Manuscript accepted on :12-05-2025
Published online on: 20-05-2025
Plagiarism Check: Yes
Reviewed by: Dr. Hassan Shora
Second Review by: Dr. Adinarayana Andy
Final Approval by: Dr. Prabhishek Singh
Abid Shaheer 1* , Mahir Jallo 2
, Mahir Jallo 2 and Shahid Akhtar3
and Shahid Akhtar3
1Department of Biomedical Sciences, College of Medicine, Gulf Medical University, United Arab Emirates.
2Department of Internal Medicine, Thumbay University Hospital, United Arab Emirates.
3Department of Clinical Biochemistry, Thumbay Labs, United Arab Emirates.
Corresponding Author E-mail: abidshaheer@gmail.com
DOI : https://dx.doi.org/10.13005/bpj/3192
Abstract
The objective of the study was to find out the effect of semaglutide add-on to metformin on the visceral adiposity index and markers of visceral adipose tissue activity in type 2 diabetic patients based on body mass. A total of 90 patients with type 2 diabetes (both genders aged 30-60 years) were included in the study. The participants were divided into three groups based on their Body Mass Index (BMI), which includes the subjects with normal body weight (BMI between 18.5 - 24.9 kg/m2), overweight (BMI between 25 - 29.9 kg/m2) and obesity (BMI ≥30 kg/m2). Enzyme-linked immunosorbent assay was used to analyze serum leptin and adiponectin. Visceral Adiposity Index (VAI) was determined by the formula: Waist Circumference/(39.68 + [1.88 × BMI]) × Triglycerides/1.03 × 1.31/High-Density Lipoproteins. VAI showed minimal changes with metformin alone but decreased significantly with semaglutide add-on (p < 0.05), with the greatest reduction in the obesity group (-0.70). Leptin levels remained unchanged after metformin but significantly declined following semaglutide treatment (p < 0.05). The reduction was most pronounced in the obesity group (-5.10 ng/mL), followed by overweight (-3.40 ng/mL) and normal-weight individuals (-2.60 ng/mL). Adiponectin levels increased significantly post-semaglutide add-on therapy (p < 0.05). The greatest increase was observed in the obesity group (+3.70 μg/mL), followed by overweight (+3.30 μg/mL) and normal-weight participants (+3.30 μg/mL). Semaglutide presents a promising long-term solution for reducing the visceral adiposity index and enhancing metabolic health by modulating markers of visceral fat activity in patients with poorly controlled type 2 diabetes. This study highlights the importance of personalized treatment approaches, suggesting that semaglutide can be strategically utilized to prevent excess fat accumulation in the abdominal cavity.
Keywords
Adipokine; Adiponectin; Diabetes; Leptin; Obesity
Download this article as:| Copy the following to cite this article: Shaheer A, Jallo M, Akhtar S. Effect of Semaglutide Add-on to Metformin on Visceral Adiposity Index and Markers of Visceral Adipose Tissue Activity in Type 2 Diabetic Patients. Biomed Pharmacol J 2025;18(2). |
| Copy the following to cite this URL: Shaheer A, Jallo M, Akhtar S. Effect of Semaglutide Add-on to Metformin on Visceral Adiposity Index and Markers of Visceral Adipose Tissue Activity in Type 2 Diabetic Patients. Biomed Pharmacol J 2025;18(2). Available from: https://bit.ly/4k3LTXt |
Introduction
Visceral obesity is an independent risk factor involved in the pathogenesis of insulin resistance that leads to type 2 diabetes mellitus. Excessive visceral fat is associated with an increased cardiovascular disease risk due to variations in blood pressure, glucose and lipid levels.1 Visceral obesity advances diabetic risk via various adipokines and hence, effective therapeutic targets are necessary to manage obesity in high-risk individuals. The dysregulation of adipokine production contributes significantly to the development of obesity-related disorders and their associated complications.1,2 Abnormal levels of adipokines such as adiponectin and leptin are associated with metabolic syndrome and obesity. The serum leptin and adiponectin are altered with body weight and adiposity variations.3,4
The International Diabetes Federation estimates that the prevalence of type 2 diabetes mellitus in the Middle East currently stands at approximately 12.8%, with projections indicating an increase to 14.2% by 2030.5,6 A recent study in the Middle East shows that the prevalence of overweight and obesity is 48% and 24% in males and the incidence is 35% and 40% in females.7,8 Visceral adiposity is associated with metabolic and cardiovascular disturbances such as insulin resistance, type 2 diabetes and dyslipidemia.9 Estimating visceral adiposity is recommended in certain situations in routine clinical practice. Waist circumference and body mass index (BMI) are accurate clinical estimates of visceral adiposity. The visceral adiposity index (VAI) measurement is recently recommended to indicate insulin sensitivity and visceral adiposity that reflect cardiometabolic risk.9,10
Metformin is one of the best choices for first-line treatment for type 2 diabetes mellitus. However, the add-on therapy is recommended for better glycemic control in current clinical practice.11,12 Metformin lowers blood glucose in inadequately controlled type 2 diabetic patients by decreasing intestinal absorption of glucose, inhibiting hepatic gluconeogenesis and improving insulin sensitivity. Metformin enhances the adenosine monophosphate-activated protein kinase activity by liver kinase or inhibiting mitochondrial respiration and controlling adenosine monophosphate/adenosine triphosphate ratio. Metformin also works in glycerol metabolism by inhibiting glycerophosphate dehydrogenase.11,13,14
Semaglutide is a glucagon-like peptide-1 (GLP-1) receptor agonist currently used for second-line treatment in type 2 diabetic cases who are inadequately maintained with metformin.15,16 Semaglutide add-on therapy with sodium-glucose cotransporter 2 inhibitors improves glycemic control and body weight in poorly controlled type 2 diabetic patients and is generally well-tolerated.17,18 Semaglutide triggers the GLP-1 receptors that help to reduce the glucose-dependent secretion of glucagon, increase glucose-dependent insulin secretion, decrease gastric emptying and improve satiety.19,20
This study focused on the effect of semaglutide add-on to metformin on visceral adiposity index and biochemical markers of visceral adipose tissue activity in type 2 diabetic cases based on body mass.
Materials and Methods
This study included 90 type 2 diabetic subjects (both genders aged 30-60 years), identified with type 2 diabetes mellitus in Thumbay University Hospital and Clinics, collaborated with Thumbay Labs, Ajman, United Arab Emirates.
Inclusion criteria: Newly diagnosed type 2 diabetic subjects on first-line therapy with metformin and identified as inadequately controlled after 3 months of metformin monotherapy. The participants on the first-line medication (metformin-1000 mg twice daily) were monitored for 3 months and the poorly controlled subjects were recruited. The participants with add-on therapy with semaglutide injection (0.25 mg once weekly for the first 4 weeks, followed by 0.5 mg once weekly) were included.
Exclusion criteria: Subjects with type 1 diabetes mellitus, gestational diabetes mellitus, cardiovascular disease, diabetic nephropathy and diabetic ketoacidosis. Those who received different dosages of metformin, other hypoglycaemic treatments and statins were excluded from the study.
The subjects were divided into three groups based on their BMI, which includes the subjects with normal weight (BMI: 18.5 – 24.9 kg/m2), overweight (BMI: 25 – 29.9 kg/m2) and obesity (BMI: ≥30 kg/m2). Type 2 diabetes patients were diagnosed based on the criteria set by the American Diabetes Association (ADA).
Data were collected before the initiation of first-line metformin therapy, three months following the commencement of metformin treatment and three months after the addition of supplementary therapy. The glycemic control was observed by measuring Hemoglobin A1C (HbA1c), Fasting Plasma Glucose (FPG) and 2-hour Postprandial Blood Glucose (PPBG). The screening tests included were serum creatinine, albumin, alanine transaminase (ALT) and alanine transaminase (AST). Study participants who experienced a reduction in HbA1c levels greater than 1% or achieved an HbA1c level of less than 7% were excluded from the study. A questionnaire with written informed consent was used to collect information such as age, lifestyle, family history and therapeutic strategies. Measurements of waist circumference (WC), height, weight, systolic and diastolic blood pressure were documented.
The study was conducted for two years after approval from the Research Ethics Committee (IRB/COM/FAC/34/OCT-2022), conducted from November 2022 to October 2024. Informed written consent was received from all subjects and their confidentiality was strictly upheld throughout the study.
The samples collected from the participants were analyzed for Adiponectin and Leptin by Enzyme-Linked Immunosorbent Assay (ELISA) using the reagent kits supplied by Euroimmun. HbA1c, FPG, 2-hour PPBG, Triglycerides (TG), Total Cholesterol (TC), LDL Cholesterol (LDLC) and HDL Cholesterol (HDLC) were analyzed by Beckman Coulter Clinical Chemistry Analyzer. Body mass index (BMI) was calculated by weight (in kilograms) divided by height (in meter square). The VAI was determined using the following formula: Waist Circumference/(39.68 + [1.88 × BMI]) × Triglycerides/1.03 × 1.31/High-Density Lipoproteins.
The data are presented as mean ± standard deviation or frequencies, depending on the variable type. Parametric and non-parametric statistical tests were utilized for comparisons, based on the distribution and nature of the data. A z-test was employed to assess differences in means between groups. The effect of semaglutide on the anthropometric measures, VAI, diabetic profile, lipid profile and markers of visceral adipose tissue activity was evaluated using repeated measures analysis of covariance, with treatment as the independent variable and changes in HbA1c and age as covariates. Data analysis was conducted using the Statistical Package for the Social Sciences, Version 29.0, with a p-value of less than 0.05 considered statistically significant.
Results
Table 1 shows the baseline demographic, anthropometric and clinical characteristics of study participants, categorized by weight status (normal weight, overweight and obesity). A total of 90 individuals were included, with 30 participants in each of the normal weight, overweight and obesity groups. Among the total population, 52% were men and 48% were women, with a higher proportion of men in the normal weight group (60%) compared to the overweight (53%) and obesity (43%) groups. Smoking prevalence was highest in the overweight group (30%) and lowest in the normal weight group (10%). Hypertension and hypercholesterolemia were more common in individuals with obesity (77% and 70%, respectively) compared to those with normal weight (17% and 33%, respectively).
Anthropometric measurements showed a progressive increase in weight, BMI and waist circumference across the weight categories. The mean weight was highest in the obesity group (83.1 ± 7.5 kg) and lowest in the normal weight group (71.6 ± 7.2 kg). Similarly, BMI increased from 22.2 ± 2.4 kg/m² in normal-weight individuals to 33.2 ± 2.8 kg/m² in those with obesity. Blood pressure values also followed an increasing trend, with the obesity group having the highest systolic (126.1 ± 7.8 mmHg) and diastolic (84.1 ± 7.5 mmHg) readings, while the normal weight group had the lowest (107.4 ± 6.7 mmHg and 76.2 ± 5.8 mmHg, respectively). Overall, the data indicate that individuals with higher BMI tend to have increased cardiovascular risk factors, including hypertension and hypercholesterolemia, compared to those with normal weight.
Table 1: Baseline Demographic, Anthropometric and Clinical Characteristics of Study Participants
| Total | Normal Weight | Overweight | Obesity | |
| n | 90 | 30 | 30 | 30 |
| Men (n, %) | 47 (52%) | 18 (60%) | 16 (53%) | 13 (43%) |
| Women (n, %) | 43 (48%) | 12 (40%) | 14 (47%) | 17 (57%) |
| Smoking (n, %) | 14 (15%) | 3 (10%) | 9 (30%) | 5 (17%) |
| Hypertension (n, %) | 43 (47%) | 5 (17%) | 15 (50%) | 23 (77%) |
| Hypercholesterolemia (n, %) | 48 (53%) | 10 (33%) | 17 (57%) | 21 (70%) |
| Weight (kg) | 78.0±7.7 | 71.6±7.2 | 78.2±8.4 | 83.1±.7.5 |
| BMI (kg/m2) | 28.0±2.5 | 22.2±2.4 | 27.2±2.2 | 33.2±2.8 |
| WC (cm) | 93.0±5.2 | 87.1±4.7 | 94.7±4.8 | 98.2±6.1 |
| Systolic BP (mm/Hg) | 117±7.8 | 107.4±6.7 | 117.2±8.9 | 126.1±7.8 |
| Diastolic BP (mm/Hg) | 81±5.0 | 76.2±5.8 | 81.6±7.6 | 84.1±7.5 |
| Data are expressed as n (%), means ± SD.
VAI: Visceral Adiposity Index, BMI: Body Mass Index, WC: Waist Circumference, BP: Blood Pressure |
||||
Table 2 presents the impact of semaglutide add-on therapy on BMI, VAI and adiposity markers in type 2 diabetic subjects, categorized by weight status. The results were recorded at baseline, after 12 weeks of metformin treatment and after an additional 12 weeks of semaglutide therapy. BMI remained relatively unchanged following 12 weeks of metformin monotherapy in all groups. However, after the add-on therapy of semaglutide, a significant reduction in BMI was observed across all weight categories (p < 0.05). The most notable decrease was seen in individuals with obesity (-0.90 kg/m²), followed by overweight (-0.70 kg/m²) and normal-weight participants (-0.60 kg/m²). VAI levels showed minimal fluctuation after metformin therapy alone but significantly decreased following the addition of semaglutide (p < 0.05). The greatest reduction was observed in the obesity group (-0.70), while smaller yet significant changes were noted in the overweight (-0.50) and normal weight groups (-0.40).
Leptin levels, an indicator of adipose tissue function, did not show considerable changes after metformin treatment. However, semaglutide add-on therapy resulted in a significant decrease in leptin concentrations across all weight groups (p < 0.05). The reduction was most pronounced in the obesity group (-5.10 ng/mL), followed by overweight (-3.40 ng/mL) and normal-weight individuals (-2.60 ng/mL). Adiponectin, an anti-inflammatory adipokine, increased after semaglutide treatment in all groups, with significant improvements compared to baseline (p < 0.05). The greatest increase was observed in the obesity group (+3.70 μg/mL), followed by overweight (+3.30 μg/mL) and normal-weight participants (+3.30 μg/mL).
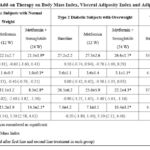 |
Table 2: Effects of Semaglutide Add-on Therapy on Body Mass Index, Visceral Adiposity Index and Adiposity Markers of Study Subjects |
Figure 1 to 4 illustrates the changes in BMI, VAI, leptin and adiponectin levels, respectively, across three groups of type 2 diabetic subjects (normal weight, overweight and obese) over three timepoints: baseline, after 12 weeks of metformin therapy and after 24 weeks of metformin with semaglutide. The results indicate the significant role of semaglutide in body weight and visceral fat regulation when used as an adjunct to metformin.
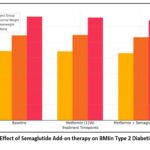 |
Figure 1: Effect of Semaglutide Add-on therapy on BMIin Type 2 Diabetic Subjects |
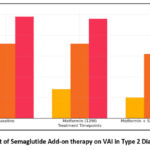 |
Figure 2: Effect of Semaglutide Add-on therapy on VAI in Type 2 Diabetic Subjects |
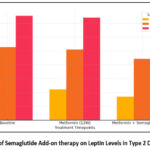 |
Figure 3: Effect of Semaglutide Add-on therapy on Leptin Levels in Type 2 Diabetic Subjects |
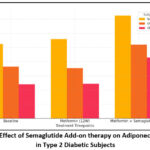 |
Figure 4: Effect of Semaglutide Add-on therapy on Adiponectic Levels in Type 2 Diabetic Subjects |
Table 3 demonstrates the impact of adding semaglutide to metformin therapy on various metabolic parameters in individuals with type 2 diabetes across different weight categories. The addition of semaglutide to metformin therapy led to significant improvements in metabolic parameters, particularly in the second phase of treatment (12 weeks to 24 weeks), as indicated by changes in glucose and lipid profiles across the different groups.
In the normal weight group, FPG decreased from 8.1 ± 1.1 to 7.0 ± 0.6 (p < 0.05), in the overweight group from 10.8 ± 1.3 to 9.3 ± 0.8 (p < 0.05) and in the obesity group from 13.5 ± 0.7 to 12.2 ± 1.2 (p < 0.05), indicating a substantial improvement in glucose control with the addition of semaglutide. PPBG levels also showed significant improvement in all groups after 24 weeks of semaglutide treatment. In the normal weight group, PPBG decreased from 11.8 ± 1.2 to 10.5 ± 1.8 (p < 0.05), in the overweight group from 14.9 ± 1.2 to 13.4 ± 2.2 (p < 0.05) and in the obesity group from 16.6 ± 1.2 to 15.1 ± 1.4 (p < 0.05). A significant reduction in both FPG and PPBG was observed after 24 weeks of semaglutide therapy across all weight groups. The greatest improvements were seen in individuals with overweight and obesity, indicating enhanced glycemic control.
The HbA1c was observed with significant reductions from 12 weeks to 24 weeks in both the normal weight and overweight groups. In the normal weight group, HbA1c decreased from 7.2 ± 0.3 to 6.8 ± 0.5 (p < 0.05) and in the overweight group, it decreased from 8.8 ± 0.8 to 7.9 ± 0.4 (p < 0.05). The obesity group also showed a significant decrease from 8.9 ± 0.5 to 8.0 ± 0.3 (p < 0.05). HbA1c levels declined significantly following semaglutide treatment, with a more pronounced effect in the overweight and obese groups. This suggests better long-term glucose regulation, reinforcing the role of semaglutide in diabetes management.
The addition of semaglutide also had a significant effect on TC in the normal weight group, where TC decreased from 5.3 ± 0.4 to 4.8 ± 0.6 (p < 0.05) after 24 weeks of treatment. LDLC was observed significant reductions were observed in all three groups from 12 weeks to 24 weeks. In the normal weight group, LDLC decreased from 3.2 ± 0.3 to 2.8 ± 0.7 (p < 0.05), in the overweight group from 4.2 ± 0.6 to 4.0 ± 0.7 (p < 0.05) and in the obesity group from 4.5 ± 0.4 to 4.2 ± 0.5 (p < 0.05). Also, TC: HDLC ratio showed a significant decrease from 12 weeks to 24 weeks in the overweight group, where the ratio dropped from 5.7 ± 0.6 to 5.3 ± 0.2 (p < 0.05) and in the obesity group, where it decreased from 6.0 ± 0.4 to 5.7 ± 0.4 (p < 0.05), indicating a more favourable lipid profile after 24 weeks of treatment with metformin and semaglutide. Overall, the data indicate that the addition of semaglutide to metformin therapy resulted in significant improvements in glucose control and lipid profiles, especially after the 12-week mark, with all marked changes having a p-value < 0.05.
Semaglutide treatment led to significant reductions in TC and LDLC, particularly in normal-weight and overweight individuals. The decrease in these markers suggests potential cardiovascular benefits associated with semaglutide use. No significant changes were observed in TG or HDLC levels across the groups. This indicates that semaglutide is effective on lipid metabolism and may be more pronounced in cholesterol regulation rather than triglycerides.
The TC/HDLC ratio, an indicator of cardiovascular risk, showed a significant reduction in overweight and obese individuals, further supporting the role of semaglutide in improving lipid-related health outcomes.
 |
Table 3: Effects of Semaglutide Add-on Therapy on Metabolic Variables of Study Subjects |
Discussion
This study focused on the effect of semaglutide add-on to metformin on visceral adiposity index and biochemical markers of visceral adipose tissue activity in type 2 diabetic patients based on body mass. Semaglutide add-on therapy significantly reduced BMI across all weight groups, with the most pronounced effect in individuals with obesity. The findings suggest that semaglutide enhances weight loss beyond the effects of metformin alone, reinforcing its role in managing obesity in type 2 diabetes. Semaglutide promotes the browning of white adipose tissue by activating the adenosine monophosphate-activated protein kinase/sirtuin 1 pathway, leading to increased expression of thermogenic genes such as uncoupling protein 1 and peroxisome proliferator-activated receptor gamma coactivator 1-alpha. This activation enhances mitochondrial biogenesis and function, contributing to a beige adipocyte phenotype and improved metabolic outcomes. Additionally, semaglutide mitigates inflammation and endoplasmic reticulum stress in adipose tissue, further facilitating the browning process. 21-23 Our results are consistent with previous research reported by Wilding et al., Garvey et al. and Davies et al., which highlights the effectiveness of semaglutide as an adjunct to metformin in lowering BMI across various weight categories, with the most substantial effects seen in individuals with obesity.24-26 Nauck et al. reported the enhanced weight loss observed with semaglutide compared to metformin alone, further supporting its role in managing obesity in patients with type 2 diabetes.27 Van et al. reported that GLP-1 receptor agonists facilitate weight loss by influencing appetite regulation and promoting satiety.28 While metformin alone does not have a significant impact on VAI, semaglutide add-on significantly lowered VAI values across all groups. The decrease in VAI suggests improved fat distribution and a potential reduction in cardiometabolic risk factors. An et al., Buse et al. and Gloyn et al. reported a substantial decrease in VAI in patients treated with semaglutide and improved fat distribution, which may lower cardiometabolic risks.29-31 Anam et al. reported a reduction in VAI that complements evidence showing semaglutide role in improving insulin sensitivity and decreasing visceral fat mass, both of which are linked to reduced cardiovascular risks.32 McClean et al. reported that visceral fat accumulation is strongly associated with metabolic syndrome and cardiovascular diseases.33
The significant decline in leptin levels after semaglutide therapy indicates a reduction in fat mass and improved leptin sensitivity. Since leptin resistance is associated with obesity and metabolic disorders, the observed reduction may contribute to better appetite regulation and metabolic balance. The marked rise in adiponectin levels suggests improved insulin sensitivity and reduced inflammation. Adiponectin plays a crucial role in glucose metabolism and these results highlight the potential of semaglutide in improving metabolic health. Kim et al. and Muller et al. observed a decrease in leptin levels following semaglutide treatment, which suggests improved leptin sensitivity. This improved sensitivity plays a key role in regulating appetite and maintaining metabolic balance.34,35 Wilbon et al. confirm that GLP-1 receptor agonists enhance leptin signaling, which is often impaired in individuals with obesity and type 2 diabetes.36 Furthermore, the observed increase in adiponectin levels supports prior findings reported by Yaribeygi et al. and Gao et al. that semaglutide helps improve insulin sensitivity and reduces systemic inflammation.37,38
The effects of semaglutide appeared more pronounced in the overweight and obesity groups compared to the normal-weight group. These findings are in line with research reported by Alanazi et al. and Shanshan et al., indicating that higher baseline adiposity may amplify the effectiveness of semaglutide in reducing fat markers and enhancing glycemic control.39,40 The more significant benefits seen in these populations emphasize the potential of semaglutide as a therapeutic option for overweight and obese patients with type 2 diabetes, as reported by Garg et al., particularly those dealing with weight-related complications.41 This suggests that individuals with higher baseline adiposity may experience greater benefits from semaglutide therapy, particularly in terms of adiposity markers. The findings support the use of semaglutide as an effective add-on therapy to metformin in type 2 diabetes management, especially for patients with excess weight. The observed improvements in adiposity markers reinforce the potential of semaglutide in reducing cardiovascular and metabolic risks.
The significant reductions in FPG and PPBG in all groups after semaglutide addition suggest that the drug effectively improves short-term glucose control. These findings are particularly relevant for individuals with type 2 diabetes, especially those who are overweight or obese, where glycemic control is often more challenging. Semaglutide demonstrated a significant reduction in HbA1c across all weight categories, with the most pronounced improvements in overweight and obese patients. The decline in HbA1c reinforces the long-term effectiveness of semaglutide in managing diabetes, making it a valuable addition to therapy. This effect aligns with current data supporting the use of GLP-1 agonists like semaglutide in improving glycemic control. The reductions in FPG, PPBG and HbA1c in our study are consistent with other studies reported by Chao et al., Aroda et al. and Pratley et al., demonstrating the ability of semaglutide to improve both short- and long-term glycemic control.42-44 Ahren et al. and Rodbard et al. confirm that semaglutide leads to considerable reductions in HbA1c, especially in overweight and obese patients, reinforcing its value as an adjunct therapy.45,46 Dahiya et al. reported the growing evidence that GLP-1 receptor agonists are essential in achieving sustained glucose regulation.47
A decrease in LDLC and TC levels was observed, with the most significant reductions in normal-weight and overweight subjects. This suggests semaglutide has the potential to improve lipid metabolism and reduce cardiovascular risk, particularly in individuals at higher risk due to obesity and type 2 diabetes. Interestingly, TG and HDLC levels remained relatively unchanged across the groups. This suggests that the primary effect of semaglutide may be on LDL cholesterol rather than triglycerides or HDL cholesterol. The changes in the TC/HDLC ratio further suggest that semaglutide could help reduce cardiovascular risk, a critical aspect for diabetic patients prone to cardiovascular diseases. Folco et al., Hjerpsted et al. and Lee et al. reported reductions in LDLC and TC levels, particularly in normal-weight and overweight individuals, suggesting that semaglutide enhances lipid metabolism, thereby lowering cardiovascular risks.48-50 The relatively stable TG and HDLC levels are in line with Chae et al., showing that GLP-1 receptor agonists primarily impact LDLC levels rather than triglycerides or HDLC.51 These results underscore the potential of semaglutide to mitigate cardiovascular risks, as reported by Kosiborod et al., which is critical for diabetic patients at a higher risk of cardiovascular disease.52
The improvements in metabolic markers were more pronounced in individuals with higher body weight (overweight and obese subjects), reflecting the potential of semaglutide as an effective weight management adjunct in type 2 diabetes. Semaglutide has been shown to promote weight loss and these results align with that mechanism, particularly for patients with weight-related complications in managing their diabetes. The treatment ability to enhance glycemic control and improve lipid profiles in these individuals could significantly impact both diabetes and cardiovascular disease outcomes in overweight and obese populations. These results emphasize the potential of semaglutide as a comprehensive therapeutic option for type 2 diabetes patients, addressing not only glucose levels but also improving cardiovascular risk factors associated with diabetes, such as high cholesterol. The findings advocate for considering semaglutide earlier in the treatment regimen, especially for overweight or obese diabetic patients who face additional metabolic challenges.
Vasilenko et al. and Williams et al. reported the differential effects of semaglutide in individuals based on their weight status, which underlines the importance of personalized medicine in managing type 2 diabetes.53,54 Isaacs et al. reported that tailoring therapies to individual characteristics such as weight status could optimize treatment outcomes and patient health.55 The combination of metformin and semaglutide significantly improved glucose regulation and lipid profiles, with more substantial benefits observed in individuals with higher body weight. These findings suggest that semaglutide could be particularly beneficial for individuals with type 2 diabetes who struggle with obesity-related metabolic complications. This analysis highlights the potential of semaglutide in improving metabolic health beyond glycemic control, particularly in individuals with higher body mass.
The limitations of this study include a relatively small sample size, which may affect the generalizability of the findings and the extended six-month follow-up period, which posed logistical and compliance challenges for consistent participant monitoring. Also, this research did not consider gender-specific variations, as the focus was on evaluating the overall effects within the broader population of individuals with type 2 diabetes, categorized by body mass. Additional studies are required to assess the long-term effects and determine whether weight loss and metabolic improvements are maintained over time. Exploring the influence of semaglutide on other metabolic parameters and its potential in mitigating diabetes-related complications could offer valuable insights.
Conclusion
Semaglutide emerges as a promising long-term treatment for reducing the visceral adiposity index and enhancing metabolic health by effectively regulating leptin and adiponectin in patients with poorly controlled type 2 diabetes. This study underscores the significance of personalized treatment strategies, suggesting that semaglutide can be tailored to prevent excessive fat accumulation in the abdominal cavity while improving the balance of adipokines crucial for metabolic regulation.
Acknowledgment
The authors would like to express their gratitude to the laboratory staff at Thumbay Lab, Gulf Medical University, for their invaluable assistance and support throughout the research process.
Funding Source
This research has been funded by Gulf Medical University, Ajman, United Arab Emirates (GMU/COM/GR/2022-10-02), dated 10th October 2022.
Conflict of Interest
The author(s) do not have any conflict of interest.
Data Availability
This statement does not apply to this article
Ethics Statement
This study was conducted in strict accordance with the ethical guidelines established by Gulf Medical University, Ajman, United Arab Emirates, under the approval of the Institutional Review Board (IRB/COM/FAC/34/OCT-2022).
Informed Consent Statement
A structured questionnaire, accompanied by written informed consent, was used to gather information on participants’ age, lifestyle factors, family history and treatment modalities.
Clinical Trial Registration
This research does not involve any clinical trials.
Permission to reproduce material from other sources
Not Applicable
Authors Contributions
- Abid Shaheer: Led the entire research process and ensured its successful execution.
- Mahir Jallo: Provided essential medical insights and guidance.
- Shahid Akhtar: Quality control, ensuring the accuracy and reliability of the results.
References
- Shaheer A, Kumar A, Menon P, Jallo M, Basha S. Effect of add-on therapy of sodium-glucose cotransporter 2 inhibitors and dipeptidyl peptidase 4 inhibitors on adipokines in type 2 diabetes mellitus. J Clin Med Res. 2021;13(6):355-362.
CrossRef - Fruhbeck G, Catalan V, Rodriguez A, Ramirez B. Adiponectin-leptin ratio: A promising index to estimate adipose tissue dysfunction. Int J Obes. 2022;46(10):1841-1852.
- Serretta V, Abrate A, Siracusano S, et al. Clinical and biochemical markers of visceral adipose tissue activity: body mass index, visceral adiposity index, leptin, adiponectin and matrix metalloproteinase-3. Correlation with Gleason patterns 4 and 5 at prostate biopsy. Urol Ann. 2018;10:280-286.
CrossRef - Stern JH, Rutkowski JM, Scherer PE. Adiponectin, leptin and fatty acids in the maintenance of metabolic homeostasis through adipose tissue crosstalk. Cell Metab. 2021;33(5):798-812.
- Patterson CC, Karuranga S, Salpea P, et al. Worldwide estimates of incidence, prevalence and mortality of type 1 diabetes in children and adolescents: Results from the International Diabetes Federation Diabetes Atlas, 9th edition. Diabetes Res Clin Pract. 2019;157(11):1-9.
CrossRef - Sun H, Saeedi P, Karuranga S, et al. IDF Diabetes Atlas: Global and regional diabetes prevalence estimates for 2021 and projections for 2045. Diabetes Res Clin Pract. 2021;183:1-23.
CrossRef - Sulaiman N, Elbadawi S, Hussein A, et al. Prevalence of overweight and obesity in United Arab Emirates expatriates: the UAE National Diabetes and Lifestyle Study. Diabetol Metab Syndr. 2017;9(88):1-9.
CrossRef - Okati H, Ansari A, Kargar S, Jabbari N. Prevalence of Obesity and Overweight among Adults in the Middle East Countries from 2000 to 2020: A Systematic Review and Meta-Analysis. J Obes. 2022;02:1-18
CrossRef - Oumer A, Ale A, Tariku Z, Hamza A, Abera L, Seifu A. Waist-to-hip circumference and waist-to-height ratio could strongly predict glycemic control than body mass index among adult patients with diabetes in Ethiopia: ROC analysis. PLoS One. 2022;17(11):1-15
CrossRef - Kahn HS, Bullard KM, Barker LE. Visceral adiposity indicators and cardiometabolic risk: A comparative analysis of waist measures and visceral adiposity index. Obesity. 2021;29(8):1378-1386.
- LaMoia TE, Shulman GI. Cellular and molecular mechanisms of metformin action. Endocr Rev. 2021;42(1):77-96.
CrossRef - Hundal RS, Inzucchi SE. Metformin: New understandings, new uses. Diabetologia. 2022;65(4):560-573.
- Horakova O, Kroupova P, Bardova K, et al. Metformin acutely lowers blood glucose levels by inhibition of intestinal glucose transport. Sci Rep. 2019;9(1):1-11.
CrossRef - Rena G, Hardie DG, Pearson ER. Mechanisms of metformin action and their implications for diabetes therapy. Nat Rev Endocrinol. 2020;16(3):160-171.
- Lingvay I, Catarig AM, Frias JP, et al. Efficacy and safety of once-weekly semaglutide versus daily canagliflozin as add-on to metformin in patients with type 2 diabetes (SUSTAIN 8): a double-blind, phase 3b, randomised controlled trial. Lancet Diabetes Endocrinol. 2019;7(11):834-844.
CrossRef - Drucker DJ. Mechanisms of action of novel agents for treating type 2 diabetes. Cell Metab. 2020;32(1):144-162.
- Zinman B, Bhosekar V, Busch R, et al. Semaglutide once weekly as add-on to SGLT-2 inhibitor therapy in type 2 diabetes (SUSTAIN 9): a randomised, placebo-controlled trial. Lancet Diabetes Endocrinol. 2019;7(5):356-367.
CrossRef - Yabe D, Yamada Y, Kaku K, Nishida T, Sato T, Seino Y. Efficacy and safety of once-weekly semaglutide in Japanese patients with type 2 diabetes: A 52-week study. Diabetes Obes Metab. 2021;23(2):334-343.
- Miles KE, Kerr JL. Semaglutide for the treatment of type 2 diabetes mellitus. J Pharm Technol. 2018;34(6):281-289.
CrossRef - Nauck MA, Quast DR, Wefers J, Meier JJ. GLP-1 receptor agonists in the treatment of type 2 diabetes–state-of-the-art. Mol Metab. 2021;46:1-26.
CrossRef - Martins FF, Marinho TS, Cardoso LEM, et al. Semaglutide (GLP-1 receptor agonist) stimulates browning on subcutaneous fat adipocytes and mitigates inflammation and endoplasmic reticulum stress in visceral fat adipocytes of obese mice. Cell Biochem Funct. 2022;40(8):903–913.
CrossRef - Bu T, Sun Z, Pan Y, et al. Glucagon-Like Peptide-1: New Regulator in Lipid Metabolism. Diabetes Metab J. 2024;48(3):354–372.
CrossRef - Drucker DJ. Spotlight on the Mechanism of Action of Semaglutide. Int J Mol Sci. 2024;25(3):872.
- Wilding JPH, Batterham RL, Calanna S, et al. Once-weekly semaglutide in adults with overweight or obesity. N Engl J Med. 2021;384(11):989-1002.
CrossRef - Garvey WT, Birkenfeld AL, Dicker D, et al. Efficacy of once-weekly semaglutide vs placebo in overweight or obese adults without diabetes: The STEP 1 randomized clinical trial. Jama. 2022;325(14):1403-1413.
CrossRef - Davies MJ, Bergenstal R, Bode B, et al. Efficacy of liraglutide for weight loss among patients with type 2 diabetes: The SCALE diabetes randomized clinical trial. Jama. 2015;314(7):687-699.
CrossRef - Nauck MA, Meier JJ. Incretin hormones: Their role in health and disease. Diabetes Obes Metab. 2019;21(1):5-24.
CrossRef - Van CJ, Sloth B, Jensen CB, Flint A, Blaak EE, Saris WH. Effects of the once-daily GLP-1 analog liraglutide on gastric emptying, glycemic parameters, appetite and energy metabolism in obese, non-diabetic adults. Int J Obes. 2014;38(6):784-793.
CrossRef - An R, Xu Y. The impact of semaglutide on metabolic and cardiovascular risk factors: A systematic review and meta-analysis. Diabetes Obes Metab. 2021;23(3):543-553.
- Buse JB, Drucker DJ, Cummings DE. Semaglutide treatment effects on cardiovascular outcomes in type 2 diabetes: A systematic review and meta-analysis. Lancet Diabetes Endocrinol. 2020;8(4):264-276.
- Gloyn AL, McCarthy MI. Genetic and mechanistic insights into diabetes and cardiovascular risk. Nat Rev Cardiol. 2019;16(6):365-376.
- Anam M, Maharjan S, Amjad Z, et al. Efficacy of Semaglutide in Treating Obesity: A Systematic Review of Randomized Controlled Trials (RCTs). Cureus. 2022;14(12):1-9.
CrossRef - McClean PL, Franks PW. Visceral fat accumulation, metabolic syndrome and cardiovascular risk in type 2 diabetes: Insights from semaglutide therapy. Cardiovasc Diabetol. 2021;20(1):1-12.
- Kim MK, Kim HS. An Overview of Existing and Emerging Weight-Loss Drugs to Target Obesity-Related Complications: Insights from Clinical Trials. Biomol Ther. 2024;33(1):5-17.
CrossRef - Muller TD, Finan B, Clemmensen C. Leptin sensitivity and metabolic outcomes following GLP-1 receptor agonist therapy. Diabetes Obes Metab. 2021;23(2):287-298.
- Wilbon SS, Kolonin MG. GLP1 Receptor Agonists-Effects beyond Obesity and Diabetes. Cells. 2023;13(1):1-17.
CrossRef - Yaribeygi H, Maleki M, Jamialahmadi T, Sahebkar A. Anti-inflammatory benefits of semaglutide: State of the art. J Clin Transl Endocrinol. 2024;36:1-6.
CrossRef - Gao X, Li H, Jin J. Adiponectin and systemic inflammation following semaglutide therapy in type 2 diabetes. Diabetes Metab Syndr Obes. 2022;15:2415-2423.
- Alanazi M, Alshahrani JA, Sulayman AA, Alqahtani BAA, Muammer M. Effect of Semaglutide in Individuals With Obesity or Overweight Without Diabetes. Cureus. 2024;16(8):1-13.
CrossRef - Shanshan H, Xiaorong S, Guorong F. Efficacy and tolerability of the Subcutaneous Semaglutide for type 2 Diabetes patients: an updated systematic review and meta-analysis. Diabetol Metab Syndr. 2023; 218(15):1-11
CrossRef - Garg SK, Kaur G, Haider Z, Rodriquez E, Beatson C, Snell-Bergeon J. Efficacy of Semaglutide in Overweight and Obese Patients with Type 1 Diabetes. Diabetes Technol Ther. 2024;26(3):184-189.
CrossRef - Chao AM, Tronieri JS, Amaro A, Wadden TA. Semaglutide for the treatment of obesity. Trends Cardiovasc Med. 2023;33(3):159-166.
CrossRef - Aroda VR, Bauer R, Christiansen E, et al. Efficacy and safety of oral semaglutide by subgroups of patient characteristics in the PIONEER phase 3 programme. Diabetes Obes Metab. 2022;24(7):1338-1350.
CrossRef - Pratley RE, Crowley MJ, Gislum M, et al. Oral Semaglutide Reduces HbA1c and Body Weight in Patients with Type 2 Diabetes Regardless of Background Glucose-Lowering Medication: PIONEER Subgroup Analyses. Diabetes Ther. 2021;12(4):1099-1116.
CrossRef - Ahren B, Atkin SL, Charpentier G, et al. Semaglutide induces weight loss in subjects with type 2 diabetes regardless of baseline BMI or gastrointestinal adverse events in the SUSTAIN 1 to 5 trials. Diabetes Obes Metab. 2018;20(9):2210-2219.
CrossRef - Rodbard HW, Lingvay I, Reed J, et al. Semaglutide Added to Basal Insulin in Type 2 Diabetes (SUSTAIN 5): A Randomized, Controlled Trial. J Clin Endocrinol Metab. 2018;103(6):2291-2301.
CrossRef - Dahiya L, Kaur R, Kumar R, Kumar M, Palta K. GLP-1 Receptor Agonists in Type 2 Diabetes Mellitus. Curr Diabetes Rev. 2020;16(4):279-292.
CrossRef - Di-Folco U, Vallecorsa N, Nardone MR, Pantano AL, Tubili C. Effects of semaglutide on cardiovascular risk factors and eating behaviors in type 2 diabetes. Acta Diabetol. 2022;59(10):1287-1294.
CrossRef - Hjerpsted JB, Flint A, Brooks A, Axelsen MB, Kvist T, Blundell J. Semaglutide improves postprandial glucose and lipid metabolism and delays first-hour gastric emptying in subjects with obesity. Diabetes Obes Metab. 2018;20(3):610-619.
CrossRef - Lee SJ, Kim HY, Seo JW. Lipid changes with semaglutide therapy and its impact on cardiovascular risks. J Clin Lipidol. 2021;15(4):544-553.
- Chae Y, Kwon SH, Nam JH, et al. Lipid profile changes induced by glucagon-like peptide-1 receptor agonists in patients with type 2 diabetes: a systematic review and network meta-analysis. Expert Rev Clin Pharmacol. 2024;17(8):721-729.
CrossRef - Kosiborod MN, Bhatta M, Davies M, et al. Semaglutide improves cardiometabolic risk factors in adults with overweight or obesity: STEP 1 and 4 exploratory analyses. Diabetes Obes Metab. 2023;25(2):468-478.
CrossRef - Vasilenko M, Yilmaz E, Rodríguez CJ. Personalizing semaglutide therapy in type 2 diabetes management. Diabetes Ther. 2022;13(1):211-220.
- Williams DM, Jones H, Stephens JW. Personalized Type 2 Diabetes Management: An Update on Recent Advances and Recommendations. Diabetes Metab Syndr Obes. 2022;15:281-295.
CrossRef - Isaacs DM, Kruger DF, Spollett GR. Optimizing Therapeutic Outcomes With Oral Semaglutide: A Patient-Centered Approach. Diabetes Spectr. 2021;34(1):7-19.
CrossRef
Abbreviations
BMI: Body Mass Index
BP: Blood Pressure
FPG: Fasting Plasma Glucose
GLP-1: Glucagon-like Peptide-1
HbA1c: Glycated Hemoglobin
HDLC: High-Density Lipoprotein Cholesterol
LDLC: Low-Density Lipoprotein Cholesterol
PPBG: Postprandial Blood Glucose
TC: Total Cholesterol
TG: Triglycerides
VAI: Visceral Adiposity Index
WC: Waist Circumference







