosterone, FSH and LH as compared to control rats. Additionally, significant rise in lipid peroxidation (TBARS) alongside a simultaneous reduction in SOD and CAT activities as well as ascorbic acid and glutathione levels within the testicular tissues were observed compared to control rats. The administration of Cassia fistula extract or glibenclamide via oral route in diabetic rat led to improvements in serum insulin and reproductive hormone concentrations. Additionally, a reversal of histopathological and histomorphometric changes was noted relative to the diabetic reference group. Furthermore, the administration with the extract decreased lipid peroxidation and increased antioxidant levels in testicular tissue in comparison to the untreated diabetic rats. The outcomes of this research reveal that the hydroalcoholic extract from Cassia fistula pod exhibits significant antioxidant activities and can also modulate testicular dysfunction in diabetic male rats.
Keywords
Antioxidant activity; Cassia fistula; Histopathology; Oxidative stress; Streptozotocin; Testosterone; Testicular dysfunction
Download this article as:| Copy the following to cite this article: Jangir R. N, Jain G. C, Jain A, Jangid T. Ameliorative Effects of Cassia fistula Pod Extract on Testicular Histopathology, Oxidative Stress and Reproductive Hormone Levels in Streptozotocin Induced Diabetic Rats. Biomed Pharmacol J 2025;18(3). |
| Copy the following to cite this URL: Jangir R. N, Jain G. C, Jain A, Jangid T. Ameliorative Effects of Cassia fistula Pod Extract on Testicular Histopathology, Oxidative Stress and Reproductive Hormone Levels in Streptozotocin Induced Diabetic Rats. Biomed Pharmacol J 2025;18(3). Available from: https://bit.ly/40nKsLC |
Introduction
Today diabetes mellitus is amongst the most significant public health concerns, and its prevalence is rising rapidly worldwide. As estimated by the IDF, approximately 536.6 million adults globally were living with diabetes in 2021, with the figure expected to rise to nearly 783.2 million by 2045.1 Diabetes mellitus is marked by raised blood sugar level due to inadequate insulin production, ineffective insulin action, or both. Without treatment, persistently elevated blood sugar can lead to damage, dysfunction, and eventual failure of multiple organs. Reproductive dysfunction emerges as a significant secondary complication of diabetes mellitus in both males and females.2,3
Evidence from both animal studies and clinical investigations strongly suggests that diabetes mellitus negatively impacts male reproductive function and fertility.4 Research conducted on experimental diabetic rats has revealed a decrease in testosterone and gonadotropin levels.5,6 Numerous studies also reported that diabetes mellitus is associated with testicular dysfunctions viz. A decline in testicular weight, degradation of germ cells and seminiferous epithelium height, along with shrinkage and degenerative alterations in the testis has been observed.7,8
Research indicates oxidative stress as a crucial factor and as the primary cause of testicular impairment, ultimately leads to male hypogonadism, bad sperm quality, and infertility.9,10 Both the sperm and testes generate insulin, and insulin production within the testes seem to be affected by diabetes. However, the negative impact of diabetes could also arise from disrupted signaling of insulin in the testes and brain, systemic consequences of deficit in insulin, or a combination of both factors.11,12 Numerous investigations have demonstrated that high blood glucose levels led to an excess of reactive ROS and free radicals to be produced, surpassing the organism’s cellular antioxidant defense capacity and disrupting the cellular reduction-oxidation balance.13,14 Oxidative stress may be lessened by taking antioxidant supplements. Natural antioxidants found in medicinal plants and herbs are abundant and can be used to treat diabetes and its consequences.14-16
In India, a significant variety of plants have been utilized for treating diabetes and its complications since the time of Charaka and Sushruth.15 Review of literature reveals that various plant extracts and their bioactive components exhibit ameliorative impacts on oxidative stress and diabetic complications. These natural products-based phytochemicals target increased insulin secretion, regeneration/protection of insulin secreting β-cells, some have extra pancreatic effect by acting directly on tissue like liver, muscles etc. and regulation of glycolysis and gluconeogenesis, inhibit carbohydrate digestion and absorption by blocking α-glucosidase activity in the intestine.17,18
Review of literature reveals that various plant extracts and bioactive compounds have shown beneficial effects in maintaining blood glucose levels, strengthening antioxidant defense mechanisms, and improving reproductive health in male diabetic rats. This emphasizes the significance of phytotherapy as a possible approach for managing diabetes and related male reproductive disorders.19,20
Cassia fistula Linn (Family–Caesalpiniaceae) is an indigenous medicinal plant and cultivated throughout India as an ornamental tree. This species is native to the Indian Subcontinent and Myanmar. It is a tree that predominantly thrives in seasonally dry tropical regions. It serves multiple purposes, including use as animal feed, traditional medicine, and poison. Additionally, it has environmental and social applications, and is utilized for food and fuel. In traditional medicine, virtually every part of Cassia fistula is utilized for the treatment of diverse ailments. The pods of plant are used as abortifacient, anodyne, anti-bilious, antidiabetic, anti-inflammatory, antipyretic, astringent, depurative, diuretic, emollient, purgative and tonic. Phytochemical research has revealed that Cassia fistula pods are an abundant source of vital nutrients such as calcium (Ca), potassium (K), iron (Fe), and manganese (Mn). The pod of Cassia fistula are known to contain various phytochemicals, such as 1,8-dihydroxy-3-anthraquinone carboxylic acid, rhein, 3-formyl-1-hydroxy-8-methoxy anthraquinone, 3B-hydroxy-17-norpimar-8(9)-en-15-one, quercetin dehydrate, dimeric proanthocyanidin CFI, (+) catechin, (-) epiafzelechin, dihydrokaempferol, kaempferol and 1,8-dihydroxy-3-methylanthraquinone.21-24
The hypoglycemic impact of Cassia fistula extract has been shown in diabetic rats induced with STZ.25-27 Furthermore, in-vitro studies and/or animal model studies have also reported the antioxidant activity of C. fistula pod/fruit pulp extract.25,28,29
As far as we are aware, research on the influence of Cassia fistula pod extract on diabetes-induced reproductive dysfunctions has been found to be very limited. Therefore, the objective of the current study is to assess the ameliorative effects of the Cassia fistula pod extract on male reproductive dysfunctions, particularly focusing on testicular dysfunctions and serum levels of reproductive hormones.
Materials and Methods
Plant collection and preparation of its extract
The pods of Cassia fistula were gathered from the University of Rajasthan campus between the months of April-June. The plant specimen was verified by botanist of herbarium, ‘University of Rajasthan’, Jaipur (Voucher No. RUBL21057). The mature pods of the plant were dried in shed then the dried pods were powder by using an electric grinder. The obtained powder was mixed with 70% ethanol and left undisturbed for 1 day at room temperature. The mixture underwent extraction using a Soxhlet apparatus at a temperature of 60°C. High-temperature and prolonged heating are avoided to prevent the decomposition of phytochemicals. After extraction, the solution underwent filtration using filter paper, and obtained filtrate was subsequently dried in the oven at a temperature of 40°C. The thick brown paste yield from Cassia fistula was approximately 33 g (13.2% of the dried powder). This residue was stored in a sealed airtight bottle within a refrigerator until needed.
Animals
In this study, Wistar strain male rats (Rattus norvegicus), weighing between 170 and 210 grams, were selected. They were housed in polypropylene cages in the department’s animal facility (1678/Go/ReBi/S/2012/CCSEA) under standard laboratory conditions. The rats were given water and nutritious pellet diet. Animal handling was conducted in accordance with the standards established by the “Committee for the Purpose of Control and Supervision of Experiments on Animals” (CPCSEA). The research received approval from the Institutional Animal Ethics Committee.
Inducing diabetes
Rats that had fasted overnight were induced with diabetes mellitus by a single intraperitoneal injection of STZ at a dosage of 60 mg/kg, mixed with freshly prepared citrate buffer (pH 4.5). Seven days following the STZ administration, fasting blood glucose levels were assessed by collecting samples of blood from the tail vein of rats that had been fasted overnight. Rats with fasting blood glucose concentrations above 250 mg/dl were classified as diabetic and selected for inclusion in the study.
Experimental protocol
The animals were split into six groups, each group containing 6 rats, and all were treated orally for 60 consecutive days. The groups were organized as follows:
Group I: Normal control rats were given 0.5 ml of DW daily.
Group II: Untreated diabetic rats were given 0.5 ml of DW daily.
Group III: Diabetic rats were provided a low dose of extract (100 mg/kg) in 0.5 ml.
Group IV: Diabetic rats were provided a medium dose of extract (250 mg/kg) in 0.5 ml.
Group V: Diabetic rats were provided a high dose of extract (500 mg/kg) in 0.5 ml.
Group VI: Diabetic rats were given glibenclamide at a dose of 5 mg/kg in 0.5 ml.
Testicular weight and Serum hormone profiles
At the conclusion of the intervention period, all rats that had fasted overnight were weighed and euthanized under light ether anesthesia for autopsy. The testes were removed, rinsed with ice-cold saline, cleaned of any residual fat and blood clots, and then weighed using an electronic balance. Samples of blood were collected via direct puncture in heart, and serum was isolated by centrifuging the blood at 3000 rpm for ten minutes. The concentrations of serum testosterone, FSH, insulin, and LH were assessed using a chemiluminescent immunoassay.
Testicular lipid peroxidation and antioxidant markers
Testicular tissue samples from rats in various groups were utilized to measure Lipid peroxidation (TBARS),30 Reduced glutathione (GSH),31 Superoxide dismutase (SOD),32 AA (Vitamin C),33 and Catalase (CAT),34. A minimum of six samples from each group were collected and averaged.
Histopathological study
For histopathological examination, testis tissue samples were initially preserved in Bouin’s solution. After fixation, the samples were rinsed with distilled water (DW) to remove any excess fixative. Next, the tissues underwent dehydration by immersing them in a series of concentrated alcohol solutions, then cleared with xylene to eliminate any remaining alcohol. The tissues were subsequently embedded in paraffin wax for structural stability. After embedding, the tissues were sliced into thin sections, each 5 µm thick. These sections were stained using eosin and hematoxylin for enhanced visualization. The prepared slides were observed under a light microscope, and images were clicked with a Nikon digital camera connected to the microscope.
Histomorphometric analysis
Light microscope, fitted with an ocular micrometer and calibrated using a stage micrometer, was utilized to assess seminiferous tubules diameter (STs) and the nuclear size of Leydig cells. At a magnification of X100, two perpendicular diameters were recorded for each seminiferous tubule (40 tubules per group). These measurements were then averaged to determine the seminiferous tubule diameter. For the Leydig cells, nuclear diameters were measured at X1000 magnification (100 cells per group), and the results were averaged to calculate the mean Leydig cell nuclear diameter.
Stage VII of the seminiferous tubule cycle was used for the quantitative evaluation of reproductive cells, such as spermatogonia and pachytene spermatocytes, preleptotene spermatocytes, and round spermatids, per cross-section (50 circular tubules per group) at X400 magnification.35 The nuclear diameter of various types of germ cells was measured by using an ocular micrometer. A correction factor was applied to determine the accurate number density of germ cells.36

Statistical analysis
The data was examined and processed using SPSS 20.0 software for Windows (SPSS Inc., Chicago, IL, USA). Results are displayed as the average ± standard error of the mean (SEM), with variation assessed. One-way analysis of variance (ANOVA) was utilized to evaluate statistical differences, followed by Tukey’s post hoc analysis. A p-value below 0.05 was regarded as statistically significant.
Results
Testicular weight
Compared to group I, the testicular weight of diabetic rats (group II) decreased and were statistically significant (P≤0.001). When Cassia fistula extract was given to diabetic rats at low, medium, and high dosages, the absolute testicular weight increased in a dose related and significant manner (P≤0.05, P≤0.01, and P≤0.001, respectively). Glibenclamide administration also results in a significant (P≤0.001) rise in the absolute testicular weight compared to the diabetic control group (group II) as illustrated in Figure 1.
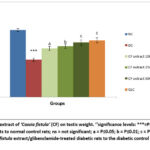 |
Figure 1: Effect of extract of ‘Cassia fistula’ (CF) on testis weight. ‘‘significance levels: ***=P≤0.001, comparing diabetic control rats to normal control rats; ns = not significant; Click here to view Figure |
Serum hormones
Diabetic rats in group II showed a significant decrease (P≤0.001) in serum insulin, testosterone, FSH, and LH levels relative to the control rats in group I. Nevertheless, administering Cassia fistula extract at doses of 250 mg/kg as well as 500 mg/kg resulted in a significant increase in insulin levels (P≤0.05 and P≤0.001, respectively) in the diabetic rats. Likewise, there was a marked rise in testosterone, FSH, and LH levels (P≤0.01 and P≤0.001, respectively) relative to diabetic control group. Similar improvements were observed in group VI diabetic rats, with a significant rise (P≤0.001) in these serum hormones relative to group II diabetic rats (Table 1).
Table 1: Effects of extract of ‘Cassia fistula’ on levels of insulin and reproductive hormone.
| Groups | Insulin(µIU/mL) | Testosterone(ng/ml) | FSH(mIU/mL) | LH(mIU/mL) |
| Group INormal Control | 14.32±0.77 | 6.30±0.48 | 2.40±0.16 | 3.31±0.14 |
| Group IIDiabetic Control | 4.18±0.29*** | 1.89±0.16*** | 0.87±0.07*** | 1.10±0.08*** |
| Group IIIDiabetic+100mg/kg of Extract | 5.91±0.36 ns | 3.02±0.19 ns | 1.42±0.11 ns | 1.65±0.13 ns |
| Group IVDiabetic+250mg/kg of Extract | 6.98±0.44 a | 3.58±0.24 b | 1.67±0.14b | 2.02±0.16b |
| Group VDiabetic+500mg/kg of Extract | 9.18±0.66 c | 4.60±0.33 c | 1.95±0.15c | 2.64±0.18 c |
| Group VI Diabetic+5mg/kg of Glibenclamide | 10.32±0.85 c | 4.70±0.38 c | 2.01±0.16 c | 2.72±0.24 c |
‘‘The values have been expressed as mean ± SEM (n=6), with the following levels of significance: *** = P≤0.001, comparing diabetic control rats to normal control rats; ns = not significant; a = P≤0.05; b = P≤0.01; c = P≤0.001, comparing Cassia fistula extract/glibenclamide-treated diabetic rats to diabetic control rats’’.
Lipid peroxidation
The testis tissue of group II diabetic rats revealed a noteworthy rise (P≤0.001) in levels of TBARS as compared to the group I normal rats. Diabetic rats which were given lower, medium, and higher doses of Cassia fistula extract exhibited a substantial dose responsive reduction (P≤0.05, P≤0.01, P≤0.001, respectively) in levels of TBARS in the testis in contrast to group II diabetic rats. Additionally, group VI rats displayed a remarkable (P≤0.001) fall in TBARS levels relative to group II diabetic rats (Figure 2).
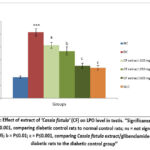 |
Figure 2: Effect of extract of ‘Cassia fistula’ (CF) on LPO level in testis. ‘‘Significance levels: ***=P≤0.001, comparing diabetic control rats to normal control rats; ns = not significant;Click here to view Figure |
Antioxidant Marker Parameters
Diabetic rats of group II exhibited a significant reduction (P≤0.001) in SOD, GSH, CAT and ascorbic acid levels in testicular tissue relative to group I normal rats. However, diabetic rats treated with medium (group IV) and high (group V) doses of Cassia fistula extract displayed a marked increase in SOD and CAT activities, likewise GSH and ascorbic acid levels (P≤0.01 and P≤0.001, respectively) as compared to group II diabetic rats. In group III, treatment with a low dose of Cassia fistula extract to diabetic rats exhibited a slight but marked increase in ascorbic acid levels. Additionally, group VI exhibited a significant improvement (P≤0.001) in all antioxidant markers (Figure 3-6).
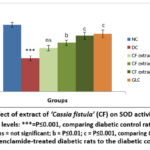 |
Figure 3: Effect of extract of ‘Cassia fistula’ (CF) on SOD activity in testis. ‘‘Significance levels: ***=P≤0.001, comparing diabetic control rats to normal control rats; ns = not significant;Click here to view Figure |
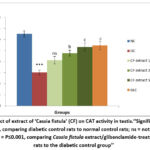 |
Figure 4: Effect of extract of ‘Cassia fistula’ (CF) on CAT activity in testis. ‘‘Significance levels: ***=P≤0.001, comparing diabetic control rats to normal control rats; ns = not significant;Click here to view Figure |
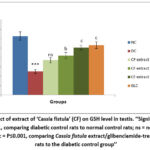 |
Figure 5: Effect of extract of ‘Cassia fistula’ (CF) on GSH level in testis. ‘‘Significance levels: ***=P≤0.001, comparing diabetic control rats to normal control rats; ns = not significant;Click here to view Figure |
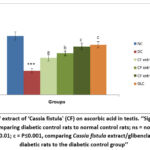 |
Figure 6: Effect of extract of ‘Cassia fistula’ (CF) on ascorbic acid in testis. ‘‘Significance levels: ***=P≤0.001, comparing diabetic control rats to normal control rats; ns = not significant;Click here to view Figure |
Histomorphometric analysis
Table 2 presents changes in the nuclear diameter of Leydig cells and diameter of STs along with the population of testicular germ cells. In diabetic group II, there was a noteworthy (P≤0.001) decline in the average diameter of STs and Leydig cell nuclei relative to control group I. Administration of Cassia fistula extract in the medium dose (group IV) and high dose (group V) resulted in a significant (P≤0.01 and P≤0.001, respectively) rise in the average diameter of seminiferous tubules and Leydig cell nuclei compared to the diabetic group II. Reference drug treated group VI also demonstrated a noteworthy (P≤0.001) increase in the average diameter of STs and Leydig cell nuclei compared to group II.
Diabetic control group II exhibited a noteworthy (P≤0.001) decline in population of spermatogonia, pachytene spermatocytes, preleptotene spermatocytes, likewise round spermatids as relative to normal control group I. There was a dose responsive rise in different types of germ cells, a non-significant increase in the low dose group III (except for round spermatids), a moderately notable (P≤0.01) increase in the medium dose group IV, and a highly significant (P≤0.001) increase in the highest dose group V compared to diabetic control group II. Glibenclamide-treated group VI also exhibited a notable (P≤0.001) rise in the numbers of all types of germ cells, relative to the diabetic control group II.
Table 2: Effects of extract of ‘Cassia fistula’ on seminiferous tubules diameter, nuclear diameter of Leydig cell and histomorphometric indices.
| Groups | Diameter of STs (µ m) | Nuclear diameter of Leydig cells (µ m) | Spermatogonia | Spermatocyte (Preleptolene) | Spermatocyte (Pachytene) | Spermatid |
| Group INormal Control | 272.67±8.32 | 6.67±0.33 | 6.33±0.25 | 20.17±1.08 | 23.67±1.52 | 64.87±2.37 |
| Group IIDiabetic Control | 168.50±9.75*** | 3.50±0.26*** | 3.83±0.31*** | 11.50±0.76*** | 13.83±1.25*** | 28.00±2.06*** |
| Group IIIDiabetic+100mg/kg of Extract | 200.83±10.15 ns | 4.65±0.27ns | 4.85±0.34ns | 14.13± 0.97 ns | 17.70±0.95 ns | 39.37±2.19 a |
| Group IVDiabetic+250mg/kg of Extract | 223.67±9.69 b | 5.27±0.21 b | 5.52±0.32 b | 16.20±0.91 b | 19.55±0.90 b | 43.97±2.50 b |
| Group VDiabetic+500mg/kg of Extract | 242.50±8.30 c | 6.10±0.32 c | 5.80±0.25 c | 17.78±0.61 c | 21.60±0.69 b | 52.90±3.06 c |
| Group VI Diabetic+5mg/kg of Glibenclamide | 248.50±8.69 c | 6.20±0.37 c | 5.85±0.35 c | 17.85±1.01 c | 22.00±1.15 c | 56.40±3.29 c |
‘‘The values are presented as mean ± SEM (n=6), with the following significance levels: *** = P≤0.001, comparing diabetic control rats to normal control rats; ns = not significant; a = P≤0.05; b = P≤0.01; c = P≤0.001, comparing Cassia fistula extract/glibenclamide-treated diabetic rats to the diabetic control group’’.
Histopahological study
Histopathological study of testis of normal control rats of group I exhibited normal testicular histoarchitecture with large no. of spermatozoa found in the lumen (Figure 7a). In contrast, testis of diabetic rats of group II showed significantly (P≤.001) reduced size of STs and the morphology of the seminiferous epithelium was severely impaired. Many tubules did not contain sperm cells or spermatozoa (Figure 7b). Group III (low dose of C. fistula extract) exhibited mild prevention of degenerative changes in the histoarchitecture compared with group II rats (Figure 7c). Administration of medium dose of extract to diabetic rats (group IV) improved testicular histoarchitecture, evidenced by increased number of spermatogenic cells and spermatozoa. Furthermore, there was a significant reduction in the degenerative and vacuolated structure of STs (Figure 7d). The testis of diabetic rats receiving high doses of C. fistula extract (group V), exhibited normal histological structure of most STs and an increase of spermatogenic density as the lumen seemed to be filled with sperm (Figure 7e). Standard drug treated diabetic rats (group VI) revealed almost normal appearance of histoarchitecture with a moderate number of spermatozoa (Figure7f).
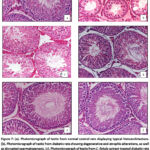 |
Figure 7: (a). Photomicrograph of testis from normal control rats displaying typical histoarchitecture. (b). Photomicrograph of testis from diabetic rats showing degenerative and atrophic alterations, as well as disrupted spermatogenesis. Click here to view Figure |
Discussion
The current study demonstrated diabetes mellitus induced reproductive dysfunctions and highlighted the protective effects of Cassia fistula pod extract on several assessed parameters. Notably, there was a marked reduction in absolute weight of testis in STZ-induced diabetic control rats. These findings are compatible with many earlier reports in which similar fall of absolute weight of testis have been reported in STZ-induced diabetic rats.37,38,39 The decrease in testicular weight in diabetic rats might be attributed to the suppression of testosterone synthesis by Leydig cells as well as loss of spermatids, spermatozoa and Sertoli cells.40,41 However, diabetic rats receiving treatment with either Cassia fistula extract or glibenclamide exhibited a noteworthy improvement in testis weight. This could be due to better glycemic homeostasis and redox state resulting in restoration of spermatogenic and steroidogenic activities.
The results of the present study indicate that the administration of streptozotocin (STZ) led to a significant reduction in the amounts of insulin, FSH, LH, and testosterone in their serum. These findings are consistent with the observations of several researchers who have documented substantial reductions in insulin levels, concomitant with decreases in testosterone, FSH, and LH levels in STZ-induced diabetic rats.42,43 Additionally, similar significant decreases in testosterone, FSH, and LH levels have been reported in the diabetic patients by various studies.44,45
The reduction in FSH as well as LH observed in the diabetic rats might be linked to deficiency of insulin, as several studies have shown a relationship between levels of insulin and the biosynthesis and secretion of pituitary’s LH and FSH hormones.46,47,48 The decline in serum testosterone level in STZ-induced diabetic rats could be attributed to reduced levels of LH which may cause both, a reduction of total number of Leydig cells and impairment of function of Leydig cell resulting in decrease of biosynthesis and secretion of androgens.49,50 Furthermore, increased oxidative stress during diabetic state may also cause impairment of steroidogenesis in Leydig cells.51
The findings of the present research also demonstrated that treating diabetic rats by Cassia fistula extract or glibenclamide led to a notable enhancement in the serum insulin, testosterone, LH, and FSH levels. These outcomes are aligned with previous reports that demonstrated a noteworthy increase in insulin level in experimental diabetic rats following treatment with the methanolic extract of various parts of the Cassia fistula52 or ethanolic extract of leaves53 or hexane extract of bark54 or ethanolic extract of flowers55 of Cassia fistula. The observed improvements in LH, testosterone, and FSH levels in serum of diabetic rats treated with Cassia fistula extracts could be linked to the rejuvenating effects of the phytoconstituents of the extract. This may lead to improved synthesis and secretion of insulin, consequently facilitating the resumption of Sertoli and Leydig cells functions in testis.56
Comparable ameliorative effects of glibenclamide on sex hormones have also been documented in diabetic rats.57,58 The useful effects of glibenclamide may be due to their insulinogenic action. Glibenclamide blocks the ATP-sensitive potassium (KATP) channels on the β-cell membrane, causing depolarization, activation of the voltage gated Ca++ and thereby insulin release.59
The study results also exhibited a significant rise in lipid peroxidation (TBARS) and a notable decrease in antioxidant markers like CAT, GSH, AA and SOD in testicular tissues. These findings are uniform with numerous prior studies that reported comparable reductions in antioxidants and elevated TBARS levels in the testicular tissue of diabetic animals.43,57,60 Hyperglycemia induced by hypoinsulinemia leads to glucose auto-oxidation, glycation of non-enzymatic proteins, activation of the polyol pathway, and the excessive formation of ROS, resulting in oxidative stress.13 The observed rise in the oxidative stress in testis may be linked to a decrease in levels of testosterone, which is associated with reduced serum insulin concentration.
Cassia fistula extract treatment exhibited a notable decline in TBARS levels and a simultaneous elevation in antioxidant parameters within the testicular tissues of diabetic rats. These findings indicate the robust antioxidant activity of the phytoconstituents contained in the extract such as catechin, epicatechin, anthraquinone, kaempferol, quercetin dehydrate etc.28,61 These results align with earlier reports that have also indicated improvements in antioxidant parameters alongside a decline in TBARS level in diabetic rats following extracts treatment derived from various parts of Cassia fistula.25,62
Histological picture of the testis in diabetic control rats revealed marked degenerative and atrophic changes in the testis. There was a marked reduction in the quantity of various types of germ cells and spermatozoa within the STs. The interstitial space was widened and the diameter of STs was also decreased. These findings align with prior research that observed similar atrophic and degenerative changes, along with impaired spermatogenesis, in diabetic rats testes.37,63,64
The degenerative and atrophic changes showed in the diabetic rats testes might be linked to the dysfunction of the hypothalamic-pituitary-gonadal (HPG) axis, leading to reduced levels of FSH, LH and testosterone due to insulin deficiency. It has been postulated that LH-mediated proliferation of Leydig cells involves a mechanism incorporating insulin as well as insulin-like growth factor (IGF-1) signaling.65 Consequently, inadequate insulin in STZ-induced diabetic rats causes a decline in both the number and metabolic functions of Leydig cells, ultimately contributing to a decline in testosterone levels. The observed reduced nuclear diameter and atrophy of Leydig cells in the present study support this mechanism. Adequate levels of testosterone in the testes are crucial for normal spermatogenesis and the maintenance of the normal morphology of STs.66 The depletion in different types of germ cell populations within the testes of diabetic rats provides additional evidence for the reduced availability of testosterone. Additionally, degenerative and atrophic changes in Sertoli cells adversely affect spermatogenic functions. Sertoli cells play a vital role in reproductive functions and spermatogenesis, secreting diverse growth factors and transport proteins.64,67
Moreover, persistent hyperglycemia and insulin deficiency can cause testicular cells to undergo metabolic adaptations and resort to alternative substrates, which induce oxidative stress.68 The overproduction of ROS and free radicals can suppress endogenous antioxidant enzymes, disrupt testicular function, and result in apoptosis of germ cells and cell death. Numerous studies have previously observed an increased occurrence of the induction of apoptosis in germ cells within the testes of type 1 diabetic rats and mice.64,69,70
Administration of Cassia fistula extract/glibenclamide to diabetic rats led to significant improvements in the histological architecture of their testes. STs showed enlargement, with active spermatogenesis and Leydig cells exhibited morphology close to normal. These beneficial outcomes are likely due to the restoration of testosterone and insulin levels, along with the alleviation of oxidative damage, achieved through the combined action of phytoconstituents present in the extract. Numerous plants with antihyperglycemic properties have demonstrated a mitigating effect on reproductive dysfunction, attributable to the presence of bioactive polyphenolic phytoconstituents.19,71 Treatment with glibenclamide in diabetic rats also significantly improved testicular histomorphology, consistent with previous findings in diabetic rats.57,60,72,73
Conclusion
The finding from the current study showed that hydroalcoholic extract of Cassia fistula pod significantly ameliorate diabetes induced testicular dysfunctions via multiple mechanism including increase insulin secretion, inhibition of lipid peroxidation, enhancement of the antioxidant activity/concentration and preservation of the concentration of hormone FSH, testosterone and LH. This research provides scientific proof for the traditional utilization of pod of Cassia fistula in the control of diabetes mellitus. Additional research is required to identify the particular bioactive compounds responsible for this therapeutic effect.
Acknowledgement
We extend our gratitude to the Head of the Department of Zoology, University of Rajasthan, Jaipur, for offering the essential resources and support.
Funding Source
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
The animals were maintained in accordance with the guidelines of the CPCSEA and experimental protocol was approved by Institutional animal ethical committee, Department of zoology, University of Rajasthan, Jaipur.
Informed Consent Statement
The informed consent was obtained for experimentation and that it conforms to the standards currently applied in the country of origin. The privacy rights of human subjects must always be observed.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not applicable
Author Contributions
- Ram Niwas Jangir: Data Collection, Methodology, and Writing – Original Draft;
- Gyan Chand Jain: Conceptualization, Analysis, Supervision & Editing;
- Aditya Jain: Review editing, Table and figure formatting:
- Tanvi Jangid: Visualization, Review and editing.
- Each author mentioned has significantly and directly contributed intellectually to the project and has given their approval for its publication.
References
- Sun H, Saeedi P, Karuranga S, et al. Erratum to “IDF Diabetes Atlas: Global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045” Diabetes Res. Clin. Pract. 2023; 204: 110945. doi: 10.1016/j.diabres.2023.110945.
CrossRef - American Diabetes Association (ADA). Diagnosis and classification of diabetes mellitus. Diabetes Care. 2014; 37(Suppl 1): S81–S90. doi: 10.2337/dc14-S081.
CrossRef - Basmatzou T, Hatziveis K. Diabetes mellitus and influences on human fertility. Int. J. Caring Sci. 2016; 9(1): 371–379.
- Jangir RN, Jain GC. Diabetes mellitus-induced impairment of male reproductive functions: A review. Curr. Diabetes Rev. 2014; 10(3): 147–157. doi: 10.2174/1573399810666140606111745.
CrossRef - Bhandari MK, Mukherjee A, Ray I and Acharya MS. Effect on the pituitary-testicular axis in streptozotocin-induced diabetic rats. World J. Pharm. Res. 2015; 4(9): 2096–2106. doi: 10.20959/wjpr20159-6181.
- Jangir RN, Jain GC. Ameliorative effect of Moringa oleifera Lam. leaves extract on the sex hormone profile and testicular dysfunctions in streptozotocin-induced diabetic Wistar rats. Pharmacog. Res. 2022; 14(2): 225–232. doi: 10.5530/pres.14.2.32.
CrossRef - Scarano WR, Messias AG, Oliva SU, Klinefelter GR and Kempinas WG. Sexual behaviour, sperm quantity, and quality after short-term streptozotocin-induced hyperglycaemia in rats. Int. J. Androl. 2006; 29(4): 482–488. doi: 10.1111/j.1365-2605.2006.00697. x.
CrossRef - Kotian SR, Kumar A, Mallik SB, Bhat NP, Souza AD and Pandey AK. Effect of diabetes on the male reproductive system—A histomorphological study. J. Morphol. Sci. 2019; 36(1): 17–23. doi: 10.1055/s-0039-1688405.
CrossRef - Amaral S, Oliveira PJ, and Ramalho-Santos J. Diabetes and the impairment of reproductive function: Possible role of mitochondria and reactive oxygen species. Curr. Diabetes Rev. 2008; 4: 46-54.
CrossRef - Shaikh H, Shrivastava VK, and Amir M. Diabetes mellitus and impairment of male reproductive function: Role of the hypothalamus-pituitary-testicular axis and reactive oxygen species. Iran. J. Diabetes Obes. 2016; 8: 41-50.
- Gomez O, Ballester B, Romero A, et al. Expression and regulation of insulin and the glucose transporter GLUT8 in the testes of diabetic rats. Horm. Metab. Res. 2009; 41: 343-349.
CrossRef - Schoeller EL, Schon S, and Moley KH. The effects of type 1 diabetes on the hypothalamic, pituitary, and testes axis. Cell Tissue Res. 2012; 349: 839-847.
CrossRef - Ahmed RG. The physiological and biochemical effects of diabetes on the balance between oxidative stress and the antioxidant defense system. Med. J. Islam. World Acad. Sci. 2005; 15(1): 31-42.
- Darenskaya MA, Kolesnikova LI and Kolesnikov SI. Oxidative stress: Pathogenetic role in diabetes mellitus and its complications and therapeutic approaches to correction. Bull. Exp. Biol. Med. 2021; 171(2): 179-189. doi: 10.1007/s10517-021-05191-7.
CrossRef - Grover JK, Yadav S and Vats V. Medicinal plants of India with anti-diabetic potential. J. Ethnopharmacol. 2002; 81(1): 81-100. doi: 10.1016/S0378-8741(02)00059-4.
CrossRef - Asadi N, Bahmani M, Kheradmand A and Rafieian-Kopaei M. The impact of oxidative stress on testicular function and the role of antioxidants in improving it: A review. J. Clin. Diagn. Res. 2017; 11(5): IE01-IE05. doi: 10.7860/JCDR/2017/23927.9886.
CrossRef - Noor A, Bansal VS and Vijayalakshmi MA. Current update on anti-diabetic biomolecules from key traditional Indian medicinal plants. Curr. Sci. 2013; 104: 721-727.
- Saad B, Zaid H, Shanak S, and Kadan S. Anti-diabetes and anti-obesity medicinal plants and phytochemicals: Safety, efficacy, and action mechanisms. Springer. 2017.
CrossRef - Jain GC and Jangir RN. Modulation of diabetes-mellitus-induced male reproductive dysfunctions in experimental animal models with medicinal plants. Pharmacog. Rev. 2014; 8(15): 113-121. doi:10.4103/0973-7847.134245.
CrossRef - Oliveira JS, Silva AAN, Junior Silva VA. Phytotherapy in reducing glycemic index and testicular oxidative stress resulting from induced diabetes: A review. Braz. J. Biol. 2017; 77(1): 68-78. doi:10.1590/1519-6984.09915.
CrossRef - Khare CP. Encyclopedia of Indian medicinal plants. Springer, Berlin, Heidelberg, New York. 2007; 128.
CrossRef - Mwangi RW, Macharia JM, Wagara IN, Bence RL. The medicinal properties of Cassia fistula L: A review. Biomed. Pharmacother. 2021; 144: 112240. doi: 10.1016/j.biopha.2021.112240.
CrossRef - POWO (2025). “Plants of the World Online. Facilitated by the Royal Botanic Gardens, Kew. Published on the Internet; https://powo.science.kew.org/Retrieved 30 April 2025.
- Jangid T, Jain A, Bhardwaj GS, Jangir RN. A Comprehensive review of phytochemical and pharmacological appraisal of Cassia fistula Linn. Pharmacog. Res. 2025;17(2):326-47.
CrossRef - Jangir RN and Jain GC. Evaluation of antidiabetic activity of hydroalcoholic extract of Cassia fistula Linn. pod in streptozotocin-induced diabetic rats. Pharmacogn. J. 2017; 9(5): 599-606. doi:10.5530/pj.2017.5.95.
CrossRef - Jangir RN and Jain GC. Evaluation of protective effects of hydroalcoholic extract of Cassia fistula Linn. pod on pancreas in streptozotocin-induced diabetic rats. Phcog. Res. 2018; 10(2): 205-212. doi:10.4103/pr.pr_95_17.
CrossRef - Patil VP, Amarshetty NA, Hugar S, Nanjappaiah HM, Navanath V. Antihyperglycemic activity of Cassia fistula fruit extracts in streptozotocin-induced diabetes. Asian J. Phytomed. Clin. Res. 2016; 4(3): 100-107.
- Maheep B, Sunil V, Yogesh V, Durgesh S and Kanika S. Antioxidant activity of fruit pulp of Cassia fistula. Pharmacogn. J. 2010; 2(8): 219-228. doi:10.1016/S0975-3575(10)80097-5.
CrossRef - Kalaiyarasi C, Karthika K, Lalithkumar P, Ragupathi G, Saravanan S. In vitro antioxidant activity of various solvent fractions of Cassia fistula L. pods. J. Pharmacogn. Phytochem. 2014; 3(4): 73-76.
- Ohkawa H, Ohishi N, Yagi K. Assay for lipid peroxide formation in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 1979; 95(2): 351-358. doi:10.1016/0003-2697(79)90738-3.
CrossRef - Moron MJ, Depieare JW, Mannivik B. Levels of glutathione reductase and glutathione transferase activities in rat lung and liver. Biochim. Biophys. Acta. 1979; 582(1): 67-68. doi:10.1016/0304-4165(79)90289-7.
CrossRef - Marklund S, Marklund G. Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur. J. Biochem. 1974; 47(3): 469-474. doi:10.1111/j.1432-1033. 1974.tb03714. x.
CrossRef - Roe JH and Kuether CA. Determination of ascorbic acid in whole blood and urine through the 2,4-DNPH derivative of dehydroascorbic acid. J. Biol. Chem. 1943; 147(2): 399-407.
CrossRef - Claiborne A. Catalase activity. In: Greenwald, R. (Ed) CRC Handbook of Methods for Oxygen Radical Research. CRC Press, Boca Raton, Florida. 1985; 283-284. doi:10.1201/9781351072922.
CrossRef - Leblond CP and Clermont Y. Definition of the stages of the cycle of the seminiferous epithelium in the rat. Ann. N. Y. Acad. Sci. 1952; 55(4): 548-573. doi:10.1111/j.1749-6632. 1952.tb26576. x.
CrossRef - Abercrombie M. Estimation of nuclear population from microtome sections. Anat. Rec. 1946; 94: 239-247. doi:10.1002/ar.1090940210.
CrossRef - Nak-ung S, Nakprom N, Maneengam C, Nudmamud-Thanoi S and Thanoi S. Changes in sperm quality and testicular structure in a rat model of type 1 diabetes. Asian Biomed. (Res. Rev. News). 2018; 12(4): 141-147. doi:10.1515/abm-2019-0014.
CrossRef - Jangir RN and Jain GC. Effects of Moringa oleifera Lam. on reproductive organs weight and tissue biochemistry in streptozotocin-induced diabetic rats. J. Nat. Remedies. 2025; 25: 321-331.
CrossRef - Nagy AM, Fahmy HA, Abdel-Hameed MF, et al. Protective effects of Euphorbia heterophylla against testicular degeneration in streptozotocin-induced diabetic rats in relation to phytochemical profile. PLoS ONE. 2025; 20: e0314781. https://doi.org/10.1371/journal.pone.0314781
CrossRef - Foley GL. Overview of male reproductive pathology. Toxicol. Pathol. 2001; 29(1): 49-63. doi:10.1080/019262301301418856.
CrossRef - Chandra AK, Sengupta P, Goswami H and Sarkar M. Effects of dietary magnesium on testicular histology, steroidogenesis, spermatogenesis, and oxidative stress markers in adult rats. Indian J. Exp. Biol. 2013; 51: 37-47.
- Schoeller EL, Schon S, Moley KH. The effects of type 1 diabetes on the hypothalamic, pituitary, and testes axis. Cell Tissue Res. 2012; 349: 839-847.
CrossRef - El-Beltagy AEFB, Abou El-Naga AM, El-Habibi ESM and Shams SESM. Ameliorative role of neem (Azadirachta indica) leaves ethanolic extract on testicular injury of neonatally diabetic rats induced by streptozotocin. Egypt J. Basic Appl. Sci. 2020; 7: 210-225.
CrossRef - Maneesh M, Jayalakshmi H, Singh TA and Chakrabarti A. Impaired hypothalamic-pituitary-gonadal axis function in men with diabetes mellitus. Indian J. Clin. Biochem. 2006; 21: 165-168.
CrossRef - Chandel A, Dhindsa S, Topiwala S, Chaudhuri A and Dandona P. Testosterone concentrations in young patients with diabetes. Diabetes Care. 2008; 31: 2013-2017.
CrossRef - Baccetti B, La Marca A, Piomboni P, et al. Insulin-dependent diabetes in men is associated with hypothalamo-pituitary derangement and with impairment in semen quality. Hum. Reprod. 2002; 17: 2673-2677.
CrossRef - El Kader MAA, Gabr MM, Khater SM, Ghanem RA, El Naga AMA. Impact of insulin producing cells derived from adipose tissue mesenchymal stem cells on testicular dysfunction of diabetic rats. Heliyon. 2021; 7: e08316, https://doi.org/10.1016/j.heliyon.2021.e08316
CrossRef - Silva AAN, Oliveira JSD, Dias FCR, et al. The effectiveness of insulin therapy in the functional and morphological recovery of Leydig cells in rats submitted to diabetes mellitus. Biosci. J. 2023, 39, e39033. https://doi.org/10.14393/BJ-v39n0a2023-65096
CrossRef - Hamden K, Jaouadi B, Carreau S, et al. Potential protective effect on key steroidogenesis and metabolic enzymes and sperm abnormalities by fenugreek steroids in testis and epididymis of surviving diabetic rats. Arch. Physiol. Biochem. 2010; 116: 146-155.
CrossRef - Reddy KP, Narayana Rao M, Murthy JSR, Reddy PS. Lead aggravates the diabetic-induced reproductive toxicity in male Wistar rats. Toxicol. Res. 2016; 5: 1465-1476.
CrossRef - Adaramoye OA, Lawal SO. Effect of kolaviron, a biflavonoid complex from Garcinia kola seeds, on the antioxidant, hormonal, and spermatogenic indices of diabetic male rats. Andrologia. 2013; 46: 878-886.
CrossRef - Einstein JW, Rais MM, Mohd AM. Comparative evaluation of the antidiabetic effects of different parts of Cassia fistula Linn, a Southeast Asian plant. J. Chem. 2013; doi:10.1155/2013/714063.
CrossRef - Vasudevan K, Manoharan S, Panjamurthy K, Vellaichamy L, Chellammal A. Evaluation of antihyperglycemic effect of Cassia fistula (Linn.) leaves in streptozotocin-induced diabetic rats. Electron J. Pharmacol. Ther. 2008; 1: 57-60.
- Nirmala A, Eliza J, Rajalakshmi M, Priya E, Daisy P. Effect of hexane extract of Cassia fistula barks on blood glucose and lipid profile in streptozotocin diabetic rats. Int. J. Pharmacol. 2008; 4: 292-296.
CrossRef - Jeyanthi KA. Beneficial effect of Cassia fistula (L.) flower extract on antioxidant defense in streptozotocin-induced diabetic rats. Int. J. Pharm. Pharm. Sci. 2012; 4: 274-276.
- Daisy P, Balasubramanian K, Rajalakshmi M, Eliza J, Selvaraj J. Insulin mimetic impact of Catechin isolated from Cassia fistula on the glucose oxidation and molecular mechanisms of glucose uptake on Streptozotocin-induced diabetic Wistar rats. Phytomedicine. 2010;17: 28-36.
CrossRef - Chatterjee K, Ali KM, De D, et al. Hyperglycemia-induced alteration in reproductive profile and its amelioration by the polyherbal formulation MTEC (modified) in streptozotocin-induced diabetic albino rats. Biomark. Genom. Med. 2013; 5: 54-66.
CrossRef - Nwokike MO, Ghasi SI, Ogbonna AO, Anusiem CA. The effect of Imperata cylindrica root aqueous extracts on serum testosterone levels of hyperglycemic rats. J. Pharma. Pharma. Sci. 2020; 4: 187. doi: 10.29011/2574-7711.100087
CrossRef - Eliasson L, Renström E, Ämmälä C, et al. PKC-dependent stimulation of exocytosis by sulfonylureas in pancreatic beta cells. Science. 1996; 271: 813-815.
CrossRef - Ebokaiwe AP, Ijomone OM, Osawe SO, et al. Alteration in sperm characteristics, endocrine balance, and redox status in rats rendered diabetic by streptozotocin treatment: attenuating role of Loranthus micranthus. Redox Rep. 2018; 23: 194-205.
CrossRef - Luximon-Ramma, A, Bahorun T, Soobrattee MA, Aruoma OI. Antioxidant activities of phenolic, proanthocyanidin, and flavonoid components in extracts of Cassia fistula. J. Agric. Food Chem. 2002; 50: 5042-5047.
CrossRef - Manonmani G, Bhavapriya V, Kalpana S, Govindasamy S, Apparanantham T. Antioxidant activity of Cassia fistula (Linn.) flowers in alloxan-induced diabetic rats. J. Ethnopharmacol. 2005; 97: 39-42.
CrossRef - Pomjunya A, Ratthanophart J, Fungfuang W. Effects of Vernonia cinerea on reproductive performance in streptozotocin-induced diabetic rats. J. Vet. Med. Sci. 2017; 79: 572-578.
CrossRef - Song J, Gao X, Tang Z, et al. Protective effect of berberine on reproductive function and spermatogenesis in diabetic rats via inhibition of ROS/JAK2/NFκB pathway. Andrology. 2020; 8: 793-806.
CrossRef - Feng HL, Sandlow JI, Sparks AE, Sandra A, Zheng LJ. Decreased expression of the c-kit receptor is associated with increased apoptosis in subfertile human testes. Fertil. Steril. 1999; 71: 85-89.
CrossRef - Sharpe RM, Donachie K, Cooper I. Re-evaluation of the intratesticular level of testosterone required for quantitative maintenance of spermatogenesis in the rat. J. Endocrinol. 1998; 117: 19-26.
CrossRef - Rato L, Alves MG, Duarte AI, et al. Testosterone deficiency induced by progressive stages of diabetes mellitus impairs glucose metabolism and favors glycogenesis in mature rat Sertoli cells. Int. J. Biochem. Cell Biol. 2015; 66: 1-10.
CrossRef - Alves MG, Martins AD, Cavaco JE, Socorro S, Oliveira PF. Diabetes, insulin-mediated glucose metabolism, and Sertoli/blood-testis barrier function. Tissue Barriers. 2013; 1: e23992.
CrossRef - Koh PO. Streptozotocin-induced diabetes increases apoptosis through JNK phosphorylation and Bax activation in rat testes. J. Vet. Med. Sci. 2007; 69: 969-971.
CrossRef - Shrilatha B, Muralidhara. Early oxidative stress in testis and epididymal sperm in streptozotocin-induced diabetic mice: Its progression and genotoxic consequences. Reprod. Toxicol. 2007; 23: 578-587.
CrossRef - Mishra R, Nikam A, Hiwarkar J et al. Flavonoids as potential therapeutics in male reproductive disorders. Futur. J. Pharm. Sci. 2024; 10: 100. https://doi.org/10.1186/s43094-024-00677-3
CrossRef - Shokri A, Pourheydar B, Farjah GhH, Krimipour M, Pourheydar M. Effects of glibenclamide and troxerutin on the sperm parameters and histopathological changes of testis in streptozotocin-induced diabetic male rats: An experimental study. Int. J. Reprod. BioMed. 2023; 21: 123–138.
CrossRef - Hakim P, Abdullah Sani H, Mat Noor M. Effects of Gynura procumbens extract and glibenclamide on sperm quality and specific activity of testicular lactate dehydrogenase in streptozotocin-induced diabetic rats. Malays. J. Biochem. Mol. Biol. 2008; 16: 10-14.
Abbreviations
CAT: Catalase,
AA: Ascorbic Acid,
GSH: Glutathione,
ROS: Reactive Oxygen Species,
NC: Normal Control,
DC: Diabetic Control,
ST: Seminiferous tubule







