Manuscript accepted on :25-04-2025
Published online on: --
Plagiarism Check: Yes
Reviewed by: Dr. Hany Akeel
Second Review by: Dr. Rajan Singh
Final Approval by: Dr. Anton R Keslav
Raveesha Peeriga1* , KrishnaveniManubolu2
, KrishnaveniManubolu2 , Venkateshan Narayanan3
, Venkateshan Narayanan3 , Prashanth Kumar Katta4
, Prashanth Kumar Katta4 , Nuraddeen Ibrahim Jaafar5
, Nuraddeen Ibrahim Jaafar5 and Nagul Meeravali Shaik1
and Nagul Meeravali Shaik1
1Department of Pharmacognosy, V. V. Institute of Pharmaceutical Sciences, Seshadri Rao Knowledge Village, Gudlavalleru, Andhra Pradesh, India.
2Faculty of Pharmaceutical Sciences, Narayana Pharmacy College, Chinthareddypalem, Nellore, Andhra Pradesh, India.
3Arulmigu Kalasalingam College of Pharmacy, Krishnan Kovil, Virudhunagar District, Tamil Nadu.
4Department of Restorative Dental Sciences, College of Dentistry, King Faisal University, Al Ahsa, Saudi Arabia.
5Department of Biomedical Sciences, College of Dentistry, King Faisal University, Al Ahsa, Saudi Arabia.
Corresponding Author E- Mail: drprsha@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3157
Abstract
The physiological and immunological profile of children makes pediatric viral infections particularly difficult. Although pediatric populations are a high risk for hospitalization, pediatric-specific antiviral therapy is largely unexplored, resulting in long delays between pediatric drug approvals and commercialization. This chapter discusses inadequacies in the antiviral drug development and administration for pediatric patients and highlights the importance of age specific approaches including regulatory hindrances to approving children's drugs, and the inhumane nature of performing trials in children. The role that Pediatric Research Equity Act (PREA) and the Best Pharmaceuticals for Children Act (BPCA) played in promoting pediatric focused drug research are highlighted. The chapter seeks to encourage cooperation between pediatricians, pharmaceutical developers, and regulatory bodies, in support of the notion that therapeutic antiviral should be truly pediatric centric. Optimizing treatment outcomes is essential to the global health of young patients and these efforts are required.
Keywords
Antiviral Therapy; Best Pharmaceuticals for Children Act; Case Studies; Pediatric Research Equity Act; Pediatrics
Download this article as:| Copy the following to cite this article: Peeriga R, Manubolu K, Narayanan V, Katta P. K, Jaafar N. I, Shaik N. M. Optimization of Antiviral Therapies among Pediatrics: Confronting Remedying Unique Challenges. Biomed Pharmacol J 2025;18(2). |
| Copy the following to cite this URL: Peeriga R, Manubolu K, Narayanan V, Katta P. K, Jaafar N. I, Shaik N. M. Optimization of Antiviral Therapies among Pediatrics: Confronting Remedying Unique Challenges. Biomed Pharmacol J 2025;18(2). Available from: https://bit.ly/3Sdo8zK |
Introduction
Modern medicine, however, focuses on the development and optimization of antiviral therapies in pediatric populations where there is an outstanding need to tackle the inherently different challenges of pediatric viral infections. Physiologically and immunologically, children are different than adults and especially infants and toddlers are further vulnerable to viral diseases. Their immune systems are still sluggish, and they may not have that same resiliency to a viral infection or the capacity to develop an effective immunology. As a result, children’s viral infections can become life-threatening, with long-lasting periods of illness, complications, and death. Yet despite the high risk of hospitalization among pediatric populations to viral infections, antiviral therapies have frequently been designed for adults, and pediatric care providers must then manage a complex, at best, inadequate treatment landscape for young patients.1-3
One major problem is that very few antiviral drugs are actually first tested and approved for use in adults with pediatric formulations and dosing instructions commonly taking much longer, or not arriving at all.4 This delay can be disastrous for child health, with doctors often unable to give children adult dosages, and children must wait for years before pediatric-specific drugs are approved. However, pediatric populations add to these delays with variations in drug metabolism, body composition, and the absence of appropriate drug formulations (e.g., chewable tablets, liquids, or age-appropriate doses).5 In the absence of pediatric-specific antiviral treatments, this can limit optimal treatment, result in poor therapeutic outcomes, and increase the potential for adverse effects that are particularly concerning given they are occurring in vulnerable populations such as the neonate, infant, and young child.6
Pediatric populations also have distinct pharmacokinetic and pharmacodynamic characteristics than adults, requiring consideration of such for antiviral drug development.7 Children’s bodies process drugs differently than an adult’s does. For example, liver and kidney functions are underdeveloped in infants and toddlers and thus require modifications in drug formulations and dosages in drug products.8 Additionally, the concentration at which a drug reaches the bloodstream may differ in children because of differences in body composition (the proportion of body fat and water). These variances necessitate more individually given treatment regimens that have similar efficacy and safety profiles in young patients.9
The recent approaches were made for increasing antiviral drug accessibility in children with repurposed drugs, age-appropriate drug formulations, and new delivery systems that will facilitate better adherence and more precise dosing.10-12 In the end, we aim to expand the knowledge of pediatric antiviral therapies and to suggest the check to enhance treatment options, to improve the clinical course, and to implement the antiviral care for children in the world in an individual manner.
Current Landscape of Antiviral Use in Pediatric Populations
In pediatric antiviral drug use, the current landscape is a complex mix of critical gaps as well as emerging chances of optimization in antiviral therapies.13 This has seen significant progress in a range of viral infections, yet the burden of these antiviral drugs on the pediatric population continues to be overlooked, with most antiviral drugs once developed and accepted in adults.14 Especially with drugs for common viral infections that disproportionately harm children, like respiratory Syncytial virus (RSV), cytomegalovirus (CMV), influenza, and HIV, this is an especially large gap. The high incidence of these infections and their potential for severity in children, further juxtaposed by the limited availability of pediatric-specific antiviral treatments for those infected observed in current pediatric care, presents a significant pediatric challenge.15
Viral Infections Affecting Pediatric Populations
Viral infections can be dangerous for children, especially when they are under the age of five. Early years are very important for growth and development, and viral infections can disrupt near-term health as well as long-term outcomes. In particular, respiratory viruses can cause serious respiratory illness, resulting in hospitalizations, long-term respiratory complications, or death.16 So Respiratory Syncytial Virus (RSV), a leading cause of bronchiolitis and pneumonia in young children, is an example of this. Because RSV is so common, there are few therapeutic options. Ribavirin is only FDA approved for the treatment of severe RSV infections and is controversial and rarely used because of its side effects and lack of efficacy. Additionally, there are expensive and not universally available but preventive options, such as the monoclonal antibody palivizumab, reserved for high-risk infants. Despite successful clinical trials, there remains a large gap in pediatric medicine — and a critical gap, given the absence of effective antiviral therapies against RSV — in the need for more targeted, safe and accessible medications for children.17-19
Cytomegalovirus (CMV) is another virus that is making a serious impact on the health of infants and young children. CMV infection normally is an asymptomatic disease in healthy persons; however, it can be a serious health problem for newborns, with particular compromise being consequent to acute CMV infection of the mother during pregnancy. Congenital CMV can lead to many different birth defects, including hearing loss, vision problems, developmental delay, and, in some cases, death. Antiviral options for neonates, especially those with symptomatic congenital CMV, are limited and current therapies with Ganciclovir and Valganciclovir have serious safety concerns, including risks of toxicity such as to kidney and to bone marrow suppression. The current lack of pediatric-specific, safe antiviral drugs for CMV has been a longstanding barrier to obtaining optimal treatment until safer and more efficacious pediatric antiviral drugs for congenital infections are developed.20
It also remains a significant issue in pediatric populations during seasonal epidemics. Vaccination has decreased the incidence of severe disease, but influenza continues to cause a large number of hospitalizations and deaths among children each year. Medications such as oseltamivir (Tamiflu) can diminish the impact of influenza, but the treatments have their problems. Commonly given off label, resulting Oseltamivir (and other Neuraminidase inhibitors such as Zanamivir) medications are prescribed for pediatric patients based on extrapolation of formulations and dosing recommendations from adult data. Oseltamivir has been approved by the FDA for use in children two weeks or older but remains untested in children who are younger than 12.21 Resistant strains of influenza, especially so of the seasonal strains, are emerging as a concern for resistance to antiviral drugs, and add to treatment decisions.22
HIV (Human Immunodeficiency Virus) also continues to be a stubborn infection in the pediatric population, including in low- and middle-income countries (LMICs), where the burden of HIV infection in children is also high. ART (Antiretroviral Therapy) has revolutionized children with HIV, but pediatric formulations and doses are not available for many pediatric guidelines, and many are extrapolated from adult guidelines.23 Lifelong ART is required in many children to control viral replication and prevent disease progression, but many drugs available to treat HIV infection are not optimized for use in children. To administer properly such formulations require the use of liquid formulations, chewable tablets, and weighting based dosing, and in many areas beyond home are not available to children of different age.24 Thus, treatment adherence is often challenging, and the chance of treatment failure or resistance is higher in children because suboptimal dosing and the complexity of lifelong therapy make children less likely.25
The Challenge of Pediatric-Specific Antivirals
Although children have a huge burden of viral diseases, treatments for children with viral diseases are limited. Off-label antiviral drugs that have demonstrated efficacy in adults are used in children in many cases.26 This practice, while often necessary, presents several risks and challenges, as stated in Table 1.
Table 1: Key Challenges for Antiviral Treatment in Pediatrics
| Challenge | Description | Implications |
| Lack of Pediatric Formulations | Most antiviral drugs lack child-friendly formulations like oral suspensions, chewable tablets, or liquids. | Without appropriate formulations, treatment adherence may decline, potentially leading to suboptimal therapy and worsened outcomes. |
| Off-Label Use and Dosing Challenges | Antiviral drugs are often used off-label in children, with pediatric dosing extrapolated from adult guidelines. Tablet splitting or crushing introduces high risks of dosing errors. | Inappropriate dosing risks treatment failure, development of drug resistance, and adverse effects, especially in neonates or younger children. |
| Regulatory and Financial Barriers | Pediatric antiviral drug development faces challenges such as limited profitability, small patient populations for clinical trials, and high development costs. | Limited financial incentives result in fewer pediatric-specific antivirals being developed, delaying the availability of effective treatments for viral infections. |
Lack of Pediatric Formulations
Most antiviral are unavailable in child-friendly formulations. Younger children may have difficulty swallowing tablets, and injectable forms may not be suitable for outpatient care, or for children below certain ages. They should have pediatric-specific oral suspensions, chewable tablets, or liquid formulations so that the treatment is safe and effective. Nevertheless, the formulation of such products requires thought on factors such as taste, stability, bioavailability, and ease of administration.
Off-Label Use and Dosing Challenges
When pediatric dosing formulations or guidelines are not available, physicians often are forced to parse recommendations from extrapolation of adult dosing or the use of adult formulations in different dosage forms (e.g., tablet splitting in half or crushing tablets). This practice presents very high risk of dosing error, with the potential for suboptimal drug levels or increased toxicity, and particularly in children or neonates whose often developing organs (liver and/or kidney) processes drugs differently from adults. Dosing that is inappropriate increases the risk of treatment failure, drug resistance, and adverse side effects.
Regulatory and Financial Barriers
Scientifically and financially, developing new antiviral therapies for children is difficult. The inability to generate large numbers of pediatric patients to study drug safety and efficacy in safety studies (and emerging biologics) has decreased potential profitability for pediatric drug development, and this does not hold true for viral infections where patients are less prevalent or more easily preventable (such as RSV and CMV). In the U.S., some barriers like the Pediatric Research Equity Act (PREA) and the Best Pharmaceuticals for Children Act (BPCA) have helped to open the door for pediatric studies but do not offer incentives for pharmaceutical companies to develop pediatric drugs, as the cost of running clinical trials is often costly and the market size is often much smaller.
Since viral infections are so common amongst young children and pediatric specific antiviral drugs are lacking, this effort to develop and optimize treatments specific to pediatric patients is critical.28 It means developing appropriate formulations and dosing guidelines for age that are done with pediatric clinical trials to prove the safety and efficacy of the antiviral treatments. Improving the regulatory framework to encourage the development of pediatric-specific antivirals and better working together between healthcare providers, pharmaceutical companies, and researchers is also necessary.27 In the end, it will reduce the global burden of viral diseases and improve measures of health for children worldwide by developing optimized antiviral therapies.29
Limited Pediatric-Specific Antiviral Therapies
A critical issue in pediatric healthcare is the limited pediatric-specific antiviral drugs. Antiviral medications are commonly used in adults for many different kinds of viral infections, but little has been done regarding pediatric formulations or guidelines for doses in children and adolescents. This is particularly a problem because the gap often means the use of medications in ways that are not appropriate—or that are extrapolated from adult data, using dosages and regimens that are based on scant evidence of safety or effectiveness in children. There is an uncertain clinical landscape for the providers of healthcare with a lack of pediatric-specific data to guide treatment, thus requiring the use of their best clinical judgment rather than evidence-based guidelines.30
Physiological Differences Between Adults and Children
They are not little adults; the way children’s bodies change physiologically as they mature affects profoundly how they respond to medications. It is crucial for pharmacokinetics and pharmacodynamics when these differences exist because of their exposure and polarity, absorption, and metabolism.31 When administering antivirals, clinicians must take into account several factors depicted in Figure 1.
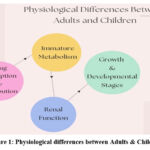 |
Figure 1: Physiological differences between Adults and ChildrenClick here to view Figure |
Drug Absorption and Distribution
In infants and young children, body water is greater than in adults, and body fat is less. That affects how drugs are distributed throughout the body. For instance, the volume of distribution of a water-soluble drug might not be the same as another and will change the concentration in the blood and could change the drug’s therapeutic effect. Alternatively, fat-soluble drugs can accumulate differently, dependent on body fat percentage at various developmental stages.
Immature Metabolism
The liver and kidneys of newborns and infants are immature and dramatically change how drugs are metabolized and are removed from the body. Many medications need to be metabolized by the liver, and this gets better over the first year of life, so babies may not be able to deal well with drugs that adults can use and are metabolized. It can ultimately lead to either drug accumulation with increased risk of toxicity or less optimal clearance so that there are insufficient therapeutic levels. For example, medications like Ganciclovir (used for CMV) or Oseltamivir (for influenza) can be metabolized differently in infants, and adjusting dosing is key, but evidence bases for the use of them in pediatric populations are lacking.
Renal Function
Like liver function, renal function is immature at birth. Clearing many drugs from the body is the job of the kidneys, and the age of the child tends to make it easier for the kidneys to excrete drugs. Slow renal clearance is more common in infants and young children (less than six months of age), especially with this age group; hence, the drug may linger longer in the body and increase the risk for adverse effects.
Growthand Developmental Stages
How we should dose these drugs depends on the child’s development, and children move along different developmental ‘stages’ in the neonatal, infant, toddler, and adolescent clinical periods. But at each stage, body composition, organ maturation, and immune development differ and can influence how safe and effective antiviral medications are. For example, drugs that are well tolerated in older children or adults may not be appropriate for the neonates or infants because of their particular metabolic and physiological differences.
The Problem with Extrapolating Adult Dosing to Children
Because there are no pediatric specific clinical trials available, pediatric patients often receive the adult dosing data32. However, this practice is fraught with risks, as depicted in Figure 2.
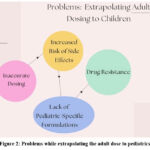 |
Figure 2: Problems while extrapolating the adult dose to pediatricsClick here to view Figure |
Inaccurate Dosing
Such a one-size-fits-all approach with adult dosing guidelines that are applied to children may be incorrect. Metabolic pathways mature in response to as well as differences in organ function, and drug absorption and distribution vary among children. Therefore, a dose that is effective and safe in adults may not be sufficient in children. Antiviral medications such as oseltamivir (Tamiflu) widely used to treat influenza are often dosed in children on the basis of weight, but these calculations are crude; they are based on little adult data and no pediatric clinical trial evidence. Meaning there are currently no rigorous pediatric studies whereby we can accurately predict dosing for children, which could result in underdosing (less efficacy) or overdosing (greater potential to cause toxicity).
Increased Risk of Side Effects
In particular, children, and infants, especially, have higher risk of adverse drug reactions because their bodies react more readily to medications. For example, drugs such as Ganciclovir may produce bone marrow suppression or nephrotoxicity at increased rates in neonates due to their immature organs’ inability to eliminate drug byproducts or metabolites as well as an older child or adult. For instance, sick children taking Oseltamivir have occasionally experienced gastrointestinal signs, including vomiting, and this may be due to dosing, as children may be being given an inappropriate formulation, or because the drug may produce side effects in children.
Drug Resistance
Drug resistance can also result from the use of improper dosing. Again, subtherapeutic Oseltamivir dosing has been associated with Oseltamivir resistance in influenza strains, such as in H5N1. If a child does not receive enough of a strong antiviral (because of inaccurate dosing), the virus can live and multiply, allowing it to become resistant. On the other hand, too high a dose may incur an unnecessary toxicity without any therapeutic benefit whatsoever.
Lack of Pediatric-Specific Formulations
A second major concern is that many antiviral drugs don’t come in formulations suitable for children. Since pediatric-specific drugs are not available, adult formulations are used, an inappropriate example being when tablets are crushed or split for the purpose of adjusting doses to children. For example, crushing tablets may lead to changes in drug stability, bioavailability, and effect; liquid formulations may require special storage; and syrups may be unpleasant to taste or difficult to dose accurately.
Case Study
Oseltamivir (Tamiflu) for Pediatric Influenza
Despite its wide use as an antiviral for the treatment of influenza, the use of Oseltamivir (Tamiflu) in children provides a classic example of the problem of pediatric antiviral therapy. No studies on Oseltamivirhave been done in children under three years of age, and the dosing of oseltamivir in children three or older is based on limited studies. The doses children are recommended to be given are usually calculated based on their body weight, and clinical guidelines recommend higher doses for children and teens. Despite this, dosing recommendations frequently result from extrapolation of data from adult studies without any evidence of optimal dose in the pediatric population. This leads to considerable uncertainty with regard to whether the prescribed dose is safe and effective for younger children, especially for infants whose pharmacokinetics may differ from that of older children.Since oseltamivir is not available in a pediatric-specific formulation, young children may refuse the medication unless it is in a liquid or chewable form that is easier to administer. In addition, the taste of the drug may not be pleasant, and its use may not be adhered to according to the reported treatment regimen. Antiviral treatment in severe influenza in children—particularly those less than five years of age—may be associated with worse outcomes and higher intensive care by nonadherence. Specifically, the challenges in using existing adult-derived dosing regimens and formulations in children make it essential to develop pediatric-specific antiviral therapies. That includes the development of age-appropriate formulations, rigorous pediatric clinical trials, and clear evidence-based dosing guidelines.33
Regulatory Framework for Pediatric Drug Approvals
Recent decades have seen significant change in the regulatory landscape for pediatric drug development in an attempt to address the long-standing problem of children being under-represented in clinical trials and of having access to safe and effective medications. The U.S. Congress, through the Pediatric Research Equity Act (PREA) and the Best Pharmaceuticals for Children Act (BPCA), has laid down the pathway for this arena. Yet, despite such regulatory progress, there continue to be substantial gaps and barriers, especially in the development of pediatric-specific antiviral therapies.34
The Pediatric Research Equity Act (PREA)
The law, the Pediatric Research Equity Act of 2003, requires pharmaceutical companies to demonstrate safety and effectiveness of new drugs for children before the U.S. Food and Drug Administration will approve them. Under PREA, the FDA also can require that sponsors perform pediatric studies on drugs to be used in both pediatric and adult populations. Thus, these studies must determine both appropriate dosing and potential drug side effects in children as well as the drug’s pharmacokinetics (absorption, distribution, metabolism, and excretion) and pharmacodynamics (how the drug acts in the body).35
The goal of PREA is to make sure potential drugs receive proper testing among children before approval, closing a gap in pediatric data that has existed for too long. Failure to comply with PREA may result in delayed approval of a drug or restrain its access to the marketplace, and hence pharmaceutical companies are likely to include pediatric populations in the clinical trials of the drug. But PREA has not ensured companies will test new drugs for use in kids or approve pediatric formulations or even pediatric guidelines for complex meds like antivirals.36
The Best Pharmaceuticals for Children Act (BPCA)
The Best Pharmaceuticals for Children Act (BPCA, 2002) supplements PREA by offering incentives to the pharmaceutical industry to perform pediatric studies for off-patent drugs or for medications that have not previously been studied in children. The most compelling incentive BPCA offers is extending market exclusivity by six months if a company performs satisfactory pediatric studies and then submits them.38 This exclusivity extension has the potential to be financially lucrative for businesses, particularly in the case of older medications for which there may not be any original patents in their possession otherwise. The goal of BPCA is to support the study of drugs in kids when companies may initially have no financial incentive to do so, say when a drug is already on the market generically.37
BPCA is encouraging firms to discover additional pharmaceuticals for children, including antiviral medications that might potentially benefit pediatric patients. The BPCA’s efficacy has been hindered by the limitation that the legislation pertains just to existing medications, rather than addressing the need for novel pediatric formulations for viral infections. Several obstacles remain: there is a deficiency of pediatric-specific formulations and dose recommendations, and numerous antiviral continue to be used off-label in children.39
Challenges in Pediatric Antiviral Drug Development
Despite those legal achievements made through PREA and BPCA, there are several significant challenges in development and authorization of the pediatric antiviral drugs.40 These issues restrict the efficacy of regulatory measures and persist in hindering advancements in the optimization of antiviral medications for children:
Protracted and Complicated Approval procedure
The approval process for pediatric uses remains slow even where clinical studies are carried out for pediatric patients. Officials employing regulating agencies often require extensive clinical trial information that is inclusive of various groups of pediatric patients, which will include neonates, babies, toddlers, and adolescents, a process that takes a lot of time to compile. Most pediatric clinical research, especially involving children with afflictions like HIV, RSR, or influenza, requires complex implementation, thus making it hard for the firms to file petitions for expedited clearance. This causes youngsters to continue using the improper dosages or formulations of successful drugs, sometimes for a long time, without adequate evidence of their safety or efficacy for their age group.
Insufficient Pediatric-Specific Formulations
Lack of suitable form experiences is one of the key challenges for the development of pediatric antiviral therapy. Some drugs that work on adults become very difficult to administer to children because of their small size and developmental milestones and their ability to judge what is palatable. Many antiviral drugs, such as oseltamivir (Tamiflu), have formulations that are not pediatric-adapted; the issue of compliance and dosing becomes compounded. It is important that water-soluble suspensions, tablets that could be chewed, or medicines that dissolve easily in saliva be developed to facilitate medication compliance in children. Nevertheless, pharmaceutical corporations often neglect these formulations, mainly because if the medicine is already available in adult formulations, there is no need for more.
Extrapolating of Adults Information to Children
Extensions of the present results from studies on adult to child groups pose major challenges because of the significant physiological differences between adults and children. The metabolism, the state of organs, and the immune system of children change with age, which is the reason why direct use of adult dosing for pediatric patients is often problematic. The PREA mandates pediatric trials; however, a number of antiviral drugs are authorized based on the adult data, the dose adjustments occasionally being made on an ad hoc basis, which puts kids in jeopardy of both underdosing and overdosing.
Cost and Access Challenges in Low-Resource Environments
Limited pediatric antiviral drugs remain a problem as a dearth of treatment options is still viewed as an essential issue across LMICs where the incidences of viral diseases like HIV, RSV, and influenza have been increasingly identified. Thus, while PREA and BPCA have made improvements in increasing the availability of pediatric medications within high-income countries, high costs of preparations, limited access, and the requisite infrastructure for the delivery of medications remain some major barriers to equitable access in LMICs. Sometimes developing and preparing original antiviral drugs for children is quite costly, and in some less-developed countries, authorities and doctors can face definite difficulties in giving patients legal access to these kinds of treatments.
Hence, there are always fresh needs for enhancing the current regulations and coming up with new approaches in the development of antiviral drugs for pediatric use. Appealing incentives such as the PREA and BPCA have enhanced outcomes; however, further modifications are required to ensure that pediatric patients access safe, effective, and appropriate antiviral medicines on time.41 These improvements may encompass further streamlining of the pediatric clinical trial process because any delays in this process cause a hold-up in the approval of pediatric antiviral medicines.
Considering these gaps, the development of specific pediatric-focused approaches with regards to pharmacological concerns, and ensuring that the legal setting of the antiviral medications’ administration will ensure the expedited, safe, and effective availability for children. This section will explain why these tactics have to be implemented and offer new solutions for overcoming the challenges associated with the development of pediatric antiviral medication.43
Pharmacological Obstacles in Pediatric Antiviral Treatment
One of the major problems that remains crucial in pediatric antiviral therapy is the lack of appropriate formulations. There is a variety of antiviral medicines developed and intended for adult people, which complicates their application to children. This brings some difficulties when babies and young children may be unable to swallow tablets, requiring special dose alterations and different pharmacokinetics as compared to adults.45 Pediatric drug metabolism in some phases is different in the newborn, baby, and toddler periods. Drugs get absorbed, distributed, metabolized, and excreted in quite a different manner in children than in adults.44
This drug is commonly used in children who have the flu; however, oseltamivir comes in capsules or as powder for an oral solution, and the small child then has problems swallowing the tablets. Similarly, in coming up with efficient strategies for dosing modifiers in regard to the kind of antiretroviral drugs in panicked HIV patients considering weight or age criteria, pediatric-specific guidelines might prove rather problematic. Up to the age of two years, children’s hepato-renal immaturity can put them in a disadvantageous position to clear drugs, essential for the metabolism of antiviral agents.
The development of easy-dissolving preparations, liquids, palatable preparations, or capsules is highly essential in progressing improved adherence to therapy regimens. Also, clear guidelines for pediatric dosage are beneficial to avoid adverse effects of the antiviral drugs, especially because research involving children should inform the dosage rates required to treat illnesses in young patients. When the formulation and dosage are not properly standardized, the therapy becomes an experimental approach that may yield poor outcomes or side effects.2 Some of the impediments to drug development and the reasons for advancing kid-appropriate drug development.47
However, even by today’s standards, several rules, including the Pediatric Research Equity Act (PREA) and the Best Pharmaceuticals for Children Act (BPCA), have been developed, but the prospect of the development of drugs designed for the use of children is still filled with immense hurdles.49 PREA calls for pediatric studies of specialty drugs; however, there are gaps in the expeditious approval of pediatric-specific antiviral remedies. It is not rare for companies of pharmacists to face challenges when registering pediatric clinical trials as a result of moral challenges, restricted patient resources, and other complexities associated with conducting research in children. However, costs and time that may be attached to the conduct of pediatric studies may be compromised when the intended drug market is principally the adult population.48
While BPCA provides such encouragements as longer market exclusivity for initiating pediatric trials, corporations seldom feel motivated to embrace pediatric research. As a result, some antiviral medications remain prescribed off-label to children based on data extrapolation from adults, which is often inaccurate in representing the drug’s impact on children. Further, pediatric-specific formulations are often not developed, and thus there is the dearth of the present pharma market to meet children’s needs.50
Taking all of the above into consideration, creative regulatory solutions are critical to bringing the pediatric-specific antiviral medications forward at a quicker pace. Pediatric-specific formulations and antivirals that have met important pediatric requirements should be served with high priority for regulatory authorities to consider incentives for such pediatric products and consideration for fast-track approval procedures for antivirals. On the other hand, the optimization of the data extrapolation process and allowing the use of real-world evidence is likely to be helpful during the linkage between clinical trials and practical treatment options.51
Pediatric Antiviral Drug Development: Collaborative Approaches
Collaboration, both inside and outside of the healthcare industry, offers a highly promising answer to these difficulties. To optimize the use of antiviral medication for children, pharmaceutical firms, healthcare providers, regulatory agencies, public health organizations, and academics need to work together. Together, these stakeholders can address deficiencies in pediatric medication development, drive better research, and deliver novel antiviral therapies to children faster.52
Global Partnerships
Addressing the obstacles to pediatric antiviral medication development is essential, especially for illnesses that primarily affect children in low- and middle-income countries (LMICs) and is particularly dependent on international collaborations. HIV, influenza, and RSV frequently affect these nations in a greater number, but these nations are under-resourced to conduct extensive pediatric clinical studies. The exchange of research data could be accelerated through international cooperation for the development and approval of antiviral medications with pediatric indications. Additionally, pediatric formulations may incur costs that collaborative initiatives may reduce, bringing such formulations to populations most in need.
Public-Private Collaborations
Public-private partnerships (PPPs) can facilitate the rapid development of pediatric antiviral drugs. As such, public-private partnerships (PPPs) can efficiently bridge governmental bodies, pharmaceutical firms, and non-governmental organizations knowledge and resources to help bridge the gap between research and practical implementation. Working with the pharmaceutical associations, however, Global Fund and Gavi, the Vaccine Alliance, have kept a close tab on favorable prices of drugs and vaccines for poor childbearing communities. Antiviral cooperation in such research may ultimately result in increased funding for research, decreased developmental cost, and improved access to medications in areas of need.
The Function of Pediatricians, Pharmacists and Public Health Entities
Promotion of improved access to pediatric-specific antiviral therapies, including prioritizing children’s needs in the drug development process, is dependent on pediatricians, chemists, and public health organizations. Healthcare professionals and pediatricians frequently identify deficiencies in antiviral pediatric medicines and can advocate for the inclusion of pediatric patients in clinical trials and for custom formulations.53
Proficient pharmacists, particularly in terms of drug formulation and patient counseling, are potential partners for healthcare practitioners so as to ensure that children receive the right doses of the antiviral medications and do follow the treatment protocols. They can be used to identify possible adverse effects and make recommendations for the most efficient method of delivery for difficult drugs.
WHO, national health agencies, and other public health bodies have an important part to play in globally defining health priorities and promoting research and development in pediatric antiviral therapies. Together these can financially contribute greatly towards the research collaboration and knowledge exchange between nations, thus fostering the development of less expensive, effective, and age-appropriate antivirals for children.
An approach that is comprehensive, yet novel, in terms of novel regulatory tactics, collaborative research, and pediatric-specific formulations is required to address deficiencies in pediatric antiviral treatment. Safe and effective antiviral therapies for children will require augmented regulatory incentives, global research collaborations, and public-private partnerships to provide for accelerated accessibility. Focusing on the divergence of pediatric requirements ensures that children everywhere get the best antiviral medicines, thereby improving their health and easing the worldwide financial expenses of viral illnesses.54
Obstacles in Formulating Antiviral Treatments for Pediatric Patients
Optimizing treatment for such a sensitive age demographic lags behind the advancement of antiviral medicines for pediatric patients, with specific challenges that complicate antiviral medicine optimization for this population. Pharmacokinetics, ethical considerations, clinical trial logistics, medication formulation, and safety are these obstacles. There are different challenges to be overcome for each of these domains to improve the efficacy and safety of antiviral therapies in pediatric patients.
Pharmacokinetic and pharmacodynamic variability
Comprehending and resolving pediatric to adult disparities in pharmacokinetics (PK), pharmacodynamics (PD), and safety for pediatric patients poses a major challenge in the design of antiviral medicines for children. Pharmacokinetics is the process of drug absorption, distribution, metabolism, and excretion, and pharmacodynamics is describing the physiological effects of a medication on the body. Children are not little replicas of adults; they draw their own lessons. Physiological changes in their bodies that influence the absorption, distribution, and excretion of medication make determining appropriate dosages for this group more complex than for adult populations.
Age-Dependent Metabolism
Hepatic and renal functions important for drug metabolism and excretion are impaired in neonates and newborns. Diminished medication clearance, increased blood concentrations, and an increase in toxicity risk can occur as a result of not appropriately adjusting doses. Newborns have underdeveloped renal function, and drugs such as ganciclovir, used to treat CMV infections, must have dosage adjustment due to this condition.
Body Composition
With growth, children’s body composition changes, including the distribution of medications in the body. Body water and body fat are relatively greater in infants and newborns versus adults, during which time the distribution volume of water-soluble vs. fat-soluble medications will be skewed. This diversity requires highly specific pharmacological doses among pediatric age categories.
Developmental Stage
Children’s immune systems are still developing and therefore may not respond as well to antiviral treatments as adults. What works great for one person may not work at all for another—in infants and children; there are different reactions to infections than adults, as well as in response to pharmaceuticals. Knowledge of the main differences of pharmacokinetics (PK) and pharmacodynamics (PD) between neonates, babies, children, and adolescents is required for the determination of appropriate dosage to achieve target drug concentration and avoid side effects.
These pharmacokinetic and pharmacodynamic disparities emphasize that clinical trials that take into account the distinct physiological features of children’s growing bodies are urgent so that dosing, safety, and efficacy for antiviral medication need to be increased.
Ethical and logistical problems to conducting clinical trials
Intrinsic to pediatric clinical trials is greater complexity due to ethical and practical challenges. Children, however, including babies and toddlers, are unable to give autonomously provided informed permission, so parental permission and, in some cases, older children’s permission is required. That raises the complexity of the trial procedure by forcing researchers to be very careful, with meticulous compliance with ethical norms and protocols for safety, to protect young participants.
Ethical Considerations
A major roadblock to pediatric clinical trials has been the ethical rub of subjecting children to experimental therapies, especially if the long-term effects of a medication are unclear. The potential benefits of involvement must be weighed against its potential harm to children and parents must continuously be educated with regards to the attendant risks of involvement. These make these ethical issues particularly relevant in antiviral medication development—who knows what effects these medications might have on a growing organ or the developing immune system?
Operational Difficulties
Sometimes it is not possible to recruit the number of pediatric patients that the therapeutic team requires for clinical studies, such as for uncommon viral illnesses. This lack of people who are eligible can result in weak, or even nonexistent, trials, leading to the trial failing to generate sufficient evidence on the effectiveness and safety of antiviral. Unlike the research on adult patients, in which the response to drug treatment is more homogeneous and variables such as age, weight, and developmental stage are less critical, the design and execution of studies of children’s responses to treatments are complicated by the heterogeneity of children’s responses. These factors retard the progression of pediatric antiviral medication development and further extend the time for new therapies to reach the market.
Considering these ethical and logistical hurdles, we consider that adaptive trial designs, increased funding for pediatric research, and interinstitutional collaborations are needed to accelerate including children in clinical trials and ensuring they get the appropriate antiviral therapy.
Pharmaceutical Formulation and Administration
Specialized drug formulations of antiviral medications are needed for children because of their special drug development needs. Unlike adults, young children—especially younger ones—often possess an inability to take pills or capsules and so require alternative, more palatable, and simple-to-administer formulations.55
Formulations Suitable for Children
Ensuring that youngsters get the appropriate amount requires the creation of antiviral drugs in liquid or syrup, or orally dispersible formulations, or chewable pills. However, these products have the formulation problem of stability, palatability, and bioavailability. The flavor must be palatable to youngsters to prevent non adherence and liquid formulations must maintain chemical stability over a diverse set of temperatures. Additionally, it is important that the formulation give an exact amount of therapeutic in each dosage, since poor dosing caused by formulation errors may lead to treatment failure or toxicity.
Age-Appropriate Dosing
Consequently, oral formulations that address a broad age spectrum (neonate, baby/toddler, child, adolescent) must be developed with doses that optimize safety, convenience of administration, and safety. A classic example would be that when one has a dose regimen for an adolescent, it may or may not be effective for a neonate, as the body digests the medication differently.
Adherence
Children under the age of six are less likely to follow through with treatment regimens requiring intricate dosage schedules or drugs that are difficult to employ. The efficacy of antiviral medications depends on adherence, inadequate adherence leading to drug resistance, relapse, or treatment failure. Thus, it is important to have antiviral medications that are safe and easy for administration to children in order for them to be utilized successfully.
Safety and Toxicity Apprehensions
The development of antiviral treatments for pediatric populations is markedly constrained by the importance of safety, as children are very sensitive to adverse effects from pharmaceuticals. The organs are still maturing, making them more susceptible to caustic, immediate, and chronic toxic damage.
Short-Term Toxicity
Antiviral agents used in treatment of CMV infections (ganciclovir; foscarnet) are associated with such serious adverse effects as nephrotoxicity and bone marrow suppression. These are alarming side effects in adults, but children—especially babies and neonates—are much more at risk because their organs are still developing. Concerns can only be mitigated with dosing modifications, vigilant oversight, and regular blood examinations, but these strategies may make it more difficult to treat.
Long-Term Toxicity
As many viral illnesses in children, such as HIV, require long treatment with antiviral drugs, it is important to assess their long-term impact on supporting development. Antiviral medications are available early in infancy, and while they can help to prevent the infection, this comes at the cost of possible long-term consequences from drug toxicity, including developmental delays, neurotoxicity, and cardiovascular abnormalities. Long-term follow-up studies and post-marketing surveillance are very important monitoring observations for any late adverse effects of antiviral medications, especially as fears grow over the safety of prolonged drug exposure in children.
If the safety and toxicity in the development of medication are mitigated, researchers can ensure that antiviral therapies are both efficacious and safe for long-term use in pediatric populations. We need to follow up, be attentive in the accessibility of professional practice, and be proactive in the way of early signs of probable adverse effects during the treatment process.
Making antiviral medicines for children is itself a complex endeavour, combining pharmacological, ethical, logistical, and safety factors. These issues must be tackled using customized clinical trials, the development of age-specific formulations, and painstakingly careful safety monitoring to improve antiviral therapies for pediatric populations. Given the challenges just mentioned, healthcare practitioners can assure the efficacy and safety of antiviral medications for young children and do so to improve health outcomes and benefit global pediatric care.57
Addressing the Obstacles in Pediatric Antiviral Drug Development: Strategies
With several hurdles to developing antiviral treatments for pediatric populations, we will discuss how several different ways can be used to overcome them. In doing so, they cover an important territory, including breakthroughs in clinical trials, regulatory incentives, personalized medicine, and formulations of pediatric-friendly pharmaceuticals necessary for the efficacy and safety of antiviral medications for children. We then look at these tactics in more detail.
Novel Clinical Trial Methodologies
The execution of clinical trials in the pediatric population entails high ethical and logistical barriers, many of which are resolved by new trial designs. These tactics can be used to supplement the efficacy of pediatric trials, lessen the danger to children, and improve the accuracy of the data being collected.
Adaptive Trial Designs
An alternative might be using adaptive clinical trials: that will permit the intermediate findings to alter the trial protocols. It can mean fewer children exposed to potentially dangerous medications and give the first few results a chance at fast dose adjustments if preliminary findings indicate unfavorable effects or possible benefits. This permits real-time dosing regimen adjustments, recruitment parameters, or outcome modifications based on empirical data, which helps the researchers to immediately overcome unexpected obstacles.
Modelling and Simulation Approaches
Population pharmacokinetic (PopPK) modelling is becoming an important tool to develop pediatric pharmaceuticals. Mathematical models let researchers mimic drug behaviour across different pediatric age groups to predict drug absorption, metabolism, and elimination in children. Therefore, these models can tell us whether full-scale clinical trials will need ‘a whole pile of children,’ potentially obviating the requirements for such extensive pediatric cohorts. This method can help reduce the number of children who have to be given experimental medications, particularly during tests for unusual illnesses or disorders.
Extrapolation of Data from Adults
This corresponds to what is called extrapolation: the use of data collected from other adults is utilized to infer what outcomes may occur in children (where conducting clinical trials on this population is difficult). Compared to illnesses with disparate pathophysiology across age demographics, there are protocols for regulatory bodies (e.g., the FDA) to extrapolate pediatric data from adult cohorts. This technique will speed up approval of antiviral medications for children without sacrificing safety and effectiveness requirements.
Adaptive trials and modelling techniques can improve the efficiency, ethics, and safety of pediatric antiviral research, shortening the time to discovery of essential medicines for children.
Regulatory Incentives and Collaboration
The financial and logistical difficulties that pharmaceutical firms face in developing pediatric antiviral medications require regulatory incentives, as well as partnerships amongst business, government agencies, and academia to overcome them.
Prolonged Patent Exclusivity for Pediatric Indications:
It has become common practice for pharmaceutical companies to be unwilling to provide funding for pediatric medication development because of the extremely high associated costs in research and regulatory approval. Incentives by regulatory bodies, e.g., prolonged patent exclusivity for pharmaceuticals licensed for pediatric use, can be provided. The possibility of granting extended market exclusivity for pediatric formulations of antiviral medicines could act as an incentive for governments to encourage pharmaceutical firms to develop pediatric antiviral treatments. In other therapeutic domains such as cancer where pediatric medication development is driven by market exclusivity incentives, this methodology has been shown to work.
Pediatric Research Equity Act (PREA) and Best Pharmaceuticals for Children Act (BPCA)
By virtue of their approval procedure, pharmaceutical firms in the U.S. are obligated to do pediatric research. However, these rules are not globally applied, and as a consequence, there are significant delays in pediatric medication approvals. Such regulatory obligations of pediatric antiviral medicines on the global scale serve to guarantee that products undergo very thorough testing prior to market introduction and to ensure better safety and effectiveness data for children.
International Regulatory Collaboration
This will accelerate the process of licensing for pediatric antiviral treatments from within the U.S. FDA, according to the European Medicines Agency (EMA), the World Health Organization (WHO), and national coordination. The standardizing of regulatory criteria internationally helps to reduce the difficulty and expense of obtaining regulatory clearance of pediatric formulations, so eliminating access to therapies for children internationally. The international initiative in support of globally recognizedpediatric—specific protocols for medication development and licensing can be easily achieved using collaborative initiatives.
Collaborations between the public and private sectors
Much more resources and knowledge of the pediatric antiviral research can be furnished by cooperation of the pharmaceutical sector and governmental and academic bodies. Meanwhile, the vaccination and antiviral treatment of illnesses, including HIV and influenza, have been effectively accelerated by public-private partnerships (PPP). Such collaborations can reduce the cost base for pharmaceutical corporations while ensuring the adequate representation of pediatric populations in the process of medical research.
Tailored Medicine and Accurate Dosing
Pediatric antiviral medication development hinges on personalizing medicine and precision dosage to overcome the hurdles so that healthcare practitioners can customize treatment programs for individual patients to improve safety and efficacy.
Advances in pharmacogenomics—the study of how genetic variation influences how medications are processed through the body—offer great promise for pediatric antiviral treatments. Such discoveries in genetics allow healthcare practitioners to find out which genetic characteristics make children prone to metabolism and sensitive to drugs so that treatment regimens can be specially designed for specific patients. Some gene variants in liver enzymes like CYP 450 influence how medications are metabolized by your liver, so understanding these genetic differences helps to determine a safe drug dose for kids, which decreases the chances of undesired effects while maximizing therapeutic outcomes.
Dosing Informed by Biomarkers
Precision dosage of antiviral medications in pediatric patients can be achieved by identifying certain biomarkers. By measuring viral loads in HIV- or hepatitis B-infected children, doctors know what drug dose is necessary to avoid both the proper therapeutic dose and undesirable side effects.
Personalized Dosing Algorithms
Using patient-specific variables such as age, weight, organ function, and genetic profile, personalized dosage algorithms informed by these can improve the efficacy of antiviral medication. Such algorithms may be welcomed by clinical practice, allowing healthcare practitioners to adjust in real time, on the basis of patient reactions, and to avoid under- or overdosing.
Personalized medication and precision dosage can be used to optimize pediatric antiviral therapies, targeting the peculiarities of each child and thus minimizing unwanted effects and increasing the chance of successful outcome.
Creation of Pediatric-Compatible Formulations
A large hindrance to the effective use of antiviral medicines in the pediatric patient relates to the difficulty of developing these treatments in a formulation that can be safely and reliably delivered to children. For improving treatment and adherence results, the development of appealing and simple-to-administer pediatric formulations is critical.
Pediatric-Friendly Dosage Forms
However, to facilitate medicine administration in children, it is necessary to create liquid formulations, syrups, oral suspensions, chewable tablets or dispersible tablets. Younger children who are unable to take the tablets or capsules are often preferred to have the liquid formulations. In these formulations, they must be stable, have a palatable flavor, and allow for precise dosage. From a volume standpoint, pediatric formulations must accommodate how much liquid a child is expected to ingest because there is so much volume to encompass.
Extended-Release Formulations
One way to increase adherence is to develop long-acting formulations—i.e., long-acting solutions or injectable antivirals. Since long-acting formulation minimizes dose frequency, it is particularly useful in pediatric patients with persistent viral diseases, including HIV and hepatitis B. These formulations also help avoid, which are often a problem among young people on difficult regimens.
Innovative Drug Delivery Systems
New opportunities in pediatric antiviral therapy arise from progress in innovative drug delivery systems. Analysing the skin, pharmaceutical companies could engineer transdermal patches to administer antiviral drugs through the skin, allowing the whole application process to occur at a needle-free grace. Medication bioavailability might be enhanced while providing safer and more effective drug administration through nanoparticles and liposomal formulations. These are especially beneficial to those youngsters who may be reluctant to ingest drugs in conventional formats.
Formulation of antiviral medications can be enhanced to improve the accessibility, palatability, and convenience of the formulations, thereby increasing adherence to antiviral medication, which improves health outcomes for pediatric patients.
Therefore, a comprehensive strategy is needed to address the obstacles in pediatric antiviral medication development. Using innovative clinical trial designs, regulatory incentives, personalized medicine, and the creation of child-friendly formulations are some potential ways to sort out the unique challenges with which the pediatric patients face. It will better assure improved health outcomes in pediatric care by focusing on these measures, which may help to improve the safety, efficacy, and accessibility of antiviral medications for children.50
Case Studies: Effective Pediatric Antiviral Treatments
Case studies demonstrate definitively how novel drug formulation and clinical testing strategies, as well as novel regulatory procedures, address the unique challenges of pediatric antiviral treatment. Each of the case studies shows the power of pediatric-specific antiviral treatments to improve health outcomes and provide important lessons in future medication design for this group.
Oseltamivir and Zanamivir (for influenza treatment)
Zanamivir and oseltamivir are neuraminidase inhibitors, antiviral agents. Extensive use of these medications for the treatment and prophylaxis of influenza in adults and children paralleled a landmark advance in the evolution of treatment with pediatric antivirals.
Development and Pediatric Application
Originally, zanamivir (inhaled) and oseltamivir (oral) were developed for use in the adult population, but because of the prevalence of influenza in pediatric populations and the severe complications associated with it, pediatric formulations were developed. In this way, oseltamivirhas been made available largely as oral suspensions and capsules and is suitable for children over all age groups. Dose recommendations for pediatric patients as young as 5 years old were established for inhalation, whereas those for peroral administration were directed only to adults.
In both cases comprehensive trials for the licensing procedure for both medications took place to determine their safety and effectiveness in pediatric populations. Finally, oseltamivir and zanamivir significantly reduced symptom duration and reduced the risk of influenza complications in pediatric patients, according to the studies. The authorization of these pharmaceuticals for pediatric use was a crucial step forward because it shows that antiviral therapies in children can be both safe and efficacious when based on adult evidence. Exhaustive clinical trials eliminated the problem of establishing a suitable dose that could be applied according to weight and developmental stage, making dosing guidelines more appropriate for pediatric patients.
Effects and Obstacles
These antiviral treatments have made influenza much less morbid and less mortal for children, especially those in high-risk pediatric groups, those with preexisting illnesses such as asthma, cystic fibrosis, or immunodeficiency. However, despite these accomplishments, optimization of dose in very young children is still challenging, in particular with respect to palatability and convenience of administration. The resolution of these concerns has occurred by means of the formulation of liquid formulations and methods of administration, including inhalation.
Modifying adult antiviral treatments for children is achievable in the context of pediatric populations where zanamivir and oseltamivir were developed for. This was due mainly to careful attention to dose, safety, and efficacy demonstrated in pediatric studies and in ongoing research to maximize dose formulation for different age groups.
Respiratory Syncytial Virus—V. Palivizumab
The only treatment indicated for high-risk newborns and young children to prevent respiratory syncytial virus (RSV) infections, palivizumab, a monoclonal antibody, has been shown to be highly efficacious. Respiratory Syncytial Virus (RSV) is a contagious viral infection that, while common, can cause significant respiratory distress, often in premature births, people living with congenital heart disease, and those with compromised immune health.
Development and Pediatric Application
Formula of palivizumab as a preventive intervention for RSV. Palivizumab differs from conventional antiviral medication used to treat active infection and is used to prevent RSV infection in high-risk pediatric groups. It is monoclonal against the RSV virus itself, sticks to the virus,, and prevents it from entering a cell and infecting it. Particularly important for children who are at risk of severe RSV consequences—such as preterm newborns and those with chronic lung illness—this prophylactic medication is crucial.
Meanwhile, palivizumabwas approved for use in children under the age of two and children at increased risk for severe RSV disease. During RSV season, the medication is given by monthly intramuscular injections—typically during winter—so there is continuous protection. But the approval was on condition that the drug reduced the chance of severe RSV-related hospitalizations by as much as 55%.51
However, palivizumab has become the reference treatment for preventing severe RSV infections in pediatric populations at increased risk, reducing the burden on the health systems and hospitalizations and admissions to the intensive care unit. Concerns have been raised over the unavailability of palivizumab among low-income countries due to the substantial expense of palivizumab and limited indication. The practicalityof supplying monthly injections is also another challengebecause we will require regular follow-up and easy access to health professionals.
The effectiveness of monoclonal antibody therapeutics in pediatric antiviral therapy, and especially their role in prophylaxis, is illustrated by palivizumab. This points to the need for tailored medicines to address at-risk populations and low-cost, easily accessible treatments in order to make this feasible for any and all healthcare environments.
Antiretroviral Therapy for HIV
Advancement of antiretroviral treatments (ART) for pediatric populations with HIV/AIDS is a major step in pediatric antiviral medicine. The advancing illness has unique obstacles that children with HIV face, particularly in relation to administering long-term treatment in a pediatric demographic.
Development and Pediatric Formulations
The treatment of HIV in children has been revolutionized by the creation of pediatric-friendly antiretroviral formulations. ART was originally not intended for children, but only a few pediatric formulations were available. In the face of the need for alternatives to effective treatment from drugs for children, the pharmaceutical firms said they took the initiative to bring in oral suspensions, chewable tablets, and liquid formulations to cater to the age requirements of young children. In addition, fixed-dose combination treatments of various antiretroviral agents were combined in one pill or liquid to make treatment easier, more streamlined, and improve adherence.
Authorization and Administration
However, integrative factors leading to an endorsement of pediatric ART formulations, thorough clinical trials to establish the effectiveness and safety of the drugs for children. The differences in the physiology between adults and children, especially metabolism and drug clearance, were accounted for in pediatric dosage recommendations in various age categories from newborns to teenagers. The pediatric preparation of nevirapine, lamivudine, and zidovudine has been successfully prepared, and the dose has been tailored to the child’s weight and age.
Antiretroviral therapy (ART) in pediatrics has tremendously improved the life expectancy and quality of life in pediatric HIV. Medication adherence is important for the success of HIV therapy, and the development of streamlined combination regimens has improved drug adherence by creating less cumbersome combination regimens. Nevertheless, challenges remain in treating treatment resistance and adverse effects and ensuring that children living in disadvantaged environments receive these medications. Financial and supply chain challenges limit access to pediatric antiretroviral therapy in many low- and middle-income countries where HIV is most common.
Pediatric ART represents a major step forward in the evolution of antiviral medications for children. The present emphasizes the need for pediatric-specific formulations and continuing research for improvements of treatment modalities available to pediatric HIV patients, particularly in resource-limited environments.
These case studies show the advances and continuing problems in pediatric antiviral drug development. The treatment and prevention of many viral infections in children have been significantly influenced by zanamivir, oseltamivir, palivizumab, and antiretroviral medicines, and the need for ongoing innovation in the development of new medical products remains clearly demonstrated. To address the unique challenges of pediatric pharmacology, such as dosing, safety, and formulation, ongoing industry, regulatory, and research community collaboration is necessary to ensure the safety and efficacy of the best available antiviral therapies for pediatric patients.53
Treatment for pediatric populations with advanced and improved antiviral treatments represents a significant problem in contemporary medicine. In this chapter, I have discussed the multiple challenges (including pharmacokinetic and pharmacodynamic variability, ethical and logistical barriers to clinical trials, and formulation and safety issues) that limit drug development of antiviral medications for children. New methodologies and case studies are emphasized that give important insights for future medication development. Pediatric antiviral treatments continue to seek more effective strategies, and this activity is essential.
Ongoing Innovation
The innovations that propel pediatric antiviral treatment also need to advance as the pediatric antiviral treatment itself progresses. So as the prevalence of viral disease rises, disproportionately afflicting children with RSV, influenza, HIV, hepatitis, and many other viruses, there is a growing need for this need to find novel antivirals, including antivirals that elevate immunity. Current deficiency in age appropriate formulations for several antiviral drugs reflects inadequacy of pediatric treatment and their need to move toward research within this domain. Effective solutions to these challenges of pediatric research include innovative clinical trial designs: adaptive trials and population pharmacokinetic modelling, which permit treatments to be efficacious and safe without unnecessary large-scale trials at risk to children. With investigations of personalized medicine, or customizing medication therapies based on a child’s genetic profile, presenting enormous promise for obtaining greater results through optimized dosages and limited side effects, may there be stronger support for the most vulnerable.57
An essential catalyst for advancement efforts to improve pediatric antiviral treatments requires collaboration. Pharmaceutical creation, particularly that of efficacious pharmaceuticals, involves contributions from many stakeholders: researchers, healthcare professionals, regulatory agencies, and pharmaceutical firms. Lacking are deficiencies in research financing, regulatory approval mechanisms, and the conversion of scientific innovations into cost-effective, accessible therapies that will be addressed through enhanced collaborations between business, academia, and government. Additionally, international cooperation between regulatory authorities (e.g., FDA, EMA, and national entities) is necessary for expediting the approval process and expeditious global market introduction of pediatric antiviral treatments—particularly in areas that are underserved—to help end the epidemic.58-61
A framework has been employed through the actions taken under the Pediatric Research Equity Act (PREA), as well as the Best Pharmaceuticals for Children Act (BPCA), which requires pharmaceutical corporations to undertake research in pediatric populations. However, these efforts should still continue to develop as they try to tackle the contours of modern-day medication research, guaranteeing that kids are not lost in the search to fight viral infections.62-64
Regulatory Assistance and Policymaking Modification
The lack of a pediatric regulatory environment has been a huge roadblock in pediatric antiviral development. While policy around the area has moved forward (the Pediatric Research Equity Act and the Pediatric Exclusivity Program, for example), there are still obstacles. Pediatric formulations sometimes have complex and complex regulatory routes, leaving children missing out on important medicines available to adults. Improvements to the regulatory process are needed to facilitate expeditedpediatric-specific formulations licensure and more consistent dosing criteria for testing and dosing in the pediatric population.65-68
Furthermore, the pharmaceutical sector might be encouraged to devote efforts to developing treatments for children by means of governmental incentives such as extended patent protection for pharmaceuticals intended for pediatric use, including funding for children’s related research on pediatric antiviral medications. This will reduce off-label use and increase available formulations for children.69-75
Accessibility and Affordability: Their Significance
The public health imperative of guaranteeing the availability, accessibility, and affordability of pediatric antiviral medications. Despite great progress in pharmaceutical discovery, many children, and especially those from low-income countries, remain denied access to lifesaving antiviral treatments. Cost hurdles to access might be mitigated by global coordination on pricing and donor-funded initiatives (pediatric HIV therapy, for example). Also, the development of affordable pediatric-friendly formulations (such as liquid suspension, chewable tablets, or dispersible tablets) will promote easier medication administration, particularly in environments with limited healthcare infrastructure.76-78
Improving Global Health Outcomes
The goal of improving pediatric antiviral medicines is to minimise a worldwide burden of viral infections in children, leading to high morbidity and mortality every year. An example of how global immunisation efforts for viral diseases, namely influenza and hepatitis B, show that the development and release of antiviral drugs tailored especially for pediatric populations will have a powerful impact for improving public health outcomes. We can move towards a future where pediatric antiviral therapies are broadly available, effective, and especially conceived to meet the developmental needs of children through cooperation, research, and regulatory endorsement.79-80
Conclusion
The principal difficulties and potential techniques to improve antiviral therapy in pediatric populations have been delineated in this chapter. Tackling scientific and practical challenges, like pharmacokinetics, ethics, medication formulations, and safety, will best help us develop the more effective and accessible therapies for children around the world. These difficulties require collective action by players from all sectors—government, business, academia, and public health organizations.
Federal investment in research, regulatory change, and international collaboration on pediatric antiviral treatments is needed, and the future is promising. If we design certain interventions, children across the world will also be receiving safe, efficacious and age appropriate antiviral therapies resulting in enhanced health outcomes and decreased burden of global viral illnesses.
Acknowledgment
The author is also profoundly grateful to the management of V. V. Institute of Pharmaceutical Sciences, Gudlavalleru.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Clinical Trial Registration
This research does not involve any clinical trials
Permission to reproduce material from other sources
Not Applicable
Author Contributions
- PR (Peeriga Raveesha), PKK (Prashanth Kumar Katta), NIJ (Nuraddeen Ibrahim Jaafar ): Conceptualization, Methodology, Writing – Original Draft.
- MK (ManuboluKrishnaveni), NV (N. Venkatesh) & NMS (Nagul Meeravali Shaik ): Data Collection, Analysis, Writing – Review & Editing.
References
- Van der Lee J. H and Vastert S. J. Pediatric antiviral drug development: Current state and future directions. Pediatr. Infect. Dis., 2020; 15: 103-112.
- Wong M. J and Lee M. Challenges in pediatric drug development. Pediatr. Pharmacol. Ther., 2019; 24: 487-494.
- Jiang Y and Tang X. Pediatric antiviral therapies: State-of-the-art and challenges. Drugs, 2021; 23: 537-548.
- Furuta Y and Komeno T. Development of antiviral agents for pediatric populations. Antiviral Res., 2020; 181: 104943.
- Bocchini J. A and DeStefano F. Regulatory challenges and the approval of pediatric drugs. Pediatrics, 2020; 145: e20191632.
- Keller L. A and Roy M. A. Pediatric drug formulations and the challenges of antiviral therapies. Pediatr. Pharm. Ther., 2020; 26: 245-251.
- Sullivan K and Gabbay R. The pharmacokinetics of antiviral drugs in children. Infect. Dis. J., 2018; 37: 1135-1142.
- Finkelstein J. A and Greenberg M. Pharmacodynamics and antiviral drugs in pediatrics. Drugs, 2019; 21: 123-135.
- Hale M and Chandler C. Extrapolation of adult data for pediatric antiviral dosing: Challenges and considerations. Pharmacother., 2019; 53: 881-888.
- Gollins S. M and Beck C. Pediatric clinical trials for antiviral drugs: Innovations and regulatory considerations. Lancet Child Adolesc. Health, 2020; 4: 410-418.
- Glazebrook P and Howard L. Safety concerns with pediatric antiviral therapy. Drug Saf., 2018; 41: 335-347.
- Baker S and MacDonald S. Ethical challenges in pediatric clinical trials of antiviral drugs. Pediatrics, 2019; 143: e20183434.
- Parks A and Beeler T. Innovative approaches to pediatric antiviral drug development. J. Pharmacol., 2020; 60: 590-597.
- Sharma D and Gupta M. Pharmacokinetic considerations for antiviral therapies in neonates and infants. Pharmacokinet., 2019; 58: 365-376.
- Bardeguez A. D and Korman A. M. Off-label use of antiviral medications in pediatric populations. Pediatr. North Am., 2020; 67: 589-607.
- Ross D. J and Mathews K. H. Formulations of antiviral medications for pediatric use: Practical challenges. Drugs, 2018; 20: 453-463.
- Berthelot D and Fisher S. M. Personalizing antiviral therapies for children. Pediatrics, 2019; 144: e20190500.
- Pérez-Álvarez L and García-Rodríguez M. Challenges in pediatric antiviral drug dosing and administration. Neonatal Drug Ther., 2020; 13: 113-118.
- Goodman J. R and Bourget C. M. Challenges in developing pediatric-specific antiviral formulations. Drug Dev., 2019; 35: 725-731.
CrossRef - Patterson A and Lorber B. Regulatory pathways for pediatric antiviral drug development: The role of PREA and BPCA. J. Ther., 2020; 27: e153-e162.
- El-Hayek S and Waldo P. Antiviral drugs in children: A review of clinical use, safety, and efficacy. Clin. Pediatr. Infect. Dis., 2019; 38: 1-12.
- Raveesha P, Balagani P. K, Raju G. E, Narayanan R, Mohammed J and Yarlagadda L. C. Findings of Steroids, Coumarins and Saponins in Ipomeasagittifolia (Burn. f.). Leaf Extract by HPTLC Fingerprints. J. Chem. Biochem. Sci., 2023; 24: 132-140.
- Raveesha P, Shaik K, Shaik A, Riaz B and Raju G. E. Investigation on ethanolic stem extract of Ipomeasagittifolia to explore the presence of Saponins by High Performance Thin Layer Chromatography. J. Membr. Sci. Technol., 2023; 10: 1012-1020.
CrossRef - Raveesha P, Shaik A, Arabath S. A. M. S, Chennuru M. L and Marimganti S. N. Assessment of Steroids in ethanolic stem extract of Morning glory by High Performance Thin Layer Chromatography. J. Chem. Biochem. Sci., 2023; 23: 361-366.
- Hogg M. T and Shelton D. Successes in pediatric antiviral drug approval. Pediatr. Infect. Dis., 2020; 39: 484-490.
- Kaufman R. P and Reynolds S. S. Optimizing dosing strategies for pediatric antiviral therapies. Lancet Infect. Dis., 2018; 18: 1179-1189.
- Wilson M and Kahn R. L. Innovative methods for pediatric antiviral drug trials: Addressing ethical, pharmacologic, and logistical barriers. Drugs, 2020; 22: 249-257.
- Bala A and Kelliher L. The impact of regulatory requirements on pediatric antiviral drug development. J. Pediatr. Pharmacol. Ther., 2018; 23: 47-56.
- Rakhmanina N. Y and Capparelli E. V. Pediatric HIV therapy: Pharmacokinetics and practical implications of current and investigational antiretrovirals. Pharmacokinet., 2020; 59: 33-48.
- Penazzato M, Lee J, Capparelli E, Zorrilla C, Amornkul P, McKinney R, TamhaneA, Kline A, andMirochnick M. Optimizing drug formulations and dosing to improve treatment outcomes in pediatric HIV. Acquir. Immune Defic. Syndr., 2016; 73: 159-168.
- Hazra R, Siberry G. K, and Mofenson L. M. Growing up with HIV: Children, adolescents, and young adults with perinatally acquired HIV infection. Rev. Med., 2010; 61: 169-185.
CrossRef - Jain V, Hansen H, Flores S, Borkowf C, Tang L and Huo X. Adherence and viral suppression in pediatric HIV patients: Review of global challenges. HIV Res., 2019; 17: 249-261.
- Nachman S, Zheng N, Acosta EP, Apolito K, Blatny M, Byerly S, Bwakura-Dangarembizi M, Capparelli E, Chaix C, Chen J, Hazra R, Hodge J, McCoig C, Monteiro J, Moultrie H, Naver L, Patel K, Saidi F, Violari A, Yogev R. Pharmacokinetics, safety, and efficacy of integrase inhibitors in HIV-infected children: Lessons learned from clinical trials. Pediatr. Infect. Dis. J., 2017; 36: 743-752.
- Puthanakit T, Ananworanich J, Vonthanak S, Kanjanavanit S, Sittiwangkul R and Chokephaibulkit K. (2010). Early versus deferred antiretroviral therapy in children aged 1-12 years. New England Journal of Medicine, 363(7), 635-646.
- Luzuriaga K and Mofenson L. M. (2016). Challenges in the elimination of pediatric HIV-1 infection. New England Journal of Medicine, 374(8), 761-770.
CrossRef - Kapogiannis B. G, Soe M. M, Nesheim S. R, Thomas L, O’Keefe A and Phelps R. (2020). Mortality trends in perinatally HIV-infected children in the United States. Journal of Acquired Immune Deficiency Syndromes, 83(5), 533-540.
- Farr A. M, Sheehan N. L, Harrington T, Roberts K and Tinsley M. (2021). Improving outcomes in pediatric HIV care through optimized antiretroviral regimens. Current Opinion in HIV and AIDS, 16(3), 191-198.
- Baxter C and Abdool Karim S. Combination antiretroviral therapy in children: Challenges and opportunities. N. Y. Acad. Sci., 2018; 1419: 47-63.
- Bagenda D, Musoke P. M, Yiannoutsos C, Kasirye R and Ndeezi G. Survival of HIV-infected children receiving antiretroviral therapy in resource-limited settings. Pediatrics, 2006; 117: e544-e552.
- Violari A, Cotton M. F, Gibb D. M, Khamduang WandMofenson L. M. Early antiretroviral therapy and mortality among HIV-infected infants. Engl. J. Med., 2008; 359: 2233-2244.
CrossRef - Gibb D. M, Goodall R. L, Giacomet V, Ahmed N and Vitoria M. Adherence to prescribed antiretroviral therapy in HIV-infected children in Europe. Pediatrics, 2003; 111: e699-e705.
- McSherry G. D., Shiau S and Abrams E. J. Long-term outcomes in pediatric HIV treatment: Advances and remaining challenges. Opin. HIV AIDS., 2020; 15: 166-175.
- Rutstein R. M and Lowenthal E. D. Adolescents and HIV: Pediatric perspectives on emerging challenges. Opin. Pediatr., 2016; 28: 535-540.
- Ananworanich J and Mellors J. W. Pediatric HIV reservoir dynamics and implications for cure strategies. Opin. HIV AIDS., 2015; 10: 85-91.
CrossRef - Cotton M. F, Violari A, Otwombe K, Nuttall J, McKerrow N and Reddy M. Early versus deferred antiretroviral treatment for HIV-infected infants and children in N Engl J Med. 2013;368(24):2285-2296.
- Nachman S, Zheng N, Acosta E. P, McAllister K and Della Torre C. Adherence and safety profiles in pediatric HIV trials. Clin Infect Dis. 2017;65(6):951-960.
- Davis N. L, Miller W. C, Hudgens M. G, Wilkins A andGeffre C. Virologic suppression and resistance patterns in children on second-line antiretroviral therapy. J Acquir Immune DeficSyndr. 2021;86(4):453-460.
- Van Dyke R. B, Patel K, Siberry G. K, Seage G. R andNachman S. Antiretroviral regimen durability and clinical outcomes in perinatally HIV-infected children. JAMA Pediatr. 2014;168(4):370-378.
- Jaspan H. B, Warneke C, Grebe E, Wessels MandArpadi S. M. Pediatric antiretroviral therapy and resistance: Where are we now? Pediatr Res. 2016;79(3):420-426.
- El-Hayek S. and Waldo P. Antiviral drugs in children: A review of clinical use, safety, and efficacy. The Journal of Clinical Pediatric Infectious Diseases, 2019; 38(4): 1-12.
- Hogg M. T. and Shelton D. Successes in pediatric antiviral drug approval. Pediatr. Infect. Dis., 2020; 39(6): 484-490.
- Kaufman R. P. and Reynolds S. S. Optimizing dosing strategies for pediatric antiviral therapies. Lancet Infect. Dis., 2018; 18(11): 1179-1189.
CrossRef - Wilson M. and Kahn R. L. Innovative methods for pediatric antiviral drug trials: Addressing ethical, pharmacologic, and logistical barriers. Drugs, 2020; 22(4): 249-257.
- Bala A. and Kelliher L. The impact of regulatory requirements on pediatric antiviral drug development. Pediatr. Pharmacol. Ther.,2018; 23(1): 47-56.
- Rakhmanina N. Y. and Capparelli E. V. Pediatric HIV therapy: Pharmacokinetics and practical implications of current and investigational antiretrovirals. Pharmacokinet., 2020; 59(1): 33-48.
- Penazzato M, Lee J, Capparelli E, Zorrilla C, Amornkul P, McKinney R, Tamhane A, Kline AandMirochnick M.Optimizing drug formulations and dosing to improve treatment outcomes in pediatric HIV. Acquir. Immune Defic. Syndr., 2016; 73(2): 159-168.
- Hazra R, Siberry G. KandMofenson L. M. Growing up with HIV: Children, adolescents, and young adults with perinatally acquired HIV infection. Rev. Med., 2010; 61: 169-185.
CrossRef - Committee on Infectious Diseases. Recommendations for Prevention and Control of Influenza in Children, 2023–2024. Pediatrics. 2023;152(4):e2023063772.
- Committee on Infectious Diseases. Recommendations for Prevention and Control of Influenza in Children, 2024–2025: Policy Statement. Pediatrics. 2024;154(4):e2024068507.
- World Health Organization. Pediatric drug optimization for antibiotics. Geneva: WHO; 2024.
- PediaTime Editorial Team. Principles of Antiviral Therapy in Pediatrics. PediaTime. 2024 Jun.
- Krishnaveni M, Raveesha P, Mohan Gandhi B, Bhaskara Raju V, Nirmala K, Edward Raju G, Devi K. Evaluation of Anti-inflammatory Activity of Millingtoniahortensis Leaf Extract. International Journal of Chemical and Biochemical Sciences. 2023; 24(6): 380-385.
- Krishnaveni M, Pavan Kumar B, Edward Raju G, Varalakshmi J, Raveesha P, Sridevi A. R. Unlocking the Potential of Aquasomes: A Comprehensive Review on Innovative Nanocarriers in Drug Delivery and Beyond. International Journal of Chemical and Biochemical Sciences. 2023; 24(6): 405-410.
- Mohan Gandhi B, Raveesha P, Krishnaveni M, Bhaskara Raju V, Nirmala K, Edward Raju G. A Bibliometric Analysis of Investigations on Black Pepper Published from 1978 to 2023. International Journal of Chemical and Biochemical Sciences. 2023; 24(6): 386-394.
- Atmakuri LR, Peeriga R, Begum S, Vallamkonda B, Baratam A. Novel bioanalytical LC-MS/MS method for determination of metoprolol in human plasma. J Appl Pharm Sci. 2024;14(12):131–138.
CrossRef - Peeriga R, VenkataBangaru GK. Implementation of the POCSO Act in Schools. In: Child Sexual Abuse: A Public Health Problem in India. p. 391–413.
CrossRef - Peeriga R, Yarlagadda LC, Alla NR. Protection of Children from Sexual Exploitation. In: Child Sexual Abuse: A Public Health Problem in India. p. 191–201.
CrossRef - Peeriga R, Shiek Arabath SAM, Manubolu K, Thelappilly BB, Yarlagadda LC. Insilico assessment of phytoconstituents in Myxopyrum smilacifolium Blume against arthritis. Biomed Pharmacol J. 2024;17(1):235–241.
CrossRef - Manubolu K, Balagani PK, Gope ER, Peeriga R, Sridevi AR. Unlocking the potential of aquasomes: A comprehensive review on innovative nanocarriers in drug delivery and beyond. Int J ChemBiochem Sci.2023;24(6):405–410.
- Manubolu K, Peeriga R, Bonthu MG, Gope ER, Kadirvel D. Evaluation of anti-inflammatory activity of Millingtonia hortensis leaf extract. Int J Chem Biochem Sci. 2023;24(6):380–385.
- Peeriga R, Chandrasekhar KB. Comparative study of anti-inflammatory activity of petroleum ether extract of Myxopyrum smilacifolium Blume and ethanolic leaf extract of Pamburus missionis J Pharm Sci Res. 2017;9(6):955–957.
- Peeriga, R., Manubolu, K. (2025). Design Strategies for Waste Reduction and Enhanced Recyclability in Pharmaceutical Packaging. In: Yingngam, B., Aslam, M.S., Haghi, A.K. (eds) Sustainable Pharmaceutical Product Development and Optimization Processes. Springer, Singapore.
CrossRef - Krishnaveni Manubolu, RaveeshaPeeriga, K. B. Chandrasekhar. A Short Guide to Clinical Pharmacokinetics. 1st 2024. Springer.Singapore.
CrossRef - Manubolu, K., Peeriga, R. (2025). Ecodesign Principles. In: Yingngam, B., Aslam, M.S., Haghi, A.K. (eds) Sustainable Pharmaceutical Product Development and Optimization Processes. Springer, Singapore.
CrossRef - SinaSalajegheh T, Pouneh H, PhelipeMagalhães D, Ahmed H, Heba A, Sarah El-N, Mohamed F. Unveiling Emerging Avenues in Antiviral Research: Navigating Clinical Trials and Regulatory Landscapes. Advances in Antiviral Research. Singapore: Springer Nature Singapore, 2024. 421-453.
CrossRef - Kyle A. Soltys, Geoff Bond, Rakesh Sindhi, Sara K. Rassmussen, Armando Ganoza, Ajai Khanna, George Mazariegos, Pediatric intestinal transplantation. Seminars in pediatric surgery. WB Saunders. 2017; 26(4): 241-249.
CrossRef - Anne E. M. K, Alasdair B, Alfredo T, Tim R. C, Adrie B, Pauline A, Hilda Angela M, Francis Ateba N, Aminata D, Alexandra C, Marc L, Angela C, Anna T. OptimisingPediatric HIV Treatment: Recent Developments and Future Directions. Pediatric Drugs2024: 1-18.
- Rampedi PN, Ogunrombi MO, Adeleke OA. Leading Pediatric Infectious Diseases—Current Trends, Gaps, and Future Prospects in Oral Pharmacotherapeutic Interventions. Pharmaceutics. 2024: 16(6), 712.
CrossRef - Siberry, George K, and Theodore Ruel. The treatment optimisation ODYSSEY for children with HIV.The Lancet HIV(2025).
CrossRef - Riccardo M, Gianluca B, Edoardo M, Davide L, Milo G, Riccardo Di S, Federico P, Susanna E. Therapeutic Drug Monitoring of Antimicrobial Drugs in Children with Cancer: A New Tool for Personalized Medicine. Pediatric Drugs, 2024: 1-16.
Abbreviations
ART: Antiretroviral Therapy,
BPCA: Best Pharmaceuticals for Children Act,
CMV: Cytomegalovirus,
LMICs: Low and Middle Income Countries,
PREA: Pediatric Research Equity Act,
RSV: Respiratory Syncytial Virus.







