Manuscript accepted on :30-04-2025
Published online on: 20-05-2025
Plagiarism Check: Yes
Reviewed by: Dr. Nitya Krishnasamy
Second Review by: Dr. Safa Mohammed
Final Approval by: Dr. Patorn Piromchai
Sheela Durairajan1* , Preetha Selva2
, Preetha Selva2 , Rajagopalan Vijayaraghavan3
, Rajagopalan Vijayaraghavan3 and Senthil Kumar Sivanesan3
and Senthil Kumar Sivanesan3 and Srinivasan Vengadassalaapathy1
and Srinivasan Vengadassalaapathy1
1Department of Pharmacology, Sri Venkateswaraa Medical College Hospital and Research Institute, Tamil Nadu, Sri Venkateswaraa University, India,
2Department of Pharmacology, Saveetha Medical College and Hospital, Saveetha Institute of Medical and Technical Science, Saveetha University, Tamil Nadu, India.
3Department of Research and Development, Saveetha University, Tamil Nadu, India.
Corresponding Author E-mail: sheela151278@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3185
Abstract
Modification in drug release provides efficient drug delivery for the benefit of the consumers in improving their convenience, compliance, safety, and efficacy. The need for self-administration of drugs has also evolved due to extreme conditions like natural and man-made disasters to reduce the malaise and fatality. The purpose of this research is to know the reliability of using an opioid analgesic through a self-injectable device during extremity conditions. The opioid analgesic used in this research was buprenorphine. This self-injectable analgesic autoinjector is a recyclable device with the advantage of dose selection. Sterile water from the cartridge was replaced with an opioid analgesic (buprenorphine hydrochloride) under a laminar flow to obtain dosage concentrations of 50μg/mL and 100 μg/mL, respectively. All quality control tests were carried out. Opioid analgesic was given intraperitoneally through an autoinjector in albino rats continuously for 7 or 14 days to evaluate the reliability of using an analgesic autoinjector by observing their oxidative stress parameters. The buprenorphine autoinjector and the drug cartridges quenched the required quality check. There was no sign of bleeding or injury in the essential organs of albino rats in the research categories, when they were autopsied after euthanasia. The oxidative stress parameters, viz., GSH, MDA, SOD, catalase, GPX, and GR of blood and GSH and MDA of liver and kidney showed the absence of any remarkable change in the experimental albino rats. The newly developed analgesic autoinjector was tolerable and reliable in albino rats and renders a scope to extend the research in human participants.
Keywords
Autoinjector; Analgesic; Buprenorphine; Self-injection; Oxidative stress; Quality control
Download this article as:| Copy the following to cite this article: Durairajan S, Selva P, Vijayaraghavan S, Sivanesan S. K, Vengadassalaapathy S. Investigation on Reliability of Administering an Opioid Analgesic Through an Autoinjector by Exploring the Oxidative Stress Parameters in Albino Wistar Rats. Biomed Pharmacol J 2025;18(2). |
| Copy the following to cite this URL: Durairajan S, Selva P, Vijayaraghavan S, Sivanesan S. K, Vengadassalaapathy S. Investigation on Reliability of Administering an Opioid Analgesic Through an Autoinjector by Exploring the Oxidative Stress Parameters in Albino Wistar Rats. Biomed Pharmacol J 2025;18(2). Available from: https://bit.ly/3F9OKyP |
Introduction
The efficient drug delivery involves an alteration in drug release to improve its bioavailability, metabolism, and elimination for the benefit of victims to enhance their convenience, compliance, efficacy, and tolerability during their usage.1 The non-invasive conventional routes are easier to administer the drugs, which include per oral, topical, nasal, buccal, sublingual, vaginal, ocular, and rectal.2 Recently, the burden of managing communicable diseases has increased globally. Therefore, the requirement for self-injectable devices has increased even in developed countries like the USA, where over 9000 people attain 65 years every day, and the percentage of adults suffering from multiple chronic diseases has increased, making the hospital unable to meet the demand for patient care.3The need for self-administration of drugs has also evolved due to emergency conditions like natural and man-made disasters. The benefit of a self-injecting device is its flexibility of injecting the medicaments at any time and in any place.4 Autoinjectors, pen injectors, and inhalers are some of the self-administering injecting devices.5 Insulin Pen injectors are used for the dispensing insulin in the management of diabetes.6The inhalers have the advantage of cost-effectiveness and portable convenience, but they can be used only in patients who are able to coordinate.7The novel drug delivery method, like transdermal patches, also has its own limitations, like local irritation, erythema, itching, and local oedema at the site of administration. The drug molecule in a transdermal patch should possess the characteristic feature of smaller size and lipid solubility for better penetration through the skin to produce the effect. The drugs given through this route are delivered in a controlled manner for providing the therapeutic effect for a longer duration.8The limitation of the conventional drug delivery is to provide adequate effect during emergency conditions like man-made disasters, during war, and natural disasters like tsunamis, earthquakes, floods, etc. This has led to the introduction of another self-injectable device, the autoinjector.
The autoinjector was first introduced in the early 1970s as an alternative to manual injection to administer the drugs through the subcutaneous route.9,10 Suitability and acceptability of the autoinjectors by the patient have given way in the treatment of many conditions like anaphylaxis, nerve gas poisoning, seizures, opioid overdose, multiple sclerosis, and rheumatoid arthritis.11Results of uninterrupted development in technologies have contributed to a simple push-type autoinjector from the earlier, more complex, and fully automatic button type. Presently, the FDA has approved an autoinjector for the treatment of adults with moderate to severe rheumatoid arthritis.12For the first time in India, a conception of a self-injectable device for buprenorphine, an opioid analgesic, was proposed. This self-injectable device was available with the replaceable cartridges and dose selection mode for better usage during crisis situations for children and adults.13
Generally, frequently used analgesics are more effective against nociceptive pain than neuroleptic pain. The mild and moderate pains were managed by nonsteroidal anti-inflammatory drugs, and chronic pains were managed with opioid analgesics. Opioid analgesics used in chronic treatment produce physical dependence and reduction in immunity. This partial agonist and antagonist, the opioid analgesic buprenorphine, was used in the development of the present analgesic autoinjector due to its advantages of prolonged effect, minimal neonatal abstinence syndrome, a ceiling effect in respiratory depression, and reduction of immunity on chronic use. Another advantage of an autoinjector is its usability in children and farm.14 Amikacin is an effective antibiotic against gram-negative as well as gram-positive bacteria. Hence, being a broad-spectrum aminoglycoside, it was used in the development of the antibiotic autoinjector.15
Acute or chronic tissue injury can produce oxidative damage to the cell membrane by releasing the free radicals. Free radicals are atoms, ions, or molecules with unpaired electrons, which are not stable and highly reactive. Generally, a healthy cell maintains an equilibrium between the formation of antioxidants and the removal of free radicals. When the equilibrium is altered, there is an increase in cell damage or death due to oxidative stress. These highly reactive ions can initiate lipid oxidation and break the double helical DNA structure and oxidize the biological membrane, resulting in tissue injury, which can result in a decrease of antioxidants like superoxide dismutase (SOD), catalase, and glutathione peroxidase activity (GPX). The decrease in these enzymes can affect the essential functions.16
This self-injectable device can release the medicament with force. The improper handling of the device and the inappropriate muscle size may produce negative efficacy of the device by venturing the patient at risk.17The present work investigates the reliability in delivering the medicaments through the self-injectable autoinjector by noticing any alterations in the oxidative stress parameters of albino rats used for this research.
Materials and Methods
Animal
The weight of the albino Wistar female rats used in this research work ranged from 180 to 240 g (5 to 6 months old), which were purchased from the animal house of Saveetha University. Fifty percent of albino rats from each category were maintained in single polypropylene cage with paddy husk as the bedding material under controlled temperature, humidity, and natural dark/light cycle. Laboratory animal feed (VRK Nutritional, India) and filtered water were given to the albino rats. The experiments were carried out as per the CPCSEA guidelines (Committee for the Purpose of Control and Supervision of Experiments on Animals, India). The research work was initiated after obtaining prior permission from the Animal Ethics Committee of Saveetha University.
Preparation of Buprenorphine Cartridges
Glass cartridges filled with sterile water and embedded inside with a 23-gauge convoluted needle were used for the preparation of buprenorphine cartridges (M/s Neon Laboratories Ltd., Mumbai, India). The capacity of each cartridge was 2.3 – 2.4 mL. When used in the autoinjector, 2.1 to 2.2 mL would be delivered without the restrictor, and with the restrictor, about 1.5 mL would be delivered (dual dose mode). Two different concentrations of 50 μg/mL and 100 μg/mL were prepared by withdrawing 0.4 mL or 0.8 mL of sterile water from the glass cartridge and replacing the same volume with buprenorphine hydrochloride from a 0.3 mg/mL vial (2 mL) of M/s Neon Laboratories Ltd., (Mumbai, India). To check the quality control parameters. A few buprenorphine cartridges were randomly taken and subjected to vibration tests, drop tests, injection efficiency tests, low-pressure tests, and sterility tests.
Autoinjector
The reusable autoinjectors are manufactured by Hindustan Industries (Gwalior), Hariom Technologies (Hyderabad) and Neon Laboratories (Mumbai). They specifically supply atropine and pralidoxime filled autoinjectors for military and paramilitary preparedness against nerve gas poisoning(Figure 1).
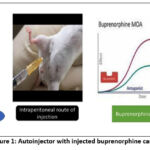 |
Figure 1 : Autoinjector with injected buprenorphine cartridge |
Experimental Categories
The following were the experimental groups.
Group 1: Control, sterile water manual injection for 7 continuous days,
Group 2 – Drug concentration 50 μg/mL, through an autoinjector for 7 continuous days,
Group 3 – Drug concentration with 50 μg/mL, through manual injection for 7 continuous days,
Group 4 – Drug concentration 100 μg/mL, through an autoinjector for 7 continuous days,
Group 5 – Drug concentration 100 μg/mL, through manual injection for 7 continuous days,
Group 6: Drug concentration 50 μg/mL, through an autoinjector for 14 continuous days.
Group 7: Drug concentration 50 μg/mL, through manual injection for 14 continuous days.
In each category, 6 animals were used. The experiment was carried out in phases I & II, containing 3 animals in each category, and the data was pooled.
Drug Delivery through Autoinjector
When an opioid partial agonist analgesic was dispensed through an autoinjector to the albino rats, the animals were held gently and firmly in a supine position on an operation table. After turning the protection lock clockwise in the autoinjector, they were placed gently in the rats, either on the left or right part of the lower abdomen, and the trigger button was pressed to release the medicament intraperitoneal route. The autoinjector was positioned on the abdomen for 10 sec for effective drug delivery. The albino rats were allowed to move freely after the smooth removal of the autoinjector. The weight of the drug cartridge was noted prior and post drug release to calculate the dose and the volume of the medicament dispensed. The expelled needle was measured by a vernier caliper. The drug was injected in albino rats in the left or right part of the lower abdomen on alternate days.
Conventional Drug Injection Technique (Manual)
After administering buprenorphine through an autoinjector to the albino rats, the drugs remaining in the cartridges were collected separately (50 μg/mL and 100 μg/mL) for manual medicament intake. The volume of drug injected intraperitoneally is around 1.2 mL using a 2.0 mL syringe with a gauge size of 23.
Sample Collection
The weight of the albino rats, food and water intake, along with their general behaviour, were monitored and noted daily during the experimental period. Blood was withdrawn from the orbital plexus on the 8th or on the 15th day after anesthetizing the rats with isoflurane. Blood was collected in a vacutainer with tri-potassium ethylenediamine tetra acetic acid to prevent coagulation. The animals were euthanized with an overdose of isoflurane, and the liver and kidney were excised out. These organs were weighed, cleaned, and stored at –80°C for biochemical analysis.
Estimation of antioxidants in blood
Superoxide dismutase (SOD) was estimated by following the procedure of Kakkar (1984). A blood sample with anticoagulant was taken, and hemolysate was prepared based on the above method. The quantification of this antioxidant was done at 560 nm using a spectrophotometer (Genesys 10S, USA). The SOD antioxidants interfere with the formation of the nicotinamide adenine dinucleotide NADH-phenazine methosulphate-nitro blue tetrazolium complex.18Catalase activity was determined as per the procedure mentioned by Luck (1974). The catalase activity was estimated by measuring hydrogen peroxide using a spectrophotometer at the wavelength of 240 nm.19 Ohkawa’s method (1979) was used to calculate malondialdehyde (MDA) in which a pink colour is formed due to the interaction of MDA with thiobarbuitric acid and measured spectrophotometrically at 535nm.20Glutathione peroxidase (GPX) and glutathione reductase (GR) activity were also estimated spectrophotometrically. Glutathione (GSH) content was estimated using DTNB (Ellman’s reagent), and the coloured byproduct was measured at 412 nm using a UV-VIS spectrophotometer.21One unit of glutathione reductase enzyme activity is described as the reduction of one μM of glutathione disulfide to glutathione per minute at 25̊ C and pH 7.6.22
Tissue Biochemical Estimation
Specimens for calculating the antioxidant levels were collected in two different parts. Each organ was weighed initially and homogenized using a Potter-Elvehjem tissue homogenizer. Phosphate buffer saline (PBS) 50 mM pH (7.4) was used for the estimation of GST, CAT enzyme activity, and GSH level, and potassium phosphate buffer 10 mM pH (7.4) was used for the estimation of MDA and GPX activity. The supernatant liquid produced during centrifugation of crude tissue homogenate at 10,000 rpm for 15 minutes was utilized for further estimations.23
Statistical Analysis
The research data was analysed by one-way ANOVA with Dunnett’s test for comparison with the control. The research data was expressed as mean + SE. The results in this work were taken to be significant when the “P” (probability) value is equal or less than 0.05. The statistical analysis was carried out using SigmaPlot 13.0, and the graphs were plotted using Systat Software, USA.
Results
The opioid analgesic buprenorphine autoinjectors are intended to be used during catastrophes and require efficient functioning in those situations. Hence, various quality control parameters like robustness, mechanical handling force, drug-delivering time, needle length, volume of drug released, and sterility of autoinjectors were carried out. The analgesic autoinjector and cartridges cleared all the above quality checks. Usage of an analgesic autoinjector in the albino rats by intraperitoneal injection showed an absence of mortality in any of the experimental categories. There was no marked difference in animal feed and water consumption and in the gross behaviour of experimental animals. When the rats were autopsied after euthanasia, there was an absence of bleeding or abdominal injury in any of the categories.
The volume of drug released through this self-injectable device was about 1.25 + 0.05 mL, and the dose was about 0.352 + 0.021 mg/kg and 0.678 + 0.040 mg/kg of 50 μg/mL and 100 μg/mL cartridges, respectively. The corresponding doses of opioid analgesic released by manual injection were 0.336 + 0.012 mg/kg and 0.649 + 0.015 mg/kg, respectively. Using the restrictor in the autoinjector, the needle length was adjusted for effective delivery, and it was around 1.5 mm.
The blood GSH level in the control was 20.5 + 2.0 mg/dL, and in the experimental albino rat, a significant decline in GSH levels was not observed. Whereas, in the research group of the 14-day study with the 50 μg/mL cartridge, GSH level in both the autoinjector and manual groups exhibited a marked increased level. The MDA levels in the control group were 2.23 + 0.19 ng/mL, and there was also no remarkable increase in any of the groups. Whereas in a 7-day study with a drug cartridge of 50 μg/mL, MDA concentration did not show any notable decrease in both autoinjector and manual groups (Figure 2).
 |
Figure 2: Blood GSH and MDA levels in control, A and M groups using two concentrations of drug cartridges (5 0μg/ mL and 100 μg/mL) and two durations, 7 d and 14 d. Mean + SE (n = 6). A: autoinjector; M: manual; d: day |
The liver and kidney GSH concentrations were not altered. However, in the manual group of 100 μg/mL with the study duration of 7 days, there is an increase in liver GSH values. A significant proliferation was exhibited in kidney GSH levels for the autoinjector group in a 7-day study with 50 μg/mL. There were no significant modifications seen in liver MDA values; meanwhile, for 100 μg/mL in manual drug administration of a 7-day study duration, the kidney MDA values showed a significant increase (Figure 3).
 |
Figure 3: MDA and GSH levels of liver and kidney in control, autoinjector (AJI) and manual(M) groups using two concentrations of drug cartridges (50μg/mL and 100μg/mL) for 7-day duration. + SE (n = 6) |
The SOD and catalase antioxidant levels in blood were modified with a notable increase in the experimental groups for the dosage of 50 μg/mL in 7 and 14 days of research duration in the self-injectable device (autoinjector) and manual group, respectively (Figure 4).
 |
Figure 4: Blood SOD and catalase levels in control, A and M groups using two concentrations of drug cartridges (0.05 and 0.10 μg/mL) and two durations, 7 d and 14 d. Mean + SE (n = 6). A: autoinjector; M: manual; d: day. |
The self-injectable device group and the manual group of 50 μg/mL cartridges for 14 days showed a marked raise in the values of blood GPX levels (Figure 4). The glutathione reductase antioxidants confirmed the absence of significant variations in any of the experimental groups (Figure 5).
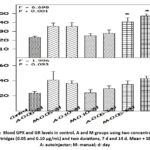 |
Figure 5: Blood GPX and GR levels in control, A and M groups using two concentrations of drug cartridges (0.05 and 0.10 μg/mL) and two durations, 7 d and 14 d. Mean + SE (n = 6). A: autoinjector; M: manual; d: day |
Discussion
The current experimental work was done to know the reliability and efficiency of buprenorphine when administered through a self-injectable autoinjector device. The drug dispensed through this technique requires pressure and it is been given continuously for 7 and 14 days. The autoinjectors intended for intramuscular administration were tested by intraperitoneal administration in albino rats. The reliability in using this device was studied by confirming the antioxidant enzymes in rats. The novel idea behind the analgesic autoinjector development was for their reliability in patients’ compliance during chronic treatment and during contingency in order to reduce the fatalities and further complications. The analgesic autoinjector developed is recyclable with changeable cartridges. The preferable route for the drug to be dispensed during injuries would be intramuscular route for better drug absorption because of hypotension due to blood loss. The analgesic autoinjector can be preferred in adults and children by selecting the dose.
Free radicals are released during physical or psychological stain by external or internal stimuli. There are drugs that can increase or decrease the release of these highly reactive ions. Dopamine, propofol, dobutamine, noradrenaline, captopril, fluvastatin, atorvastatin, topiramate, and zonisamide are some of the drugs that increase the antioxidant capacity.24 Drugs like phenytoin, valproic acid, carbamazepine25, doxorubicin, 5-fluorouracil, cyclophosphamide26, and opium drugs like morphine, heroin, and buprenorphine reduced the antioxidant level when they were used for long-term therapy. Genotoxicity was evident in morphine and noscapine, whereas in buprenorphine and pentazocine it was absent.27
Oxidative stress is produced when the reactive oxygen species levels have been markedly increased and the antioxidants are released to neutralize these reactive ions. The balance between the antioxidant enzymes and the reactive ions can be disturbed by the psychological stress in humans and the restrain stress in rats.28 SOD, catalase, glutathione peroxidase plays a pivotal role in defending these reactive species.29 In this experimental study, SOD and catalase levels have raised in autoinjector and manual medicament dispense groups with same dose of 50μg/mL but with different study duration. In autoinjector, it was in 7 days study duration and in manual group it was 14 days study duration (Figure 4).
GPX is a stable enzyme, and its activity is directly explained by the content of GSH since it is a substrate and cofactor of GPX.30,31 Glutathione is the earliest catalyst released to overcome the excess release of highly unstable ions. The GR levels were not altered in all the experimental categories, whereas the GPX was elevated in both experimental drug administration group (50μg/mL,14 days) (Figure 5).
The decrease in concentration of GSH is an index for the liver damage.32 The present experimental study has shown a marked raise in liver and kidney GSH levels ,when the drug was given in conventional intraperitoneal injection having higher dose and in self-injectable device having lesser dose (Figure 3). Blood GSH levels have elevated in both the self-injectable device and conventional injection groups having same dose of 50 μg/mL and study duration of 14-day (Figure 2).
Malondialdehyde is a compound released from polyunsaturated fatty acid peroxidation and arachidonic acid metabolism. Lipid peroxidation is an indicator for oxidative stress in which membrane lipids of plants and animals’ break down, and during this, the most abundant unstable compound is MDA. Due to this, measuring MDA is considered to be an indicator for lipid peroxidation. Many studies supported the increase in MDA content as a sign of major depressive disorder.32 In the present experimental study, there was no significant raise in MDA level; instead, there was a decline in MDA level in both the experimental groups for the study period of 7 days with the dosage of 50 μg/mL when compared with control (Figure 3).
The buprenorphine autoinjector did not produce any adverse effect on various haematological and biochemical parameters when administered intraperitoneally to rats for 7 and 14 days.11 The oxidative stress enzymes like superoxide dismutase, catalase and glutathione peroxidase have shown the difference in their proportions for both the autoinjector and manual drug administration groups. Thus, the self-injectable device can be used to manage the pain during extremity conditions.
Conclusion
The newly developed opioid analgesic autoinjector proved to be tolerable and reliable in albino rats by producing no damage to essential organs. The dose selection facility in the developed analgesic autoinjector can find its benefit even in children and also in farm and pet animals. The present research strengthens their suitability for usage in individuals during any disaster and for mass casualty management. Thus, the research on the opioid analgesic through the self-injectable device even encourages to extent the concept in producing different drug cartages for managing the chronic pain. Thus, the present experimental research can be extended in humans for their reliability and tolerability.
Acknowledgment
The authors thank M/s Neon Laboratories for providing water-filled cartridges. I also thank Pharmacology Department of Saveetha Medical College and Hospital who shared my departmental work during my research work.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
The present study was carried after the approval from Institutional Ethical Committee of Saveetha Medical College and Hospital (SU/BRULAC/RD/001/2013).
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Permission to reproduce material from other sources
Not Applicable
Clinical Trial Registration
This research does not involve any clinical trials.
Author Contributions
- Sheela Durairajan : Data Collection, Analysis, Writing
- Preetha Selva: Review & Editing the writing
- Rajagopalan Vijayaraghavan: Conceptualization, Methodology,
- Senthil Kumar Sivanesan: Supervision of project
- Srinivasan Vengadassalaapathy : Statistical Analysis
References
- Jha MK. Modified release formulations to achieve the quality target product profile (QTPP). Int J Pharm Sci Res. 2012;3(8):2376.
- Jitendra PK, Bansal S, Banik A. Noninvasive routes of proteins and peptides drug delivery. Indian J Pharm Sci. 2011;73(4):367.
- Zimmer PA, Braun LT, Fraser R, Hecht L, Kelliher F. Promoting success in self-injection: listening to patients. MedSurg Nurs. 2015;24(4):279.
- Makwana S, Basu B, Makasana Y, Dharamsi A. Prefilled syringes: An innovation in parenteral packaging. Int J Pharm Investig. 2011;1(4):200.
CrossRef - Thompson I, Lange J. Pen and autoinjector drug delivery devices. In: Sterile Product Development. Springer; 2013:331-356. doi:10.1007/978-1-4614-7978-9_13
CrossRef - Lange J, Richard P, Bradley N. Usability of devices for self-injection: results of a formative study on a new disposable pen injector. Med Devices (Auckl). 2014;7:195-203.
CrossRef - Lavorini F. The challenge of delivering therapeutic aerosols to asthma patients. ISRN Allergy. 2013;2013:1-17.
CrossRef - Ehdaie B. Enhanced delivery of transdermal drugs through human skin with novel carriers. J Pharm Biomed Sci. 2011;1(8):161-166.
- Lange J, Richard P, Bradley N. Usability of a new disposable autoinjector platform device: results of a formative study conducted with a broad user population. Med Devices (Auckl). 2015;8:255-262.
CrossRef - Raffa RB, Taylor R, Pergolizzi JV, et al. Application of human factors engineering (HFE) to the design of a naloxone auto-injector for the treatment of opioid emergencies. Drug Deliv Transl Res. 2017;7(1):1-10.
CrossRef - Sheela D, Vijayaraghavan R, Senthilkumar S. A study on the safety evaluation of buprenorphine administered through an autoinjector compared with manual injection using haematological and biochemical variables in rats. Hum Exp Toxicol. 2017;36(9):901-909.
CrossRef - Schiff M, Koo J, Jin E, et al. Usability and acceptability of the abatacept pre-filled autoinjector for the subcutaneous treatment of rheumatoid arthritis. Adv Ther. 2016;33(2):199-213.
CrossRef - Roy A, Vijayaraghavan R, Geetha RV, et al. A comparative study of the effect of amikacin administered through autoinjector and manual injection on biochemical parameters in rats. J Appl Pharm Sci. 2016;6(2):109-114.
CrossRef - Sheela D, Geetha RV, Krishna Mohan S, Vijayaraghavan R. A concept on the development of buprenorphine autoinjector for self and emergency administration. Int J Pharm Pharm Sci. 2015;7:253-257.
- Geetha RV, Roy A, Senthilkumar S, Bhaskar AS, Vijayaraghavan R. A Concept of a Probable Autoinjector for Bio-threat Agents. Def Sci J. 2016;66(5):521-524.
CrossRef - Pham-Huy LA, He H, Pham-Huy C. Free radicals, antioxidants in disease and health. Int J Biomed Sci. 2008;4(2):89-96.
CrossRef - Song TT, Nelson MR, Chang JH, Engler RJ, Chowdhury BA. Adequacy of the epinephrine autoinjector needle length in delivering epinephrine to the intramuscular tissues. Ann Allergy Asthma Immunol. 2005;94(5):539-542.
CrossRef - Kakkar P, Das B, Viswanathan PN. A modified spectrophotometric assay of superoxide dismutase. Indian J Biochem Biophys. 1984;21(2):130-132.
- Krishna RK, Krishnakumar S, Chandrakala S. Evaluation of antioxidant properties of different parts of Amorphophallus commutatus, an endemic aroid of Western Ghats, South India. Int J Biomed Sci. 2012;3(3):443-455.
- Divakaran N. Responses of antioxidants and lipid peroxidation in Villorita cyprinoides to mercury exposure. Int J Innov Res Dev. 2014;3(9):38-44.
- Mitra EL, Ghosh DE, Ghosh AK, et al. Aqueous Tulsi leaf (Ocimum sanctum) extract possesses antioxidant properties and protects against cadmium-induced oxidative stress in rat heart. Int J Pharm Pharm Sci. 2014;6(1):500-513.
- Parayanthala Valappil M, Santhakumar S, Arumugam S. Determination of oxidative stress related toxicity on repeated dermal exposure of hydroxyapatite nanoparticles in rats. Int J Biomater. 2014;2014:1-8.
CrossRef - Noeman SA, Hamooda HE, Baalash AA. Biochemical study of oxidative stress markers in the liver, kidney and heart of high fat diet-induced obesity in rats. Diabetol Metab Syndr. 2011;3(1):17.
CrossRef - Bucalen CF. Effects of xenobiotics on total antioxidant capacity. Interdiscip Toxicol. 2012;5(3):117-122.
CrossRef - Nazıroğlu M, Yürekli VA. Effects of antiepileptic drugs on antioxidant and oxidant molecular pathways: focus on trace elements. Cell Mol Neurobiol. 2013;33(5):589-599.
CrossRef - Singh G, Maulik SK, Jaiswal A, Kumar P, Parshad R. Effect on antioxidant levels in patients of breast carcinoma during neoadjuvant chemotherapy and mastectomy. Malays J Med Sci. 2010;17(2):24-31.
- Puli LK, Patil PA. Genotoxic evaluation of morphine, buprenorphine, pentazocine, and noscapine by micronucleus and comet assay in albino mice. Indian J Pharmacol. 2007;39(6):265-269.
CrossRef - Ighodaro OM, Akinloye OA. First line defence antioxidants—superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX): Their fundamental role in the entire antioxidant defence grid. Alexandria J Med. 2018;54(4):287-293.
CrossRef - Zafir A, Banu N. Induction of oxidative stress by restraint stress and corticosterone treatments in rats. Indian J Biochem Biophys. 2009;46:53-58.
- Rahbani-Nobar ME, Rahimi-Pour A, Rahbani-Nobar M, Adi-Beig F, Mirhashemi SM. Total antioxidant capacity, superoxide dismutase and glutathione peroxidase in diabetic patients. Med J Islam Acad Sci. 1999;12(4):109-114.
- Nwogueze BC, Ojieh AE, Aloamaka CP, Igweh JC, Onyesom I. Levels of glutathione-related antioxidants in some tissues of stressed Wistar rats. Indian J Physiol Pharmacol. 2021;65(3):167-176.
CrossRef - Nagasaka H, Inoue I, Inui A, et al. Relationship between oxidative stress and antioxidant systems in the liver of patients with Wilson disease: hepatic manifestation in Wilson disease as a consequence of augmented oxidative stress. Pediatr Res. 2006;60(4):472-477.
CrossRef - Bajpai A, Verma AK, Srivastava M, Srivastava R. Oxidative stress and major depression. J Clin Diagn Res. 2014;8(12):CC04.







