Manuscript accepted on :27-08-2025
Published online on: 22-09-2025
Plagiarism Check: Yes
Reviewed by: Dr. Soujania Singh
Second Review by: Dr. Adinarayana Andy
Final Approval by: Dr. Prabhishek Singh
Onobrudu, Divine Avwerosuoghene* , Acha, Joy Onyewonuwa, Orororo, Osuvwe Clement
, Acha, Joy Onyewonuwa, Orororo, Osuvwe Clement , Enudinisu, Gabriel Nduka
, Enudinisu, Gabriel Nduka , and Onyesom, Innocent
, and Onyesom, Innocent
Department of Medical Biochemistry, Faculty of Basic Medical Sciences, Delta State University, Abraka, Nigeria
Corresponding Author Email:daonobrudu@delsu.edu.ng
DOI : https://dx.doi.org/10.13005/bpj/3271
Abstract
Malaria chemotherapy, the mainstay of treatment is currently failing due to the emergence and spread of resistance of the causative parasites to conventional drugs endangering public health. The most severe complication of malaria is pulmonary involvement which occurs in 3-10% of the infection with about 70% mortality. So, discovery of antimalarial therapeutic agent with the capacity to combat associated lung complications is imperative. In Nigeria, herbs, like Ocimum gratissimum are used to treat malaria and associated fevers. Hence, this study evaluated the qualitative phytochemical constituents, toxicity, and antimalarial properties of O. gratissimum with its capacity to resolve the malarial-induced disturbances in lungs. The curative murine models and standard methods were adopted for the study. Results indicates that Plasmodium berghei (NK65) malarial parasite induced biochemical derangement in lungs of experimental adult male BALB/c mice, traceable to free radical-elicited lipid peroxidation of lung’s membrane, occasioned by the down regulation of the tissue’s Nuclear Factor Erythroid 2 Related Factor (Nrf2) as evidenced by its immunohistochemical expression. However, the ethanol extract of O. gratissimum (EEOg) was observed to be non-toxic with rich medicinal antimalarial phytochemicals capable of curing malaria and combating the associated lung complications by stimulating the expression of Nrf2 in lung tissue which enhanced the level of reduced glutathione (GSH) and activities of superoxide dismutase (SOD) and catalase antioxidants in the lungs of malarial infected mice treated with EEOg (125, 250, 500 mg/kg) and standard drug Artemether-lumefantrine (LDS). Our computational study identified eugenol as the most active compound with drug-likeness property which strongly inhibited Plasmodium falciparum lactate dehydrogenase (PfLDH), and modulated Keap1-Nrf2/p62 pathway. These properties indicate that eugenol is a potential antimalarial and antioxidant compound which could be optimized for enhanced performance as potent antimalarial agent, capable of resolving the associated lung impairments and oxidative stress.
Keywords
Antimalaria; Antioxidant; Eugenol; Lungs; Ocimum gratissimum
Download this article as:| Copy the following to cite this article: Avwerosuoghene O. D, Onyewonuwa A. J, Clement O. O, Nduka E. G, Innocent O. A Study on Toxicity and Antimalarial Efficacy of Ocimum Gratissimum, Influence on Lung Antioxidant Defense and Nrf2 Expression in Plasmodium Berghei (Nk65) Infected Mice. Biomed Pharmacol J 2025;18(3). |
| Copy the following to cite this URL: Avwerosuoghene O. D, Onyewonuwa A. J, Clement O. O, Nduka E. G, Innocent O. A Study on Toxicity and Antimalarial Efficacy of Ocimum Gratissimum, Influence on Lung Antioxidant Defense and Nrf2 Expression in Plasmodium Berghei (Nk65) Infected Mice. Biomed Pharmacol J 2025;18(3). Available from: https://bit.ly/4nG9PBt |
Introduction
The escalating worldwide prevalence of drug-resistant Plasmodium strains, particularly in endemic areas, is a critical concern to global health.1 Malaria infection induces oxidative stress, which is characterized by an imbalance in the host’s redox system as a result of excessive formation of reactive nitrogen and oxygen species (RNS-OS).2 RNS-OS play dual effect in the human system, serving as signaling molecules under physiological conditions, but inducing oxidative stress when produced uncontrollably. 3,4 The source of these oxidizing compounds includes the degradation of haemoglobin by the parasite and the host’s immune response, leading to respiratory burst and ischemia-reperfusion syndrome.5 This oxidative stress contributes to systemic and tissue damage, particularly in organs like the lungs and brain, with severe manifestations which include acute respiratory distress syndrome acute lung injury.6 Pulmonary alterations occur in 3-10% of the cases of P. falciparum malaria and represents the most severe complications of malaria with 70% lethality. 6
Consequently, the Nrf2 emerges as a crucial moderator of resistance to oxidative stress, with studies highlighting its role in cellular protection.7 In humans, Nrf2 is encrypted by nfe2l2 gene and it belongs to the cap ‘n’ collar family due to the basic leucine zipper in its structural composition.8 Initially identified as a homolog of the nuclear factor-erythroid 2 p45 (NF-E2) associated with β-globin gene expression.7, 9 Nrf2 was later found to bind to the antioxidant responsive element (ARE) in deoxyribonucleic acid, activating the expression of antioxidant enzymes and detoxifying enzymes (Phase II) which include cysteine ligase, glutamate NAD(P)H dehydrogenase (quinone 1) and haem oxygenase-1 (HO-1).10, 11
The ARE, also known as the electrophile response element, is a crucial cis-acting enhancer sequence located in the promoter section of most genes coding for phase II and antioxidant enzymes. It is involve in the expression of inducible gene which respond to oxidative stress and associated with low-level basal gene expression to antagonize RNS-OS formed during cellular respiratory processes.2, 13
Under basal conditions, Nrf2 associates with the Kelch-like ECH-associated protein 1 (Keap1), leading to Nrf2 ubiquitination and subsequent degradation through the proteasome.14 However, under cellular stress, this association is disrupted, preventing Nrf2 degradation and allowing its accumulation within the cell. This results in elevated translocation of Nrf2 into the nucleus, heterodimerization occurs with nuclear factors and then binds to AREs, thereby activating antioxidant gene transcription. The complex transcriptional machinery involved in this process includes co-activators like receptor-associated co-activator (RAC3) and co-regulators such as CREB binding protein.15, 16 The activation of antioxidant genes, then, lowers oxidative stress.
In murine study, Plasmodium berghei-mediated Nrf2 activation in infected hepatocytes enhanced parasite survival.17 However, it has been demonstrated that Nrf2 protects against cerebral malaria (CM) by inducing HO-1,18 but protects the spleen by inducing p62 expression which binds to Keap1 and increases free Nrf2 level.17 It has been observed that respiratory symptoms are common among African children with uncomplicated malaria (ECDC, 2017), and in adults, a frequency of 4-18% has been reported.18 Therefore, the role of Nrf2 in protecting the lungs during malarial infection is not clear. Hence, the need to search for inexpensive, more effective, easily accessible and safer bioactive compounds from natural products, particularly, herbal plants used in traditional treatment of malaria has become urgent and imperative.
Ocimum gratissimum, commonly known as clove basil or African basil is one of such medicinal plants.19 Ocimum gratissimum has been recognized for its diverse therapeutic properties, and its leaves have been traditionally employed for various medicinal purposes. Notably, plants extract exhibit antioxidant potential, attributed to their phytochemical constituents.19 Hence, this research aims to explore the in vivo antimalarial activity of Ocimum gratissimum and its relationship to the expression of lung’s Nrf2 and associated oxidative damage. The overall objective is to contribute valuable insights into the potential of Ocimum gratissimum as a natural resource for combating malaria and understanding its impact on oxidative stress and parasite survival in the lungs.
Materials and Methods
Plant collection and authentication: The plant (Ocimum gratissimum) was harvested fresh from their natural habitat in Abraka, Ethiope East Local Government Area of Delta State, Nigeria, in the month of May, 2023. It was identified and authenticated by Mr. O.E. Michael, Delta State University Herbarium, Abraka, Nigeria (Voucher Number, DELSUH 196).
Preparation of Ethanol Extract: The plucked fresh leaves were washed, air-dried at room temperature, pulverized, macerated in ethanol (1:8 w/v) for 72 h and filtered. The obtained filtrate was concentrated 45 °C with the aid of rotary evaporator and water bath, labelled and, refrigerated at 4oC.
Phytochemical Profiling
The phytochemical composition of the prepared EEOg was investigated using standard procedures and documented methods. Tannins, phlobatannins, saponins, steriods and terpeniods were screened following the procedures outlined by Ezeonu and Ejikeme et al.20 Furthermore, flavonoids, alkaloids and glycosides were estimated using the methods described by Sofowara,21 while, anthraquinones, phenols, and triterpenoids were identified according to the techniques stated by Sharma et al,22 Shah and Yadav,23 and Hasmi et al,24 respectively.
Ethical Approval: All procedures were carried out in accordance with the approved guidelines for animal use by the Research, Ethics and Grants Committee, Faculty of Basic Medical Sciences, Delta State University (RBC/FBMC/DELSU/25/794).
Oral Acute Toxicity
Oral acute toxicity was assessed in vivo through a single oral administration of the EEOg at a dose of 5,000 mg/kg to five (5) mice. Thereafter, the mice were monitored hourly for 3 hours and then, observed for 14 days in line with the protocol described by Pouaha.25
Malarial Curative Test
Thirty (30), adult male BALB/cmice were maintained under standard housing condition, fed with chow and water ad libitum and acclimatized for a week before the study was commenced.
Study Design: Twenty-five (25) mice were peritoneally inoculated with P. berghei (NK 65) malarial parasite infected blood containing about 1×107 parasites/mL. Inoculation success was confirmed 72 hours after, and the mice were grouped (n= 5) as follows:
Group 1 Mice (Naïve); were neither infected nor treated (NINT). Group 2 Mice (Sham); were infected, but not treated (INT). Group 3 Mice (Standard treatment); were infected and treated with 10 mg/kg b.wt of the standard ACT drug used for the treatment of malaria, containing; Artemether (80mg) and Lumefantrine (480mg). Groups 4-6 Mice; were infected and treated with 125, 250 and 500 mg/kg body weight of the ethanol extract of Ocimum gratissimum.
Animal euthanization and sample collection: Blood samples were collected from the cut tail tip at Day 0 (end of acclimatization and inoculation), Day 3 (confirmation of parasitaemia and commencement of the 4-day treatment), Day 6 (end of treatment), Day 9 (third day post treatment) and Day 12 (sixth day post treatment), for determination of malarial parasite.
After the Day 12, the mice were euthanized by cervical decapitation and blood samples were collected by ocular puncture using capillary tube and then, placed in plain, sterile tubes and centrifuged (10,000 xg for 10 mins at room temperature) to obtain serum for lung function (bicarbonate, glucose and lactate) assays. Laparotomy was carried out to expose the internal organs and lungs were excised and prepared for parasite count, tissue antioxidant and immunohistochemical evaluations.
Parasitemia Assessment
Thin smears were made on a microscope slide from the collected blood samples, smears were fixed with methanol, stained with field stain A and B, and viewed microscopically using a X 100 objective lens to determine the parasite density.26
Collection of bronchoalveolar lavage fluid (BALF)
Tracheostomy was performed, cannula was inserted into the trachea, 2 mL of sterile saline was instilled into the lungs and the BALF was collected and centrifuged (500 x g for 10 min) to remove debris and cells.27
Estimation of lung function markers
Markers of lung function (bicarbonate, glucose, and lactate) in both serum and BALF were determined using kits (Sigma-Aldrich) according to the manual instructions.
Determination of lung tissue antioxidants and marker of oxidative stress
The collected lungs were washed in an ice cold 1.15% potassium chloride solution, homogenized with 0.1M phosphate buffer (pH 7.2), centrifuged at 2500 rpm for 15 min. and the supernatant was decanted. The resultant was analysed for antioxidants and oxidative stress marker.
Lung Antioxidant Assay
The activities of superoxide dismutase (SOD), catalase (CAT) and reduced glutathione (GSH) in the lung tissue homogenate were estimated spectrophotometrically in line with the method of Misra and Fredorich, 28 Cohen et al 29 and Ellman, 30 respectively. The breakdown product of lipid peroxidation, malondialdehyde (MDA) was evaluated in line with the method of Gutteridge and Wilkins.31
Immunohistochemical staining of lung tissue for Nrf2 expression
Method of Kansanen et al 32 was adopted for the staining of formalin-fixed and paraffin-embedded (FFPE) lung tissue sections (4-6 μm thickness. The stained sections were developed and counterstained with haemotoxylin and mounted with permount mounting medium. Nrf2 staining intensity and distribution in the lung tissue sections were evaluated using light microscope.
Data Analysis
Results were presented as Mean ± Standard Error of Mean (SEM) for n=5 mice per group. Data were subjected to ANOVA using GraphPad prism (v. 10.4.0; 2024), and p<0.05 was taken as statistically significant.
Results
Qualitative Phytochemistry of Ocimum gratissimum Ethanol Leaf Extract
The findings from the qualitative phytochemical evaluation of Ocimum gratissimum ethanol extract (EEOg) are shown in Table 1.
Table 1: Qualitative phytochemical contents of Ocimum gratissimum ethanol leaf extract
| Phytochemical | Inference |
| Saponins | + |
| Cardiac glycoside | +++ |
| Flavonoids | ++ |
| Tannins | + |
| Terpenoids | +++ |
| Alkaloids | +++ |
| Phenols | +++ |
+ Low, ++ Moderate, +++ High
The results (Table 1) indicate high amounts of cardiac glycosides, triterpenoids, phenols and alkaloids, but moderate level of flavonoids with low saponin content.
Toxicity Assessment
The oral acute toxicity of EEOg was evaluated to determine the safety limits for selection of appropriate dosing regimens for the murine study. During the assessment, no mortality was recorded within the first day and the tracked duration of 14 days following the administration of 5,000 mg/kg body weight of the mice. Also, the animals did not display any outward sign of abnormalities like posture, diarrhoea, lacrymation, movement, salivation, tremors, loss of appetite, and depression. These observations showed that the LD50 of EEOg is ≥ 5,000 mg/kg; indicating non-toxicity. Therefore, 2.5% (125 mg/kg), 5% (250 mg/kg), and 10% (500 mg/kg) doses of EEOg were selected for the curative study using murine model.
Parasite levels, suppression and mean survival time of infected mice treatment with EEOg
Changes in parasite levels (Figure 1a) and parasite suppression (Figure 1b) in both blood and lungs induced by the treatment with EEOg, and associated mean survival time, MST, (Figure 1c) as shown as indicated.
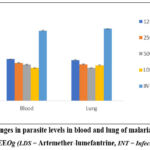 |
Figure 1a: Changes in parasite levels in blood and lung of malarial-infected mice treated with EEOg (LDS = Artemether-lumefantrine, INT = Infected, not treated). |
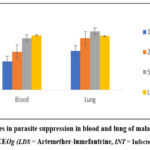 |
Figure 1b: Changes in parasite suppression in blood and lung of malarial-infected mice treated with EEOg (LDS = Artemether-lumefantrine, INT = Infected, not treated) |
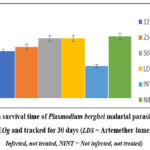 |
Figure 1c: Mean survival time of Plasmodium berghei malarial parasite infected mice treated with EEOg and tracked for 30 days (LDS = Artemether-lumefantrine, INT = Infected, not treated, NINT = Not infected, not treated) |
Expression of Nrf2 in lungs of infected mice
Next, we examined the impact of parasite survival in the lung on the immunohistochemical expression of Nrf2 (Figure 2) and histological grading (Table 2) of the infected lungs.
Figure 2 shows the staining pattern in lungs of malarial infected mice treated with Ocimum gratissimum varying doses (125, 250, 500 mg/kg) and controls.
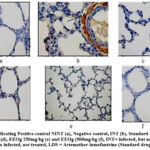 |
Figure 2. (a-f) indicating Positive control NINT (a), Negative control, INT (b), Standard Control, LDS (c), EEOg 125mg/kg (d), EEOg 250mg/kg (e) and EEOg (500mg/kg (f), INT= infected, but not treated, NINT: No infected, not treated, LDS = Artemether-lumefantrine (Standard drug). |
The INT group showed strong staining indicating consistent expressions of Nrf2 but those of the other groups were low. This expression induced inflammation in infected lungs (Table 2) and modified parasite sequestration receptors (Figure 3 a-d) which altogether promoted parasite survival, growth, and infectivity (Table 1).
Histological grading
The histological grading of the lung tissue obtained from the malarial parasite infected mice, with and without treatment, including the controls are shown in Table 2. Also, data for the sequestration receptors are indicated in Figure 3a-d
Table 2: Histological grading of lung tissue after staining with haematoxylin
| Grading | NINT | LDS | INT | EEOg (mg/kg) | ||
| 125 | 250 | 500 | ||||
| Peribronchal inflammation | 0 | 1 | 2 | 1 | 0 | 0 |
| Perivascular inflammation | 0 | 1 | 3 | 1 | 1 | 1 |
| Total lung inflammation | 0/6 | 2/6 | 5/6 | 2/6 | 1/6 | 1/6 |
Peribronchial inflammation for NINT reveals no detectable inflammation, LDS shows occasional cuffing with inflammatory cells. However, INT indicates that most bronchi or vessels were surrounded by a thin layer (1-5 cells thick) of inflammatory cells, but 125 mg/kg like the LDS showed occasional cuffing with inflammatory cells, and 250 mg/kg like the NINT, had no detectable inflammation. So all so, with the 500 mg/kg group.
Sequestration receptors
Levels of parasite sequestration receptors (eosinophils, lymphocytes, macrophages, and neutrophils) in lungs and BALF of malarial-infected mice, treated with EEOg are shown in Figure 3 a-d.
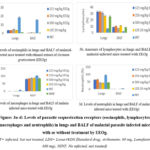 |
Figure 3a-d: Levels of parasite sequestration receptors (eosinophils, lymphocytes, macrophages and neutrophils) in lungs and BALF of malarial parasite infected mice, with or without treatment by EEOg. |
(INT= infected, but not treated, LDS= Lonart®DS (Standard drug; Arthemeter, 80 mg, Lumefantrine 480 mg), NINT: No infected, not treated)
Malarial parasite infection in experimental mice induced sequestration of parasites via increase in receptors in lung tissue, hence, the increased level in parasitized mice without treatment (INT), indicating favourable reception, evasion of host immune surveillance and remodelling of the host environment for survival in the lungs (Figure 1a), and this significantly reduced MST (Figure 1c).
Infection of experimental mice with P. berghei (NK 65) caused increase amounts of parasite sequestration receptor in infected–lungs and BALF (Figure 3 a-d) hence the elevated load of parasites in the tissue (Figure 1). Such sequestion induced inflammation (Table 2) and oxidative stress (Figure 4 a-d).
Changes in lung and BALF oxidative stress marker induced by the treatment of malarial infected mice with ethanol leaf extract of Ocimum gratissimum
The changes in lung and BALF oxidative stress marker induced by the treatment of malarial infected mice with ethanol leaf extract of Ocimum gratissimum are shown in Figure 4a-d.
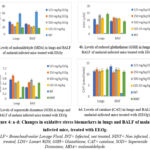 |
Figure 4 a-d: Changes in oxidative stress biomarkers in lungs and BALF of malarial infected mice, treated with EEOg. |
BALF= Bronchoalveolar Lavage Fluid, INT= Infected, not treated, NINT= Non infected, Not treated, LDS= Lonart ®DS, GSH= Glutathione, CAT= catalase, SOD= Superoxide Dismutase, MDA= malonaldehyde.
MDA level for the treated groups decreased significantly (p<0.05) when compared with INT. GSH, SOD and catalase for the treated groups increased significantly (p<0.05) when compared with INT. This indicate that EEOg treatment enhanced antioxidant molecules and thus, reduce oxidative stress in lungs and BALF of malarial infected mice.
Parasite sequestration in lungs induced inflammation and oxidative stress which affected lung functions (Figure 5 a-c).
Evaluation of biochemical function in lung and BALF of malarial infected mice treated with or without EEOg
Malarial infection in experimental mice induced oxidative stress in lungs and BALF by hampering the synthesis of antioxidant molecules, and the effect of such stress on biochemical markers (bicarbonate, glucose and lactate) of lung function were estimated and results are presented in Figure 5a-c
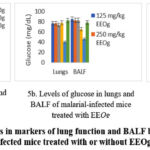 |
Figure 5a-c: Changes in markers of lung function and BALF balance in malarial-infected mice treated with or without EEOg. |
In the BALF, malarial infection significantly (p<0.05) reduced glucose content, but increased (p<0.05) bicarbonate and lactate levels. Similar pattern was observed in the lung. Malarial-infection disturbs biochemical function, possibly due to the increased oxidative stress (Figure 4) and inflammation (Table 2).
Effect of treatment of malarial infection with EEOg in mice
The treatment of P. berghei (NK65) parasite induced malaria in experimental mice showed good anti-malarial activity (Figure 1b) with the additional capacity to significantly (p<0.05) decrease parasite sequestration receptors in lungs (Figure 3), which in turn, reduced (p<0.05) parasite load in the tissue (Figure 1a). The significant reduction in lungs’s parasite level halted tissue (lung) inflammation (Table 2), improved antioxidant capacity (Figure 4) and restored cellular biochemical function (Figure 5) which enhanced the mean survival time (MST) of the treated when tracked for 30days (Figure 1c). These observations compared well with the standard ACT treatment (Lonart®DS).
Drug Property Test
To evaluate the drug properties of the already identified eighteen (18) compounds in EEOg [33], their drug likeness scores were computationally determined in order to select the compounds with likelihood of being developed into drugs. The drug properties obtained for two prioritized compounds (eugenol and carvacrol are presented in Table 3.
Table 3: Drug likeness scores for the two prioritized compounds
| Scores | Prioritized compounds | |
| Eugenol | Carvacrol | |
| Lipinski’s Rule | ||
| · Molecular weight (g/mol) | 164.21 | 150.22 |
| · LogP | 2.73 | 3.38 |
| · HBD | 1 | 1 |
| · HBA | 2 | 1 |
| · TPSA | 2 | 1 |
| · Rotable bonds | 4 | 3 |
| · Molar refractivity | 55.11 | 48.41 |
| · LogD | 2.53 (pH 7.4) | 3.18 (pH 7.4) |
| Pharmacokinetic properties | ||
| · Solubility (water) (mg/mL) | 1.9 | 0.12 |
| · Permeability (HIA) (%) | 85.14 | 92.11 |
| · Bioavailability (%) | 65-80 | 70-90 |
| · Volume of distribution Vd (L/kg) | 1.23 | 1.54 |
| · Clearance (ml/min/kg) | 1.15 | 0.81 |
| · Half-life (hours) | 2.5 | 3.5 |
| Pharmacodynamic properties | ||
| · Target | COX-2, Receptor (TRPV1) | COX-2, Receptor (TRPV1) |
| · Mechanism | Antioxidant, anti-inflammatory, analgesic | Antimicrobial, antioxidant, anti-inflammatory |
| · Binding affinity, Ki (µM) | 10-50 | 5-20 |
| Toxicity and Safety | ||
| · LD50 (oral) (g/kg) rat | 2.65 | 3.4 |
| · LD50 (dermal) (g/kg) rabbit | >2 | >2 |
| · Genotoxicity | Non-genotoxic | Non-genotoxic |
| · Carcinogenicity | Non-carcinogenic | Non-carcinogenic |
| Drug-likeness scores | ||
| · DL-Score | 0.43 (moderate) | 0.53 (good) |
| · QED | 0.61 (good) | 0.73 (excellent) |
| · Drug-likeness probability | 0.71 | 0.85 |
Following the drug properties analysis, two compounds (eugenol and carvacrol) were prioritized and so, selected based on their profile ranking (Table 3). Eugenol possessed moderate solubility, clearance, lipophilicity and polar surface area, but good oral bioavailability with analgesic, antioxidant and anti-inflammatory properties, and low toxicity.
Carvacrol has moderate lipophilicity, polar surface area, and clearance, but good oral bioavailability and poor solubility. It possesses antimicrobial, antioxidant and anti-inflammatory characteristics with low toxicity. Eugenol and carvacrol, obtained from EEOg demonstrated promising drug-likeness properties, and analysis indicate potential as therapeutic agents in the treatment of especially inflammatory and infectious diseases.
EEOg has been shown to possess good antimalarial activity and two compounds (eugenol and carvacrol) have been identified with drug-likeness properties with potential for combating infectious diseases and associated inflammatory and oxidative stress complications. Following, therefore, the two compounds were dock against Plasmodium falciparum protein targets, important for parasite survival and perpetuation of the malarial infection and results (Table 4) showed that eugenol is topmost and inhibited Pf LDH (Plasmodium falciparum lactate dehydrogenase) the more.
Table 4: Dock scores for eugenol against important parasite (P. falciparium) targets (Pf LDH)
| Targets | PDB 1D | Docking score (kCal/mol) | XP Gscore (kCal/mol) | MMGBSA dG Bind (kCal/mol) |
| PfLDH | 1TQJ | -3.962 | -3.963 | -38.900 |
Results (Table 4) indicates that eugenol showed high binding affinities for PfLDH; suggesting interruptions of P. faciparium energy metabolism
In all, PfLDH seems to be the primary target for eugenol. So, the post dock binding analysis, and bond interactions between eugenol and PfLDH are shown in Figures 6 and 7, respectively.
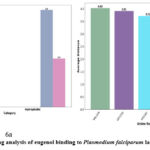 |
Figure 6: Post docking analysis of eugenol binding to Plasmodium falciparum lactate dehydrogenase. |
This analysis presents insights into the binding interactions between eugenol and Plasmodium falciparum lactate dehydrogenase (PfLDH). The first bar chart (6a) categorizes the types of bonds involved in the binding interaction, showing both hydrogen bonds and hydrophobic interactions. Two hydrogen bonds were identified: one conventional hydrogen bond and one carbon hydrogen bond, each with a count of 1. These hydrogen bonds contribute to the stability and specificity of eugenol’s binding with PfLDH. Hydrophobic interactions are also significant, with one alkyl and one π-alkyl interaction, indicating the role of nonpolar interactions in stabilizing eugenol within the enzyme’s binding pocket.
The second bar chart (6b) displays the average distances between eugenol and specific amino acid residues in the PfLDH binding site. The residues VAL129 and LEU150 show the longest interaction distances (4.03 Å and 3.91 Å, respectively), while SER234 has the shortest interaction distance at 1.73 Å, suggesting it may play a key role in the binding affinity due to closer proximity to eugenol. Other notable residues include HIS182 and THR83, with distances of 3.72 Å and 2.93 Å, respectively. These distances indicate that various amino acids contribute differently to the binding, with closer residues potentially having stronger interactions with eugenol.
In summary, both hydrogen bonding and hydrophobic interactions are essential in the binding of eugenol to PfLDH, with specific amino acid residues at varying distances playing crucial roles in stabilizing the complex. The presence of close hydrogen bond interactions (e.g., with SER234) and multiple hydrophobic contacts supports the potential effectiveness of eugenol as an inhibitor of PfLDH.
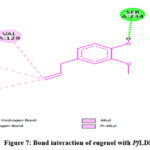 |
Figure 7: Bond interaction of eugenol with PfLDH |
These bond interactions reduce PfILDH activity which disturbs glycolysis and decrease the ability of P. faciparium to produce energy and defend oxidative stress.
Alongside malaria, P. faciparium induces oxidative stress in the host. To investigate the ability of eugenol to combat this malarial associated complications, molecular docking was conducted with host’s vital targets (Keap1-Nrf2 pathway) and results are shown in Table 5.
Table 5: Molecular docking scores for eugenol against Keap1
| Targets | PDB 1D | Docking score (kCal/mol) | XP Gscore (kCal/mol) | MMGBSA dg Bind (kCal/mol) |
| Keap1 | 4CXI | -7.784 | -7.784 | -35.220 |
Keap1= Kelch-like ECH-associated protein 1, inhibition of Keap1-p62 by eugenol =75.2%, Nrf2 activation by eugenol=82.1%, Inhibition of Keap1-Nrf2 interactions by eugenol =85.6%
Date (Table 5) reveal that eugenol strongly inhibits Keap1 and its association with Nrf2 and p62, but activates Nrf2. These evidences indicate that eugenol is a potent antioxidant.
 |
Figure 8: Post docking analysis of eugenol binding to Keap1 [Kelch-like ECH-associated protein 1 [AF-Q14145] |
The analysis identifies several key interactions between the ligand and specific amino acid residues, revealing a mix of hydrogen bonding and hydrophobic interactions (Figure 8a). ALA536 forms a conventional hydrogen bond with the ligand, although with a relatively large average distance of 4.72 Å, indicating a weaker or spatially distant interaction. GLY605 and VAL418 both engage in carbon hydrogen bonds, with average distances of 2.82 Å and 2.73 Å, respectively, suggesting stronger, closer interactions than ALA536. Also, GLY367 and VAL606 exhibit close contact with the ligand, with distances of 2.29 Å and 2.25 Å, respectively, further stabilizing the interaction network. Lastly, a single R-Alkyl hydrophobic interaction is observed, adding stability to the overall binding complex. Together, these interactions underscore a binding profile dominated by hydrogen bonds, supported by hydrophobic contacts, which collectively enhance the ligand’s binding affinity (Figure 8b and 9).
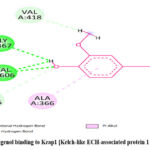 |
Figure 9: Eugenol binding to Keap1 [Kelch-like ECH-associated protein 1 [AF-Q14145] |
Evidence from the computational data suggest that eugenol, the most abundant compound in Ocimum gratissimum possesses drug likeliness, antimalarial and antioxidant properties, potentially the capable of combating malaria and associated inflammatory and oxidative stress in the lungs.
Discussion
Malaria is a hemoparasitic infection, caused by the Plasmoidum sp and transmitted by an infected female Anopheles mosquito, leading to severe life-threatening disease, ranging from mild to severe clinical forms depending on host’s infection history, immune status, parasite virulence, and genetic variations in both host and parasite.1,3,32,
Respiratory disturbances have been observed in uncomplicated malaria, and are common among African children (ECDC, 2019). However, in adults, respiratory involvement has also been reported in uncomplicated P. faciparium malaria with a frequency of 4-18%, and such pulmonary alterations include: impaired ventilation, increased pulmonary phagocytic activity, obstruction of airflow and reduced gas transfer18. But, in severe and complicated malaria, multi-organ dysfunctions occur and these include: acute lung injury, ALI, and acute respiratory distress syndrome (ARDS) which contribute to patient’s high mortality.
In our present experimental murine study, we observed that P. berghei malarial parasite infection in mice increased the expression of Nrf2 in lungs of the infected mice and this resulted in parasite sequestration and induction of oxidative stress and inflammation in mice (host) pulmonary tissues. Malaria parasite reside within a vacuole created during invasion which act as the main interface between the host and the parasites. These parasites coordinate several activities (uptake of host cell cytosol, nutrient import, effector protein export, and waste efflux) to subvert host cell and support their own metabolism, survival and virulence.5 Evidence for the involvement of Nrf2-signalling pathway leading to Plasmodium parasite survival has been provided.36
During quiescent conditions, Nrf2 is negatively regulated by Kelch-like ECH associated protein 1 (Keap 1), a redox-sensitive E3 ubiquitin ligase substrate adaptor.37 Keap 1 mediates the ubiquitination of Nrf2 via E3 ubiquitin ligase Cullin3 (CUL3) by binding to it, and this elicits the degradation of Nrf2 by the ubiquitin proteasome pathway.38, 39 However, in response to oxidative stress, the oxidation of cysteine residues in Keap1 causes a change in conformation and that releases Nrf2 from Keap1 which protects it from degradation.40, 41 Consequently, the free Nrf2 is translocated to the nucleus where it heterodimerizes with the Maf protein family, inducing transcription of its target genes. Besides, the stability of Nrf2 can be determined by p62/sequestosome-1 (p62) which functions in Nrf2 regulation via interaction with Nrf2 binding site of Keap1 and hence, competitive inhibition of ther Keap1-Nrf2 binding which leads to Nrf2 stability and then, nuclear translocation.42, 43 Phosphorylation of p62 at serine 349 increases its binding with Keap1, freeing Nrf2 whose activation supports Plasmodium faciparium survival during its liver stage.44
Nrf2 inhibits ferroptosis (accumulation of lipid peroxidation products and ROS from iron metabolism) by regulating the expression of SLC7a11.45 This controls intracellular oxidation homeostasis of the host cell which favours parasite survival.43, 46, 47 It has been demonstrated that the high oxidative stress in brain of experimental cerebral malaria induced Nrf2 activiation 47 which in turn stimulated heme oxygenase-1 (Hmox1) that enhanced the breakdown of heme and reduction of ROS, leading to parasite survival and perpetuation of infection in both brain [48] and liver.49 Upregulation of genes controlled by Nrf2; including p62, was seen in all infected cells; indicating the vital role of this transcription factor for parasite persistence and survival.50
Essentially, Nrf2 activation that promotes parasite survival and infectivity seems not be restricted to the liver and brain cells, our study shows that sequestrated pulmonary parasites survived and induced Nrf2 expression, high oxidative stress and inflammation with accompanying increase in sequestration receptors. It seems that the Keap1-Nrf2 pathway plays critical role in protecting against parasites’ oxidative stress and promoting their survival in lungs of P. berghei infected mice. Activation of the pathway appears to enable the parasites to neutralize harmful ROS and maintain cellular homeostasis.35 Also Nrf2, a key transcription factor, regulates the expression of antioxidants and detoxification genes that provide protection for parasite survival. The Keap1-Nrf2 pathway, in addition, influences host immune response, modulating pro-inflammatory cytokines and chemokines productions which creates a favourable environment for parasite growth and survival, enabling parasites’ evasion of host immune system.35 Furthermore, the pathway regulates biochemical processes (glucose metabolism, amino acid biosynthesis, and lipid synthesis) important for parasite survival and growth in the host. These speculations have nonetheless provided valuable insights regarding the role of Keap1-Nrf2 pathway in P. faciparium survival in lungs of malarial infected mice.
Albeit, treatment of the malarial infection with EEOg, a non-toxic extract (LD50 ≥ 5,000 mg/kg) containing important medicinal phytochemicals (Table 1) reduced parasitemia (Figure 1), lungs’ parasite load (Figure 1), oxidative stress (Figure 4), inflammation (Table 2), sequestration receptors (Figure 3) and Nrf2 expression (Figure 2), but improved lung function (Figure 5) in manners that compared well with the standard treatment and the naïve mice.
The in silico evaluation of compounds in EEOg identified eugenol and carvacrol as the topmost bioactive compounds with drug properties (Table 3). However, docking analysis with protein targets (PfLDH, PfDHFR, Pf SUB1, and Pf ASL) important for parasite growth, survival and infectivity (Table 4) indicated that eugenol interacted the more with PfLDH with both hydrogen bonding and hydrophobic interaction (Figure 6). It bonded closely and strongly with SER234 of PfLDH via hydrogen bonding, and the other multiple bond interactions contribute to the stability of the binding complex (Figure 7), supporting the potential effectiveness of eugenol as a strong inhibitor of PfLDH which is expressed in all stages of the parasite life cycle. PfLDH aids the parasites production of lactate from pyruvate, and this generates energy for the parasite required for multiplication, growth and survival. Also, it yields NAD+ from NADH which maintains parasites’ redox balance. Therefore, the eugenol in EEOg may have inhibited PfLDH, affecting parasites’ energy supply and ability to maintain redox homeostasis which hamper survival, hence, the reduction of parasitaemia and sequestration in pulmonary tissues. Other natural compounds (quercetin, apigenin, curcumin, and berberine) that inhibit PfLDH have been demonstrated to possess antiplasmodial activity and our study aligns with theirs. 51
In order to investigate the roles of eugenol on the Keap1-Nrf2 pathway, docking was performed and we discovered that eugenol strongly inhibited Keap1 via a mix of hydrogen bonding and hydrophobic interactions (Figure 8) and such binding particularly with Gly 367 and Val 606 inhibited Keap1’s association with Nrf2 (85.6%) and p62 (75.2%) (Table 5). This frees Nrf2 which is then activated to increase the production of antioxidants (Figure 8) improved oxidative stress defence in lungs of infected mice (Figure 4). So, eugenol binds to Keap1, distorting its association with p62 known to favour parasites’ survival. Eugenol’s binding with Keap1 prevents the association with p62, the parasites’ machinery employed to maintain redox balance and evade host’s immune system, 36 and stimulation of the released (free) Nrf2 to favour host’s redox maintenance and hence, parasites’ inability to survive due to oxidative stress and immune capture. The study of Annina et al. 36 has also shown that Nrf2 activation is driven by p62 recruitment of Keap1 into PVM (parasitophorous vacuole membrane) of P. berghei parasites, and this activation was demonstrated to be essential for optimal parasite development, suggesting it to be another example of how parasites evade signalling for survival.
Conclusion
EEOg is a non-toxic, culminary plant, containing several important classes of phytochemicals, and our study has identified its eugenol compound as the most active antiplasmodial agent which strongly inhibits PfLDH and modulates the Keap1-Nrf2/p62 pathway. These properties regulate host’s redox system and induce oxidative stress in invading parasites which hampers lung sequestration. Therefore, eugenol can be optimized for further study as an antiplasmodial and antioxidant compounds, capable of resolving malarial associated lung symptoms, the commonest complication of malaria.
Acknowledgment
We wish to acknowledge the contrributions of the laboratoy staff members of the Department of Medical Biochemistry, Delta state University, Abraka, and the technical assistance of the CEO and Management of Affemefune Biomedical Consullt, Abraka, Delta State, Nigeria.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
An Ethical approval with the number RBC/FBMC/DELSU/25/794 was obtained from the Research, Ethics and Grant Committee, Faculty of Basic Medical Sciences, Delta State University, Abraka, Nigeria. The study was conduct in line with guidelines provided.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Authors’ Contribution
Onyesom I: Conceptualization and supervision.
Onobrudu DA: Writing, review and editing
Acha JO: Data collection
Orororo OC: Methodology and data analysis
Enudinisu GN: Laboratory investigations and data collection
References
- World Health Organization (WHO). World Malaria Report 2023. Geneva https://www.who.int. 2024 [Accessed 30 May 2025].
- Sawadogo H, Soulama I, Zida A, et al. Plasmodium falciparum Genetic Diversity and Resistance Genotype Profile in Infected Placental Samples Collected after Delivery in Ouagadougou. Drug Resist. 2023; 16:6673–6680.
CrossRef - Opajobi AO, Onobrudu DA, Ojugbeli ET, et al. Levels of Cerebral Malaria Indices, Free radical Scavengers and Preventive Antioxidants in Plasmodium berghei Infected Mice Treated with Phytochemical Extracts of Phyllanthus amarus Schum and Thonn. 2022;20(6):374-390
- Onobrudu DA, Ikewuchi JC and Onyeike EN. Moderation of ocular markers of oxidative stress of selenite-induced cataractous pups by Boswellia dalzielii hutch extract. Global Adv. Res. J. Med. Plant. 2016;4(2):007-011)
CrossRef - Percário S, Moreira DR, Gomes BA, et al. Oxidative stress in malaria. Int J. Mol. Sci. 2012;13:16346-16372
CrossRef - Marsh K, Forster D and Waruiru C. Indicators of life-threatening malaria in African children. New England J. Med. 1995;332:1399–1404
CrossRef - Moi P. Isolation of NF-E2-related factor 2 (Nrf2), a NF-E2-like basic leucine zipper transcriptional activator that binds to the tandem NF-E2/AP1 repeat of the beta-globin locus control region. Nat. Acad. Sci. USA. 1994;91(21):9926-9930
CrossRef - Silva-Islas CA and Maldonado PD. Canonical and non-canonical mechanisms of Nrf2 activation. Res.2018;134:92-99
CrossRef - Silva JC, Pereira RLS, Sampaio de Freitas, T, et al. Keap1 represses nuclear activation of antioxidant responsive elements by Nrf2 through binding to the amino-terminal Neh2 domain. Genes Dev. 1999;13(1):76–86
CrossRef - El-Mahrouk SR, El-Ghiaty MA and El-Kadi AOS. The role of nuclear factor erythroid 2-related factor 2 (NRF2) in arsenic toxicity. Environ. Sci. 2024;150:632-644
CrossRef - Tripathi W, Kharkwal G, Mishra R and Singh G. Nuclear factor erythroid 2-related factor 2 (Nrf2) signaling in heavy metals-induced oxidative stress. Heliyon. 2024;10(18):e37545
CrossRef - Tanigawa S, Fujii M, and Hou DX. Action of Nrf2 and Keap1 in ARE-mediated NQO1 expression by quercetin. Free Rad. Biol. Med. 2007;42(11):1690-1703
CrossRef - Rushmore TH, Morton MR and Pickett CB. The antioxidant responsive element Activation by oxidative stress and identification of the DNA consensus sequence required for functional activity. Biol. Chem.1991;266:11632–11639
CrossRef - Dinkova-Kostova AT, Kostov RV, and Canning P. Keap1, the cysteine-based mammalian intracellular sensor for electrophiles and oxidants. Biochem. Biophy. 2017;617:84-93
CrossRef - Tong KI. Keap1 recruits Neh2 through binding to ETGE and DLG motifs: characterization of the two-site molecular recognition model Cell Biol. 2006;26(8):2887-2900
CrossRef - Zhang J, Zhang G, Zeng M, et al. Nuclear Factor Erythroid 2 and Treatment of Metabolic Syndrome, Redox Signal. 2024;(10-12):744-768
CrossRef - Bindschedler A, Schmuckli-Maurer J, Wacker R, et al. Plasmodium berghei-Mediated NRF2 Activation in Infected Hepatocytes Enhances Parasite Survival. Cell Microbiol. 2022;1:7647976
CrossRef - Boccolini D, Menegon, M and Di Luca, M. Non-imported malaria in Italy: Paradigmatic approaches and public health implications following an unusual cluster of cases in 2017. BMC Public Health. 2020;20:857
CrossRef - Silva JC, Pereira RLS, Sampaio de Freitas T, et al. Rodrigues FFG, Santos GJG. Evaluation of antibacterial and toxicological activities of essential oil of Ocimum gratissimum L and its major constituent eugenol. Food Sci. 2022;50:e102128
CrossRef - Ezeonu CS and Ejikeme, CM. Qualitative and quantitative determination of phytochemical contents of indigenous Nigerian softwoods. New J. Sci. 2016;5(1):633-640
CrossRef - Sofowora EA. Medicinal plant and traditional medicine in Africa University of Ife Press, Ile –Ife:1994;20-23
- Sharma AK, Anupam K, Swaroop V, Lal PS and Bist V. Pilot scale soda-anthaquinone pulping of palm oil empty fruit bunches and elemental chlorine free bleaching of resulting pulp. Cleaner Prod. 2015;106:422-429
CrossRef - Shah RK and Yadav RNS. Qualitative phytochemical analysis and estimation of total phenols and flavonoids in leaf extract of Sarcochlamys pulcherrima. Global J. Bio-Sci. Biotechnol. 2015;4(1):81-84
- Hashmi HF, Bibi S, Anwar M and Khan MR. Qualitative and quantitative analysis of phytochemicals in Lepidium pinnatifidum Int. J. Tradit. Complement. Med. 2021;4(5):67-75
- Pouaha CLC, Lienou LL, Dongmo FFD, et al. Evaluation of acute and sub-acute oral toxicities of Momordica foetida Schumach (Cucurbitaceae) leaves methanol extract in Wistar rats. Biosci. Med. 2023;11:123-142
CrossRef - Adu-Gyasi D, Adams M, Amoako S, et al. Estimating malaria parasite density: assumed white blood cell count of 10,000/μl of blood is appropriate measure in Central Ghana. J. 2012;11(1)
CrossRef - Cheng C, Ho WE, Goh FY, et al. Anti-malarial drug artesunate attenuates experimental allergic asthma via inhibition of the phosphoinositide 3-kinase/Akt pathway. PLoS One. 2011;6:e20932
CrossRef - Misra, HP and Fridovrich, I. The role of superoxide anion in autoxidation of epinephrine and a simple assay for superoxide dismutase. Biol. Chem. 1972;247:3170-3175
CrossRef - Cohen G, Dembiec D, and Marcus J. Measurement of catalase activity in tissue extracts. Biochem. 1970;34:30–38
CrossRef - Ellman GL. Tissue sulfhydryl groups. Biochem. Biophy. 1959;82:70-77
CrossRef - Gutterridge JMC and Wilkins C. Copper dependent hydroxyl radical damage to ascorbic acid Formation of thiobarbituric acid reactive product. Letters. 1982;137:327-340
CrossRef - Akoniyon OP, Akiibinu M, Adeleke MA, et al. Comparative study of genetic diversity and multiplicity of infection in uncomplicated Plasmodium falciparum infections in selected regions of pre-elimination and high transmission settings usingmsp1 and msp2 genes. 2024;13:e172
CrossRef - Onobrudu DA, Toloyai PE, Awhin EP, Ibobo GO, Dunkwu CC, Onyesom I. Genetic structure and diversity of Plasmodium falciparum merozoite surface proteins 1 and 2 among children below 5 years in Delta state, Nigeria. Biotechnol. Res. J. 2025;9:152-157.
CrossRef - Graham H, Bakare AA and Ayede AI. Diagnosis of pneumonia and malaria in Nigerian hospitals: A prospective cohort study. Pulmonol. 2020;55: 37–50
CrossRef - Beck JR and Ho CM, Transport mechanism at the malaria parasite host cell interface. PLoS Pathogens. 2021;17(14): 1009394
CrossRef - Annina B, Schmuckli-Maurer J, Rahel W, et al. Plasmodium berghei-mediated NRF2 activation in infected hepatocytes enhances parasite survival. Cell Biol. 2022
CrossRef - Itoh K. An Nrf2/small Maf heterodimer mediates the induction 991 of phase II detoxifying enzyme genes through antioxidant response elements. Biophysiol. Res. 1997;236(2): 313-322
CrossRef - Cullinan SB, Gordan JD and Jin J. The Keap1-BTB protein is an adaptor that bridges Nrf2 to a Cul3-based E3 ligase: oxidative stress sensing by a Cul3-Keap1 ligase. Cell Biol. 2004;24(19):8477–8486
CrossRef - Furukawa M and Xiong Y. BTB protein Keap1 targets antioxidant transcription factor Nrf2 for ubiquitination by the Cullin 3-Roc1 ligase. Cell Biol. 2005;25(1):162–171
CrossRef - Dinkova-Kostova AT, Holtzclaw WD and Cole RN. Direct evidence that sulfhydryl groups of Keap1 are the sensors regulating induction of phase 2 enzymes that protect against carcinogens and oxidants. Natl. Acad. Sci. USA. 2002;99(18):11908–11913
CrossRef - Levonen AL, Landar A and Ramachandran A. Cellular mechanisms of redox cell signalling: Role of cysteine modification in controlling antioxidant defences in response to electrophilic lipid oxidation products. J. 2004; 378(2):373–382
CrossRef - Copple IM, Lister A and Obeng AD. Physical and functional interaction of sequestosome 1 with Keap1 regulates the Keap1-Nrf2 cell defense pathway. Biol. Chem. 2010;285(22):16782–16788
CrossRef - Lau A, Wang XJ and Zhao F. A noncanonical mechanism of Nrf2 activation by autophagy deficiency: direct interaction between Keap1 and p62. Cell Biol. 30(13):3275–3285
CrossRef - Komatsu M, Kurokawa H and Waguri S. The selective autophagy substrate p62 activates the stress-responsive transcription factor Nrf2 through inactivation of Keap1. Cell Biol. 2010;12(3):213–223
CrossRef - Feng L, Zhao K and Sun L, 2021, SLC7A11 regulated by NRF2 modulates esophageal squamous cell carcinoma radiosensitivity by inhibiting ferroptosis. Transl. Med. 2021;19(1): 367
CrossRef - Abdalkader M, Lampinen R, Kanninen KM, Malm TM, and Liddell JR. Targeting Nrf2 to suppress ferroptosis and mitochondrial dysfunction in neurodegeneration. Neurosci. 2018;12:466
CrossRef - Song X and Long, D. Nrf2 and ferroptosis: A new research direction for neurodegenerative diseases. Neurosci. 2020;14:267
CrossRef - Imai T, Iwawaki T and Akai R.Evaluating experimental cerebral malaria using oxidative stress indicator OKD48 mice. J. Parasitol., 2014;44(10): 681-685
CrossRef - Epiphanio S, Mikolajezak SA, Goncalves LA, et al.. Heme oxygenase-1 is an anti-inflammatoy host factor that promotes murine plasmodium live infection. Cell Host Microbe. 2008;3(5): 331-338
CrossRef - Ruberto AA, Maher SP and Vantaux A, Joyner CJ, Bourke C, Balan B, Jex A, Mueller I, Witkowski B and Kyle DE. Single-cell RNA profiling of Plasmodium vivax-infected hepatocytes reveals parasite and host-specific transcriptomic signatures and drug targets. Cell Infect. Microbiol. 2022;25(12): 986314
CrossRef - Kumar S, Chauhan R and Agnihotri N. Quercetin inhibits Plasmodium falciparum lactate dehydrogenase: A potential antimalarial target. Nat. Prod. 2018;81(7): 1735–1740
Abbreviations List
SOD: Superoxide dismutase
Nrf2: Nuclear factor erythroid 2 related factor
EEOg: Ethanol extract of Ocimum gratissimum
GSH: Reduced glutathione
PfLDH: Plasmodium falciparum lactate dehydrogenase
RNS-OS: Reactive nitrogen and oxygen species
ARE: Antioxidant responsive element
Keap1: Kelch-like ECH-associated protein 1
CAT: Catalase
FFPE: Formalin-fixed and paraffin-embedded
BALF: Bronchoalveolar lavage fluid
INT Infected, but not treated
NINT: Neither infected nor treated
LDS Artemether-lumefantrine
BALB/c: Bagg albino
WHO: World Health Organization







