Manuscript accepted on :02-04-2025
Published online on: 29-05-2025
Plagiarism Check: Yes
Reviewed by: Dr. Audrey Jacob
Second Review by: Dr. Tetty A
Final Approval by: Dr. Patorn Piromchai
Fahima Aktar1 , Md. Naymor Rahaman2
, Md. Naymor Rahaman2 , Hasina Akhter Simol3
, Hasina Akhter Simol3 , Mahfuza Afroz Soma4
, Mahfuza Afroz Soma4 and Md. Zakir Sultan3,*
and Md. Zakir Sultan3,*
1Department of Pharmaceutical Chemistry, Faculty of Pharmacy, University of Dhaka, Dhaka, Bangladesh
2Department of Chemistry, University of Dhaka, Dhaka, Bangladesh
3Centre for Advanced Research in Sciences (CARS), University of Dhaka, Dhaka, Bangladesh
4Cancer research graduate program, University of Nebraska Medical Center, Omaha, NE, USA.
Corresponding Author E-mail:zakir.sultan@du.ac.bd
DOI : https://dx.doi.org/10.13005/bpj/3176
Abstract
Seaweeds provide a range of crucial vitamins, among them water-soluble B complex vitamins such as thiamine (vitamin B1) and riboflavin (vitamin B2). The study focuses on the accurate quantification of thiamine and riboflavin in seaweed samples through an accurate and precise analytical process involving acid and enzymatic hydrolysis followed by HPLC analysis with a fluorescence detector. The applied methods were very accurate and precise as it performed recovery percentage about 99.06% for thiamine and 95.81% for riboflavin and RSD percentage less than 2% on average. Our findings reveal significant levels of thiamine and riboflavin present in the seaweeds which emphasis the nutritional value of seaweeds. Our study says that seaweeds contain up to 2.3 mg thiamin /100g and 0.9 mg riboflavin/100 g. The level of vitamins content varied with the variety of species and the environmental condition. Although with relatively lower riboflavin content, Seaweeds are very good source of vitamins. This study highlights the nutritional value of seaweeds which can be consider as powerhouse in nutritional context. Further exploration into other vitamins (water and fat soluble) and essential minerals would enhance our understanding of their nutritional significance.
Keywords
Fluorescence Detector; HPLC; Nutritional value; Riboflavin; Seaweeds; Thiamine
Download this article as:| Copy the following to cite this article: Aktar F, Rahaman M. N, Simol H. A, Soma M. A, Sultan Z. Quantification and Evaluation of Thiamine and Riboflavin in Marine Resources of the Bay of Bengal. Biomed Pharmacol J 2025;18(2). |
| Copy the following to cite this URL: Aktar F, Rahaman M. N, Simol H. A, Soma M. A, Sultan Z. Quantification and Evaluation of Thiamine and Riboflavin in Marine Resources of the Bay of Bengal. Biomed Pharmacol J 2025;18(2). Available from: https://bit.ly/4dzlOwG |
Introduction
The Bay of Bengal is known for its exciting ecosystems and diverse marine life. Seaweeds are one of the natural treasures of the Bay of Bengal, which occur in a variety of colors, forms, and sizes. These marine algae are not only amazing to look at, but they also perform an important function in maritime ecosystems. They are considered primary producers of the marine ecosystem because they feed and shelter an extensive diversity of marine species.1 Seaweeds are extremely useful and are gathered for a variety of applications across sectors. They are utilized in food, fertilizers, and a variety of industrial applications, such as agar and biofuel production.2
Seaweeds are highly valued for their nutritional density. According to various studies seaweeds are abundant in protein, fat, vitamins, and minerals.3-6 They provide significant amount of protein, vitamins and minerals which are essential for the growth, repair and maintenance of the body tissue. The edibility of seaweeds is determined by the quantity of nutrients they possess, alongside the essential vitamins they contain.7
Vitamins are regarded as necessary nutrients for our bodies to function properly. Seaweeds contain both hydrophilic and lipophilic vitamins.8 Naturally seaweeds contain higher levels of the water-soluble vitamin B and vitamin C content.9 Thiamine (commonly known as vitamin B1) and riboflavin (also known as vitamin B2) are B complex vitamins that are both water soluble.10 Thiamine is well known for its di-phosphorylated derivatives (ThDP), which act as important coenzymes in energy metabolism. However, various activities other than coenzyme activity have been proposed for this vitamin for some years.11 Thiamine triphosphate (ThTP) and thiamine pyrophosphate (ThPP) are essential for neuronal function, membrane transport, and energy conversion. Riboflavin produces the coenzymes flavin adenine dinucleotide and riboflavin mononucleotide, both of which are involved in energy metabolism.12 The conventional technique for extracting Thiamine and Riboflavin from food matrices requires an initial treatment with acid, usually hydrochloric acid.13-20 Enzymatic hydrolysis is subsequently applied.13-15, 19-22
Thiamine and riboflavin both are UV active compounds. So those can be analyzed by HPLC assembled with UV detector.23-25 Those two essential vitamins can also be detected by fluorescence detector.26-28 Riboflavin is naturally fluorescent compound while thiamine is not fluorescent in nature. Thus, thiamine needs to be oxidized to thiochrome to detect with fluorescence detector. Fluorescence detector is more preferable over UV detector for the analysis of those vitamins as it is faster, more specific and sensitive than UV.
In this research work, thiamine and riboflavin were extracted from seaweeds through a step wise process involving hydrolysis with HCl followed by treatment with taka-diastase enzyme. Detection of the two vitamins were achieved by using a fluorescence detector. Fluorescence property of thiamine was induced with an oxidant containing potassium ferricyanide in sodium hydroxide which was introduced before the detector. This oxidant served to oxidize thiamine to thiochrome, enabling sensitive detection. By using this method, we aim to offer detailed insights into the thiamine and riboflavin content of seaweeds, highlighting their nutritional value and potential applications.
Materials and Methods
Chemicals
Thiamine and riboflavin standards were purchased from Supelco (Bellefonte, USA). Taka-diastase,1-Hexanesulphonate sodium salt, potassium ferricyanide, sodium acetate trihydrate, sodium bicarbonate, sodium hydroxide, hydrochloric acid and acetic acid were bought from Sigma Aldrich (St. Louis). Methanol and ethanol (HPLC grade) were purchased from Supelco (Burlington). Distilled water was supplied by CARS, DU.
Concentration of the solutions used throughout this research were as follows: 0.1N HCl, 10% taka-diastase in 2.5M sodium acetate, 20% acidified ethanol (pH=4.0), 0.02N acetic acid, 0.005M 1-Hexane-sulphonic acid sodium salt in 85:15 methanol-water, 0.001M potassium ferricyanide in 0.375M NaOH. All the solutions were prepared on the day of use.
Sample collection
All the fresh samples were collected from the vicinity of Cox’s Bazar and Teknaf area of the Bay of Bengal in Bangladesh and identified by a seaweed specialist. The fresh samples namely Sargassum platycarpum, Ulva fasciata, Ipomoea pescaprea, Ulva reticulata, Eucheuma spinosum, Enteromorpha clathrata, Caularpa sp., Sargasum sp., Glacilaria sp., Gracilaria sp. were collected, stored in polyethylene bags and then carried to the labs for further processing.
Preparation of samples
All samples were subjected to thorough cleaning and drying for several days before any chemical treatment. The dried samples were grinded and homogenized by using a blender. Then, 5 g of each dried homogenized samples were transferred into individual conical flasks. For extraction, approximately 60 mL of 0.1 N HCl solution were added to each flask and stirred to ensure thorough mixing. Then the flasks were tightly sealed with aluminium foil and placed to a boiling water bath at 80 ℃ for approximately 30 minutes. After being removed from the water bath, the flasks were cooled until the temperature dropped below 50 ℃. Once cooled, 5 mL of a 10% taka-diastase enzyme solution (prepared in 2.5 M sodium acetate) were added into all the flasks. The flasks were resealed and then incubated in a water bath at 37 ℃ overnight for additional processing.
The following day, the samples underwent filtration, and the resulting filtrate was carefully collected into 100 mL volumetric flasks and the volume of each solution was adjusted up to the mark by adding distilled water. Lastly, the samples were passed through a 0.22 µm syringe filter and collected into 1.5 mL amber glass vials, rendering them ready for analysis.
Preparation of standards
11.2 mg of thiamine-HCl (99.9% purity) was dissolved with 20% acidified ethanol in a 50 mL volumetric flask. Separately, 10 mg of riboflavin (99.9% purity) was dissolved with 0.2 N acetic acid in another 50 mL volumetric flask, the mixture gently heated in a water bath until the riboflavin was completely dissolved. Then the volume was made up to the mark by adding more 0.2 N acetic acid.
To prepare a composite standard, 10 mL of each standard solution was mixed in a 100 mL volumetric flask and diluted with distilled water. Following this, 5 mL of the resulting composite mixture was transferred into another 100 mL volumetric flask and treated with 0.1 N HCl. The flask was then placed in a water bath at 80 ℃. Once removed from the water bath, the flask was cooled to below 50 ℃ and treated with a 10% taka-diastase enzyme solution. Subsequently, it was incubated at 37 ℃ overnight, mirroring the treatment of the samples. The following day, the treated standard composite solution was filtered and further diluted to 100 mL with distilled water. From this composite stock solution, composite standards of five different concentrations (200 ppb, 400 ppb, 600 ppb, 800 ppb, 1000 ppb) were prepared for calibration.35
Instrumentation
A dual pump reversed phase high performance liquid chromatography (HPLC) was used for this research work. The sample was injected automatically by using an autosampler (SIL-20AC HT). Anon-polar C-18 ODS 250 × 4.60 mm, 5µ column was used for the separation of the compounds based on their hydrophobic interaction. Detection of the compounds were achieved by using a fluorescence detector (RF-10AXL). The output results were monitored by using LC solution software. This whole set up were used for the analysis of all the samples and the standards.
Mobile phase, Oxidant and Chromatographic condition
For the determination of thiamine, a mobile phase was used which consists of 0.005 M Hexane-sulphonic sodium salt in a mixture of 85% methanol and 15% water (pH ≈ 6.0) while a mobile phase comprising 50% methanol in water was employed for riboflavin determination [26]. Since thiamine is not naturally fluorescent, an oxidant solution containing 0.001 M potassium ferricyanide in 0.375 M NaOH was introduced before the detector to induce fluorescence properties. Unlike thiamine, riboflavin does not require an oxidant for detection as it is a naturally fluorescent compound. The injection volume was 10 µL for the whole analysis in both cases. the isocratic flow rate of both the mobile phase and oxidant was maintained at 1.0 mL/min throughout the analysis conducted at 28 ℃.35
Results
Optimization of HPLC conditions
Successfully obtaining acceptable separation between peaks of vitamin B1 and B2 simultaneously without experiencing any interference between them has proven to be a difficult issue, as emphasized in previous studies.13, 18, 30
Therefore, in this studies, thiamine and riboflavin were analyzed separately using different methodologies by HPLC. Since HPLC is one of the sophisticated instruments for analysis of vitamins, drugs and so on.31-32 For the analysis of thiamine, the optimized mobile phase consisting of 0.005M Hexane-sulphonic sodium salt in a mixture of 85% methanol and 15% water at pH 6.0 was used, along with an oxidizing agent containing 0.001M potassium ferricyanide in 0.375M NaOH. It is noteworthy that thiamine determination is particularly pH-sensitive, and at pH 6.0, it exhibits optimal peak symmetry Graph 1. On the other side, riboflavin determination the optimized mobile phase was 50% methanol in water.
 |
Graph 1: Chromatograms for the Standards and Samples analysis.(A) Thiamine std, (B) Riboflavin std., (C) Thiamin in Ulva fasciata and (D) Riboflavinin Sargasum sp.Click here to view Graph |
Presence of Thiamine and Riboflavin in the seaweed samples
Samples from both acid and enzymatic hydrolysis showed identical retention times as the reference composite of thiamine and riboflavin (3.276 ± 0.017 min for thiamine and 4.557 ± 0.011 min for riboflavin) when analyzed on a C-18 ODS 250 × 4.60 mm, 5µ column with a fluorescence detector and mobile phase running at a 1.0 mL/min isocratic flow rate. Thiamine standards and all 10 samples showed a single peak at 360 nm excitation and 435 nm emission while riboflavin standards and samples showed a single peak at an excitation wavelength of 440 nm and an emission wavelength of 530 nm, which corresponded to particular retention durations.
These consistent results act as a proof that the seaweed samples definitely contain both thiamine and riboflavin.
Limit of detection (LOD), Limit of quantification (LOQ), Accuracy and Precision
The limit of detection (LOD) of this study was calculated from the signal to noise ration of the chromatogram of standards. The LOD was found to be 4.0 ppb as it showed 3:1 signal to noise ration while the LOQ was determined to be 12.0 ppb (Table 1). The precision of this study was accessed through the relative standard deviation. All the sample and standards were analyzed at least for three times and the RSD were calculated. It was observed that the RSD value were varied in between 0.29 to 5.95% (Table 2). The average RSD or coefficient variation for thiamine was 1.012% while it was 1.62% for the riboflavin analysis indicating high precision of the analysis.
Table 1: Analytical parameters employed to assess of thiamine and riboflavin content in marine seaweed samples.
| parameters | Thiamine | Riboflavin |
| Slope (m) | 4504.3 | 1220.3 |
| Y- Intercept | -569327 | -2469.2 |
| Regression coefficient (R2) | 0.9989 | 0.9974 |
| LOD(ppb) | 4.0 | 12.0 |
| Recovery (%) | 99.06 | 95.81 |
Table 2: Levels of thiamine and riboflavin found in the seaweed samples.
| Samples Name | Average Concentration (ppb) | RSD (%) | Level of thiamine and riboflavin (mg/100g) | |||
| Thiamine | Riboflavin | Thiamine | Riboflavin | Thiamine | Riboflavin | |
| Sargassum platycarpum | 178.73 | 243.94 | 0.52 | 0.43 | 0.353 ± 0.002 | 0.481 ± 0.002 |
| Ulva fasciata | 162.43 | 32.51 | 0.69 | 5.01 | 0.324 ± 0.002 | 0.065 ± 0.003 |
| Ipomoea pes-caprea |
691.89 | 138.38 | 0.11 | 5.95 | 1.382 ± 0.002 | 0.276 ± 0.015 |
| Ulva reticulata | 320.57 | 131.81 | 0.63 | 1.31 | 0.639 ± 0.004 | 0.263 ± 0.001 |
| Eucheuma spinosum | 219.29 | 231.98 | 1.04 | 0.64 | 0.438 ± 0.005 | 0.463 ± 0.003 |
| Enteromorpha clathrata | 1162.96 | 181.99 | 3.53 | 0.29 | 2.326 ± 0.082 | 0.364 ± 0.001 |
| Caularpa sp. | 268.74 | 39.35 | 0.5 | 1.31 | 0.535 ± 0.003 | 0.078 ± 0.001 |
| Sargasum sp. | 336.68 | 50.49 | 1.56 | 0.99 | 0.669 ± 0.011 | 0.100 ± 0.001 |
| Glacilaria sp. | 552.14 | 401.79 | 1.06 | 0.001 | 1.093 ± 0.012 | 0.795 ± 0.001 |
| Gracilaria sp. | 465.98 | 447.70 | 0.48 | 0.27 | 0.924 ± 0.004 | 0.891 ± 0.002 |
The accuracy of the analysis was tested by spiking of the sample with known concentration of standard solution. The recovery percentage of the spiked sample indicates the accuracy of the current analysis and it was found to be 99.06 % for thiamine and 95.81% for the riboflavin (Table 1).
Discussion
Thiamine content in sea weeds samples, Sargassum platycarpum, Ulva fasciata, Ipomoea pescaprea, Ulva reticulata, Eucheuma spinosum, Enteromorpha clathrata, Caularpa sp., Sargasum sp., Glacilaria sp., Gracilaria sp. which were analyzed, ranged from 0.324 to 2.326 mg/100 g. The Enteromorpha clathrate seaweed contains the highest concentration of thiamine. Although, Glacilaria sp. also contains higher level of thiamine which makes it a good source of those vitamin. Many factors like location of the study, age of sea weeds, drying, preservation and treatment condition may influence the content of thiamine which may vary from earlier results. Vitamin B1 or thiamine is crucial for balanced activities of nervous system and metabolism of energy. RDA requirement of thiamine for adult male is 1.2 mg/day and 1.1mg/day for adult female.33Sea side people suffering from malnutrition can be benefited from these natural sources of thiamine studied here.
Riboflavin content varied between 0.065 and 0.891 mg/100g in the studied samples. Among these Glacilaria sp. contains a greater quantity of riboflavin. Factors like age of sea weeds, drying procedure and analyses conditions can influence the content of the vitamin. Riboflavin, Vitamin B2 is very much necessary in human body for growth and energy production and maintenance of healthy skin and eyes and dried fruits are reputed source of riboflavin as the vitamin is heat stable. RDA requirements for riboflavin in adult men and women are consecutively 1.3 mg/day and 1.1 mg/day.34
The thiamine content dominated over riboflavin in most samples. This finding aligns with earlier research 29,30 which similarly noted higher concentrations of thiamine relative to riboflavin in algae. From the study we found that the seaweeds are rich source of these vitamins.
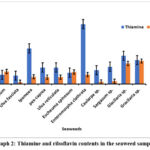 |
Graph 2: Thiamine and riboflavin contents in the seaweed samples.Click here to view Graph |
Conclusion
In this study, we used precise analytical methods to accurately assess the quantities of thiamine and riboflavin in seaweed samples. We accurately quantified these essential vitamins using the acid and enzymatic hydrolysis approach, followed by HPLC analysis. We found that Enteromora clathrata contains the highest amount of thiamine i.e., 2.326 ± 0.082 mg/100 g whereas Gracilaria sp. contains the highest amount of riboflavin i.e., 0.891 ± 0.002 mg/100 g. Our findings highlight seaweed’s nutritional richness, particularly in thiamine, while also revealing a relatively low riboflavin level. This study focuses on critical insights into the nutritional profile of seaweeds, emphasizing their potential as a source of important vitamins in diets. Furthermore, the difference in thiamine and riboflavin concentrations seen amongst seaweed samples highlights the significance of doing further investigation into factors influencing nutritional composition, such as species diversity and environmental conditions. Further exploration into other vitamins (water and fat soluble) and essential minerals would advance our understanding of seaweed’s nutritional value and enlighten their prospective usefulness in nutritional cases.
Acknowledgement
We would like to convey our heartfelt gratitude to UGC Grant 2022-2023, University of Dhaka, Dhaka 1000 for their kind financial assistance, which enabled this research. Their funding was critical in aiding our work and allowing us to achieve significant results. We are also grateful to Department of Pharmaceutical Chemistry, University of Dhaka and Centre for Advanced Research in Sciences (CARS), for giving access to critical resources and equipment. Their assistance greatly aided the effective execution of this investigation. The technical help and facilities provided by them were invaluable in carrying out our tests and analysis. Finally, we thank everyone who helped or encouraged us with this research, whether directly or indirectly.
Funding Sources
Financial Supports for the work from the UGC Grant 2022-2023, University of Dhaka, Dhaka 1000, Bangladesh for the fiscal year of 2022-2023 (Memo No.: Reg./Adm.-3/75706; Date 17/4/2023).
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Author Contributions
- Fahima Aktar: Originating the concept, Supervision of the overall work.
- Naymor Rahaman: Labwork, Manuscript reviewing and editing
- Hasina Akhter Simol: Critically reviewed the whole activities.
- Mahfuza Afroz Soma: Critically reviewed the whole activities.
- Zakir Sultan: Critically reviewed the whole activities.
References
- Dinakarkumar Y, Kothandaraman G. Impact of seaweeds in agriculture. 2017. Available: https://www.researchgate.net/publication/317765252.
- Cabral EM, Oliveira M, Mondala JRM, Curtin J, Tiwari BK, Garcia-Vaquero M. Antimicrobials from seaweeds for food applications. Marine Drugs, 2021; 19(4): 211. doi: 10.3390/md19040211.
CrossRef - Marinho-Soriano E, Fonseca PC, Carneiro MAA, Moreira WSC. Seasonal variation in the chemical composition of two tropical seaweeds. Bioresour Technol. 2006; 97(18): 2402–2406. doi: 10.1016/j.biortech.2005.10.014.
CrossRef - Norziah MH, Ching CY. Nutritional composition of edible seaweed Gracilaria changgi. Food Chem. 2000; 68(1): 69–76. doi: 10.1016/S0308-8146(99)00161-2.
CrossRef - Mishra VK, Temelli F, Ooraikul B, Shacklock PF, Craigie JS. Lipids of the red alga, Palmaria palmata. Botanica Marina. 1993; 36(2): 169-174. doi: 10.1515/botm.1993.36.2.169.
CrossRef - Chan JC-C, Cheung PC-K, Ang PO. Comparative studies on the effect of three drying methods on the nutritional composition of seaweed Sargassum hemiphyllum (Turn.) C.Ag. J. Agric. Food Chem. 1997; 45(8): 3056–3059. doi: 10.1021/jf9701749.
CrossRef - Vinothkumar R, Murugesan S, Kotteswari M, Shanthi N. Biochemical composition of marine red alga Champia parvula (C. Agardh). Int. J. Pharmtech. Research. 2020; 13(3): 186–191. doi: 10.20902/IJPTR.2019.130308.
CrossRef - Škrovánková S. Seaweed vitamins as nutraceuticals. Adv. Food Nutr. Res. 2011; 64: 357–369. doi: 10.1016/B978-0-12-387669-0.00028-4.
CrossRef - Bekah D, Thakoor AD, Ramanjooloo A, et al. Vitamins, minerals and heavy metals profiling of seaweeds from Mauritius and Rodrigues for food security. J. Food Compos. Anal. 2023; 115: 104909. doi: 10.1016/j.jfca.2022.104909.
CrossRef - Cornell L, Arita K. Water Soluble Vitamins: B1, B2, B3, and B6. Geriatric Gastroenterology. Cham: Springer International Publishing; 2021. p. 569–596. doi: 10.1007/978-3-030-30192-7_21.
CrossRef - Bettendorff L, Wins P. Biological functions of thiamine derivatives: Focus on non-coenzyme roles. OA. Biochemistry. 2013; 1(1): 2–18.
CrossRef - Saedisomeolia A, Ashoori M. Riboflavin in human health: a review of current evidences. Adv. Food Nutr. Res. 2018; 83: 57–81. doi: 10.1016/bs.afnr.2017.11.002.
CrossRef - Ali MA, Sultan MZ, Mia ML, Islam A, Ria UH, Salam MA. Evaluation of thiamine and riboflavin in different varieties of rice grown in Kurigram district, Bangladesh. Heliyon. 2024; e39868. https://doi.org/10.1016/ j.heliyon.2024.e39868.
CrossRef - Dawson KR, Unklesbay NF, Hedrick HB. HPLC determination of riboflavin, niacin, and thiamin in beef, pork, and lamb after alternate heat-processing methods. J. Agric. Food Chem. 1988; 36(6): 1176–1179. doi: 10.1021/jf00084a012.
CrossRef - Ndaw S, Martine B, Claude H. Extraction procedures for the liquid chromatographic determination of thiamin, riboflavin and vitamin B6 in foodstuffs. Food Chem. 2000; 71(1): 129–138. doi: 10.1016/S0308-8146(00)00135-7.
CrossRef - Valls F, Sancho MT, Fernández-Muiño MA, Checa MA. Determination of total riboflavin in cooked sausages. J. Agric. Food Chem. 1999; 47(3): 1067–1070. doi: 10.1021/jf9806006.
CrossRef - Fernando SM, Murphy PA. HPLC determination of thiamin and riboflavin in soybeans and tofu. J. Agric Food Chem. 1990; 38(1): 163–167. doi: 10.1021/jf00091a035.
CrossRef - Sims A, Shoemaker D. Simultaneous liquid chromatographic determination of thiamine and riboflavin in selected foods. J. AOAC. Int. 1993; 76(5): 1156–1160. doi: 10.1093/jaoac/76.5.1156.
CrossRef - Fox JB, Lakritz L, Thayer DW. Thiamin, riboflavin and α‐tocopherol retention in processed and stored irradiated pork. J. Food Sci. 1997; 62(5): 1022–1025. doi: 10.1111/j.1365-2621.1997.tb15028.x.
CrossRef - Hägg M. Effect of various commercially available enzymes in the liquid chromatographic determination with external standardization of thiamine and riboflavin in foods. J. AOAC. Int. 1994; 77(3): 681–686. doi: 10.1093/jaoac/77.3.681.
CrossRef - Orzáez Villanueva MT, Díaz Marquina A, Franco Vargas E, Blázquez Abellán G. Modification of vitamins B1 and B2 by culinary processes: Traditional systems and microwaves. Food Chem. 2000; 71(4): 417–421. doi: 10.1016/S0308-8146(00)00131-X.
CrossRef - Valls F, Checa MA, Fernández-Muiño MA, Sancho MT. Determination of thiamin in cooked sausages. J. Agric. Food Chem. 1999; 47(1): 170–173. doi: 10.1021/jf980593l.
CrossRef - Li HB, Chen F. Simultaneous determination of twelve water- and fat-soluble vitamins by high-performance liquid chromatography with diode array detection. Chromatographia. 2001; 54: 270–273. doi: 10.1007/BF02492256.
CrossRef - Koly SF, Kundu SP, Kabir S, Amran MS, Sultan MZ. Analysis of aceclofenac and bovine serum albumin interaction using fluorescence quenching method for predictive, preventive, and personalized medicine. The EPMA J. 2015; 6: 10.1186/s13167-015-0047-x.
CrossRef - Nahar K, Rahman MS, Sajjad MMR, et al. Evaluation of some physicochemical properties as quality control parameters of an ayurvedic preparation “Khadirarishta”. European J. Preven. Med. 2015; 3: 17–21. doi: 10.11648/j.ejpm.20150302.11.
CrossRef - Mia ML, Sultan MZ, Ali MA, Simol HA, Salam MA. Quantification of riboflavin and thiamine in GI (Geographical Indication) branded yogurts collected from Bogura, Bangladesh using HPLC equipped with a fluorescence detector. Bang. Pharma. J. 2024; 27(1): 59–66. doi: 10.3329/bpj.v27i1.71155.
CrossRef - Rahmani N, Muller HG. The fate of thiamin and riboflavin during the preparation of couscous. Food Chem. 1996; 55(1): 23–27. doi: 10.1016/0308-8146(95)00065-8.
CrossRef - Augustin J. Simultaneous determination of thiamine and riboflavin in foods by liquid chromatography. J. AOAC. Int. 1984; 67(5): 1012–1015. doi: 10.1093/jaoac/67.5.1012.
CrossRef - De Roeck-Holtzhauer Y, Claire C, Bresdin F, Amicel L, Derrien A. Vitamin, free amino acid and fatty acid compositions of some marine planktonic microalgae used in aquaculture. Botanica Marina. 1993; 36(4): 321-326. doi: 10.1515/botm.1993.36.4.321.
CrossRef - Brown MR, Mular M, Miller I, Farmer C, Trenerry C. The vitamin content of microalgae used in aquaculture. J. Appl. Phycol. 1999; 11(3): 247–255. doi: 10.1023/A:1008075903578.
CrossRef - Begum R, Sultan MZ, Chowdhury JA, Amran MS. In vitro pharmaceutical equivalence study of three brands of atenolol tablets available in Bangladesh. Dhaka University J. Pharm. Sci., 2019; 18(1): 43–48. doi: https://doi.org/10.3329/dujps.v18i1.41426.
CrossRef - Aktar F, Hossain MJ, Sultan MZ, Rashid MA. Iron (II) and Zinc (II) complexes of gemifloxacin mesylate: synthesis, characterization, serum binding profiling, and evaluation of antimicrobial activity. J. Bang. Acad. Sci., 2022; 46(2): 203–211. doi: 10.3329/jbas.v46i2.63622.
CrossRef - Williams RD, Mason HL, Wilder RM. The minimum daily requirement of thiamine of man. The J. Nut., 1943; 25(1): 71-97. DOI: 10.1093/jn/25.1.71.
CrossRef - Suwannasom N, Kao I, Pruß A, Georgieva R, Bäumler H. Riboflavin: The health benefits of a forgotten natural vitamin. Int. J. Mol. Sci., 2020; 21(3):950. https://doi.org/10.3390/ijms21030950
CrossRef - Islam A, Sultan MZ, Ali MA, Mia ML, Salam MA. Assessment of thiamine and riboflavin levels in common green leafy vegetables grown in Shariatpur, Bangladesh. Heliyon, 2025; 11(1):e41419. https://doi.org/10.1016/j.heliyon.2024.e41419
CrossRef







