Manuscript accepted on :23-05-2025
Published online on: 13-06-2025
Plagiarism Check: Yes
Reviewed by: Dr. Rajendra Jangde
Second Review by: Dr. Sharath B S
Final Approval by: Dr. Patorn Piromchai
Ni Wayan Tianing1* , I Wayan Sudarsa2
, I Wayan Sudarsa2 and Ni Putu Sriwidyani3
and Ni Putu Sriwidyani3
1Department of Biochemistry, Faculty of Medicine, Udayana University, Bali, Indonesia.
2Surgical Oncology Division, Department of Surgery, Faculty of Medicine, Udayana University/Prof Ngoerah General Hospital, Bali, Indonesia.
3Department of Anatomical Pathology, Faculty of Medicine, Udayana University/ Prof Ngoerah General Hospital, Bali, Indonesia.
Corresponding Author E-mail: wtianing@yahoo.com
DOI : https://dx.doi.org/10.13005/bpj/3197
Abstract
Metastatic breast cancer is a serious health problem with a high mortality rate. Matrix metalloproteinase-9 (MMP-9) gene polymorphisms (rs17576) and Vascular Endothelial Growth Factor (VEGF) expressions enhance the process of angiogenesis and the risk of metastatic breast cancer. However, there is still a lack of studies on this topic. Therefore, the purpose of the study was to evaluate the association between MMP-9 gene polymorphism and VEGF levels with the risk of metastasis in breast cancer patients. This is a cross-sectional study involving breast cancer patients in Bali. The Polymerase Chain Reaction and DNA sequencing techniques are used to analyze the MMP-9 gene polymorphisms (rs17576), while the ELISA method is used to measure the VEGF levels. The data was then analyzed using SPSS ver.26. This study involved 64 breast cancer patients. The median (IQR) of the VEGF level is 85.17 (57.82 – 124.73) pg/mL. It was found as much as 42.2% of the patients experienced MMP-9 gene polymorphism. Receiver operating curve (ROC) analysis found the optimum cut-off of VEGF level is 98.11 pg/mL with a sensitivity of 66.7% and specificity of 81.4% (p=0.007). This study found that breast cancer patients with high VEGF levels ( 98.11 pg/mL) (p=0.001; AOR: 8.93; 95%CI: 2.41 – 33.08) have a higher risk of metastasis. In conclusion, breast cancer patients with high VEGF levels have a higher risk of developing metastasis.
Keywords
Breast cancer; MMP-9 gene; Metastasis; Polymorphism; VEGF
Download this article as:| Copy the following to cite this article: Tianing N. W, Sudarsa I. W, Sriwidyani N. P. High Vascular Endothelial Growth Factor (VEGF) Level and Matrix Metalloproteinase-9 (MMP-9) Gene Polymorphism (rs17576) Increase Metastasis Risk in Breast Cancer: A Cross-sectional Study. Biomed Pharmacol J 2025;18(2). |
| Copy the following to cite this URL: Tianing N. W, Sudarsa I. W, Sriwidyani N. P. High Vascular Endothelial Growth Factor (VEGF) Level and Matrix Metalloproteinase-9 (MMP-9) Gene Polymorphism (rs17576) Increase Metastasis Risk in Breast Cancer: A Cross-sectional Study. Biomed Pharmacol J 2025;18(2). Available from: https://bit.ly/4dXpqJc |
Introduction
Breast cancer is the most common cancer in women worldwide and has become a global problem in both developed and developing countries. In Indonesia, breast cancer ranks first in women and is generally diagnosed at an advanced stage with an increasing incidence of more than 5% per year.1,2 Factors that affect the clinical outcome of breast cancer are known such as; age, tumor size, clinical stage, metastatic status, histopathological stage, genetic information, and the role of angiogenesis factors. Research and methods of treatment and care have been carried out but the mortality rate of breast cancer remains high, this is mainly due to the process of breast cancer metastasis has not been pursued continuously.3–5
Progression and metastasis in cancer are influenced by angiogenesis factors. In normal cells, physiologically angiogenesis plays a role in the synthesis of new capillary blood vessels which are the basis for the process of reproduction, development, and tissue repair by carrying oxygen and nutrients and cleaning injured tissue.6,7 This occurs because of the balance between pro and antiangiogenic factors, which involve several cells and cytokines.3,5 In pathological conditions such as cancer, angiogenesis is a major requirement for cancer growth, invasion, and metastasis.5 Cancer cells can also secrete acidic or basic fibroblast growth factors (bFGF, aFGF) which increase endothelial cell proliferation and the formation of new capillaries. MMPs can also be produced by tumor cells, endothelial cells, or stromal cells (fibroblasts and inflammatory cells). Blood vessel proliferation is required for the growth and metastasis of cancer cells and hematologic malignancies.3,8
Analysis of angiogenesis ability plays a role in determining the prognosis and management of cancer patients.2,3,9,10 Research on breast cancer has shown that angiogenesis correlates with prognosis. The number of blood vessels in the specimen correlates with the tumor stage and can predict prognosis independently. Cancer cells can release several substances as proangiogenic factors such as Vascular Endothelial Growth Factor (VEGF), endothelin, Fibroblast Growth Factor (FGF), Transforming Growth Factor-β (TGF-β), or other substances as antiangiogenic factors such as endostatin, angiostatin, thrombospondin-1, Tissue Inhibitor of Metalloproteins (TIMPs).3,10 Vascular Endothelial Growth Factor is an active homodimeric protein, a member of platelet-derived growth factor (PDGF). VEGF is a Vascular Permeability Factor (VPF) and a specific mitogen for endothelial cells because the tyrosine kinase receptors, namely Kinase Domain Receptor (KDR or VEGFR-2), are exclusively expressed in endothelial cells.11
VEGF stimulation plays a role in angiogenesis. Angiogenesis and neovascularization play an important role in tumor progression and metastasis.2,3,5 VEGF expression has been detected in various types of cancer and proves it has a very important role in tumor biology and carcinogenesis processes, especially related to cancer growth and invasion.2,5 Examination of VEGF in cancer tissue by immunohistochemistry or immunoassay methods shows a significant correlation with MVD. The discovery of various angiogenesis markers secreted into the circulation or other body fluids is important because the measurement is more objective, faster, and does not require invasive procedures. Angiogenesis factors such as VEGF can be measured through blood, urine, or cerebrospinal fluid in cancer patients.3
Several studies have been conducted to evaluate VEGF levels with diagnosis, growth, metastasis, recurrence, survival, and prognosis in breast cancer, but there is still disagreement in the results. Several studies have shown an increase in VEGF levels along with the stage and prognostic factors. VEGF levels in stage IV breast cancer patients increase significantly when compared to stage I, II, and III breast cancer. VEGF levels increase along with increasing tumor size and breast cancer stage. VEGF levels can be used as a prognostic marker to determine recurrence and metastasis of breast cancer, but do not play much role in the early stages of the disease so they do not play a role in disease screening.12 Studies that have been conducted have shown that serum VEGF-C levels are not related to axillary lymph node metastasis and clinicopathological factors. Serum VEGF-C levels increase significantly in early-stage breast cancer.3,12 In Indonesia, research on the relationship between VEGF levels and metastasis involving genes is still very limited, therefore, researchers want to conduct this study in breast cancer and involve genes related to the process of angiogenesis and breast cancer metastasis such as the MMP-9 gene.
Materials and Methods
Study design
This study was a cross-sectional study. Sampling was carried out at Prof IGNG Ngoerah Hospital, Denpasar, while the analysis of MMP-9 gene polymorphism (rs17576) and VEGF level was carried out at the Biochemistry Laboratory, Faculty of Medicine, Udayana University.
Sample and Population
This study included female breast cancer patients who were treated at Prof IGNG Ngoerah Hospital, Denpasar, and underwent surgery or biopsy in 2018. The inclusion criteria in this study are age 35-65 years are willing to be a sample by signing the informed consent. The samples were selected by consecutive sampling. Meanwhile, the exclusion criteria in this study are patients with comorbidities of diabetes mellitus, stroke, and infarction.
Variables and Measurement
The data regarding age, stage, tumor size, nodal status, and metastasis status were collected through medical records. The patient blood sample was collected as much as 7 mL. Three milliliters of blood were then centrifuged at 3000 rpm for 5 minutes to separate the serum and cell components of the samples. The ELISA techniques were used to measure VEGF levels using serum samples that were previously described. The rest 4 mL of blood then underwent DNA isolation followed by Polymerase Chain Reaction (PCR) using MMP-9 gene (rs17576) primer F: 5’-ATGGGTCAAAGAACAGGA-3’ and R: 5’-GGTAGACAGGGTGGAGG-3’. The process then continued to sequencing to analyze the MMP-9 gene polymorphisms (rs17576).
Data Analysis
The data were analyzed using SPSS ver.26. The descriptive analysis was used to determine age and other clinicopathological factors. Receiver operating curve (ROC) analysis was used to determine the best cut-off value for VEGF levels. Bivariate analysis using chi-square test and multivariate analysis using logistic regression were used to analyze the differences between VEGF levels and MMP-9 gene polymorphism with metastasis status of breast cancer patients. P value <0.05 was declared significant.
Results
This study included 64 patients, with a median (IQR) age of 46 (42-51) years. Based on tumor size, patients with T4b (53.1%) had the highest proportion, followed by T4c (15.6%), and T4a (14.1%). Based on the status of tumor spread to the lymph nodes (N), the highest proportion was N1 (48.4%), followed by N2 (21.9%), and N0 (18.8%). A total of 67.2% of patients were found to have no metastasis, while the rest (32.8%) had metastasis. Based on stage, patients with advanced stage (stage IV) had the highest proportion compared to patients with stage II (10.9%) and stage III (4.3%) (Table 1).
Table 1: Baseline characteristics of the samples
| Variables | N=64 |
| Age, (years) median (IQR) | 46 (42 – 51) |
| T, n (%) | |
| T2 | 6 (9.4%) |
| T3 | 4 (6.3%) |
| T4a | 9 (14.1%) |
| T4b | 34 (53.1%) |
| T4c | 10 (15.6%) |
| T4d | 1 (1.6%) |
| N | |
| N0 | 12 (18.8%) |
| N1 | 31 (48.4%) |
| N2 | 14 (21.9%) |
| N3 | 7 (10.9%) |
| M | |
| M0 | 43 (67.2%) |
| M1 | 21 (32.8%) |
| Stage | |
| II | 7 (10.9%) |
| III | 3 (4.3%) |
| IV | 54 (84.4) |
| MMP-9 polymorphism | |
| Positive | 27 (42.2%) |
| Negative | 37 (57.8%) |
| VEGF level, (pg/mL) | 85.17 (57.82 – 124.73) |
VEGF: vascular endothelial growth factor; MMP-9: matrix metalloproteinase 9
The results of the ROC test of the VEGF level variable against metastasis status can be seen in Figure 1. From the ROC test, it was found that with a cut-off value of 98.11 pg/mL, a sensitivity of 66.7%, a specificity of 81.4%, with an area under the curve (AUC) value of 0.708 (p: 0.007; 95%CI: 0.55 – 0.86) (Table 2).
Table 2: ROC analysis of VEGF level
| Cut-off value | Sensitivity | Specificity | AUC | p-value | 95%CI |
| 98.11 | 66.7% | 81.4% | 0.708 | 0.007 | 0.55 – 0.86 |
AUC: area under the curve; CI: confidence interval
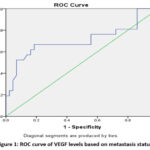 |
Figure 1: ROC curve of VEGF levels based on metastasis status |
Based on metastasis status, it was found that 66.7% of patients with high VEGF levels experienced metastasis, more than patients with low VEGF levels (33.3%). Meanwhile, patients without metastasis were dominated by patients with low VEGF levels (81.4%). These results were statistically significantly different (p<0.001; OR: 3.81; 95%CI: 1.81 – 8.05). Similar results were also found in the MMP-9 polymorphism variable, where patients with metastasis were dominated by patients with positive polymorphism (61.9%), while non-metastatic patients were more patients without MMP-9 polymorphism (67.4%). These results were statistically significantly different (p: 0.026; OR: 2.22; 95%CI: 1.07 – 4.61) (Table 3).
No statistically significant differences were observed in the distribution of age, tumor size, nodal status, or cancer stage between the two groups. Although patients aged ≥60 years were only found in the second group (11.6%), this did not reach statistical significance (p = 0.163; OR = 1.55; 95% CI: 1.28–1.87). The majority of tumors in both groups were categorized as T4 (Group 1: 85.7%; Group 2: 83.7%), with a non-significant association (p = 1.000; OR = 1.16; 95% CI: 0.26–5.05). Nodal status (≥N1 vs N0) was also comparable between groups (p = 0.736; OR = 1.58; 95% CI: 0.38–6.61), with most patients presenting with nodal involvement. Advanced-stage cancer was predominant in both groups (Group 1: 95.2%; Group 2: 79.1%); however, the difference was not statistically significant (p = 0.146; OR = 5.29; 95% CI: 0.62–44.93). (Table 3).
Table 3: Bivariate analysis of VEGF level and MMP-9 polymorphism with metastasis status
| Variables | Metastasis Status | p-value | OR | 95%CI | |
| Metastasis | Non-Metastasis | ||||
| VEGF level | |||||
| High ( 98.11 pg/mL) | 14 (66.7%) | 8 (18.6%) | <0.001* | 3.81 | 1.81 – 8.05 |
| Low (<98.11 pg/mL) | 7 (33.3%) | 35 (81.4%) | |||
| MMP-9 polymorphism | |||||
| Positive | 13 (61.9%) | 14 (32.6%) | 0.026* | 2.22 | 1.07 – 4.61 |
| Negative | 8 (38.1%) | 29 (67.4%) | |||
| Age | |||||
| 60 years old | 0 (0%) | 5 (11.6%) | 0.163 | 1.55 | 1.28 – 1.87 |
| <60 years old | 21 (100%) | 38 (88.4%) | |||
| Tumor size | |||||
| T4 | 18 (857%) | 36 (83.7%) | 1.000 | 1.16 | 0.26 – 5.05 |
| T2-T3 | 3 (14.3%) | 7 (16.3%) | |||
| Nodal status | |||||
| N1 | 18 (85.7%) | 34 (79.1%) | 0.736 | 1.58 | 0.38 – 6.61 |
| N0 | 3 (14.3%) | 9 (20.9%) | |||
| Stage | |||||
| Advanced stage | 20 (95.2%) | 34 (79.1%) | 0.146 | 5.29 | 0.62 – 44.93 |
| Early stage | 1 (4.8%) | 9 (20.9%) | |||
VEGF: vascular endothelial growth factor; MMP-9: matrix metalloproteinase 9; OR: odd risk; CI: confidence interval; *: statistically significant
In the multivariate logistic regression analysis, high VEGF levels (≥98.11 pg/mL) were found to be significantly associated with the outcome of interest, with an adjusted odds ratio (aOR) of 8.93 (95% CI: 2.41–33.08; p = 0.001), indicating that patients with elevated VEGF levels had nearly nine times higher odds compared to those with lower levels. Although the presence of MMP-9 polymorphism showed a trend toward significance (aOR = 3.45; 95% CI: 0.93–12.70; p = 0.062), this association did not reach statistical significance. Similarly, the advanced cancer stage was not significantly associated with the outcome (aOR = 5.37; 95% CI: 0.47–60.47; p = 0.173). Interestingly, age ≥60 years was not a significant predictor (p = 0.999) and displayed an anomalously low odds ratio (<0.01; 95% CI: 0.13–0.35), likely due to model instability or data sparsity for that subgroup (Table 4).
Table 4: Multivariate analysis of VEGF level and MMP-9 polymorphism with metastasis status
| Variables | B | p-value | Adjusted OR | 95%CI |
| High VEGF level ( 98.11 pg/mL) | 2.18 | 0.001* | 8.93 | 2.41 – 33.08 |
| MMP-9 polymorphism positive | 1.23 | 0.062 | 3.45 | 0.93 – 12.70 |
| Advanced stage | 1.68 | 0.173 | 5.37 | 0.47 – 60.47 |
| Age 60 years old | -20.07 | 0.999 | <0.01 | 0.13 – 0.35 |
VEGF: vascular endothelial growth factor; MMP-9: matrix metalloproteinase 9; OR: odd risk; CI: confidence interval; *: statistically significant
Discussion
Cancer metastasis in general and breast cancer, in particular, is very frightening, especially for patients, because it is the main cause of increased morbidity and risk of death in breast cancer. Feelings of worry may also be felt by doctors who treat patients because metastatic breast cancer requires more complex treatment than if the cancer is detected at an early stage and with a low cure rate. The discovery of early-stage cancer is a very important factor both in terms of treatment including prevention of metastasis and the cost of treatment. The cure rate is also higher if cancer is found at an early stage compared to if cancer is diagnosed as metastatic, which has a cure rate of around 30%.13,14
Breast cancer is influenced by many factors, one of which is hereditary (genetic) factors. MMP-9 gene promoter polymorphism is one of the genetic factors that plays a role in the occurrence of cancer metastasis. Several studies have reported a relationship between cancer metastasis and polymorphisms in the MMP-9 gene promoter, but some studies report no significant relationship. This difference is due to genetic variation because the studies analyzed were of different ethnicities. The high levels of VEGF in cases compared to controls prove that there is an increase in MMP-9 enzyme activity in cases, thus facilitating the formation or process of angiogenesis which will later facilitate breast cancer metastasis. The results of this analysis indicate a genetic role that influences the increase in VEGF levels.12,15,16
The results of the characteristic analysis of the samples analyzed in this study reflect that the median (IQR) age in this study is 46 (42-51) years old. This reflects that the age between 41-55 years is very risky for breast cancer. The risk of breast cancer increases in proportion to age. This is thought to be related to hormonal status, namely the length of exposure to estrogen and progesterone hormones that affect breast tissue proliferation and exposure to other risks that require a long time to induce cancer.17 This incident is closely related to late diagnosis caused by many factors such as patient ignorance about how to detect cancer early and the lack of health infrastructure for early detection screening of breast cancer.18,19
This study found that patients with T4b (53.1%), N1 (48.4%), and M0 (67.2%) were the highest proportion among others. This study also found that most of the patients (84.4%) came in the advanced stage (stage IV). This reflects that the awareness of patients in carrying out early detection is still lacking, in addition to the lack of knowledge about the risks of breast cancer if detected at an advanced stage is also still lacking. The lack of knowledge about metastatic cancer may be caused by a lack of counseling, understanding, and education in the community or a lack of infrastructure for early detection of breast cancer, especially in rural communities.20,21
As is known, tests for early detection are still centered in urban areas so most people are less aware of early detection or other screening tests. This is also due to limited funds and perhaps time because they have to go to the city.22 Seeing this reality, the government, both at the central and regional levels through the Health Service, must prioritize this handling through counseling and screening tests in the community. Another cause is that people know about the dangers of cancer if found at an advanced stage, but are still lazy to do early detection. Early detection of breast cancer can be done using mammography or other tests such as Ultrasonography (USG). Prevention of the metastasis process is the main goal of breast cancer management. Early detection is important, in addition to being able to find cancer in its early stages, it is also very important to ensure that patients get the right therapy.22
Detection of metastasis can now be done with chest X-ray, bone scan, Ultrasonography (USG), Magnetic Resonance Imaging (MRI), and Computed Tomography Scan (CT-Scan).23 However, until now, most of these examination techniques require expensive costs, and special skills and are only available in certain health service centers, so it is necessary to develop new biomarkers with easy-to-do examinations, for example with non-invasive and easily accessible blood tests to complement existing screening tools.21,22,24
This study found patients with metastasis have a higher proportion (66.7%) of high VEGF levels. These findings indicate that samples with metastasis cause enzyme activity to increase or become more active which can activate the angiogenesis process which is indicated by increased VEGF levels in cases. High VEGF levels facilitate the angiogenesis process so that they can increase and/or be a risk factor for metastasis.25–27 This study also found that patient with MMP-9 gene polymorphism has higher risk of metastasis compared to non-polymorphism patients. The results of this analysis prove that the MMP-9 gene plays a major role in the process of breast cancer metastasis.28
The results of this study further strengthen the relationship between the MMP-9 gene and VEGF to increase metastasis in cancer, especially breast cancer, because the presence of polymorphisms in the MMP-9 gene and also followed by high VEGF levels further increases the risk of cancer metastasis. The significant difference between MMP-9 levels in cases compared to controls indicates that polymorphisms in the MMP-9 gene cause the MMP-9 gene to be more expressive and MMP-9 enzyme activity to increase. Increased enzyme activity will affect the degradation of elastin and proteoglycan proteins so that it can increase the concentration of MMP-9 in these individuals. MMP-9 as gelatinase B or collagenase type IV, has a special role in the basement membrane, in the form of the ability to degrade collagen.15,28
MMP-9 also called matrixins is a Zn-dependent endopeptidase and is a major protease against the degradation of extracellular matrix (ECM) and degrades almost all components of the ECM and is an important stage for tumor growth, invasion, and metastasis.12 Invasion of the ECM is an active process consisting of several stages until the invasion and metastasis process.29 MMP-9 and MMP-2 are collagenases that can break down type IV collagen in the epithelium and basement membrane of blood vessels. These MMPs play a role in angiogenesis, growth, and motility of tumor cells. All MMPs have an active site containing Zn2+ and based on their structure and ability to degrade type IV collagen.12
MMP-9 is a key proteolytic enzyme that remodels the extracellular matrix (ECM) and plays an indirect role in promoting angiogenesis through the modulation of vascular endothelial growth factor (VEGF) activity. The MMP-9 gene polymorphisms can alter its expression and enzymatic activity, affecting the bioavailability of VEGF. MMP-9 acts by cleaving ECM components, which releases sequestered VEGF that stimulates angiogenic signaling in various tissues.30
Elevated levels of MMP-9 due to such polymorphisms have significant implications in tumor angiogenesis and other pathological neovascularization processes. For instance, in gastric carcinoma, patients with high MMP-9 mRNA and increased VEGF protein expression exhibit more aggressive tumor angiogenesis and poorer prognoses.30 Additionally, studies indicate that MMP-9-mediated cleavage of ECM components not only releases VEGF but also facilitates the migration of endothelial cells and the formation of new blood vessels.31 In cancerous tissues, hypoxia can further upregulate both MMP-9 and VEGF, creating a microenvironment conducive to an “angiogenic switch” critical for tumor growth and metastasis.32
In addition to its direct enzymatic role, MMP-9 polymorphisms have been implicated in gene-gene interactions that can influence the overall angiogenic phenotype. Interactions among polymorphisms in MMP-9, VEGF, and endothelial nitric oxide synthase (eNOS) have been observed in the context of childhood obesity, suggesting the genetic background can modulate angiogenesis through complex regulatory networks.33 These interactions highlight the multifaceted role of MMP-9 polymorphism not only as an independent variable affecting VEGF levels but also within broader genetic networks governing vascular remodeling in both physiological and pathological contexts.34
However, this study has several limitations such as limited sample size, only using cross-sectional design, and limited to single-center sampling. Also, since we only have limited time and resources, we cannot take samples through different stages of the patients. Future research addressing these limitations can help further validate the role of VEGF and MMP-9 gene polymorphisms in breast cancer metastasis and improve prognostic and therapeutic strategies.
Conclusions
In conclusion, this study demonstrates that high VEGF levels significantly increase the risk of metastasis in breast cancer patients. While MMP-9 gene polymorphism was also associated with metastasis, only VEGF levels remained statistically significant in multivariate analysis. These findings suggest a synergistic role of angiogenic markers and genetic predisposition in breast cancer progression. The results underscore the potential utility of VEGF as a non-invasive biomarker for metastasis risk stratification. Further research with larger, multi-center cohorts is recommended to validate these associations and explore targeted interventions.
Acknowledgment
The authors would like to thank the Biochemistry Department staff for helping with the ELISA and PCR procedures.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This study has received ethical clearance from the Ethics Commission of Medical Faculty, Udayana University with number 1649/UN14.2.2IPD|KEP 12018.
Informed Consent Statement
This study has received written informed consent from all participants.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not applicable
Author Contributions
- Ni Wayan Tianing: Conceptualization, Methodology, Writing – Original Draft.
- I Wayan Sudarsa: Data Collection, Analysis, Writing – Review & Editing.
- Ni Putu Sriwidyani: Data Collection, Analysis, Writing – Review & Editing.
References
- Schneider BP, Miller KD. Angiogenesis of Breast Cancer. Journal of Clinical Oncology. 2005;23(8):1782-1790. doi:10.1200/jco.2005.12.017
CrossRef - National Cancer Institute. Surveillance, Epidemiology, and End Results Program (SEER) Cancer Statistics Review. 2021.
- Fidler IJ, Ellis LM. Neoplastic Angiogenesis — Not All Blood Vessels Are Created Equal. New England Journal of Medicine. 2004;351(3):215-216. doi:10.1056/nejmp048080
CrossRef - Mirshahidi HR, Mirshahidi S, Abraham J. Can eribulin change the treatment algorithm in the management of breast cancer? Breast Cancer Manag. 2013;2(4):339-346. doi:10.2217/bmt.13.31
CrossRef - Bhoo Pathy N, Yip CH, Taib NA, et al. Breast cancer in a multi-ethnic Asian setting: Results from the Singapore–Malaysia hospital-based breast cancer registry. The Breast. 2011;20:S75-S80. doi:10.1016/j.breast.2011.01.015
CrossRef - Yayan J, Franke KJ, Berger M, Windisch W, Rasche K. Adhesion, metastasis, and inhibition of cancer cells: a comprehensive review. Mol Biol Rep. 2024;51(1):165. doi:10.1007/s11033-023-08920-5
CrossRef - Gerstberger S, Jiang Q, Ganesh K. Metastasis. Cell. 2023;186(8):1564-1579. doi:10.1016/j.cell.2023.03.003
CrossRef - Hughes S, Agbaje O, Bowen RL, et al. Matrix Metalloproteinase Single-Nucleotide Polymorphisms and Haplotypes Predict Breast Cancer Progression. Clinical Cancer Research. 2007;13(22):6673-6680. doi:10.1158/1078-0432.ccr-07-0884
CrossRef - Bayramoglu A, Gunes HV, Metintas M, Deǧirmenci I, Mutlu F, Alataş F. The association of MMP-9 enzyme activity, MMP-9 C1562T polymorphism, and MMP-2 and -9 and TIMP-1, -2, -3, and -4 gene expression in lung cancer. Genet Test Mol Biomarkers. 2009;13(5):671-678. doi:10.1089/gtmb.2009.0053
CrossRef - Fletcher CDM, World Health Organization., International Agency for Research on Cancer. WHO Classification of Tumours of Soft Tissue and Bone. IARC Press. 2013.
- Brogowska KK, Zajkowska M, Mroczko B. Vascular Endothelial Growth Factor Ligands and Receptors in Breast Cancer. J Clin Med. 2023;12(6). doi:10.3390/jcm12062412
CrossRef - Rybakowski JK. Matrix Metalloproteinase-9 (MMP9)—A Mediating Enzyme in Cardiovascular Disease, Cancer, and Neuropsychiatric Disorders. Cardiovasc Psychiatry Neurol. 2009;2009:1-7. doi:10.1155/2009/904836
CrossRef - Smolarz B, Zadrożna Nowak A, Romanowicz H. Breast Cancer—Epidemiology, Classification, Pathogenesis and Treatment (Review of Literature). Cancers (Basel). 2022;14(10). doi:10.3390/cancers14102569
CrossRef - Wang J, Wu SG. Breast Cancer: An Overview of Current Therapeutic Strategies, Challenge, and Perspectives. Breast Cancer: Targets and Therapy. 2023;15:721-730. doi:10.2147/BCTT.S432526
CrossRef - Grieu F, Li WQ, Iacopetta B. Genetic polymorphisms in the MMP-2 and MMP-9 genes and breast cancer phenotype. Breast Cancer Res Treat. 2004;88(3):197-204. doi:10.1007/s10549-004-0595-6
CrossRef - Tianing NW, Wibawa IBT, Artha A. C-1589T and G-1665A Polymorphisms Of Matrix Metalloprotein-9 Gene Promoter Increased Level Of Matrix Metalloproteinase-9 Enzyme As A Risk Factor Metastatic Of Breast Cancer In Balinese Tribe. Bali Medical Journal. 2016;5(1):80. doi:10.15562/bmj.v5i1.252
CrossRef - Takeiwa T, Mitobe Y, Ikeda K, Horie-Inoue K, Inoue S. Roles of splicing factors in hormone-related cancer progression. Int J Mol Sci. 2020;21(5). doi:10.3390/ijms21051551
CrossRef - Juranah J, Widaningsih Y, Hamdani W, Pakasi RD, Bahrun U. Interleukin-8 Levels in Breast Cancer. Indonesian Journal of Clinical Pathology and Medical Laboratory. 2014;20(3).
- Azamris A, Irwan I, Bachtiar H. Prognosis Comparison Molecular Subtype Breast Cancer between Young and Adult Women at General Hospital Dr. M Djamil Padang. Biomed J Sci Tech Res. 2017;1(6). doi:10.26717/bjstr.2017.01.000508
CrossRef - Shah R, Rosso K, David Nathanson S. Pathogenesis, prevention, diagnosis and treatment of breast cancer. World J Clin Oncol. 2014;5(3):283-298. doi:10.5306/wjco.v5.i3.283
CrossRef - Tahmooresi M, Afshar A, Rad BB, Nowshath KB, Bamiah MA. Early Detection of Breast Cancer Using Machine Learning Techniques. Journal of Telecommunication, Electronic and Computer Engineering. 2018;10(3):21-27.
CrossRef - Lim YX, Lim ZL, Ho PJ, Li J. Breast Cancer in Asia: Incidence, Mortality, Early Detection, Mammography Programs, and Risk-Based Screening Initiatives. Cancers (Basel). 2022;14(17):4218. doi:10.3390/cancers14174218
CrossRef - Park M, Kim D, Ko S, Kim A, Mo K, Yoon H. Breast Cancer Metastasis: Mechanisms and Therapeutic Implications. Int J Mol Sci. 2022;23(12). doi:10.3390/ijms23126806
CrossRef - Rosen EL, Blackwell KL, Baker JA, et al. Accuracy of MRI in the detection of residual breast cancer after neoadjuvant chemotherapy. American Journal of Roentgenology. 2003;181(5):1275-1282.
CrossRef - Panigoro SS, Muhammad, Kartini D, Andinata B, Sabarguna BS, Harahap AS. The potential role of MMP-9 and VEGF-C as predictors of lymph node involvement in papillary thyroid carcinoma. Bali Medical Journal. 2023;12(1):187-191. doi:10.15562/bmj.v12i1.3805
CrossRef - Verheul HM, Pinedo HM. The role of vascular endothelial growth factor (VEGF) in tumor angiogenesis and early clinical development of VEGF-receptor kinase inhibitors. Clin Breast Cancer. 2000;1 Suppl 1(September):S80-S84. doi:10.3816/cbc.2000.s.015
CrossRef - Carmeliet P. VEGF as a key mediator of angiogenesis in cancer. Oncology. 2005;69(SUPPL. 3):4-10. doi:10.1159/000088478
CrossRef - Huang H. Matrix metalloproteinase-9 (MMP-9) as a cancer biomarker and MMP-9 biosensors: Recent advances. Sensors (Switzerland). 2018;18(10). doi:10.3390/s18103249
CrossRef - Koboldt DC, Fulton RS, McLellan MD, et al. Comprehensive molecular portraits of human breast tumours. Nature. 2012;490(7418):61-70. doi:10.1038/nature11412
CrossRef - Yang Q, Ye ZY, Zhang JX, Tao HQ, Li SG, Zhao ZS. Expression of matrix metalloproteinase-9 mRNA and vascular endothelial growth factor protein in gastric carcinoma and its relationship to its pathological features and prognosis. Anatomical Record. 2010;293(12):2012-2019. doi:10.1002/ar.21071
CrossRef - Ebrahem Q, Chaurasia SS, Vasanji A, et al. Cross-talk between vascular endothelial growth factor and matrix metalloproteinases in the induction of neovascularization in vivo. American Journal of Pathology. 2010;176(1):496-503. doi:10.2353/ajpath.2010.080642
CrossRef - Chetty C, Bhoopathi P, Rao JS, Lakka SS. Abstract 3468: MMP-9 inhibition abolishes hypoxia-induced angiogenesis in medulloblastoma. Cancer Res. 2011;71(8_Supplement):3468-3468. doi:10.1158/1538-7445.am2011-3468
CrossRef - Belo V, Luizon M, Carneiro P, Souza-Costa D, Lanna C, Tanus-Santos J. Abstract 459: Gene-Gene Interaction Among Endothelial Nitric Oxide Syntase (eNOS), Matrix Metalloproteinase-9 (MMP-9) and Vascular Endothelial Growth Factor (VEGF) in the Childhood Obesity. Hypertension. 2012;60(suppl_1). doi:10.1161/hyp.60.suppl_1.a459
CrossRef - Bausch D, Pausch T, Krauss T, et al. Neutrophil granulocyte derived MMP-9 is a VEGF independent functional component of the angiogenic switch in pancreatic ductal adenocarcinoma. Angiogenesis. 2011;14(3):235-243. doi:10.1007/s10456-011-9207-3
CrossRef







